Abstract
Transient ischemic attacks (TIAs) before an acute ischemic stroke (AIS) could induce ischemic tolerance (IT) phenomena. with an endogenous neuroprotective role (Ischemic preconditioning. IPC). A consecutive prospective cohort of patients with AIS were recruited from 8 different hospitals. Participants were classified by those with non-previous recent TIA vs. previous TIA (within seven days. TIA ≤7d). A total of 541 AIS patients were recruited. 40 (7.4%). of them had previous TIA ≤7d. In line with IPC. patients with TIA ≤7d showed: 1) a significantly less severe stroke at admission by NIHSS score. 2) a better outcome at 7–90 days follow-up and reduced infarct volumes. 3) a specific upregulated metabolomics/lipidomic profile composed of diverse lipid categories. Effectively. IPC activates an additional adaptive response on increasing circulation levels of structural and bioactive lipids to facilitate functional recovery after AIS which may support biochemical machinery for neuronal survival. Furthermore. previous TIA before AIS seems to facilitate the production of anti-inflammatory mediators that contribute to a better immune response. Thus. the IT phenomena contributes to a better adaptation of further ischemia. Our study provides first-time evidence of a metabolomics/lipidomic signature related to the development of stroke tolerance in AIS patients induced by recent TIA.
Keywords
Introduction
Ischemic preconditioning (IPC) consists of brief periods of ischemia that confers protection against later episodes of prolonged ischemia (ischemic tolerance [IT]). 1 The IT-induced mechanisms evoked by previous brief ischemia appear complex and involve unique cellular gene expression changes and metabolic and signaling pathways. 1 The IT could be induced either by local. global. and remote IPC.1–3
IT could be activated in clinical scenarios by a recent transient ischemic attack (TIA) before an acute ischemic stroke (AIS).4–7 It has been reported that patients with previous TIA before an AIS had better outcome4–7 and smaller infarct volumes6,8 than patients without previous TIA. although data from two studies showed no association between previous TIA and low stroke severity.9,10
Metabolomics. including lipidomic analysis. is the comprehensive analysis of a biological sample with high-sensitivity to characterize changes in low-molecular-weight molecules. The plasma metabolome study arises as a powerful functional tool to describe phenotypes in both physiological and pathological states. 11 Consequently. metabolomics emerges as a promising approach to propose potential prognostic and recurrence biomarkers of cerebrovascular diseases 12 and a plasma biomarker related to neuroimaging findings. 13
The present study was designed to address the IT phenomenon by the effect of a previous TIA on AIS patients' outcome and to describe its metabolic signature.
Materials & methods
Study participants – cohort description
The present study follows the STROBE statement. 14 A cohort of AIS patients was consecutively and prospectively recruited. Patients were recruited between January 2011 and January 2016 following the same procedure (TIA/IS diagnose. neuroimaging confirmation. blood extraction time) from eight Hospitals in Spain (Hospital Universitari Arnau de Vilanova-Lleida. Hospital Universitario-Pamplona. Hospital del Mar-Barcelona. Hospital de son Espases-Mallorca. Hospital Parc Taulí-Sabadell. Hospital de Sant Pau-Barcelona. Hospital Ramón y Cajal-Madrid. Hospital de Albacete). A neurologist attended AIS patients during the first 24 h after the onset of symptoms. All recruited patients underwent neuroimaging diagnosis. Patients with symptoms that persisted less than 24 h without clear evidence of acute ischemic lesion in the neuroimaging were excluded from the study. Other exclusion criteria were a modified Rankin Scale Score (mRS) greater than 2. more than 24 h after symptoms onset. or patients under 18 years old. Blood from patients was collected immediately upon hospital arrival at the emergency room by standard venipuncture. Plasma was obtained after centrifugation 4°C at 1500 g for 10 min and aliquoted into cryovials for prompt storage at −80°C. In all cases. blood samples were obtained before intravenous fibrinolysis or thrombectomy.
Patients were classified etiologically according to TOAST criteria (Trial of Org 10172 in Acute Stroke Treatment) 15 as a large-artery occlusive disease (LAA). small-vessel disease (SV). cardioembolism (CE). other cause (OC). or undetermined cause (UND). The following patient's characteristics were collected: age at admission. gender. vascular risk factors. prior treatments. premorbid mRS. and stroke severity on the National Institutes of Health Stroke Scale (NIHSS) on admission and after seven days. Oxfordshire topographic classification was also assessed.
The Ethics Committee approved this study protocol of the Hospital Universitari Arnau de Vilanova (approval code: 928). Written informed consent was obtained from the patients or patients' surrogates in all participating Hospitals. All procedures followed the Declaration of Helsinki guidelines developed by the World Medical Association regarding ethical principles for medical research involving human subjects.
According to previous evidence. 7 patients were classified and analyzed into two groups according to the time interval between prior TIA and IS: i) TIA within the preceding seven days. and ii) non-TIA or TIA before the preceding seven days. Families of patients with language impairment or disturbances of consciousness and their general practitioners were interviewed to identify prior TIA events.
Previous TIA was defined as a reversible episode of neurological deficit of ischemic origin that resolved totally within 24 h. 16 All baseline data were centrally monitored. and queries were sent to each center's enrolling stroke neurologists. The primary outcome was the degree of functional impairment at three months. measured by mRS ≤1 at 90 days. We employed a face-to-face interview in each hospital to follow up with patients.
Final infarct volume was measured in 396 (73.2%) patients by diffusion-weighted magnetic resonance imaging (DWI) performed within seven days (median 2.8 days: interquartile range: 1.6 to 4.6). One neuroradiologist blinded to clinical features established the presence of DWI abnormalities. Furthermore. OsiriX v4.0 semi-automated segmentation tool and imaging software (Bernex. Switzerland) was used to calculate the total volume of DWI abnormal signal intensity. Polygon tool was used to assess the presence of abnormalities. regions of interest (ROIs) delineated between segmented slices and automatically interpolated. and the infarct volume determined. 13
Clinical lipid and inflammatory biomarkers panel
Lipid profile (total cholesterol, HDL cholesterol, calculated LDL and triglycerides), fibrinogen, leukocytes, glycemia, and creatinine levels were obtained from clinical history reports by a routine blood test at hospital admission. Interleukin-6 (IL-6), N-terminal pro-B type natriuretic peptide (NT-proBNP), high-sensitivity C-reactive protein (hsCRP), high sensitive troponin (hsTroponin), neuron-specific enolase (NSE), and S-100b protein levels were assessed in plasma samples (Hoffmann-La Roche. Basel. Switzerland) by an electrochemical chemiluminescence immunoassay using the COBAS 6000 e601 (Hoffmann-La Roche. Basel. Switzerland) following previously published protocol. 17
Metabolomic and lipidomic sample processing
Non-targeted metabolomic and lipidomic profiling was performed on an Agilent 1290 LC system coupled to an electrospray-ionization quadruple time of flight mass spectrometer (Q-TOF. 6520 instrument. Agilent Technologies. Barcelona. Spain). All samples were analyzed together. We applied two different protocols to cover as many metabolites as possible. from more polar ones like amino acids to more apolar like cholesterol esters (CE) or triglycerides (TG).
For metabolomics analysis. plasma samples were processed as described previously. 12 Briefly. 30 μl of cold methanol (containing phenylalanine-C13 as an internal standard) were added to 10 μl of plasma. incubated for one hour at −20°C. and centrifuged for three minutes at 12.000 g. The supernatant was recovered. filtered with a 0.2 μm Eppendorf filter. Two µL of the extracted sample were applied onto a reversed-phase column (Zorbax SB-Aq 1.8 µm 2.1 × 50 mm; Agilent Technologies) equipped with a pre-column (Zorbax-SB-C8 Rapid Resolution Cartridge 2.1 × 30 mm 3.5 µm; Agilent Technologies) with a column temperature of 60°C. The flow rate was 0.6 mL/min. Solvent A was composed of water containing 0.2% acetic acid. and solvent B was composed of methanol 0.2% acetic acid. The gradient started at 2% B and increased to 98% B in 13 min and held at 98% B for 6 min. Post-time was established in 5 min.
For lipidomics analysis. plasma samples were processed as described previously. 18 Briefly. to precipitate the protein fraction. five μl of Mili Q water and 20 μl of methanol were added to 10 μl of a plasma sample. After the addition. samples were shaken for 2 min. Then. 250 μl of MTBE plus internal standards were added. and samples were ultra-sounded in a water bath (ATU Ultrasonidos. Valencia. Spain) with a frequency and power of 40 kHz and 100 W, respectively, at 10°C for 30 min. Then. 75 μL of Mili Q water was added to the mixture, and the organic phase was separated by centrifugation (1.400 g) at 10°C for 10 min. The upper phase. containing all the extracted lipid species. was collected and subjected to mass spectrometry. A pool of all lipid extracts was prepared and used as quality controls (QC) as previously described. 19 Ten μl of lipid extract was applied onto 1.8 μm particle 100 × 2.1 mm id Waters Acquity HSS T3 column (Waters. Milford. MA. USA) heated at 55°C. The flow rate was 400 μl/min with solvent A composed of 10 mM ammonium acetate in acetonitrile-water (40:60. v/v) and solvent B composed of 10 mM ammonium acetate in acetonitrile-isopropanol (10:90. v/v). The gradient started at 40% B and reached 100% B in 10 min and held for 2 min. Finally, the system was switched back to 40% B and equilibrated for 3 min. as previously described. 20
Metabolomic and lipidomic data acquisition and analysis
Data were collected in positive electrospray mode time of flight operated in full-scan mode at 50–3000 m/z in an extended dynamic range (2 GHz), using N2 as the nebulizer gas (5 L/min. 350°C). The capillary voltage was 3500 V with a scan rate of 1 scan/s. The ESI source used a separate nebulizer for the continuous. low-level (10 L/min) introduction of reference mass compounds 121.050873 and 922.009798. used for continuous. online mass calibration. Mass Hunter Data Analysis Software (Agilent Technologies. Barcelona. Spain) was used to collect the results. and Mass Hunter Qualitative Analysis Software (Agilent Technologies. Barcelona. Spain) to obtain the molecular features of the samples. representing different. co-migrating ionic species of a given molecular entity using the Molecular Feature Extractor algorithm (Agilent Technologies. Barcelona. Spain). as described. 12 We selected samples with a minimum of 2 ions. MassHunter Mass Profiler Professional Software (Agilent Technologies. Barcelona. Spain) was used to select. align. and filter molecular features. Multiple charge states were considered for metabolite profiling only in lipidomics. Compounds from different samples were aligned using a retention time window of 0.1% ± 0.25 minutes and a mass window of 30.0 ppm ± 2.0 mDa. We selected only those present in at least 50% of one group's samples and corrected for individual bias.
Identities were confirmed based on exact mass. retention time. isotopic distribution. and MS/MS spectrum using public databases such as Metlin. 21 HMDB. 22 and LipidMatch. 23
Metaboanalyst platform 24 was used to perform multivariate statistics (partial least squares discriminant analyses (PLS-DA) of the extracted features. R software was used to find potential biomarkers of TIA neuroprotection prior to AIS and perform Spearman correlations. Robust linear models were performed using the MASS package's rlm function. considering the metabolite or lipid intensity as a response to the group (previous TIA vs. non-previous TIA). P-values for the regression coefficients were obtained by bootstrapping using the car package's Boot function and were adjusted by Benjamini-Hochberg's False Discovery Rate. In order to adjust for potential confounding. samples were weighted using the overlap weighting method. which has been demonstrated to produce an unbiased treatment effect estimate. with lower variance compared with other propensity score based weighting methods. 25 Propensity scores for TIA ≤7 days were estimated using a logistic regression. which included baseline characteristics (age. sex. smoking habits. hypertension. diabetes mellitus. hyperlipidemia. previous atrial fibrillation and ischemic heart disease). previous treatment (anticoagulants. antiplatelets. statins. antihypertensives and antidiabetics) and stroke etiology (LAA/non-LAA). Chord diagram for correlational analyses between selected lipid species was performed using Circos Table Viewer (http://mkweb.bcgsc.ca/tableviewer/visualize/).
Statistical analysis
Sample size was initially calculated considering studies with a clinical endpoint of mRS < 2 or similar: Moncayo et al. 26 Schaller et al. 27 and Wieh et al. 28 We initially calculated a sample size of 490 subjects to achieve 80% power to detect differences of 15% between groups using a two-sided binomial test at significance level of 0.05. assuming that assuming that 56% of non-previous TIA patients will experience good outcome. Finally. we determine a temporal endpoint to increase the number of recruited patient in order to analyse previous TIA patients subgroups.
Data are presented either as the mean ± standard deviation. the median (interquartile range). or percentage. Categorical variables were expressed as numbers and percentages and compared with Fisher's exact or ordinal chi-square tests. as appropriate. Continuous variables were expressed as medians and interquartile ranges or as means and standard deviations and were compared with the use of Student's t-test. ANOVA. and Mann-Whitney test. Univariate analyses were performed to detect variables associated with the presence of previous TIA ≤7 days and with a mRs ≤1 at 3 months. We used multivariable logistic backward stepwise regression analysis to obtain adjusted odds ratios. Variables for which P < 0.10 in univariate testing were included. Statistical analysis was performed using SPSS software version 20 (SPSS. Chicago. IL) or Stata 11 statistics package (StataCorp. College Station. TX). Statistical significance was considered when p < 0.05.
Data availability
Anonymized data will be shared by request from any qualified investigator on reasonable requests.
Results
The cohort included 541 patients (151 excluded patients). Of the 541 enrolled patients. 40 (7.4%) patients had a previous TIA ≤7 days (Figure 1). Previous TIA was ipsilateral to the subsequent stroke in 37 (92.5%) cases and unique in 23 (59.0%) cases (Table 2).
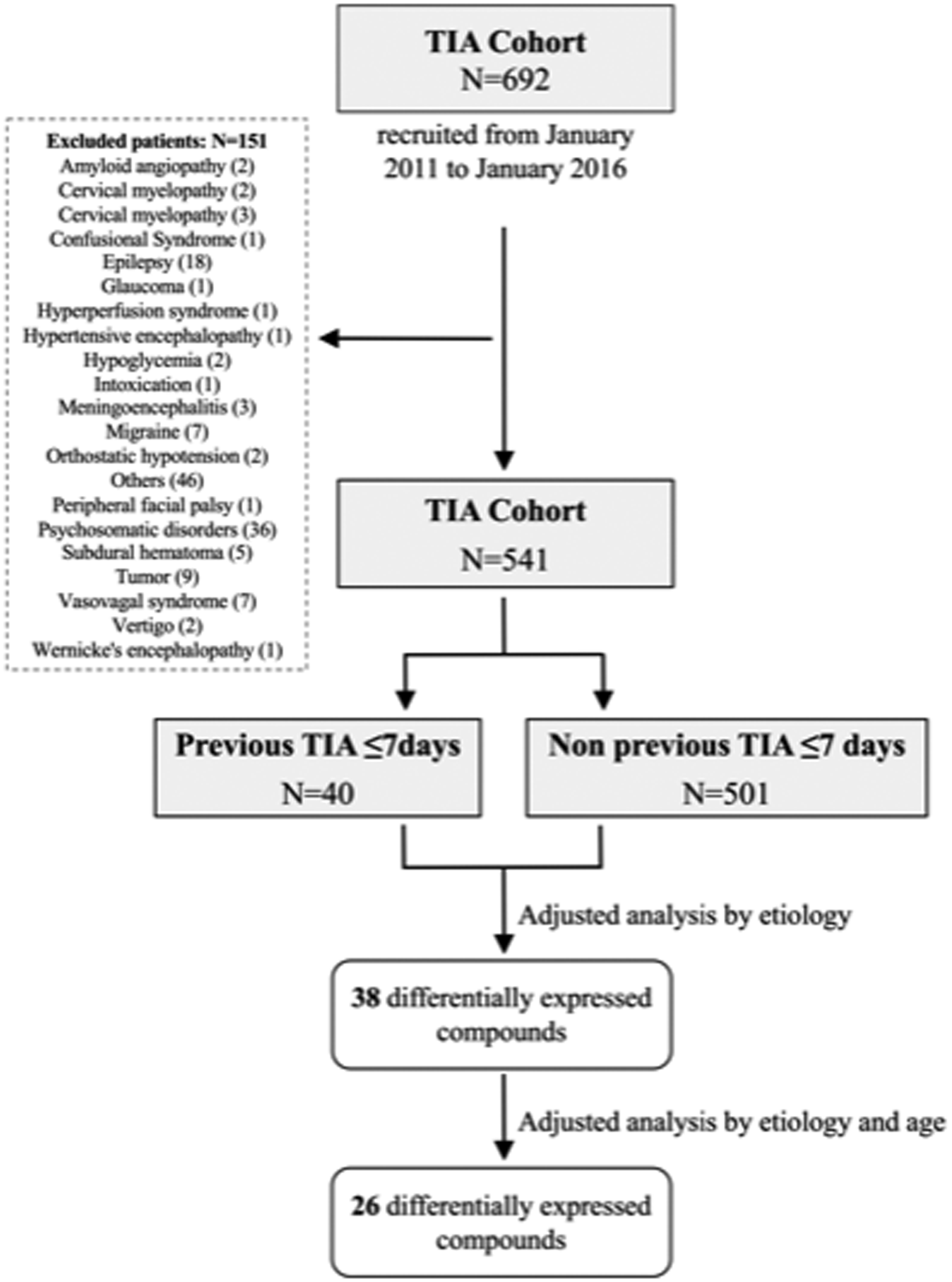
Flow diagram of recruited cohort. excluded patients. and the number of final included patients in the metabolomic and lipidomic analysis.
Age. basal NIHSS and LAA were independently related to previous TIA: Odds ratio 0.97 (95% CI: 0.95–1.00). P = 0.041; OR 0.88 (95% CI 0.81–0.95). P = 0.002; and OR 2.11 (1.01–4.43). P = 0.048 respectively. Furthermore. the outcome at 7 and 90 days measured by disability or dependence in daily activities (mRS) and NIHSS at seven days was better in previous TIA ≤7d patients than non-previous TIA patients (Table 1). Moreover. previous TIA ≤7d patients' infarct volumes were significantly reduced (75% lower. median volume). Additionally. IL-6. NT-proBNP. S100B. and hs-troponin were decreased in the previous TIA ≤7d group (Table 1).
Clinical characteristics of stratified participants by non-previous TIA vs. previous TIA ≤7 days.
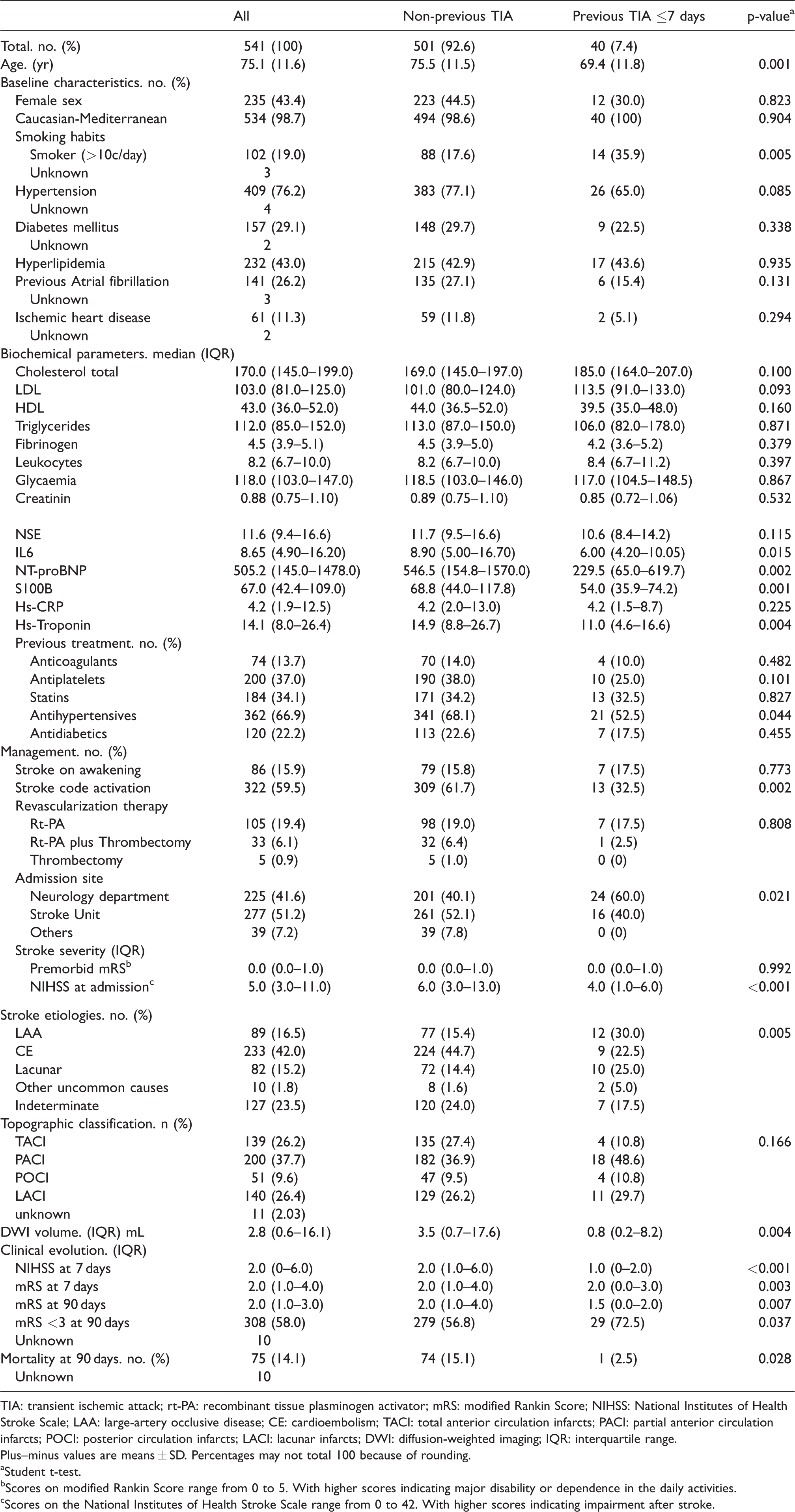
TIA: transient ischemic attack; rt-PA: recombinant tissue plasminogen activator; mRS: modified Rankin Score; NIHSS: National Institutes of Health Stroke Scale; LAA: large-artery occlusive disease; CE: cardioembolism; TACI: total anterior circulation infarcts; PACI: partial anterior circulation infarcts; POCI: posterior circulation infarcts; LACI: lacunar infarcts; DWI: diffusion-weighted imaging; IQR: interquartile range.
Plus–minus values are means ± SD. Percentages may not total 100 because of rounding.
aStudent t-test.
bScores on modified Rankin Score range from 0 to 5. With higher scores indicating major disability or dependence in the daily activities.
cScores on the National Institutes of Health Stroke Scale range from 0 to 42. With higher scores indicating impairment after stroke.
Comparison of clinical characteristics according to 90-day modified Rankin score outcome.
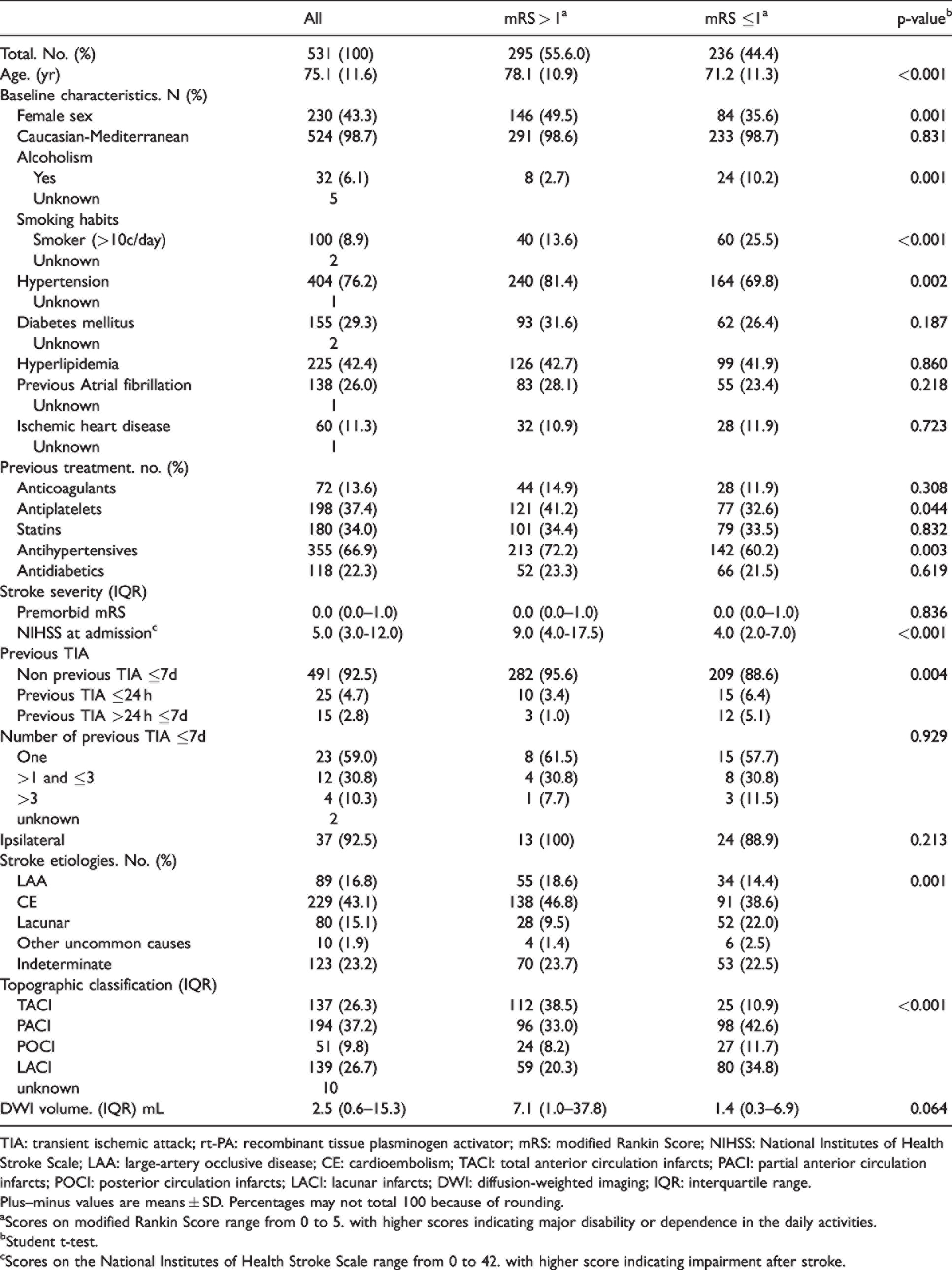
TIA: transient ischemic attack; rt-PA: recombinant tissue plasminogen activator; mRS: modified Rankin Score; NIHSS: National Institutes of Health Stroke Scale; LAA: large-artery occlusive disease; CE: cardioembolism; TACI: total anterior circulation infarcts; PACI: partial anterior circulation infarcts; POCI: posterior circulation infarcts; LACI: lacunar infarcts; DWI: diffusion-weighted imaging; IQR: interquartile range.
Plus–minus values are means ± SD. Percentages may not total 100 because of rounding.
aScores on modified Rankin Score range from 0 to 5. with higher scores indicating major disability or dependence in the daily activities.
bStudent t-test.
cScores on the National Institutes of Health Stroke Scale range from 0 to 42. with higher score indicating impairment after stroke.
Table 2 shows variables associated with mRs ≤1 at 3 months. In the multivariate regression analysis. age (OR 0.96 [95% CI: 0.94–0.98]. P < 0.001). basal NIHSS (OR 0.87 [95% CI: 0.84–0.90]. P < 0.001). LAA (OR 0.50 [95% CI: 0.29–0.85]. P = 0.011) and previous TIA ≤7d (OR 2.18 [95% CI: 1.01–4.73]. P = 0.049) were predictors of mRs ≤1 at 3 months.
Untargeted metabolomics and lipidomics analysis were performed to investigate whether previous TIA ≤7d patients had a plasma metabolome signature expressing an adaptive metabolic response with a potential neuroprotective role. The metabolomics approach revealed 18.332 molecular features. whereas in the lipidomic approach. we found 3.215. To define the most robust and reliable molecules. we only considered those present in more than 50% of the samples. Then. metabolomics approach revealed 134 molecules and lipidomics approach 201 molecules.
Multivariate statistical approach (PLS-DA) of plasma metabolites and lipids did not reveal the existence of a specific circulating signature of TIA ≤7d patients in both metabolomics and lipidomics analysis when the global metabolome and lipidome were analyzed (Figure 2(a) and (b)). However, univariate analysis revealed 53 metabolites and lipid species differentially expressed in TIA ≤7d patients (Table 3). and 4 of them obtained FDR p-value <0.05. Among the 53 differential species. 27 (51.0%) were identified based on exact mass. retention time. and/or MS/MS spectrum. mainly lipid metabolism-related species (23/27. 85%). All of the 27 identified species were increased in the TIA ≤7d group. For graphic representation. see https://aitmetllip.pythonanywhere.com/. When identified molecules were evaluated according to their families. we found 5 molecules belonging to glycerolipids (GL). 14 glycerophospholipids (GPL). 3 sphingolipids (SL). Additionally. we identified 3 indoles and derivatives. an isoleucine and an hydroxydodecanoic acid. Regarding GL. we found 2 diacylglycerides (DG(18:1/16:0). DG(34:2)). 2 oxidized triacylglycerides (OxTG(18:1/18:1/18:2);1O. OxTG(55:1);2O) and one triacylglyceride (TG(21:0/18:1/18:2)). Among GPL. we found 4 phosphatidylethanolamine (LysoPE(18:4) LysoPE(20:4). LysoPE(22:4). PE(P-38:6)). 8 phosphatidylcholine (PC(32:2). PC(34:1). PC(38:6). PC(40:6) . PC(42:5). PC(42:6). PC(O-36:4)/PC(P-36:3). PC(O-42:8)). 1 phosphatidylinositol (PI(38:4)) and 1 phosphatidylserine (PS(O-29:0)). Three of these species were lysophospholipids (LysoPE(18:4) LysoPE(20:4). LysoPE(22:4)). Concerning SL we identified two sphingomyelins (SM) (SM(d35:1). SM(d36:2) and one ganglioside. Ganglioside GM3 (d38:0)).
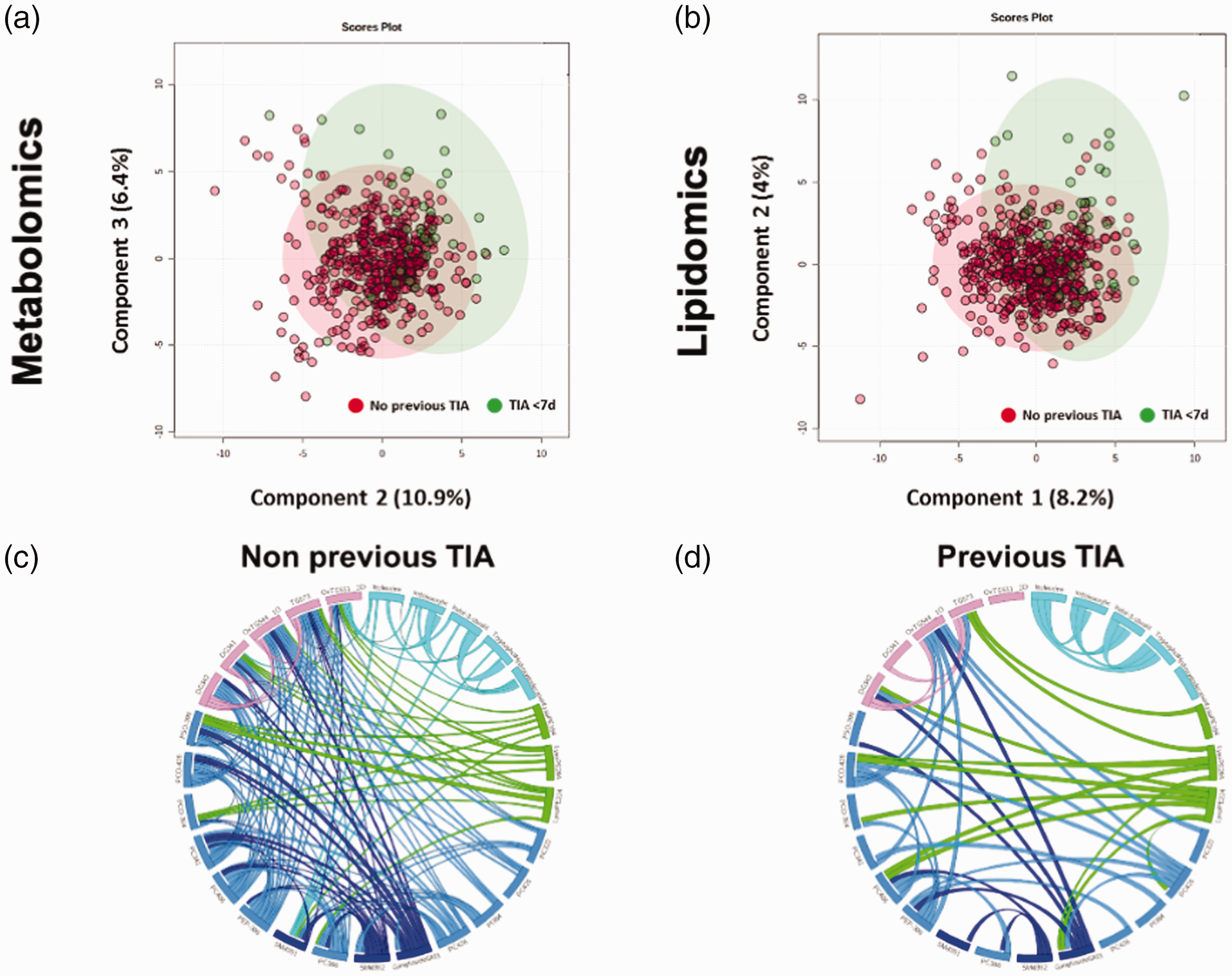
Multivariate statistics revealed that suffering a TIA ≤7 days before IS determines specific plasma metabolomics (a); and lipidomics (b) signatures. Previous TIA ≤7d patients are represented in green spots and non-previous TIA in red. PLS-DA cross-validation details are: metabolomics (Accuracy: 0.93. R2: 0.054. Q2: −0.05) and lipidomics (Accuracy: 0.92. R2: 0.063. Q2: −0.03). (c) Graphical visualization of significant differential lipid correlation in non-previous TIA compared to prior TIA ≤7 days patients (d). Each ribbon indicates a significant lipid correlation. Ribbon thickness refers to the degree of correlation (p values for Spearman's correlation <0.01).
Metabolite and lipid species differentially expressed in TIA ≤7d patients adjusted by baseline characteristics, previous treatment and stroke etiology.
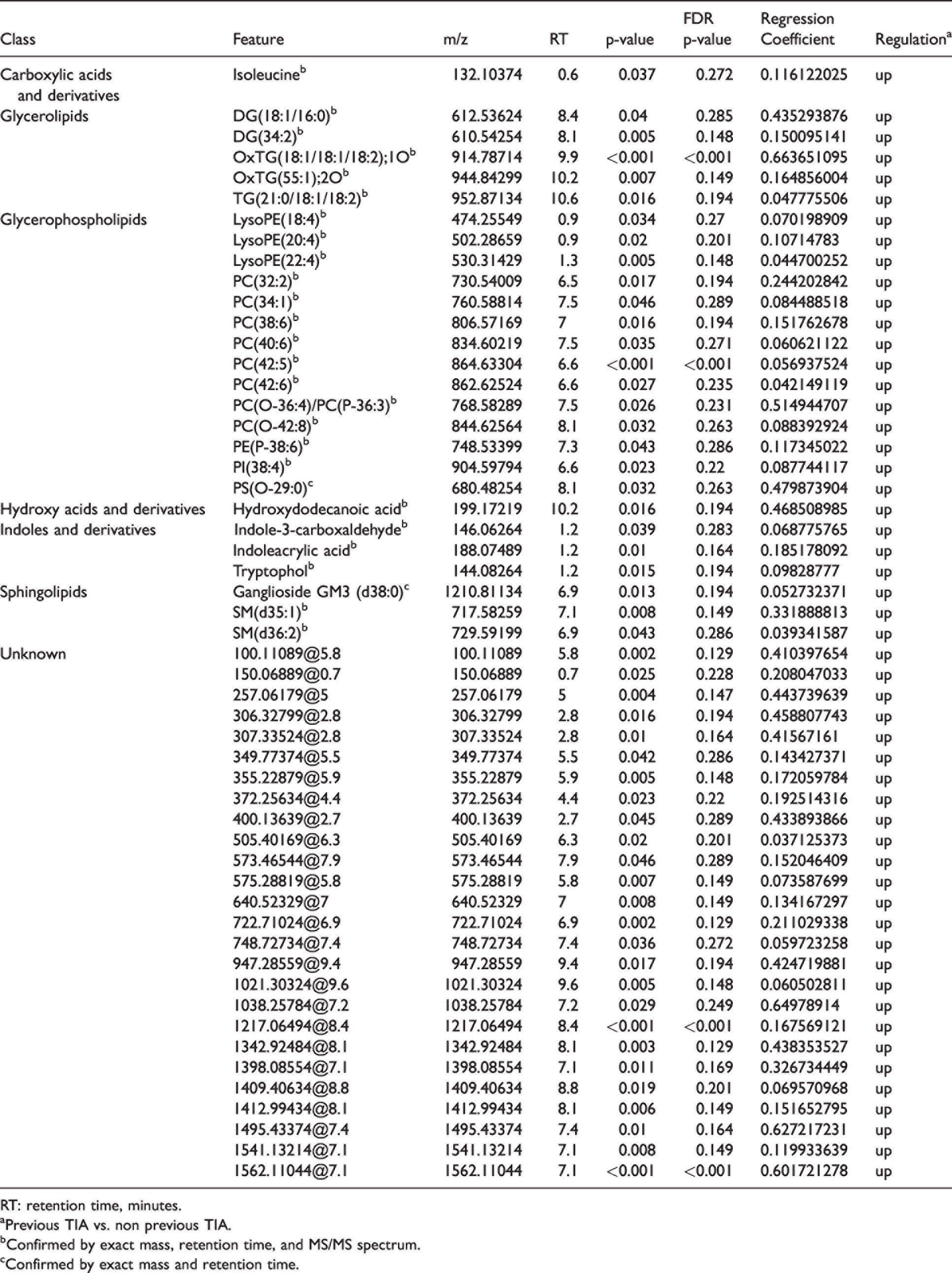
RT: retention time, minutes.
aPrevious TIA vs. non previous TIA.
bConfirmed by exact mass, retention time, and MS/MS spectrum.
cConfirmed by exact mass and retention time.
To discern whether the identified molecules could have a neuroprotective function. we further evaluated the relationship between the differential molecules identified in previous TIA ≤7d group vs. non-previous TIA ≤7d group comparison with AIS severity indicators using Spearman correlations (Figure 3). One of the identified molecules (DG(34:2)) showed a significant inverse correlation (FDR p-value <0.05) with DWI infarct volume. Twenty of them presented a significant inverse correlation with NIHSS at admission (FDR p-value <0.05): DG(18:1/16:0). DG(34:2). Hydroxydodecanoic acid. Indole-3-carboxaldehyde. Indoleacrylic acid. Isoleucine. LysoPE(18:4). LysoPE(20:4). OxTG(18:1/18:1/18:2);1O. PC(38:6). PC(40:6) . PC(42:5). PC(42:6). PC(O-36:4)/PC(P-36:3). PC(O-42:8). PE(P-38:6). PI(38:4). SM(d36:2). TG(21:0/18:1/18:2). Tryptophol. and 5 of them maintained this correlation with NIHSS 7 days after admission (DG(34:2). Indole-3-carboxaldehyde. PC(42:5). PE(P-38:6). PI(38:4)). 13 of them presented a significant inverse correlation with mRS 90 days after admission (FDR p-value <0.05): DG(34:2). Indole-3-carboxaldehyde. Isoleucine. LysoPE(18:4). LysoPE(20:4). OxTG(18:1/18:1/18:2);1O. PC(42:5). PC(O-42:8). PE(P-38:6). PI(38:4). SM(d36:2). TG(21:0/18:1/18:2). Tryptophol. In all the reported cases. those molecules upregulated in the previous TIA group showed an inverse correlation with NIHSS. mRS and DWI infarct volume except for the correlation between hydroxydodecanoic acid and NIHSS at admission.
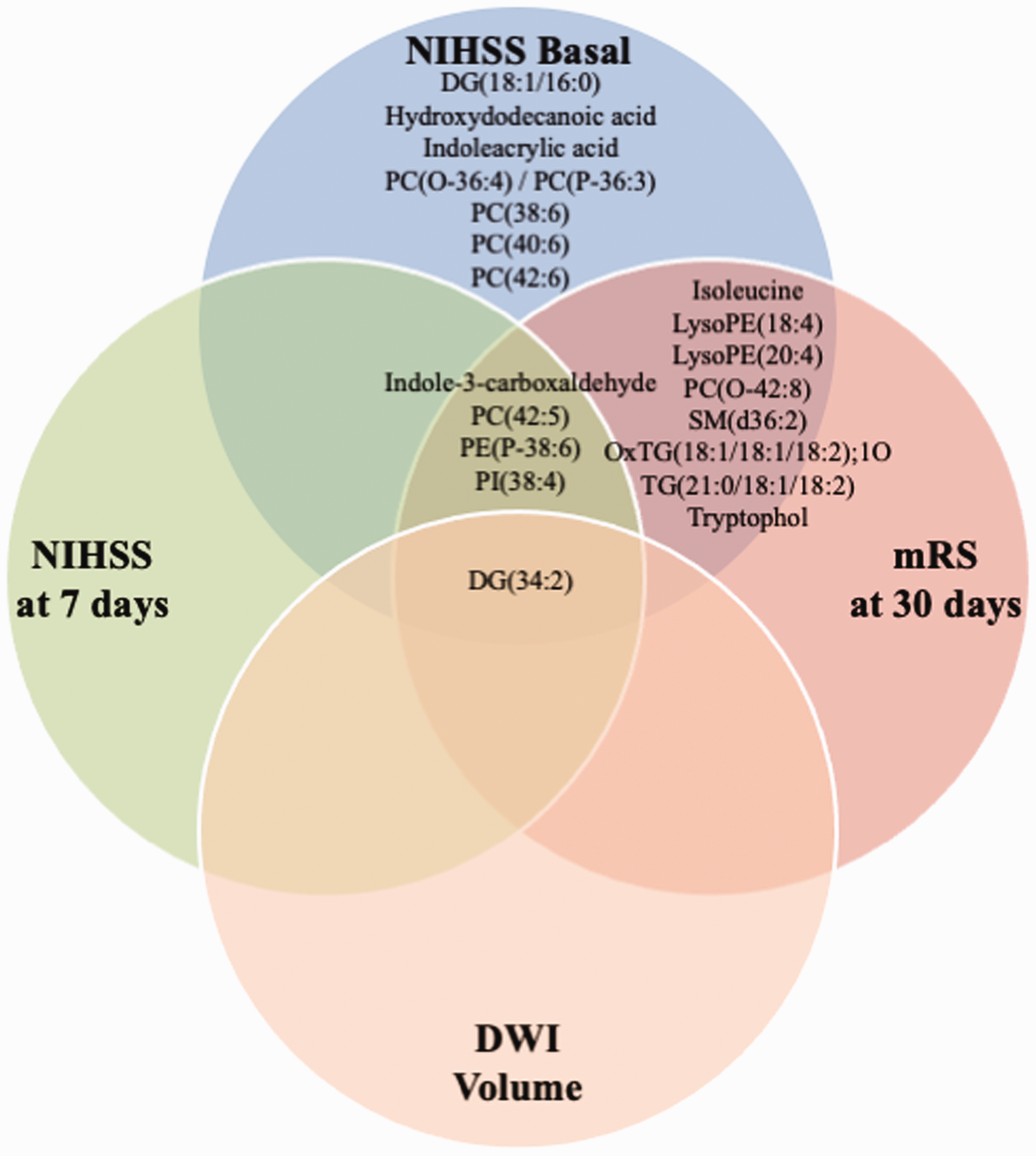
Euler diagram of differentially expressed metabolites in TIA ≤7d patients (adjusting by etiology) correlating with IS severity indicators (FDR p values for Spearman’s correlation <0.05). aconfirmed by exact mass. retention time. and MS/MS spectrum; bconfirmed by exact mass and retention time. Differentially expressed metabolites in TIA ≤7d patients when adjusted by etiology and age are highlighted in bold.
To know whether the relationship between lipid species was also altered in previous TIA ≤7d group. a chord diagram including identified differential molecules between groups was performed (Figure 2(c) and (d)). Correlations between the differential lipids showed a different distribution between the previous TIA ≤7d group and non-previous TIA ≤7d group. For instance. in the previous TIA group. all correlations of OxTG(55:1);2O with other differential metabolites disappear in the previous TIA ≤7d group. Similarly. OxTG(54:4);1O correlates in both groups with many different lipids. though a correlation with SM(d36:2) is lost in the previous TIA ≤7d group. Regarding also SL. while in non-previous TIA group. SM(d35:1) correlates significantly with several aminoacids and related compounds (isoleucine. indoleacrylic acid. indole-3-carboxaldehyde). this relationship is lost in the previous TIA group. Of note. isoleucine relationships with the metabolites of degradation (indoleacrylic acid. and indole-3-carboxaldehyde) seem mainly restricted to the previous TIA group. Reinforcing that these differences could be related to switches in lipid metabolism. for instance. ganglioside GM3 levels are correlated with highly unsaturated lysophosphatidylethanolamines (LysoPE(22:4) and LysoPE(20:4)) in the previous TIA group. but not in the non-previous TIA group. Similarly. these lipids correlate also with an ether containing lipid (PC(O-42:8)). as well as a highly unsaturated phosphatidylcholine (PC(40:6)) in the previous TIA group. but this relationship is not present in the non-previous TIA group. These data supported the idea that previous TIA before AIS induces a specific regulation of lipid metabolism resulting in a specific metabolomics/lipidomic signature.
Discussion
AIS patients with a previous recent TIA had a better clinical and neuroimaging outcome in comparison with AIS patients without prior recent TIA. This clinical translation of the phenomenon of IT induced by an episode of TIA before AIS has been described previously.4–7 Interestingly. our study provides for the first-time evidence of a metabolomic-lipidomic signature. This signature suggests the induction of a specific regulation of lipid metabolism by recent TIA.
The phenomenon of IPC is well documented in animal models. 1 although IT's physiological mechanisms are still unknown. It depends on complex signaling pathways involving ligands of G-protein-linked receptors. membrane growth factor receptors. mitochondrial ATP-sensitive potassium channels. signaling kinases such as PKC and PKG -elicited by changes in phospholipids-. modulation of reactive oxygen species. TNF-α. and sphingosine-1-phosphate production. 29
In our study. IPC impacts the plasma metabolome and lipidome. increasing specific molecules. mainly involved in lipid metabolism. Furthermore. the relationship between these lipid species is also altered by a brief ischemic period. reinforcing the idea of a specific regulation of lipid metabolism associated with preconditioning.
Globally. we described a plasma lipid profile of individuals who had a previous recent TIA characterized by a significant increase in selected GPL. GL and SL species. mainly involved in structural. cell signaling and bioenergetics roles. GPLs are the main components of neural (neurons and glia) cell membranes. 30 Specifically. PA is the precursor for “de novo” generation of different types of GPL such as PC. PE. PS. glycerophophatidylinositols (PI). PG. and cardiolipin (CL). 31 PC are the main components of mammalian cell membranes. but other lipid species such as PE. PS. SM. CL and PI are also important as cell membrane constituents. 32 Our findings demonstrated specific changes affecting mainly PC. but also PE. PI and PS. Previous papers have also described changes in brain and plasma phospholipid profile after an ischemic event such as stroke in both humans and animal models33–36 In addition. we also found three lysophospholipids (specifically lysoPE) statistically different between groups. Lysophospholipids are made from phospholipids via “remodeling pathways” by specific phospholipases generating a wide variety of lysophopholipids which are involved not only in membrane structural properties but also in cell signaling. 31 In line with our results. LysoPE(20:4) has been previously related to ischemia resistance in a rodent model. 37
Among the 14 GPL. 4 of them are plasmalogens. which have antioxidant properties and have been previously related to response to hypoxia in primary neuronal cultures. 38 Additionally. the specific lipid species identified contained fatty acids with a high unsaturation degree. in line with the highly unsaturated fatty acid profile characteristic of neural lipid species. 39 GPL containing polyunsaturated fatty acids are mainly the result of the “remodeling pathway” and are known as major sources of fatty acid-derived lipid mediators. 31 Specifically. these species have important roles determining membrane fluidity. stability and activity. and oxidative stress status. among others. Therefore. our results suggest that previous TIA induced changes in lipid composition via “remodeling pathway” to obtain specific lipid mediators potentially important for preconditioning process.
Notably. PC and PS. which are metabolically interconnected maintain a relationship with mitochondria. Thus. PC and PS synthesis involve the mitochondria-associated-membranes (MAM). 32 Therefore. it is proposed that previous TIA induces an adaptive response to promote structural membrane biosynthesis. repair and neural cell survival to facilitate functional recovery after AIS. Additionally. this adaptive response probably involves mitochondria for a dual function: improvement of neural bioenergetics and GPL biosynthesis. to maintain the membrane integrity and potential lipid-derived survival signaling pathways.
Furthermore. among these GPL. we described some other lipid forms that are related to neuronal membrane integrity. 30 Reinforcing this concept. we also reported an increment of structural SL (two SMs. and the ganglioside GM3). Considering that SM is basically present in plasma membrane and lipid rafts. and that its biosynthesis is mainly derived from ceramide and PC. it can be suggested that the increase in SM content is concomitant with the increase in PC also observed in the previous recent TIA group. GM3 deserves special attention. Gangliosides are glycosphingolipids featured by the presence of sialic acid residues. They also are essential structural components of the lipid rafts and plasma membrane. 40 Functionally. gangliosides maintain interactions with intra- and extracellular signaling molecules. participates in physiological mechanisms of both differentiation and proliferation. and they are altered in pathological conditions such as neurodegeneration and oncogenesis. 40 A-series gangliosides – including GM3. GM2. GM1. and GD1a – are the primary. structurally simplest group of gangliosides and represent the most abundant species in brain. In physiological conditions. the complex gangliosides GM1 and GD1a are more abundant in the brain than the simple species GM2 and GM3 and have neuroprotective properties. GM3 is structurally the simplest member of the ganglioside family and acts as precursor to the other more complex ganglioside species and regulates the level of expression of all other gangliosides in cells. being expressed only in very low concentrations in the healthy adult brain. 40 The accumulation of GM3 in neural tissue generates toxic effects and apoptosis.40,41 An increase in GM3 expression has been described in both human and animal models of Alzheimer disease. 42 as well as in rodent models of stroke and in human stroke with previous recent TIA. in line with observations in experimental models. suggesting the activation of an adaptive response from brain injury in order to offer precursors for the biosynthesis of more complex gangliosides such as GM1 and GD1a which have neuroprotective effects to preserve neuronal function. 43
Ischemia phenomena is tightly related to oxidative stress. 44 In this work we described two oxidized triglycerols [OxTG(18:1/18:1/18:2);1O and OxTG(55:1);2O] and four plasmalogens [PC(O-36:4). PC(O-42:8). PE(P-38:6) and PS(O-29:0)] up-regulated in previous TIA group. These results suggest that TIA phenomena induces an increase in cellular oxidative stress that is compensated generating “antioxidant” molecules. This response could contribute to better response to AIS and the better outcome described.
It is important to highlight that the metabolomics analyses revealed the different regulation of three interesting molecules related to indole metabolism: indole-3-carboxaldehyde. indoleacrylic acid and tryptophol. These three molecules are microbiota products of tryptophan and present important immune functions. Specifically. indole-3-carboxaldehyde stimulates the production of interleukin-22 which facilitates mucosal reactivity. 45 indoleacrylic acid promotes intestinal epithelial barrier function and mitigates inflammatory responses 46 and tryptophol production is negatively associated with interferon-gamma production (IFNγ) which suggests that tryptophol has anti-inflammatory properties. 47 The relation between gut microbiota and TIA/AIS is described elsewhere.48–50 In line with our results. an alteration in the composition of gut microbiota after acute central nervous system injury has been previously described. 48 This alteration damages the equilibrium of the bidirectional gut-brain axis and leads to poor prognosis by triggering pro-inflammatory responses in both peripheral circulation and central nervous system. Thus. the present results suggest a better response of intestinal immune system in previous TIA patients that should contribute to mitigate the inflammatory process associated with AIS.
Previous studies pointed aminoacids in general and isoleucine in particular as potential biomarkers of ischemic stroke.51,52 However. there is no information about its role in ischemia preconditioning. Our study demonstrated increased levels of isoleucine in previous TIA group. These results are in line with a previous work where the levels of leucine-isoleucine were related to good functional recovery after suffering stroke. 53
Notably. those lipid species that were differentially regulated in the previous TIA group presented a correlation with parameters associated with AIS severity and poor outcome (mRS at 90 days. infarct volume. basal NIHSS and NIHSS at 7 days). indicating that this metabolic adaptation observed in previous TIA patients has a neuroprotective role. Among the differential molecules described in previous TIA patients. DG(34:2) correlates with all the clinical outcomes. and Indole-3-carboxaldehyde. PC(42:5). PE(P-38:6) and PI(38:4) correlate with all the outcomes except from DWI volume. arising as the potential molecules with highest neuroprotective power.
The observation of low levels of inflammatory cytokines and biomarkers of a brain injury like IL-6 54 and S100b 55 suggested a neuroprotective environment of preceding TIA IS patients. The phenomena of remote ischemic preconditioning might enhance most of the pathways of focal IPC.
Some limitations of our study should be highlighted. The size of the two cohorts could limit the potential generalization of the obtained results. Further. the lower number of patients with a previous recent TIA contributed to obtaining only 4 molecules with FDR adjusted p-value <0.05. so further studies are needed to confirm our results. We reported significantly fewer biomarkers related to cardioembolism like hs-troponin 56 and NT-proBNP 17 in previous TIA IS patients than in non-previous TIA. We are aware of the possibility that some information on previous TIA would be lost on severe stroke patients who suffered disturbances of consciousness or aphasia beside our efforts.
Also. we used the time frame of seven days for the preceding TIA before AIS according to the previous evidence. 7 Again. the low proportion of patients with recent TIA cases in our cohorts prevented us from performing some sub-analyses. Moreover. MRI was not performed. and lesion volume was not analyzed in up to 26.8% of patients. This lack of patients could be a limitation. as those patients who underwent MRI were younger. were less cardioembolic etiology. had a less severe stroke. and had better outcomes than patients who did not undergo MRI. In no cases. MRI was performed within 24 hours of the onset of symptoms. So. infarction volumes were not representative of the early acute phase of the stroke. Outcomes of patients with recent previous ischemic heart disease or peripheral artery disease 3 were analyzed. but the number of patients with these antecedents was shallow and statistical analysis was not possible to perform (data not shown).
In conclusion. preceding TIA induces IT which clinical translated as better outcome and smaller ischemic lesion. Previous recent TIA before AIS seems to activate an additional adaptive response based on increasing circulation levels of structural and bioactive lipids to facilitate functional recovery after AIS. Furthermore. changes after TIA event seems to facilitate the production of anti-inflammatory mediators before the definitive AIS that contribute to a better immune response. The IT phenomena contribute to a better adaptation of further ischemia. Our study provides first-time evidence of a metabolomic-lipidomic signature related to the development of stroke tolerance in AIS patients induced by recent TIA and defined by specific regulation of lipid metabolism. Our findings open the way for the future discovery of biomarkers and metabolomics targets for new therapies of neuroprotection.
Footnotes
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was supported by the Government of Catalonia-Agència de Gestió d'Ajuts Universitaris i de Recerca (FP: 2017 SGR 1628; RP: 2017 SGR 696); Instituto de Salud Carlos III and co-funded by European Union (ERDF/ESF. “Investing in your future” and “A way to build Europe”) (FP: PI17-01725. PI14/01574; M.P-O: PI 17–00134. 20-0155); the Spanish Ministry of Science. Innovation and Universities (RP: RTI2018-099200-B-I00); the Department of Health (RP: SLT002/16/00250); and the INVICTUS plus Research Network (Carlos III Health Institute) (FP. CT and GA: RD16-0019-0017; TS: RD16-0019-0012; AO and JR: RD16-0019-0012; AC: RD16-0019-0006. M.J. is a professor under the Serra Hunter program (Generalitat de Catalunya).
Acknowledgements
We are grateful to all recruited patients and the members of Clinical Neuroscience group at IRBLleida. personnel of Neurology Department at Hospital Universitari Arnau de Vilanova de Lleida for scientific discussions and instrumental help. Rosa Gómez and Ikram Benabdelhak for their technical assistance. We also acknowledge the use of the facility of Biobank (B.0000682) at IRBLleida and Plataforma Biobancos PT17/0015/0027.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
GA MJ FP conceived the study. JS RP MPO MJ FP designed experiments. GM. JMS. NA. AO. JR. ST. MGC. JMF. ACC. TS. FP cohorts' recruitment. clinical data and blood samples acquisition. JS LCC CT DGL RP MPO MJ FP sample processing and data analysis. JS RP MPO GA MJ FP participated on data interpretation and draft the manuscript. All authors critically revised the final version of the manuscript. All authors approved the final version to be published. FP procured funding.
