Abstract
Multi-parametric photoacoustic microscopy (PAM) has emerged as a promising new technique for high-resolution quantification of hemodynamics and oxygen metabolism in the mouse brain. In this work, we have extended the scope of multi-parametric PAM to longitudinal, cortex-wide, awake-brain imaging with the use of a long-lifetime (24 weeks), wide-field (5 × 7 mm2), light-weight (2 g), dual-transparency (
Keywords
Introduction
Capable of imaging the cerebral vasculature with comprehensive functional and metabolic insights, photoacoustic microscopy (PAM) shows increasing promise in basic and translational brain research.1–3 In particular, multi-parametric PAM has demonstrated simultaneous imaging of hemoglobin concentration, blood oxygenation, and blood flow in the awake mouse brain at the microscopic level4,5 and has been applied to study a wide variety of brain diseases, including stroke, 6 traumatic brain injury, 7 neurodegenerative disease, 8 and multiple sclerosis. 9
However, it remains an unmet challenge for multi-parametric PAM to carry out functional and metabolic imaging of the same mouse brain
In contrast to the rapid and exciting advances in longitudinal, cortex-wide, high-resolution TPM of neuronal morphology and activity, PAM of cerebrovascular structure and function with micron-level resolution and similar spatial and temporal coverage remains to be demonstrated. In this work, we extended the scope of PAM to longitudinal, cortex-wide, functional-metabolic imaging of the awake mouse brain through the development, assessment, and application of a long-lifetime (24 weeks), wide-field (5 × 7 mm2), light-weight (2 g), and dual-transparency (
Material and methods
3D-Printed cranial window frame
For longitudinal, cortex-wide imaging of the awake mouse brain, a head-mounted cranial window frame was designed and optimized to achieve light weight (2 g, which is ∼5% of the animal’s body weight and thus wearable) while retaining stiffness and durability. The window frame can be 3 D printed using the PA12 Multi-Jet Fusion Plastic (Shapeways, New York City, New York), which meets the requirements on weight, stiffness, and durability. As shown in Figure 1(a), the window frame has a large central opening of 6 × 8 mm2 for convenient imaging access to the bilateral mouse cortex. The bottom of the frame is curved to fit the average skull contour of adult mice, so that the frame can be securely installed onto the exposed skull. Each of the two extended lateral wings of the frame has a bore to thread a #4–40 screw into a metal arm-piece to restrain the mouse head for awake-brain imaging (Figure 1(b)).
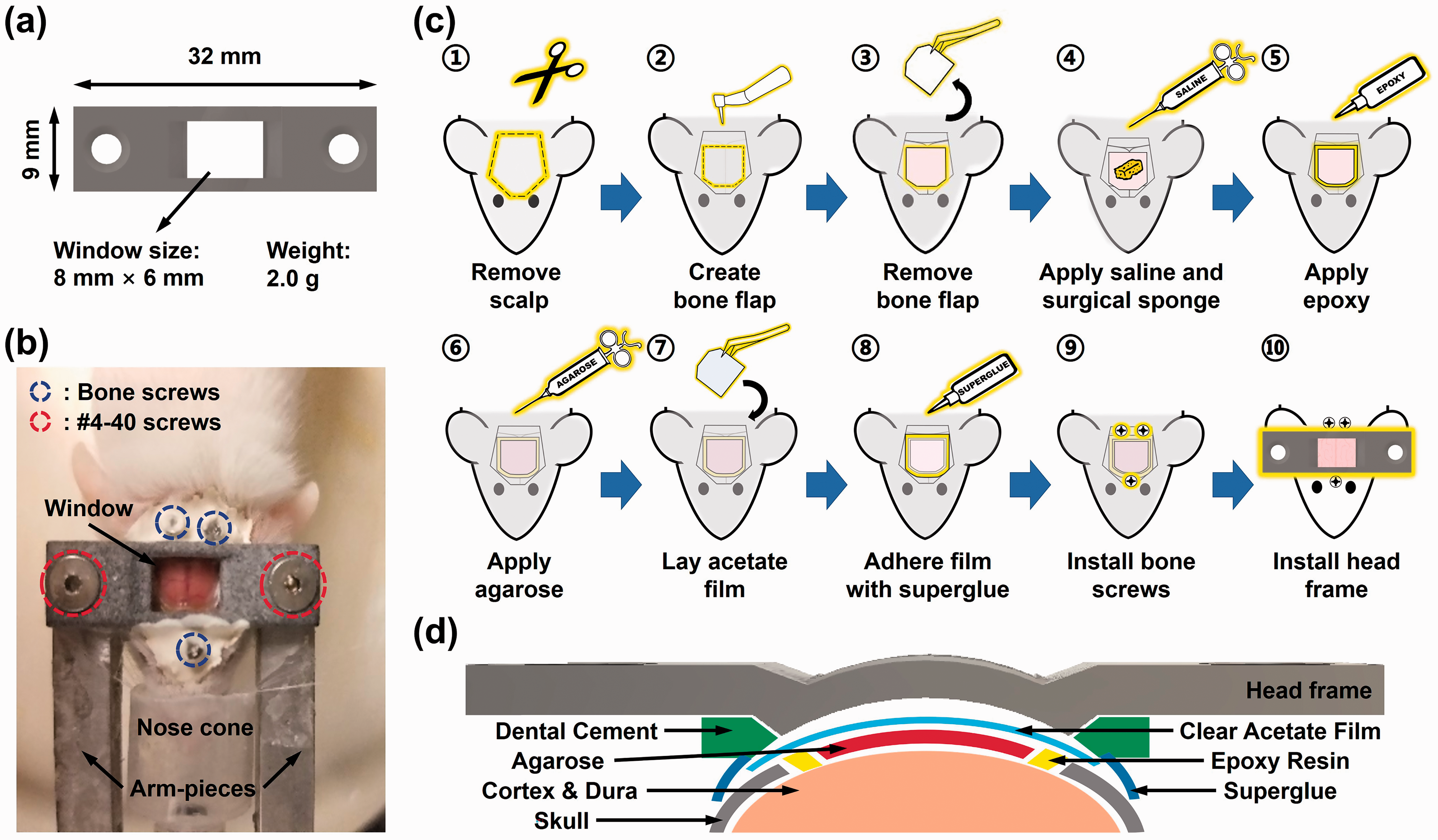
Dual-transparency cranial window. (a) Dimensions and weight of the 3D printed cranial window frame. (b) Top view of a window-installed mouse in the head-restraint setting ready for awake-brain imaging. (c) Major steps of the cranial window preparation procedure. (d) Detailed layout of the cranial window.
Cranial window preparation
CD-1 mice (male, 6–10 weeks old, 30–40 grams; Charles River Laboratories, Wilmington, Massachusetts) were used. First, the animal was anesthetized with vaporized isoflurane in an induction chamber (EZ-SA800, E-Z Systems, Palmer, Pennsylvania). Toe pinch was performed to ensure effective anesthesia. Then, it was secured in a homemade stereotaxic surgical station, and the body temperature was maintained at 37 °C using a homeothermic monitoring system (No. 69020, RWD life science, Dover, Delaware). Anesthesia was maintained throughout the preparation process by 1–2% vaporized isoflurane, which was delivered with the inhalation gas through a nose cone in the stereotaxic setup (Figure 1(b)). Carprofen (5 mg/kg) and dexamethasone (0.2 mg/kg) were administered to mitigate inflammation and swelling of the brain in response to craniotomy. The mouse eyes were lubricated with ophthalmic ointment (Stye™, Lynchburg, Virginia) to avoid corneal drying.
Before craniotomy, hair on the mouse scalp was removed by a hair trimmer (9990-1301, Wahl Clipper, Sterling, Illinois). Then, the scalp was disinfected with povidone-iodine solution, wiped with 70% ethanol prep pads, and removed with surgical scissors (MDS10030, Medline Industries, Mundelein, Illinois)—starting with an incision along the base of the head, then two cuts in the rostral direction toward the eyelids, and finally two oblique cuts converging at the midline (Figure 1(c)①). After treatment with a mixture of lidocaine (HCl 1%) and epinephrine (1:100,000) to avoid bleeding or pain, the periosteum was removed with a scalpel (6008 T-10, Medline Industries, Mundelein, Illinois). Also, neck muscles were gently retracted to expose the interparietal bone. The surgically exposed skull was then scraped to create a dry surface for good adhesion of the glue when applied later (Figure 1(c)①). The desired boundaries of the cranial window were delineated using a fine-point marker, showing a 7 × 5 mm2 trapezoid with the longer base at 1 mm rostral to the lambda and the shorter base at 2–3 mm rostral to the bregma. During craniotomy, a shallow groove was created along the boundaries by using a dental drill (K.8350-H.30, Foredom, Bethel, Connecticut) with a 0.7-mm drill bit (FG, Shofu Dental, San Marcos, California) under a stereo microscope (SM-3B, AmScope, Irvine, California) after a second lidocaine-epinephrine treatment. Note that the drilling was paused every 30 seconds to avoid overheat of the skull. Once the shallow groove was created, a finer drill bit with a 0.5-mm diameter (CN1, Shofu Dental, San Marcos, California) was used to deepen the groove until a very thin layer of the bone remained (Figure 1(c)②). Then, the bone flap was immersed in sterile saline and peeled off with fine-tip forceps (Figure 1c③). After craniotomy, surgical sponges were applied to the exposed mouse brain to stop bleeding (Figure 1(c)④).
Once the dura mater was dried and no bleeding was observed, the mixture of epoxy resin and hardener (Starter Kit, ArtResin, Carrollton, Texas) was carefully applied along the edge of the cranial window with a fine-tipped applicator to prevent bone regrowth. After curing of the epoxy mixture (∼5 minutes), the exposed mouse brain was flushed with sterile saline to prevent drying (Figure 1(c)⑤). Then, the surrounding intact skull was carefully dried with Kimwipes, and a few drops of 0.6% agarose (pre-prepared and then stored in a 55 °C water bath) was applied onto the exposed brain with a 0.3 cc syringe (Figure 1(c)⑥). A pre-cut acetate film (76.2 µm, Clear Dura-Lar, Grafix, Maple Heights, Ohio) was gently laid on top of the agarose over the window area. Using a pair of thumb forceps, the two non-parallel sides of the film were pressed down until flush against the skull following its natural curvature. Agarose outside the window area was carefully removed with Kimwipes, while ensuring sufficient agarose under the film so that no air bubble was generated. The film was recentered with the window as needed, allowing ∼1-mm overlap with the skull on each side (Figure 1(c)⑦). Then, the gel-based cyanoacrylate glue (2,34,790 Loctite, Hartford, Connecticut) was applied to all edges of the window and allowed for one minute to cure, before the removal of the forceps (Figure 1(c)⑧). With the 0.7-mm dental drill, three deep indents were created in the remaining skull, two on each side of the interparietal bone near the caudal end and one on either side of the nasal bone near the coronal suture. Three bone screws (90065A013, McMaster-Carr, Elmhurst, Illinois) were installed in these indents. Caution was taken to ensure that the screws did not penetrate the skull to damage the underlying brain tissue (Figure 1(c)⑨). A layer of cyanoacrylate was applied along the bottom of the curved surface of the head frame. Then, the 3 D printed window frame was placed at the center of the window, and gentle downward pressure was applied for ∼30 seconds to allow for the glue to cure. Once the frame was attached to the skull and the acetate film, additional glue was applied along the outer edges of the frame, where it met the skull, to reinforce the adhesion. After the glue was cured, adhesive cement (S396, S398, and S371, Parkell, Edgewood, New York) was applied throughout the remaining skull surface. Additional cement was applied between the gap of the window frame and the skull to create a tight seal (Figure 1(c)⑩).
After the window installation, the animal was subcutaneously injected with Ketoprofen (5 mg/kg) and returned to its home cage once awakening from anesthesia. The entire procedure took ∼3 hours. Ketoprofen (5 mg/kg) was administered daily for 3 days post-surgery to reduce inflammation and pain. The animal was closely monitored for signs of distress and pain and to ensure easy access to food and water.
Then, the animal was subject to five daily training sessions to help acclimate to the head restraint and relieve stress. Each session lasted 45 minutes. Detailed training procedure can be found in our previous report. 5 The animals failed to acclimate to the restraint after training (3 out of 29, due to stress reactions to the head restraint) were excluded from imaging studies.
All experiments were carried out in conformity with the animal protocol approved by the Institutional Animal Care and Use Committees at the University of Virginia and Washington University in St. Louis, and were conducted in compliance with the Animal Welfare Act (AWA), National Institute of Health Guide for the Care and Use of Laboratory Animals, and ARRIVE (Animal Research: Reporting in Vivo Experiments). 19
Imaging through the cranial window vs. the intact skull
Under anesthesia, a structural image of the cerebral vasculature in an 8-week CD-1 mouse was acquired by PAM through the intact skull. Throughout the experiment, the temperatures of the animal body and brain were both maintained at 37 °C. After that, the animal was transferred to the stereotaxic surgical station for cranial window installation. Following the installation and solidification of the glue and cement, the animal was transferred back to the imaging system, where the same region of interest was reimaged with the same PAM setting and under the same physiological state. After the completion of the experiment, the animal was euthanized.
In vivo assessment of cerebrovascular permeability
After baseline PAM imaging under awake condition, the animal was anesthetized with 2% isoflurane and intravenously injected with 2% (w/v) Evans blue dye at the dose of 4 ml/kg body weight. After 2 hours of dye extravasation, the animal was transferred to the head-restrained PAM for awake-brain imaging at 532 nm, where both hemoglobin and Evans blue show strong optical absorption. The same procedure was utilized to assess the cerebrovascular permeability at different time points, including 0, 7, 14, and 18 days after installation of the cranial window, in four different animals from the same cohort. The animals were euthanized immediately after completion of the imaging experiments. Side-by-side comparison of the PAM images acquired before and 2 hours after Evans blue injection was performed to assess dye extravasation, which is indicative of cerebrovascular permeability. A detailed description of the
Immunohistochemistry analysis of astrogliosis and microgliosis
Deeply anesthetized with isoflurane, the animals were transcardially perfused with 0.01-M Phosphate-buffered saline (PBS). The brains were harvested, fixed in 4% paraformaldehyde for 24 hours, and transferred to 30% sucrose in 0.2-M PBS. After snap‐frozen on dry ice and stored at –80°C, the brains were sectioned (50-µm thickness) using a sliding microtome and stored in 0.1-M PBS with 30% sucrose and 30% ethylene glycol at −20°C. After treatment with the Rabbit anti‐Iba‐1 antibody (1: 1000; Wako Pure Chemicals Industries, Tokyo, Japan) and mouse anti-GFAP antibody (1:1000, Sigma, St. Louis, Missouri) overnight at 4 °C, the brain sections were incubated with Alexa Fluor 488‐conjugated donkey anti‐rabbit antibody (1:400; Molecular Probes, Grand Island, New York) and Cy3‐conjugated donkey anti-mouse antibody (1: 800; Jackson ImmunoResearch Laboratories, West Grove, Pennsylvania). Then, they were washed, nuclear counterstained with DAPI, mounted, and examined by a confocal microscope (Nikon A1Rsi, Nikon Instruments, Melville, New York).
Imaging under awake condition vs. general anesthesia
To demonstrate the utility of the cranial window for high-resolution, awake-brain imaging, multi-parametric PAM was continuously performed on the same mouse brain, during which the physiological state of the animal was altered between wakefulness and general anesthesia. Specifically, after 30-day recovery from the cranial window installation, the mouse head was secured in the head-restraint setting under a brief anesthesia with 2% isoflurane (<2 minutes). After the animal regained wakefulness for 30 minutes, multi-parametric PAM images were acquired. Then, the animal was anesthetized with 2% isoflurane. After 15 minutes under anesthesia to reach equilibrium, 5 a second set of multi-parametric PAM images were acquired over the same region of interest.
Longitudinal multi-parametric PAM imaging
After recovering from the window installation for 2–3 weeks and passing the head-restraint training, the animal was ready for awake-brain imaging by our head-restrained PAM system,5,20 which used two nanosecond-pulsed lasers (wavelength: 532 and 558 nm; repetition rate: 6 kHz; pulse energy: 100 nJ on the tissue surface) for optical excitation and a customized ring-shaped ultrasonic transducer (inner diameter: 2.2 mm; outer diameter: 4.0 mm; focal length: 6.0 mm; center frequency: 35 MHz; 6-dB intensity bandwidth: 70%) for acoustic detection. Throughout the imaging experiment, the animal was supplied with medical air (AI M-T, Praxair, Danbury, Connecticut). The brain temperature was maintained at 37 °C using a temperature-controlled water bath, which was attached to the mouse head for acoustic coupling.
5
After acquisition of the multi-parametric images over a 5.5 × 3.5 mm2 region (step sizes: 0.1 and 10 µm along the
Photothrombotic stroke
The photothrombotic stroke experiment was performed 30 days after the cranial window installation to ensure brain recovery. Specifically, after baseline PAM imaging under awake condition, the animal was briefly anesthetized with 2% isoflurane and administered with 1-mL Rose Bengal (20 mg/mL) intravitreally. Then, a 1.5 × 1.5 mm2 region of interest in the mouse brain was exposed to 1-mW, 543.5-nm laser light (25-LGP-193-249, Melles Griot, Carlsbad, California) through a mask for 15 minutes to induce focal photothrombotic stroke. After 3 hours, the animal was transferred to the head-restrained PAM for post-stroke multi-parametric imaging. Subsequent PAM imaging was performed on day 4, 7, 14, 21, and 28. The animals were euthanized at the conclusion of the stroke monitoring.
Segmentation analysis
A vessel segmentation algorithm we developed before was used for quantitative analyses at the single-vessel level. 21 Briefly, feeding arteries and draining veins in the region of interest were manually identified. Then, the Otsu thresholding method 22 was applied to acquire proper threshold values, with which vessel boundaries were precisely determined. By isolating these vessels, average values of the vessel diameter, total concentration of hemoglobin (CHb), oxygen saturation of hemoglobin (sO2), and blood flow speed within each segment were calculated. A detailed description of the vessel segmentation method can be found in our previous report. 21
The segmentation analysis also enables the quantification of microvessel density, which is defined as the number of pixels occupied by the microvasculature in the image divided by the total number of image pixels. To estimate the microvessel density, the microvasculature was first isolated by removing the non-vessel background as well as trunk arteries and veins, whose diameters are larger than 40 µm,5,20 using vessel segmentation. Then, the number of pixels occupied by the microvasculature was obtained, based on which the microvessel density was computed by taking the ratio of the microvascular pixel number to the total pixel number.
Statistical analysis
All animal-research data were reported in accordance with the ARRIVE guidelines.
19
Paired
Results
Cranial window improves PAM visualization of cortical vasculature
To demonstrate that the cranial window is essential for PAM visualization of the cortical microvasculature in adult mice, a side-by-side comparison between PAM of the mouse brain through the intact skull
As shown in the left column of Figure 2(a), through the intact skull of the 8-week-old mouse, the superior sagittal sinus (SSS) underneath a piece of relatively thick skull is not visible, and the entire transcranial image shows a weak contrast to the non-vessel background. Further, the zoom-in inset shows that only a small number of microvessels can be observed transcranially with limited signal strengths and blurred boundaries, due to the adverse influence of the skull. Moreover, the skull vasculature (indicated by arrowheads in Figure 2(a)), which appears in a thin, bright, and tortuous structure, obstructs the underlying cortical vessels of interest. In contrast, the image acquired through the cranial window shows much improved contrast and resolution (Figure 2(a), right column). Specifically, the SSS and its bifurcations can be clearly visible, along with significantly increased microvessel density throughout the entire cortical region of interest. Indeed, in the zoom-in view, more microvessels are observed with clearly resolved boundaries, showing a substantial improvement over the observation through the intact skull.
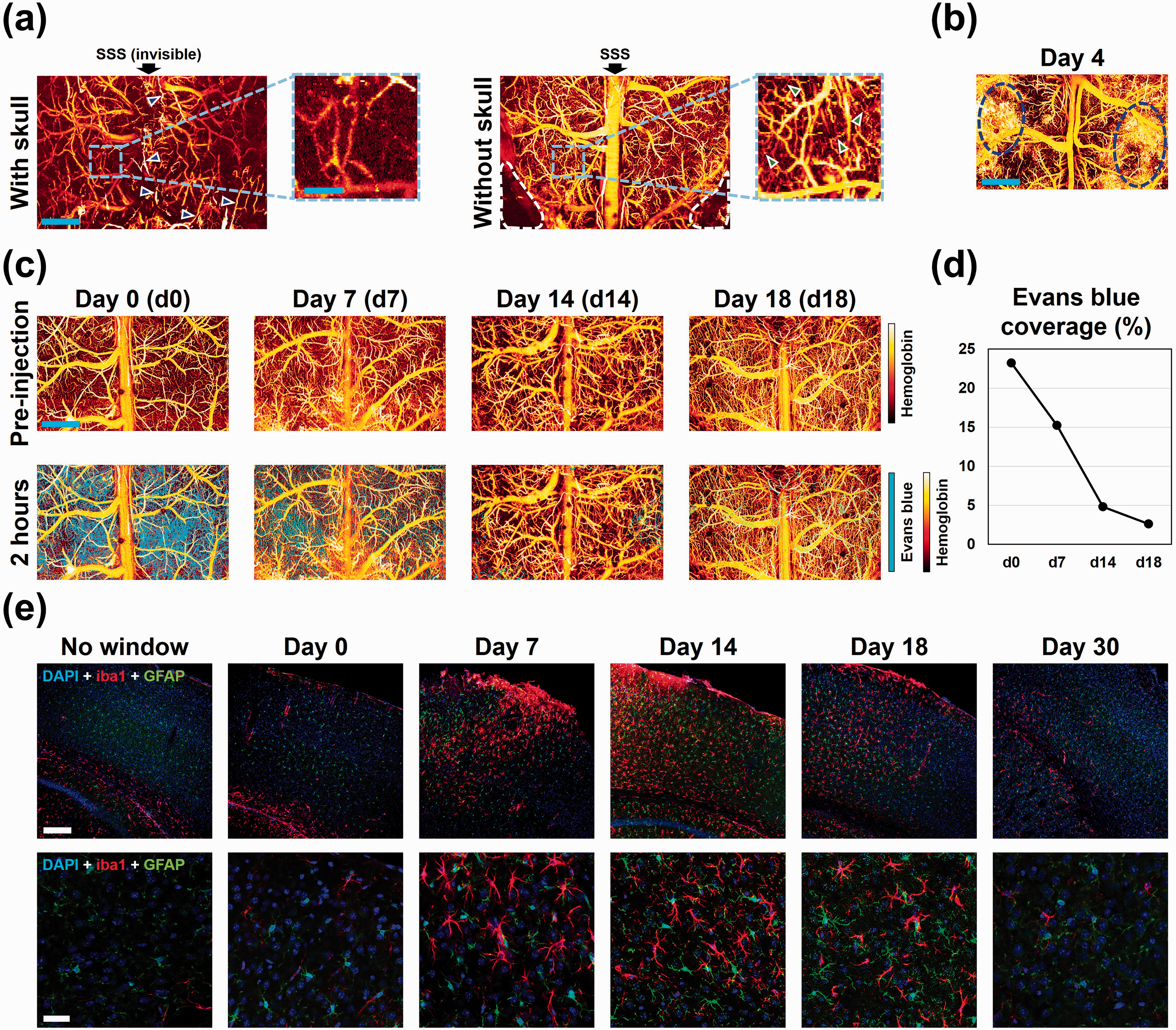
Cranial window characterization. (a) PAM of cerebrovascular structure through the intact skull (left)
Cranial window induces BBB breakdown and microgliosis that recover over time
To guide the proper use of the dual-transparency cranial window for longitudinal imaging, the vascular and inflammatory responses of the brain to the window installation, as well as their recovery over time, were comprehensively characterized.
After the window installation, hemorrhage was occasionally observed within the first few days (circled regions in Figure 2(b)). However, it typically disappeared over the ensuing week and none was visible after 14 days (
Also, the time course of the brain inflammatory response to the window installation was examined using GFAP and Iba1-1 immunohistochemistry analysis to visualize astrogliosis and microgliosis, respectively. As shown in Figure 2(e), an acute activation of astrocytes and microglia was observed on the day of window installation (
Cranial window enables high-quality cortex-wide awake-brain imaging
As previously demonstrated, 5 awake-brain PAM imaging without the use of anesthesia is crucial for objective quantification of cerebral hemodynamics and oxygen metabolism. Thus, our design of the cranial window took into consideration of the head-restraint requirement. To test the utility of the cranial window for high-quality, wide-field, awake-brain imaging, a side-by-side comparison of the imaging performance under wakefulness and anesthesia was carried out in five window-carrying mice without any other intervention.
As shown in the cortex-wide view (Figure 3(a)) and regional zoom-in view (Figure 3(b)), the multi-parametric images sequentially acquired in the same mouse brain under the awake condition and isoflurane anesthesia presented similar high qualities with clearly resolved microvascular structures. More importantly, anesthesia-induced hemodynamic changes were observed in the bilateral cortex, including increased blood flow speed and venous sO2, which echoed our previous observation in a much smaller cortical region (2.5 × 2.5 mm2) through a thinned-skull window.
5
Then, vessel segmentation was applied to extract individual trunk vessels (arteries and veins were labeled in red and green in Figure 3(c), respectively) for quantitative analysis. The segmentation-based analysis showed that, under general anesthesia, the average diameters of arteries and veins increased by 11 µm (25.7%, p < 0.0001) and 5 µm (8.4%, p < 0.05) compared to the baseline values, respectively (Figure 3(d)). While the arterial sO2 remained unchanged under anesthesia (
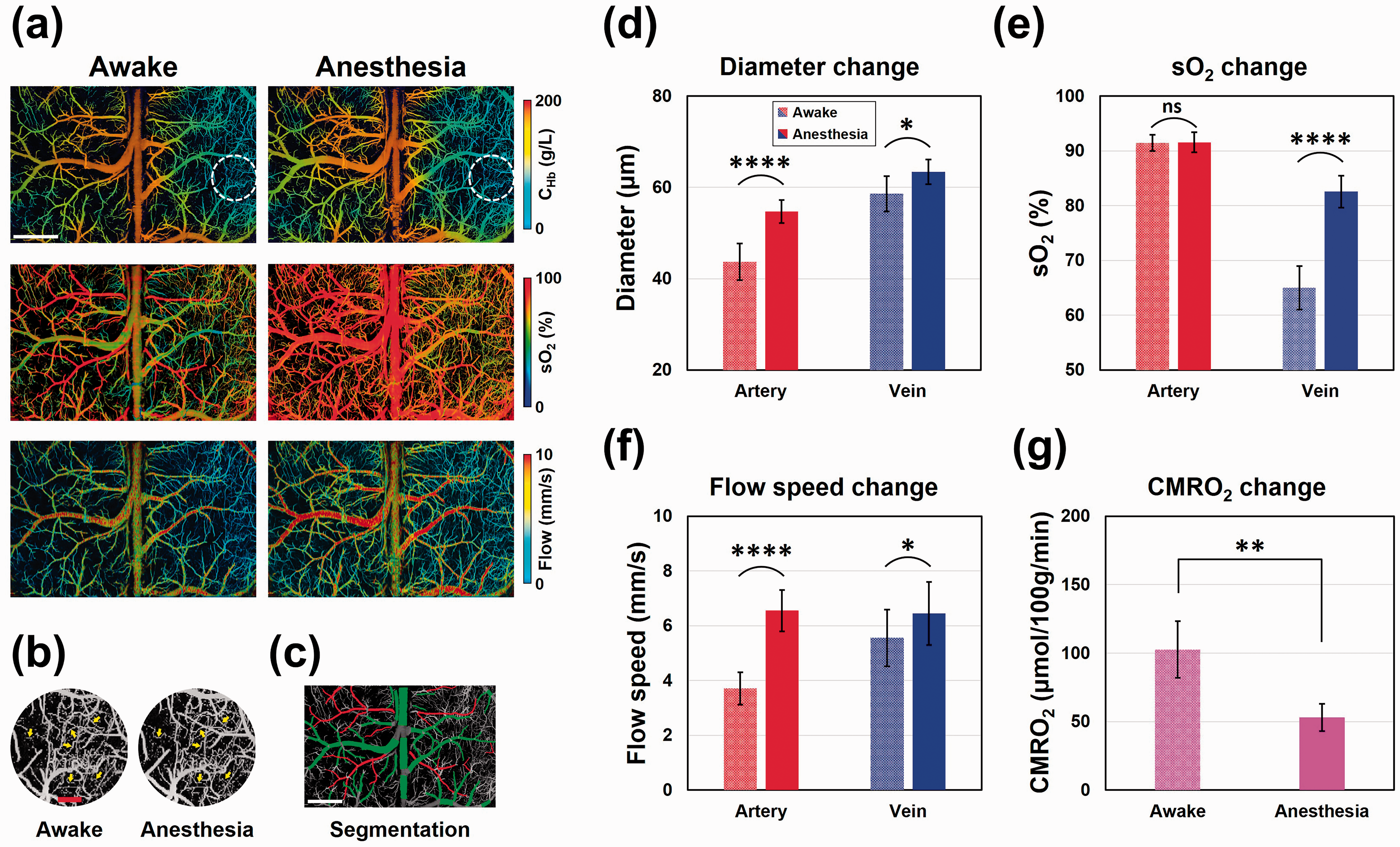
Cranial window enables awake-brain imaging. (a) Multi-parametric PAM images of total concentration of hemoglobin, oxygen saturation of hemoglobin (sO2), and blood flow speed acquired in the same mouse brain under wakefulness (left) and anesthesia (right). Scale bar: 1 mm. (b) Zoom-in views of the white dashed-circle region in (a), showing clearly resolved microvessels. Yellow arrows: capillaries. Scale bar: 200 µm. (c) Twenty-nine arterial segments and 60 venous segments extracted by vessel segmentation. Scale bar: 1 mm. (d–g) Quantitative analysis of anesthesia-induced changes in vessel diameter, sO2, blood flow speed, and cerebral metabolic rate of oxygen (CMRO2). Data are presented as mean ± standard deviation (n = 5). *p < 0.05; **p < 0.01; ****p < 0.0001.
Cranial window enables longitudinal imaging with consistent performance
Besides the cortex-wide coverage, another key feature of the present cranial window is the long lifetime. To determine how long the window quality can be maintained for high-resolution, multi-parametric imaging, a longitudinal monitoring study was performed in window-carrying mice without any other intervention.
As shown in Figure 4(a), both the multi-parametric images acquired by PAM and the photos taken by the stereo microscope showed high-quality and consistent results throughout the 22-week monitoring, with clearly resolved microvasculature shown in the PAM images. No signs of dura thickening or bone regrowth were observed. Moreover, the vessel segmentation-aided quantitative analysis of 13 feeding arteries and 27 draining veins (labeled in red and green in Figure 4(b), respectively) revealed that there were no significant changes in the average vessel diameter, CHb, sO2, and blood flow speed at any time point throughout the 22-week period, compared to the initial measurements taken at week 2 (Figure 4(c)).
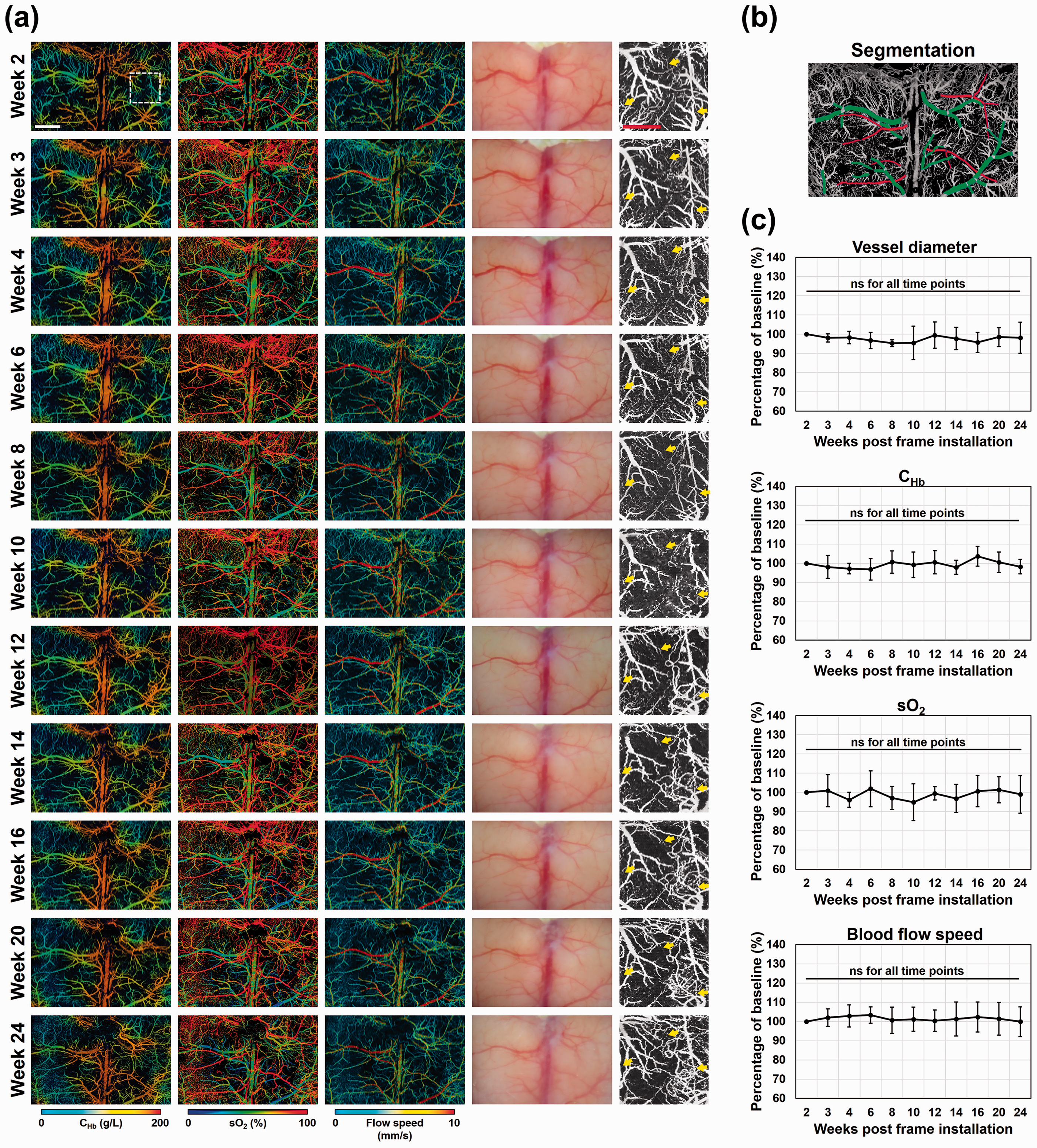
Longitudinal PAM of the awake mouse brain through the cranial window. (a) Time-lapse images acquired by multi-parametric PAM (Column 1–3) and a stereo microscope (Column 4) through the cranial window over an extended period of 22 weeks. Scale bar: 1 mm. Column 5: zoom-in views of the white dashed-rectangle region in Column 1. Yellow arrows: capillaries. Scale bar: 500 µm. (b) Feeding arteries (n = 13) and draining veins (n = 27) in the region of interest identified and isolated by vessel segmentation. (c) Quantitative analyses of the cerebrovascular structure and function measured at different time points over the 22-week period, showing no changes in the vessel diameter, total concentration of hemoglobin (CHb), oxygen saturation of hemoglobin (sO2), and blood flow speed. Data are presented as mean ± standard deviation.
Longitudinal multi-parametric PAM of photothrombotic stroke
After the demonstration of its long-term monitoring capability, this PAM technique was used to study chronic changes in the cerebrovascular structure, function, and associated oxygen metabolism during brain recovery from ischemic injury in a mouse model of ischemic stroke.
As shown in Figure 5(a), high-resolution, multi-parametric images of the CHb, sO2, and blood flow speed were acquired before (
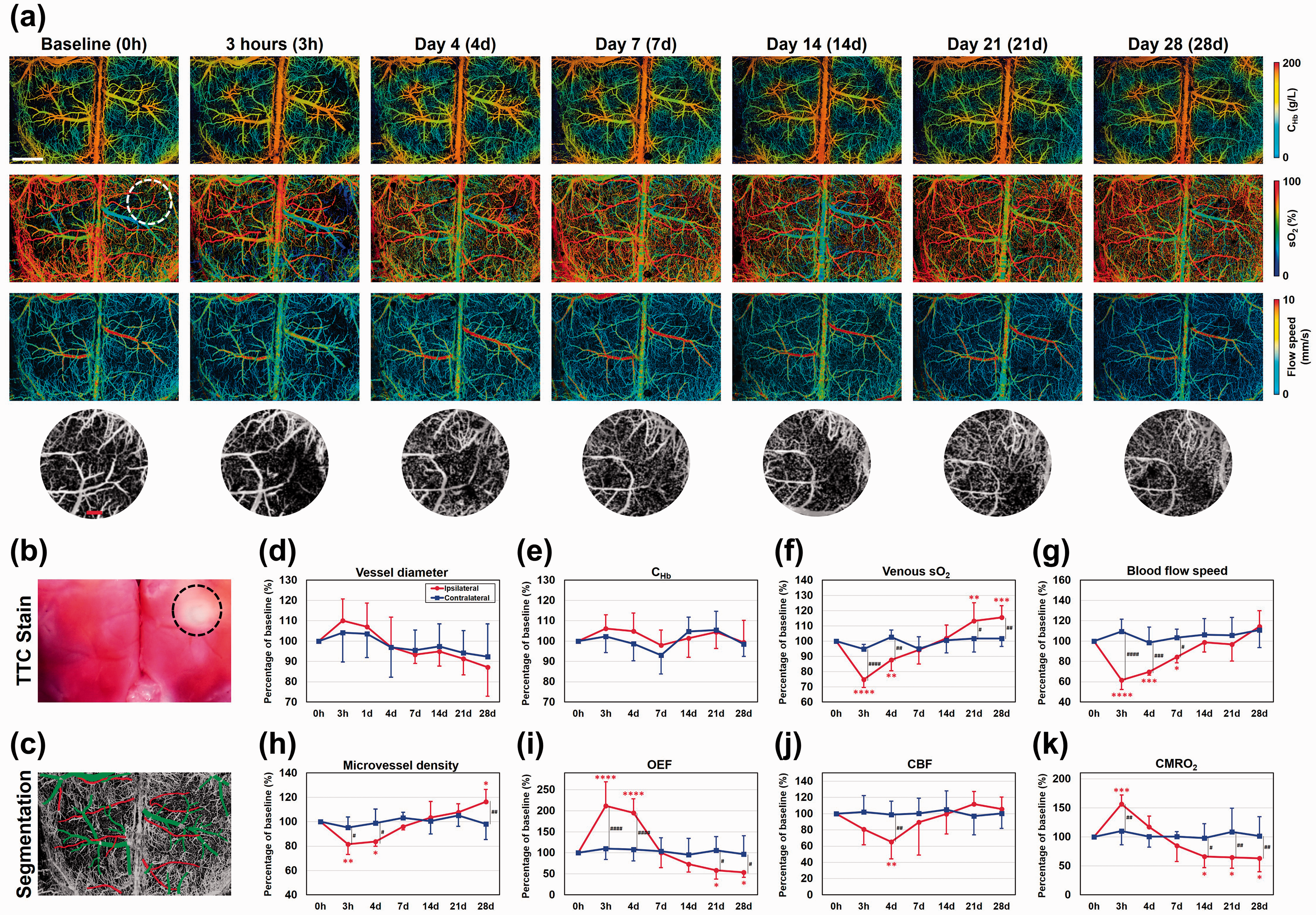
Longitudinal multi-parametric PAM of photothrombotic stroke. (a) Time-lapse, multi-parametric PAM images acquired throughout the acute (3 hours), subacute (day 4), and chronic (day 7, 14, 21, and 28) stages of photothrombotic stroke. Dashed circle: region treated with 543.5-nm laser light for photothrombosis. Scale bar: 1 mm. Row 4: zoom-in views of the white dashed-circle region. Scale bar: 200 µm. (b) TTC analysis of the brain at the conclusion of the PAM monitoring, showing tissue infarction highlighted by the dashed circle. (c) Feeding arteries and draining veins in the region of interest identified and isolated by vessel segmentation. (d–k) Time-dependent vascular, hemodynamic, and oxygen-metabolic responses of the ipsilateral and contralateral cortices to the stroke, including vessel diameter, total concentration of hemoglobin (CHb), venous oxygen saturation of hemoglobin (sO2), blood flow speed, blood-perfused microvessel density, oxygen extraction fraction (OEF), cerebral blood flow (CBF), and cerebral metabolic rate of oxygen (CMRO2). Data are presented as mean ± standard deviation (n = 6). *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
Structurally, no change in the trunk vessel diameter was observed in the ipsilateral cortex throughout the 28-day monitoring (Figure 5(d)), but significant changes in the microvessel density were observed (Figure 5(h)). Specifically, photothrombosis-induced microvascular occlusion led to reduced density of blood-perfused microvessels in the acute (81.5% of the baseline at 3 hours,
Discussion
Longitudinal, high-resolution imaging of the mouse brain represents a challenge for optical microscopy, due to increasing opaqueness of the skull with aging.
12
To address this challenge, transparent imaging windows have been developed to offer high-quality optical access to the underlying brain tissue. Owing to the minimally invasive nature, the thinned-skull window has been widely used, allowing repeated microscopic imaging in the first few days after the initial surgery. However, due to bone regrowth, longer-term monitoring beyond 2–3 days requires re-thinning of the skull, which is difficult to perform repeatedly because optical properties of the skull may gradually deteriorate with repeated thinning and eventually become irretrievable.
13
Alternatively, part of the skull can be surgically removed and replaced by transparent materials (
By combining the dual-transparency cranial window and multi-parametric PAM, our work achieves longitudinal imaging of cerebrovascular structure and function at the microvascular level, bridging the apparent gap between long-term large-scale recording of neuronal activity 15 and attendant hemodynamics and metabolism. The reported technique has three key features: long-term monitoring capability (22 weeks), cortex-wide spatial coverage (5 × 7 mm2 as shown in Figure 2(a)), and accessibility to the awake mouse brain. Moreover, it is worth noting that the dual-transparency cranial window is also compatible with fluorescence imaging. As shown in Supplementary Figure 2, our TPM system can clearly resolve the microstructure of GFP-labeled microglia in the live mouse brain through the window.
In the longitudinal study, the longest PAM monitoring on the same mouse brain to date was demonstrated, showing high and consistent imaging performance throughout the 22-week period. We tend to attribute the longevity of the cranial window to several key components in the surgical preparation, which prevent dura thickening and bone regrowth. Dura thickening is typically caused by the lack of pressure from the cranial window. 25 By taking advantage of the gelatin texture of the agarose, we were able to apply gentle pressure on the dura underneath the cranial window to prevent possible thickening. To prevent bone regrowth, we applied epoxy resin along the edge of the skull window, which proved to be effective.
The stroke study was carried out in the bilateral cortex under awake condition, which not only enabled the use of the contralateral side as an intra-animal control but also eliminated the neuroprotective effect of anesthesia.
26
In contrast to the unchanged vascular structure, function, and metabolism in the contralateral cortex throughout the entire monitoring period (blue curve in Figure 5(d) to (k)), significant and time-dependent changes in microvessel density, OEF, CBF, and CMRO2 were observed on the ipsilateral side across the acute, subacute, and chronic stages of photothrombotic stroke (red curve in Figure 5(d) to (k)). Our observations agree with previous reports, but with new, important insights into blood oxygenation and metabolism. Specifically, the acute declines in the flow cross-sectional area and vessel area density one day after photothrombotic stroke followed by a recovery by day 7, as observed by optical coherence tomography,
27
echo our observations on the flow speed (Figure 5(g)) and microvessel density (Figure 5(h)) in the acute and subacute stages. The decline in microvessel density was likely due to photothrombosis-induced impairment of blood perfusion and/or microvessel compression by tissue edema. By extending the monitoring to the chronic stage of stroke, our study revealed a significant rebound of the microvessel density surpassing the baseline in the ipsilateral cortex (Figure 5(h)), indicating possible angiogenesis during stroke recovery as reported before.28,29 Interestingly, despite the recovery of CBF by day 7 (Figure 5(j)), long-lasting deficits in OEF and CMRO2 were observed by PAM in the chronic stage (Figure 5(i) and (k), respectively). These observations, together, indicate that part of the ischemic tissue proceeded to permanent infarction,
30
which is confirmed by the TTC analysis at the conclusion of the longitudinal PAM monitoring (Figure 5(b)). It is worth noting that, to shorten the image acquisition time, the field of view was intentionally reduced from 7 × 5 mm2 to 5.5 × 3.5 mm2 in the stroke study. Even though, it took ∼50 minutes to acquire a set of multi-parametric images. The lengthy acquisition might be problematic when recording the acute responses of the brain to stroke (
For longitudinal imaging using the cranial window, it is important to allow for at least two weeks of recovery from craniotomy and window installation. It has been shown in our studies that acute micro-hemorrhage and increased cerebrovascular permeability can be induced by the window installation and are typically attenuated in the first couple of weeks (Figure 2). In addition, for head-restrained awake-brain imaging, it is necessary to train the animal for a few days to attenuate the restraint-induced stress. After two weeks of surgical recovery and head-restraint acclimation, the animal should be ready for longitudinal cerebrovascular imaging. However, it is worth noting that the window-induced non-vascular responses may last as long as four weeks (Figure 2(e)), including inflammation,14,32,33 activation of astrocyte and microglia, 34 and the loss of dendritic spines in neurons. 35 Thus, for longitudinal studies beyond the cerebral vasculature, it is necessary to extend the recovery time to four weeks.
In summary, we have demonstrated longitudinal (22 weeks), cortex-wide PAM imaging of vascular structure, function, and oxygen metabolism in the awake mouse brain with the use of a self-developed, dual-transparency (
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211034096 - Supplemental material for Longitudinal cortex-wide monitoring of cerebral hemodynamics and oxygen metabolism in awake mice using multi-parametric photoacoustic microscopy
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211034096 for Longitudinal cortex-wide monitoring of cerebral hemodynamics and oxygen metabolism in awake mice using multi-parametric photoacoustic microscopy by Vincent M Sciortino, Angela Tran, Naidi Sun, Rui Cao, Tao Sun, Yu-Yo Sun, Ping Yan, Fenghe Zhong, Yifeng Zhou, Chia-Yi Kuan, Jin-Moo Lee and Song Hu in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Institutes of Health (NS099261), National Science Foundation (2023988), and American Heart Association (15SDG25960005).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
SH, VS, and RC conceived the study. VS, AT, NS, RC, TS, YYS, PY, FZ, and YZ performed the experiments and data analyses. VS, AT, NS, PY, CYK, JML, and SH wrote the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
