Abstract
Focused ultrasound combined with circulating microbubbles (FUS+MB) can transiently enhance blood-brain barrier (BBB) permeability at targeted brain locations. Its great promise in improving drug delivery to the brain is reflected by a rapidly growing number of clinical trials using FUS+MB to treat various brain diseases. As the clinical applications of FUS+MB continue to expand, it is critical to have a better understanding of the molecular and cellular effects induced by FUS+MB to enhance the efficacy of current treatment and enable the discovery of new therapeutic strategies. Existing studies primarily focus on FUS+MB-induced effects on brain endothelial cells, the major cellular component of BBB. However, bioeffects induced by FUS+MB expand beyond the BBB to cells surrounding blood vessels, including astrocytes, microglia, and neurons. Together these cell types comprise the neurovascular unit (NVU). In this review, we examine cell-type-specific bioeffects of FUS+MB on different NVU components, including enhanced permeability in endothelial cells, activation of astrocytes and microglia, as well as increased intraneuron protein metabolism and neuronal activity. Finally, we discuss knowledge gaps that must be addressed to further advance clinical applications of FUS+MB.
Introduction
The combination of focused ultrasound and microbubbles (FUS+MB) is an emerging technology that has been used to deliver various therapeutic agents to the brain. 1 In 2001, FUS+MB treatment was first introduced by Hynynen et al. to enhance the permeability of the blood-brain barrier (BBB). 2 Over the past two decades, interest in FUS+MB has increased due to its unique capability to noninvasively, locally, and reversibly enhance BBB permeability for the delivery of therapeutic agents across the BBB. Preclinical studies have succeeded in delivering a wide range of therapeutic agents to the brain using FUS+MB, including small molecules, 3 proteins,4–6 viruses,7,8 and even cells.9,10 The success in preclinical studies, predominantly conducted using healthy animals, has led to several clinical trials in patients with glioblastoma,11,12 Alzheimer’s disease, 13 and amyotrophic lateral sclerosis. 14 As clinical applications of FUS+MB continue to expand, there is a pressing need to fully understand FUS+MB-induced biological effects and underlying mechanisms to improve the efficacy of current treatment as well as enable discoveries of new therapeutic strategies for brain diseases.
To date, studies of biological effects of FUS+MB treatment have primarily focused on changes in blood vessels and associated physiological effects. These biological effects include but are not limited to increased BBB permeability and altered cerebral blood flow, as covered by several excellent reviews.15–17 Although exact mechanisms of FUS+MB-induced BBB opening are still under investigation, it is considered that safe, transient BBB opening is attributed to the expansion and contraction of MBs in the ultrasound field, a phenomenon also known as MB cavitation.18–20 MB cavitation can generate mechanical stress on the brain endothelium, resulting molecular, cellular, and structural changes that are associated with BBB permeability enhancement.21–23
Can FUS+MB affect other brain cells surrounding blood vessels? The answer is yes. Increasing evidence indicates that the impact of FUS+MB reaches all cell types in the neurovascular unit (NVU), an essential structure composed of brain endothelial cells, pericytes, astrocytes, microglia, and neurons. This extensive impact results in systemic bioeffects, including the sterile inflammatory response, waste clearance, and even neurogenesis. 24 Despite the broad impact FUS+MB has on brain physiology, a complete understanding of biological effects on different NVU cell types is lacking. In addition to having differential effects on different cell types, FUS+MB-induced bioeffects are dependent on various factors, such as FUS pressure, time post sonication, target location in the brain, and healthy/diseased animal model.25–30 A better understanding of FUS +MB-induced effects on the NVU can help delineate underlying mechanisms of systemic physiological changes in the brain after FUS+MB treatment. This knowledge will be critical for improving the safety and effectiveness of FUS+MB-enhanced therapeutic applications.
Here, we first provide a brief introduction to the FUS+MB technique. We then briefly discuss the roles of each component in the NVU. For each NVU component, we summarize existing evidence on molecular-level changes induced by FUS+MB and discuss remaining knowledge gaps. We conclude with an outlook for studies needed to understand the full extent of biological effects induced by FUS+MB in the NVU and their underlying mechanisms.
FUS+MB technique
Although neurosurgical, pharmacologic, and physiologic strategies have been developed to circumvent the BBB for brain drug delivery, FUS+MB is the only established technique that can induce localized and reversible BBB opening. 15 Several excellent articles review the FUS+MB technique with the focus on its application in brain drug delivery.15,31,32 In this review, we provide an overview of critical parameters involved in this technique and discuss its limitations.
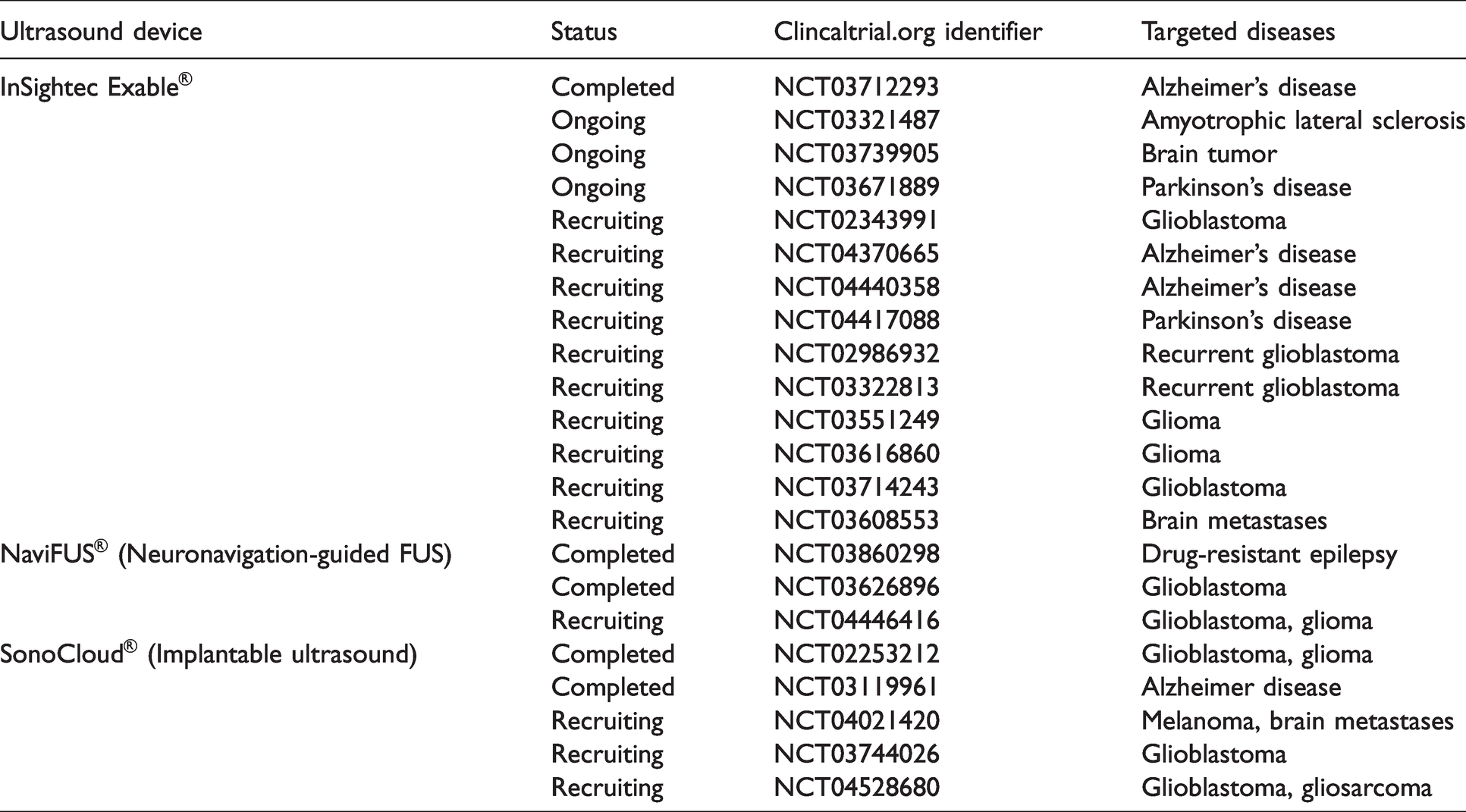
Technological advancement in past decades has led to the development of transcranial FUS devices that are capable of noninvasively delivering ultrasound energy inside the brain with a maximum depth at the scale of 10 centimeter, and a focal region of 1 × 1 × 10 mm3.33,34 MBs, which are gas-filled bubbles coated by protein or lipid shells (diameters range from 1 to 10 µm), were originally used as blood-pool ultrasound contrast agents in clinics. When encountering an ultrasound beam, MBs cavitate—a broad term for various ultrasound-induced bubble activities, including their volumetric oscillation and collapse. MB cavitation can generate microstreaming or jetting that induces shear stress on surrounding tissue. MB expansion and contraction can also induce vessel distention and invagitation. 18 MB cavitation is believed to be the main physical mechanism that causes bioeffects, which can last a few hours to weeks. Hundreds of preclinical studies have reported on optimizing FUS+MB experimental parameters, developing imaging tools to monitor and assess the treatment, improving FUS targeting accuracy, and demonstrating its short and long-term safety. Building on extensive and promising preclinical findings, the first-in-human study was conducted in 2018 to evaluate feasibility and safety of FUS+MB-induced BBB opening in patients with Alzheimer’s disease. 13 Since then, there have been 5 completed and 17 ongoing FUS+MB clinical trials in patients with different types of brain diseases (Table 1)11,13,35–38
Clinical trials using FUS+MB to treat brain diseases.
Preclinical studies have identified a large parameter space that impact FUS+MB results, including MB parameters (e.g., type, dosage, size, and injection method), FUS parameters (e.g., frequency, ultrasound pressure, burst parameters, sonication duration), and other experimental variables (e.g., targeted brain region and disease condition).
Three commercially available ultrasound contrast agents are currently used in FUS+MB studies: Definity, Optison, and SonoVue. Definity are perfluorocarbon gas-filled lipid shells (mean diameter = 1.1–3.3 µm; maximum concentration = 6 × 109 MBs/mL; recommended clinical dose = 10 µl/kg). Optison are perfluorocarbon gas-filled albumin shells (mean diameter = 3.0–4.5 µm; concentration = 5–8 × 108 MBs/mL; recommended clinical dose = 0.5 mL per adult). SonoVue are sulfur hexafluoride gas-filled lipid shells (mean diameter = 1.5–2.5 µm; concentration = 1–5 × 108 MBs/mL; recommended clinical dosage = 2–2.4 mL per adult). While these commercially available MBs have a large size variance (i.e., polydisperse), custom-made monodisperse MBs have also been used in FUS+MB preclinical studies to achieve more consistent BBB opening. 39 More recently, Nanobubbles with long circulation time (i.e., 10 min in rats) were shown to successfully induce BBB opening. 40
As for FUS parameters, a wide range has been used to induce BBB disruption, and there is a lack of standard and consent for optimal parameters. The most well-characterized FUS parameters are peak negative pressure (PNP) and mechanical index (MI, which is PNP divided by square root of central frequency). McDannold et al. found that the threshold for BBB disruption (probability of disruption was estimated to be 50%) was between a MI of 0.4 and 0.5 using a rabbit model. 41 In comparison, a lower MI threshold for BBB disruption was reported in primates as 0.32 (0.15 MPa, 0.22 MHz transducer). 41 The authors argued that the discrepancy may be due to different detection sensitivity of MRI versus histological analysis for BBB disruption and differences between small animal models and primates. 41 It is difficult to specify the clinical range of PNP as most reported clinical trials used 50-60% “cavitation threshold” power but did not specify PNP or MI.13,14,31,37 In a clinical trial of recurrent GBM, BBB disruption was observed by MRI using MI of 0.48–0.68 and PNP of 0.34–0.48 MPa. 42 Although most existing studies focus on characterizing the threshold and safety range of PNPs to induce BBB disruption, emerging evidence suggests PNPs lower than the threshold can still affect the NVU without disrupting the BBB or inducing an inflammation response. For example, FUS+MB treatment at 0.1 MPa achieved localized endothelial cell transfection on cerebral blood vessels by injecting MBs conjugated with plasmid. 43 As PNP increased from 0.1 MPa to 0.4 MPa, transfected cell types shifted from cells on blood vessels (endothelial cells, pericytes, astrocytes) to cells distal from the blood vessels (neurons, microglia). 44 These results show that FUS+MB affects cells in the NVU at different PNPs depending on the distance between the affected cell and blood vessels, suggesting that FUS parameters may be tuned to transfect specific brain cell types.
Other experimental parameters, for example, the targeted brain region, anesthesia, and disease condition of the experimental model, can also affect FUS+MB-induced bioeffects in the brain. FUS+MB can induce BBB disruption in a wide range of brain regions with varying vasculature and structural properties, such as hippocampus, 45 striatum, 46 and brainstem. 47 A recent study showed that FUS+MB induced a lower level of BBB opening in the caudate putamen compared to the thalamus in a rat brain, possibly due to the difference in ultrasound’s incidence angle at these targeted locations. 48 Another study reported that astrocytes and microglia in the cerebral cortex and hippocampus were affected differently by FUS+MB treatment in a healthy rat model.25,49 Furthermore, neurogenesis was observed in the sonicated frontal lobe of healthy rats but not the sonicated hippocampus after 6 weekly FUS+MB treatments; however, this local difference in neuronal response was reversed in rats that received one-time treatment. 25 Moreover, the anatomical difference between healthy and pathological brain tissue can result in significant differences in FUS+MB-induced effects. For example, one study using a mouse model of Alzheimer’s disease found that FUS+MB-induced BBB permeability enhancement was lessened in transgenic (TgCRND8) mice compared to non-transgenic mice, suggesting that amyloid plaque may weaken FUS+MB-induced effects on vessel permeability. 50 Changes in the brain tumor microenvironment such as blood vessel disruption, impaired blood flow, and increased endocytosis can also lead to a reduced effect from FUS+MB, which may explain the overall higher PNP used to enhance BBB permeability changes in brain tumors compared to healthy tissue.42,51–53
There are several limitations with the FUS+MB technique that should be mentioned. Due to a lack of complete understanding of biological effects induced by FUS+MB and their underlying mechanisms, FUS+MB treatments may cause unwanted biological effects. For example, multiple FUS+MB treatments have been shown to elevate hyperphosphorylated tau protein in neurons. 25 Secondly, the efficacy of the FUS+MB technique can be influenced by many technical variables; therefore, clinical studies should be aware of the degree of variability between patients. In a phase I safety trial for treating patients with Alzheimer’s disease, while all patients had successful BBB opening following the MRgFUS treatment, results were most uniform in 2 out 5 patients. 13 Thirdly, there is a lack of understanding of long-term bioeffects induced by FUS+MB, rendering safety evaluation of the technique incomplete. Finally, microhemorrhage and edema can happen when relatively higher PNPs are used, however, safety should be evaluated for each specific FUS setup and animal model to determine the threshold for PNP. 54
NVU components
The concept of the NVU initially emerged to describe the unique interrelation between cerebral blood vasculature and brain cells.55–57 The most studied role of the NVU is neurovascular coupling, which links neuronal activity to blood flow and became the basis for functional MRI (fMRI). 57 Additionally, the NVU regulates BBB permeability, the glymphatic system, and neurovascularization. Dysfunction of the NVU contributes to numerous brain pathologies, including neurodegenerative diseases, brain tumors, cerebral ischemia, and inflammation.56–58 Given that FUS-activated MBs must first interact with the NVU for any effect in the NVU to happen, it is important to understand its biological function when investigating effects of FUS+MB (Figure 1). In this section, we briefly review the biological role of each NVU component and how brain diseases affect their function.
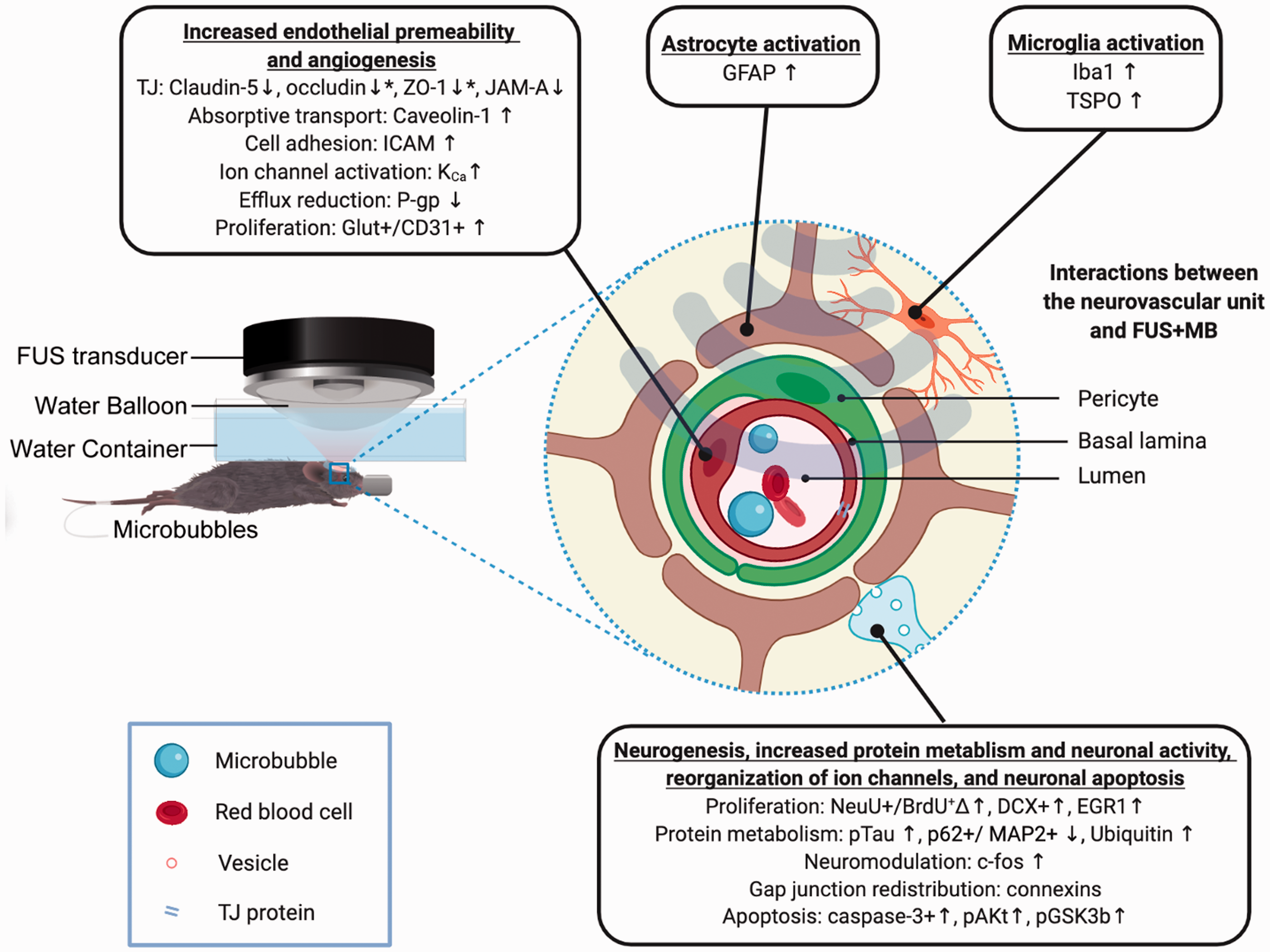
Bioeffects of FUS+MB on neurovascular unit (NVU) components. An illustration of the experimental setup for applying the FUS+MB technique on small animals is provided on the left. Molecular changes and functional effects induced by FUS+MB for each NVU component are listed in boxes. *Contradictory results were reported; ΔSpatial heterogeneity of the effect on this protein was observed; Molecular markers in red have been studied in diseased animal models. Details regarding focused ultrasound (FUS) parameters, methods of measurement, brain regions where molecular changes were observed, the time post sonication when molecular changes were observed, and how long these changes lasted please refer to Chapter 5 to 8 and Tables 2 to 5.
Endothelial cells
The endothelial cells lining cerebral blood vessels regulate the trafficking of molecules and cells between blood and brain by forming highly specialized paracellular and transcellular barriers. 59 Altered expression and dysfunction of elements of these barriers can result in BBB permeability changes.
The paracellular barrier of the BBB is formed by tight junctions and adherent junctions between cerebral endothelial cells, limiting the diffusion of ions, macromolecules, and other solutes across the BBB. 60 The tight junction structure is composed of transmembrane tight junction proteins (e.g., claudins, occludins, and junctional adhesion molecules [JAMs]) and their cytoplasmic adaptors (e.g., zonula occludens-1 [ZO-1] and ZO-2), which are linked to the cytoskeletal protein actin. 61 The adherent junction structure is established through transmembrane proteins such as vascular endothelial cadherin (VE-cadherin) and platelet-endothelial cell adhesion molecule (PECAM), which are coupled to the cytoskeleton via catenins. 62 Reduced expression and altered morphology of these structural barriers have been observed along with enhanced BBB permeability and BBB dysfunction under several pathological conditions, including brain tumors, Alzheimer’s disease, and stroke.63–65
The transcellular barrier is formed by highly-regulated transcytosis and efflux transporters. During transcytosis, molecules are internalized by caveolin- or clathrin-coated vesicles and shuttled across the endothelial cell from its luminal to abluminal surface. Once on the abluminal side, vesicular contents are exocytosed into the brain parenchyma. 66 Vesicles in brain endothelial cells are more than 10-fold fewer than those in blood vessels of other tissues, resulting in low rates of transcytosis. 67 Changes in expression and dysfunction of caveolins in brain endothelial cells can alter the permeability of macromolecules, regulate intracellular signal transduction, and have been linked to several brain pathologies, including inflammation, Alzheimer’s disease, ischemic stroke, and brain cancer.68–71 Additionally, polarized expression of influx and efflux transporters, ion channels, and ion transporters are involved in transcellular transport across the BBB. These transporters ensure that nutrients, such as glucose and amino acids, are delivered to the brain and that solutes and metabolite waste products are effluxed from the brain to the blood. 72 For example, the multidrug resistance efflux transporter P-glycoprotein (P-gp), a type of ATP-binding cassette (ABC) transporter, is mainly present on the luminal membrane surface, preventing the passage of drugs and toxins across the BBB into the brain from the blood. 73 Increased expression of P-gp in the brain has been observed following ischemic stroke, which could significantly reduce the BBB permeability, posing another challenge for drug delivery to the brain. 74 A negative correlation between P-gp expression and the presence of amyloid deposition was observed in brain samples of Alzheimer’s disease. 75 Together, this evidence suggests that transcytosis and P-gp play an important role in mediating BBB permeability in both health and brain diseases.
Pericytes
Pericytes encircle cerebral endothelial cells and the vascular lumen. In this anatomical position, they contribute to the barrier function and communicate directly with endothelial cells through specialized junctions. 76 Pericyte:endothelial cell interactions regulate microvascular permeability, remodeling, and angiogenesis. 62 Pathological conditions like hypoxia (e.g., in tumor environment) or brain trauma can cause detachment of pericytes from endothelial cells and, consequently, impaired BBB barrier function along with other endothelial abnormalities. 62
Astrocytes
Astrocytes provide a cellular link between the neuronal circuitry and blood vessels; they are involved in maintaining vascular integrity, neuronal metabolism, and perivascular clearance mechanisms. 56 Their extended endfoot processes provide ∼99% abluminal vessel coverage. 56 Astrocytic endfeet express high levels of aquaporin-4 (AQP4), a type of water channel proteins. Displaced astrocytic endfeet from brain endothelial cells can be caused by infiltrating glioma cells, resulting in a localized breach in the BBB. 77 On the other hand, injury and stress in the CNS cause astrocytes to become activated and secrete a variety of protective factors, including angiopoietin-1, sonic hedgehog, glial-derived neurotrophic factor, retinoic acid, apolipoprotein E, and insulin-like growth factor-1; these factors attenuate BBB permeability and promote BBB function after brain injury.78,79 Aquaporin 4 (AQP4), a type of water channel proteins, are highly expressed by astrocytic endfeet; this protein is involved in the perivascular clearance mechanisms via the newly discovered glymphatic system. 80
Microglia
Microglia cells are resident macrophages of the brain and contribute to brain function by modulating innate immune activity within the perivascular region. 62 Under pathologic conditions, microglia become activated, highly mobile, phagocytic, and can secret numerous soluble factors, including pro- and anti-inflammatory cytokines, growth factors, chemokines, and neurotrophins.56,81,82 A recent study showed that activated microglia could have opposing effects on BBB permeability: Their initial response to pathological conditions can help maintain BBB integrity via the expression of tight junction protein claudin-5 and physical contact with endothelial cells; however, chronic inflammation can result in microglia phagocytosis of astrocyte endfeet, impairing BBB function. 84
Neurons
The physical contact between neuronal processes with the cerebral vasculature is critical for mediating cerebral blood flow in response to the neuronal metabolic demand. 56 Recent studies have shown that neuronal signals contribute to the stabilization of the structure of the vascular network and the regulation of efflux transporters.85,86 The integrity of the neuronal network depends on gap junction-mediated intercellular communication. Gap junctions connect the cytoplasms of adjoining cells through extracellular interactions between plaques of connexin hemichannels (i.e., connexons) formed in each cell. 87 Thus, gap junctions allow for direct passage of cytoplasmic inorganic ions and other small water-soluble molecules between neighboring neurons.
FUS+MB-induced BBB opening
BBB permeability increases immediately after FUS+MB, then gradually returns to normal within a few hours or days, depending on the experimental condition. 24 It is difficult to untangle cellular effects on the rest of the NVU and to verify whether these effects are caused by FUS+MB-induced BBB opening, mechanical forces generated from MB cavitation, or both. What occurs following BBB disruption during brain insults such as trauma and stroke may offer some insights into how NVU elements are affected by FUS+MB-induced BBB opening. BBB disruption following brain insults can result in downregulated tight junction proteins, increased transcytosis of plasma proteins (e.g., albumin), and vasogenic edema.28,88,89 Extravasated albumin following BBB disruption can activate astrocytes and microglia, inducing an inflammation response.88,90 Kovacs et al. found that MB cavitation induced increased transient expression of cytokines, chemokines, and trophic factors (CCTFs) associated with NFκB pathways as well as cell adhesion molecules, which is comparable to a sterile inflammation response following ischemia or trauma.24,28 In particular, FUS+MB induced protein expression for pro-inflammatory cytokines TNFα, IL-1α, IL-1β, IL18, and IFNγ as well as heat-shock protein 70 (HSP70) within 5 minutes of sonication, suggesting the close involvement of NVU components in response to the treatment. 28 Increased CCTFs associated with NFκB pathways occurred as early as 0.5 hours and lasted 24 hours post sonication. 28 Transient vasospasm and hypoxia following FUS+MB treatment can also lead to production and release of TNFα and other proinflammatory factors. Vasospasm in mouse arteries and arterioles was observed immediately after sonication and recovered within a few minutes.24,91 Using functional MRI and an arterial spin labeling (ASL) MRI, a more recent study found that FUS+MB significantly reduced the blood oxygen level dependent (BOLD) and cerebral blood flow signal changes in response to hind paw stimulation, which returned to baseline after 24 hours. 92 In the following chapters, we review current findings on molecular changes induced by FUS+MB for each NVU component separately to gain more cell-type-specific insights into biological effects of FUS+MB.
Effects of FUS+MB on endothelial cells
When activated by FUS, MB cavitation can induce mechanical stress on endothelial cells, resulting in the temporary and local increase of BBB permeability.18, 92 Studies in vivo have found that FUS+MB causes the reconfiguration of paracellular junctions, increased transcytosis, and reduced efflux transporter activity, and the opening of endothelial cell ion channels.21,25,29, 93 A long-term effect of endothelial cell proliferation has also been reported following FUS+MB, suggesting that FUS+MB may induce lasting effects on the NVU. In this section, we review literatures on molecular effects of FUS+MB on brain endothelial cells and discuss functional changes these effects can lead to. Table 2 provides a summary of molecular changes in brain endothelial cells and experimental conditions used in each study.
Effects of FUS+MB on endothelial cells in the NVU.
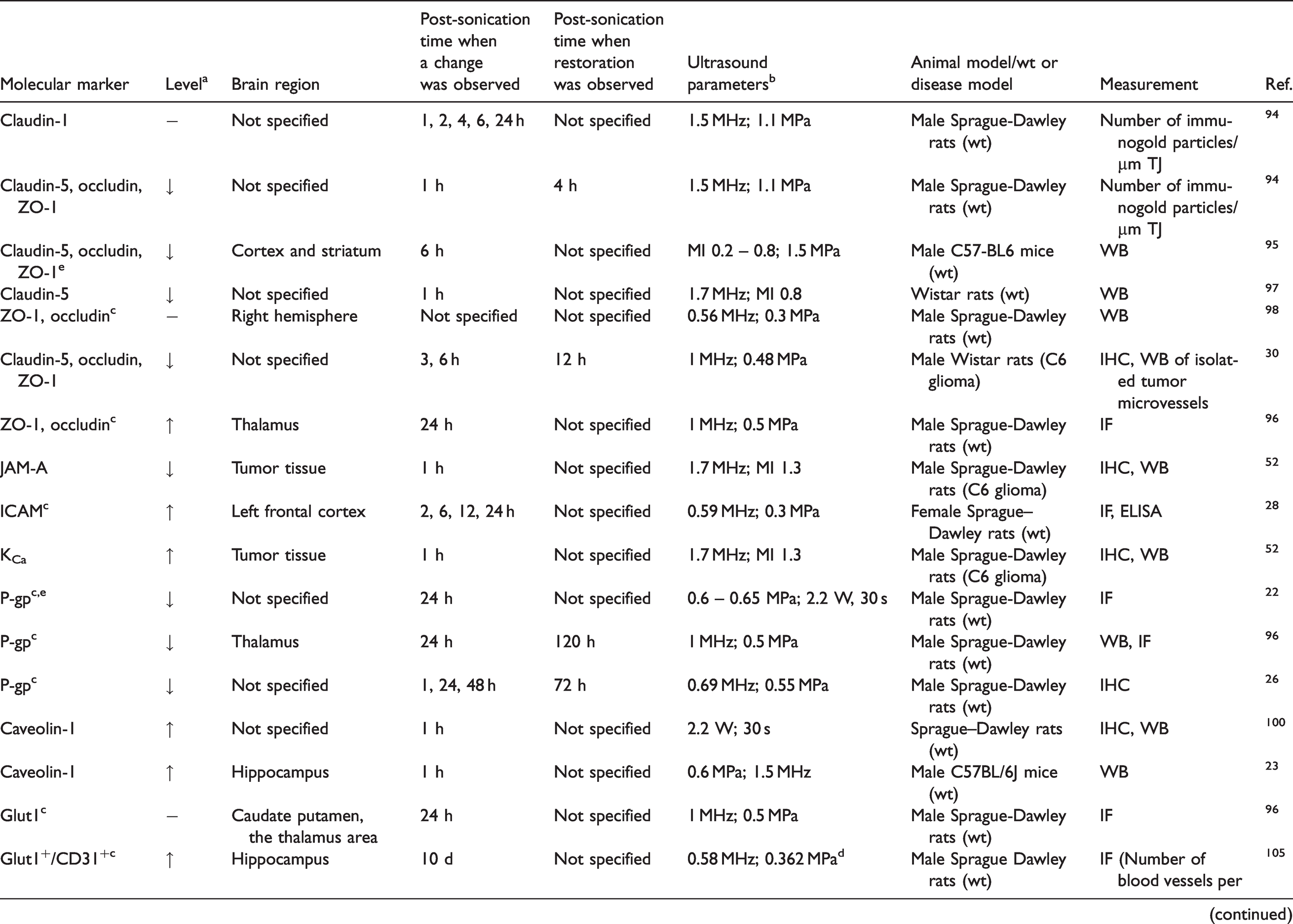
aChanges in protein levels include increase (↑), decrease (↓), or no change (−).
bUltrasound parameters include center frequency of transducer in MHz, peak negative pressure (PNP) of the transducer in MPa, and mechanical index (MI) = PNP//√ (center frequency of the transducer).
cLarge volume treatment (multiple treatments in one region).
dCavitation feedback-controlled the FUS.
eVessel damage or hemorrhage was observed.
TJ: tight junction; WB: western blot; IHC: immunohistochemistry; IF: immunofluorescence; ELISA: enzyme-linked immunoassay; DG: dentate gyrus; CA1: cornu ammonis 1; CA3: cornu ammonis 3.
Paracellular junction breakdown and re-distribution
The first direct evidence of FUS+MB-induced alterations in tight junction proteins was demonstrated by electron microscopy and immunogold labeling of these proteins. 94 This study found decreased expression of occludin, claudin-5, and ZO-1 following sonication, while claudin-1 expression was less affected. 94 These effects, observed as early as 1 hour after sonication, were accompanied by the extravasation of normally-excluded compounds (e.g. Evans Blue) into the brain parenchyma. 94 Tight junction protein expression was restored within 4 hours post sonication. 94 Another study using a commercialized, diagnostic ultrasound system combined with MBs reported immediate BBB opening following sonication. While the BBB permeability measured by Evans Blue extravasation was restored within 6 hours, reduction in occludin, claudin-5, and ZO-1 protein levels persisted 6 hours following sonication. 95 Interestingly, one study found that the fluorescent intensity of immunolabeled ZO-1 and occludin increased rather than decreased in sonicated brain regions at 24 hours post-sonication. 96 It remains to be determined whether this is due to the recovery of tight junction proteins as protein levels return and eventually surpass the baseline level. Another study compared continuous and intermittent launches of FUS and found that claudin-5 protein levels were more reduced as compared to non-FUS controls, by intermittent, rather than continuous sonication. 97 These data suggest that FUS+MB-induced tight junction downregulation may be due to structural changes, recycling, or relocalization of tight junction proteins.
In contrast, several other studies showed that tight junction protein reduction does not always accompany FUS+MB-induced BBB disruption. One study showed that while occludin and ZO-1 protein levels did not decrease, the physical interaction between these two proteins was reduced in sonicated brain regions, as demonstrated through co-precipitation. 98 The timeframe for these measurements post sonication was not specified in this study.
To date, only two preclinical studies of FUS+MB-induced effects on tight junction proteins in diseased animal models have been reported. In the first study, a significant reduction in mRNA and protein levels of junctional adhesion molecule-A (JAM-A) was observed in a rat model of glioma in which diagnostic ultrasound was used in combination with MBs. 52 The second study, also in a rat glioma model, showed similar reduction in mRNA and protein expression of claudin-5, occludin, and ZO-1 within 3 hours after FUS+MB. 30 These transcriptional and proteomic changes in tight junction proteins lasted for 9 hours and were nearly restored within 12 hours following sonication. 30 Given the complexity of the tumor environment and heterogeneity among different types of tumors, it is clear that more preclinical studies are needed to better understand both the short-term and long-term effects of FUS+MB on tumor endothelial cells and their junctional proteins.
Despite the important function of adherent junctions in regulating BBB, little is known about how they are affected by FUS+MB. Intercellular adhesion molecule 1 (ICAM-1) is expressed on brain endothelial cells where it is critical for the firm arrest and transmigration of leukocytes from blood to brain tissues. ICAM-1 expression can be increased by pro-inflammatory cytokines. 99 Only one study so far has reported that ICAM-1 protein levels increased within 2 hours, and the elevation persisted through 24 hours after FUS+MB treatment. 28 A better understanding of how FUS+MB affects ICAM-1 expression and immune cell migration might identify novel targets for brain tumor immunotherapy.
Transcellular transport enhancement
FUS+MB has been shown to increase the number of caveolae in the cytoplasm of brain endothelial cells in healthy rodent models as well as a stroke/ischemic murine model.21,94,100,101 Several studies using molecular-level characterization showed that FUS+MB upregulates the expression of caveolin-1, an essential mediator of caveolae-mediated endocytosis.23,100 Particles of different sizes may employ different transcytosis mechanisms following FUS+MB treatment. A recent study showed that FUS+MB delivered significantly less 500 kDa dextrans in caveolin knockout mice when compared to the wide type mice, while no significant difference was observed for 3 kDa and 70 kDa dextrans, suggesting that caveolin-1 plays an important role in FUS+MB-mediated transcellular transport for large particles. 23 Future studies are needed to investigate underlying mechanisms by which FUS+MB affects transcellular transport. In addition, FUS parameters and drug characteristics for optimized FUS-enhanced transcellular transport remain undefined.
Efflux transporter reduction
FUS+MB changes efflux transporter expression in the brain. Several studies have reported that the P-gp protein levels in endothelial cells were suppressed for up to 24 hours after sonication but restored within 72 hours when moderate FUS PNP was used (∼0.55 MPa), temporarily preventing drug efflux back into the blood circulation.22,26,29,96,102 In addition, downregulation of mRNA expression level of ABC and SLC transporters Abcb1a, Abca9, Slc22a6, Slc22a8, and Slc6a13 was observed 6 hours following sonication. 29 The expression level of these transcripts was restored to basal levels within 24 hours. When high FUS pressure (0. 81 MPa) was applied, an increased and diffuse expression of P-gp was observed in tissues near damaged blood vessels. Under these conditions, basal P-gp expression was not restored within 120 hours, suggesting that higher FUS pressure may cause irreversible damage to the P-gp transporter pathway. 26 Together, these findings suggest that the effect of FUS+MB on the efflux transporter expression is dependent on the FUS pressure.
Ion channel activation
Modulation of ion channels, especially K+ channels, has been recognized as an early response in the signaling cascade induced by FUS+MB. This FUS+MB-induced ion channel activation was observed 1 hour after treatment in a rat C6 glioma model. The expression and protein level of the α-subunit of calcium-activated potassium channels (KCa) were increased significantly, concomitant with increased BBB permeability. 52 FUS+MB -induced ion channel opening has also been observed in vitro in several cell types, including endothelial cells 92 and breast cancer cells, 93 where an increased cellular influx of Ca2+ and K+ followed treatment.
Exact mechanisms of ion channel activation following FUS+MB remain to be revealed. Structural and physiological changes in ion channels activated by mechanical forces have been studied extensively. 103 During FUS+MB treatment, MB cavitation and altered blood flow can both induce changes in the mechanical stress sensed by endothelial cells. Current research on mechanotransduction in the endothelium offers several insights regarding mechanisms of FUS+MB-induced ion channel activation 104 : (1) a direct physical interaction link may exist between the mechanical force on the endothelial cell surface and the ion channel, in which case the force pushes the ion channel open, (2) mechanical forces acting on endothelial cells change the tension on cytoskeleton or cell membrane fluidity, which in turn changes channel function and activity, and (3) the response of ion channels may be secondary to the activation of cellular proteins (e.g., kinases and phosphatases), which modulate ion channel activity. Future studies are needed to better understand the exact mechanism of ion channel activation following FUS+MB treatment and associated functional effects on the NVU.
Endothelial cell proliferation
FUS+MB treatments can induce endothelial cell proliferation and angiogenesis in the brain tissue. Following FUS+MB treatment, researchers have observed an acute upregulation in the expression of pro-angiogenic factor, vascular endothelial growth factor (VEGF),28,105,106 and angiogenesis-related genes, including egr3, ltgb2, lgals3, and mmp9. 29 Importantly, this increase in VEGF and other angiogenesis-related proteins may also increase BBB permeability.107,108 Endothelial cell proliferation may not happen right away, as suggested by one study in which no significant changes in the fluorescent intensity from staining Glut1, a brain endothelial cell marker, were detected 24 hours post sonication. 96 However, increased phosphorylation (activation) of c-jun N-terminal kinase (pJNK), which is involved in endothelial cell proliferation, apoptosis, and differentiation, was detected in the thalamus of sonicated brains at 24 hours post sonication, suggesting that endothelial cell proliferation may occur at a later timepoint. 96 Delayed angiogenesis is further supported by several studies that report a late-stage upregulation in the density of blood vessel segments and newborn endothelial cells 7 – 21 days after FUS+MB treatment.105,106 One study found an increased number of blood vessels stained by Glut1 and CD31 (also known as PECAM-1) in the hippocampus 10 days post sonication. 105 Another study showed an increased area of fluorescence from Glut1 staining in the hippocampus of sonicated brains between 7 and 14 days. 106 The study also found an increased number of BrdU+/Glut1+ cells, a marker of endothelial cell proliferation, in the sonicated hippocampus up to 21 days, suggesting that FUS+MB can affect endothelial cells long after sonication. This increase in endothelial cell proliferation may be triggered by changes in shear stress applied on the cerebral endothelium caused by FUS+MB.104,109 Signals arising through mechanotransduction have been shown to mediate diverse cellular processes, including cell growth, angiogenesis, apoptosis, migration, and differentiation.104,110 It remains to be determined whether FUS+MB-induced endothelial cell proliferation and angiogenesis can produce functional, perfused blood vessels. This bioeffect could significantly impact the treatment of cerebral ischemia, where damage to the brain issue is caused by blockage in blood vessels.
Effects of FUS+MB on astrocytes
It is known that FUS+MB can induce astrocyte reactivity and microglia activation as a result of a sterile inflammatory response in healthy mice.28,105 Both single-location and large volume FUS+MB treatment led to an increase in the area or intensity of GFAP and Iba1 signals in the brain as early as 3 hours post-sonication. This effect can last for as long as 7 weeks (Table 3).25,28,49,105,111 For some cases, astrocyte reactivity was reported to recover within 2 weeks for single location treatment and within 13 weeks for large volume treatment.25,111 In a high-grade murine glioma model, moderate and heterogeneous astrocyte activity (GFAP expression) was observed in tumor regions without FUS+MB treatment, whereas increased astrogliosis and astrocyte activity were observed in regions of tumors targeted with FUS. 53
Effects of FUS+MB on astrocytes in the NVU.
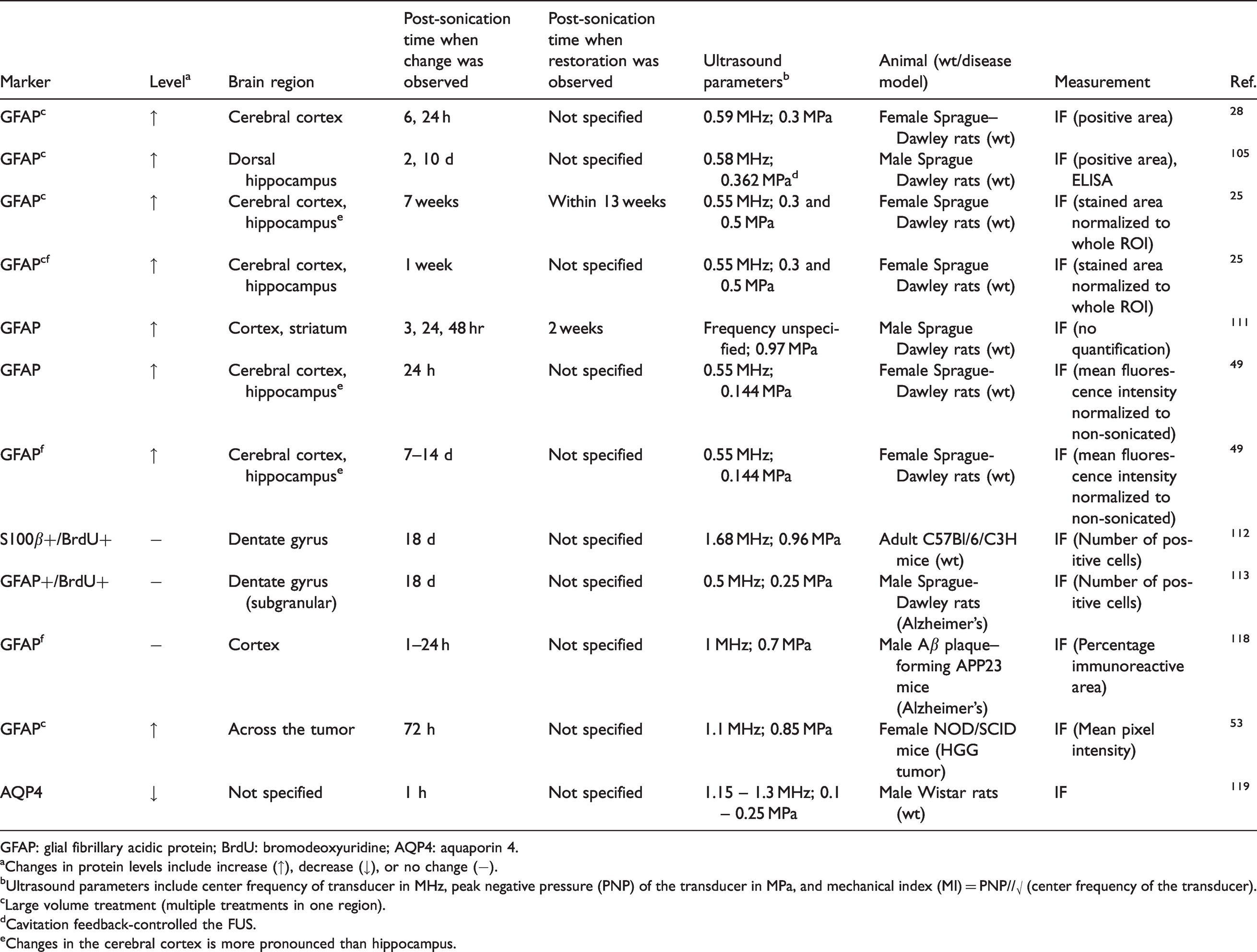
GFAP: glial fibrillary acidic protein; BrdU: bromodeoxyuridine; AQP4: aquaporin 4.
aChanges in protein levels include increase (↑), decrease (↓), or no change (−).
bUltrasound parameters include center frequency of transducer in MHz, peak negative pressure (PNP) of the transducer in MPa, and mechanical index (MI) = PNP//√ (center frequency of the transducer).
cLarge volume treatment (multiple treatments in one region).
dCavitation feedback-controlled the FUS.
eChanges in the cerebral cortex is more pronounced than hippocampus.
Spatial heterogeneity of astrocyte reactivity in response to FUS+MB has been observed. In one study where healthy rats received FUS in both the cerebral cortex and hippocampal regions, an increase in GFAP expression 24 hours post-sonication was more pronounced in the cerebral cortex than hippocampus for both one-time FUS treatment and multiple treatments at different time points. 49 Another study reported similar spatial heterogeneity in astrocyte activation at a later time point, where astrocytes in the sonicated cerebral cortex showed increased levels of GFAP 7 weeks following sonication while no significant change was observed in the sonicated hippocampus. 25
Although short-term astrogliosis has been reported after FUS+MB treatment, long-term astrogliosis has not been observed, suggesting that FUS-induced astrocyte proliferation may be temporary. One study found no significant astrogliosis in FUS-treated adult mice after 18 days by counting positive cells stained with an astrocyte marker (S100β) and a cell proliferation marker (BrdU). 112 Another study using a rat model of Alzheimer's disease, found no astrogliosis at 18 days post sonication. 113
Recent reports have shown that FUS+MB may enhance the clearance of waste products, such as Amyloid β, in the brain.114–118 It is suspected that astrocytes play a role in this FUS+MB-enhanced clearance. One hypothesis is that the CSF travels along peri-arterial spaces, enters the brain interstitium partly through aquaporin-4 (AQP4) channels on astrocyte endfeet, and transports across the glymphatic space by diffusion and convection. At the same time, it mixes with interstitial fluid and pulls along waste products and drains via the peri-venous spaces. 77 However, this hypothesis is contrary to the observation that AQP4 protein levels reduced after FUS+MB due to the loss of astrocytic endfeet. 119 The role of astrocyte end-feet and AQP4 remains unclear in the regulation of solute transport within parenchyma extracellular spaces. 120 Recently, a study used FUS+MB to study the glymphatic system in human subjects and observed glymphatic efflux. 35 The authors suspected that it may be due to changes of AQP4 levels on astrocyte endfeet following FUS+MB treatment. Further studies are needed to understand whether astrocytes contribute to enhancing waste clearance following FUS+MB treatment and, if yes, how.
Effects of FUS+MB on microglia
Several preclinical studies have observed FUS+MB-induced microglia activation using methods from immunohistochemistry staining of Iba1 to noninvasive PET imaging with [18F]-DPA714, a biomarker of translocator protein (TSPO) at the site of BBB disruption.25,28,49,53,111,121 FUS+MB-induced microglia activation occurred as early as 1 hour after sonication and may last for as long as 7 weeks while the recovery time ranges from 24 hours to 13 weeks (Table 4). Both single and multiple treatments of FUS+MB have been shown to activate microglia in the sonicated cortex. 49 Additionally, upregulation of translocator protein, a highly inducible protein localized on the mitochondrial outer membrane upon microglial activation, was observed in brain regions treated by FUS+MB throughout 14 days. 49
Effects of FUS+MB on microglia in the NVU.
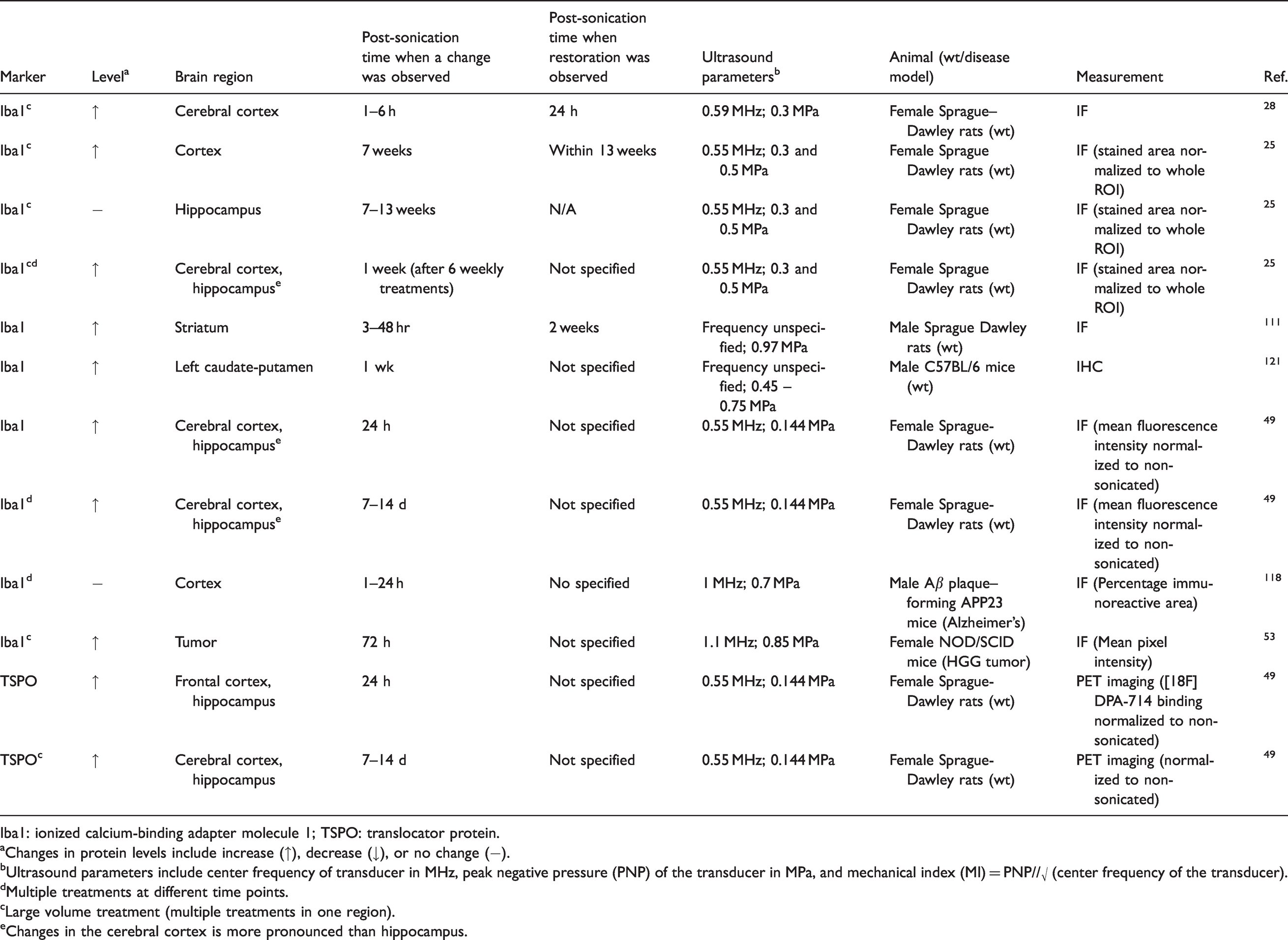
Iba1: ionized calcium-binding adapter molecule 1; TSPO: translocator protein.
aChanges in protein levels include increase (↑), decrease (↓), or no change (−).
bUltrasound parameters include center frequency of transducer in MHz, peak negative pressure (PNP) of the transducer in MPa, and mechanical index (MI) = PNP//√ (center frequency of the transducer).
dMultiple treatments at different time points.
cLarge volume treatment (multiple treatments in one region).
eChanges in the cerebral cortex is more pronounced than hippocampus.
Activated microglia may have opposing effects on BBB permeability: their initial expression of TJ proteins can promote BBB integrity, while chronic inflammation can cause microglia phagocytosis of astrocyte endfeet and impair BBB function.84,122,123 Given that FUS+MB only causes transient microglia activation, the former effect may dominate, however, further studies are needed to understand the complete effect of FUS+MB-induced microglia activation and establish safety limits. In addition, activated microglia secret numerous soluble factors, including pro- and anti-inflammatory cytokines, growth factors, chemokines, and neurotrophins, which can impact a wide range of cells in the brain.56,82,83
FUS+MB-activated microglia were shown to take up albumin-binding amyloid β by phagocytosis in mouse models of Alzheimer's disease.118,124 In addition, FUS+MB increased Iba1 immunoreactivity and induced “activated” microglia morphology in wild type and Alzheimer's disease models, respectively. 118 Although it is needed to establish safety limits and optimize treatment protocol, FUS+MB holds promise to be applied in clinics to clear waste products, such as Amyloid β, in aging brains and neurodegenerative brain diseases.
Similar to astrocytic activation, spatial heterogeneity of Iba1 signal following FUS+MB treatment was evident in the brain. One study showed that an increase in Iba1 expression 24 hours post-sonication was more pronounced in the FUS-treated cerebral cortex than the FUS-treated hippocampus. 49 One study reported an increase of Iba1 signal in the cerebral cortex at 7 weeks following sonication, while no increase of Iba1 was observed in the hippocampus. 25 Given that the activation of microglia and astrocytes shares similar spatial heterogeneity, the FUS+MB-induced activation of these two cell types may be coupled. The density of microglia in different brain regions may also contribute to such spatial heterogeneity of Iba1.
The differential Iba1 expression in microglia may only reflect a small part of the intrinsic heterogeneity of microglia. Recent transcriptome analyses have uncovered diverse functional phenotypes of microglia, ranging from pro-inflammatory “M1-like” phenotypes (i.e., upregulates inflammatory mediators such as Tnf, Il1b) to immunosuppressive “M2-like” phenotypes (i.e., upregulates Chil3, Fzd1, and Arg1). 125 Existing studies found upregulation of Tnf and Il1b in the brain regions targeted by FUS,27–29 suggesting that FUS+MB may enhance the “M1-like” phenotype in microglia. A diverse population of microglia was found post sonication using single-cell RNA sequencing, 43 but it is unclear whether FUS+MB increased or reduced such cell heterogeneity. Since this is still an understudied area, we do not know the full extent of FUS+MB effects on microglia except that some microglia can be activated. It is unclear why FUS+MB “selectively” activates some microglia but not others. Understanding the effects of FUS+MB on cell heterogeneity could have clinical value, as cell heterogeneity has long been recognized as a hallmark for cancer and tumor environment.
Effects of FUS+MB on neurons
Although neurons are not in direct contact with FUS-activated MBs, FUS+MB can still have biological effects on neurons through several mechanisms. First, vasospasm, vasoconstriction, and interrupted local blood flow induced by FUS+MB may cause ischemic neuronal damage; however, no direct evidence of neuronal damage has been seen so far, suggesting that this effect from transient vasoconstriction and blood flow changes may be limited.91,127 Second, mechanical forces from blood vessel expansion and contraction, as well as the ultrasound waves themselves, may activate mechanotransduction pathways in surrounding neurons due to the proximity of neurons and blood vessels (<10 – 20 µm 128 ) Third, BBB disruption following FUS+MB treatment causes the exchange and release of cytokines and solute molecules between the blood and brain tissue, which may affect neuronal function. One of the physiological responses following FUS+MB, acute inflammation, is caused by rapid and early activation of the glial cells as a response to the mechanical and biological stimuli, leading to neuroprotective effects such as clearing of abnormal protein deposits. 129 In this section, we review existing studies on neuronal responses following FUS+MB treatment and remaining knowledge gaps. Table 5 provides a summary of molecular changes in neurons and experimental conditions used in each study.
Effects of FUS+MB on neurons in the NVU.
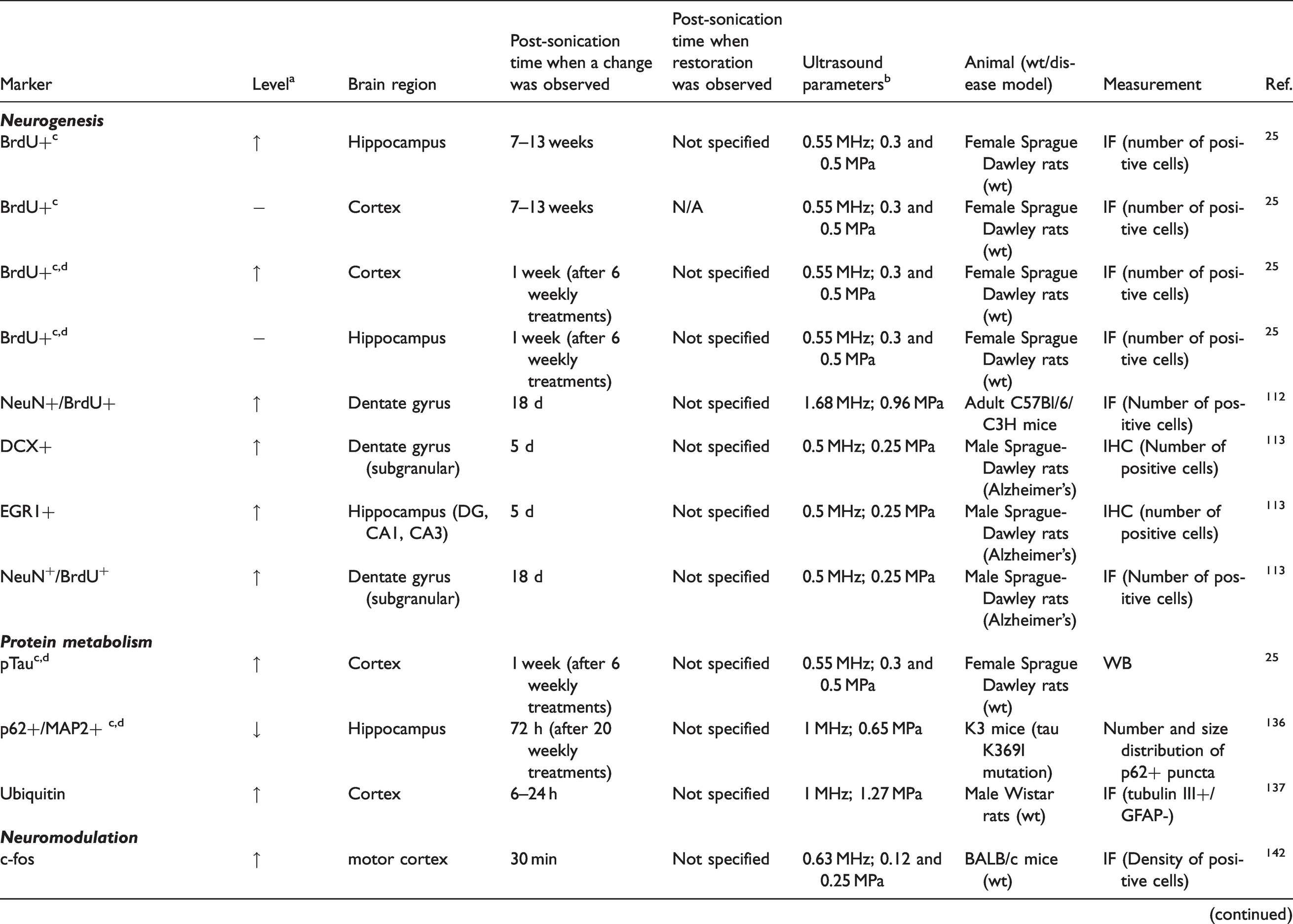
NeuN: neuronal nuclear protein; DCX: doublecortin; EGR1: early growth response protein 1; p62: sequestosome-1; MAP2: microtubule-associated protein 2; Akt: protein kinase B; GSK3b: glycogen synthase kinase 3 beta.
aChanges in protein levels include increase (↑), decrease (↓), or no change (−).
bUltrasound parameters include center frequency of transducer in MHz, peak negative pressure (PNP) of the transducer in MPa, and mechanical index (MI) = PNP//√ (center frequency of the transducer).
cLarge volume treatment (multiple treatments in one region).
dMultiple treatments at different time points.
Neurogenesis
Neurogenesis, the persistent formation of functional, mature neurons from neural stem cells, appears to be decreased in aged adults and those with Alzheimer's disease.130,131 As a result, multiple efforts are underway to study whether FUS+MB treatment can enhance neurogenesis in the adult brain or interrupt the neurodegenerative process that leads to neuronal death. Several preclinical studies reported promising results. One study showed that FUS treatment significantly increased the number of proliferating neurons (NeuN+/BrdU+) in the dentate gyrus of adult mice 18 days after sonication. 112 Another study reported upregulated EGR1, a marker for neuronal plasticity, and an increased number of neuroblasts (DCX-positive) in a dementia rat model 5 days after sonication, along with a significant increase of NeuN+/BrdU+ cells in the subgranular zone (SGZ) and the granule cell layer (GCL) of the hippocampal dentate gyrus at 18 days after sonication. 113 Spatial heterogeneity of FUS-induced neurogenesis has been observed in adult rats. Kovacs et al. showed that 6 weekly FUS+MB treatments significantly increased the number of BrdU+ cells in the sonicated frontal lobe compared to contralateral brain regions, while no significant increase was detected in the sonicated hippocampus. 25 In contrast, an increased number of BrdU+ cells was detected in the sonicated hippocampus but not the cerebral cortex at 7 or 13 weeks after a single FUS+MB treatment. 25 This study also reported co-localization of NeuN+ and BrdU+ cells, suggesting that these proliferating cells were neurons. 25
Several studies have shed light on the mechanisms of FUS+MB-induced neurogenesis. Recent work suggests that this effect is dependent on increasing the permeability of the BBB, as sonication at low pressures with MBs, as well as at high pressures in the absence of MBs, both failed to affect the density of BrdU+ cells in the dentate gyrus. 132 One possibility is that neurogenesis is stimulated by cytokines such as neuromodulators and growth factors that extravasate from the blood. Indeed, FUS+MB-induced upregulation of growth factors, including brain-derived neurotrophic factor (BDNF) and early growth response 1 (EGR1) was associated with neurogenesis in a rat dementia model 18 days post-sonication. 113 Furthermore, a sustained increase in phosphorylation (activation) of Akt and GSK3β was observed for 24 hours after sonication in neuronal cells flanking the region of the FUS+MB-induced BBB opening. 98 These data suggest that the activation of proliferation pathways in neuronal cells is involved in FUS+MB-induced neurogenesis.
Intraneuron protein metabolism
A prominent feature of neurodegenerative disorders is the aggregation of misfolded endogenous proteins into insoluble inclusions. 133 These misfolded proteins and inclusion bodies are believed to play a critical role in disease propagation, neuronal death, and dysfunction. For instance, hyperphosphorylated tau protein species and pathological tau aggregations are implicated in the spread of neuropathology and neurodegeneration in Alzheimer's disease and other tauopathies.134,135
FUS+MB can be used to promote clearance of pathological tau from the brain. In line with this, a recent study demonstrated that repeated FUS+MB treatments (over 15 weeks) induced a three-fold reduction of hippocampal phosphorylated tau-positive deposits and a two-fold reduction in the number of both cortical and hippocampal neurofibrillary tangles in a K3 mouse model (carrying K369I mutant tau). 136 The absence of a reduction in phosphorylated tau-positive deposits in the cerebral cortex was attributed to greater dispersion of these deposits and dilution of FUS+MB effect in the cerebral cortex, compared to the hippocampus. 136 While a previous study showed that FUS+MB enhanced ubiquitin expression, 137 the change in tau deposits was not due to tau ubiquitination or a decrease in protein kinase activity, 136 which in turn could result in proteasome-mediated protein clearance and decreased tau phosphorylation, respectively. In contrast, FUS+MB induced neuronal autophagic activity with increased interaction between tau and p62 (an autophagy adaptor and flux marker), increased beclin-1 expression (an autophagosomal nucleation factor), and decreased mTOR activity (a key regulator of autophagy). 136 In another study using a different tau mouse model (rTg4510 mice carrying P301L mutant tau), a shorter course of FUS+MB treatment (4 weeks) also resulted in decreased phosphorylated tau density both on the sonicated side and, to a lesser degree, on the contralateral side. 124 Furthermore, FUS+MB resulted in microglial activation, and immune cell density was associated with lower phosphorylated tau density. 124
Contrary to the above findings that FUS+MB decreased phosphorylated tau density in Alzheimer’s mouse models, multiple FUS+MB treatments in wild-type rats were reported to increase the number of phosphorylated tau-positive neurofibrillary tangles in the sonicated cerebral cortex (but not the hippocampus). 25 Underlying mechanisms of FUS+MB-induced neurofibrillary tangle accumulation as well as its potential implications in humans remain elusive. More studies are needed to further our understanding of cellular and molecular pathways by which FUS+MB can affect neuronal protein homeostasis. It is also unclear whether FUS+MB can have beneficial effects on other intracellular proteinopathies (e.g., synucleopathies and TDP-43 proteinopathies). This is particularly relevant in patients who have mixed neuropathologies, such as in older patient groups. 138 Finally, animal models only recapitulate certain aspects of neurodegenerative disorders. Clinical studies are needed to determine the efficacy of FUS+MB-mediated BBB opening and provide mechanistic insight into FUS+MB-induced bioeffects in neurodegenerative disorders.
Gap junctions between neurons
Sonication does not appear to alter total gap junction connectivity between neurons but does appear to differentially affect small- versus large-sized gap-junctional plaques between neurons and astrocytes. Small neuron-astrocyte plaques were reduced in number, while large plagues were increased at 3 to 6 hours after sonication (1.25 MPa) with MBs. Basal plaque size distribution was restored within 24 hours. 139 These results indicate that FUS+MB leads to a reorganization of gap-junctional plaques in both neurons and astrocytes. Interestingly, similar gap-junctional changes have been observed as a response to ischemia in the brain,140,141 suggesting that this re-distribution may be crucial for the maintenance of homeostasis in the face of tissue stressors. Additional gap-junctional changes may also be indicative of cell damage. When cells are damaged by high-pressure FUS (1.25 MPa), ions (e.g., Ca2+ and K+) present at high concentrations in the extracellular fluid move into the cell. This can induce gap junction channel closure, potentially limiting the spread of damage to neighboring cells. Further studies are needed to assess whether FUS+MB-induced gap junctional changes lead to functional changes in neuronal integrity, and, if so, whether gap junctions between neurons can be used as a safety measure for FUS+MB effects on neurons.
Neuronal circuit modulation
Although FUS alone can modulate neuronal activity and cognitive function, FUS+MB may induce a stronger effect in neurons. A recent study demonstrated that expression of c-fos, a molecular marker for neuronal activity, was enhanced when FUS and MBs were applied together compared to FUS alone in the sonicated brain region. 142 Interestingly, FUS+MB also significantly increased the success rate of eliciting electromyogram compared to FUS alone, which may be explained by the enhanced expression of c-fos at the motor cortex. 142 One non-human primates study showed that FUS+MB improved performance in a reward-based reaching task by decreasing reaction times and touch error. 143 Another study reported that FUS+MB targeting at the somatosensory cortex significantly reduced the somatosensory-evoked potential in non-human primates, and the effect lasted from less than one hour to 7 days depending on the FUS parameter (mechanical index). 144 These results suggest that FUS+MB could have a positive and sustained impact on cognitive function. However, the underlying mechanisms remain unknown due to the lack of molecular and cellular studies.
It is suspected that FUS+MB-induced neuromodulation is attributed to the extravasation of blood constituents from FUS+MB-induced BBB opening and mechanical effects from oscillating MBs. FUS+MB-induced BBB opening can enrich the parenchymal concentration of oxygen, glucose, ions, nutrients, and hormones, which can affect neuronal function. 145 On the other hand, oscillating MBs may radiate acoustic energy into the surrounding medium, thereby affecting neurons through mechanotransduction pathways. Challenges remain to elucidate mechanisms involved in neuromodulation from FUS+MB treatment. To that end, further studies are needed to link neuroplasticity at the molecular and cellular level with the behavioral outcome to understand mechanisms of FUS effects on neuronal function.
Concluding remarks
In this review, we summarized existing evidence on cellular and molecular effects induced by FUS+MB treatment in the NVU. To understand the full extent of bioeffects associated with FUS+MB and to further advance its clinical applications, several key questions remain to be answered. First, how differently do diseased brains respond to FUS+MB compared to healthy ones? Second, how does cellular and spatial heterogeneity within the brain affect FUS+MB-induced bioeffects? Third, how long does it take for different bioeffects to be induced and how long do they last? And fourth, after gaining this new knowledge, what new therapeutic applications can FUS+MB have?
There is a lack of preclinical studies on FUS+MB-induced biological effects and underlying mechanisms in diseased animal models. Considering that the BBB and the tissue environment in brain diseases are altered from those in a healthy brain, it is crucial to gain more knowledge about how brain diseases modify FUS+MB-induced bioeffects. For example, reduced expression and activity of efflux transporters, P-gp and Glut1, are known to occur in patients with Alzheimer’s disease. 146 The impact that such reduced efflux activity has on the efficacy of FUS+MB-induced BBB opening remains unclear.
Existing studies largely focus on averaged bioeffects at the bulk tissue level, despite increasing evidence showing significant local differences in BBB permeability in both healthy and diseased brains. 147 In healthy brains, local differences in BBB permeability may be caused by the difference in white matter versus grey matter, differences in vessel types and density, and other anatomical features in different regions of the parenchymal. For brain tumors, a portion of the BBB can be disrupted by infiltrating tumor cells to become the blood tumor barrier. This type of local differences and intratumoral heterogeneity may translate into a differential response to FUS+MB treatment. Indeed, a recent study reported that the caudate putamen had a lower level of FUS+MB-induced BBB opening compared to the thalamus, possibly due to the difference in ultrasound’s incidence angle at these targeted locations. 48 Spatial heterogeneity of microglia and astrocyte activation following FUS+MB has also been reported in several studies.25,49 Therefore, a better understanding of the location and cell-specific bioeffects induced by FUS is desired. Traditional omics tools such as RNA sequencing (RNA-seq) can shed light on the bioeffects of FUS+MB without being biased and confounded by limited a priori knowledge (as in candidate pathway studies). However, RNA-seq is blind to the cell type of origin and location concerning the FUS beam, making it difficult to be used in studying cell and location-specific FUS+MB effects. While immunohistochemical staining is widely used to gain spatial information, its low throughput and low multiplexity make it unsuitable for mechanistic studies. The key to solving this problem may lie with advanced spatial transcriptomics and proteomics tools such as 10x Genomics’s Visium Spatial Gene Expression system and Nanostring’s GeoMx™ Digital Spatial Profiler system.148,149
As Todd et al. pointed out in a recent review, different bioeffects of FUS+MB can happen at various time points following treatment. 24 For example, changes in BBB permeability happen immediately after sonication and last up to 24 hours, while it may take 4–5 days to observe clearance of Amyloid β and up to 3 weeks to observe neurogenesis. 24 In our review, we noted that the timing and duration of several bioeffects have not been determined or specified (Tables 2 and 5). A better understanding of the temporal dynamics of FUS+MB-induced bioeffects is critical to understand underlying mechanisms. To this end, advanced technologies such as deep tissue live imaging with multi-photon microscopy may offer insights into the temporal profile of FUS+MB-induced bioeffects through live imaging.50,116 Finally, as we translate FUS+MB treatments from animal models to clinical studies, it is important to develop non-invasive tests to monitor both short-term and long-term effects of FUS+MB on the brain. These biomarkers may be detected by FUS+MB-enabled liquid biopsy, a new technique recently introduced for noninvasive detection of brain-specific biomarkers by releasing these biomarkers from the brain to the blood.150,151
Many of the biological effects in the NVU besides BBB opening have only been discovered relatively recently and are still under investigation. As a more complete picture of cellular and molecular effects induced by FUS+MB emerges, new ways to apply FUS+MB treatment beyond drug delivery may become apparent. For example, FUS-enhanced transcytosis is now considered to play a bigger role in transporting agents/drugs across BBB than paracellular pathways, suggesting drugs targeting caveolin or receptors involved in transcytosis in combination with FUS-activated MBs may achieve higher efficacy in drug delivery. Effects of FUS+MB on pericytes remain largely unknown despite the important roles pericytes play in regulating BBB permeability.152,153 Furthermore, FUS+MB-enhanced waste clearance via microglia and neurons is still an understudied field, despite its promising clinical value in treating Alzheimer's disease.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Institutes of Health (R01EB027223, R01EB030102, and R01MH116981), the Charlie Teo Foundation, and the Little Legs Foundation.
Acknowledgements
The authors would also like to thank Charlotte Weixel for proofreading the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
