Abstract
Alterations in the concentration of nitric oxide (NO) and L-arginine metabolites have been associated with the pathophysiology of different vascular diseases. Here, we describe striking changes in L-arginine metabolism after hemorrhagic stroke. Blood and cerebrospinal fluid (CSF) samples of patients with intracerebral hemorrhage (ICH) and/or intraventricular hemorrhage were collected over a ten-day period. Liquid chromatography-tandem mass spectrometry was used to quantify key substrates and products of L-arginine metabolizing enzymes as well as asymmetric (ADMA) and symmetric dimethylarginine (SDMA). Changes in the plasma were limited to early reductions in L-ornithine, L-lysine, and L-citrulline concentrations. Intrathecally, we observed signs of early NO synthase (NOS) upregulation followed by a decrease back to baseline accompanied by a rise in the level of its endogenous NOS-inhibitor ADMA. SDMA demonstrated increased levels throughout the observation period. For arginase, a pattern of persistently elevated activity was measured and arginine:glycine amidinotransferase (AGAT) appeared to be reduced in its activity at later time points. An early reduction in CSF L-arginine concentration was an independent risk factor for poor outcome. Together, these findings further elucidate pathophysiological mechanisms after ICH potentially involved in secondary brain injury and may reveal novel therapeutic targets.
Introduction
Intracerebral hemorrhage (ICH) is a common subtype of stroke that is associated with high mortality and poor functional outcome. 1 In addition to the detrimental consequences of intraparenchymal ICH, extension into the ventricular system or intraventricular hemorrhage (IVH) has been identified as an independent predictor of poor outcome. 2 Secondary to the initial intracerebral bleeding event, brain injury is caused by various pathways including cytotoxicity of blood components, excitotoxicity, oxidative stress, and inflammation. 3
Nitric oxide (NO) is a crucial mediator of vascular autoregulation including cerebral blood flow. 4 Disturbances in NO metabolism are associated with cerebrovascular disease and brain injury.5,6 All the isoforms of NO synthase (NOS) catalyze the oxidation of L-arginine to NO and L-citrulline.6,7 Arginase I and II, enzymes that are located in the cytosol and the mitochondria, respectively, catalyze the metabolism of L-arginine to L-ornithine and urea. 8 Arginine:glycine amidinotransferase (AGAT) may use L-lysine as an alternative guanidine group acceptor to form L-homoarginine and L-ornithine. 9 Notably, L-homoarginine has recently been associated with stroke outcome. 10 Finally, methylation of L-arginine leads to the synthesis of dimethylarginines, which include asymmetric dimethylarginine (ADMA) and symmetric dimethylarginine (SDMA). 11 ADMA serves as a reversible competitive inhibitor of NOS and is a cause of endothelial dysfunction, 12 whereas SDMA acts as a competitor for L-arginine transport mediated by human cationic amino acid transporter 2B (hCAT-2B).11,13 Interestingly, SDMA has been associated with outcome after ischemic stroke.14,15
The actual role of NO in the context of ICH is controversial – poor outcome was associated with high CSF levels of NO in one study, 16 whilst poor outcome was related to low plasma concentrations of NO metabolites as well as L-arginine and L-citrulline in another study. 17 Dimethylarginine levels after ICH have only been measured in two studies that exclusively reported their peripheral blood levels but not CSF concentrations.18,19 Results were discrepant: Wanby and co-workers observed no changes in dimethylarginine levels, 18 whereas Worthmann et al. observed elevated ADMA levels and an independent association of SDMA concentration with outcome. 19
No comprehensive analysis of intrathecal metabolites of L-arginine catabolism after ICH has been reported yet. Moreover, CSF data on these metabolic pathways is also sparse for other acute neurological disorders. Therefore, our study aimed to provide a deeper understanding of systemic and CNS-specific L-arginine metabolism in patients suffering from ICH with the hypothesis that L-arginine metabolism is altered in hemorrhagic stroke. This involved serial measurements of substrates and products of the key enzymes of L-arginine catabolism, NOS, arginases, and AGAT, as well as dimethylarginines up to ten days after hemorrhagic stroke.
Material and methods
Patient recruitment and study design
This prospective cohort study was performed in accordance with institutional guidelines, international ethical standards, and the Helsinki Declaration of 1975 (and as revised in 1983). Study approval was granted by the local ethics committee (Ethik-Kommission der Ärztekammer Hamburg, PV5497). Written informed consent for study participation was obtained from the patient or, if not possible for medical reasons, from the healthcare proxy.
Patients with ICH admitted to our university medical center from 2017 to 2019 were screened for enrollment. Patients with ICH related to aneurysmal subarachnoid hemorrhage or traumatic brain injury were excluded. Inclusion criteria were external ventricular drain (EVD) placement within the first three days after onset and age ≥18 years. Clinical indication for EVD placement at our center was made by the senior consultant on duty in patients with ICH/IVH with a decreased level of consciousness and radiological signs of hydrocephalus. Patients ≥18 years admitted for evaluation of normal pressure hydrocephalus or ventriculoperitoneal shunt dysfunction were recruited as a control cohort. Patients presenting with clinical or laboratory parameters of infection were excluded as control patients.
In ICH patients, blood samples and CSF samples from the EVD were collected at early, intermediate and late time points that were defined as days 0–3, 4–7, and 9–11, respectively. For the control cohort, CSF was collected either from spinal tap test or intraoperatively from shunt placement/revision. Fresh CSF samples were spun down, supernatant was collected and stored at -80°C until analysis. Plasma was separated from EDTA whole blood via centrifugation and stored at −80°C as well.
Demographic, clinical and outcome data were collected for all ICH patients. Medical history was screened for oncological or autoimmune disease as potential confounders. Primary outcome parameter was the extended Glasgow Outcome Scale (eGOS) after six months. 20 Initial Glasgow Coma Scale (GCS) and ICH Score were used to evaluate clinical ICH severity.2,21 ICH and IVH volumes on the initial CT scan were determined by volumetric analysis (Brainlab Elements, Brainlab AG, Munich, Germany). Extent of intraventricular blood was additionally evaluated via the modified Graeb score. 22
Analysis of L-arginine and its metabolites by LC-MS/MS
Established and validated protocols for liquid chromatography-tandem mass spectrometry (LC-MS/MS) were used to quantify plasma and CSF L-arginine and its metabolites.23–25 Briefly, 25 μl of plasma or CSF were diluted in methanol to which stable isotope labeled internal standards had been added. Thereafter, the analytes were converted into their butyl ester derivatives and quantified by LC-MS/MS. Analyte concentrations were calculated using calibration curves based on four concentration levels of each analyte. Quality controls (QC) were run in two levels by triplicates. The second analysis was done on the QC samples to assess coefficient of variation and bias of QC, which was below 15% for all analytes.
Ratios of known substrates and products of L-arginine metabolizing enzymes were calculated as surrogate parameters for enzyme activity. This was conducted for NO synthase by using the L-arginine/L-citrulline ratio (NOS-ratio), for arginase with L-arginine/L-ornithine (arginase-ratio), and for AGAT with L-lysine/L-homoarginine (AGAT-ratio).7,9 As a marker of substrate availability for NO synthesis, the ratio L-arginine/ADMA (Arg/ADMA ratio) was used. 11
Statistical analyses
Statistical analysis and visualization were performed using Python 3.7.4 language including the modules pandas, seaborn, SciPy, NumPy, scikit-learn and statsmodels. For binary logistic regression analysis, IBM® SPSS® Statistics Version 24.0 (IBM Corporation, Armonk, NY, USA) was used. Outliers in the MS dataset were defined by a Z score of 4 within all measurements of a single biomolecule separately for either CSF or blood and removed from the analysis. Patients demonstrating more than one outlier were completely excluded from analysis. Continuous data is presented as mean ± standard deviation (SD), except for eGOS, GCS and ICH score which were shown as median with interquartile range (IQR). Mann–Whitney U test was used for univariate analyses between control and ICH patients after confirmation of non-normal distribution of data. Distribution of data was assessed by Kolmogorow-Smirnow-Test and inspection of histograms. A level of statistical significance of p < 0.05 was applied. Correction for multiple testing was performed as described in the results. Hierarchical agglomerative cluster analysis was based on the UPGMA method. Correlation matrices were based on the Spearman correlation coefficient. A binary logistic regression model was used to examine the independent effect of variables on dichotomized outcome and was adjusted for ICH score, which is based on the predictors age, initial GCS, hematoma volume, presence of intraventricular blood, and infratentorial location. For CSF compounds, modified Graeb score as a more accurate estimate for IVH extent was additionally added as a covariate. L-Arginine derivatives were included if they showed a significant correlation that was confirmed by univariate testing.
Results
Included patients and samples
A total of 26 patients with ICH and 20 control patients were recruited for this study. Time points of sample collection were early, intermediate, and late in 26, 21, and 13 patients, respectively. In two patients, two samples were collected each during the early time period (day 0 and day 1 or 2).
One ICH patient and one control patient were removed from analysis due to multiple outliers in the MS dataset. The ICH patient had a history of rheumatoid arthritis and showed a complicated clinical course with unclear disseminated intravascular coagulation. Two more measurement values of a single biomolecule from other patients were classified as outliers and excluded from analysis.
The final dataset for analysis consisted of 25 ICH and 19 control patients. The total CSF sample number was 78 including 19 control samples. For ICH, 27, 20, and 12 were collected at early, intermediate, and late time points, respectively (Table 1). The number of blood samples was 11 for control samples and 26, 19, and 12 for ICH at early, intermediate, and late time points, respectively. A reduced sample number at later time points was mostly due to earlier removal of the EVD based on clinical decision making.
Demographic and clinical characteristics of the study cohort.

Description of demographic and clinical characteristics of the study cohort
ICH was spontaneous in 18 patients and secondary in 7 patients (Table 1). One control patient suffered from ICH app. 1.5 years before and presented now with suspected ventriculo-peritoneal shunt dysfunction.
Age and sex distribution were comparable between the ICH and control cohorts (Table 1). Mean age was 66.3 ± 12.6 and 70.7 ± 15.3 years (p = 0.07) and number of female patients was 10 (40.0%) and 10 (52.6%) in the ICH and control cohort (p = 0.405), respectively. Mean ICH volume was 24.3 ± 28.2 ml and mean IVH volume was 12.3 ± 11.7 ml with a mean modified Graeb score of 10.7 ± 5.8. ICH was located infratentorial in 7 (28.0%) cases. As a measure of overall ICH severity, median ICH score was 2 (IQR 2–3). A craniotomy for hematoma evacuation was received by 8 patients (32.0%). This was performed before collection of the first sample in all cases. Four patients (16.0%) received a ventriculo-peritoneal shunt implantation during the 6 months of follow-up.
Overall outcome was poor with a median eGOS of 3 (IQR 1–3) after 6 months. Eight patients (32.0%) deceased (eGOS 1) and one patient (4.0%) demonstrated a persistent vegetative state (eGOS 2). With 15 patients (60.0%), the majority of the ICH cohort was classified as severely disabled (eGOS 3&4). Only one young patient suffering from an AVM-related hemorrhage achieved a good recovery (eGOS 7).
Temporal signature of changes in L-arginine metabolism after intracerebral hemorrhage
We measured the concentrations of L-arginine and its main derivates in the CSF and blood of ICH and control patients. Not only did we see marked differences between control and ICH group samples, but also a differential temporal pattern and striking differences between intrathecal and systemic environment (Figure 1). Interestingly, we did not observe major differences between different etiologies (Figure 2). Moreover, statistical testing did not show significant differences in metabolite levels between patients with spontaneous ICH (without relevant comorbidities) and the remaining cohort at any time point (Figure 3(a)). Therefore, we assumed a well-conserved alteration of L-arginine metabolism after ICH independent from the underlying cause and conducted our downstream analysis under inclusion of all etiologies.
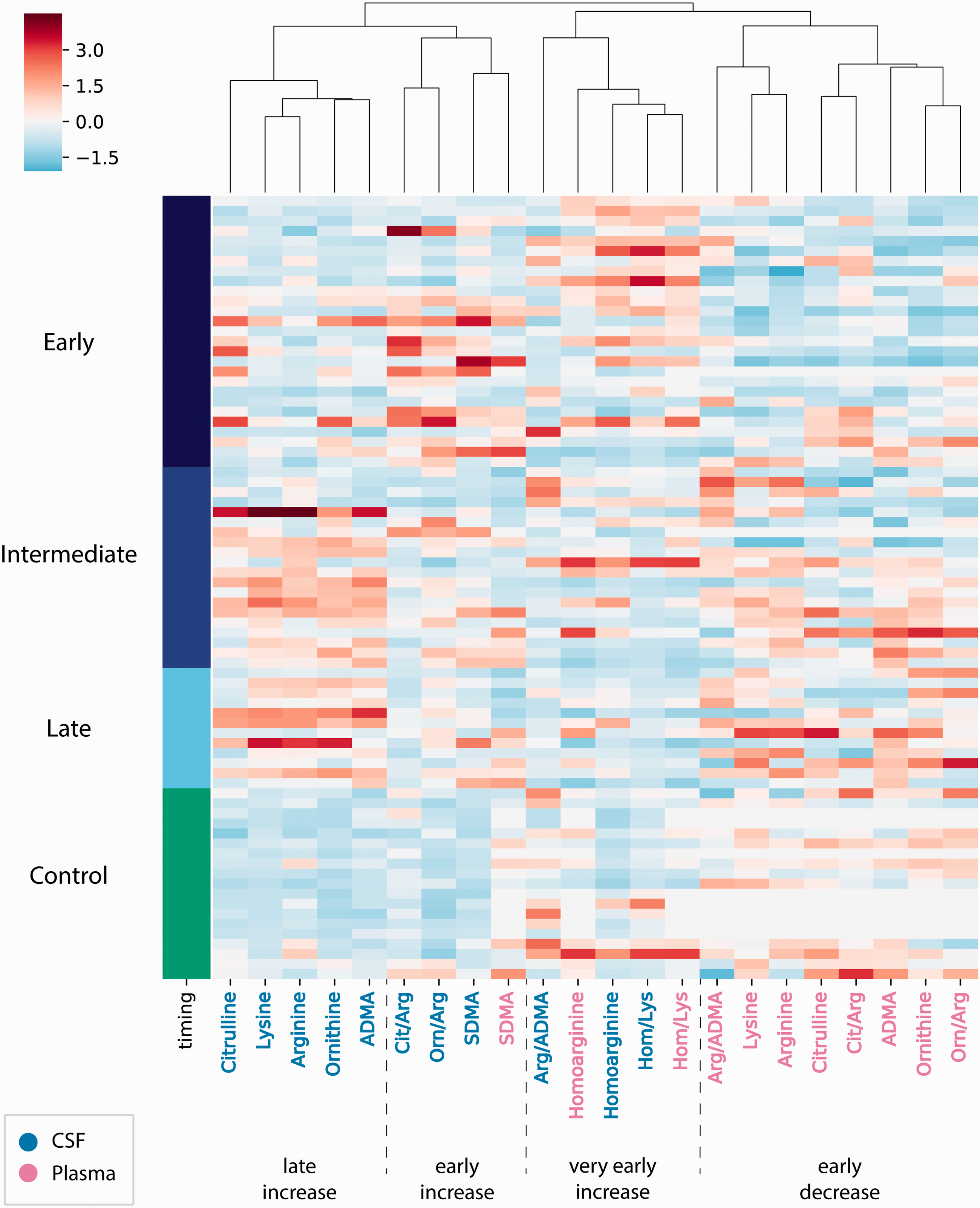
The heatmap demonstrates normalized measurement values of L-arginine derivates and their ratios. Rows are sorted by ICH and control patients and within the ICH group by day of sample collection after ICH onset. Columns are hierarchically clustered and depict the different biomolecules and their ratios separately for CSF and plasma.
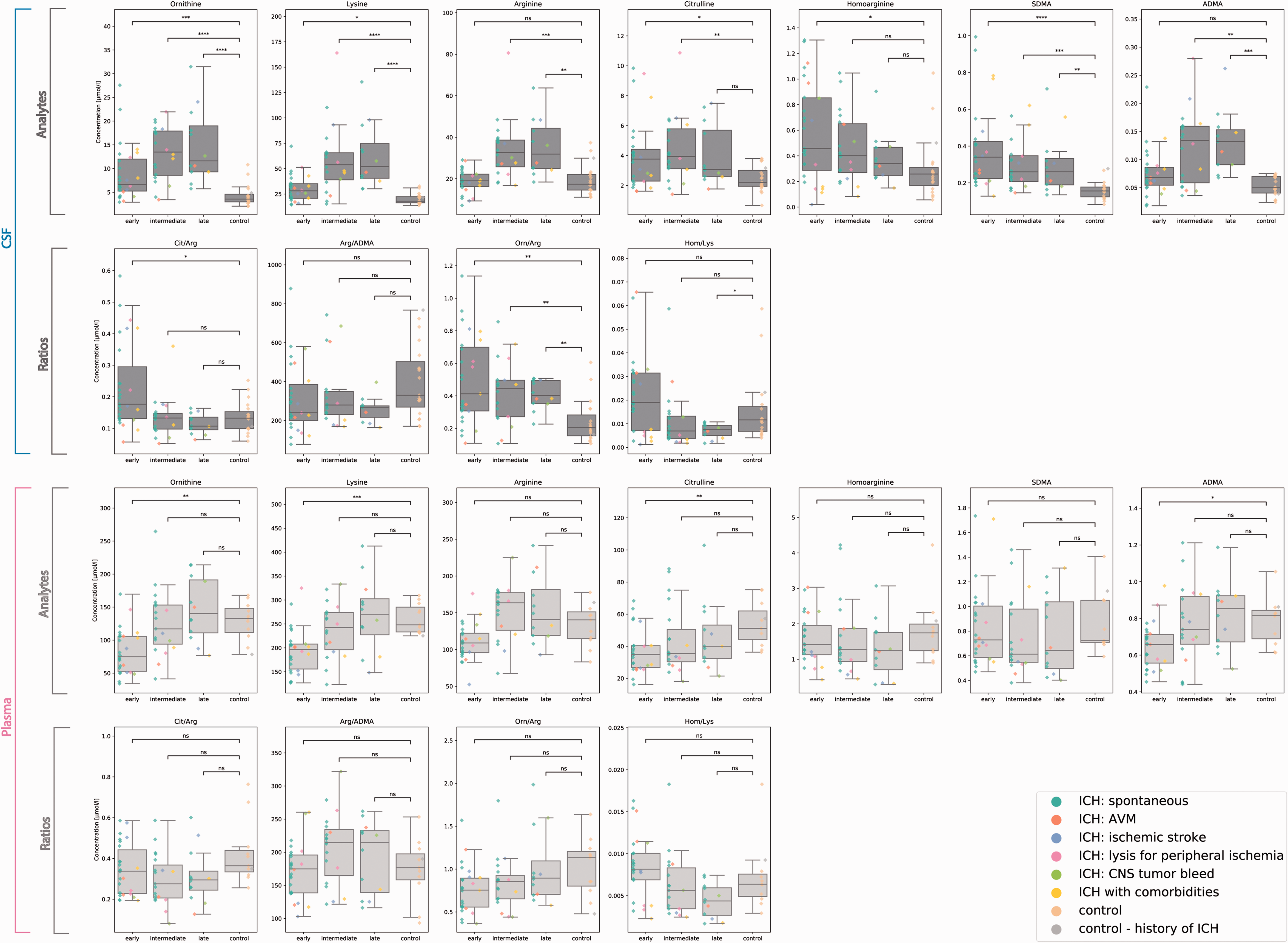
The boxplots demonstrate the distribution of measurement values of L-arginine derivates and selected ratios for different timepoints after ICH and control patients. Samples derived from CSF are shown in the upper, samples from plasma in the lower rows. Single data points are color labeled for different etiologies. Differences between ICH and control values are tested for their statistical significance (two-sided Mann-Whitney U test with Bonferroni correction within subplots). ns: p > 0.05; *: p < 0.05; **: p < 0.01; ***: p < 0.001; ****: p < 0.0001.
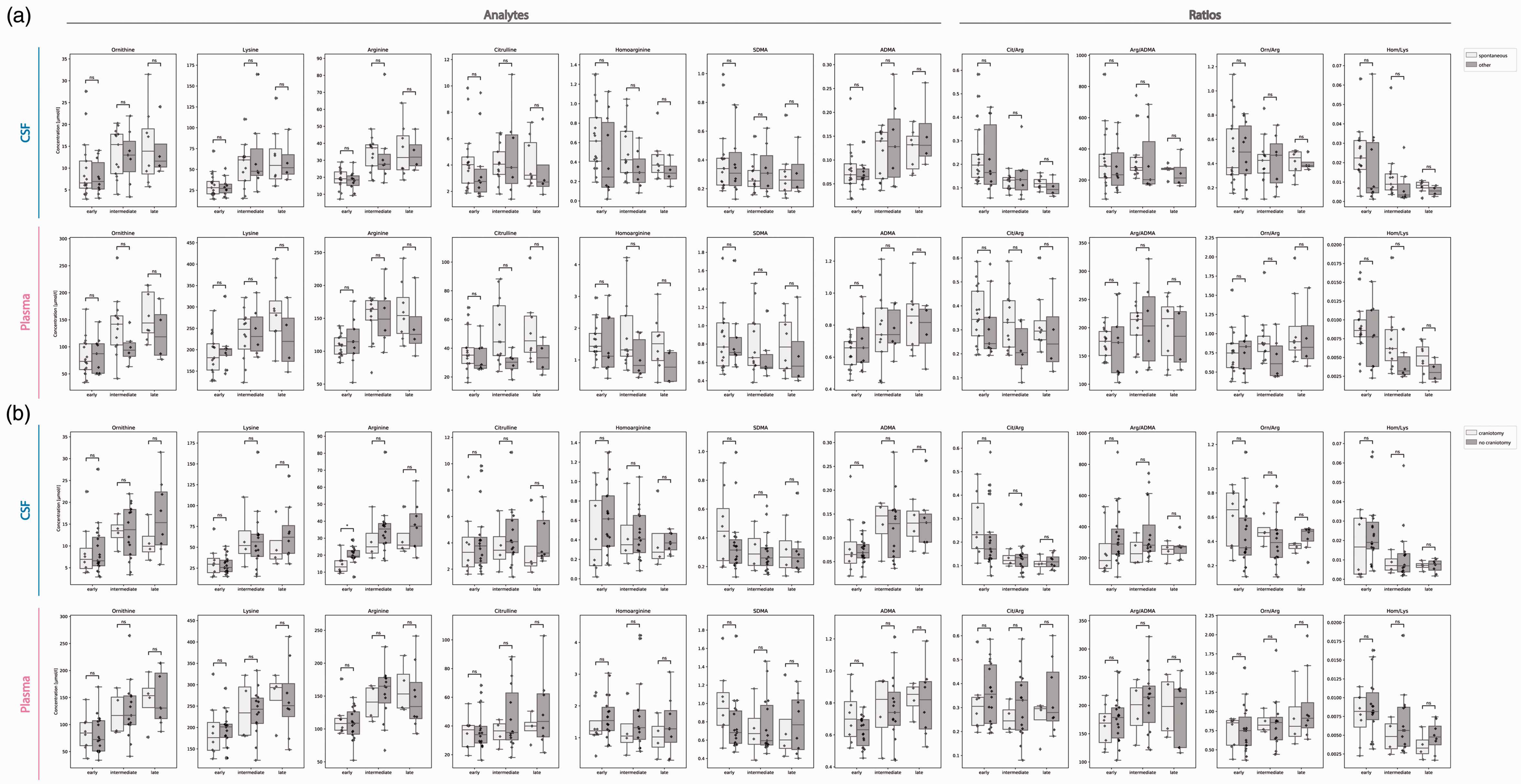
The boxplots show the distribution of measurement values of L-arginine derivates and selected ratios for CSF and plasma at different timepoints. (a) Differences between patients with spontaneous ICH without relevant comorbidities (n = 15) and the remaining cohort (n = 10) as well as (b) differences between patients who received a craniotomy with hematoma evacuation (n = 8) and patients with EVD placement only (n = 17) are demonstrated and tested for their statistical significance (two-sided Mann-Whitney U test with Bonferroni correction within subplots). ns: p > 0.05; *: p < 0.05; **: p < 0.01; ***: p < 0.001; ****: p < 0.0001.
Hierarchical clustering of normalized values returned four main groups with distinct patterns (Figure 1). Three groups showed an increase in concentrations compared to control patients either more pronounced at a very early, early or later time point and were mostly reflected by the intrathecal compartment. The ‘very early cluster’ was the least specific and did not separate between control and ICH consistently. Weak significant differences in this cluster were only found for early L-homoarginine and late AGAT-ratio (Figures 1 and 2). The ‘early increase cluster’ included SDMA of both CSF and plasma as well as NOS-ratio and arginase-ratio in CSF while the ‘late increase cluster’ consisted of L-citrulline, L-lysine, L-arginine, L-ornithine and ADMA in the CSF. Univariate testing confirmed a significant difference between the control and ICH cohorts for L-ornithine, L-lysine, L-arginine, L-citrulline and ADMA as well as for NOS-ratio, arginase-ratio and SDMA in CSF but not plasma (Figure 2). Notably, L-citrulline showed a significant elevation at the early and intermediate but not at the late time point and thus is distinct from the other late-increase biomolecules as also shown by the subcluster in Figure 1. A fourth main cluster demonstrated an early decrease and was exclusively apparent in the plasma (Figure 1). Significant effects in this ‘early decrease cluster’ were confirmed for L-ornithine, L-lysine, L-citrulline and ADMA (Figure 2).
Except for early intrathecal L-arginine levels, no significant differences were observed between the eight patients who received a craniotomy with hematoma evacuation and patients who were treated solely with EVD placement (Figure 3(b)). L-arginine was significantly lower in patients that were allocated to craniotomy (p = 0.026).
Correlation between L-arginine derivatives and demographic, clinical and outcome parameters
Next, we sought to examine the association between alterations of L-arginine metabolism and clinical parameters. Given the profoundly different patterns of early and late phases after ICH onset, we calculated correlation matrices separately for these time points (Figure 4).
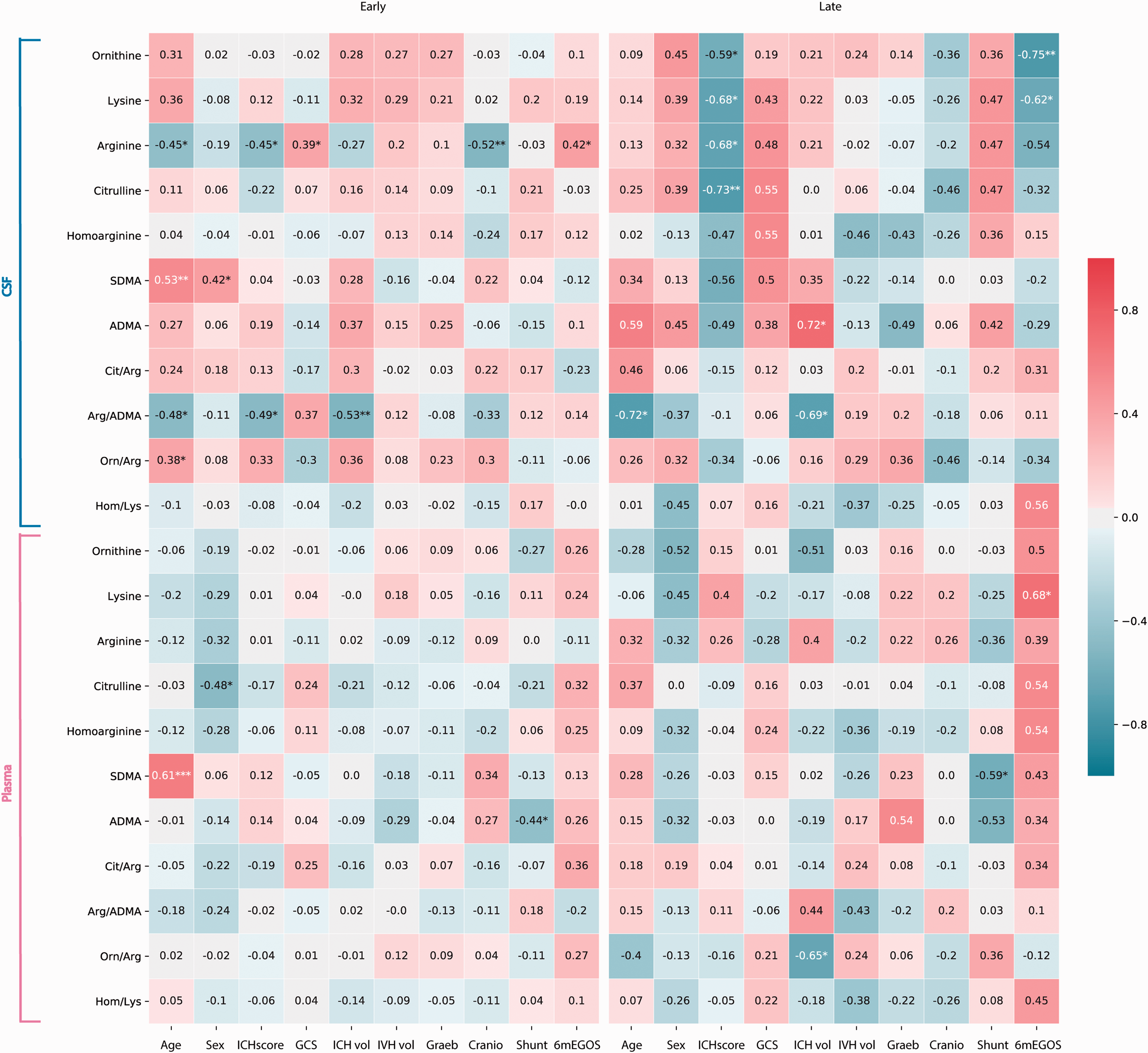
The heatmaps show a correlation matrix based on the Spearman correlation coefficient between demographic, clinical and outcome parameters and L-arginine derivates and substrate/product ratios for CSF and plasma samples. The left map represents early, the right map late sampling timepoints. The significance of the correlation is expressed by asterisks (*: p < 0.05; **: p < 0.01; ***: p < 0.001).
We observed the most consistent pattern of correlations for early L-arginine concentration in CSF. Early L-arginine exhibited a negative correlation with the ICH score and associated parameters such as higher age and lower initial GCS. Moreover, low L-arginine was also associated with a higher rate of craniotomies. Interestingly, no significant correlations were seen for IVH volume or modified Graeb score at early and late time points. Only Arg/ADMA ratio correlated with ICH volume at the early time point. Further, L-arginine, L-ornithine, L-lysine, and L-citrulline concentration in the CSF correlated negatively with the ICH score in the late phase.
In line with the relationship of early L-arginine CSF levels and ICH severity, we observed an early positive correlation of CSF L-arginine with eGOS (Figures 4 and 5(a)). Interestingly, this positive correlation swapped to a negative but not significant correlation at the late time point. This was accompanied by significant correlations of L-ornithine (CSF) and L-lysine (CSF and plasma).
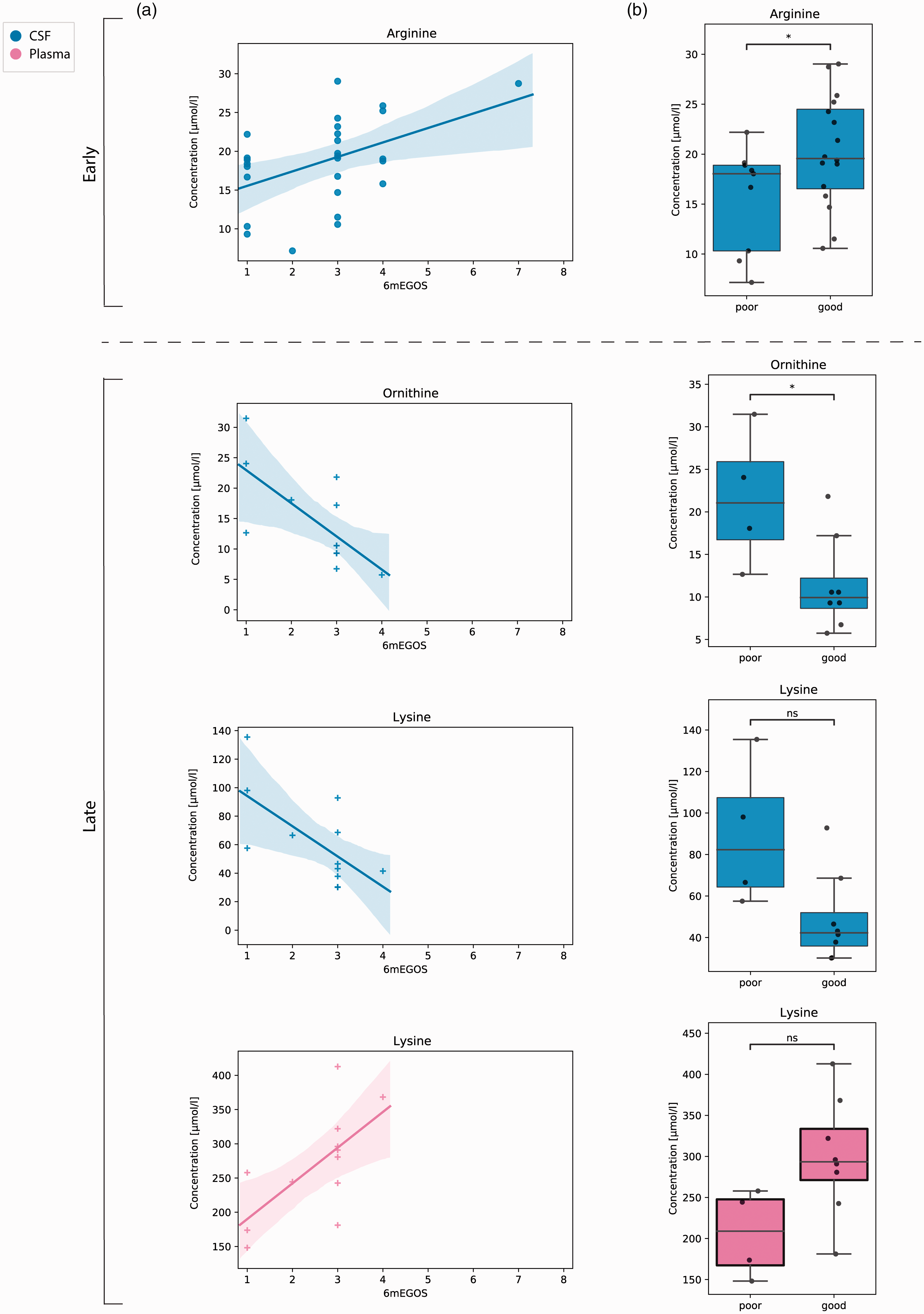
(a) Scatterplots show timepoint specific (dot marker: early; cross marker: late) concentrations of biomolecules and substrate/product ratios in CSF (blue) and plasma (magenta) with a significant Spearman correlation coefficient against eGOS. Linear regression lines are displayed with a 95% confidence interval. (b) Same biomarker constellations are illustrated by boxplots against a dichotomized outcome variable (eGOS ≤2 = poor; eGOS ≥3 = good). Annotations demonstrate significant differences of a two-sided Mann-Whitney U test (*p < 0.05).
In order to confirm these findings, we dichotomized the outcome based on the distribution of our data into eGOS ≤2 (n = 9) vs eGOS ≥3 (n = 16) to allow for univariate (Figure 5(b)) and logistic regression analysis between these groups. Values derived from samples that were collected twice from same patients in the early period were averaged for further analysis. In the early phase, a significant difference was confirmed for CSF L-arginine concentration (15.6 ± 5.2 vs 20.3 ± 5.5 µmol/l (p = 0.036)). For the late phase, univariate analysis confirmed elevated levels of L-ornithine in patients with bad outcome (21.6 ± 8.1 vs 11.4 ± 5.4 µmol/l (p = 0.027)).
In order to confirm an independent influence on outcome in the early phase, we used a binary logistic regression model. The limited patient number of only twelve at the late time point was not deemed suitable for regression modelling. After adjustment for ICH score and modified Graeb score, early CSF L-arginine concentration retained a significant association with outcome (Odds ratio 1.516 (95% Confidence interval 1.03–2.23) per µmol/l; p = 0.045). Accordingly, low concentrations were predictive for poor outcome.
Discussion
Alterations of L-arginine metabolism in CSF and plasma after ICH
Our data provide new and additional insight into the alterations of L-arginine metabolism in CSF and plasma after ICH (Figure 6(a)). We observed early increased L-citrulline concentrations in the CSF with, in comparison to the control group, unchanged L-arginine concentrations, which is suggestive for an early increase in NOS activity. Later on, however, accumulating L-arginine together with a decrease in L-citrulline back to a normal level might be interpreted as a decline in the activity of NOS with consecutively less NO production. Increasing CSF levels of ADMA, a dimethylated analogue of L-arginine and known inhibitor of NOS, 26 at intermediate and late time points could potentially cause this late drop in NOS activity. A similar pattern was observed for L-arginine and ADMA in CSF after subarachnoid hemorrhage. 27 Moreover, an association between CSF concentrations of ADMA and delayed cerebral ischemia after subarachnoid hemorrhage has been established.25,28
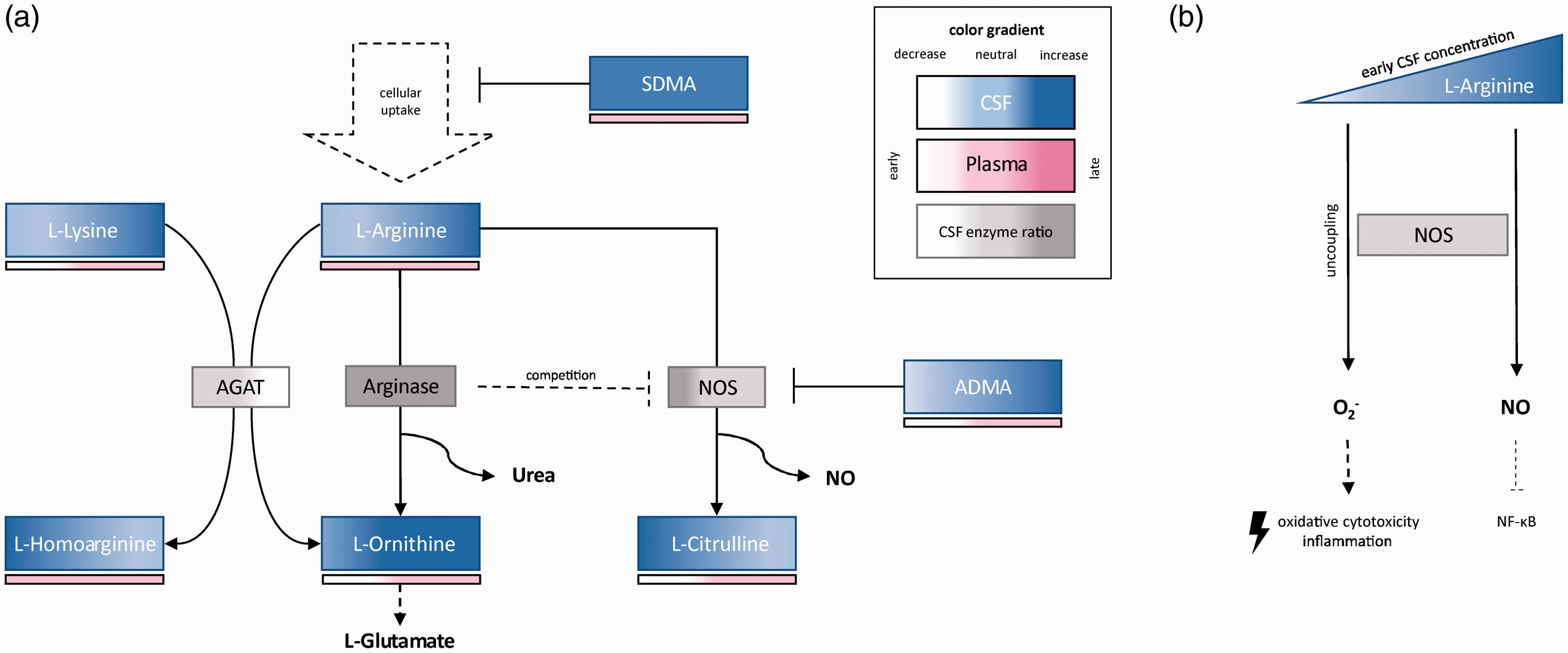
The flow charts illustrate the biochemical findings and pathomechanistic hypotheses generated by this study. (a) Biomolecules measured by mass spectroscopy in the CSF are displayed in blue boxes and a bar located below reflects plasma values in red. Color gradients schematically depict concentration changes over time in comparison to control samples with a left to right temporal orientation per box. Key enzymes of the L-arginine metabolism are shown in grey boxes with color gradients demonstrating assumed activity levels in the CSF based on substrate/product ratios. (b) Uncoupling of NOS and increased NF-κB mediated inflammation in relation to low L-arginine concentration are illustrated as possible pathomechanisms of secondary brain injury after ICH.
We observed elevated concentrations of SDMA in CSF throughout the observation period. As another dimethylated L-arginine analogue, SDMA competes with cellular uptake of L-arginine and leads to an intracellular L-arginine depletion. 13 This may be a second mechanism leading to a declining NOS activity over time while extracellular L-arginine is increasing and can be detected in the CSF while not necessarily reflecting an accurate estimate of the intracellular concentration. Acute elevations in intrathecal SDMA concentrations have previously been described also for ischemic stroke. 29
At later time points, rather high L-lysine and low L-homoarginine concentrations lead to the assumption of reduced AGAT activity.9,10 By contrast, an increasing concentration of CSF L-ornithine over time with a persistently elevated L-ornithine/L-arginine ratio may point to an increased activity of arginase as a main pathway of L-arginine catabolism. 8 Arginase is therefore a direct competitor with NOS, and high arginase activity in macrophages was shown to reduce NO production.30,31 Interestingly, arginase demonstrates a gene expression that is regulated in a complex manner by different agents including cytokines, suggesting that secondary inflammatory processes could lead to an upregulation. 8 It was indeed shown that experimental tissue trauma can lead to increased arginase activity that is able to outcompete simultaneously upregulated NOS activity. 32 Similarly, patients with severe trauma demonstrated elevated plasma arginase I levels accompanied by decreased L-citrulline and NO metabolite levels. 33 With regards to CNS lesions, increased arginase I levels have been detected after experimental spinal cord injury associated with an increase in macrophages and reactive astrocytes. 34 Hence, increased arginase activity, which occurs possibly as a sequel of neuroinflammation, may be a third underlying mechanism of the late reduction of NOS activity observed in our cohort.
In plasma, an early significant decrease of the amino acids L-ornithine, L-lysine, and L-citrulline was detected. We also observed lower L-arginine values but without statistical significance. A significant drop in plasma L-arginine after ICH has been described before17,18 but, similar to our findings, only showed a trend to decrease in a different study. 19 Furthermore, significant decreases for L-citrulline and L-ornithine after ICH have been measured as well. 17 Similarly, significantly decreased plasma levels of L-arginine, L-citrulline and L-ornithine without changes in dimethylarginines have been reported after severe traumatic brain injury. 35 In our cohort, we also did not observe any alterations in plasma dimethylarginines, which is in line with a previous study of ICH patients. 18 However, this is in contrast to a study by Worthmann et al. describing elevated ADMA levels through day seven after ICH. 19
Impact of early alterations in L-arginine metabolism on neurological outcome
We discovered several correlations between neurological outcome after six months and alterations in L-arginine metabolism (Figure 5(a)). For the early phase, we confirmed the L-arginine concentration in the CSF to be significantly lower in patients that were dead or in a persistent vegetative state after six months. Consistently, we observed also a negative correlation between early CSF L-arginine and age, hematoma volume, and impaired consciousness. Moreover, L-arginine concentration retained an independent effect on neurological outcome even after adjustment for ICH severity and IVH extent. There are several pathophysiological mechanisms that might explain this relationship (Figure 6(b)). Low levels of L-arginine have been demonstrated to lead to oxidative stress due to an uncoupling of NOS with oxygen radical formation like superoxide (O2–) and increased downstream nitration products like nitrotyrosine, suggestive for the cytotoxic oxidant peroxynitrite (ONOO−). 36 Moreover, endothelial NOS has an important role for the maintenance of cerebral blood flow after CNS injury. 37 Lack of nitric oxide has been identified as an important mechanism of post-hemorrhagic microvascular dysfunction in experimental subarachnoid hemorrhage. 38 A similar role of improved microperfusion after ICH is imaginable and would be impaired by L-arginine depletion. Reduced production of NO may also lead to enhanced neuroinflammation given the anti-inflammatory properties of NO by redox regulation of NF-κB. 39
Our data may provide evidence for a potential beneficial role of early increase in L-arginine concentration in the CNS compartment. Indeed, a protective effect for L-arginine administration after experimental traumatic brain injury or vasospasm after subarachnoid hemorrhage has previously been reported.40,41 L-Arginine substitution after brain injury has been shown to increase brain tissue NO levels and cerebral blood flow. 42 However, L-arginine supplementation in experimental ischemic stroke showed no benefit despite increased cortical blood flow whereas other NO donors reduced lesion volume. 43 Interestingly, poly-arginine and L-arginine-rich peptides have been demonstrated to be neuroprotective in experimental stroke even though a protective role in ongoing intracerebral bleeding was not confirmed.44,45 Our data encourage further experimental studies to explore therapeutic strategies aiming at early CNS L-arginine increase in the setting of ICH and IVH.
Besides low L-arginine levels, NOS uncoupling has previously been observed in the presence of elevated ADMA levels.46,47 However, it was also reported that ADMA can inhibit O2– generation in the absence of L-arginine in a tetrahydrobiopterin-dependent manner. 48 Our current data depict no correlation between systemic or intrathecal dimethylarginine concentrations and outcome, thus could neither support a detrimental or protective role of ADMA in ICH. Worthmann et al. investigated the influence of plasma dimethylarginine concentrations on the outcome in ICH patients and found an increase in patients with unfavorable outcome. 19 However, only an elevation of SDMA and not ADMA was confirmed in a logistic regression model. We were not able to confirm this finding in our cohort.
Impact of late alterations in L-arginine metabolism on neurological outcome
Data derived from late sampling time points in our cohort need to be interpreted cautiously due to a limited patient number and possible selection bias. Early dropouts were due to removal of the EVD based on clinical decisions or death of the patient. Accordingly, we doubted the validity of a regression analysis in this small patient subset and only correlation and univariate analyses were performed. Intrathecally, we observed particularly higher concentrations of L-ornithine and L-lysine in patients with poor outcome, which was significant only for L-ornithine in the univariate analysis. As outlined above, L-ornithine is the product of L-arginine catabolism by arginase. L-ornithine can also be a product of AGAT from the substrates L-arginine and L-lysine.9,10
It might be speculated that elevated L-lysine levels are due to a decrease in AGAT activity as we observed a significant correlation of the AGAT ratio with outcome, which was however not confirmed in the univariate testing. Interestingly, treatment with L-lysine has previously been associated with a protective role after experimental ICH. 49 An explanation might be that an exogenous increase in L-lysine improves the production of L-homoarginine, given that low levels of L-homoarginine have been related to poor outcome after ischemic stroke. 10
The late increase in L-ornithine in patients with poor outcome could be interpreted as a sign of increased arginase activity. As outlined above, arginase activity can be an indicator of an inflammatory reaction and compete with NOS for the substrate L-arginine, potentially promoting oxidative stress.8,30–33,36 Moreover, it has been shown that arginase itself can promote inflammation by decreasing cellular content of NO resulting in an enhancement of the NF-κB pathway, which is reversed by arginase inhibition. 50 By inhibiting NO production via several mechanisms, arginase upregulation has been demonstrated to contribute to endothelial dysfunction in different diseases. 51 The product L-ornithine is furthermore the substrate for L-glutamate synthesis, 7 and a deterioration of excitotoxicity would be imaginable. Indeed, arginase inhibition by L-citrulline or L-ornithine demonstrated a protective effect after experimental ischemic stroke with improved functional outcome and reduced markers of oxidative stress. 52
Taken together, the findings in this exploratory study let us hypothesize that similar to other conditions L-arginine metabolism needs more attention for its possibility to be pharmacologically manipulated to improve outcome for ICH patients.
Limitations, generalizability, and value of the study
The current study is a pilot study with a limited number of patients. For the first time, a comprehensive description of L- arginine metabolism after ICH has been provided for both, the systemic and intrathecal, compartments. Our cohort was comparable in size to related studies that only examined plasma.18,19 It exhibited sufficient power for the establishment of a pathophysiological hypothesis that, however, needs to be confirmed in larger patient cohorts. The generalizability of our study cohort is limited to ICH/IVH patients requiring external CSF drainage. A possible selection bias may be present for late sampling time points. Overall, the patient cohort was severely affected and showed mostly a poor outcome after 6 months, which is expected in ICH patients requiring EVD placement. 53 However, this might have limited the power of the outcome related analysis due to reduced selectivity of the dependent variable. Moreover, correlations were calculated without adjustment for multiplicity and the final functional role and causality of the shown correlations remain to be clarified. Differentiation of metabolic changes between different cell populations and enzyme isoforms might be important for deeper pathophysiologic understanding 54 but is not possible to achieve from patient-derived biomaterials like CSF or plasma. Moreover, extracellular concentrations may be different from intracellular concentrations particularly in the context of uptake inhibitors like SDMA. 13
Conclusions
L-arginine metabolism is strikingly altered after ICH. Whereas changes in the plasma were limited to early reductions in L-ornithine, L-lysine, and L-citrulline concentrations, intrathecal alterations were more drastic. Hallmarks were (i) signs of early NOS upregulation followed by a decrease back to baseline accompanied by a rise in the level of its endogenous inhibitor ADMA, (ii) persistent increase in SDMA, (iii) signs of persistently elevated activity of arginase and (iv) signs for a delayed reduction in AGAT activity. Early reduction in CSF L-arginine was identified as an independent risk factor for poor outcome.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contribution
M. Mader: Study design. Patient recruitment and sample collection. Statistical analysis and interpretation of data. Drafting the manuscript. R. Boeger: Study design. Acquisition of MS data. Interpretation of data. Critically revising the manuscript. D. Appel: Interpretation of data and critically revising the manuscript. E. Schwedhelm: Acquisition of MS data. Interpretation of data and critically revising the manuscript. M. Haddad: Interpretation of data and critically revising the manuscript. M. Mohme: Patient recruitment and sample collection. Interpretation of data and critically revising the manuscript. K. Lamszus: Supervision of sample deposition. Interpretation of data and critically revising the manuscript. M. Westphal: Interpretation of data and critically revising the manuscript. P. Czorlich: Study design. Patient recruitment and sample collection. Interpretation of data and critically revising the manuscript. J. Hannemann: Study design. Acquisition of MS data. Interpretation of data. Critically revising the manuscript. All authors have read and approved the final version of the manuscript.
Acknowledgments
We want to thank Mareike Holz, Katharina Kolbe, Svenja Zapf, Sarah Matulla, and Mariola Kastner for their excellent technical support. We are grateful for the support of the study participants and their relatives.
