Abstract
Blood-brain barrier (BBB) disruption is a pivotal pathophysiological process in ischemic stroke. Although temporal changes in BBB permeability during the acute phase have been widely studied, little is known about the chronic phase of cerebrovascular changes that may have a large impact on the long-term outcome. Therefore, this study was aimed to measure cerebral vascular abnormalities using CT perfusion in nine rhesus monkeys subjected to transient middle cerebral artery occlusion (tMCAO) for ≥1 year (MCAO-1Y+). The level of cerebral perfusion demonstrated by mean transit time was significantly higher in the ipsilateral caudate nucleus, white matter, thalamus, hippocampus, and contralateral thalamus in MCAO-1Y+ compared with the other nine age-matched control monkeys. The increase in BBB permeability measured through the permeability surface was found in the same ten regions of interest ipsilaterally and contralaterally. We also found decreased levels of Aβ 42/40 ratio in the cerebrospinal fluid (CSF), suggesting a potential link between post-MCAO cognitive decline and Aβ metabolism. Overall, we demonstrated significant cerebral hypoperfusion, BBB disruption, and CSF Aβ decrease during the rehabilitation stage of ischemic stroke in a non-human primate model. Future studies are needed to elucidate the cause-effect relationship between cerebrovascular disruptions and long-term neurological deficits.
Keywords
Introduction
Ischemic stroke remains the second leading cause of death and the third leading cause of disability in adults worldwide. 1 Up to 75% of stroke survivors require some level of long-term assistance, which imposes a significant burden on global health and the economy. 2 Although the cascade of molecular events that occur during the acute phase of ischemic stroke have been widely studied, only a few studies have focused on chronic pathophysiology that may significantly contribute to the long-term outcome and functional recovery following this devastating disease. 3
A considerable proportion of stroke survivors (>25%) suffer from dementia and neuropsychiatric disorders. 4 Dysfunction of cerebral autoregulation and neurovascular coupling (NVC) in otherwise healthy brain regions may significantly affect post-stroke cognitive and neuropsychiatric impairment. Normal brain function critically depends on the proper regulation of cerebral blood flow (CBF) that matches brain metabolic demands and the delivery of oxygen and nutrients. 5 A sudden drop in CBF is a hallmark of acute ischemic stroke (AIS) and is the leading cause of hypoxic injury. 6 Post-reperfusion CBF can be mostly restored, but it remains unclear if it will ever return to its original level.7–9 CBF is regulated at different levels, including 1) cerebral autoregulation, the cerebral vascular response to perfusion pressure; 2) NVC, a mechanism that results in local CBF regulation in response to localized active neurons; 3) vascular reactivity at arteriole or capillary level (including carbon dioxide).5,9–11
The blood-brain barrier (BBB) is an essential structure in the neurovascular unit (NVU) and plays a vital role in NVC. 12 The maintenance of BBB structure and function is crucial for providing neuroprotective effects in many neurovascular and neurodegenerative diseases. 13 The BBB opens during various stages of ischemic stroke, either continuously or biphasically.14–16 Previously, we showed that temporal changes in BBB permeability that occur within the first seven days following longitudinal transient middle cerebral artery occlusion (tMCAO) using rhesus monkeys, and demonstrated the strong correlation between BBB disruption and stroke outcome. 17
Even though many studies have focused on the acute and subacute stroke stages, only a few of these studies have found impaired CBF regulation and BBB disruption at the chronic stage. Most of these studies have described the situation for only three months following a stroke.3,18,19 In human patients, Krainik et al. described regional impairment of cerebrovascular reactivity and blood-oxygen-level-dependent (BOLD) signal in 8 fully recovered patients at least 32 months after the onset of stroke. 20 In a post-stroke case examined after one year, Naqvi et al. reported chronic BBB disruption and white matter hyperintensities spreading into white matter regions with a normal appearance. 21 Yang et al. showed disturbed BBB in the non-infarcted basal ganglia and thalamus three months post lacunar/subcortical ischemic stroke. 22 In rodent models of stroke, Morgan et al. demonstrated the significant late opening of the BBB 14 days post-MCAO in rats using gadolinium-enhanced MRI. 23 Garbuzova-Davis et al. demonstrated compromised BBB integrity in rats three months post-ischemic attack. 24
Though the effects of cerebrovascular conditions on long-term brain recovery after stroke are of immense importance, very few studies have described how the conditions change spatially or temporally during the rehabilitation process. Therefore, this study aimed to evaluate cerebral perfusion and BBB disruption in rhesus monkeys that had undergone tMCAO more than one year earlier. We found an overall decrease in cerebral perfusion using CTP scanning and increased BBB permeability in different brain regions that were remote from the original infarction site.
In addition, we looked at cerebrospinal fluid (CSF) levels of amyloid β 1-42 (Aβ42), amyloid β 1-40 (Aβ40), total Tau proteins (tTau), and phosphorylated Tau Thr181 (pTau181). Although post-stroke dementia can always be categorized as vascular dementia, it is of interest to investigate the relationship between vascular impairment and Aβ metabolism. 25 The two-hit vascular hypothesis suggests that predisposed vascular damage leads to impaired Aβ clearance and increased accumulation in the brain, 26 which in turn usually leads to a decrease in Aβ42, as well as in the Aβ42/Aβ40 ratio, in CSF. In this study, although we did not detect any cognitive changes in post-MCAO monkeys, there was a significant drop in the Aβ42/Aβ40 ratio in CSF, which may indicate a potential future cognitive decline associated with Alzheimer’s disease (AD), especially the cerebral amyloid angiopathy (CAA) form. Taking advantage of the easily longitudinal assessment of the non-human primate (NHP) stroke model, we hope to carefully design and carry out serial imaging, neurobehavioral, and biochemical studies in the future, to further investigate cerebral vascular abnormalities and the potential link between post-stroke vascular dementia and AD.
Materials and methods
Experimental animals
Nine 4–7 years old male rhesus monkeys, and nine age-matched controls (AMC) that weighed 4–7 kg each, were used in the current study. Initially, these monkeys were provided by Sichuan Green-House Biotech Co., Ltd, Sichuan Province, China, and were used in drug efficacy studies as controls (such as therapeutic antibodies, Amoxicillin, and cannabidiol) at Kangcheng Biotech (Table 1), as shown in Supplementary Table 1. After the acute tMCAO studies were conducted, they were sent back to the original breeding base (Green-House Biotech) and stayed with other monkeys. Prior to imaging and CSF sampling, the monkeys were brought back to the Kangcheng facility [animal production license number: SYXK (Chuan) 2019-215] and housed in monkey cages for about one month under a controlled temperature: 22 ± 2 °C; humidity: 60%∼70% under a 12 hours-alternate light/dark cycle (lights on at 8:00 am and off at 8:00 pm). Standard food and water were provided ad libitum throughout the experiment. All experimental procedures were conducted following guidance for the Care and Use of Laboratory Animals provided by the Ministry of Science and Technology of the People's Republic of China. All animal experiments were performed in agreement with the Animal Research: Reporting in Vivo Experiments (ARRIVE) guidelines and were approved by the Institutional Animal Care and Use Committee (IACUC) of the West China Hospital of Sichuan University (Approval No. 2019192 A). The original infarction volume was comparatively small and accounted for only about 4% of brain volume. 17 After tMCAO, the monkey usually regains its eating ability within 24 hours and regains normal mobility in 7–14 days. 17 Rather than being euthanized, these monkeys should be returned to their colony in accordance with the animal welfare code.
The basic information of rhesus monkeys subjected to tMCAO.
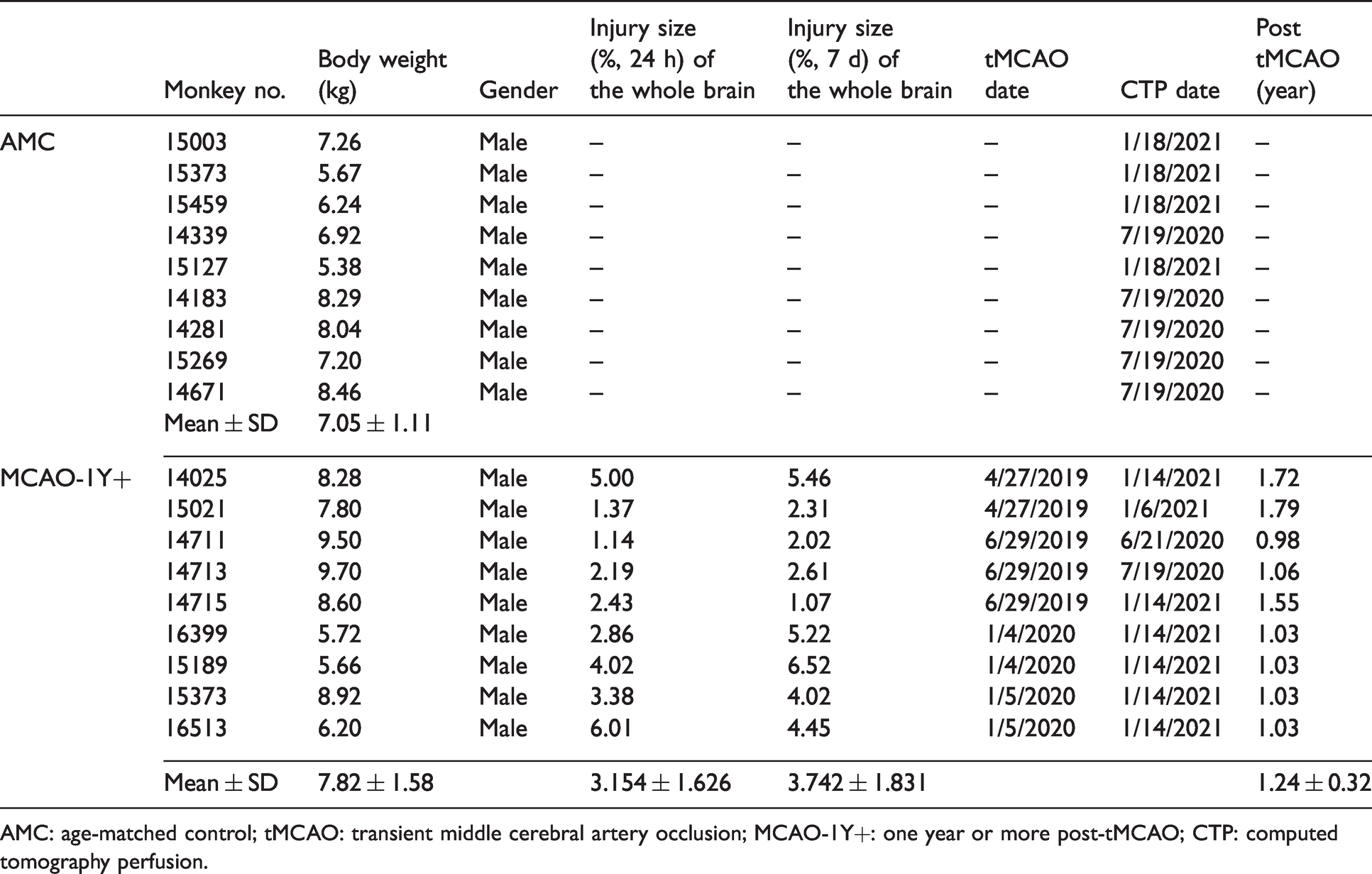
AMC: age-matched control; tMCAO: transient middle cerebral artery occlusion; MCAO-1Y+: one year or more post-tMCAO; CTP: computed tomography perfusion.
Transient Middle cerebral blood artery occlusion
A minimally invasive catheterization method was used to induce tMCAO in these monkeys, as previously described. 17 In brief, the animals were anesthetized using constant-rate propofol (AstraZeneca, 10 mg/mL) administered (0.3–0.4 mg/kg/min) into the upper vein and was monitored using an anesthetic monitor (BeneView T8, Mindray, China). Based on the guidance provided by the manufacturer of the digital subtraction angiography (DSA) system (Allura Xper FD20, Philips, the Netherlands), a 5F guiding catheter (Envoy, Johnson & Johnson, USA) and a 5 F arterial sheath (Terumo Corporation, Japan) were placed using the Seldinger technique from the right femoral artery to the right internal carotid artery. Thereafter, a microcatheter (Excelsior SL-10, Stryker, USA) was inserted using a microwire (Synchro 14, Stryker, USA) into the distal end of the M1 segment of MCA. Subsequently, a Guglielmi Detachable Coil (GDC, 102 × 4, Stryker, USA) was deployed into the M1 segment without detachment to occlude MCA and locally induce embolus formation. The right MCA was blocked for 2 hours, and the GDC was removed from the M1 segment to restore MCA blood flow. After surgery, the animals’ pain was assessed and managed in accordance with the code of practice for the housing and care of laboratory animals.27–30
Magnetic resonance imaging (MRI)
The ischemic brain injury was imaged using MRI scanning before tMCAO, at seven days, and about one year post-tMCAO using a 3.0 Tesla scanner (Signa-Excite, GE, USA). The T1-weighted image (T1WI), T2-weighted image (T2WI), T2 fluid-attenuated inversion recovery (T2 FLAIR), diffusion-weighted image (DWI), and susceptibility-weighted imaging (SWI) were captured. The infarction volumes were analyzed using MRIcro software based on the T2 FLAIR images. Abnormal hyperintensity and total brain parenchyma area were obtained using each MRI slice image, and the volume was derived from the area and thickness. 17 Injury size (% of the whole brain volume) = area of abnormal hyperintensity/total brain area × 100%.
Computed tomography (CT) scan
CT imaging was performed using a second-generation Dual Source Double 64-Detector Row CT scanner (Siemens Healthineers GmbH, SOMATOM Definition Flash, Germany) 0.6 × 64. A 24 G indwelling needle was placed into the experimental animal median cubital vein before the subject was laid supine on the CT scan table. Cerebral imaging included baseline plain CT and CT perfusion imaging (CTP).
The CTP settings were 70 kV tube voltage, 150 mA tube current, and 32 × 1.2 mm collimation detector. The CTP scan parameters were as described below: 39.14 seconds, 1.5 mm slice thickness, and 0.28 seconds rotation time. After scanning 30 times, 1380 images were obtained. The window level and width changes included a 200 mm view field, 80 window size, 35 window level, and H20f smooth kernel. The total volume of contrast injected was 10 mL iodinated at an infusion rate of 1.3 mL/s into each monkey per scan.
CTP post-processing was performed using Siemens VPCT-neuro software. CTP parameters were computed based on CTP datasets, while relative mean transit time (MTT), time to peak (TTP), cerebral blood volume (CBV), and CBF were calculated. The parameter MTT was extremely sensitive to cerebral blood flow.
Blood-brain barrier permeability assessment
CTP is commonly used to measure BBB permeability in vivo. Using the permeability surface area product (PS), the BBB was measured in terms of its permeability to contrast material, which has been confirmed to be accurate by many research studies.22,31,32 PS was determined by CTP, which is abbreviated as the BBB-PS thereinafter. The post-processing of BBB-PS was performed using an adiabatic approximation to the Johnson and Wilson model based on CTP datasets using Siemens VPCT-tumor software.
CSF, blood sampling and analysis
CSF and blood sampling
CSF was extracted from the cisterna magna. This collection method was performed using the following steps: The monkeys were anesthetized using 0.1 mL/kg Zoletil 50 (Virbac. France) administered through intramuscular injection. Then, a puncture was made between the occipital and atlas to cisterna magna until the CSF was slowly flowed and collected using 1.5 mL EP tubes. To prevent the excessive loss of CSF, an equivalent artificial cerebrospinal fluid solution (Coolaber Biotech Co., Ltd Beijing, China) was used for replenishment. The blood sample was obtained using a standard procedure, with about 3 mL of venous blood obtained from the median cubital vein. CSF and blood were collected before MCAO (pre-MCAO) and one year after MCAO (MCAO-1Y+) for albumin, Aβ, and tau analysis.
Quantification of albumin
The CSF and serum albumin levels were measured using enzyme-linked immunosorbent assay (ELISA) (ab108788, abcam, USA) and colorimetric methods respectively. The following formula was used to calculate the CSF/serum albumin ratio (AR) = albumin in CSF (mg/L)/albumin in serum (g/L).
Quantification of Aβ40, Aβ42, tTau, and pTau181 in CSF
Levels of Aβ40, Aβ42, tTau, and pTau181 in CSF were determined using a Luminex system (Merck Millipore, Germany) and a human amyloid beta and tau magnetic bead panel (Cat. # HNABTMAG-68K, MILLIPLEX, Germany). The detection ranges used were as follows: Aβ40, 21–15000 pg/mL; Aβ42, 3–2000 pg/mL; tTau, 11–8000 pg/mL; and pTau181, 0.7–500 pg/mL. Each sample was analyzed in duplicate. The average value of the final data was plotted against the standard curve generated using the kit's human Aβ and tau magnetic bead panel standard.
Statistical analysis
All data exhibited a normal distribution as determined by the statistical test of normality (Kolmogorov-Smirnov test and Shapiro-Wilk test) using SPSS 24.0 software (Chicago, IL) and the statistical variables are presented as the mean ± standard deviation (SD). All graphs were generated using GraphPad Prism software (version 8.0, San Diego, CA, USA). The difference between pre-MCAO and MCAO-1Y+ was determined using paired t-test, while AMC and MCAO-1Y+ were compared using unpaired t-test. All statistical analyses were performed using SPSS 24.0. A P < 0.05 was considered statistically significant, while a P value between 0.05 and 0.1 was considered to indicate a change in the trend.
Results
tMCAO model descriptions
All nine rhesus monkeys that had undergone tMCAO more than one year prior had recovered to an indiscernible level, as determined using routine neurological behavioral assessments (data not shown). During the first seven days following ischemia/reperfusion, the injury volume determined using T2-FLAIR was at 3.15 ± 1.63% at 24 hours but increased to 3.74 ± 1.83% at seven days. The infarct volume diminished to an undeterminable level (<0.3% of the whole brain) after 1 to 1.5 years of rehabilitation (1.24 years on average, Figure 1), although certain after-effects such as encephalomalacia and enlarged anterior horn of right lateral ventricle were still manifested in the T2WI and FLAIR (Figure 1). The total brain volume decreased by about 4.1% in three monkeys one year post-MCAO and 7.6% in three monkeys two years post MCAO (Supplementary Table 2). No cerebral microbleeds were observed, as shown in the SWI scanning.
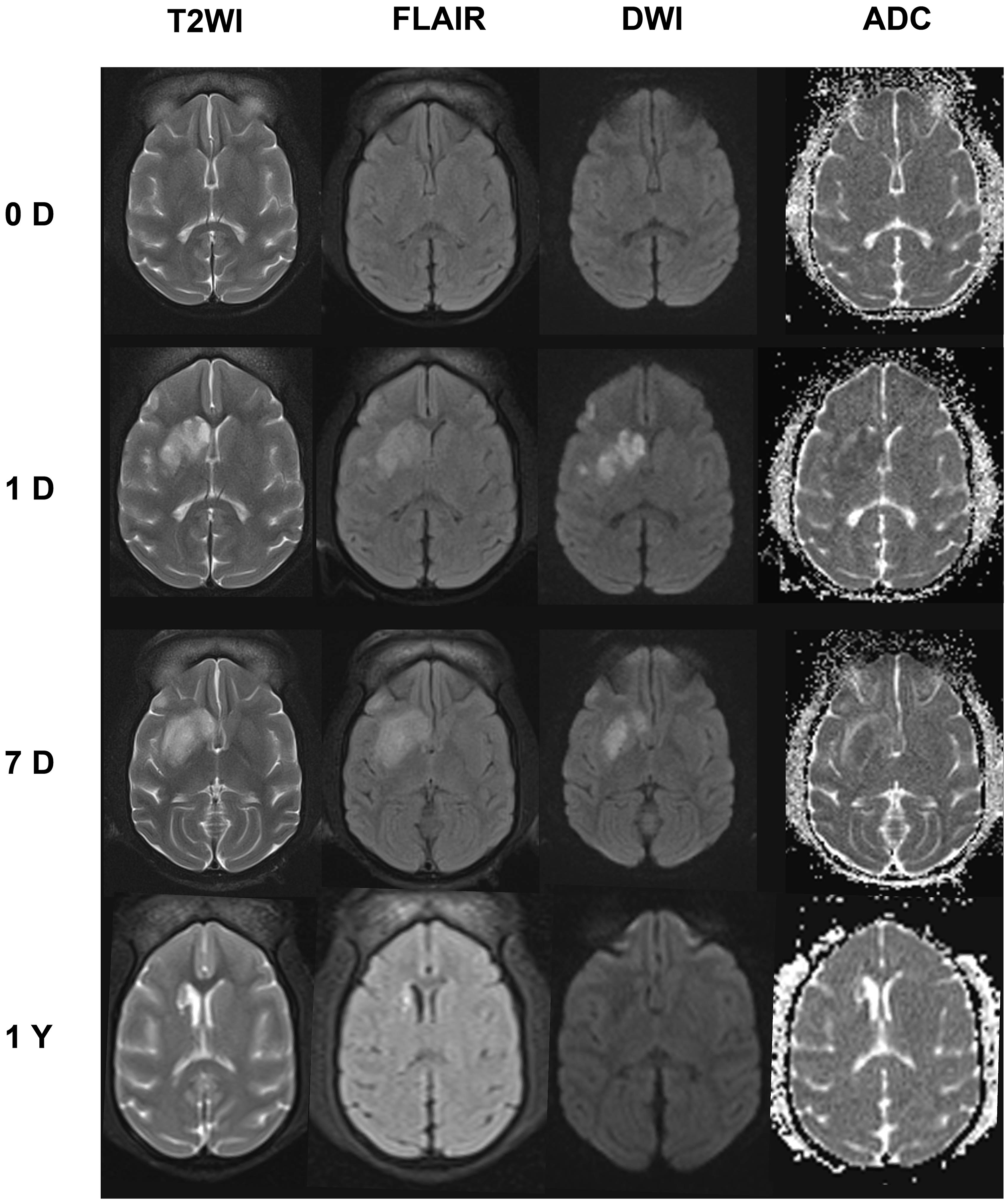
Representative MRI images from 16399 at 0 D (pre-MCAO), 1D (one day after tMCAO), 7D (seven days after tMCAO), and 1 Y (one year after tMCAO). The T2WI, FLAIR, DWI, and ADC maps showed the infarcted area in the right basal ganglia region.
Hypoperfusion occurs in different brain areas
Based on CTP, we measured three parameters (MTT, CBF, and CBV) associated with cerebral perfusion, as described in the methods section. During the acute phase, MTT increased after tMCAO, peaked at day one after tMCAO, and gradually decreased during the following 28 days (Supplementary Figure 1). Although MTT levels reduced over time, they did not return to baseline levels after one year in most monkeys (Figure 2(a) and (b)). MTT in the MCAO-1Y+ group (3.18 ± 0.16 s) was significantly higher than that in AMC group (3.03 ± 0.03 s) (P = 0.008, Figure 2(b)).
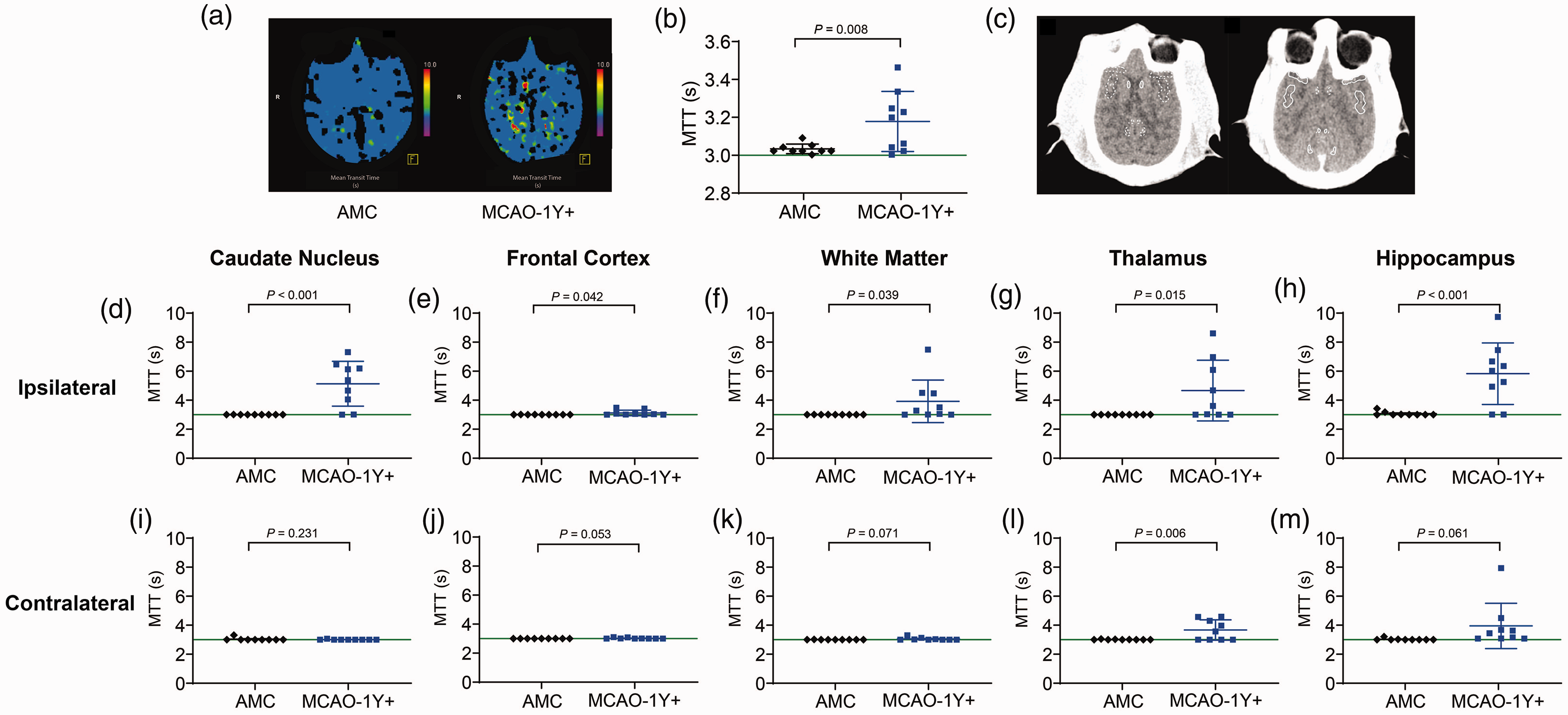
MTT decreased in the whole brain and different areas in the brain one year post tMCAO. (a) Representative MTT images in AMC and tMCAO groups. (b) MTT value of the whole brain. (c) Representative ROIs in non-contract CT maps. (d–m): MTT value in the ipsilateral and contralateral ROIs (d and i: caudate nucleus; e and j: frontal cortex; f and k: white matter; g and l: thalamus; h and m: hippocampus).
Since we observed that MTT did not show a homogenous high signal in the whole brain, we studied five pairs of regions of interest (ROIs) in both hemispheres, as shown in Figure 2(c), which included the caudate nucleus (1), frontal cortex (2), white matter (3), thalamus (4), and hippocampus (5), both ipsilaterally and contralaterally. The ROIs of the frontal cortex, white matter, thalamus, and hippocampus were generated from the suprasellar cistern section (Figure 2(c) left), and the caudate nucleus was circled in the body of the lateral ventricle section (Figure 2(c) right). Compared with AMC, MTT values were significantly higher in the ipsilateral caudate nucleus (P < 0.001, Figure 2(d)), frontal cortex (P = 0.042, Figure 2(e)), white matter (P = 0.039, Figure 2(f), thalamus (P = 0.015, Figure 2(g)), and hippocampus (P < 0.001, Figure 2(h)). On the contralateral side, no difference was observed in MTT in the caudate nucleus (P = 0.231, Figure 2(i)), which was significantly higher in the thalamus (P = 0.006, Figure 2(l)), and showed a similar trend in the frontal cortex (P = 0.053, Figure 2(j)), white matter (P = 0.071, Figure 2(k)), and hippocampus (P = 0.061, Figure 2(m)).
Regarding CBF, a parameter that indicates more severe hypoperfusion than MTT, we found a downward trend when the whole brain was included in the calculation (Figure 3(a), P = 0.067). When we analyzed the five pairs of ROIs separately, we found similar downward trends in the ipsilateral frontal cortex (Figure 3(c), P = 0.093), white matter (Figure 3(d), P = 0.082), and the contralateral frontal cortex (Figure 3(h), P = 0.085). No significant changes of CBF were found in the bilateral caudate nucleus (Figure 3(b) and (g)), contralateral white matter (Figure 3(l)), bilateral thalamus (Figure 3(e) and (j)), and ipsilateral hippocampus (Figure 3(f)). A large increase was found in the contralateral hippocampus (P = 0.028, Figure 3(k)).
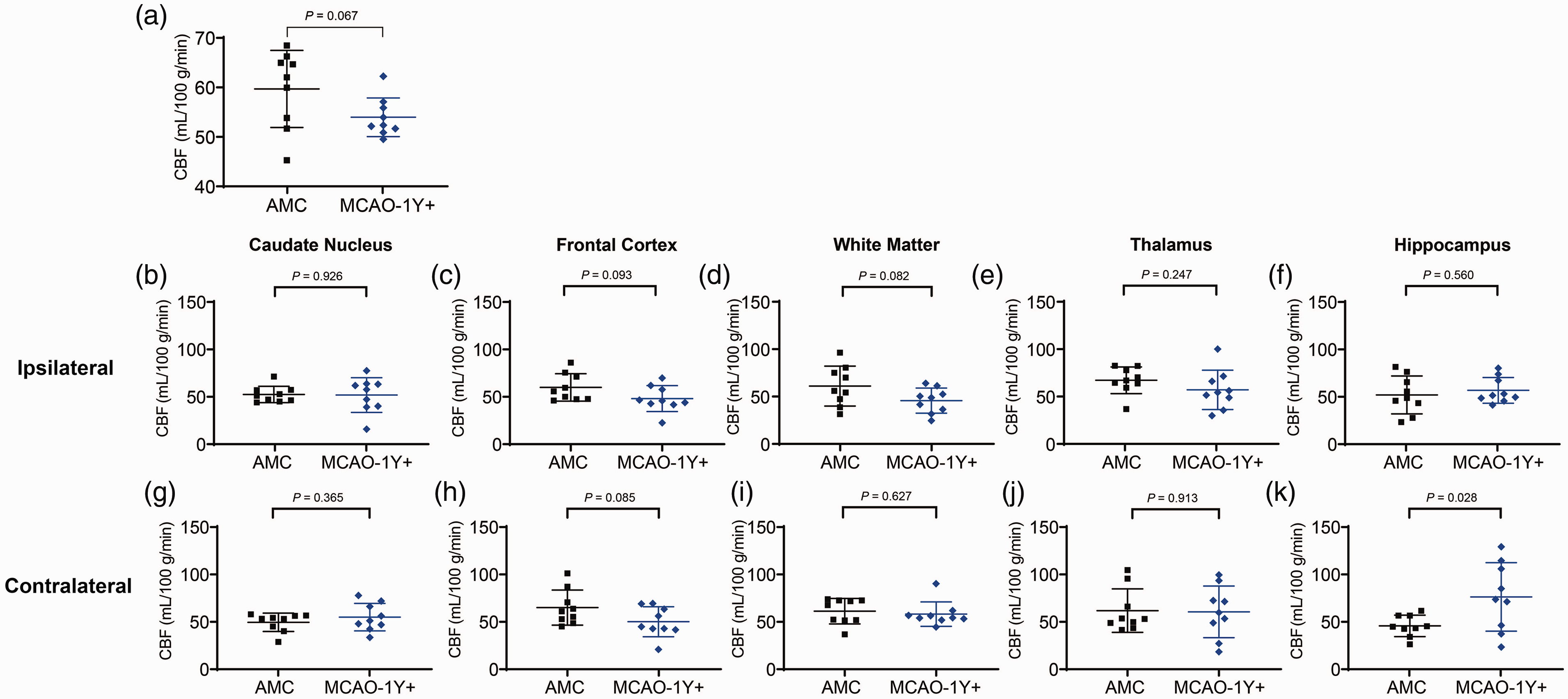
CBF was unchanged in most ROIs. (a) CBF value in the whole brain. (b–k) CBF value in the ipsilateral and contralateral brain of five ROIs (b and g: caudate nucleus; c and h: frontal cortex; d and i: white matter; e and j: thalamus; f and k: hippocampus).
BBB permeability differs in different brain areas
We measured the albumin ratio of the monkeys in the MCAO-1Y+ group and compared it with baseline levels obtained before tMCAO, but no differences were found (P = 0.489, Figure 4(a)). However, the more sensitive parameter BBB-PS showed that BBB permeability in the MCAO-1Y+ monkeys were significantly higher than that in AMC (1.27 ± 0.65 mL/100 mL/min vs. 0.53 ± 0.32 mL/100 mL/min, P = 0.007, Figure 4(b) and (c)). In the ipsilateral caudate nucleus, when the infarction was induced more than one year prior, the BBB-PS was significantly higher than that in AMC (P = 0.006, Figure 4(d)). The contralateral caudate nucleus showed changes similar to the ipsilateral caudate nucleus (P = 0.008, Figure 4(l)). BBB-PS values were higher in contralateral frontal cortex (P = 0.028, Figure 4(j)), but not in the ipsilateral frontal cortex (P = 0.234, Figure 4(e)). A higher BBB-PS also appeared in the white matter, thalamus, and hippocampus ipsilaterally and contralaterally (Figure 4(f), (k), (g), (l), (h) and (m)). The BBB-PS increase was the most pronounced in the ipsilateral hippocampus (0.67 ± 0.54 vs. 5.32 ± 3.27 mL/100 mL/min, P < 0.001, Figure 4(h)) and contralateral hippocampus (0.17 ± 0.19 vs. 4.39 ± 2.85 mL/100 mL/min, P < 0.001, Figure 4(m)).
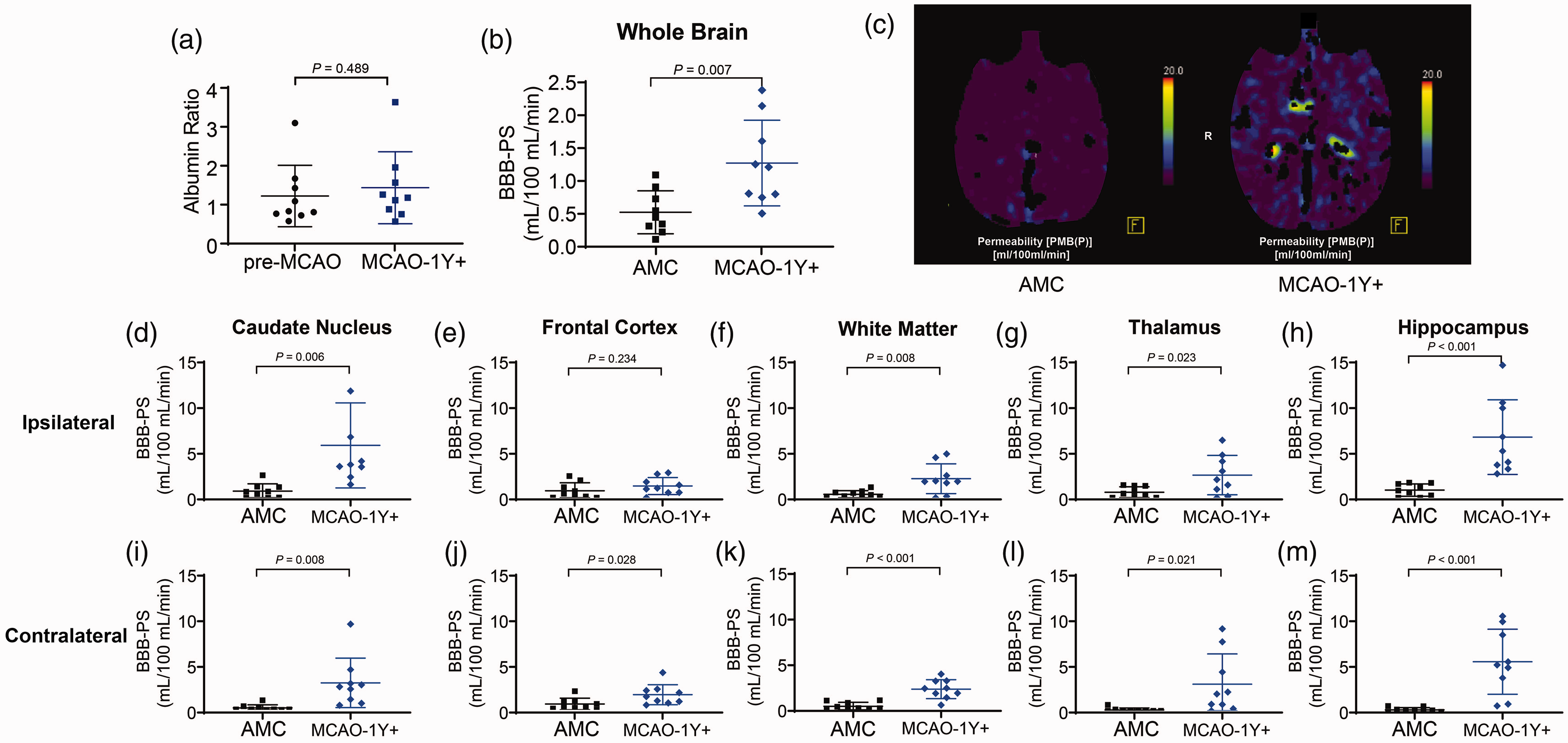
BBB-PS in MCAO-1Y+ increased significantly in most ROIs compared with AMC. (a) Albumin ratio between the pre-MCAO and MCAO-1Y+ groups. (b) BBB-PS value of the whole brain. (c) Representative images of BBB-PS in AMC and tMCAO groups. (d–m) BBB-PS value in ipsilateral and contralateral ROIs (d and i: caudate nucleus; e and j: frontal cortex; f and k: white matter; g and l: thalamus; h and m: hippocampus).
Chronic cerebral hypoperfusion and BBB disruption reduce Aβ42 concentration in CSF
Although none of the MCAO-1Y+ monkeys showed pronounced cognitive dysfunction based on the routine neurological and behavioral assessments conducted (data not shown), we speculated that the long-term hypoperfusion and BBB disruption might lead to changes in Aβ metabolism and future Aβ-related mild cognitive impairment (MCI). Therefore, we assayed four well-accepted cognitive biomarkers: Aβ42, Aβ40, tTau, and pTau181 in the CSF samples collected before tMCAO and after CTP assessment. We found that Aβ42 in CSF decreased from 209.2 ± 157.6 pg/mL to 105.9 ± 71.06 pg/mL one year after tMCAO (P = 0.006, Figure 5(a)); Aβ40 in CSF reduced from 1404 ± 699.5 pg/mL before tMCAO to 761.3 ± 516.5 pg/mL (P = 0.042, Figure 5(b)); Aβ42/Aβ40 ratio in CSF declined significantly one year post-MCAO (P = 0.019, Figure 5(c)), while no significant changes were found in CSF tTau and pTau181 levels (Figure 5(d) and (e)).
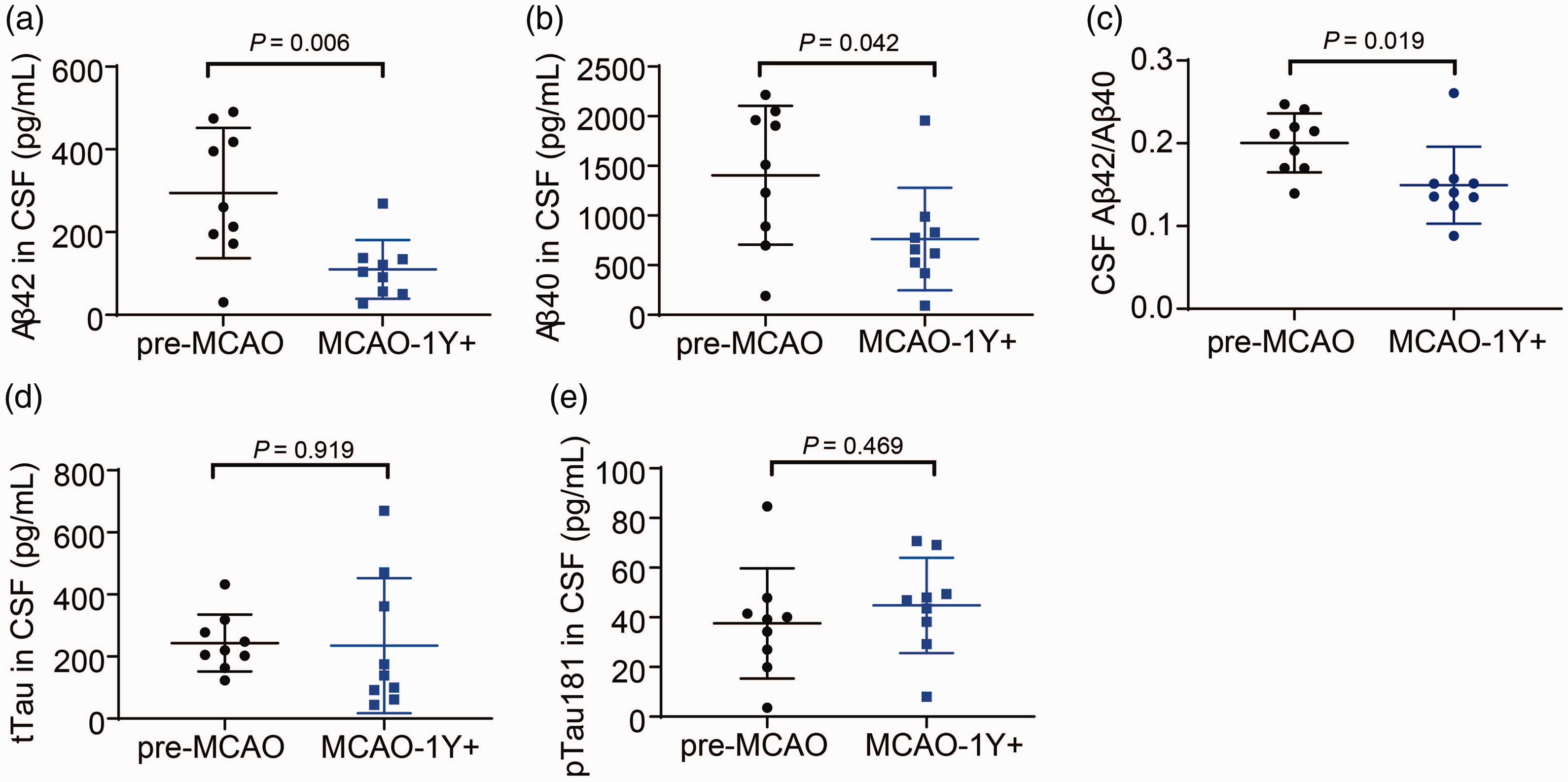
Changes in cognitive biomarkers of CSF before tMCAO (pre-MCAO) and one year after MCAO (MCAO-1Y+). (a–c) Concentrations of Aβ42 (a), Aβ40 (b) and Aβ42/Aβ40 ratio (c) in CSF; (d, e) Changes in tTau (d) and pTau181 (e) in CSF.
Discussion
In this study, we found that experimental tMCAO in rhesus monkeys led to hypoperfusion and higher BBB permeability one year later or more after the transient ischemic attack, even though the monkeys underwent a relatively satisfactory level of rehabilitation. The anatomic aspect of the injury almost disappeared, and normal neurobehavioral assessments were achieved. Despite all the satisfactory levels of recovery reported, each animal included in this study showed non-negligible levels of cerebral hypoperfusion and BBB disruption, as shown through CTP scanning.
Using NHPs for stroke modeling provides distinct advantages compared with rodents. First, NHPs are physically more similar to humans, especially in the central nervous system (CNS) anatomy, such as the gyrenecephalic brain and the gray-to-white-matter ratio. 33 Second, the larger vessel size facilitates the application of minimally invasive endovascular surgeries, providing the opportunity for tMCAO models similar to human patients and providing a higher translational value. For example, we demonstrated that the NHP tMCAO model developed in our lab provided an infarction size of about 4% in the whole brain, 17 which is very similar to clinical cases. 34 Third, the Stroke Therapy Academic Industry Roundtable (STAIR) committee recommended “to demonstrate efficacy in at least two species”, which should be “rodent then gyrencephalic species”, for pre-clinical studies in their 1999 guidelines 35 and updates in 2019 and 2021. 36 The advantages mentioned above are expected to improve the notorious low translational efficiency of stroke therapy development. 37 Fourth, and perhaps most importantly, NHP allows for the serial sampling of CSF due to its larger size and increased CSF availability, 38 making longitudinal analysis of brain metabolism possible and increasing the statistical power of the data obtained.
Although it has been well established that angiopathological changes occur during the acute and subacute stages of ischemic stroke, changes to brain blood vessels during the chronic stage (>1 year) after cerebral ischemia remains uncertain. Very few studies on human patients have reported cerebral vascular-related abnormalities for more than one year post-stroke. In rodents’ models, hypoperfusion and BBB disruption have generally been observed weeks or months after stroke.23,24,39,40 Few studies have attempted to observe stroke rehabilitation affected rodents for a longer term, partially due to animal welfare concerns. Notably, the infarction size of rodent acute tMCAO is usually 30-50% of the whole brain. On the contrary, the reduction in the percentage of animals that have had to be sacrificed and being sent-back-to-the-colony-after-the-study are two priorities that are almost mandatory in NHP studies. Therefore, we can use these advantages to observe the long-term pathological changes in post-MCAO monkeys used for pre-clinical efficacy studies (Supplementary Table 1).
An ischemic attack is usually preceded by acute hypoperfusion. TTP first indicates a slight reduction in blood flow under CTP. Once blood flow is further reduced, the MTT increases. However, the brain can still retain its cerebral circulation reserve (CCR) through the autoregulation.8,41 If hypoperfusion reaches a point of no return, the brain enters a stage called “misery perfusion” and the CCR can no longer remain balanced, leading to the damage of adjacent neurons and other parts of the NVU. All these changes are indicated by the change in CBF, and finally the CBV, by conducting CTP scanning. 42 In this study, compared with age-matched controls, overall MTT levels were significantly higher while CBF levels remained comparatively normal. It is noteworthy that CBF showed a “downward trend” in the ipsilateral frontal cortex and white matter, as well as the contralateral frontal cortex, suggesting poor cerebral perfusion, but the brain vasculature was still trying to maintain its normal physical tone. However, hypoperfusion did not occur homogeneously in all brain regions. The absurd increase in contralateral hippocampal CBF may be a compensated effect of false positive. However, we will still need to monitor CBF changes in the future to obtain a clearer result.
The BBB disruption manifested as a spatial-specific pattern. Although the increase in BBB permeability was significant in most ROIs, the absolute value of BBB-PS showed that the hippocampus and thalamus were more severely affected than the frontal cortex and white matter, both ipsilaterally contralaterally. However, it is not uncommon to observe ischemic stroke that leads to later damage in areas remote to the original occlusion site, which is known as “diaschisis”.43,44
The thalamus is located next to the subcortical MCA region, with the posterior cerebral artery providing its primary blood supply. The ipsilateral thalamus diaschisis (ITP) is commonly observed in acute MCA stroke, particularly in cases that involve the basal ganglia. Correlations between ITP and stroke outcomes are controversial. Reilder showed that ITP did not have an independent impact on patient outcome, 44 while Craig showed that contralesional thalamic volume is inversely correlated with clinical motor function. 45
The hippocampus plays pivotal roles in learning, short-term memory, and other cognitive abilities. 46 Vascular abnormalities in the hippocampal area are associated with MCI or AD,47,48 and the richness of hippocampal vascular supply was correlated with better cognitive function. 47
Even though vascular dementia is the most usual form of dementia post-stroke, it is still interesting to consider the possible causal relationship between vascular impairment and Aβ metabolism. A substantial amount of evidence shows the presence of impaired cerebral autoregulation and NVC in rodent models of AD,49,50 although studies of patients with AD or MCI have so far shown no or only mild such impairment. 5 As we age, cerebral blood vessels undergo structural and functional changes which disrupt the BBB and CBF. These normal aging processes are accelerated and occur in MCI and AD patients. 51 Similar to the monkeys of the current study, dysregulated and decreased CBF is visible in various brain areas, particularly in the cortex, hippocampus, thalamus, and parahippocampal gyrus. 49 In some patients, microbleeds have been observed in their brains. 49 We tried to look for a similar vascular pathological pattern in our monkeys using SWI but did not find any. However, it will be no surprise to see microbleeds emerge gradually in the future, along with more severe impairment of cerebral vasculature. CBF decreases and BBB disruptions have also been reported in animal models. CBF deficit has been observed in mice with mutant human APP driven by the endogenous mouse APP promoter. 52 The mice carrying the human APOE4 allele showed decreased CBF levels in the cortex, hippocampus, thalamus, and white matter,53–55 and this change was found to be associated with BBB breakdown. 55
To further investigate the link between post-MCAO vascular abnormalities and the potential of developing dementia later, especially its AD form, we measured CSF levels in Aβ40, Aβ42, tTau, and pTau181. The Aβ42 and Aβ42/Aβ40 ratio both showed a significant decrease compared with levels before tMCAO. Although there still is a lack of evidence for a causal relationship, the current study supports the hypothesis that chronic CBF dysregulation and BBB impairment may change Aβ metabolism and result in a cognitive decline apart from typical vascular dementia.
Observations on these nine monkeys are ongoing to determine whether these vascular changes are permanent or reversible. Further investigations, including but not limited to neurobehavioral studies to examine cognitive decline, PET-CT imaging, and CSF biochemical studies to examine Aβ metabolism, and myelin damage determination to show the effect of hypoperfusion, 56 will be carefully designed and carried out. We believe that longitudinal studies on these post-MCAO monkeys will provide us with an abundance of valuable information to help us further understand the vascular regulating mechanism after stroke and its relationship with vascular dementia, AD, and other types of dementia.
Conclusion
In conclusion, we used CTP imaging to determine that cerebral hypoperfusion and BBB disruption were sustained for more than one year after tMCAO in rhesus monkeys despite the good physical recovery. These cerebrovascular abnormalities mainly manifest in remote areas, such as the thalamus and hippocampus, accompanied by decreased levels of Aβ42 and the Aβ42/Aβ40 ratio in CSF.
Supplemental Material
sj-jpg-1-jcb-10.1177_0271678X221078065 - Supplemental material for Chronic cerebral hypoperfusion and blood-brain barrier disruption in uninjured brain areas of rhesus monkeys subjected to transient ischemic stroke
Supplemental material, sj-jpg-1-jcb-10.1177_0271678X221078065 for Chronic cerebral hypoperfusion and blood-brain barrier disruption in uninjured brain areas of rhesus monkeys subjected to transient ischemic stroke by Yingqian Zhang, Bangcheng Zhao, Qi Lai, Qinxi Li, Xun Tang, Yinbing Zhang, Zhixiang Pan, Qiang Gao and Zhihui Zhong in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X221078065 - Supplemental material for Chronic cerebral hypoperfusion and blood-brain barrier disruption in uninjured brain areas of rhesus monkeys subjected to transient ischemic stroke
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X221078065 for Chronic cerebral hypoperfusion and blood-brain barrier disruption in uninjured brain areas of rhesus monkeys subjected to transient ischemic stroke by Yingqian Zhang, Bangcheng Zhao, Qi Lai, Qinxi Li, Xun Tang, Yinbing Zhang, Zhixiang Pan, Qiang Gao and Zhihui Zhong in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Scientific Foundation of China (81771310, 82071349), National Key Research and Development Program (2017YFA0505903), and West China Hospital of Sichuan University Discipline Excellence Development 1·3·5 Engineering Project (ZYYC08005 and ZYJC18041).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Yingqian Zhang helped design the research, coordinated all experiment operations, and helped to prepare the manuscript. Bangcheng Zhao was accountable for data post-processing. Qi Lai, Xun Tang, and Yinbing Zhang were responsible for tMCAO model establishment and animal welfare. Qinxi Li performed the statistical analysis. Zhixiang Pan collected data and analyzed images. Qiang Gao helped to design the CTP measurement. Zhihui Zhong designed and supervised all the studies and wrote the manuscript. All authors read and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
