Abstract
Cerebrovascular reactivity (CVR) deficits in adolescents with concussion may persist after resolution of neurological symptoms. Whether or not CVR deficits predict long term neurological function is unknown. We used adolescent mice closed head injury (CHI) models (54 g, 107 cm or 117 cm drop height), followed by blood oxygenation level dependent (BOLD)-functional MRI with CO2 challenge to assess CVR and brain connectivity. At one week, 3HD 107 cm mice showed delayed BOLD responses (p = 0.0074), normal striatal connectivity, and an impaired respiratory rate response to CO2 challenge (p = 0.0061 in ΔRmax). The 107 cm group developed rotarod deficits at 6 months (p = 0.02) and altered post-CO2 brain connectivity (3-fold increase in striatum to motor cortex correlation coefficient) by one year, but resolved their CVR and respiratory rate impairments, and did not develop cognitive or circadian activity deficits. In contrast, the 117 cm group had persistent CVR (delay time: p = 0.016; washout time: p = 0.039) and circadian activity deficits (free-running period: 23.7 hr in sham vs 23.9 hr in 3HD; amplitude: 0.15 in sham vs 0.2 in 3HD; peak activity: 18 in sham vs 21 in 3HD) at one year. Persistent CVR deficits after concussion may portend long-term neurological dysfunction. Further studies are warranted to determine the utility of CVR to predict chronic neurological outcome after mild traumatic brain injury.
Introduction
Concussions, defined as mild traumatic brain injuries (mTBIs) with neurological symptoms, are common in the United States, 1 with children and adolescents accounting for roughly half of sports related concussions. 2 Recent studies suggest protracted recovery of cognitive and sleep function in concussed youth athletes, 3 an association between contact sports and dementia, and earlier onset of cognitive impairment in professional athletes when contact sports are begun at younger ages.4,5 Growing awareness of the potential dangers of repeated mTBI during childhood and adolescence have resulted in rule changes in youth sports designed to limit the number of head impacts. While avoiding athletics until clinical symptoms resolve is currently the mainstay of therapy, 6 it is unknown whether this strategy prevents chronic neurological sequelae of mTBI, in part because validated biomarkers to prognosticate long term outcomes are lacking.7–11
Cerebrovascular reactivity (CVR) changes have been identified as a physiological biomarker of severe12,13 and mTBI.14–19 Clinical studies examining CVR using fMRI/BOLD show changes in the magnitude and timing of the blood oxygen level dependent (BOLD) responses to inhaled CO2 in concussed adults and adolescents.17,19–22 In the largest fMRI study to date of adolescents with mTBI and post-concussion symptoms, abnormal CVR responses to CO2 challenge were present at one week and 4 months after injury, even after most of the post-concussion symptoms had resolved, suggesting persistent physiological brain injury. 23 It remains unknown, however, whether persistent CVR abnormalities may predict development of neurological sequelae later in life- a key question that has implications for return to play after mTBI in youth sports that is difficult to address in humans because prospective studies involving human subjects would take decades to complete. 9
Here, we used two models of repetitive mild closed head injury (CHI) in adolescent mice: One that we previously reported induces long term cognitive deficits (54 g, 117 cm drop height) 24 and one that does not (54 g, 107 cm) to test the hypothesis that that persistent CVR deficits (assessed by fMRI/BOLD with inhaled CO2 challenge) would be associated with development of chronic neurological sequelae.
Methods
Mice
Adolescent (day of life 38), male wild-type (WT) C57/BL6J mice (Jackson Laboratories, Bar Harbor, ME), were used for experiments. Females were not used because they weigh significantly less than males and would have differences in injury severity that cannot be easily estimated and controlled by scaling. Mice were housed for 12-hour day/night cycles in laminar flow racks in a temperature-controlled room. Food and water was given ad libitum. All procedures were approved by the Massachusetts General Hospital Institutional Animal Care and Use Committee and performed in accordance with the NIH Guide for Care and Use of Laboratory Animals. Studies were performed according to ARRIVE guidelines and data were obtained by investigators blinded to study groups. Mice were randomized to sham or closed head injury in blocks of 4 and housed in the same cage.
Closed head injury (CHI) model
The CHI model was used as previously described with minor modifications. 25 Our goal was to use repetitive CHI models that induce cognitive and circadian deficits, depending on injury severity, to examine their associations with CO2 reactivity. Our prior study 24 showed that spacing injuries one week apart did not produce cognitive deficits in the 117 cm drop height model, hence we used three daily injuries that remained within the 14-day period of mouse adolescence. The 3HD paradigms were intended to model a number and severity of repetititve mild TBIs in human adolescents that might result in long-term neurological sequelae, with the understanding that no mouse model replicates the loading conditions, brain vulnerability, timing, and genetic and environmental variance inherent in human mild TBI. Mice were anesthetized with 2.5% Isoflurane (Anaquest, Memphis, TN) for 90 seconds in a 70% N2O-30% O2 Fluotec 3 vaporizer (Colonial Medical, Amherst, NH). Mice were placed on a Kimwipe (Kimberly-Clark, Irving, TX) and grasped by the tail, and the head was positioned under a hollow tube (diameter 10 mm). A metal bolt (54 g) was dropped either 107 cm (42”) or 117 cm (46”) onto the dorsal aspect of the skull directly above the right (day 1, 3) or left (day 2) ear between the coronal and lambdoid sutures. The head readily penetrated the Kimwipe following impact in the anterior-posterior plane. Sham injured mice were subjected to anesthesia without weight drop. Injured mice were recovered in room air in their cages. Three-hit (3HD) mice were injured once daily for three consecutive days. Latency to spontaneous righting from a supine position was recorded for all mice on day 1.
Morris water maze
On each day of testing, mice were acclimatized to the room for at least 30 minutes. Mice were first tested in a Morris Water Maze (MWM) task 1 month after the last injury and again in a reverse MWM paradigm at 6 and 12 months. The MWM was performed as previously described with minor modifications. 26 Each mouse was subjected to 7 hidden platform trials (1-2 trials per day). Probe trials were performed 24 h after the last hidden platform trial by allowing the mice to swim for 30 s with the platform removed and recording the time spent in the target quadrant. For each reverse MWM, the platform location was moved to a unique quadrant and mice were subjected to 4-7 hidden platform trials (1-2 trials per day).
Rotarod
At 1, 6, and 12 months after injury, mice were subjected to rotarod testing. Mice were tested for three days with three trials per day on an accelerating rotarod (0-40 revolutions per minute over 200 seconds). Time on the rotarod and maximum speed attained before falling were recorded for each mouse.
Activity monitoring
Mice were single cage housed with running wheels (Med Associates, Inc., St. Albans, VT) for one week in 12 h day/night cycles and then for two weeks in 24 h darkness. Activity was monitored electronically with running wheels (revolutions per minute) and data binned every minute. Free running periods, amplitudes, and phase of peak activity were calculated from the dark period by χ2 periodogram in ClockLab software (Actimetrics, Wilmette, IL).
Histology
Mice were anesthetized, transcardially perfused with PBS followed by 4% paraformaldehyde (PFA) and brains were post-fixed in 4% PFA at 4 °C for 24 h, cryoprotected in 30% sucrose for two days, and snap frozen in isopentane/dry ice and stored at -80°C. Brains were sectioned in the coronal plane at 500 um intervals from anterior to posterior. Cryostat sections (12 um) were mounted on poly-L-lysine-coated slides. For immunohistochemistry, sections were washed in PBS, blocked in 5% normal goat serum in PBS for 1 h, and incubated overnight at 4 °C with rabbit anti-Iba1 antibody (1:200; Wako Pure Chemical Industries Ltd, Osaka, Japan) or rabbit anti-GFAP (Sigma, St. Louis, MO), rat anti-CD31 antibody (1:100; BD Pharmingen) followed by anti-rabbit IgG-Cy3 conjugated secondary antibody (1:300; Jackson ImmunoResearch, West Grove, PA) or anti-rat-IgG-Cy3 conjugated secondary antibody (1:300; Jackson ImmunoResearch). Slides were analyzed using a Nikon Eclipse Ti-S fluorescence microscope (Micro Video Instruments; Avon, MA) using the appropriate excitation/emission spectra.
Assessment of cell counts
Coronal sections were stained and analyzed by a blinded investigator using ImageJ software (NIH). Brain regions assessed were ×200 microscopic fields (1100 × 1100 um) in cortex (n = 2 fields/section, 4 sections per mouse = 8 fields/mouse), hippocampus (16 fields in 4 sections per mouse), corpus callosum (12 fields in 4 sections per mouse), thalamus and striatum (16 fields in 4 sections/mouse) for GFAP and IBA1+cells; in cortex, hippocampus and striatum (6 fields/mouse) for CD31+ cells. Cell count data for each mouse were the average of the total brain fields counted.
MRI
MRI experiments were carried out in a 9.4 T Bruker scanner (Bruker BioSpin, Billerica, MA) with a quadruple volume transmit coil, and a 4-channel surface receiving coil. mTBI mice were scanned at 1 week (acute group) and 1 year (chronic group) after three daily injuries in adolescence (3HD) or sham injury. MRI was carried out under isoflurane anesthesia in O2 (30%) enriched medical air. Respiration was measured over the whole scanning session using MR-compatible small animal monitoring system (Model 1025, SA Instruments, Inc., NY, USA). The level of isoflurane was adjusted dynamically to maintain respiratory rate in the range of 90 to 120 breaths per minute. Body temperature was monitored with a rectal probe. A small gas sampling tube was placed in the nose cone to measure the partial pressure of CO2 (PCO2) in the breathing circuit via a calibrated CO2 analyzer with a CO2 sensor (SprintIR-W, CO2Meter, Inc., FL, USA). Real-time recordings of respiration were exported to a Powerlab (AdInstruments, Inc., CO, USA) using a signal breakout module (SA Instruments, Inc., NY, USA) and synchronized with the PCO2 measurements.
Diffusion tensor images (24 diffusion directions, B = 1200 s/mm2) were acquired to assess structural brain damage. Diffusion data were processed for B0/T2-weighted, diffusion weighted (DTW), and apparent diffusion coefficient (ADC) images using diffusion tool kit (http://trackvis.org/dtk). Changes in brain connectivity and cerebrovascular reactivity were assessed by resting state fMRI (rsfMRI) and fMRI with hypercapnic challenges, respectively. Each mouse had a basal rsfMRI (10 min), followed by fMRI with hypercapnia challenges, followed by post hypercapnia rsfMRI (10 min). rsfMRI and fMRI scans were repetitive EPI images (TR 1 sec, TE 20 ms, field of view of 2.0 cm and twenty-two 0.5 mm slices, in-plane resolution 0.25 mm × 0.25 mm). For rsfMRI, imaging and physiological data were acquired when the animals were at rest and had free breathing of medical air through the nose cone on the breathing circuit. For hypercapnic challenge, the fraction of inspired CO2 was adjusted to produce steady-state conditions of normocapnia (medical air) and hypercapnia (7% CO2 balanced with medical air). The paradigm consisted of a two-minute baseline period (normocapnia), and 1-minute hypercapnia epochs followed by 2-minute epochs with medical air, repeated a total of 3 times. The total duration of the challenge lasted 11 minutes.
All functional images were registered to a standard mouse brain atlas.27–30 All rsfMRI/fMRI images were first pre-processed with standard fMRI analysis stream (time-slicing, despiking, motion correction, etc.) and then we used a generalized logistic regression model (GLM) to fit for kinetic responses to hypercapnic challenges, as described in literature,27–30 on a pixel by pixel basis. Region of interest (ROI) seed-based rsfMRI analysis was processed as previously described. 31 Non-ROI based rsfMRI analysis was also performed as it was challenging to segment detailed brain structures from mouse brains. For this, we simply reordered all voxels from the whole brain into a one-dimensional array and then clustered elements from similar brain structures together. We then performed cross-correlation of the 1-D array.
Hypercapnic BOLD data were first analyzed by GLM on a pixel by pixel basis.32 Respiratory rate in each mouse was maintained stable at baseline by adjusting the amount of isoflurane delivered, which could lead to variability in the absolute value of PaCO2 at baseline, and affect the degree of BOLD responses to hypercapnic challenge. To avoid this potential confound on the magnitude of the BOLD response, we focused on the delay in BOLD response in relation to the timing of CO2 challenge. This approach has been used by Poublanc et al. (2013) on patients with steno-occlusive cerebrovascular disease and Dodd et al. (2020) on adolescents with mild TBI.23,33 Since hypercapnia typically led to significant BOLD increases over the whole brain, and the BOLD differences in different brain structures tended to vary among animals, we performed time delay analysis for each hypercapnia epoch on the whole brain level, but not on individual brain structures. In addition to time delay analysis, CO2-fMRI data was also analyzed for functional connectivity using a one-dimensional array cross-correlation approach, as described in the rsfMRI analysis. We did not determine changes in the amplitude of the CBF response to CO2 because we did not have blood gas measurements of PaCO2 when the BOLD signal was acquired. This is because the amount of phlebotomy required causes hypotension in mice that would complicate interpretation of the BOLD signal.
Respiratory physiological data were analyzed using Matlab R2014a (Mathworks, Inc., Natick, MA, USA). A low pass filter of 3 Hz was applied to respiratory data to reduce noise and artifacts. End inspiration peak and end expiration trough was determined using custom Matlab functions and corrected on the graphical user interface incorporated in the function. Peaks and troughs on the respiration time series showing persistent artifacts were excluded from further analysis. The duration of each respiratory cycle was derived by subtracting the time in seconds of the 2 consecutive end inspiration markers, and the time series of respiratory rate in breaths per second was calculated by taking the reciprocal of the duration of respiratory cycles. Because large inter-individual variations of absolute respiratory rates existed among mice, the percent change of respiratory rate relative to the baseline value was derived for each 11-minute hypercapnic run. The mean respiratory rate for a period of 30 seconds immediately before the first epoch of hypercapnic challenge was chosen as the baseline. The percent change of maximum respiratory rate (Rmax) and minimum respiratory rate (Rmin) from baseline was measured for each cycle of hypercapnia-normocapnia on the time series of percent change of respiratory rate in the hypercapnic run. In each of the 10-minute resting state runs acquired before and after hypercapnic challenge, the coefficient of variation (i.e. standard deviation/mean) of respiratory rate (Rcv) was calculated. The percent change in coefficient of variation (ΔRcv) of respiratory rate of resting state runs before and after hypercapnic challenge was then derived for each animal.
Data analysis
For fMRI/BOLD and physiological (respiration rate) data, which were repeated measures because CO2 was administered in three epochs, and for behavior tests that were repeated over several trials (rotarod, MWM), data were analyzed using repeated measures ANOVA (group x time). Histological data, weight and righting time, circadian activity and ▵Rcv data were analyzed by t-test with comparisons made between respective sham and injured groups, rather than between 3HD 107 cm and 3HD 117 cm groups since these groups were not assessed altogether at the same time. For all RM ANOVA assessments, comparisons were made separately between sham and 3HD 107 cm groups and between sham and 3HD 117 cm groups. Since behavior and CVR analyses were performed in separate groups of mice to avoid confounds of prolonged anesthesia exposure on behavioral outcomes, it was not possible to analyze dose-response curves for behavioral and CVR outcomes. The primary outcomes for the study were delay in fMRI/BOLD response to CO2 challenge and behavioral data. Power calculations and sample sizes for these data, assessed by RM ANOVA, were based on our prior studies and effect sizes of 25-50%. 24 Normality for single measure data was assessed by Anderson-Darling (Figure 1(a) and (b)) and Shapiro-Wilk (Supplemental Figures 2 and 3) tests.
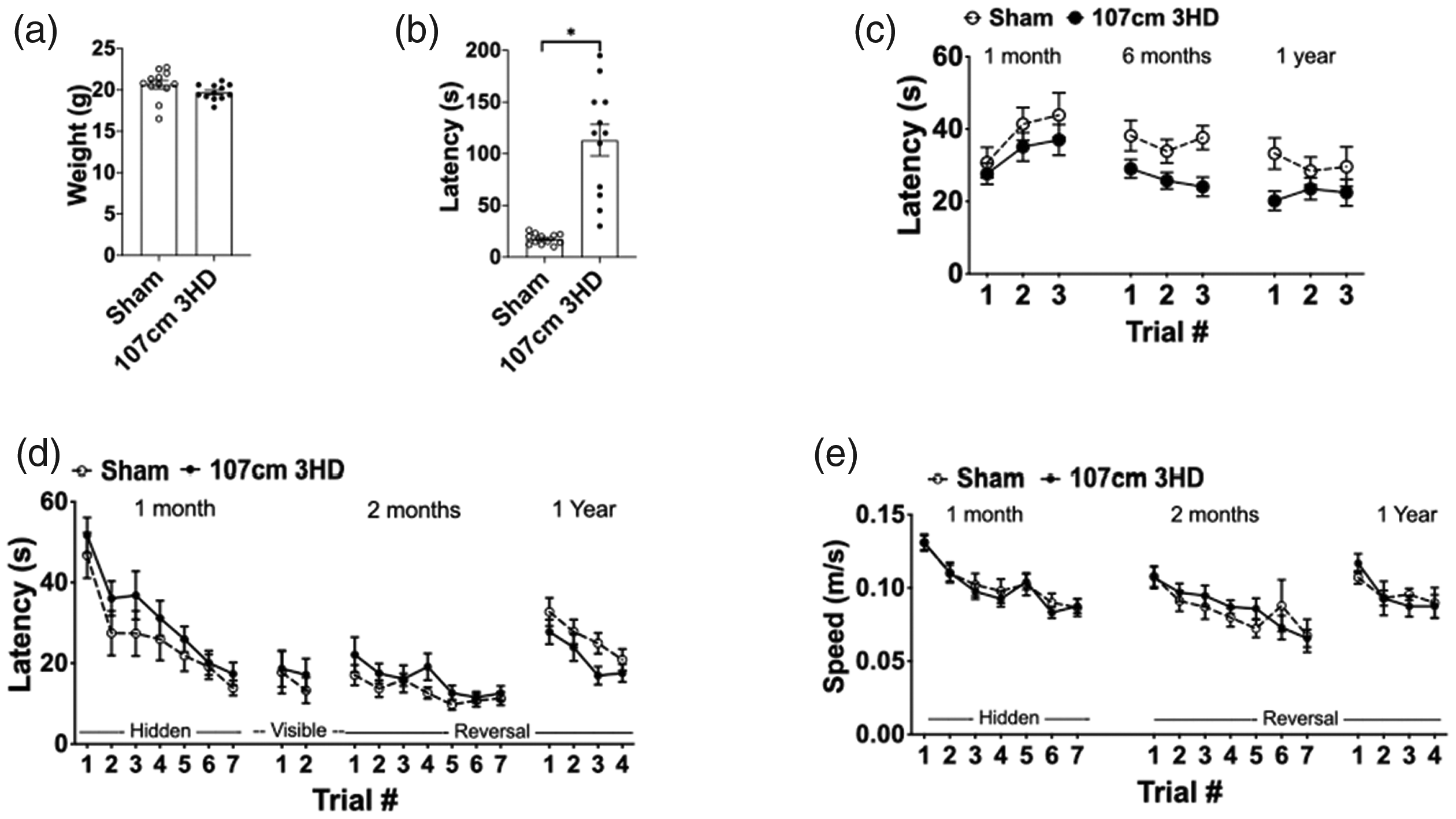
Physiological and behavioral outcomes in sham and 3HD (107 cm drop height) adolescent mice up to one year. (a) Body weights at day of life 38 were not different between sham and 3HD mice (p = 0.12, n = 12/group). (b) Time to righting after injury was significantly greater in 3HD vs. sham (*p < 0.0001, n = 12/group). (c) In mice followed for one year and used in fMRI analyses, rotarod performance in 3HD was similar to sham at 1 month (p = 0.33 for group) and 1 year (p = 0.11 for group) but significantly worse at 6 months (p = 0.02 for group, n = 15-16/group, RM ANOVA). (d) MWM performance (hidden platform trials) did not differ at any time between sham and 3HD (1 month, p = 0.23 for group; 2 months, p = 0.24 for group; 1 year, p = 0.05 for group, n = 15-16/group, RM ANOVA). Visible platform trials also did not differ between groups at 1 month (p = 0.65, n = 15-16/group, RM ANOVA). (e) Swim speed did not differ between groups at all time points examined (p = ns for group, n = 12-16/group, RM ANOVA).
Results
Body weight did not differ between sham and injured 107 cm groups at the time of CHI (Figure 1(a)). Righting time (assessed after the first CHI) was significantly greater in injured 107 cm vs. sham injured mice (Figure 1(b)). No structural brain damage was detectable by T2 weighted or diffusion weighted MRI in any mice. At two weeks after injuries, Iba-1+, GFAP+ cell counts and % area of CD 31+ cells did not differ between sham and 3HD 107 cm in any brain region examined (Supplemental Figure 2, 3). We previously reported increased righting time at time of injury and lack of histopathology at one year after injuries in the 117 cm 3HD model. 24 Together, these data confirm that both the 107 cm and 117 cm 3HD models recapitulate expected features of mild TBI.
Behavior data
In rotarod tests, 3HD 107 cm mice had performance similar to sham at 1 month but developed rotarod deficits by 6 months after injuries (Figure 1(c)). In 3HD 107 cm mice, performance in hidden and visible platform trials in the MWM was similar between sham and 3HD groups at 1, 2, and 12 months (Figure 1(d)). Likewise, swim speeds did not differ between groups at any time (Figure 1(e)). Because sleep disruption is a prominent feature of the post-concussive syndrome, we hypothesized that the chronic injury model could result in changes to circadian patterns of rest and activity. Interestingly, 3HD 117 cm demonstrated a lengthening of circadian period and a surprising increase in circadian amplitude with a concomitant change in the circadian phase of peak activity at one year post-injury whereas the 107 cm group did not (Figure 2). These data suggest that the more severe 117 cm model results in a re-programming of circadian behavior.
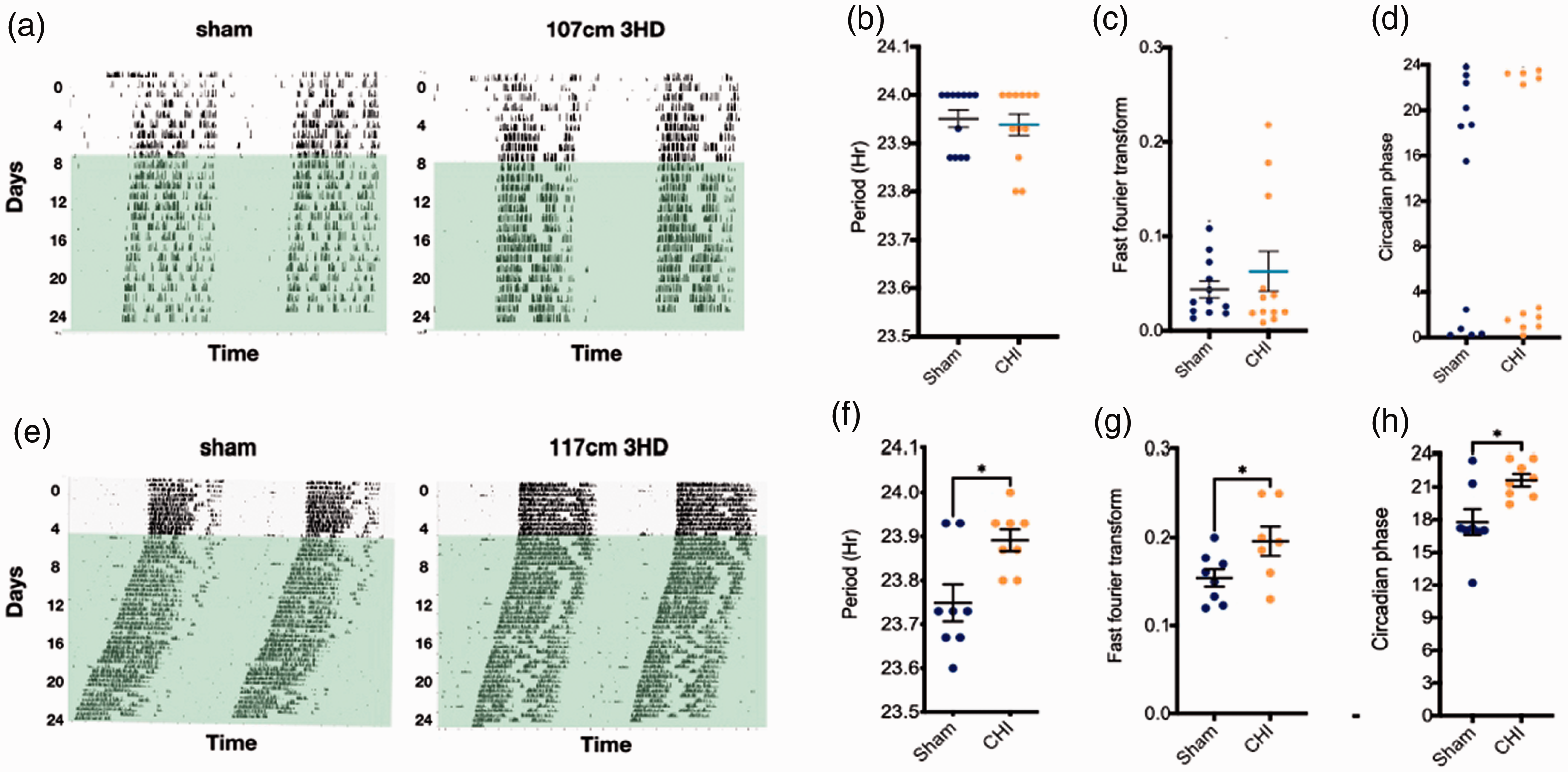
Circadian activity in sham and 3HD adolescent mice at one year after injury. (a) Representative double-plotted actograms of wheel running activity in indicated conditions in both 12 h/12h light/dark cycle and complete darkness (D/D), (b) free-running period (calculated during D/D), (c) amplitude, and (d) circadian phase of peak activity showing similar circadian activity between sham and 107 cm 3HD mice at 1 year after injury (p = ns, n = 12/group). (e) Representative double-plotted actograms, (f) free-running period, (g) amplitude, and (h) peak activity showing significant difference in circadian rhythm between sham and 3HD (117 cm drop height) mice at 1 year after injury (*p < 0.05, n = 7-8/group, t-test).
fMRI/BOLD responses to hypercapnia challenge
All animals had significant BOLD responses to the 3 hypercapnia challenges over the whole brain. However, the 3HD groups exhibited heterogenous response profiles across different brain structures. We thus performed time profile analyses covering major brain regions including caudate/putamen and thalamus. Figure 3(a) and (d) and Supplement Figure 4 shows changes in BOLD signal intensity during the 2nd half of each hypercapnia challenge and the following 30 s in medical air in representative animals at acute (1 week) and chronic (1 year) phases after injury in sham, 107 cm 3HD and 117 cm 3HD groups. At the acute stage, 107 cm 3HD mice showed significant delays in responding to hypercapnia challenges when compared to sham (Figure 3(b)) in the absence of differences in washout time (Figure 3(c)). These delays were not seen at one year in the 3HD 107 cm group vs. sham, however the 3HD 117 cm group had significant delays in response (Figure 3(e)) and washout (Figure 3(f)) to hypercapnia challenge at one year after injuries vs. sham.
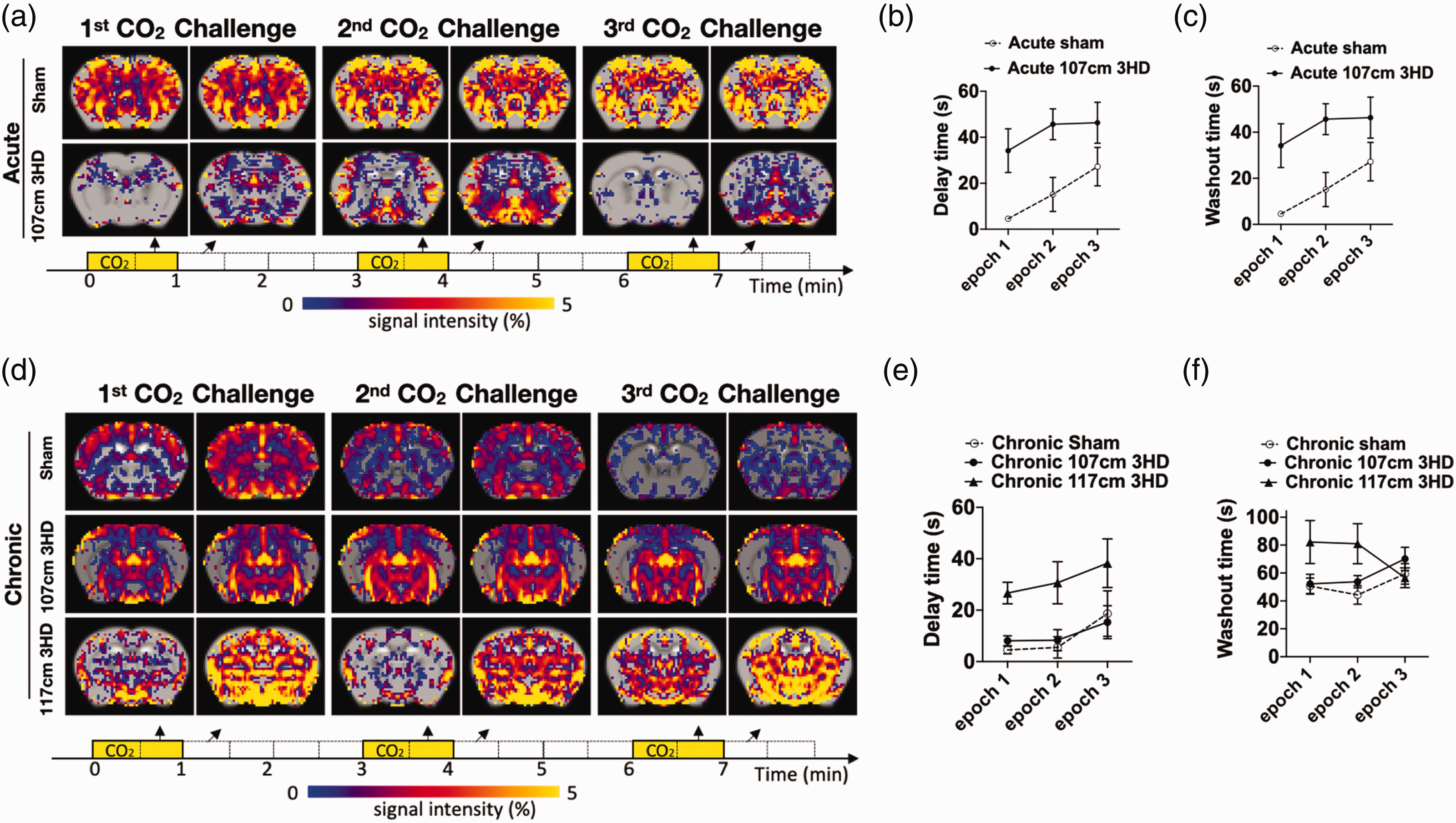
Blood oxygen level dependent (BOLD) response to hypercapnia challenge in sham and 3HD adolescent mice. (a) Representative image of Change in BOLD signal intensity (SI) in the last 30 seconds (arrows) of CO2 challenge (yellow boxes) and first 30 seconds (arrows) of air (white boxes) during each 60 s epoch of CO2 challenge in acute sham and acute 107 cm 3HD group. (b) Delay in onset of whole brain BOLD responses to hypercapnia challenge in 107 cm 3HD group was significantly greater than sham at 1 week after injury (p = 0.0074 for group, RM ANOVA, n = 6-9/group). (c) Washout time in 107 cm 3HD vs sham was similar at 1 week after injury (p = 0.22 for group, RM ANOVA, n = 6-9/group). (d) Representative image of change in BOLD SI in the last 30 seconds (arrows) of CO2 challenge (yellow boxes) and first 30 seconds (arrows) of air (white boxes) in sham, 107 cm 3HD and 117 cm 3HD group at chronic. (e) Delay time in onset of whole brain BOLD responses to hypercapnia challenge in sham, 107 cm 3HD and 117 cm 3HD groups at 1 year after injury. No differences were observed between sham chronic and 107 cm 3HD chronic groups (p = 0.83 for group, RM ANOVA, n = 6-10/group), however chronic 117 cm 3HD mice had significantly greater delay times vs. sham chronic (p = 0.016 for group, RM ANOVA, n = 6/group). (f) Washout time in 107 cm 3HD was not different vs. sham at 1 year after injury (p = 0.20 for group, RM ANOVA, n = 6-10/group), while 117 cm 3HD group had greater washout time compared to sham (p = 0.039 for group, RM ANOVA, n = 6-8/group).
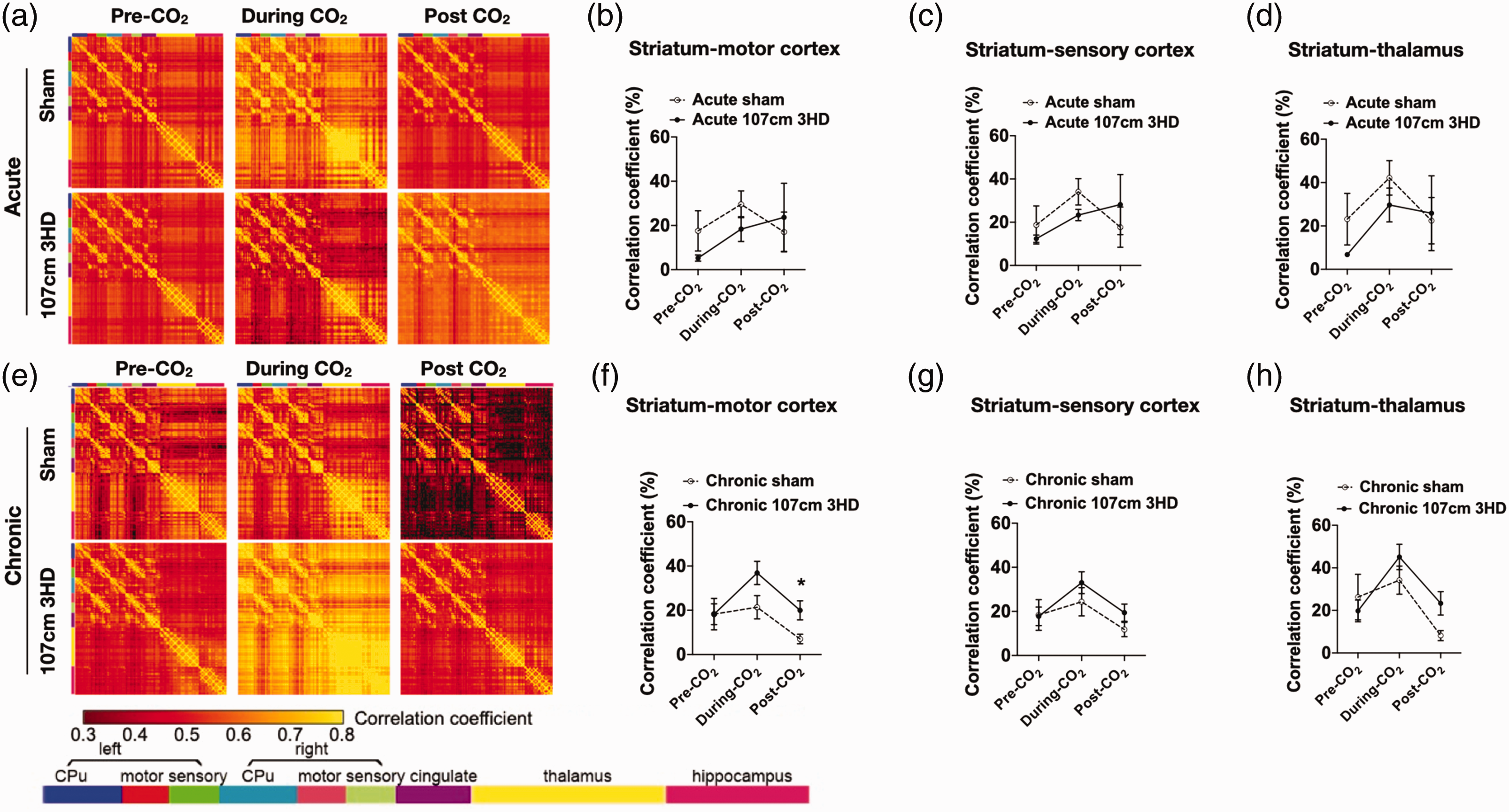
Functional brain connectivity before and after CO2 challenge in sham and 107 cm 3HD injured mice. (a) Map of group averages of cross-correlation matrix of major brain structures in sham and 107 cm 3HD groups at the acute phase (1 week) after injury. Cross correlation was carried out by rearranging 3 D brain voxels into a 1 D array, with similar brain structures clustered together. The one-dimensional array included Caudate/Putamen (CPu), motor cortex, sensory cortex, cingulate cortex, thalamus, hippocampus, and other brain regions (not shown). Those clustered brain structures were represented by color codes. Analysis of connectivity from striatum to (b) motor cortex (p = 0.60 for group), (c) sensory cortex (p = 0.84 for group), and (d) thalamus (p = 0.55 for group) before, during, and after hypercapnia challenge at the acute phase was not different between sham and 107 cm 3HD groups (n = 5-8/group, RM ANOVA). (e) Representative map of cross-correlation matrix of major brain structures at 1 year after 3HD (107 cm). 3HD had stronger connectivity vs sham from striatum to (f) motor cortex (p = 0.12 form group, *p = 0.046 for post CO2, Sidak’s multiple comparistions test), but similar from striatum to (g) sensory cortex (p = 0.34 for group) and (h) thalamus (p = 0.34 for group) before, during, and after hypercapnia challenge at one year (n = 6-15/group, RM ANOVA).
Functional connectivity
All rsfMRI and fMRI data for the 107 cm 3HD model were analyzed for functional connectivity by rearranging the 3-dimensional brain volume into a 1-dimensional array, with similar brain structures clustered together. Figure 4(a) and (e) show maps of the correlation coefficients between major brain structures for the pre-hypercapnia resting state fMRI/BOLD signals, hypercapnia fMRI/BOLD signals, and post-hypercapnia fMRI/BOLD signals in representative mice from the acute (Figure 4(a)) and chronic (Figure 4(e)) stages. For the sham and 3HD mice, there was fair cross-correlation among the homologous major brain structures in the left and right hemispheres. Figure 4(b) to (d) shows that correlation coefficients between striatum and motor cortex, sensory cortex, and thalamus did not differ between sham and acute 3HD 107 cm groups before, during or after the first CO2 epoch. However, In the chronic period, 3HD 107 cm mice had significantly increased correlation between striatum and motor cortex after the third CO2 epoch (Figure 4(f) to (h)).
Respiratory response to CO2 challenge
Averaged time series of the percent change in respiratory rate in response to three epochs of hypercapnic challenge in 3HD 107 cm and sham groups (acute and chronic stages) are depicted in Figure 5. A biphasic change in respiratory pattern was observed in each cycle of hypercapnia-normocapnia in all animals; the respiratory rate increased to a maximum during hypercapnia, began decreasing before the end of CO2 epoch, and reached a minimum (with undershoot) after each CO2 epoch (Figure 5(a) and (e)). The ΔRmax in 3HD 107 cm acute mice was significantly less than sham (Figure 5(b)). In contrast, no differences were observed in ΔRmax during the chronic period in sham and 3HD 107 cm groups (Figure 5(f)). ΔRmin was similar in sham and 3HD groups both at acute (Figure 5(c)) and chronic (Figure 5(g)) phases. No significant difference in Rcv was found between 3HD and sham groups at baseline prior to hypercapnic challenge. The ΔRcv (percentage change in coefficient of variation of respiratory rate at rest before and after hypercapnic challenge) also did not differ between groups (Figure 5(d) and (h)).
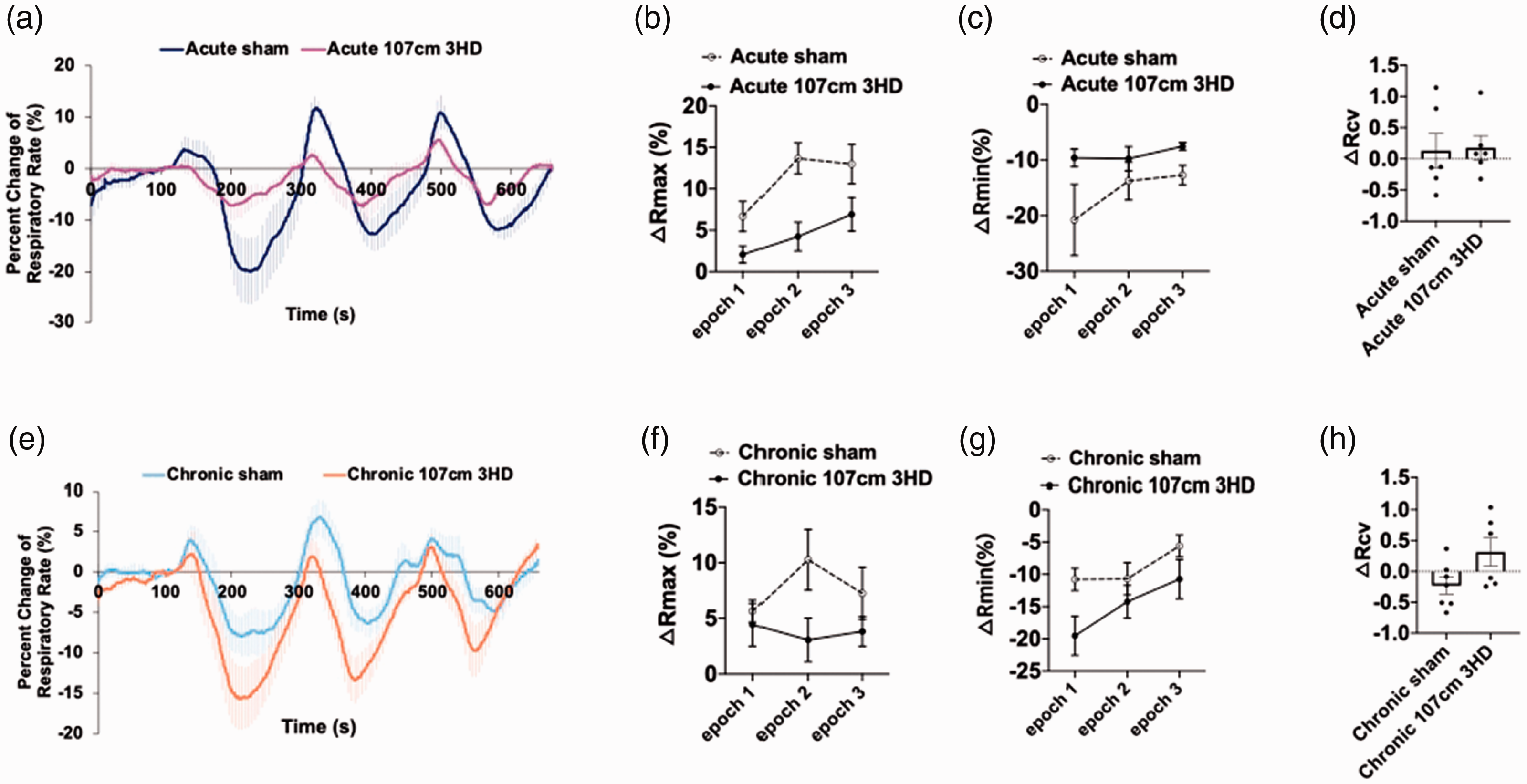
Time series of respiratory rate in response to hypercapnic challenge in sham and 3HD 107 cm injured mice. (a) Average biphasic change in respiratory rate at each epoch of hypercapnia. In sham and 3HD mice, the respiratory rate increased above baseline during hypercapnia but decreased below baseline during normocapnia after each CO2 epoch. (b) 3HD 107 cm group had lower ΔRmax (percent change of maximum respiratory rate) in the acute phase of injury vs sham (p = 0.0061 for group, n = 6/group, RM ANOVA). (c) 3HD 107 cm mice had no difference vs. sham in ΔRmin (percent change of minimum respiratory rate) after each CO2 epoch at the acute phase (p = 0.12 for group, n = 6/group, RM ANOVA). (d) ΔRcv (percent change in coefficient of variation of respiratory rate during resting state before and after hypercapnic challenge) was similar between sham and acutely injured 3HD 42” mice (p = 0.89, n = 6/group, t test). (e) Biphasic change of respiratory rate at 1 year after injuries. No differences were observed in (f) ΔRmax, (g) ΔRmin and (h) ΔRcv between sham and 3HD 107 cm groups at 1 year (p = ns, n = 6-7/group, RM ANOVA for f and g, t test for h).
Discussion
fMRI/BOLD with CO2 challenge has documented early and persistent CVR deficits in adolescents with mTBI, but the significance of these CVR deficits with regard to long term neurological prognosis is unknown. In an exploratory study, we showed that CVR deficits in the 3HD 107 cm model at one week resolved by one year and were not associated with cognitive or circadian activity deficits. However, CVR deficits were associated with circadian activity deficits in the 3HD 117 cm at one year, and we previously reported that this model induces persistent cognitive deficits as well. 24 The data suggest that persistent CVR deficits may contribute to an endophenotype of repetitive mTBI in adolescence that is associated with motor and cognitive deterioration in the chronic period, but whether or not the changes in circadian activity are adaptive or maladaptive is unknown and requires further investigation.
T2 weighted and diffusion tensor imaging revealed no structural brain damage at any time in 3HD mice, and IHC studies showed no gliosis or difference in vessel density at 2 weeks in the 107 cm model, validating a mild injury level as we previously reported for the 117 cm model. 24 However, despite a lack of histopathology and structural damage in the acute phase of injury, cerebrovascular function was an even more sensitive guage of response to 3HD 107 cm than MWM performance, suggesting that CVR deficits are particularly sensitive to mild TBI in rodents as well as in humans.17,19–23
In the largest reported longitudinal human study of CVR in adolescents with sports concussions, CVR deficits were detected concomitantly with neurological symptoms at one week and persisted at 4 months, despite resolution of most symptoms. 23 Another prior pilot study also suggested that CVR deficits may persist after clinical recovery in concussed adolescents. 16 However, the implications of CVR deficits with regard to long term neurological function in these studies remains unknown. Results from the current study suggest that the presence of CVR deficits after multiple concussions in adolescents may be associated with poor neurological outcome later in adulthood, at least in some functional domains. This possibility could have important epidemiological and clinical implications because of the high prevalence of mTBI in adolescents (estimated at one million cases per year in the US 34 ). On the other hand, resolution of CVR deficits was dissociated from persistent motor deficits assessed by rotarod performance in the chronic period after 3HD 107 cm, suggesting that unique cellular and molecular mechanisms drive these two injury phenotypes.
Another interpretation of the current data is that resolution of CVR deficits after repetitive mTBIs might predict normal cognitive and circadian function (but not necessarily striatal dependent motor function, indicated by rotarod testing) in adulthood, as adolescent mice in our study did not acquire altered MWM or circadian activity function for up to one year after 3HD 107 cm. This conclusion would be tempered by the caveats that the genetically inbred mice used herein do not represent the genetic diversity of humans with mTBI, and the fact that we have not modeled lifestyle and comorbid factors such as hypertension, diabetes, western diet, obesity, and others that could potentially interact with repetitive mTBI to produce cognitive deficits, even in the context of resolved CVR deficits. Moreover, it is also possible that our findings are unique to this inbred population of C57Bl/6J mice, and that CVR deficits may not predict poor outcomes as reliably in a genetically diverse human population. Our exploratory study is also limited by use of only one cognitive test in the chronic period, and no studies beyond one year- both of which would need to be addressed to more conclusively demonstrate possible negative predictive value of resolved CVR deficits on long term cognitive outcome. Nonetheless, data from the current study support the premise that increasing severity of mTBIs may lead to development of motor, cognitive and circadian deficits that are associated with failure to resolve CVR deficits.
Cerebral hemodynamics in healthy brain are regulated by three major processes: cerebral autoregulation, neurovascular coupling and cerebrovascular reactivity. Cerebral autoregulation ensures that cerebral blood flow (CBF) is maintained approximately constant across a wide range of mean arterial blood pressure. 35 Adult patients with mild and severe TBI in the chronic period demonstrate abnormal dynamic cerebral autoregulation to blood pressure changes that correlates with cognitive dysfunction. 36 Neurovascular coupling adapts regional CBF in response to neural activation; 37 patients with chronic TBI may also exhibit abnormal neurovascular coupling during performance of cognitive tasks.38,39 Cerebrovascular reactivity refers to the capacity of the entire cerebral vascular system to respond to a vasoactive stimulus, whereas cerebral autoregulation and neurovascular coupling are controlled at the level of small-vessel resistance. 40 Increase in arterial partial pressure of CO2 (PaCO2) normally causes vasodilation and increased CBF. Impaired CVR indicates damage to the cerebral blood vessels with resulting effects on CBF regulatory mechanisms, including CBF reduction and/or neurovascular uncoupling. 41 Studies in adults with severe TBI suggest that altered cerebrovascular reactivity to CO2 is an enduring injury phenotype detectable in the chronic stages,12,13 implicating persistent changes in cerebrovascular function. However, no data exist describing CVR changes in the chronic period after mTBI in human adolescents, and ours is the first experimental study that we know of to examine such changes in the chronic period using a preclinical repetitive concussion model.
The underlying pathophysiological bases for CVR deficits following mTBI remain largely undefined. 20 Hypercapnia is a powerful cerebral vasodilator and the normally strong positive relationship between PaCO2 and cerebral blood flow is impaired in neurodegenerative disease and aging, and predicts cognitive deficits.42,43 When CO2 reacts with carbonic anhydrase, protons accumulate outside brain cells, reducing extracellular pH that subsequently causes cerebral vasodilation,44–46 though the exact mechanisms that transduce pH signaling remain largely undefined. Recent studies implicate a role for neuronal acid-sensing ion channels (ASICs) that, when activated by extracellular H+, evoke Na+ and Ca2+ currents leading to vasodilation.47,48 Neuron-specific knockout of ASIC 1 A reduced cerebral vasodilation to elevated PaCO2, implying neuronal regulation of CO2 vascular reactivity. 49 ASICs are also involved in synaptic signaling, learning, and memory.47,48 Hence, it is tempting to speculate that repetitive mild TBI might target ASICs to cause CVR and learning deficits, however this hypothesis needs to be tested in future studies. Testosterone-induced impairments in endothelial vasodilation are prevented by inhibition of the NLRP3 inflammasome, suggesting that IL-1 beta generated in cerebral endothelium might contribute to altered cerebral vasodilatory responses to CO2 in CHI models. 24 ASIC 1 A contributes to inflammasome activation and inflammatory cytokines increase ASIC 1 A activity, providing a possible link between brain injury, IL-1 and ASIC activation.50–52 Further work to test this hypothesis in repetitive mild TBI models is currently ongoing in our laboratory.
Under normal homeostatic mechanisms in the brain, increase in arterial partial pressure of CO2 is sensed by peripheral and central chemoreceptors and ventilation is increased by feedback control of diaphragm and respiratory muscles from the brainstem respiratory center.53,54 In our study, the reduced increase in respiratory rate (ΔRmax) in acute 3HD mice suggests altered brainstem function. Notably, reduced respiratory rate of acute 3HD 107 cm mice may lead to a reduction of CO2 removal and an increase in the accumulation of arterial CO2. Compared with respiratory changes to hypercapnic challenge, no significant difference was detected in the respiratory variability (ΔRcv) among the four groups at baseline (prior to CO2 challenge), suggesting intact basal regulation of respiration in 3HD animals but impaired regulation during hypercarbia. A recent study of adolescents with mTBI54, 23 reported reduced respiratory rates with CO2 challenge in injured adolescents as well as uninjured controls, but minute ventilation (defined as respiratory rate x tidal volume) was not assessed nor was blood PaCO2 level. 23 In another study, 5 patients with post-concussion syndrome demonstrated increased end tidal CO2 levels compared to 17 controls at rest and during cognitive and physical tasks. 55 The findings in the current study that 3HD 107 cm mice had acute changes in their respiratory response to inhaled CO2 that resolved in the chronic phase are novel, and are consistent with effects of mTBI described for cardiovascular and other autonomic functions.56–58
We used a rotarod task to assess striatal dependent motor deficits after 3HD. The rotarod test has been shown to be more sensitive that beam walking or beam balance tasks in mild central fluid percussion injury models. 59 Rotarod deficits were not present at one month but developed by 6 months after 3HD 107 cm. To our knowledge, the current study is the first to demonstrate delayed striatal motor dysfunction in an adolescent repetitive mild CHI model. Previous studies of repetitive CHI in adolescent and adult mice using similar repetitive CHI models showed early rotarod deficits 60 that persisted in the chronic period in adult mice. 61 In rats subjected to multiple daily sub-concussive CHIs for up to 12 weeks, beam walk but not rotarod deficits developed between 2 and 12 weeks. 62 The finding that motor learning deficits may develop in the chronic period after repetitive mTBI independent of cognitive, circadian, or persistent CVR dysfunction suggests that the striatum or cerebellum may be particularly vulnerable to 3HD injury.
Disruption of sleep is one of the most commons symptoms of the post-concussive syndrome but still remains poorly understood.63,64 Changes in circadian timekeeping have been reported in humans and animal models of TBI.65,66 The circadian timekeeping system organizes rhythms of sleep/wake cycles, metabolism, and physiology in synchrony with the daily variation in the light/dark cycle. Surprisingly, we observed a modest lengthening of circadian free-running period and an increase in circadian amplitude in the 3HD 117 cm model but not the 3HD 107 cm model. These data suggest a dose-response by which TBI may elicit a reorganization of circadian behavior, quite possibly at the level of the central clock in the suprachiasmatic nucleus of the hypothalamus. It is unclear whether these changes represent a compensatory adaptation to TBI or another mechanism that promotes its detrimental effects on neurological function. There is strong evidence that CVR is regulated by the circadian clock and may gate the effectiveness of neuroprotective stategies sugesting that future studies could focus on the connections between TBI-related circadian re-programming and its physiological sequelae.67,68
This exploratory study has several important limitations in addition to those already mentioned. Ideally, we would have collected fMRI, physiological, and behavioral data in the same cohort of mice over time, particularly in the 117 cm group for which we only have Morris water maze data from a prior study, to compare with the 107 cm group in the current report. However, the amount of anesthesia exposure required for imaging could adversely affect behavior test performance, thus mice not involved in behavioral testing were subjected to fMRI. We did perform fMRI assessments on 6 of the same sham and 3HD 107 cm mice at 1 week and 1 year (also not included in behavioral analyses) to document resolution of CVR deficits. A second limitation is lack of a single injury control group to distinguish between effects of a single vs. 3HD. Thus, results from the current study can only be applied to repetitive mTBI. Third, we only studied male mice, because we have not yet characterized behavioral and physiological responses to 3HD in female adolescent mice. Those studies are ongoing in our laboratory. Fourth, we did not assess blood PaCO2 levels during fMRI because the phlebotomy would have caused significant changes in blood pressure. This approach mirrors the clinical studies 23 but as a result the endpoints for CVR data are restricted to time but not amplitude domains. Finally, the 3HD models undoubtedly fail to fully recapitulate the timing, severity, and age effects of repetitive mTBI in human adolescents. Rather, they serve as useful experimental platforms to explore potential outcomes that might take decades to elucidate in human studies and to explore mechanisms of neurological dysfunction in adolescents who may be particularly vulnerable to repetitive TBI.
In conclusion, we demonstrate proof of principle in an animal study that persistent CVR deficits are associated with chronic neurological dysfunction in an adolescent repetitive mTBI model. The data suggest that CVR to CO2 may be used to stratify adults at higher risk of developing chronic neurological sequelae after repetitive mild TBI in adolescence.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH/NINDS R01NS096550 (MJW) and the National Football Players Association grant to Harvard College (MJW, WPM, RM).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Meehan receives royalties from 1) ABC-Clio publishing for the sale of his books, Kids, Sports, and Concussion: A guide for coaches and parents, and Concussions; 2) Springer International for the book Head and Neck Injuries in the Young Athlete and 3) Wolters Kluwer for working as an author for UpToDate. His research is funded, in part, by philanthropic support from the National Hockey League Alumni Association through the Corey C. Griffin Pro-Am Tournament and a grant from the National Football League.
Authors’ contributions
LW, YC, SC, JL, and MW designed research, analyzed and interpreted the data, and wrote the manuscript; LW, YC, SC, GJ, EL, JL, FC, and W.E. performed analyses; RM, WM, and KK assisted in writing and revising the manuscript and approved the final version.
Supplemental material
Supplemental material for this article is available online.
