Abstract
Children born from women with preeclampsia have alterations in cerebral neurovascular development and a high risk for developing cognitive alterations. Because cerebral blood vessels are critical components in cerebrovascular development, we evaluated the brain microvascular perfusion and microvascular reactivity (exposed to external stimuli of warm and cold) in pups born to preeclampsia-like syndrome based on the reduction of uterine perfusion (RUPP). Also, we evaluate the angiogenic proteomic profile in those brains. Pregnant mice showed a reduction in uterine flow after RUPP surgery (−40 to 50%) associated with unfavorable perinatal results compared to sham mice. Furthermore, offspring of the RUPP mice exhibited reduced brain microvascular perfusion at postnatal day 5 (P5) compared with offspring from sham mice. This reduction was preferentially observed in females. Also, brain microvascular reactivity to external stimuli (warm and cold) was reduced in pups of RUPP mice. Furthermore, a differential expression of the angiogenic profile associated with inflammation, extrinsic apoptotic, cancer, and cellular senescence processes as the primary signaling impaired process was found in the brains of RUPP-offspring. Then, offspring (P5) from preeclampsia-like syndrome exhibit impaired brain perfusion and microvascular reactivity, particularly in female mice, associated with differential expression of angiogenic proteins in the brain tissue.
Introduction
Preeclampsia (PE) is a human pregnancy multisystem disorder of unknown etiology characterized by impaired placental blood flow and maternal endothelial dysfunction. 1 The global prevalence of PE has increased over the past 30 year. 2 Especially in developing countries where this condition ranges from 1.8% to 16.7% of pregnancies. 3 This maternal condition is associated with an increased risk of fetal death and severe neonatal disturbances, including prematurity and intrauterine growth retardation. 4 Moreover, the harmful consequences of PE can be extended beyond delivery, 3 showing a series of sequels in the offspring's health, such as the increased risk for obesity and cardiovascular and neurovascular disorders.5,6
Children of mothers with PE have a higher risk of developing neurological, psychological, or behavioral disorders than children born to normotensive pregnancies,7–9 independently associated with PE.10–12 In turn, these developmental and neurological alterations in children of women with PE have been associated with structural abnormalities in specific areas of the brain (such as the cerebellum, brainstem, temporal lobe, right and left amygdala), as well as narrower radii in vessels of parietal, and occipital lobes, 13 or even with impaired functional connectivity in the left and right amygdala and medial prefrontal cortex. 14 Biological confirmation of these perturbations came from animal models of PE15–17 showing perturbations in spatial learning and memory, associated with impaired neurogenesis and structural alterations in the brain cortex, hippocampus, and brain blood vessels in offspring born to PE-like models.
On the other hand, brain blood vessels actively control different neurodevelopmental processes. 15 After birth, this vasculature develops via angiogenesis, generating an extensive network of capillaries. 16 Despite no evidence regarding brain angiogenesis in children born to PE, indirect evidence has shown a reduced angiogenic capacity in human umbilical vein endothelial cells exposed to plasma from women with PE. 17 Nevertheless, deletion of the placental growth factor (Plgf), a well-described growth factor reduced in PE, generates an aberrant vascular branching at the early embryonic stage. 18 Furthermore, after birth, analysis in another experimental model of PE generated using a high-fat diet showed that the lumen diameter of middle cerebral arteries (MCA) from PE offspring was smaller and stiffer biomechanically than mice of normal pregnancy. 19 Therefore, offspring born to PE exhibit signs of impaired brain angiogenesis.9,20
In addition, since the adverse placental environment,9,21,22 is critical in the fetal brain disturbances, 23 it is intriguing why current findings in the PE offspring brains came from experimental models that do not necessarily recapitulate reduced placental blood perfusion. In the present work, we use a reduced uterine perfusion pressure (RUPP) mice model generated by clipping the uterine and ovarian arteries 24 to evaluate the brain microvascular perfusion and microvascular reactivity (exposed to external stimuli of warm and cold) in pups. In addition, we further explore the angiogenic proteomic profile in the brain of these pups. We hypothesize that RUPP-offspring presents dysfunctional brain perfusion associated with alterations in the brain angiogenic environment.
Materials and methods
RUPP model
The bioethics and Biosafety Committee of the Universidad del Bío-Bío approved the animal management and supervision protocols used in this study in agreement with the Guide for Care and Use of Laboratory Animals guidelines 25 and 3 R principles referring to humanized use of animals. In addition, all experiments are reported following the ARRIVE guidelines 2.0 (Animal Research: Reporting in Vivo Experiments) for how to report animal experiments. Female and male C57BL/6 mice were housed in a 25°C temperature and humidity-controlled room in the Vivarium belonging to Universidad del Bío-Bío. Inbred strains of mice obtained in our animal facility were used for all experiments. Light–dark cycles were 12:12 hours, and mice were fed with balanced food (Prolab RMH 3000, Labdiet, St. Luis, Missouri, USA) and water ad-libitum.
At the age of 3–5 months, female mice were crossbreeding with C57BL/6 male mice of similar age. The presence of a vaginal plug was designated as 0.5-day postcoitum (dpc). Pregnant mice at 14.5 dpc (Sham, n = 16; RUPP, n = 16) were anesthetized with isoflurane 3% before laparotomy, which was performed on a thermoregulated platform (Kent Scientific, Torrington, Connecticut, USA). Surgical exposition of the uterine horns allowed the analysis of uterine blood vessels and the presence of embryos. In the RUPP group, uterine arterial and ovary arteries were partially cauterized on both sides of the uterus following a validated model. 26 In a subset of pregnant mice (n = 8 per group), the uterine blood flow was evaluated using laser Doppler (Perimed PSI, Stockholm, Sweden) on 14.5 and 18.5 dpc. After respective surgery, the mid-abdominal incision was sutured. Analgesia post-surgery was conducted using ketoprofen (5 mg/kg, subcutaneously) every 24 hours for three days.
Measurement of blood pressure (BP)
Systemic BP was noninvasively measured from 14.5 to 18.5 dpc. Thus, a volume pressure recording sensor was placed in the tail of pregnant mice, and BP measurements were recorded in real-time (CODA, Kent Scientific, Torrington, Connecticut, USA). Systolic and diastolic blood pressure was used to calculate a mean arterial pressure (MAP) described elsewhere. 27
Proteinuria analysis
Urine samples were collected between 14.5 and 18.5 dpc. For urine collection, mice were placed in metabolic cages. Urine proteins were extracted with a RIPA buffer (Thermo Scientific, Waltham, Massachusetts, USA) and quantified by absorbance (Biotek, Winooski, Vermont, USA). Then, urine samples (6 µL) of 14.5 and 18.5 dpc were loaded on SDS–PAGE gel (10% acrylamide) and stained with Coomassie® Blue to visualize the entire protein content in each sample. The intensity of protein bands was measured using ImageJ software (NIH, Rockville, Maryland, USA).
Blood sample collection
We extracted blood samples at terminal management of pregnant mice and offspring via an intracardiac puncture. Blood samples were stored at room temperature for 2 hours and then centrifuged at 2000 g for 20 minutes to serum withdrawal. Serum samples were diluted in distilled water (1:1, v/v), and circulating proteins were quantified by absorbance as indicated above. Serum samples were stored at −20°C for subsequent ELISA analysis.
Placenta morphometry and measurements
On day 18.5 dpc, placentas (and offspring, see below) were removed from the uterus and weighted. The placental efficiency was calculated as the fetal body weight/placenta weight ratio. 26 The placentas were fixed in paraformaldehyde (4% in buffer phosphate, v/v) for 24 hours and subsequently embedded in paraffin for histological analysis.
Offspring morphometry and measurements
Similarly, at 18.5 dpc and postnatal day 5 (P5), morphometric parameters were registered in pups, including weight and size (determined by cephalo-caudal length). Pups were sexed by visual evaluation and confirmed by PCR using the expression of the jarid gene, as previously described. 28 Then, the weight-for-length ratio and weight-for-weight ratio of the brain were calculated individually. After that, brains were collected in 300 µl of RIPA buffer and frozen at −20°C until further analysis. All analyses of the pups included one to three males and females per litter in each group.
Circulating plgf and flt-1
Maternal circulating levels of Plgf were measured using a Quantikine ELISA kit (MP200, R&D Systems, Minneapolis, Minnesota, USA) following the manufacturer's protocol. Intra and inter-assay variability was 3 and 1%, respectively. The limit of detection was 1.14 pg/mL.
A dot blot technique was used to estimate vascular endothelial growth factor 1 in the serum (i.e., sFlt-1), as described elsewhere. 29 A selective antibody was used to detect Flt-1 in mice (Santa Cruz Biotechnology, Dallas, Texas, USA; sc-316, dilution 1:1000, v/v). A horseradish peroxidase-conjugated secondary antibody was used for visualization (rabbit, Thermo Scientific, Waltham, Massachusetts, USA; #31450, dilution 1:20000, v/v).
Evaluation of kidneys and placentas
Paraformaldehyde fixed-paraffin embedded placentas and kidneys (18.5 dpc) were sectioned in a Multicut 2045 (Leica, Allendale, New Jersey, USA) microtome at three μm thickness and stained with periodic acid-Schiff (PAS). For kidney analysis, ten randomly selected glomeruli from each female were analyzed using a photomicroscope Axioscop 40 (Zeiss, Oberkochen, Germany) and image processor Axiovision Release 4.6 software (Zeiss, Oberkochen, Germany). The measurement included the following parameters: glomerular Tuft area (total glomerular area); glomerular extracellular matrix (ECM) expressed as PAS-positive mesangial area; and glomerular capillaries area (Cap). 30
Placental histology was analyzed using hematoxylin and eosin (H&E) stained samples. Analyzed parameters included: areas of maternal decidua, junctional zone, and labyrinth layer tightness, as previously described. 26 Percentages of each area/zone were calculated considering the whole placental area. All histological analyses were performed by an independent researcher (DR), who was blinded to the group and sex of mice.
Brain edema
Postmortem transcranial ultrasound was performed in some randomly selected mice (n = 10 per group) (Mindray, Shenzhen, China). Thus, intracranial longitudinal distance (from the inner frontal to the occipital bone); and transverse distance (from the right and left temporal bones) were measured. For accurate measurements, the longitudinal section considered the recognition of the olfactory bulb and cranial portion of the spinal cord. The transverse section was the most significant distance between temporal bones. The inner skull circumference was estimated by dividing the measured distance (a: longitudinal and b: transverse) and measurement of the ellipse perimeter: 2π√((a2 + b2)/2). The ratio between the brain weight and the inner circumference of the skull was used to estimate brain edema. 31
Brain cerebral perfusion
In vivo, cerebral perfusion analysis in mice (P5) was performed using the Pericam® PSI-HR system (Perimed Ltd., Stockholm, Sweden). Briefly, P5 mice were anesthetized with isoflurane 4%, and then the dorsal skin of the head was removed to evaluate cerebral blood flow. The analysis matrix includes four corresponding areas (regions of interest, ROIs) of small vessels (i.e., microcirculation) at the mid-level of the brain encompassing anterior and posterior areas of interest in both hemispheres. ROIs were circular with a constant ratio for all analyses. To avoid the potential effect of the variability that may lead to the use of the different locations of the ROI in the brain cortex, we analyzed each ROI separately, showing no significant differences in either intergroup or intragroup (data no shown). The experimental protocol consisted of basal perfusion analysis (3–4 minutes), followed by vasodilation induced using a warm superficial stimulus generated by direct application of saline solution (warm stimuli, 0.9% NaCl, w/v, 37°C) on the exposed brain. After that, vasoconstriction was induced by direct application of cold saline solution (cold stimuli, 0.9 NaCl w/v, 4°C). Perfusion was continuously recorded during the whole experiment (10 minutes approximately). Time of interest (TOIs) was defined considering continuous 512 data points in each experimental window (basal, warm, and cold stimuli). Those TOIs were selected in a trace section immediately after applying the saline solution. We used 512 data points because Fourier Transformed analysis (see below) requires data rose to a power of two. Brain perfusion data in response to warm or cold stimuli were normalized for each animal's respective mean value of basal perfusion. Then, the fold of change with respect to the basal brain perfusion of the sham group was calculated in each animal. Two researchers (EL and AR) performed the perfusion analysis. To avoid observer’s bias, experiments were performed head-by-head, including pups from the two experimental groups. At the same time, we performed raw data analysis in a pup's sex-blinded manner.
Wavelet analysis
Subsequently, cerebral perfusion was used for the wavelet spectral analysis. Each experimental condition was analyzed and separated with a 2 minutes time window. By applying the Discrete Fourier Transform (DFT) function in Excel (Microsoft, Washington, DC, USA), we transform the data from the time domain to the frequency domain, calculating the amplitude-frequency spectrum of the waveform. This analysis portrays signal intensity distributed over frequencies. The wavelet data was sampled every 0.2 seconds, obtaining a frequency spectrum between 0.0095–2.5 Hz. This range allows us to analyze the contribution of each regulatory component in the perfusion. Each component has a specific band frequency range: metabolic (0.0095–0.016 Hz), neurogenic (0.02–0.06 Hz), myogenic (0.06–0.15 Hz), respiratory (0.15–0.4 Hz) and cardiac bands (0.6–2 Hz). 32 Once the new frequency spectrums were obtained, each spectrum band contribution was calculated by the area under the curve. Each contribution was displayed as a percentage of the relative energy of all the bands analyzed.
Sample size
We used the mean-differences formula to calculate sample size considering the risk of 0.05, the statistical power of 95%, precision (d, minimal value of difference), and 20% of expected loss. Differences in total neocortex volume in children born from women with PE 13 were used to calculate the sample size. Therefore, sample size calculation indicates n = 8 offspring from RUPP and sham for brain analysis. Experiments were performed head-to-head, including randomly chosen animals in each group.
Angiogenesis protein array
The proteome profiler mouse angiogenesis array kit (ARY015, R&D, Minneapolis, Minnesota, USA) was used to assess the relative amount of 53 mouse angiogenesis-related proteins. Briefly, 500 ng of brain lysates (18.5 dpc and P5) were used to react with biotinylated detection antibodies in nitrocellulose membranes overnight (at four°C). Then, membranes were washed using 1× wash buffer and incubated with streptavidin-HRP (R&D, Minneapolis, Minnesota, USA). The captured proteins were visualized using chemiluminescence. Relative expressions of the proteins were estimated using densitometry of the detected dots using Image J software (NIH, Bethesda, Maryland, USA), as previously described. 33 Functional protein networks and biological processes analysis used the web String database. 34 In silico analyzed parameters included protein cluster, biological process, and signaling pathways.
Statistical analysis
Quantitative variables are presented as mean ± standard deviation, while qualitative variables in percentage as appropriate. Also, D'Agostino & Pearson omnibus normality test was performed to analyze data distribution. Results were analyzed through one-way or two-way ANOVA, followed by the Bonferroni test for multiple comparisons. In addition, pre and post-surgery analysis of the blood perfusion and blood pressure was performed using paired T-test. Finally, chi-square tests were used to compare percentages. Statistical analyses were performed using GraphPad Prism 6.00 (GraphPad Software, San Diego, California, USA). P < 0.05 was considered statistically significant.
Results
Reduced uterine blood perfusion
PE-like syndrome using the RUPP model was generated as described elsewhere 26 (Figure 1(a)). Thus, previous to surgery, the uterine blood perfusion was similar between RUPP and sham pregnant mice (401.6 ± 47.87 units versus 353.1 ± 65.54 perfusion units, respectively) (Figure 1(d)). However, immediately after RUPP surgery, uterine perfusion decreased 30–40% compared to respective basal measurements (p = 0.02). In addition, compared to respective basal measurements, there was an increase in blood perfusion in sham pregnant mice at 18.5 dpc (p = 0.01), but it was less evident in the RUPP pregnant mice (Figure 1(e)). We also performed a paired analysis comparing sham and RUPP groups at the same time moment. No differences were found in this analysis.
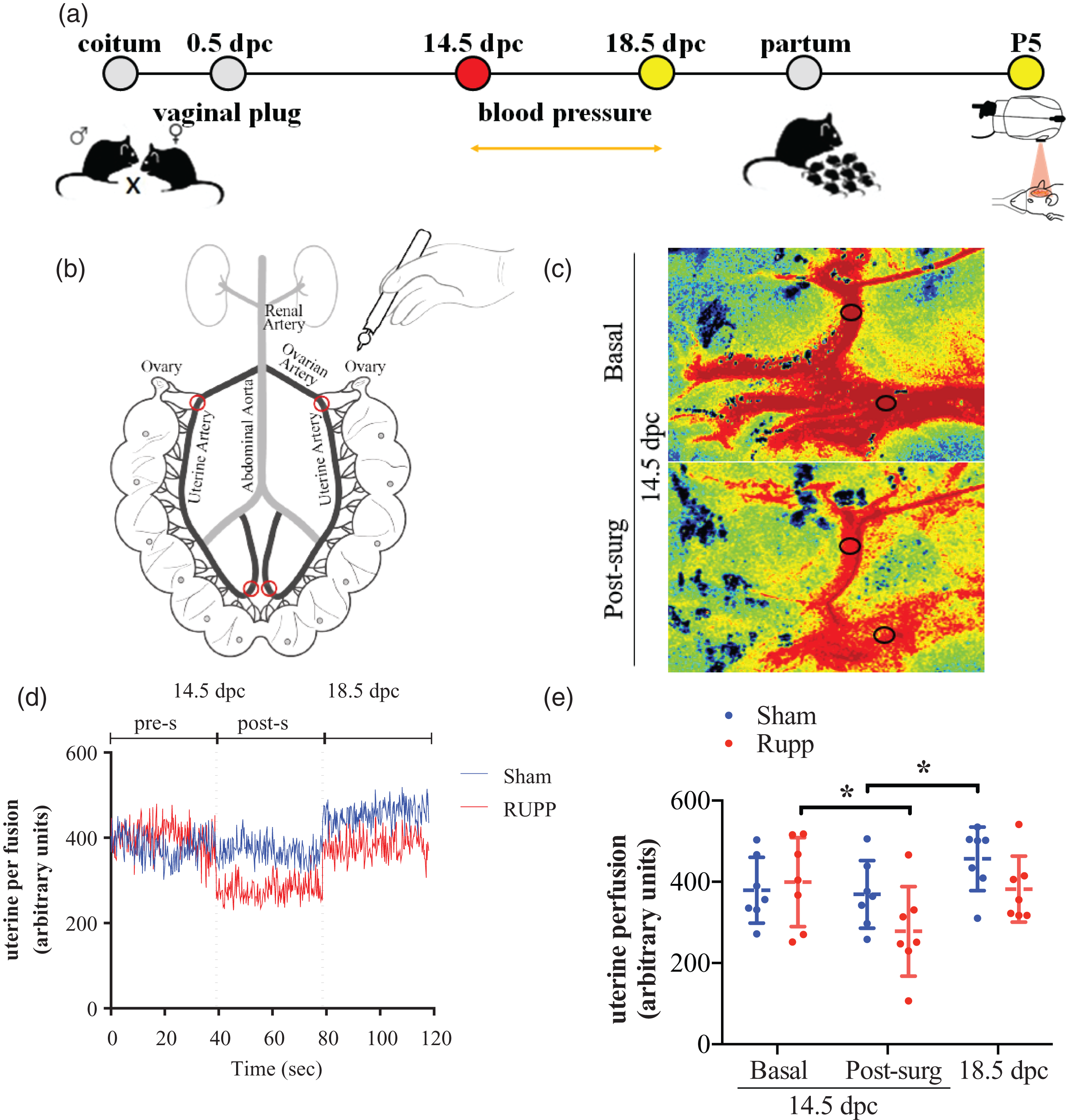
Reduced uterine perfusion pressure (RUPP) model in mice. (a) Schematic representation of the experimental design indicates the moment of surgery (red circle) and evaluation of mice (yellow circles). (b) Bilateral partial cauterization of uterine and ovarian arteries (red circles). (c) Image of the RUPP group's uterine perfusion at 14.5 dpc (Basal) and post-surgery (post-surg). (d) Representative trace of uterine perfusion before (pre-s) and after surgery (post-s) (e) In Sham and RUPP groups, basal and post-surgery uterine perfusion at 14.5 and 18.5 dpc. dpc: days post coitum, P5: postnatal day 5. Data in the plot graph represent the mean ± standard deviation. n = 8 per group. (*p < 0.05).
PE-like syndrome characterization
RUPP has been extensively characterized as a PE-like model.24,26,35 To validate this model in our laboratory, we have measured blood pressure before and after RUPP surgery, kidney alterations, proteinuria, and circulating levels of Plgf and Flt-1. Thus, before surgery (at 14.5 dpc), MAP was similar between the sham and RUPP groups (99.69 ± 10.75 versus 99.21 ± 13.81 mmHg, respectively). However, MAP significantly increased at 18.5 dpc only in the RUPP pregnant mice (116.8 ± 9.5 mmHg, p = 0.0004), which was statistically different compared to the respective value in the sham group (104.4 ± 7.6 mmHg, p = 0.03) (Figure 2(a)).
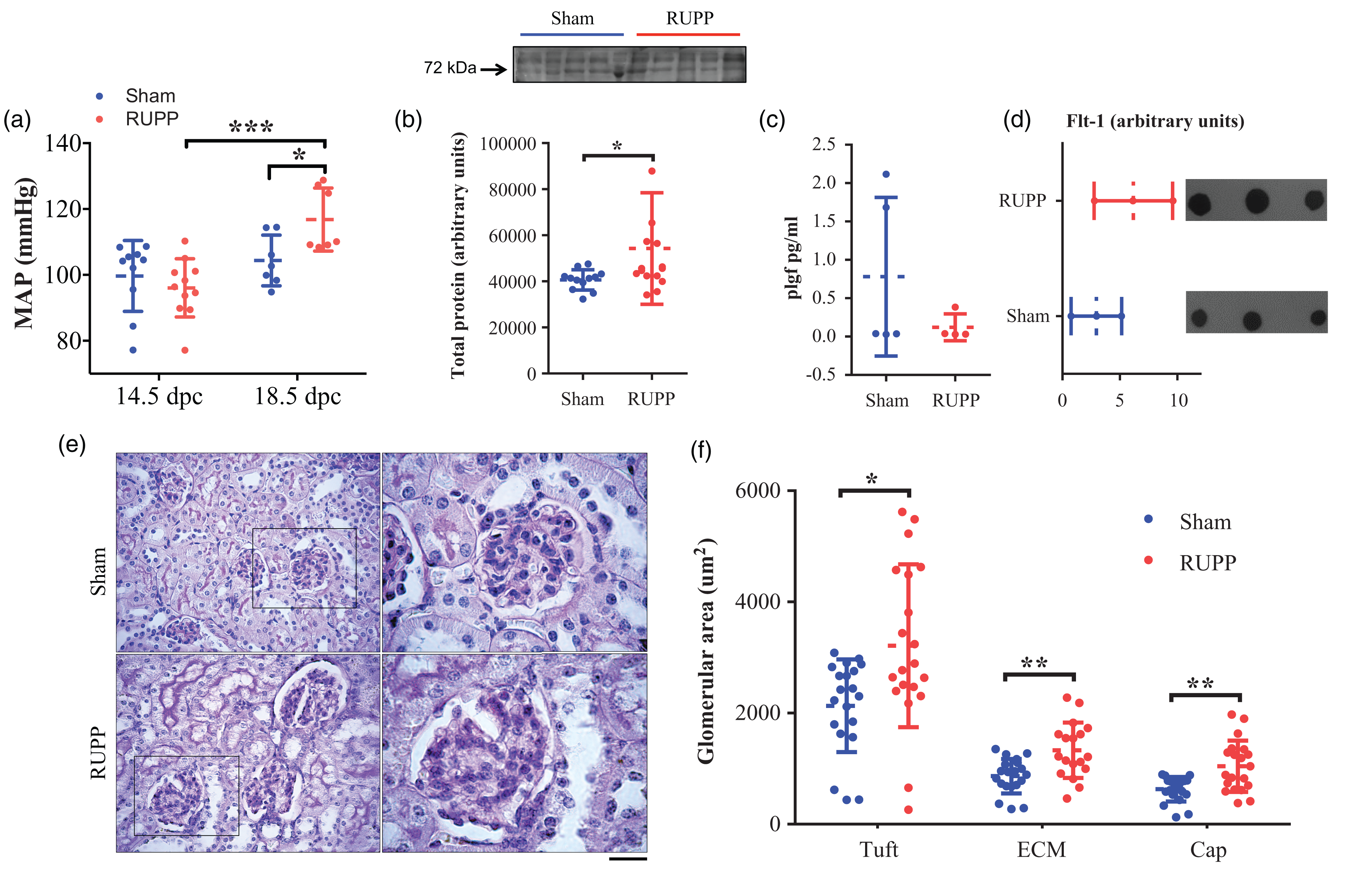
Hypertension and glomerular lesions in dams of RUPP model in mice. (a) Tail-cuff blood Mean Arterial Pressure (MAP) after partial cauterization of uterine and ovarian arteries. (b) Total urinary protein in Sham and RUPP group at 18.5 dpc. (c) Circulating levels of Plgf and (d) sFlt-1 in dams at 18.5 dpc. Representative dot-blots are included. (e) Representative images of periodic acid Schiff staining. Rights panels (100x) represent insets in left panels (40x). Scale bars represent 50 µm on left panels and 20 µm on right panels and (f) Glomerular area of the zones tuft (Tuft), extracellular matrix (ECM), and capillaries (Cap) in kidneys isolated at 18.5 gestational days. dpc: days post coitum. Data in the plot graph represent the mean ± standard deviation. n = 8 per group. (*p < 0.05; **p < 0.01, ***p < 0.001).
The total urinary protein at 18.5 dpc was higher in RUPP pregnant mice than in the sham group (p = 0.01). While the relative serum levels of Plgf and sFlt-1 were similar between groups (Figure 2(b) to (d)). Although a trend for reduced Plgf and high sFlt-1 was observed in the RUPP group.
Histological evaluation of the kidneys at 18.5 dpc showed some glomerular lesions, including increased glomerular size (Figure 2(e)), tuft (p = 0.01), extracellular matrix, and capillaries areas (Figure 2(f), p < 0.01) in comparison with the sham group.
Alterations in pregnancy, placental dysfunction, and fetal growth restriction
RUPP model generates impaired placental and fetal development and growth. 26 We found a lower gestational length in the RUPP than in the sham group (Fig S1A). Also, the percentage of reabsorption (45.7 versus 12.2%, respectively) and postnatal death (33.0 versus 5.3%, respectively) were higher in the RUPP than in the sham group (Fig S1B).
Placental weight was higher (Figure 3(a)), but the fetal weight was lower in the RUPP than in the sham group. Therefore, the placental efficiency was lower in the RUPP compared with the sham group (Figure 3(b), p = 0.01). However, this difference was independent of the fetal sex (Figure 3(b)). Accordingly, fetuses in the RUPP group were smaller than those in the sham group, with a weight/length index of 0.45 ± 0.04 versus 0.42 ± 0.04, respectively (p = 0.01) (Figure 3(e)).
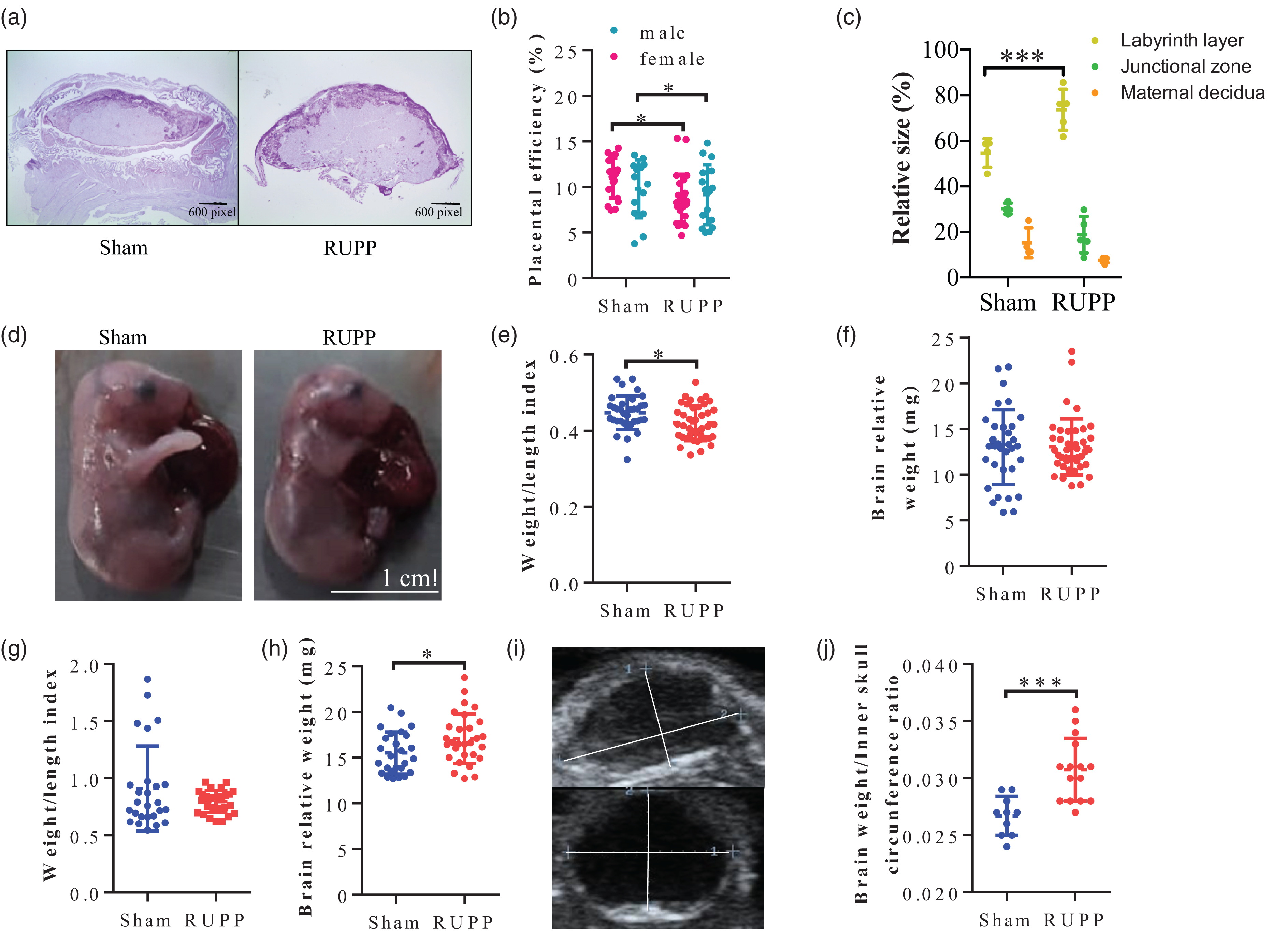
Placental dysfunction and fetal growth restriction in RUPP model in mice. (a) Representative placental images stained with periodic acid Schiff from sham and RUPP mice. (b) Placental efficiency was calculated as indicated in Methods in male and female mice. (c) The relative size of placental sections in sham and RUPP groups. (d) Representative images of the fetus at 18.5 dpc. The scale line indicates 1 cm. (e) Weight/length index in fetuses from both analyzed groups. (f) Fetal brain weight relative to respective weight at 18.5 dpc. (g) Weight/length index in pups at P5. (h) Neonatal brain weight relative to respective weight at P5. (i) Representative images of transcranial ultrasound and (j) Brain edema is estimated by brain weight and inner skull circumference ratio. dpc: days post coitum. Data in the plot graph represent the mean ± standard deviation. n = 8 dams per group. And n = 32 neonates per group. All analyses of the pups included one to three males and females per litter in each group. (*p < 0.05; ***p < 0.001).
Histological analysis of the placenta showed that the percentage spanned by the labyrinth was more prominent in the RUPP than in the sham group (73.6 ± 9.0% versus 54.6 ± 6.3%, respectively, p < 0.001) (Figure 3(c)). In contrast, no significant difference between groups was observed among other placental regions, including maternal decidua and junction zone.
Reduced fetal weight in RUPP offspring was no longer significant at P5 compared with sham offspring (Figure 3(g)). At this stage, the relative brain weight was higher in pups from RUPP than in the sham group (17.08 ± 2.72 versus 15.52 ± 2.29 mg, respectively, p = 0.03) (Figure 3(h)). While echography analysis of the ratio between brain weight and inner skull circumference, indicative of cerebral edema, was significantly higher in pups of RUPP mice than in their respective counterpart in the sham group (p < 0.001) (Figure 3(i) and (j)).
Cerebral perfusion in offspring of RUPP
Blood perfusion in the neonatal brain microcirculation (P5) was evaluated using Laser Doppler in 4 regions of interest (ROI, see Methods). In addition, we considered an experimental approach of basal perfusion followed by vasoactive examination using warm and cold solutions applied directly over the pup’s brain (Figure 4(a)). Figure 4 also presents the particular locations of the analyzed ROIs (Figure 4(b)) and representative brain images in the different experimental stimuli (Figure 4(c)).
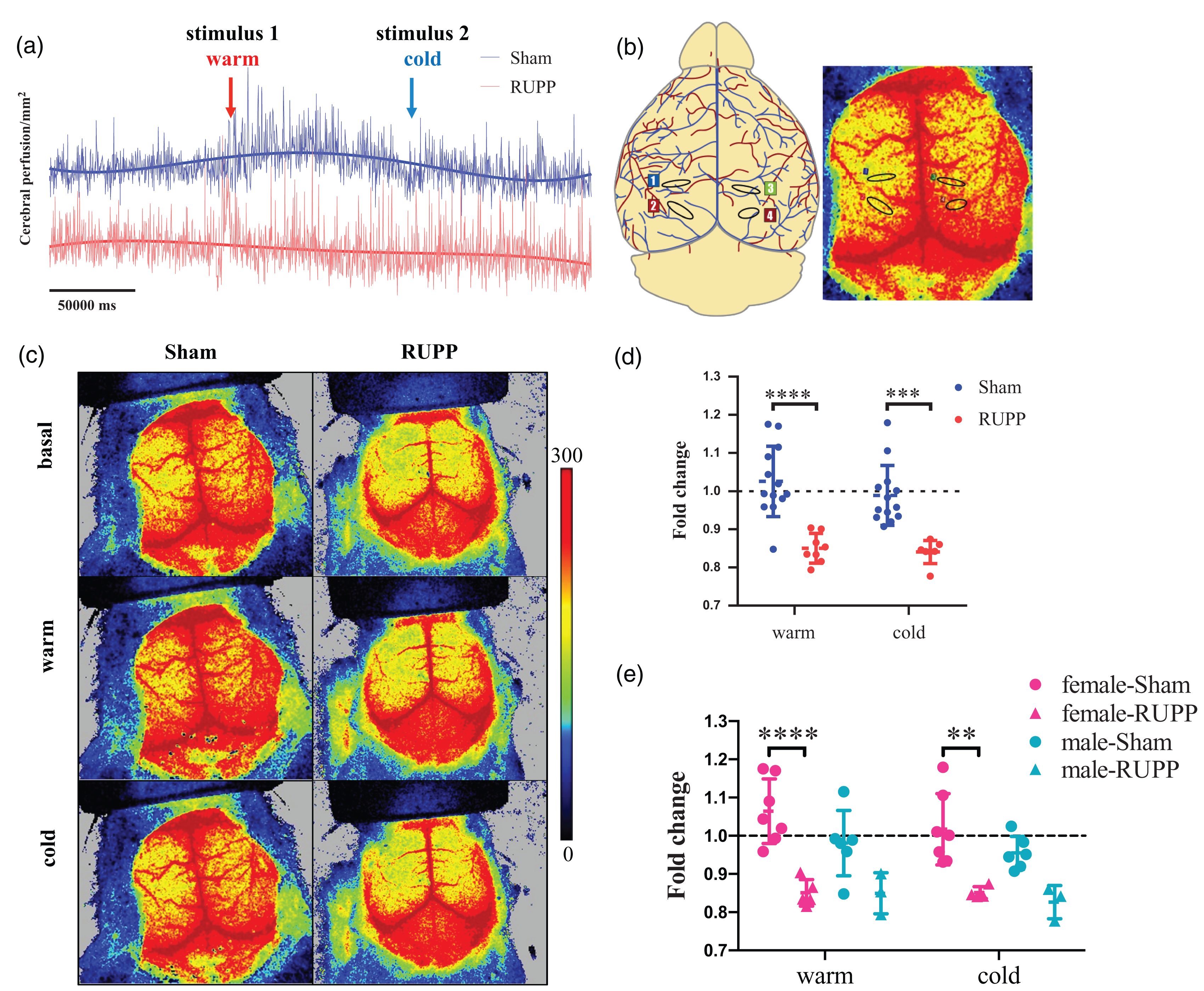
Reduced cerebral perfusion in pups of RUPP mice at postnatal day 5 (P5). (a) Representative trace of cerebral blood perfusion in basal condition, as well as after warm (1) and cold (2) stimuli. Data are normalized as indicated in Methods. (b) Regions of interest (ROI) for brain perfusion analysis (circles). (c) Representative images of cerebral perfusion in RUPP and sham mice pups. (d) Fold change of cerebral perfusion and (e) Sex-associated differences in the vascular reactivity to warm and cold stimulus. Data in the plot graph represent the mean ± standard deviation. n = 16 per group. All analyses of the pups included one to three males and females per litter in each group. (**p < 0.01; ***p < 0.001; ****p < 0,0001).
Basally, pups of RUPP mice exhibited reduced cerebral microvascular perfusion compared with their respective counterpart in the sham group (1.00 ± 0.09 versus 0.85 ± 0.04, respectively, p < 0,0001). These differences were independent of the analyzed ROI (Fig S2).
Further analysis of the frequency components in the basal brain microvascular perfusion signal using DFT found a lower metabolic component (2.07% versus 1.26%, respectively, p < 0,0001); while a higher myogenic component (4.33% versus 3.86%, respectively, p = 0.01) in pups of RUPP mice than sham mice (Figure 5(a)). Interestingly, a sex-differential response was found only in the myogenic component. Thus, while it was significantly increased in RUPP's male pups, there were no significant changes in the female pups compared to their respective counterpart in the sham group (Figure 5(b)). Therefore, basally reduced brain perfusion observed in pups of RUPP mice was more likely due to low contribution of the metabolic component associated with lack of contribution of the myogenic component in the female pups.
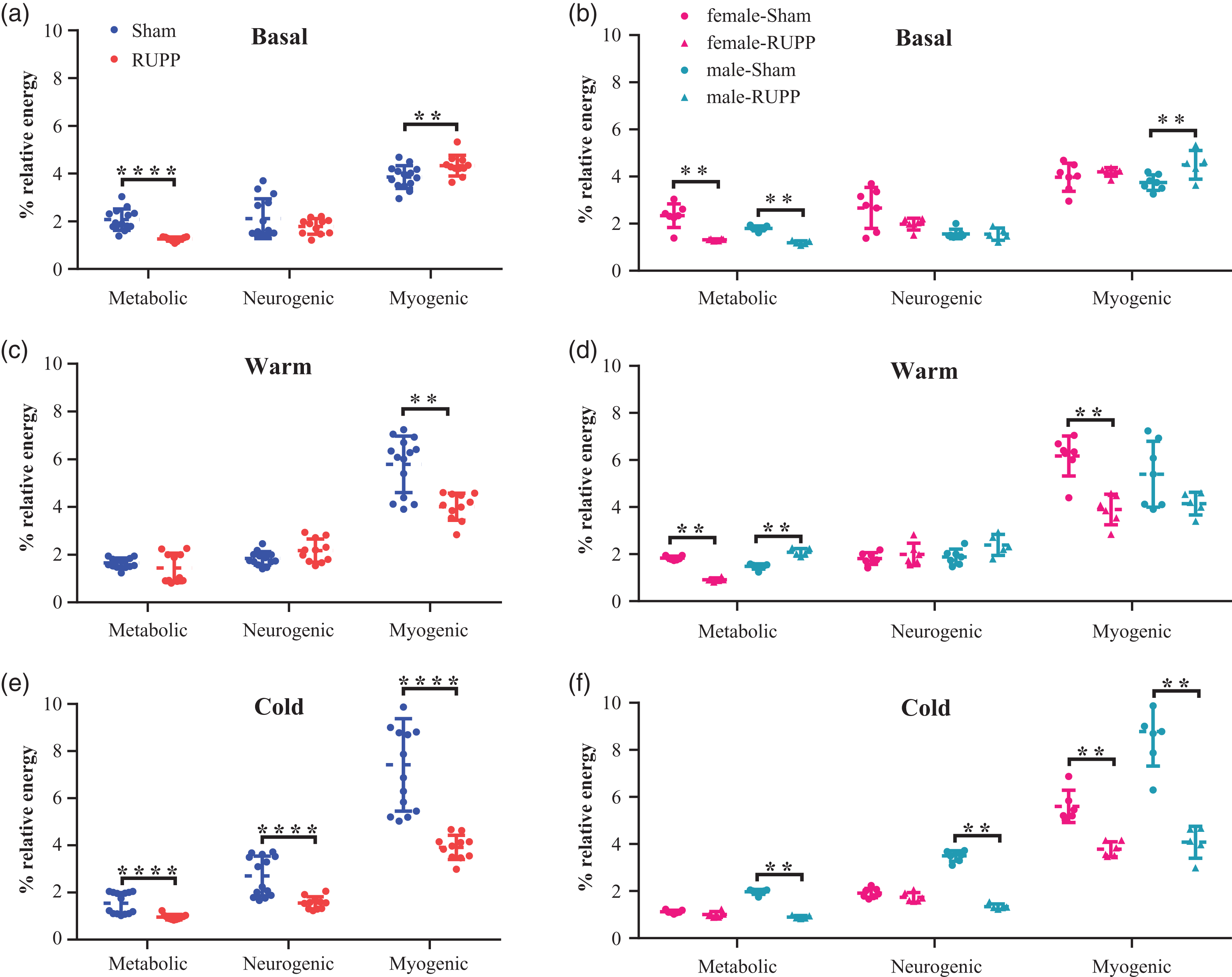
Analysis of the frequency components in the brain microvascular perfusion signal using discrete Fourier Transform (DFT). Relative energy distribution of wavelet components, including metabolic, neurogenic, and myogenic, of the perfusion signal evidenced in RUPP and sham pups. (a) Component analysis at Basal conditions. (b) Or Basal condition separated by sex. (c) Component analysis after warm-induced vascular response. (d) Or warm-induced vascular response separated by sex. (e) Component analysis after cold-induced vascular response and (f) Or cold-induced vascular response separated by sex. Data in the plot graph represent the mean ± standard deviation. n = 16 per group. All analyses of the pups included one to three males and females per litter in each group. (**p < 0.01; ****p < 0.0001).
In addition, impaired brain microvascular vasoreactivity in pups of RUPP mice was also observed. Thus, reduced response to warm (0.85 ± 0.03 versus 1.02 ± 0.09, respectively, p < 0,0001) and cold stimulus (0.84 ± 0.03 versus 0.98 ± 0.07, respectively, p < 0.001) (Figure 4(d)) was found in pups of RUPP mice compared with the respective counterpart in the sham group. Furthermore, when we analyzed the data considering the pup’s sex, the impaired microvascular reactivity remains significantly reduced only in female pups (Figure 4(e)). In addition, no significant differences were found between female and male pups within the groups in any stimuli.
When we analyzed the frequency components using DFT, differential changes in the myogenic, neurogenic and myogenic components were observed between the warm (Figure 5(c)) and cold (Figure 5(e)) stimuli in pups of RUPP mice. Thus, the reduced perfusion induced with warm stimuli was most likely associated with a reduced myogenic component in pups of RUPP mice than in sham mice (4.01% versus 5.78%, respectively, p = 0.0001) (Figure 5(d)). Interestingly, when we performed the analysis by sex, the reduced participation of the myogenic component observed in RUPP offspring after the warm stimulus (Figure 5(c)) remained significantly low only in female pups of RUPP mice (Figure 5(d)). Nevertheless, contrary in male pups of RUPP mice, reduced participation of the metabolic component was observed in the female offspring of RUPP mice compared with their respective counterpart in the sham group. Therefore, reduced warm-induced vascular response in female pups of RUPP mice was more likely associated with the reduced myogenic component. Although, the metabolic component may also contribute.
Cold stimuli generated sex-dependent significant differences, which were less evident in the basal condition or warm stimuli (see Table Supplementary S1). Thus, a significant difference in all three analyzed wavelets was observed after the cold stimuli between female and male pups in the sham group. Interestingly, this sex dimorphism was absent in the RUPP group.
Furthermore, the reduced cold-induced perfusion found in female pups of RUPP mice was associated with lower participation of the myogenic component. Additionally, a sex-related response was evident with this stimulus. Then, although all three analyzed components of the perfusion signal were reduced in male pups of RUPP mice (Figure 5(e)), there were no significant differences in the metabolic and neurogenic components in the female pups. Therefore, reduced vascular response to cold stimulus in female pups of RUPP is more likely mediated by reduced participation of the myogenic component than the participation of metabolic or neurogenic components (Figure 5(f)). Also, sex-dimorphism in the cold-induced response was absent in RUPP offspring.
Angiogenic-related proteins and pathways involved in cerebral tissue response
To analyze whether observed changes in the brain perfusion were associated with changes in the brain angiogenesis environment, we performed an angiogenic protein array using brain extraction in 18.5 dpc and P5. Of 53 proteins analyzed, two proteins at 18.5 dpc, while 17 proteins at P5 were differentially expressed in the pup’s brain of RUPP mice compared with the sham group (Figure 6(a) and (b)) (Table Supplementary S2). Thus, downregulation of Adamts-1 and Igbfbp1 at 18.5 dpc was found in RUPP mice. Furthermore, nine proteins were down-regulated (Tymp, Serpinf1, Cxcl10, Dll4, Areg, Serpine1, Adamts1, Fgf1, Dpp4), and eight proteins were up-regulated (Spp1, F3, Igfbp3, Mmp9, Pf4, Hgf, Igfbp1, Timp4) at P5 (Table Supplementary S2).
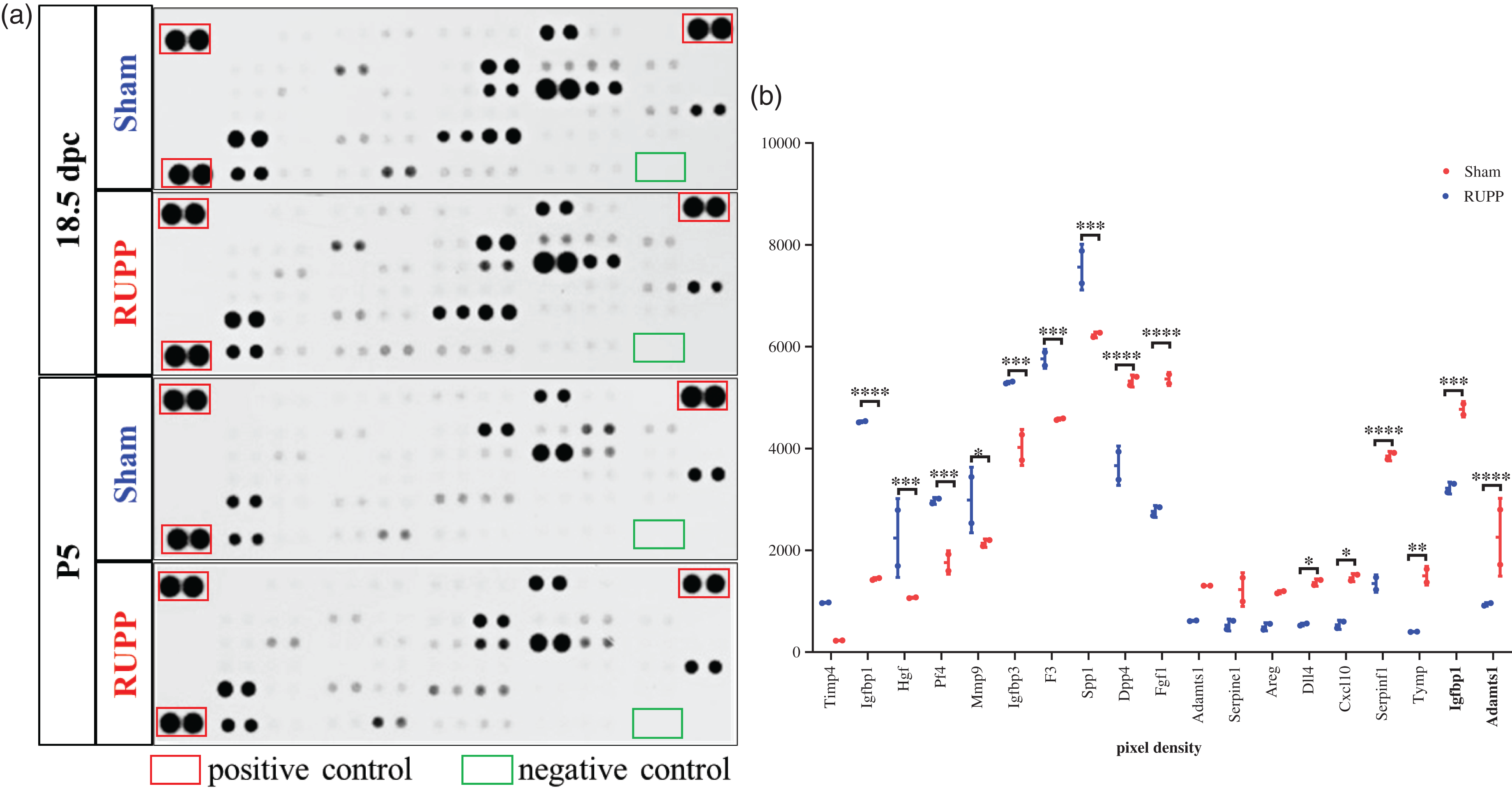
Angiogenesis-related protein hallmark in the pup’s brain of RUPP mice. (a) Representative images of dot-blot analysis of the angiogenesis-related proteins at 18.5 dpc and postnatal day 5 (P5) in RUPP and sham mice pups and (b) Pixel densitometry of the protein dots as indicated in A. Data in the plot graph represent the mean ± standard deviation. n = 3 per group. (*p < 0.05; **p < 0.01; ***p < 0.001, ****p < 0.0001).
The deregulated proteins were grouped into two biological process clusters (Figure 7(a)). At the same time, the top five signaling pathways included Cancer, PI3K-Akt, Hippo, MAPK, and Cytokine-cytokine receptor interaction (Figure 7(b)). In addition, deregulated angiogenic proteins in the pup’s brain of RUPP mice were related to the central regulatory processes, including inflammation, extrinsic apoptotic, cancer, stress, and cellular senescence.
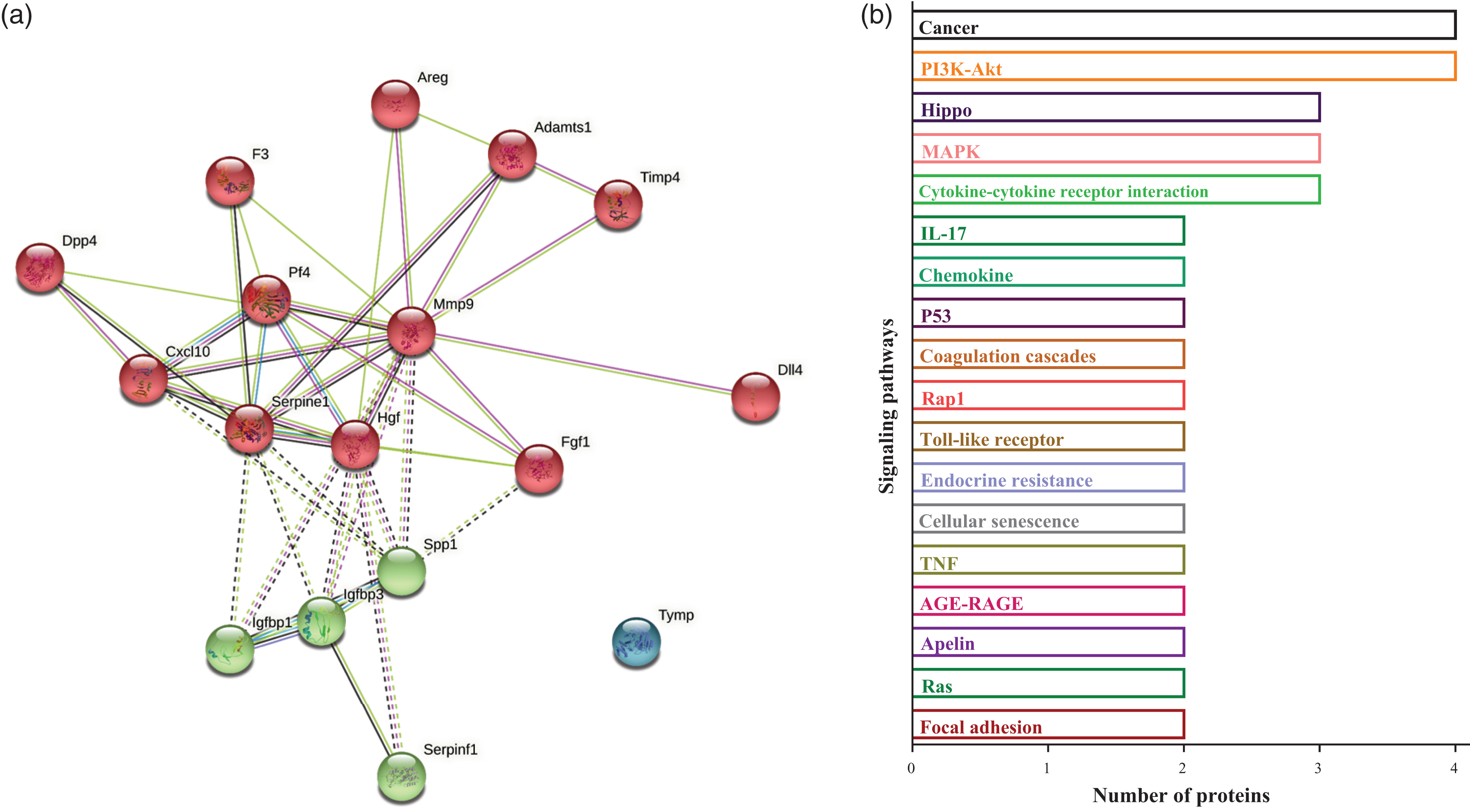
Network and pathway involved in protein dysregulation in the pup’s brain of RUPP mice. (a) Functional protein association networks for differentially expressed proteins in pup’s brain of RUPP mice at 18.5 dpc and postnatal day 5 (P5). The colors of connections suggest the type of interaction as listed in the legend. (For interpretation of the references to color in this Fig legend, the reader is referred to the web database String) and (b) Signaling pathways associated with deregulated proteins in brains of offspring RUPP.
Discussion
We present for the first time that offspring (five days postnatal, P5) from RUPP mice showed reduced basal perfusion and vasoreactivity at the brain microcirculation, particularly in female pups. The reduction in the basal perfusion observed in pups of RUPP was most likely associated with reduced participation of the metabolic component. Nevertheless, some sex-depended vascular response to warm and cold stimuli was found. Thus, while a reduction in the metabolic and myogenic components was found in female pups of RUPP mice during the warm-induced vascular response, an elevation in the metabolic component was found in male offspring. Additionally, while the reduced cold-induced vascular response observed in female pups of RUPP mice was more likely mediated by reduced participation of the myogenic component, the compensatory response (no change compared to sham) observed in male offspring was associated with reduced participation of all the three analyzed components. These changes were associated with an altered angiogenic protein profile in the pup’s brain, mainly related to inflammation, cancer, cellular stress, senescence, and apoptotic. Therefore, these results connect reduced uterine blood perfusion with altered cerebral microvascular perfusion that could generate structural and functional alterations in brain development.
RUPP as a PE model to study fetal programming
The RUPP model results in placental ischemia, a critical event in the pathophysiology of PE. 36 This damage leads to oxidative stress in the placenta that produces secretion of factors involved in endothelial cell dysfunction 37 and multisystem alterations, including structural and functional kidney alterations leading to proteinuria. 38 In the placenta, structural compensatory adaptations, including increased placental weight and enlargement of the labyrinth area, are present 35 and confirmed in our study. Although we did not further explore the cellular mechanisms responsible for these adaptations, other authors have described increased placental angiogenesis 24 or the pericytes content of the placental vessels. 26 In addition, those adaptations affect the development of placental vasculature, thereby leading to impaired maternal-fetal interactions.24,26
Under this adverse placental environment, the normal development of pregnancy is severely affected. Results showed a high rate of fetal resorption, intrauterine growth restriction, and low fetal brain weight, confirming previous findings.24,35 These findings bring the translational interpretation of fetal growth restriction and high perinatal morbidity and death observed in human preeclamptic pregnancies. 38 However, although this model has been used previously to evaluate cardiovascular programming in the offspring, 39 as far as we know, no previous publications have used it for investigating cerebrovascular adaptations/programming. RUPP model may offer a more pathophysiological point of view compared with other pharmacological,40,41 or hypoxic models 42 of PE used to study the cognitive or neurovascular impact on the offspring.
Brain vascular alterations in offspring of PE
Reduced brain weight at birth has been associated with reduced expression of neurogenesis markers or neuron proliferation in the cortex and severe impairments in spatial learning and memory. 43 In addition, we observed a high postnatal death in offspring of RUPP mice, which also presented evidence of brain edema in the echography. This result agrees with the high risk of brain edema observed in preterm babies born to women with PE. 44 Underlying mechanisms of brain edema in RUPP newborns were not investigated. However, it is feasible that placental ischemia leads to increased blood-brain barrier permeability (BBB) and neuroinflammation during gestation, which may remain after birth. 45
Nervous and vascular systems form a structural and functional interaction in the neurovascular unit. This unit comprises endothelial cells, pericytes, glia, and neurons, which enable efficient oxygen and nutrient flow across the brain. 46 In this regard, adequate brain microvascular formation (i.e., brain angiogenesis) and function are critical for cerebral development. 45 Brain angiogenesis is an active process that starts in the embryo at 9.5 days and remains postnatal. 47 After birth, the peak of brain angiogenesis in mice (particularly in the brain cortex) reaches approximately seven days postnatal. It is mainly due to increased migration rather than proliferation of brain endothelial cells. 48 Currently, no information regarding brain angiogenesis in offspring born to PE is available. Although aberrant maternal angiogenic signaling milieu typical in PE may adversely affect brain vascular growth, reduced vessel radii in occipital and parietal lobes were found, 13 and probably preceding brain structural alterations observed in the offspring. 13
This study observed a reduced basal brain blood perfusion and impaired brain microvascular vasoreactivity in pups of RUPP mice. We interpreted these findings as reduced angiogenesis and/or vascular remodeling in offspring of RUPP mice. Furthermore, in another model of PE using a high cholesterol diet in rats, the function of cortical parenchymal arterioles was impaired and compromised the ability of blood flow to match neuronal metabolic demand in the offspring. 22 These brain-vascular alterations observed early in life could contribute to white matter lesions and early-onset cognitive decline. 11 Indeed, an adverse environment can lead to small cerebral vessels losing their physiological capacity to dilate and constrict in response to variations of systemic blood pressure and contributing to ischemic damage of the brain parenchyma and BBB disruption. 23
Furthermore, there is a differential brain vascular response in female and male offspring of RUPP mice, being female mice the more affected with reduced cerebral perfusion. In addition, sex dimorphism was present in the wavelet analysis of the cold-induced vascular response only in the sham group. Although no studies have directly measured sex differences in cerebral blood perfusion in offspring of PE, previous research hypothesized that females have shown more significant cognitive impairments on frontal-dominant tasks and reported more symptoms of post-brain injury. 49 Even female fetal sex was associated with an altered maternal inflammation, which may be associated with early preterm PE. 50 Also, sex-specific alterations in fetal growth, microvascular structure, and function were found in children born to PE. 51 We remark that these sex-specific differences are evident in the immediate newborn period, a time of physiologic instability after birth, which pathophysiology and consequences need further investigation. However, they may constitute a consequence of structural microvascular changes (microvascular density and microvascular compliance) and differences in endothelial function observed in offspring from PE.5,51 Despite this, the precise role of sex in normal neurodevelopmental or programming by PE exposure is not clear yet and requires more in-depth clinical and preclinical studies. 52
In general, cerebral blood flow is regulated by metabolic, glial, neural, and vascular factors. 53 In the case of neurons, they regulate vascular function via signaling through astrocytes, smooth muscle cells, and pericytes in response to neuronal activity. 46 In this study, analysis of the components of perfusion signal allows identifying that RUPP offspring showed reduced participation of the metabolic but increased participation of the myogenic components. Interestingly, this elevated participation of the myogenic component was not evident in female pups of RUPP. Therefore, they suggest that this component would most likely be impaired in female pups. Indeed, when we analyzed the vascular reactivity, either vasodilation (warm solution) or vasoconstriction (cold solution) stimuli, the myogenic component was severely reduced in female pups of RUPP. We hypothesize that this reduction in the myogenic component may reflect structural alterations in the microcirculation.
Results suggest sex-specific perfusion differences after inducing vasoreactivity with warm or cold stimuli. We remark participation of the myogenic component, in which sex-specific differences were found during the basal and warm-induced vascular response in offspring of RUPP mice. While in the cold-induced vascular response, sex-specific differences were found in the metabolic and neurogenic components. These findings generate intriguing questions, including; 1) whether brain vascular maturation in pups of RUPP mice is sex-depended. 2) Whether vascular remodeling is also impaired in RUPP offspring. 3) Whether coupling between endothelial and smooth vascular cells (or pericytes), the critical cells involved in the myogenic response, would be mainly affected in female offspring. 4) Whether the potential negative impact of these sex-associated differences observed in pups of RUPP mice impairs the autoregulatory capacity of brain vessels or the function of the BBB. Future research should take into account these questions.
Potential brain parenchyma response to brain vascular alterations
The neurobiology of cognitive alterations in offspring of mothers with PE7,53,54 is poorly understood. 55 Compatible with described alterations in brain perfusion, we also found a differential expression of angiogenic proteins that participate in signaling pathways such as inflammation, cancer, cellular stress, senescence, and apoptotic in the pup's brain of RUPP mice. In PE, cellular senescence, apoptosis resistance, and pro-inflammatory phenotype have been related mechanistically to mesenchymal stem cell dysfunction. 56 Moreover, impaired inflammation and nutrient access could constitute critical factors in brain development and cognitive deficiencies observed in offspring born to PE pregnancies. 9 Furthermore, angiogenesis is a complex process that requires regulation of sprouting, endothelial cell proliferation, migration, and regulation branch elongation of an epithelium, keratinocyte migration, and monocyte chemotaxis; 57 then it is not surprising that multiple metabolic pathways would be affected in offspring from PE. In addition, the interaction between neuronal activity and brain microcirculation is critical for adequate angiogenesis and vascular remodeling.48,57 The brain adapts to adverse conditions during gestation, secreting a series of molecules to maintain adequate neurovascular development. However, it may also generate cellular adaptations, leading to impaired brain development. We present indirect evidence that these brain responses persist after delivery.
We acknowledge some limitations in our study. For instance, the RUPP model generated in our lab showed no differences in the levels of Plgf or sFlt-1. Since those markers were analyzed using different techniques, we could not calculate a ratio as was used in other studies. In addition to that, future studies should analyze brain vascular alterations from fetal to postnatal age. Our laboratory is currently working in this area and will be presented in future manuscripts. Likewise, we evaluated cerebral blood perfusion in surviving mice to bring our findings closer to what would happen in postnatal life in the brains of children of mothers with PE. Then if these alterations are adaptive or pathophysiological responses need to be further investigated.
Additionally, since there was a significantly reduced gestational length in the RUPP group and preterm birth affects normal brain development, we acknowledge this limitation in the analysis of the offspring. Therefore, future studies should correct postnatal analysis considering the gestational age at birth. Also, we acknowledge that future studies should include cognitive analysis of the offspring of RUPP dams to correlate them with the alterations observed in brain perfusion.
In summary, we presented the first evidence of abnormal cerebral microvascular perfusion and reactivity in female offspring of RUPP mice, which are associated with impaired participation of the myogenic component. Also, a particular hallmark of brain angiogenic protein expression was found in pups of RUPP mice (P5) associated with the critical regulatory process, including inflammation, cancer, cellular stress, senescence, and apoptosis. In a pathophysiological model of PE, our results reinforce the participation of brain vascular alterations as an underlying mechanism that could explain brain structural and cognitive alterations observed in children born to PE.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221121872 - Supplemental material for Abnormal cerebral microvascular perfusion and reactivity in female offspring of reduced uterine perfusion pressure (RUPP) mice model
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221121872 for Abnormal cerebral microvascular perfusion and reactivity in female offspring of reduced uterine perfusion pressure (RUPP) mice model by Evelyn Lara, Nathaly Rivera, Alejandro González-Bernal, Daniela Rojas, Daniela López-Espíndola, Andrés Rodríguez and Carlos Escudero in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Fondecyt Postdoctorado 3190475 (EL) and Fondecyt Regular 1200250 (CE).
Acknowledgements
The authors would like to thank the researchers belonging to Vascular Physiology Laboratory, Universidad del Bio-Bio, and GRIVAS health researchers for their valuable input during this project.
Authors’ contributions
EL model establishment. EL and NR experimental work, data collection, and analysis. A.G-B. Transcranial ultrasound. DR and DL histochemical analysis. EL wrote the draft of the manuscript. AR performs Fourier analysis. CE conceptualized the study and led the research group. All co-authors approved the final version of this manuscript.
Data availability
Raw data and original images are available upon request. Please contact Dr. Carlos Escudero.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest concerning this article's research, authorship, or publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
