Abstract
Whether preceding transient ischemic attack (TIA) can provide neuroprotective benefits in subsequent acute ischemic stroke (AIS) caused by large vessel occlusion remains unclarified. This study aimed to investigate the association between preceding TIA and functional outcomes in AIS patients with endovascular therapy (EVT). Eligible patients were divided into TIA and non-TIA groups according to whether they experienced TIA within 96 hours prior to stroke. Two groups were balanced using propensity score matching (PSM) analysis at a 1:3 ratio. Onset stroke severity and 3-month functional independence were evaluated. A total of 887 patients were included. After PSM, 73 patients with and 217 patients without preceding TIA were well matched. Onset stroke severity was not different between the groups (p > 0.05). However, the TIA group had a lower systemic immune-inflammation index (SII) (median, 1091 versus 1358, p < 0.05). Preceding TIA was significantly associated with 3-month functional independence (adjusted odds ratio, 2.852; 95% confidence interval [CI], 1.481–5.495; adjusted p < 0.01). The effects of preceding TIA on functional independence were partially mediated by SII (average causal mediation effects 0.02; 95% CI, 0.001–0.06, p < 0.05). In AIS patients treated by EVT, preceding TIA within 96 hours was associated with three-month functional independence but not with reduced onset stroke severity.
Keywords
Introduction
In patients with acute ischemic stroke (AIS), preceding transient ischemic attack (TIA) is not a rare phenomenon, and its incidence ranges from 6% to 20%.1–3 Interestingly, it has been postulated that prior TIA can provide neuroprotective benefits for its subsequent AIS, due to its similarity to ischemic preconditioning which is a short, sublethal episode of ischemia that could improve the resistance of cells and organs against subsequent severe ischemic insult.1,2,4,5 Several clinical studies have found that, compared with AIS patients without preceding TIA, those with preceding TIA had a smaller volume of cerebral infarction, 6 less severe neurological deficit at onset,1,7 and better neurological recovery. 2
TIA is similar to intermittent ischemic stimulation to brain tissue, of which the phenomenon has been investigated as ischemic preconditioning in situ during the past decades. 8 It has been found that ischemic preconditioning in situ could provide neuroprotective effects on ischemic stroke in animal models by reducing infarction volume and preventing ischemic-reperfusion injury, etc.9,10 Experimental studies proved that the protective effects of once ischemic preconditioning could last for 96 hours with a 12–24 hours “no-protection” interval. 4 However, due to the inherent risk of ischemic preconditioning in situ procedures, remote ischemic preconditioning has been proposed and widely investigated in clinical studies, which has been demonstrated to provide clinical benefits to stroke patients. 11 Recently, one large multicenter randomized clinical trial found that repeated remote ischemic preconditioning, if performed regularly, could significantly reduce the recurrence of ischemic stroke, which may indicate the neuroprotective effects and clinical benefits of ischemic preconditioning. 12 Furthermore, the effects of ischemic preconditioning for AIS have been proposed to be most effective in those treated with endovascular thrombectomy (EVT), as the protected salvageable brain tissue eventually evolves into brain infarction if the occluded arteries are not recanalized. 13
It has been found that preceding TIA is more frequent in patients with AIS caused by large vessel occlusion (LVO), 2 of which effective therapy has been demonstrated to be endovascular thrombectomy (EVT).14,15 Nevertheless, it remains unknown whether preceding TIA could provide neuroprotective effects for patients with AIS undergoing EVT. It has been found that approximately 70% of TIA occurred within seven days immediately preceding the stroke, and a relatively narrow time window for patients to recall their reversible neurological deficits prior to stroke could reduce memory bias. 3 Meanwhile, since the protective time window of once ischemic preconditioning is 96 hours, we aimed to explore the association between preceding TIA within 96 h of AIS and functional outcomes in patients with AIS who were treated with EVT, of which the results might provide guidance to research and development of neuroprotective therapies for AIS.
Methods
Study design and participants
This was an observational study at a high-volume stroke center. All patients with AIS secondary to LVO treated with EVT between January 2013 and July 2021 were screened. This study was approved by the Ethics Committee of Xuanwu Hospital of Capital Medical University. All patients or their legally authorized representatives provided written informed consent upon hospital admission. The present study was guided by the Declaration of Helsinki of 1975 (and as revised in 1983).
The eligibility criteria for this study were as follows: 1) AIS caused by LVO (internal carotid artery, M1 and M2 segments of the middle cerebral artery, A1 segment of the anterior cerebral artery, intracranial segment of the vertebral artery, basilar artery, and P1 segment of the posterior cerebral artery) and treated with EVT; 2) age ≥18 years; and 3) availability and accessibility of information on auxiliary examination data and clinical outcomes after EVT. The exclusion criteria were as follows: 1) incomplete data of preoperative serum examination and lack of imaging examination assessing outcomes; and 2) lack of three-month follow-up.
All EVT procedures and management of patients followed the recommendation of corresponding guidelines.16–18 All patients received guideline-recommended treatment (such as anti-hypertension, blood glucose-controlling, antiplatelet or anticoagulant therapy) during the perioperative period of EVT.
Data collection
Demographics (age and sex), vascular risk factors (hypertension, diabetes, hyperlipidemia, smoking status, atrial fibrillation), stroke etiology (according to TOAST [Trial of Org 10 172 in Acute Stroke Treatment] criteria), and occlusive sites (anterior circulation including the internal cerebral artery, middle cerebral artery, and anterior cerebral artery; posterior circulation including posterior cerebral artery, basilar artery, and vertebral artery) were documented. Admission characteristics including systolic blood pressure (SBP), diastolic blood pressure (DBP), and stroke severity were measured. Stroke severity was assessed using the National Institute of Health Stroke Scale (NHISS) and Alberta Stroke Program Early Computed Tomography Score (ASPECTS) or posterior circulation ASPECTS (pc-ASPECTS), evaluated by qualified neurologists. Previous medication was also recorded. Characteristics involved in the treatment procedures, including intravenous thrombolysis, anesthesia, time interval from symptom onset to puncture (OTP), time interval from symptom onset to recanalization (OTR), and EVT type were also recorded. Recanalization status was evaluated using the modified thrombolysis in cerebral infarction (mTICI) scale, and an mTICI of 2 b-3 was defined as successful recanalization.
Patients who experienced TIA within 96 h ahead of subsequent AIS were included in the TIA group, and the other patients were allocated into the non-TIA group. The TIA was diagnosed based on the traditional criteria of TIA, which was based on the clinical manifestations alone and defined as any recoverable focal cerebral ischemic event with symptoms lasting not more than 24 hours. 19
Routine blood examinations
Routine blood examinations before EVT were performed to calculate inflammatory indices. Blood samples were collected on admission, before EVT. An automated blood cell counter (MEK-7222K, NIHON KOHEN, JAPAN) was used to determine the absolute leukocyte, neutrophil, lymphocyte, monocyte, platelet, and red blood cell counts. The neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR) were calculated by dividing the absolute counts of neutrophils and platelets by that of lymphocytes. The systemic immune-inflammation index (SII) was calculated using the following formula: absolute neutrophil count ×absolute platelet count/absolute lymphocyte count. 20
Outcome measurements
The outcomes before EVT were onset stroke severity (NHISS score and ASPECT score before EVT) and the level of plasma inflammatory biomarkers (NLR, PLR, and SII). The outcomes after EVT were the proportion of symptomatic intracranial hemorrhage (sICH), re-occlusion of the culprit's vessel, and the proportion of functional independence. The modified Rankin Scale (mRS) was used to assess the functional outcome, and functional independence was defined as a mRS score of 0–2. And sICH after EVT was diagnosed according to the ECASS-III study, which defined sICH as any apparent extravascular blood within the brain or cranium that correlates with clinical deterioration (an increase of at least 4 points on the National Institutes of Health Stroke Scale [NIHSS]). 21 Post-operative re-occlusion of the culprit's vessel was assessed by computed tomography angiography or transcranial Doppler ultrasound within 72 h after EVT.
All outcomes were measured by two experienced stroke neurologists, and any discrepancy was resolved by discussion and re-evaluation.
Statistical analysis
SPSS Statistics version 23 (IBM, Armonk, New, USA) was used to analyze the majority of the data. Variables of patients with AIS with and without preceding TIA were analyzed using descriptive statistics and univariate comparisons. Continuous variables analyzed by the independent sample t-test or Mann-Whitney U test are depicted as means ± SD or medians (interquartile range), respectively. Categorical variables are shown as frequency and percentage, and comparisons were analyzed using the chi-squared test or Fisher’s exact test. All tests were two-sided, and statistical significance was set at P < 0.05.
Propensity score matching (PSM) is often used to match subjects with a similar distribution of confounders to achieve an estimation of treatment effects with minimal bias. In this study, PSM analysis was used to balance baseline variables (including demographic information, vascular risk factors, stroke etiology, occlusive sites, and previous drug use) between the two groups. The principle for variable selection used in PSM analysis should be variables not being influenced by preceding TIA. In this study, sex, hypertension, hyperlipidemia, stroke etiology, and occlusive sites (anterior or posterior circulation) that showed distinct discrepancies between the two groups at univariate analysis were used to form a propensity score for each subject in this study. PSM was performed by R statistical software (version 4.2.0) using nearest-neighbor matching with a caliper distance of 0.1 by matching patients in the two groups (TIA and non-TIA groups) at a 1:3 ratio. After PSM, comparisons of the variables between the two groups were again analyzed using SPSS statistics, as mentioned previously. Then, the influence of preceding TIA on onset stroke severity, inflammation biomarkers, and functional outcomes was evaluated.
Considering the onset severity, treatment procedures, and recanalization status of stroke which were also assessed as efficacy outcomes in this study could also affect functional outcomes after EVT as well. Therefore, we also applied multivariable logistic regression analyses to determine the independent effect of preceding TIA on functional outcomes by adjusting these confounders after TIA. The variables after TIA (onset severity, treatment procedures, and recanalization status of stroke), which were considered to distinctively affect functional outcomes in previous studies or had a significant difference (p < 0.05) in univariate analyses between the two groups, were further adjusted as covariates in multivariate logistics regression. Adjusted odds ratio (OR) and 95% confidence intervals (CI) were calculated.
The mediating effect of inflammation markers in preceding TIA for functional outcomes was demonstrated by average causal mediation effects (ACME), and the direct effect of preceding TIA on functional outcomes was reflected by average direct effects (ADE). The above analysis was performed by the “mediation” package in R software with 100 bootstrap samples.
Results
Demographic and baseline characteristics
After screening 1103 registered AIS patients secondary to LVO, a total of 887 eligible patients with 624 (70.3%) males and a mean age of 63.22 ± 12.76 years were included in this study (Supplementary figure 1). The median mRS score before stroke was 0 (ranged from 0 to 2, IQR: 0–0) in patients with preceding TIA, and 0 (ranged from 0 to 3, IQR:0–0) in the non-TIA group; there was no significant difference between the two groups. Before matching, baseline variables were compared between 73 patients with preceding TIA and 814 patients without TIA. The results showed that patients in the TIA group had a higher proportion of male patients (63 [86.3%] vs. 561 [68.9%], p = 0.002), hypertension (59 [80.8%] vs. 569 [69.9%], p = 0.049), hyperlipidemia (48 [65.8%] vs. 430 [52.8%], p = 0.034), stroke etiology of large artery atherosclerosis (60 [82.2%] vs. 475 [58.1%], p < 0.001), and posterior circulation stroke (35 [47.9%] vs. 214 [26.3%], p < 0.001) as compared with patients in the non-TIA group. By matching sex, hypertension, hyperlipidemia, stroke etiology, and occlusive sites (anterior or posterior circulation), 73 cases with TIA were 1:3 matched with 217 cases without preceding TIA. After PSM, demographic variables, vascular risk factors, stroke etiology, and occlusive sites were again compared between the two groups by univariate analysis, and no significant differences in these variables were detected. Detailed information was summarized in Table 1.
Baseline characteristics of AIS patients before and after PSM.
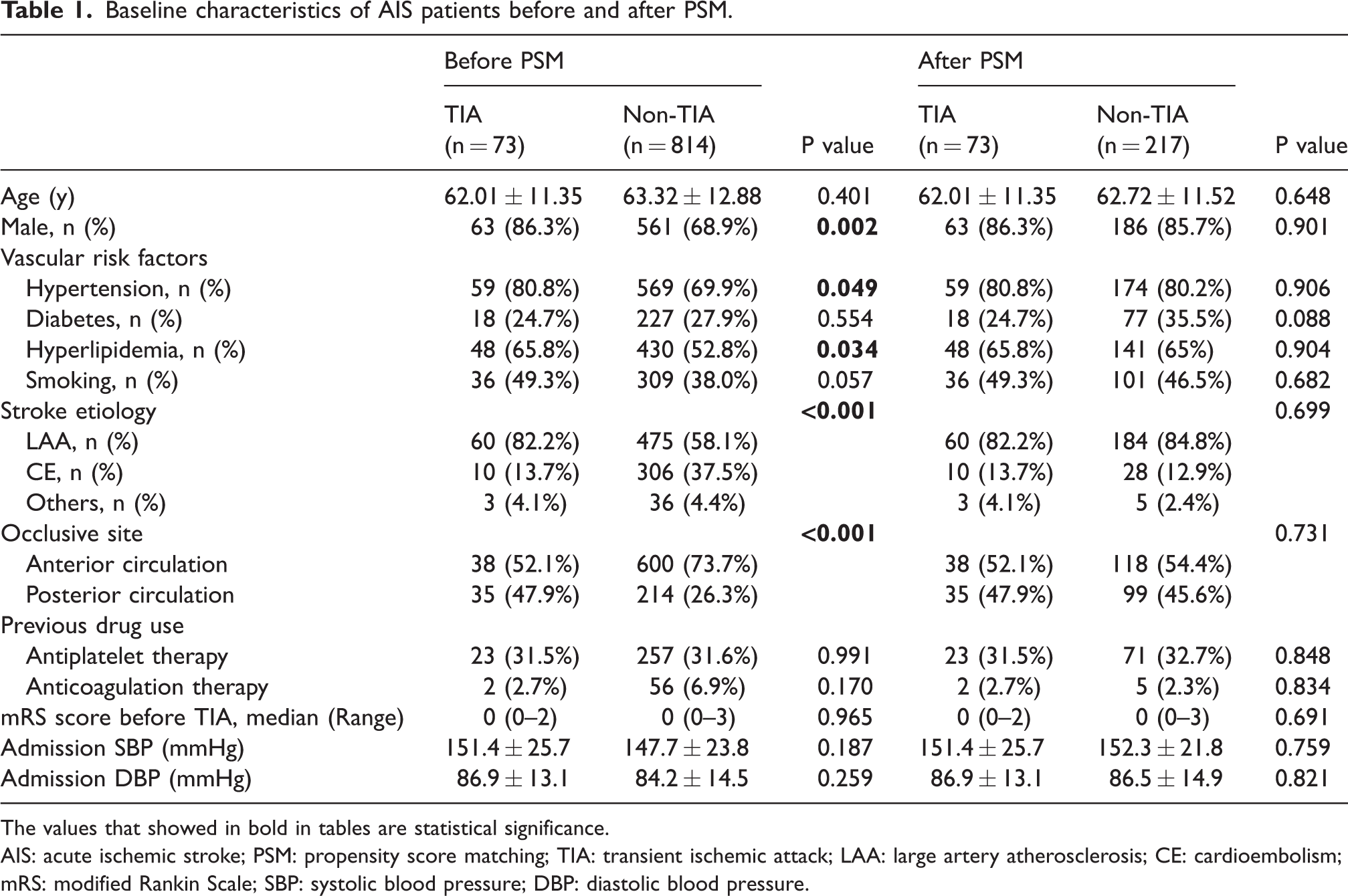
The values that showed in bold in tables are statistical significance.
AIS: acute ischemic stroke; PSM: propensity score matching; TIA: transient ischemic attack; LAA: large artery atherosclerosis; CE: cardioembolism; mRS: modified Rankin Scale; SBP: systolic blood pressure; DBP: diastolic blood pressure.
Characteristics of treatment procedure
In the PSM cohort, the TIA group had a comparable (p > 0.05) median OTP (median: 444 min, IQR: 327–735 min) and OTR (median: 518 min, IQR: 373–809 min) to the non-TIA group (OTP [median: 450, IQR: 345–610 min]; OTR [median: 518 min, IQR: 410–715 min]). Meanwhile, the proportion of patients who underwent stent implantation was not significantly different between the two groups (TIA vs. non-TIA:33 [45.2%] vs. 80 [36.9%], p > 0.05). Intravenous thrombolysis was performed in 19 (26.0%) patients with preceding TIA and 71 (32.7%) patients without TIA (p = 0.285). Successful recanalization (mTICI 2 b-3) was achieved in 68 (93.2%) patients with preceding TIA and 190 (87.6%) patients without TIA (p > 0.05). The detailed information is summarized in Table 2.
Treatment procedures and recanalization status of AIS patients.
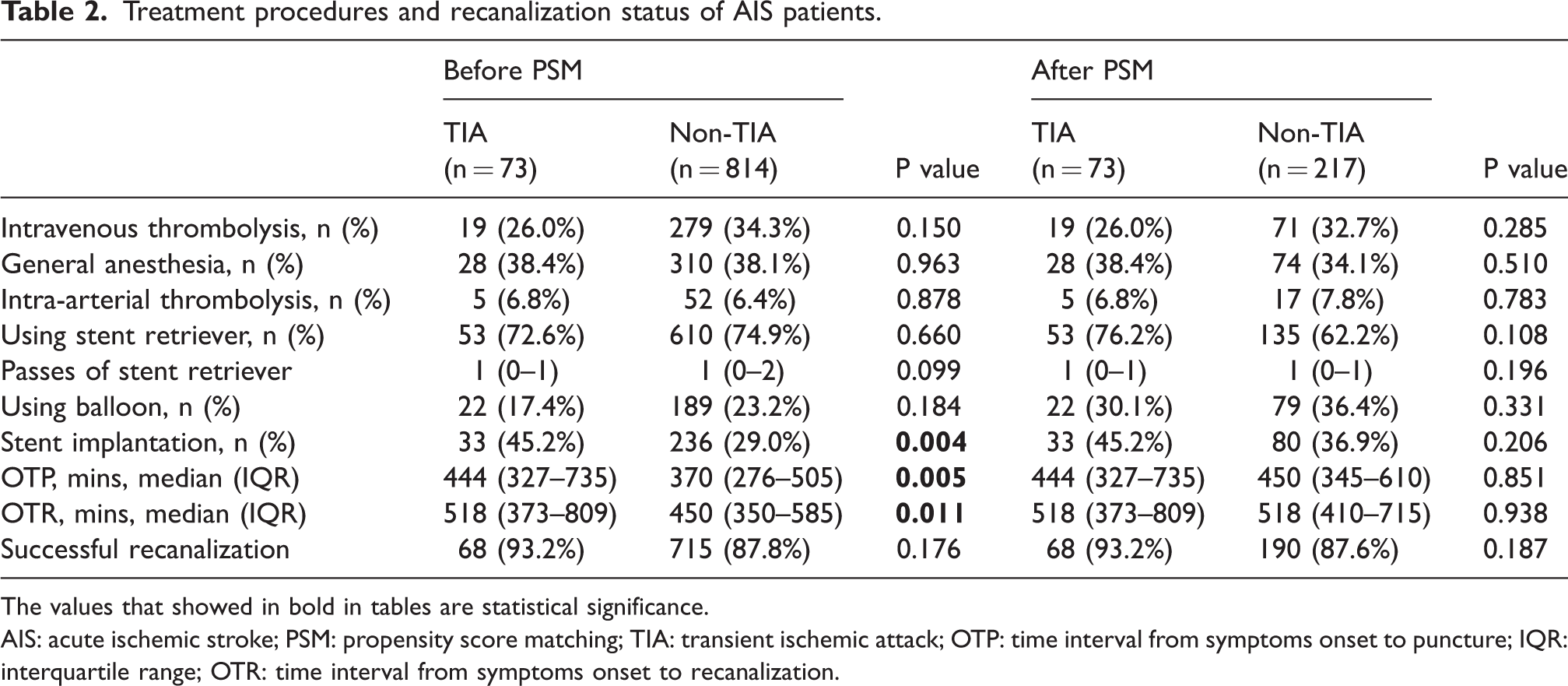
The values that showed in bold in tables are statistical significance.
AIS: acute ischemic stroke; PSM: propensity score matching; TIA: transient ischemic attack; OTP: time interval from symptoms onset to puncture; IQR: interquartile range; OTR: time interval from symptoms onset to recanalization.
Outcomes before endovascular therapy
Before PSM, the median admission NIHSS score was 15 (IQR: 11–22.75) points in patients with preceding TIA and 16 (IQR: 12–21) points in patients without preceding TIA (p > 0.05), and the median (pc-)ASPECT score was 9 (IQR: 8–9.75) points in patients with preceding TIA and 9 (IQR: 7–10) points in patients without preceding TIA (p > 0.05). After PSM, the median admission (pc-)ASPECT score was 9 (IQR: 8–9.75) in the TIA group and 8 (IQR: 7–9) in the non-TIA group (p = 0.156), and the admission NHISS scores were 15 (IQR: 11–22.75) points and 16 (IQR: 12–25) points, respectively (p > 0.05). The stroke severity was not ameliorated by preceding TIA. Moreover, a significantly lower level of inflammatory indices at baseline, including PLR (TIA vs. non-TIA:137.4 9 [IQR: 96.1–227.5] vs. 178.7 [IQR: 124.4–238.2], p = 0.021) and SII (TIA vs. non-TIA:1091 [IQR: 617–1642] vs. 1358 [IQR: 837–2300], p = 0.009), were found in the PSM cohort with preceding TIA. The data are summarized in Table 3.
Outcomes of AIS patients before and after endovascular therapy.
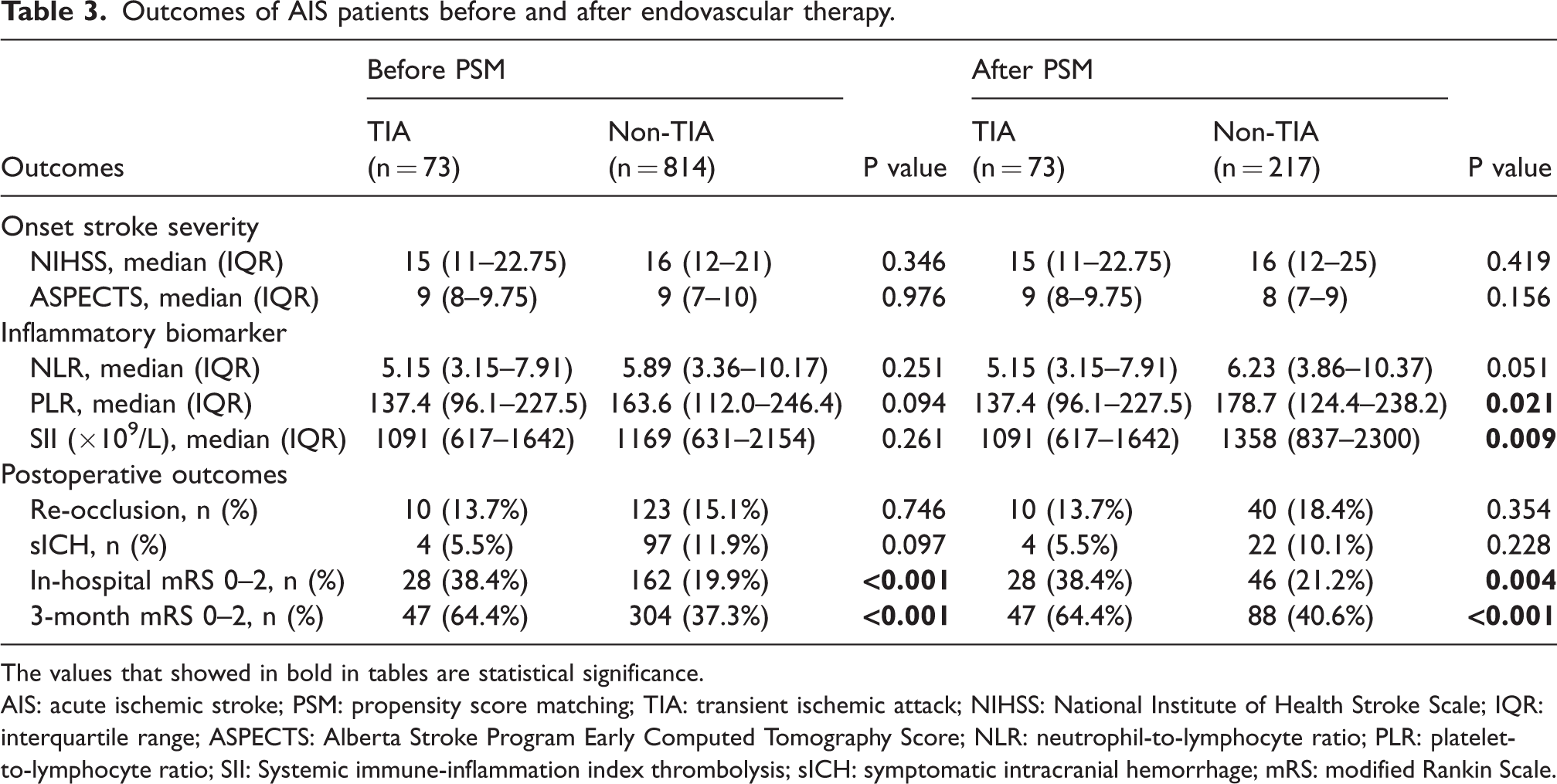
The values that showed in bold in tables are statistical significance.
AIS: acute ischemic stroke; PSM: propensity score matching; TIA: transient ischemic attack; NIHSS: National Institute of Health Stroke Scale; IQR: interquartile range; ASPECTS: Alberta Stroke Program Early Computed Tomography Score; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; SII: Systemic immune-inflammation index thrombolysis; sICH: symptomatic intracranial hemorrhage; mRS: modified Rankin Scale.
Outcomes after endovascular therapy
In the PSM cohort, 47 patients (64.4%) with preceding TIA and 88 patients (37.3%) without preceding TIA achieved functional independence (p < 0.001) at three-month follow-ups. The distribution of the three-month mRS scores of the two groups of patients is depicted in Figure 1. Twenty-eight patients (38.4%) in the TIA group and 162 (19.9%) in the non-TIA group achieved in-hospital functional independence (p < 0.001). In addition, four patients (5.5%) in the TIA group and 97 patients (11.9%) in the non-TIA group experienced sICH (p > 0.05), and 10 patients (13.7%) in the TIA group and 123 patients (15.1%) in the non-TIA group experienced re-occlusion of the culprit artery (p > 0.05). Detailed information is listed in Table 3.
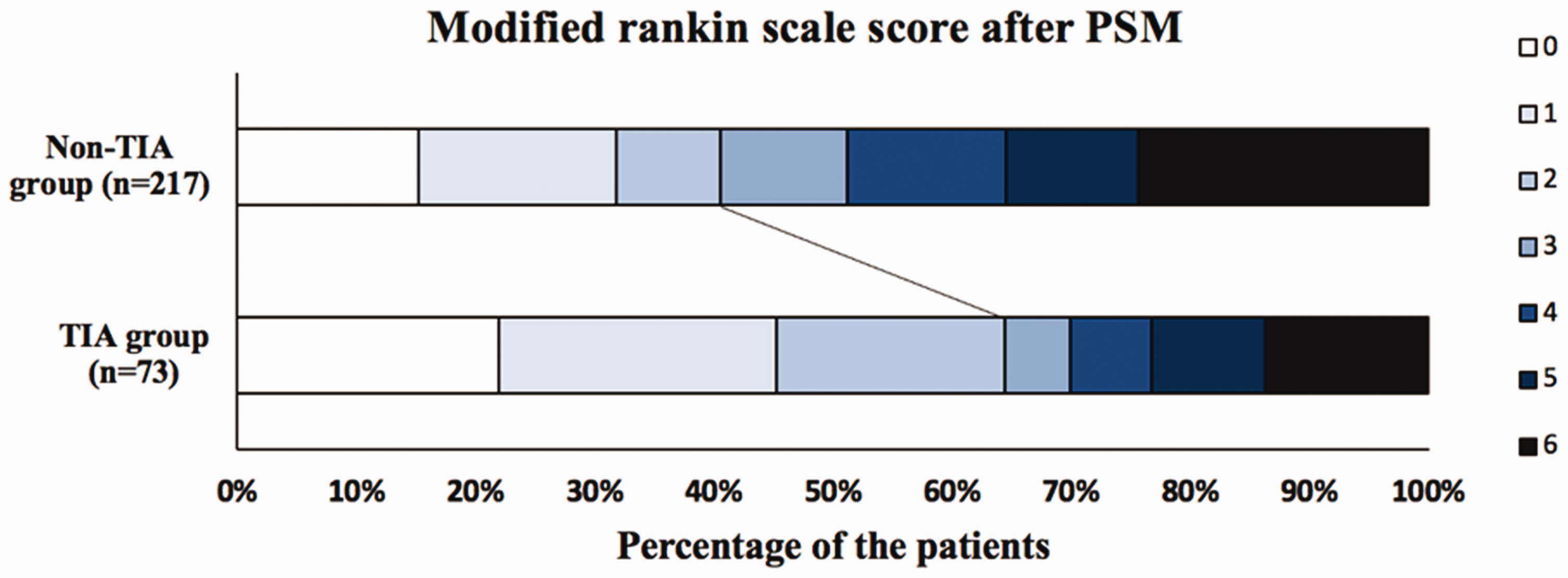
distribution of Modified Rankin scale score after PSM.
After multivariate analysis, preceding TIA was distinctively associated with three-month functional independence (OR, 2.852; 95% CI, 1.481–5.495; p = 0.002) and in-hospital functional independence (OR, 2.626; 95% CI, 1.310–5.264; p = 0.007). Meanwhile, preceding TIA was not significantly associated with re-occlusion of the culprit artery (OR, 0.771; 95%CI, 0.357–1.668; p = 0.510) or sICH (OR, 0.552; 95%CI, 0.179–1.698; p = 0.300). Detailed information is summarized in Table 4.
multivariate analysis of preceding TIA in PSM cohorts.
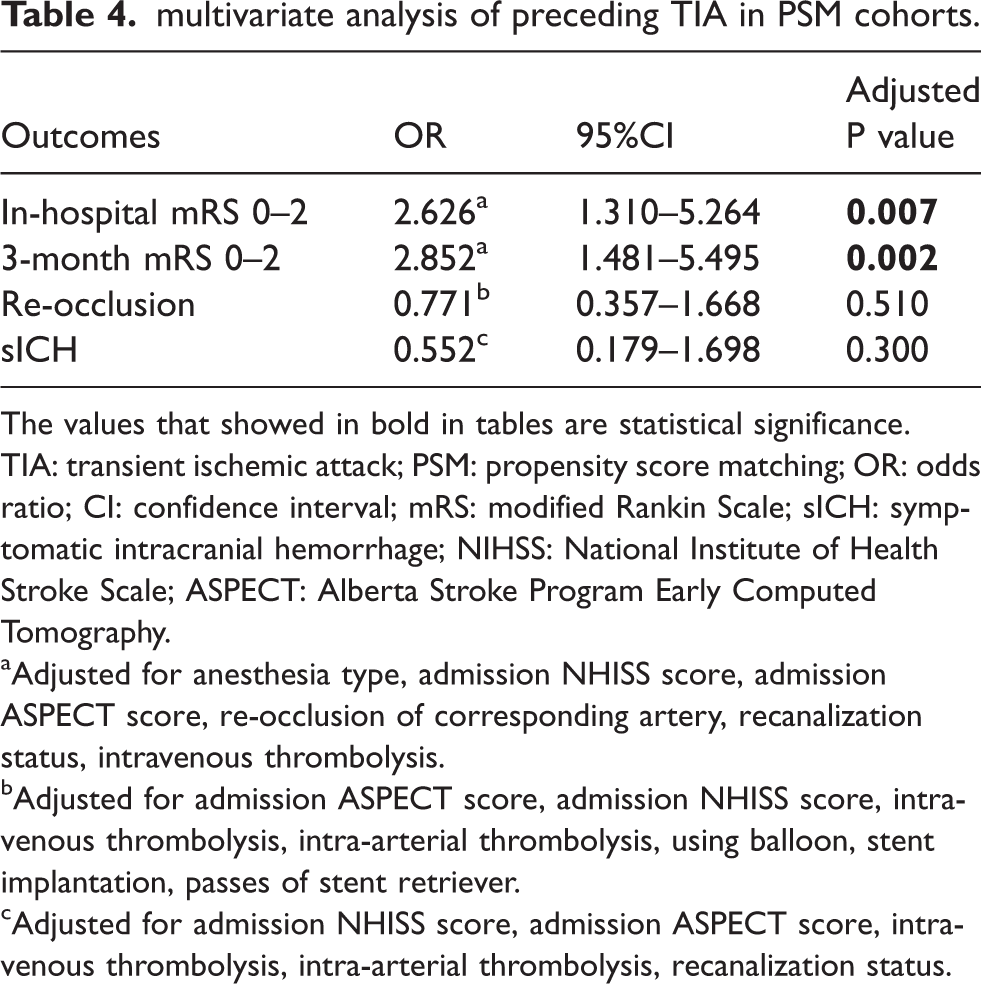
The values that showed in bold in tables are statistical significance.
TIA: transient ischemic attack; PSM: propensity score matching; OR: odds ratio; CI: confidence interval; mRS: modified Rankin Scale; sICH: symptomatic intracranial hemorrhage; NIHSS: National Institute of Health Stroke Scale; ASPECT: Alberta Stroke Program Early Computed Tomography.
aAdjusted for anesthesia type, admission NHISS score, admission ASPECT score, re-occlusion of corresponding artery, recanalization status, intravenous thrombolysis.
bAdjusted for admission ASPECT score, admission NHISS score, intravenous thrombolysis, intra-arterial thrombolysis, using balloon, stent implantation, passes of stent retriever.
cAdjusted for admission NHISS score, admission ASPECT score, intravenous thrombolysis, intra-arterial thrombolysis, recanalization status.
Mediation analysis showed that SII (ACME 0.02; 95%CI, 0.001–0.06, p = 0.04) partially mediated the effects of preceding TIA on three-month functional independence. However, PLR (ACME 0.01; 95%CI, –0.002–0.03; p > 0.05) had no mediation effects on preceding TIA for outcomes. The results are depicted in Figure 2.
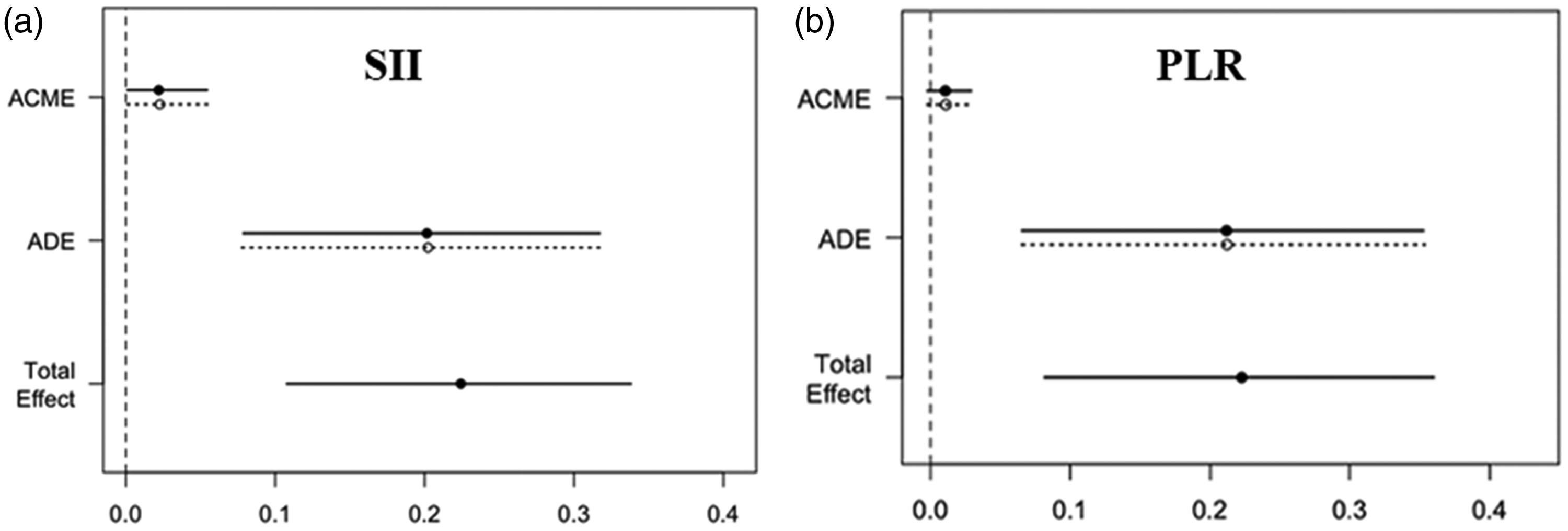
the average causal mediating effects of pheripheral inflammation markers.
Discussion
In this study, we found that in patients with AIS secondary to large-vessel occlusion and treated with EVT, preceding TIA was associated with higher ratios of three-month and in-hospital functional independence. In addition, serum biomarkers of systemic inflammation were found to be much lower in patients with preceding TIA, and SII could partially mediate the beneficial effects of preceding TIA on functional outcomes.
Compared to previous studies, the novelty of this study is that all recruited patients with AIS were secondary to large-vessel occlusion and treated with EVT, and the results of this study showed that preceding TIA was associated with favorable outcomes in this population. In addition, before PSM, the results showed that in patients with preceding TIA, the OTP and OTR were much longer, and the proportion of stent implantation was much higher than that in patients without preceding TIA. This phenomenon may be attributable to the great discrepancy in the distribution of stroke etiology, as 82.2% of patients with preceding TIA had atherosclerosis-related strokes and only 13.7% had cardioembolic strokes. The higher proportion of atherosclerosis-related strokes may have led to a delay in reperfusion due to the increased challenge of operation caused by atherosclerotic lesions.22,23
Although the benefits of preceding TIA on functional recovery were confirmed in the present study, we did not observe attenuated stroke severity at onset, which was inconsistent with most previous studies. After a literature review of previous studies that investigated the effects of preceding TIA in patients with acute ischemic stroke (summarized in Supplementary table 1), we found that stroke severity at onset could be attenuated by preceding TIA in patients with mild stroke, whereas it could not be alleviated in patients with moderate-to-severe stroke.6,7,24–26 Two previous studies investigated preceding TIA in patients with moderate-to-severe AIS who were treated with intravenous thrombolysis or intra-arterial thrombolysis, but their three-month outcomes were conflicting.25,27 The low proportion of participants who achieved recanalization after therapy may be an important reason, as salvageable brain tissue eventually evolves into brain infarction if the occluded arteries are not recanalized. Therefore, we contend that in patients with acute LVO who had severe stroke, the neuroprotection induced by prior TIA was not adequate to prevent brain damage caused by persistent obstruction of blood flow until the culprit’s vessel was recanalized. This phenomenon has also been reported in preclinical studies investigating the neuroprotective effects of ischemic conditioning in animal models of transient ischemia.28,29
Although the results of this study may indicate the neuroprotective effects of preceding TIA, the underlying mechanisms remain to be clarified. In recent years, it has been shown that secondary inflammation plays an important roles in exacerbating brain damage post-stroke,27,30–33 and therapies aimed to reduce the inflammatory response could decrease the stroke severity and improve neurological functional outcomes. 34 Peripheral inflammation, which can interact with the brain via the gut-brain axis, gradually became a focus in stroke patients because it’s easy to detect.33,34 In the present study, the results showed that there were significantly lower levels of plasma PLR and SII in patients with preceding TIA and SII may mediate the beneficial effects of preceding TIA on functional outcomes. Given the similarity of preceding TIA and ischemic preconditioning and that ischemic preconditioning regulates the immune-inflammatory response,35,36 thus the mechanisms of neuroprotection preceding TIA may be involved in the regulation of inflammation, which are urgently needed to be investigated.
Nowadays, despite full recanalization following thrombectomy for acute ischemic stroke, about half of the patients could not achieve functional independence. 37 It is important to note that full recanalization of proximal large vessels does not guarantee complete restoration of cerebral blood flow in the distal previously ischemic tissue, for recent studies have found that over 30% of patients who achieved a modified Thrombolysis in Cerebral Ischemia (mTICI) score of 3 after thrombectomy still suffered from sustaining hypoperfusion in certain area.38,39 Such residual perfusion deficiency after therapy is known as the no-reflow phenomenon (NRP) or incomplete microvascular reperfusion, which might result from the functional and structural alteration of the cerebral microcirculation during the ischemia-reperfusion process. 40 Although the detailed mechanisms of no-reflow phenomenon remains to be explored, studies have found that tissue no-reflow may result from pericyte disruption, pre-capillary small muscle cell constriction, endothelium swelling or microemboli, which may be reversible by activating the NO pathway, inhibiting oxidative stress, neutrophil depletion and so on.41,42 Intriguingly, ischemic preconditioning has been proved to activate the NO pathway, promote endothelium function, reduce inflammation and regulate fibrinolysis-coagulation system,35,36,43 which may alleviate the injuries of cerebral no-reflow. Due to the similarity to ischemic preconditioning, preceding TIA may provide benefits to patients with AIS that treated with EVT by attenuating cerebral no-reflow, which are needed to be investigated in further studies.
Given the dangers of inducing brain ischemia by preceding TIA, safe and effective methods for inducing preconditioning ischemia for AIS neuroprotection are required. In recent years, remote ischemic conditioning has emerged as a potential method for conferring neuroprotective factors to the brain via repeated ischemia-reperfusion of the upper limbs. A pilot clinical study confirmed the safety and feasibility of remote ischemic conditioning in patients with AIS who underwent EVT, 44 and preliminary efficacy results favored the use of remote ischemic conditioning in this population, although no significant difference was detected. Preceding TIA ahead of AIS is a phenomenon of ischemic preconditioning in situ and the rudiment of remote ischemic conditioning. The findings of this study may further support the application of remote ischemic conditioning in patients with AIS treated with EVT.
Our study had some limitations. First, in this retrospective study, TIA was diagnosed based on clinical manifestations alone, and as all patients finally developed AIS, this may have overestimated the proportion of TIA according to the currently recognized diagnostic criteria of TIA. Second, the effects of TIA times and duration of ischemia on functional outcomes were not investigated in the present study; further prospective studies with larger sample sizes are needed. In addition, due to the low incidence of preceding TIA before AIS and the retrospective setting of this study, the sample size was relatively small, especially in the TIA group, which may limit the representation of this cohort and influence the generalization of the results. Other potential biases may also be caused to the results due to the relatively small sample size of the TIA group. Therefore, the findings of this study should be interpreted with caution, and more powerful studies are needed to confirm these results.
Conclusions
In patients with AIS secondary to LVO and treated with EVT, preceding TIA within 96 h was associated with favorable outcomes, and the underlying mechanism may be involved in the regulation of inflammation. Further investigations are needed to confirm these results and clarify the underlying mechanisms.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231167924 - Supplemental material for Preceding transient ischemic attack was associated with functional outcome after stroke thrombectomy: A propensity score matching study
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231167924 for Preceding transient ischemic attack was associated with functional outcome after stroke thrombectomy: A propensity score matching study by Jiali Xu, Wenting Guo, Jin Ma, Qingfeng Ma, Jian Chen, Haiqing Song, Changhong Ren, Sijie Li, Yuchuan Ding, Wenbo Zhao and Xunming Ji in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Beijing Natural Science Foundation (JQ22020), Beijing Nova Program (No. Z201100006820143), National Natural Science Foundation of China (No. 82001257 and 81971114), and General Project of Science and Technology of Beijing Municipal Education Commission (No. KM202110025018).
Data availability
The data that support the findings in this article are available from the corresponding authors on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
WTG, JLX conceived of the study idea, collected and analyzed the data, and drafted the manuscript. JM participated in the data collection. WBZ, and XMJ conceived of the study idea and design, drafted and revised the manuscript. SJL, XMJ, JC, YCD, QFM, HQS and CHR participated in the coordination of the study. All authors read and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
