Abstract
Preeclampsia (PE) is a hypertensive disorder of pregnancy associated with neurovascular dysfunction, cognitive impairment and increased seizure susceptibility. Here, we sought to determine if treatment of experimental PE (ePE) rats with apocynin could prevent hippocampal arteriolar (HA) dysfunction and impaired seizure-induced hyperemia within the hippocampus, a brain region central to cognition and seizure generation. Isolated and pressurized HAs from Sprague Dawley rats that were normal pregnant (Preg; n = 8), ePE (n = 8) or ePE treated with apocynin for 2 weeks of gestation (ePE + apo; n = 8) were compared. Hippocampal blood flow (n = 6/group) was measured using hydrogen clearance before and during seizure. Aorta elastin was quantified using histochemistry. ePE was associated with HA dysfunction including reduced contraction to endothelin-1 and diminished dilation to the endothelium-dependent vasodilator NS309 that was prevented by apocynin. However, apocynin had no effect on ePE-induced impairment of dilation to the nitric oxide donor sodium nitroprusside, but increased myogenic tone and substantially increased HA distensibility. Seizure-induced hyperemia was impaired in ePE rats that was restored by apocynin. Aorta from ePE rats had reduced elastin content, suggesting large artery stiffness, that was unaffected by apocynin. Thus, while apocynin partially prevented HA dysfunction, its restoration of functional hyperemia may be protective of seizure-induced injury during eclampsia.
Introduction
Preeclampsia (PE) is a multisystem hypertensive disorder of pregnancy affecting 4-8% of pregnancies worldwide. 1 PE adversely affects numerous maternal organs and organ systems, including the brain and cerebral circulation. For example, PE can cause neurological symptoms during the index pregnancy, including severe and persistent headache, visual disturbances, and seizure (eclampsia), and is associated with an increased risk of stroke.1,2 In addition, women with a prior history of PE have brain atrophy and report memory impairment at relatively young ages (e.g., in their 40 s) that is associated with greater white matter lesion burden.3 –7 In animal models of PE, cerebral microvascular dysfunction has been shown in cortical arterioles supplying the white matter and in hippocampal arterioles (HAs) perfusing the hippocampus that may underlie memory dysfunction and cognitive decline during the index pregnancy.8,9 If persistent and untreated, cerebrovascular dysfunction during PE could also contribute to early onset cognitive decline and memory deficits in formerly PE women.
One of the underlying mechanisms by which PE causes widespread vascular dysfunction, including in the cerebrovasculature, is maternal dyslipidemia.8 –12 In PE women, levels of very low density lipoprotein (vLDL) cholesterol are considerably elevated.10,11 vLDL is a form of LDL that is highly susceptible to oxidation to form oxidized LDL (oxLDL). In fact, oxLDL levels are increased 2.6-fold in PE women. 10 Importantly, elevated oxLDL is a driver of oxidative stress through high affinity binding to the lectin-like oxidized LDL receptor-1 (LOX-1) in endothelium. 13 LOX-1 binding activates nicotinamide adenine dinucleotide phosphate (NADPH) oxidase and generates superoxide (O2−). 13 In addition to high levels of oxLDL, women with PE have increased expression of LOX-1 in the vasculature, leading to endothelial dysfunction and oxidative stress thought to underlie many of the cardiovascular complications of PE. 12
High oxidative stress and endothelial dysfunction not only affect the microcirculation but also large arteries such as the aorta that can progress cardiovascular dysfunction and brain injury in PE women. Repeated oxidative and mechanical stress during hypertension can degrade elastin fibers in the vascular wall, stiffening large arteries.14,15 Large artery stiffness is particularly detrimental to the brain that has high flow and low impedance as transmission of the pulsatile load is conducted deeper into the brain parenchyma.16,17 The propagation of high pulsatile flow to the cerebral microvasculature in response to large artery stiffness promotes vascular remodeling and microvascular damage that underlies white matter hyperintensities, cerebral microbleeds and lacunar infarcts.16,18,19 Increased large artery stiffness is present in PE women, 20 including enhanced carotid pulse wave velocity21,22 that persists postpartum.23 –26 While the relationship between large artery stiffness and cerebral microvascular damage is largely unknown in PE women, increased oxidative stress caused by binding of oxLDL to LOX-1 is associated with arterial stiffness in aging and other cardiovascular diseases.13 –15
In the current study, we used a model of PE that includes dyslipidemia and high oxLDL to study the association between large artery stiffness (aorta) and brain microvessel (hippocampal arterioles) structure and function. We previously showed that this model of PE demonstrated cognitive decline several weeks after pregnancy that was exacerbated by eclampsia-like seizures. 27 In addition, this model had HAs that were stiffer, and lacked a hyperemic response to seizure. 9 Vascular remodeling of this nature could serve to impair hippocampal blood flow and predispose the microcirculation to injury by failing to dampen pulse waves traveling along the vascular tree from the heart.16,19,28 We treated PE rats with apocynin, an inhibitor of NADPH oxidase and antioxidant, to determine the role of oxidative stress in mediating hippocampal vascular remodeling and dysfunction.29,30 Lastly, we measured hippocampal blood flow prior to and during seizure using hydrogen clearance to determine if treatment with apocynin could restore the hyperemic response to seizure in ePE rats.
Materials and methods
Animals and model of experimental PE (ePE)
Timed-pregnant female Sprague Dawley rats between 14 and 16 weeks of age were used for all experiments (Charles River, Canada). Rats were housed in pairs with environmental enrichment in the University of Vermont Animal Care Facility, an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) accredited facility. Rats were maintained on a 12-h light/dark cycle and allowed access to food and water ad libitum. All procedures were approved by the Institutional Animal Care and Use Committee at the University of Vermont and conducted in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals. Rats were randomly assigned to treatment group and experiments were conducted in a randomized fashion. ARRIVE 2.0 guidelines were followed for the reporting of results.
Three groups of animals were used for experiments. Normal pregnant (Preg), ePE and ePE treated with the NADPH oxidase inhibitor apocynin (ePE + apo). To model ePE, we used an established model that involves maintaining pregnant rats on a high cholesterol diet starting on gestational day 7. This model has previously been shown to have hyperlipidemia, including elevated total and LDL cholesterol and triglycerides. 10 This model of PE is also associated with increased blood pressure, endothelial dysfunction, fetal growth restriction, placental disease and mild cognitive impairment.9,10 Briefly, pregnant rats were fed ad libitum a high cholesterol diet (Prolab 3000 rat chow with 2% cholesterol and 0.5% sodium cholate; Scotts Distributing Inc., Hudson, NH, USA) on days 7–20 of gestation. One group of ePE rats were given apocynin, a NADPH oxidase inhibitor (1.5 mM; ∼12 mg/kg) in drinking water on days 7–20 of gestation (ePE + apo). This dose has been shown to prevent atherosclerotic lesions in high cholesterol fed rabbits. 31 Two sets of animals were used for the study. The first set (n = 8/group) was used for isolated vessel experiments on HAs and aorta histochemistry. The second set (n = 6/group) was used for blood pressure measurements and hippocampal blood flow measurements via hydrogen clearance.
Non-Invasive blood pressure measurements
Blood pressures were measured non-invasively via tail-cuff, as described previously. 11 Briefly, animals were allowed to acclimate to the environment in their home cages for 1 hr before measurements were taken. Rats were placed in individual tube holders, blood pressure tail-cuffs were applied near the base of the tail and rats were kept warm with heating pad (CODA system, Kent Scientific, Torrington, CT, USA). Systolic and diastolic blood pressures were recorded. Blood pressures were taken on gestational days 18, 19 and 20.
Isolated hippocampal arterioles
Brains were removed and place in ice-cold, freshly bubbled artificial cerebrospinal fluid (aCSF). HAs were dissected and mounted on glass cannulas in an arteriograph chamber (Living Systems Instrumentation, Burlington, VT, USA), as previously described. 9 HAs were equilibrated at 40 mmHg for one hour while maintaining physiologic conditions (pH 7.38–7.42, 36.8–37.2 °C). To measure spontaneous myogenic tone, intravascular pressure was increased in 20 mmHg increments to 120 mmHg in a stepwise manner while lumen diameter and wall thickness were recorded using video dimensional analysis. Intravascular pressure was returned to 40 mmHg for the remainder of the experiment. Reactivity to the following pharmacological agents was measured: NS309, a small- and intermediate-conductance calcium-activated potassium (SK/IK) channel agonist (10−8–10−5 M), endothelin-1 (ET-1), a vasoconstrictor (10−11–10−9 M), and sodium nitroprusside (SNP), a nitric oxide donor (10−8–10−5 M). After reactivity was measured, passive structural measurements were made by using aCSF containing zero calcium, papaverine (10−4 M) and diltiazem (10−5 M) to fully relax the vascular smooth muscle. Passive structural measurements were made within the pressure range of 5 – 120 mmHg. These included measuring lumen diameter and wall thickness at each pressure once stable, ∼3 minutes. Passive wall tension and wall stress were then calculated (please see below).
Measurement of hippocampal blood flow
Absolute blood flow in the hippocampus was determined under baseline conditions and during seizure using a hydrogen clearance method, as described previously. 9 Separate groups of Preg, ePE and ePE + apo rats (n = 6/group) were anesthetized with isoflurane (1–3% in O2) for instrumentation. Femoral arteries were cannulated to obtain blood samples for blood gas measurements and for continuous blood pressure recordings via a pressure transducer (BIOPAC Systems Inc., Goleta, CA, USA). Femoral veins were cannulated for administration of the anesthetic chloral hydrate (50 mg/mL), infusion of the paralytic vecuronium, and the chemoconvulsant pentylenetetrazole (PTZ). Rats were mechanically ventilated to maintain blood gases and pH within the normal physiological range and body temperature maintained at 37.0 ± 0.2 °C.
After instrumentation, rats were secured in a stereotaxic apparatus and tapered off isoflurane anesthesia and maintained on chloral hydrate anesthesia for the remainder of the experiment. Chloral hydrate was used because it is less vasoactive than isoflurane and has previously been used in studies measuring hippocampal blood flow via hydrogen clearance.9,32,33 Through a burr hole, a 50 µm tip glass hydrogen microsensor (Unisense, Aarhus, Denmark) was inserted into the CA3 region of the hippocampus (−3.5 mm posterior, 3.0 mm lateral, 3.5 mm ventral to bregma). 34 The hydrogen microsensor was calibrated daily, and the hydrogen current sampled at 5 Hz and recorded using a multimeter (Unisense, Aarhus, Denmark). After microsensor placement, rats inhaled 4% hydrogen gas until saturation was reached, as evidenced by the hydrogen current reaching a steady state. Hydrogen gas was then turned off and tissue desaturation recorded under baseline conditions. Absolute hippocampal blood flow was calculated from the rate of hydrogen clearance using the initial slope to determine the first-order clearance rate in which: flow (mL/sec) = 0.693/t1/2, where t1/2 is the time in seconds to reach half of the maximal tissue concentration of hydrogen, and then converted to mL/100g tissue/min using following equation: blood flow = (0.693 * 100 g *60sec)/t1/2. To measure hippocampal blood flow during seizure, hydrogen was again inhaled until saturation reached. PTZ was then infused at doses twice that of seizure threshold, hydrogen gas turned off and tissue desaturation recorded during seizure, as previously done.9 To account for differences in PTZ-induced seizure threshold between groups and to elicit hippocampal seizures sufficient to cause a hyperemic response, each group was administered a dose of PTZ twice that of their threshold dose: 74 mg/kg in Preg rats and 40 mg/kg in ePE rats. 9 These doses elicited seizures of similar severity, as detected via intrahippocampal EEG in preliminary studies. Vecuronium was used to initiate paralysis (0.05 mg/kg) to avoid physical convulsion during seizure. From tissue desaturation measurements, the half-life of hydrogen was determined and used to calculate absolute blood flow.
Elastin content of aorta
Verhoeff-van Gieson (VVG) staining of thoracic aorta was used to measure elastin content and cross-sectional area, as previously described. 32 Sections of aorta were removed and fixed in 10% buffered formalin overnight at 4 °C. After fixation, aortic rings were placed in 0.1 M phosphate buffered solution (PBS) 4 °C, then paraffin embedded oriented in cross-section. Three sections 5 µm thick were taken 200 µm apart from each aortic ring and stained for elastin. VVG staining was performed using standard techniques on paraffin sections. One image was captured at 4X and four images captured at 40X magnification from each ring using an Olympus BX50 microscope. Elastin content was quantified from the 40X images by calculating the mean grey scale value of the medial layer of the aortic wall using ImageJ (NIH, Bethesda, MD, USA) and averaged within animal and group, as previously done. 32 Cross-sectional area was measured off 4X images using ImageJ (National Institutes of Health, Bethesda, MD). Images were analyzed by an investigator blinded to group.
Measurement of tumor necrosis factor-α (TNF-α) and 8-Isoprostane
Serum levels of TNF-α and 8-Isoprostane were measured in serum from Preg (n = 6), ePE (n = 7) and ePE + apo (n = 6) via commercially available enzyme linked immune-sorbent assay (ELISA) kits (TNF-alpha: R&D Systems RTA00 8-Isoprostane: Cayman Chemicals 516351). Samples were ran undiluted, in duplicate and according to manufacturer’s instructions.
Drugs and solutions
Apocynin, chloral hydrate, PTZ, SNP, NS309, papaverine, diltiazem and ingredients for aCSF and 0.1 M PBS were purchased from Sigma Aldrich (St. Louis, MO, USA). ET-1 was purchased from Enzo Life Sciences. 10% formaldehyde solution was purchased from Santa Cruz Biotechnology (Dallas, TX, USA). Chloral hydrate and PTZ were made daily in sterile lactated Ringer’s solution. Vecuronium was purchased from the University of Vermont Medical Center Pharmacy Services (Burlington, VT, USA) and diluted in sterile lactated Ringer’s solution and used for 5 days. Apocynin (1.5 mM) treated drinking water was made weekly by dissolving apocynin in hot (∼60°C) tap water while mixing. Apocynin treated drinking water was then allowed to cool to room temperature before given to rats. Stock solutions of SNP, papaverine and diltiazem were made weekly and stored at 4°C until use. NS309 and ET-1 stock solutions were aliquoted and stored at −20°C until use. aCSF was made weekly (mM): NaCl 122.0, NaHCO3 26.0, NaH3PO4 1.25, KCl 3.0, MgCl2 1.0, CaCl2 2.0. Calcium-free aCSF was also made weekly, using the same ingredients as above, excluding CaCl2 and with the addition of 0.5 EGTA. Both aCSF solutions were stored at 4 °C without glucose. On the day of the experiment, 4.0 mM glucose was added, and aCSF was aerated with 5% CO2, 10% O2, and 85% N2 to maintain pH at 7.4.
Data calculations and statistical analyses
The number of animals used in each experiment was justified by statistical power calculation based on our previous studies using similar methodology.9,32 Results are presented as mean ± SD. Using GraphPad Prism 8.0 software (GraphPad Software Inc., La Jolla, CA), differences between the three groups were determined by D’Agostino-Pearson Omnibus normality test and either one-way analysis of variance (ANOVA), paired t-test, or Dunnett’s test with a post-hoc Bonferroni’s test to correct for multiple comparisons when appropriate. Differences in CBF in response to seizure were compared to baseline within group using a parametric paired t-test. Differences were considered significant at p < 0.05.
Myogenic tone was calculated at each pressure as a percent decrease in diameter from the fully relaxed diameter in calcium-free aCSF with diltiazem and papaverine by the equation: [1−(ϕtone/ϕpassive)] × 100%; where ϕtone is the inner diameter of the vessel with tone and ϕpassive is the inner diameter of fully relaxed vessel. Percent constriction to ET-1 was calculated as a percent change in diameter from baseline by the equation: [1−(ϕdrug/ϕbaseline)] × 100%; where ϕdrug is the diameter of vessel after drug exposure, and ϕbaseline is the diameter before giving drug. Percent reactivity to NS309 and SNP was calculated from the equation: [(ϕdose−ϕbaseline)/(ϕpassive−ϕbaseline)] × 100%; where ϕdose is the diameter at a specific concentration of drug. For wall stress and wall tension all diameter and wall thickness (WT) measurements were converted to cm. Wall stress was calculated by the equation: Wall Stress = wall tension/wall thickness, where wall tension was calculated by converting pressure (mmHg) into dynes/cm2 × (φinner/2).
Results
Effect of ePE and apocynin on HA function and structure
To investigate the role of NADPH oxidase in mediating HA dysfunction and remodeling in ePE rats, isolated and pressurized HAs were studied. One Preg animal was excluded due to technical difficulties. Figure 1(a) shows that the diameter of HAs was not different between Preg and ePE rats at any pressure studied. Preg and ePE rats had similar levels of myogenic tone across the intravascular pressure range studied, however, ePE rats treated with apocynin had significantly increased myogenic tone (Figure 1(b)).
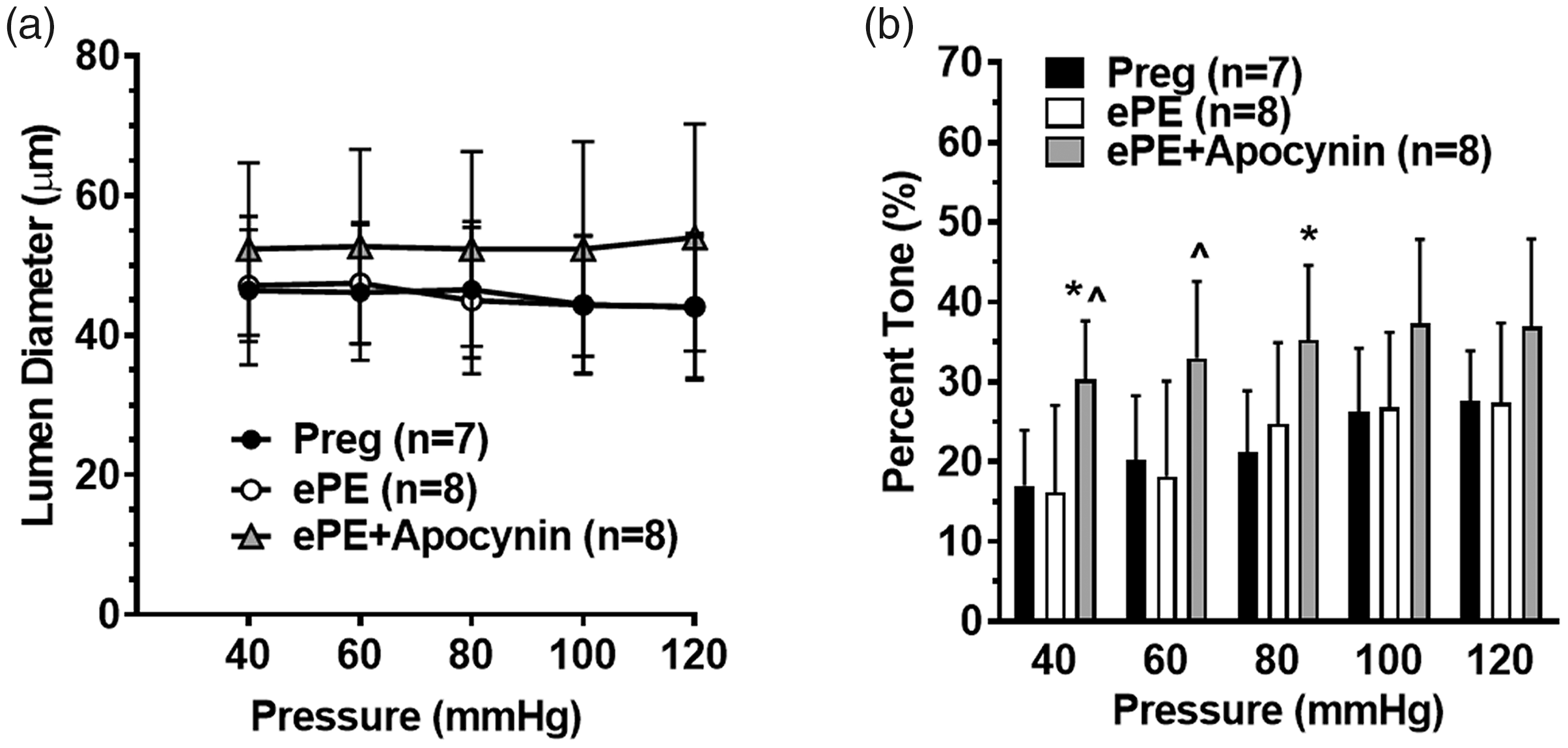
Active lumen diameters and myogenic tone of HAs from Preg, ePE and ePE + apo rats. (a) Lumen diameter of HAs over a wide range of pressures. There was no difference in lumen diameter between groups. (b) Myogenic tone of HAs from all groups. There was a significant increase in tone in HAs from ePE + apo animals compared to the other two groups at 40, 60 and 80 mmHg. Data are presented as mean ± SD; *p < 0.05 vs. Preg; ^p < 0.05 vs. ePE by one-way ANOVA and post hoc Bonferroni test for multiple comparisons.
Although there was no difference found in baseline diameters or tone of HAs in ePE vs. Preg rats, there was both endothelial and smooth muscle dysfunction seen in arterioles from ePE rats. Figure 2 shows the concentration responses to ET-1, NS309 and SNP. HAs from ePE rats were less reactive to ET-1 and SNP compared to Preg rats, demonstrating smooth muscle dysfunction. In addition, reactivity to the endothelium-dependent vasodilator NS309 was also diminished in HAs from ePE rats, suggesting endothelial dysfunction as well. Treatment with apocynin restored reactivity to ET-1 (Figure 2(a)) and NS309 (Figure 2(b)), but not SNP (Figure 2(c)).
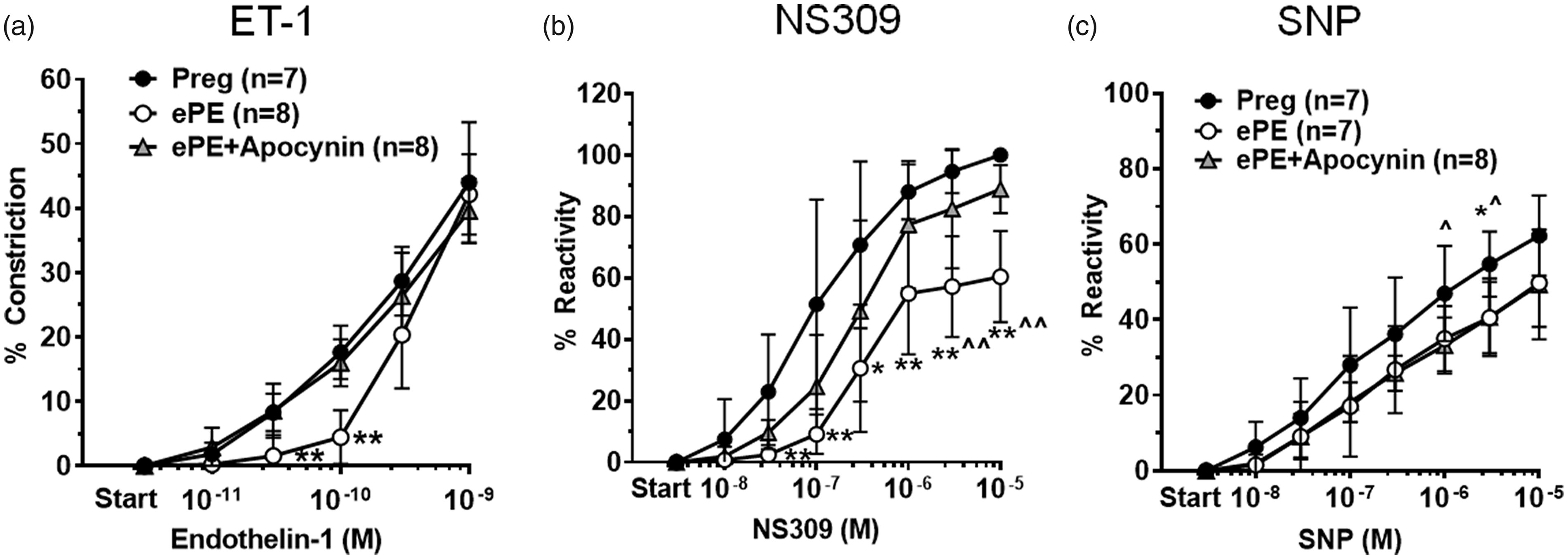
Concentration-response curves to ET-1, NS309 and SNP in HAs. (a) Percent constriction to ET-1. HAs from all groups of animals constricted in a dose-dependent manner to ET-1; however, HAs from ePE rats constricted less to lower concentrations of ET-1 than arterioles from Preg. Preg **p < 0.01 vs. Preg and ePE + apo; (b) Dilation to NS309. HAs from all groups dilated to NS309. Dilation was impaired in HAs from ePE rats compared to Preg. Treatment with apocynin restored the dilation to NS309 in HAs from ePE rats. *p < 0.05 and **p < 0.01 vs. Preg; ^p < 0.05 and ^^p < 0.01 vs. ePE+apo by one-way ANOVA and post hoc Bonferroni test for multiple comparisons. (c) Dilation of HAs to SNP was impaired in ePE compared to Preg that was not restored by apocynin. ^p < 0.05 vs. ePE+apo; *p < 0.05 vs. ePE by one-way ANOVA with posthoc Bonferroni test for multiple comparisons. Data are presented as mean ± SD.
Figure 3 shows the structural and biomechanical properties of HAs from all groups. HAs from ePE and Preg rats were not different in diameter (Figure 3(a)) or wall thickness (Figure 3(b)). However, arterioles were considerably stiffer from ePE rats as shown by the leftward shift in the stress-strain curves (Figure 3(c)). Treatment of ePE rats with apocynin caused a significant increase in passive lumen diameter of ePE + apo HAs and increased wall thickness at lower pressure (Figure 3(a) and (b)). The increase in lumen diameter in ePE + apo rats was associated with a substantial rightward shift in the stress-strain curve, demonstrating apocynin treatment decreased ePE-induced HA stiffness and increased distensibility (Figure 3(c)).
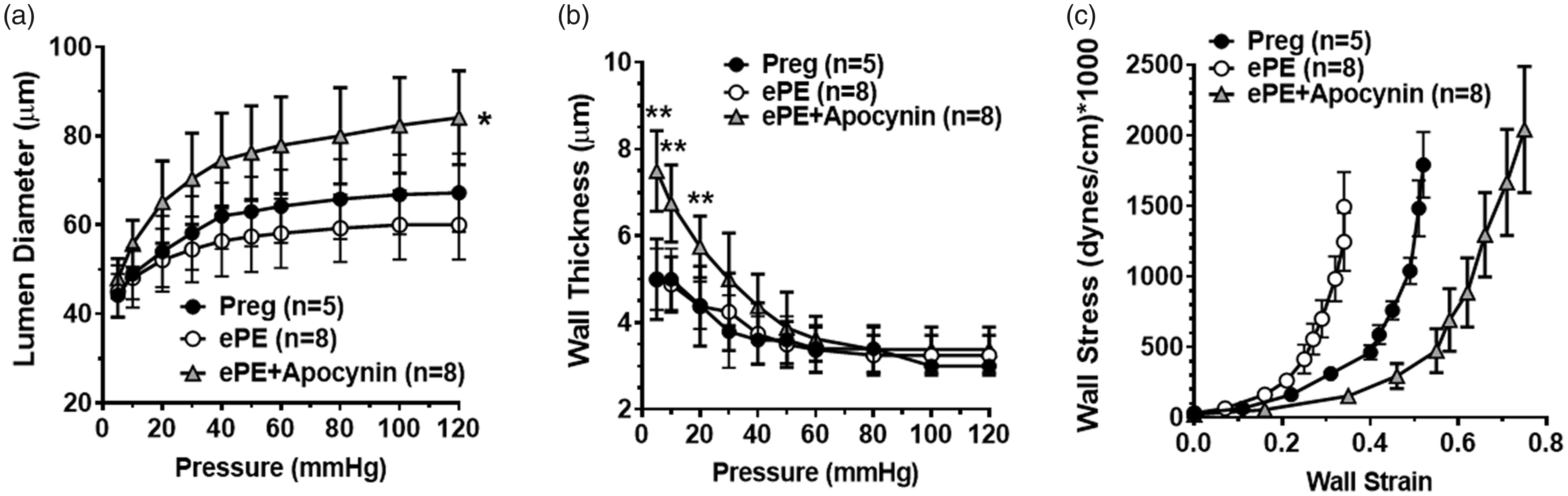
Passive structural and biomechanical properties of HAs from all groups. (a) Passive lumen diameters of HAs over a wide range of pressures. There was no difference in passive HA diameters between Preg and ePE; however, apocynin treatment significantly increased lumen diameter. *p < 0.05 vs. Preg and ePE groups by one-way ANOVA and post hoc Bonferroni test for multiple comparisons. (b) Wall thickness was not different between HAs from Preg and ePE but was significantly greater in ePE + apo HAs at pressures below 40 mmHg. **p < 0.01 vs. Preg and ePE groups. (c) Stress-strain curves of HAs from all groups. There was a leftward shift in the curve from ePE vs. Preg, suggesting increased vascular stiffness. However, treatment with apocynin shifted the curve to the right, demonstrating decreased vessel stiffness compared to both groups. Data are presented as mean ± SD.
Seizure-induced hyperemia was restored by apocynin in ePE rats
Our previous study found that the hyperemic response to seizure in the hippocampus was absent in ePE rats and that blood flow decreased during seizure by ∼15%. 9 In the current study, we also found hippocampal blood flow decreased in ePE rats during seizure. Figure 4(a) to (c) shows absolute blood flow measured in the hippocampus at baseline and again during PTZ-induced seizure plotted as individual rats. Seizure increased hippocampal blood flow in Preg rats that was decreased in ePE; treatment with apocynin restored the hyperemic response to seizure in ePE + apo rats that was not as robust as in Preg animals. Figure 4(d) and (e) show the percent change and absolute change in CBF during seizure from baseline in all groups. Baseline blood flow was not different between groups (in mL/100g/min): 61.8 ± 4.7 for Preg (n = 6), 61.4 ± 4.2 for ePE (n = 6) and 60.0 ± 4.2 for ePE + apo (n = 6; p > 0.05).
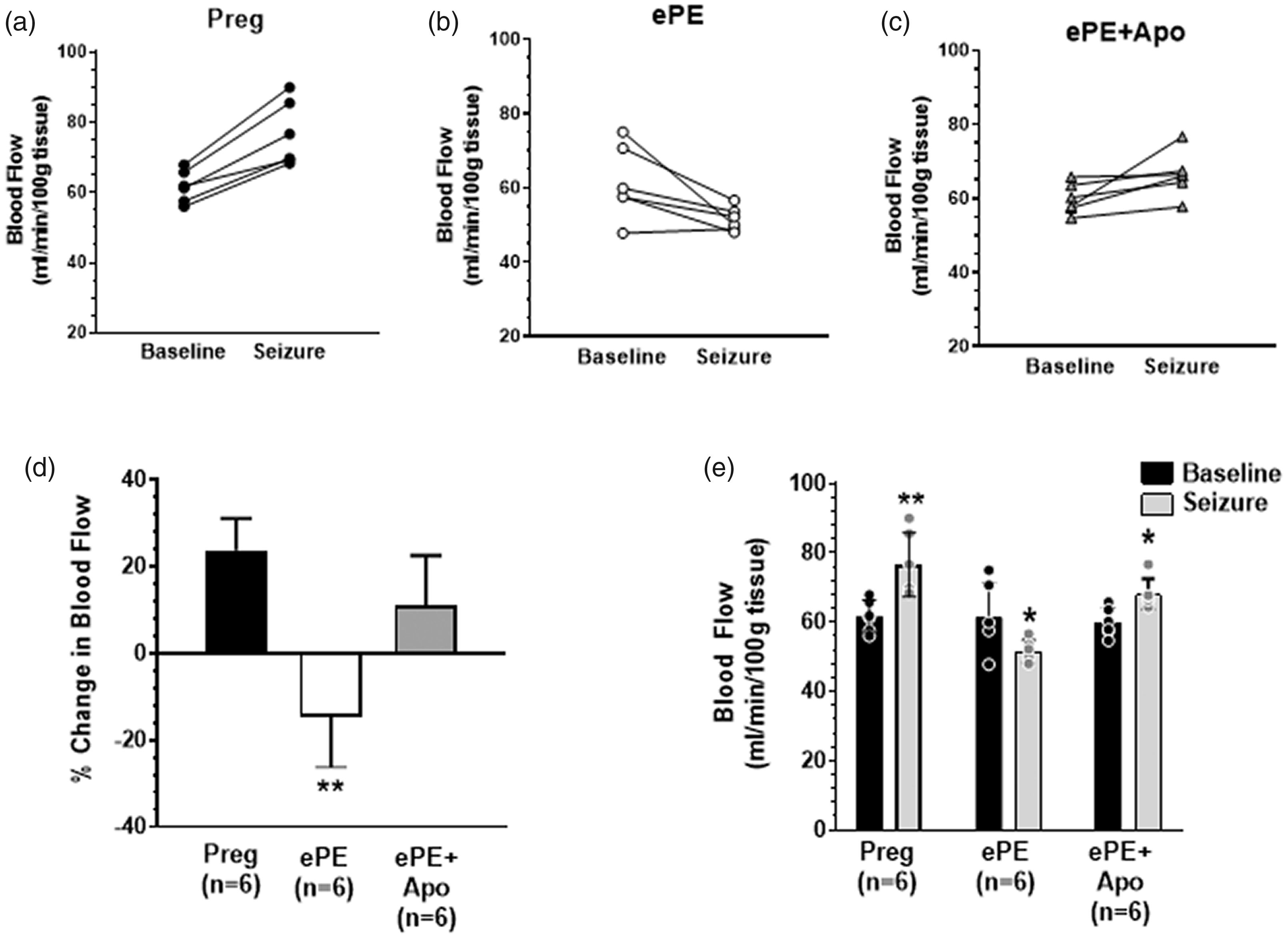
Changes in CBF in hippocampus from all groups at baseline and during seizure. (a–c) CBF measured by hydrogen clearance at baseline and during seizure in individual animals. CBF increased in Preg but decreased in ePE animals; apocynin treatment restored the hyperemic response to seizure, but was not as robust as in Preg animals. (d) Summary data showing the percent change in CBF in response to seizure calculated from baseline. **p < 0.01 vs. Preg and ePE + apo by one-way ANOVA with posthoc Bonferroni test for multiple comparisons. (e) Summary data showing absolute CBF in all groups both at baseline and in response to seizure. *p < 0.05 and **p < 0.01 vs. baseline by paired t-test. Data are presented as mean ± SD.
Apocynin did not prevent decreased elastin content in aorta from ePE rats
Hypertension and oxidative stress can degrade elastin fibers of large arteries, causing increased stiffness. We therefore measured elastin content in the media of aorta from all groups of animals. Figure 5(a) shows representative photomicrographs of cross-sections of aorta from all groups stained for elastin with VVG stain. The elastin lamellae show as prominent dark striations. The summary graph in Figure 5(b) shows that elastin content was decreased in aorta from ePE animals, suggesting large vessel stiffness in this model of PE. Treatment of ePE animals with apocynin did not prevent the elastin degradation, as elastin content was similar between ePE and ePE + apo groups. There was no difference in aortic cross-sectional area between groups (mm2): 0.498 ± 0.068 for Preg (n = 8); 0.498 ± 0.025 (n = 8) for ePE and 0.488 ± 0.017 (n = 8) for ePE + apo; p > 0.05.
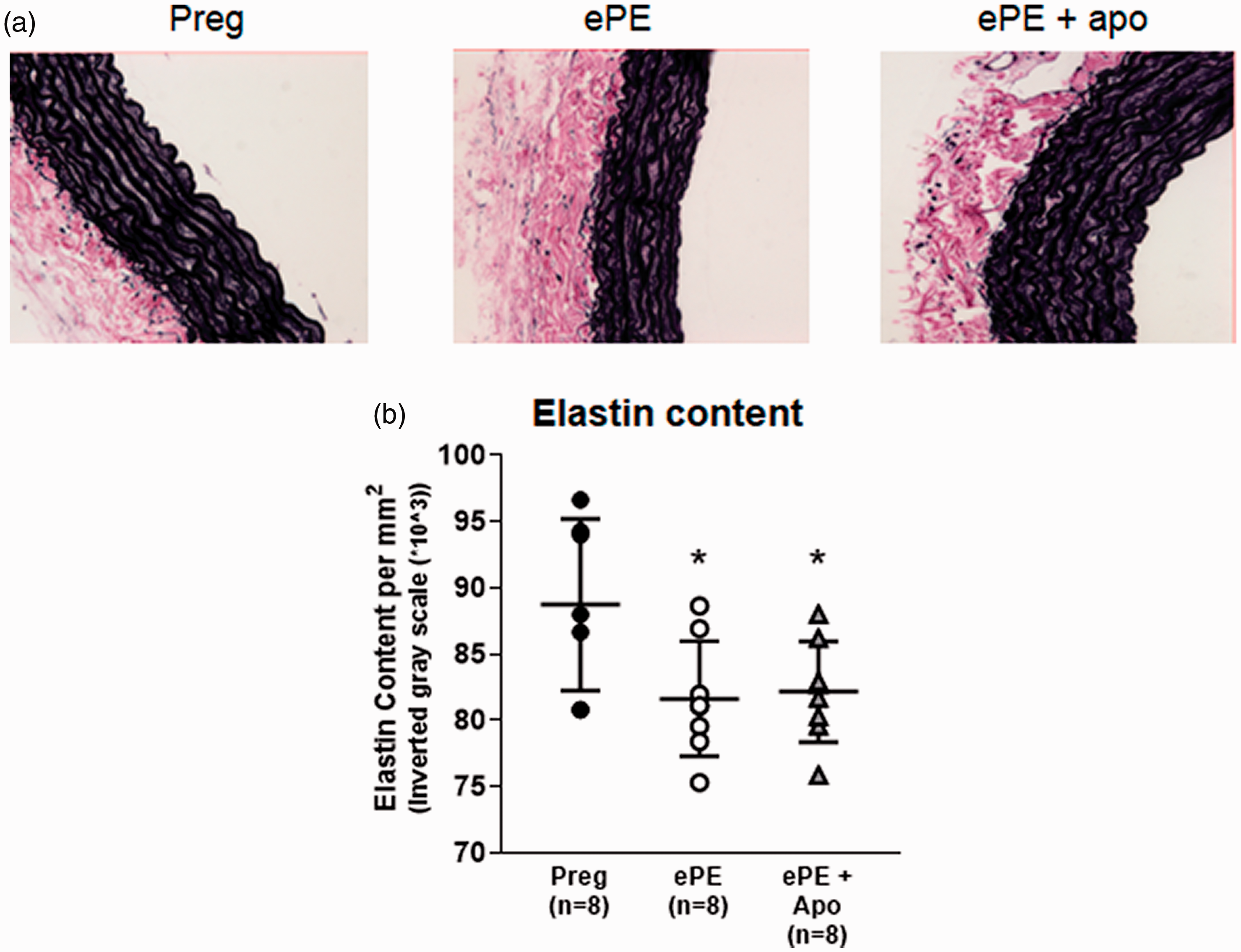
Elastin content of aorta from all groups. (a) Representative photomicrographs of aortic cross-sections stained for elastin with VVG stain. The dark lamellae are prominent in medial layer. (b) Elastin content of aorta quantified by image analysis. There was a decrease in elastin content in aorta from ePE vs. Preg that was not restored by apocynin. *p < 0.05 vs. Preg by one-way ANOVA with posthoc Bonferroni test for multiple comparisons. Data are presented as mean ± SD.
Effect of ePE and apocynin on circulating inflammatory factors
Figure 6 shows serum levels of 8-isoprostane and TNF-α from all groups of animals, markers of oxidative stress and inflammation, respectively. ePE rats had increased 8-isoprostane and TNF-α, as we have shown previously. Interestingly, treatment with apocynin prevented the increase in 8-isoprostane, but increased the level of circulating TNF-α.
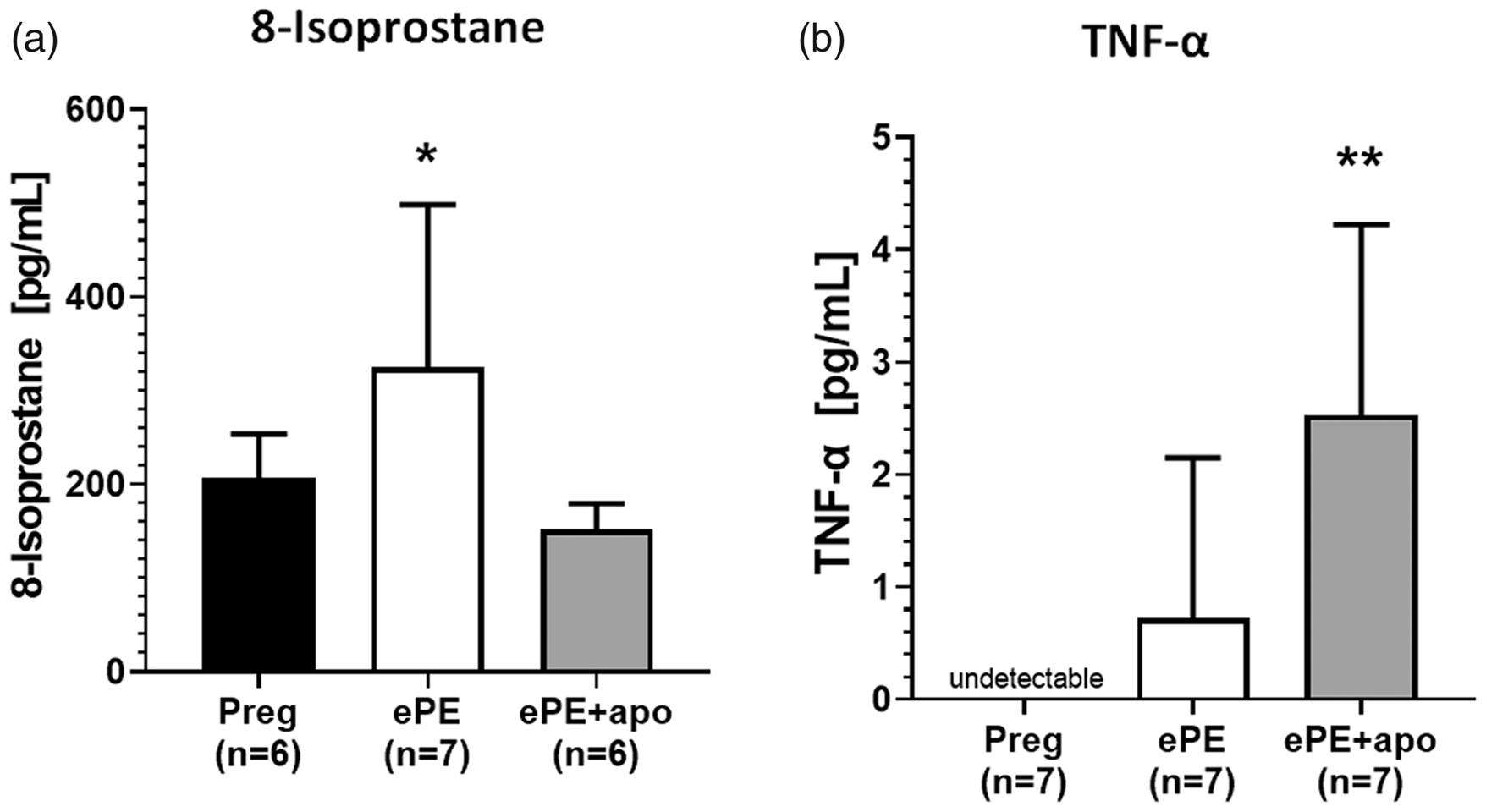
Serum levels of inflammatory markers. (a) There was a significant increase in serum levels of 8-isoprostane from ePE that was prevented by treatment with apocynin. *p < 0.05 vs. ePE by one-way ANOVA and Tukey’s test for multiple comparisons. (b) Serum levels of TNF-α were undetectable in Preg animals and increased in ePE. Apocynin treatment increased TNF-α in ePE + apo animals. **p < 0.05 vs. Preg by Dunnett’s test. Data are presented as mean ± SD.
Blood pressures
Table 1 shows blood pressures taken by tail cuff for all groups of animals. Systolic, but not diastolic, blood pressure was higher in ePE and ePE + apo groups compared to Preg that also increased mean blood pressure. Thus, apocynin did not lower elevated blood pressure in ePE rats.
Non-invasive blood pressure measurements of normal pregnant (Preg), experimental preeclamptic (ePE) and ePE rats treated with apocynin (ePE + Apo) on day 20 of gestation.

Results are mean ± SD. **p < 0.01 vs. Preg by one-way ANOVA with Bonferroni’s post-hoc test for multiple comparisons.
Discussion
In the present study, we found that HAs from ePE rats were stiffer and had vascular dysfunction compared to HAs from normal Preg rats as well as impaired seizure-induced hyperemia in the hippocampus. Treatment of ePE rats with apocynin, a purported NADPH oxidase inhibitor and antioxidant, improved endothelial and vascular smooth muscle function of HAs, decreased HA stiffness and restored seizure-induced hyperemia in the hippocampus. The increase in vessel stiffness in ePE rats appeared to be widespread as there was decreased elastin content in aorta. However, while apocynin substantially decreased vessel stiffness in HAs it did not affect elastin content in aorta or lower blood pressure, suggesting a selective effect of apocynin on resistance vessels, and possibly cerebral vessels.
HAs perfuse the hippocampus, a brain region important for memory and cognition that is also highly susceptible to seizure and seizure-induced injury.9,27 Thus, the structure and function of HAs are of interest to conditions that are associated with cognitive decline such as PE.2 –7 In the current study we found that HAs from ePE dams had endothelial and vascular smooth muscle dysfunction compared to HAs from normal Preg dams, including impaired reactivity to ET-1 and NS309. Treatment with apocynin restored the ePE-induced HA dysfunction back to the level of arterioles from Preg rats, suggesting a role for oxidative stress in mediating HA dysfunction during ePE. Apocynin did not prevent impaired HA dilation to SNP that was diminished in ePE rats, suggesting selectivity in apocynin’s effect. Similar to our previous study, we did not find increased myogenic tone in HAs from ePE dams that was similar to those from Preg animals. Unexpectedly, apocynin increased tone in HAs from ePE animals in the face of considerably reduced stiffness even compared to normal Preg dams (Figure 3). It is unlikely that the reduced stiffness had a role in increasing pressure-induced tone since there was not a similar increase in other vasoconstrictor stimuli (e.g., ET-1). The increase in myogenic tone in HAs from ePE + apo rats appeared to have normalized active lumen diameters in the face of increased distensibility since there was no difference in active diameters between groups (Figure 1(a)). In addition, baseline hippocampal blood flow was similar between groups supporting the concept that increased tone offset the increase in distensibility to maintain hippocampal perfusion (Figure 4). However, the increase in HA distensibility may have been important for restoring seizure-induced hyperemia as those animals had greater vasodilatory capacity.
In vivo apocynin treatment had effects on both the structure and function of HAs in ePE + apo rats, including increasing myogenic tone, normalizing ET-1 constriction and NS309 dilation as well as substantially increasing passive distensibility. These effects on HAs are not likely due to inhibition of NADPH oxidase directly because apocynin inhibits NADPH oxidase only in the presence of myeloperoxidase (MPO) to produce its active metabolite diapocynin.35,36 Endothelial and smooth muscle do not contain MPO and therefore the effects of apocynin on HAs are more likely due to its antioxidant and/or radical scavenging properties.30,35 In support of this, apocynin normalized increased 8-isoprostane levels in ePE + apo, a marker of lipid peroxidation that results from oxidative stress, confirming its antioxidant properties. In a previous study, chronic apocynin treatment of spontaneously hypertensive rats (SHR) improved mesenteric artery and aortic reactivity through increased endothelial cell calcium and endothelial nitric oxide synthase (eNOS) expression, without affecting soluble guanylyl cyclase (GC) levels. 30 It is possible that apocynin’s effect on restoring NS309 dilation in HAs was due to increased endothelial cell calcium since NS309 activates SK/IK channels that are calcium-dependent. Improved ET-1 reactivity, but not dilation to SNP, could be related to apocynin’s ability to scavenge ROS in vascular smooth muscle without affecting GC levels. 30 If apocynin also increased vascular smooth muscle calcium, this could explain the increase in myogenic tone in HAs from ePE + apo rats. Apocynin has also been shown to prevent reactive oxygen species (ROS)-sensitive activation of voltage-operated calcium channels. 37 However, because myogenic tone was increased in HAs from apocynin treated rats, it is unlikely that the effect of apocynin was related to inhibition of calcium channels in vascular smooth muscle since that would cause relaxation and less tone. The effect of apocynin on HA distensibility was substantial and beyond that of arterioles from Preg rats. It is unclear how apocynin had such an effect on HA structure, but it may be related to apocynin scavenging ROS that may be high in the physiological state of pregnancy. A limitation to this study is that we did not treat Preg rats with apocynin; however, our focus was on treating HA dysfunction during in ePE rats and restoring seizure-induced hyperemia, not on the mechanism of action of apocynin.
We measured the effect of apocynin treatment of ePE rats on the vasculature and functional responses to seizure during the index pregnancy because seizure occurs more frequently in PE women, a condition known as eclampsia. Eclampsia is not benign and is associated with retrograde and anterograde amnesia. 27 Women with prior PE or eclampsia have long lasting consequences on the brain, including cognitive decline at young ages remote from pregnancy. 27 In the current study, we found that in addition to restoring HA function, apocynin treatment also restored the hyperemic response to seizure in the hippocampus of ePE dams, potentially due to HAs being larger in size passively, with substantially increased distensibility, and therefore having increased vasodilatory capacity. These findings demonstrate a potential role for oxidative stress and NADPH oxidase in impaired seizure-induced hyperemia in the hippocampus during ePE.
A previous study found that eclampsia-like seizures caused acute and long-lasting hippocampal dysfunction in ePE; 27 however, whether restoring HA function with apocynin treatment improves seizure-induced memory deficits requires further investigation. Further, it is not clear if the changes noted here in HAs and activity-induced increases in hippocampal blood flow in ePE rats persist post-partum. If these changes are persistent into later life, they could contribute to early onset cognitive dysfunction that formerly PE women report. In fact, although memory appears intact during the index pregnancy, this model of ePE demonstrates memory decline that occurs several weeks after pregnancy independently of seizures due to changes in hippocampal neuroplasticity. 27 Whether there is a role for HA dysfunction in the progressive cognitive decline after ePE, and whether restoring HA function with apocynin treatment could slow such decline remains to be investigated. Given the important role of the hippocampus and hippocampal perfusion in memory and cognition, HAs seem like an important therapeutic target.
Apocynin treatment had no effect on aortic elastin content that was decreased in ePE rats. Our previous study reported a strong correlation between aortic stiffness and elastin content by directly measuring elastin content in the aorta and stiffness of isolated and pressurized aorta. 32 Thus, the decrease in elastin is likely related to and potentially causal to an increase in aortic stiffness in ePE rats. PE women are also known to have increased aortic stiffness, measured by pulse wave velocity.20,21,26 Elastin can degrade from higher mechanical pulsatility during hypertension, causing increased stiffness. 38 The importance of finding large artery stiffness in PE and ePE rats is that it has been associated with long-term cardiovascular disease risk and early-onset cognitive decline.19,23,39 In addition, HAs had increased stiffness from ePE rats, suggesting widespread vascular dysfunction and structural damage. That apocynin had no effect on aortic elastin content but decreased stiffness in HAs suggests regional differences in the response to treatment; HAs responding without affecting aorta. Regional differences in the effectiveness of treatment may be beneficial and allow targeting of vascular segments and particular pathophysiologic processes. However, it could also limit usefulness during pregnancy if reproductive organs are adversely affected such as the uteroplacental circulation.
Conclusions
PE women are more prone to eclamptic seizures and are at risk of early onset cognitive decline. Formerly PE women are also at increased risk of cardiovascular disease and stroke later in life. Finding that ePE rats had HA dysfunction and impaired seizure-induced hyperemia suggests neurovascular complications occur during PE that could have long-lasting consequences, particularly in the setting of eclamptic seizures. Treatment with apocynin seemed to selectively protect the hippocampal vasculature by improving both endothelial and vascular smooth muscle function and restored seizure-induced hyperemia. However, treatment was ineffective at preventing decreased SNP reactivity, lowering blood pressure and restoring elastin content in the aorta in ePE rats. Thus, this study showed that oxidative stress may be an underlying mechanism by which ePE caused HA dysfunction and impaired hippocampal blood flow during seizure that could potentiate cognitive decline associated with PE and exacerbate seizure-induced hippocampal injury. Finding treatments that prevent cerebral microvascular damage during PE may protect PE women from neurocognitive issues and protect the hippocampus from eclamptic seizures.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Institutes of Health, National Institute of Environmental Health Sciences (NIEHS); National Institute of Neurological Disorders and Stroke (NINDS); National Institute on Drug Abuse (NIDA); National Institute of Mental Health (NIMH) grant no. 1R01 NS108455 (MJC) and the National Center for Research Resources grant no. 1S10OD025030-01. This project was also supported by the American Heart Association Career Development Award 20CDA35310239 (ACJ). Imaging work was performed at the Microscopy Imaging Center at the University of Vermont (RRID# SCR_018821).
Acknowledgements
We gratefully acknowledge the support of the Cardiovascular Research Institute of Vermont and the Totman Medical Research Trust.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
MJC designed the study, analyzed and interpreted the data and wrote the manuscript; ST performed experiments, analyzed data and wrote parts of the manuscript; NB performed experiments and analyzed data; DA performed experiments and analyzed data; ACJ designed the study, performed experiments, analyzed and interpreted the data and wrote parts of the manuscript.
