Abstract
Despite early thrombectomy, a sizeable fraction of acute stroke patients with large vessel occlusion have poor outcome. The no-reflow phenomenon, i.e. impaired microvascular reperfusion despite complete recanalization, may contribute to such “futile recanalizations”. Although well reported in animal models, no-reflow is still poorly characterized in man. From a large prospective thrombectomy database, we included all patients with intracranial proximal occlusion, complete recanalization (modified thrombolysis in cerebral infarction score 2c–3), and availability of both baseline and 24 h follow-up MRI including arterial spin labeling perfusion mapping. No-reflow was operationally defined as i) hypoperfusion ≥40% relative to contralateral homologous region, assessed with both visual (two independent investigators) and automatic image analysis, and ii) infarction on follow-up MRI. Thirty-three patients were eligible (median age: 70 years, NIHSS: 18, and stroke onset-to-recanalization delay: 208 min). The operational criteria were met in one patient only, consistently with the visual and automatic analyses. This patient recanalized 160 min after stroke onset and had excellent functional outcome. In our cohort of patients with complete and stable recanalization following thrombectomy for intracranial proximal occlusion, severe ipsilateral hypoperfusion on follow-up imaging associated with newly developed infarction was a rare occurrence. Thus, no-reflow may be infrequent in human stroke and may not substantially contribute to futile recanalizations.
Keywords
Introduction
In acute ischemic stroke (AIS) due to large vessel occlusion (LVO), mechanical thrombectomy (MT), preceded or not by intravenous thrombolysis, affords >80% recanalization rates and significantly improves outcome. 1 However, >50% of treated patients do not recover to an independent life. 1 Understanding the mechanisms underlying such “futile recanalizations” 2 may lead to novel therapies as adjuncts to thrombectomy.3,4 Tissue reperfusion is a stronger predictor of clinical outcome than recanalization,5–7 but numerous adverse events may compromise reperfusion following thrombectomy, including per-procedural distal embolization, early re-occlusion, parenchymal hemorrhage (PH), and the “no-reflow” phenomenon. 8
No-reflow refers to the absence of tissue reperfusion despite complete recanalization, due to impaired microvascular perfusion. First described in rabbits following global cerebral ischemia, 9 it was subsequently confirmed in monkeys subjected to transient occlusion of the proximal middle cerebral artery (MCA)10–13 in the form of extensive areas with complete lack of capillary filling following carbon black perfusion associated with tissue infarction, as well as more recently in rodents.14–16 No-reflow is currently believed to reflect capillary constriction secondary to pericyte disruption in previously ischemic areas,14,17 possibly associated with pre-capillary small muscle cell constriction. 18 Disruption of capillary flow facilitates microvascular obstructs containing trapped erythrocytes and leukocytes, fibrin deposits, and aggregated platelets.14,17,19,20 Importantly, no-reflow may be prevented or counter-acted, for instance by acting on the NO pathway. 14
Despite these experimental data, no-reflow remains poorly characterized in man, and while an incidence around 25% is frequently cited, 19 its very existence is still debated. Three main factors, which all directly derive from its definition, may explain this situation. First, documentation of complete arterial recanalization, which is afforded only by digital subtraction angiography (DSA), is required in order to reliably investigate no-reflow. However, DSA has only recently become part of routine acute stroke care, after thrombectomy became standard-of-care. 1 Second, complete recanalization on DSA has been variably defined over time, and a strict definition as modified thrombolysis in cerebral infarction (mTICI) score 2c or 3 21 has only lately been widely accepted. 22 Third, and finally, using an imaging technique that directly assesses tissue perfusion is required to reliably evaluate reperfusion. To this day, no published study on no-reflow fulfills these three key criteria (see Discussion).
In the present proof-of-principle study, we therefore aimed to assess the incidence of no-reflow in a strictly selected sample of thrombectomized LVO stroke patients with DSA-proven TICI2c–3 recanalization. Based on available pre-clinical, particularly non-human primate, literature, no-reflow was operationally defined as severe focal hypoperfusion causing tissue infarction. Importantly, post-recanalization perfusion was evaluated by means of arterial spin labeling (ASL), a non-invasive MR-based technique that provides truly quantitative perfusion maps without the limitations of contrast agents23,24 and that has been validated against conventional methods in both healthy controls and AIS patients.25–30
Methods
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Operational definition of no-reflow
No-reflow by definition entails severe tissue hypoperfusion despite full recanalization of the previously occluded territory.9,10,12,13,19 For the present study, we operationally defined no-reflow as ≥40% reduction in cerebral blood flow (CBF) affecting anatomical regions of the affected hemisphere on 24-h ASL maps. This cut-off was based on a previous ASL study that demonstrated that ≥40% CBF reduction relative to contralateral regions best identifies critical hypoperfusion, with high correlation with standard first-pass gadolinium perfusion-weighted imaging (PWI) sequence. 26 Anatomical regions were based on Alberta Stroke Program Early CT Score (ASPECTS), 31 which was recently used by Yu et al. to validate a score of ASL-based reperfusion after AIS. 32
The other key operational component to identify no-reflow was tissue outcome. As no-reflow is consistently associated with tissue infarction,10,12,13,19 we considered no-reflow to be present only if the severely hypoperfused ASPECTS area as defined above also exhibited infarction on 24 h follow-up MRI.
Study population
For the present single-center study, we retrospectively searched the Sainte-Anne Hospital prospective database of all consecutive patients treated by MT, which includes patients either directly admitted (“mothership”) or referred for MT to Sainte-Anne from other centers (“drip and ship”). As per current French regulations, prospective inclusion of patients in this database has received formal regulatory agreement. Because the present study only implied retrospective analysis of anonymized data collected as part of routine care, formal approval by an Ethics Committee was not required, but patients were offered the possibility to withdraw.
Inclusion criteria for the present study were as follows: (1) anterior circulation AIS treated between January 2014 and May 2019; (2) intracranial internal carotid artery (ICA) or first segment MCA occlusion (i.e. proximal LVO, the pre-clinical situation where no-reflow has been best documented10,12,13,16); (3) successful recanalization by means of MT, defined as a modified TICI 2c or 3 21 according to an experienced neuroradiologist (WBH) who reviewed the DSA dataset blinded to findings on 24 h ASL; and (4) availability of MRI both before MT (“MRI-1”) and ∼24 h after MT (“MRI-2”; including an ASL sequence). Thrombolysis performed prior to MT according to international guidelines was not a cause of exclusion.
Exclusion criteria were (1) presence on MRI-2 of parenchymal hematoma (ECASS classification 33 ), which may hinder assessment of ASL maps; 34 (2) re-occlusion, new intracranial occlusion or ipsi- or contralateral ICA or MCA ≥ 50% stenosis on MR angiography (MRA)-2, which may affect brain perfusion; and (3) un-interpretable ASL images due to movement artifacts, technical problems, or inordinately low whole brain CBF (<20 mL/100 g/min).
Because no-reflow is expected to expand tissue necrosis, a secondary objective of the study was to assess MRI-1 to MRI-2 infarct growth. To limit the chance of marked pre-recanalization infarct growth, an additional exclusion criterion was MRI-1-to-groin puncture delay ≥3 h.
MRI protocol
In France, MRI is recommended as first line diagnostic imaging work-up in candidates to recanalization and is implemented 24/7 at the Sainte-Anne comprehensive stroke center. At Sainte-Anne hospital, a follow-up MRI (MRI-2) is carried out around 24 h after treatment whenever feasible. All MRI data were acquired using a 1.5 T MRI scanner (General Electric Healthcare). As per the above inclusion criteria, patients were included in the present study only if they had undergone both the acute and follow-up MRI sessions, and if the latter included ASL.
The following standard sequences were acquired: (i) diffusion-weighted imaging (DWI) with b = 0 s/mm2 and b = 1000 s/mm2, 6-mm-thick axial slices; (ii) fluid-attenuated inversion recovery (FLAIR); (iii) T2*, to determine the presence of hemorrhages; and (iv) 3D time-of-flight MRA, to determine the presence and localization of intracranial occlusion on MRI-1 and the absence of re-occlusion on MRI-2. In addition, ASL perfusion maps were obtained at MRI-2 using a 3D pseudo-continuous sequence acquired using the following parameters: 4.5 min duration, 3.5 × 3.5 × 4 mm resolution, TR/TE = 4600/10.5 ms, and pairs of control/label=3, post-labeling delay 2000 ms, and 40 slices with a 4.0-mm thickness with whole brain coverage and background suppression in order to suppress signal from static tissue and hence improve signal-to-noise ratio while reducing sensitivity to motion artifacts. ASL-CBF parametric perfusion maps were automatically calculated from the labeled and unlabeled ASL images (M0) with the READY VIEW® software (GE Healthcare).
Whenever feasible, MRI-1 also included a PWI sequence as well as, whenever needed, a contrast-enhanced cervical MRA to guide decision making regarding MT.
Identification of no-reflow on ASL maps
Visual analysis
Visual analysis was performed independently by two investigators with ASPECTS training (AtS and CP), blinded to clinical information except stroke side. Because anatomical identification may be difficult on ASL maps, the latter were rigidly co-registered to the 6 mm-thick 24-h DWI and FLAIR images, using General Electric Volume Viewer® software. Then, replicating the previously validated approach, 32 the investigators were instructed to select the two axial slices best matching the standard ASPECTS slices 31 and to assess the affected hemisphere across the 10 ASPECTS regions (which have no formal boundaries 31 ) for the presence of severe hypoperfusion as compared to contralateral mirror ASPECTS region. Any ASPECTS region with old infarction on MRI-1 FLAIR or hemorrhagic infarction type-2 on MRI-2 T2* was excluded. To confirm their judgment, the investigator positioned on the co-registered DWI image a circular or elliptic two-dimensional region of interest (ROI) encompassing the hypoperfused region, and mirrored the ROI in the contralateral hemisphere. As per the operational criteria above, severe hypoperfusion was retained if mean CBF in the affected ROI was reduced ≥40% relative to mirror ROI. Discrepancies between readers were to be resolved by consensus. As per the operational definition, any eligible ASPECTS region was then assessed for the presence of infarction on MRI-2. In addition, they were also assessed for the presence on MRI-1 of DWI hyperintense signal as well as hypoperfusion (whenever PWI was available; no-reflow being expected to develop in severely hypoperfused areas 19 ).
Automatic analysis
To underpin the results of the visual analysis, we implemented an independent, fully quantitative and automated analysis replicating Yu et al.’s method. 32 To this end, an ASPECTS ROI template was created using the MANGO software, version 4.0.1 (Research Imaging Institute, University of Texas Health Science Center), in the T2 Montreal Neurological Institute (MNI) 152 space, which contains 152 axial sections each 2 mm thick. In order to match the visual analysis, three contiguous MNI sections (thickness: 2 mm) were selected by the first author for each of the two standard ASPECTS cuts, and the template was created by drawing the ASPECTS ROIs on each of the six resulting sections, 32 based on the AAL-2 atlas 35 and the study by Tatu et al. 36
To apply the MNI-space ASPECTS template onto each patient’s ASL images, we implemented the post-processing pipeline illustrated in the Supplemental Figure, using Matlab R2018b (The MathWorks, Inc., Natick, MA, USA) via SPM12 (Wellcome Center for Human Imaging, UCL, UK) and the Clinical Toolbox (CRNL, University of South Carolina, USA). As initial normalization step, we elected to normalize the b = 0 s/mm2 maps into the MNI space rather than directly the ASL into this space, because ASL maps are functional, as opposed to b = 0 s/mm2 being anatomical. This approach is recommended for efficient normalization. 37 To co-register the ASL maps into MNI space, we then registered the b = 0 s/mm2 image with the M0 ASL sequence rather than the ASL derived CBF maps, as the former contains greater anatomical information. Once the ASL dataset was co-registered to the MNI 152 template, the ASPECTS ROI template was simply projected on the six ASL slices. The CBF value for each ASPECTS region was then obtained by computing the weighted average of the CBF values of the three original ROIs belonging to this region. As with the visual analysis, the presence of no-reflow was retained whenever CBF in an ipsilesional ASPECTS ROI was reduced ≥40% relative to contralateral mirror ROI.
Clinical and imaging variables
The following data were collected:
Clinical data: age, sex, vascular risk factors, history of atrial fibrillation, pre-stroke modified Rankin scale (mRS); time since last-seen-well, time of admission, and National Institutes of Health Stroke Scale (NIHSS) score. Imaging data: occlusion site (ICA, proximal or distal MCA-M1, tandem), DWI lesion site (deep or cortical), and volume for both MRI sessions: using MANGO software, DWI lesions were semi-automatically segmented, and volumes were extracted. Thrombolysis and time of thrombolysis start, groin puncture time, and mTICI score. Cause of stroke (TOAST criteria).
38
NIHSS at 24 h; early neurological improvement or deterioration (≥8-point decrease in 24 h NIHSS relative to baseline or 24 h NIHSS ≤4 and ≥4-point increase on 24 h NIHSS, respectively39,40). Three-month follow-up data: mRS; favorable outcome was defined as mRS ≤ 2.
Statistical analysis
Data were analyzed using SPSS Statistics Version 25 (IBM, Armonk, NY). Kappa coefficients were calculated to characterize inter-rater agreement for the visual analysis. To analyze differences between included and non-included patients, continuous variables were compared using the Wilcoxon rank-sum test, and categorical data were analyzed by means of Fisher exact test. Tests were bilateral and acceptable type I error was set a priori at α = 0.05. Means are presented as mean ± SD, medians as median (IQR).
Results
Study population
Thirty-three patients fulfilled the inclusion criteria. Figure 1 presents the study flowchart including the causes for exclusion. Because in our center most drip-and-ship patients return to their referring stroke center immediately following the procedure, only three drip-and-ship patients had MRI-2 including ASL in the registry; however, they were ineligible to the present study because the delay from pre-MT MRI and groin puncture was >3 h. Consequently, only mothership patients were included in the analysis.
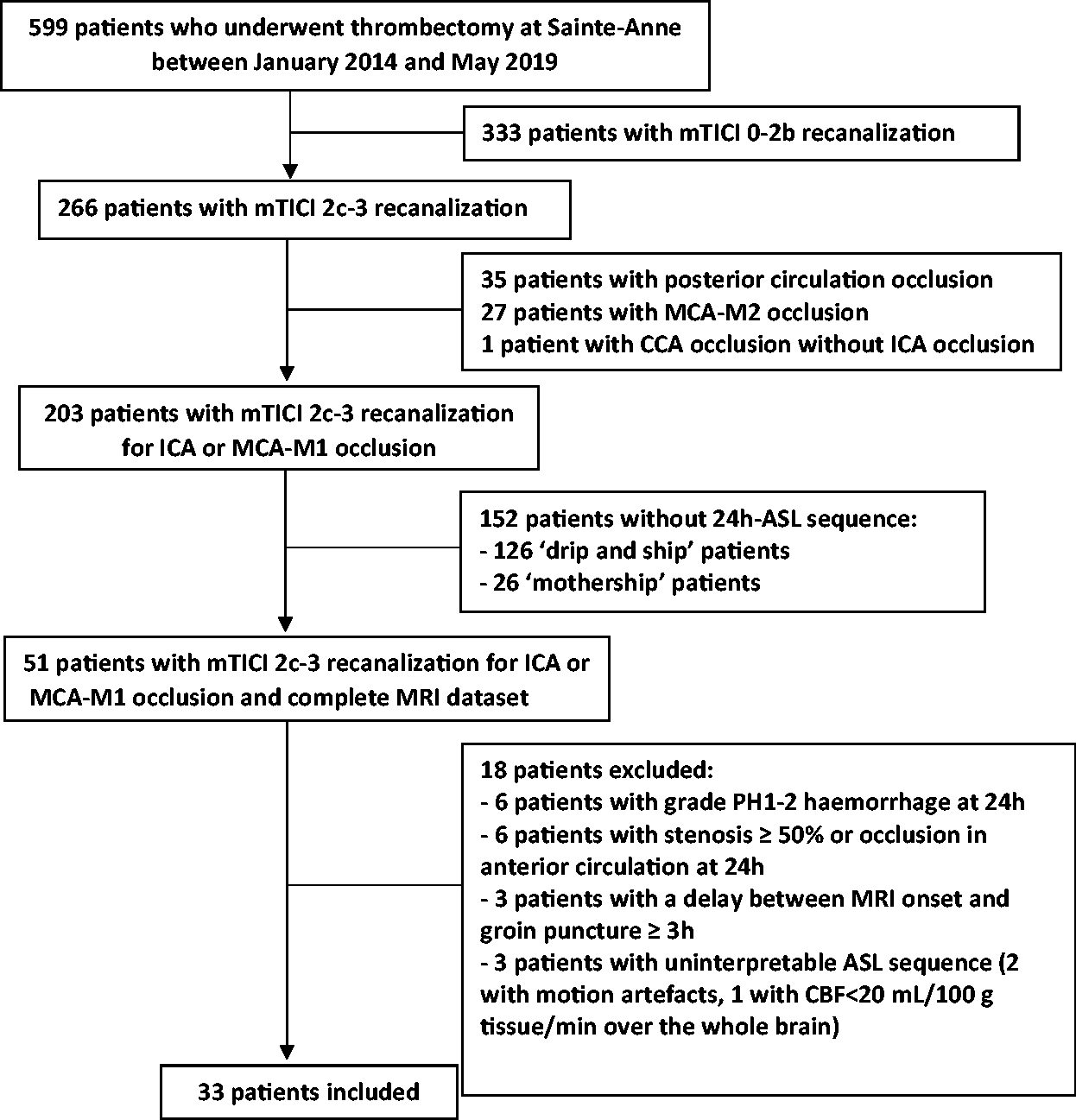
Study flowchart.
The baseline characteristics, clinical and imaging data, and treatment received and outcome data are summarized in Tables 1 and 2. Unfavorable outcome (mRS > 2) was present in 12 patients. There was a significant association between unfavorable outcome and larger admission DWI lesion volume (p = 0.001; Mann–Whitney).
Clinical and radiological characteristics.
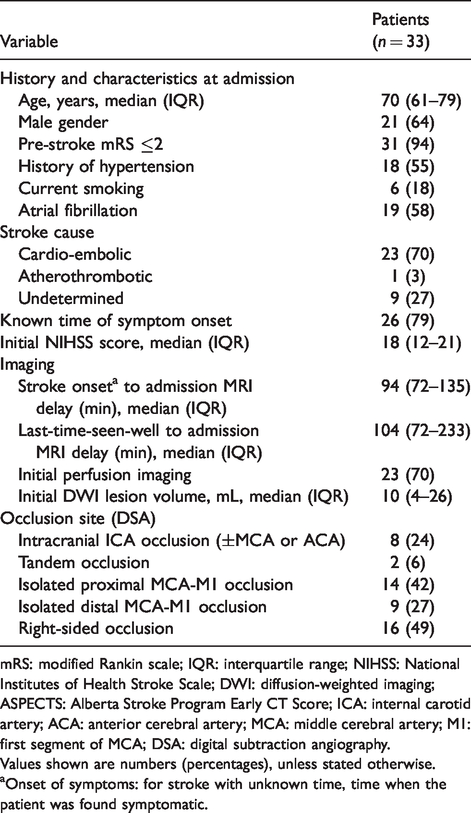
mRS: modified Rankin scale; IQR: interquartile range; NIHSS: National Institutes of Health Stroke Scale; DWI: diffusion-weighted imaging; ASPECTS: Alberta Stroke Program Early CT Score; ICA: internal carotid artery; ACA: anterior cerebral artery; MCA: middle cerebral artery; M1: first segment of MCA; DSA: digital subtraction angiography.
Values shown are numbers (percentages), unless stated otherwise.
aOnset of symptoms: for stroke with unknown time, time when the patient was found symptomatic.
Treatment and follow-up data.

IQR: interquartile range; mTICI: modified thrombolysis in cerebral infarction; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin scale.
Values shown are numbers (percentages), unless stated otherwise.
aOnset of symptoms: for stroke with unknown time, time when the patient was found symptomatic.
b≥8-Point decrease in 24 h NIHSS relative to baseline, or 24 h NIHSS ≤ 4.
c≥4-Point increase on 24 h NIHSS.
Twenty-six mothership patients with TICI 2c–3 recanalization for ICA or M1 occlusion did not undergo follow-up ASL imaging (Figure 1), of which 14 fulfilled the other inclusion/exclusion criteria for this study. None of the baseline and follow-up characteristics of these 14 patients significantly differed from those of the 33 included patients (Supplemental Table 1).
No-reflow analysis
Across the 33 included patients, eight ASPECTS regions were excluded a priori because of ipsilateral old infarct or hemorrhagic transformation, or contralateral old infarct (n = 3, 3, and 2, respectively). Accordingly, 322 ipsilateral ASPECTS regions were available for final analysis.
Visual analysis
No instance of severe focal hypoperfusion was identified in 32/33 patients. In the remaining patient, both investigators independently identified severe hypoperfusion in the same two ASPECTS regions, namely the caudate and lentiform (Figure 2). Inter-observer reproducibility was therefore perfect (kappa = 1). Both regions were infarcted on MRI-2, and therefore fulfilled the operational criteria for no-reflow. Importantly, neither exhibited DWI hyperintensity on MRI-1, indicating they underwent infarction subsequently. Likewise, they did not exhibit perfusion abnormalities on baseline PWI. This patient had early neurological recovery (24-h NIHSS = 1) and excellent outcome (three-month mRS = 0). Further clinical and imaging details are provided in the figure legend.
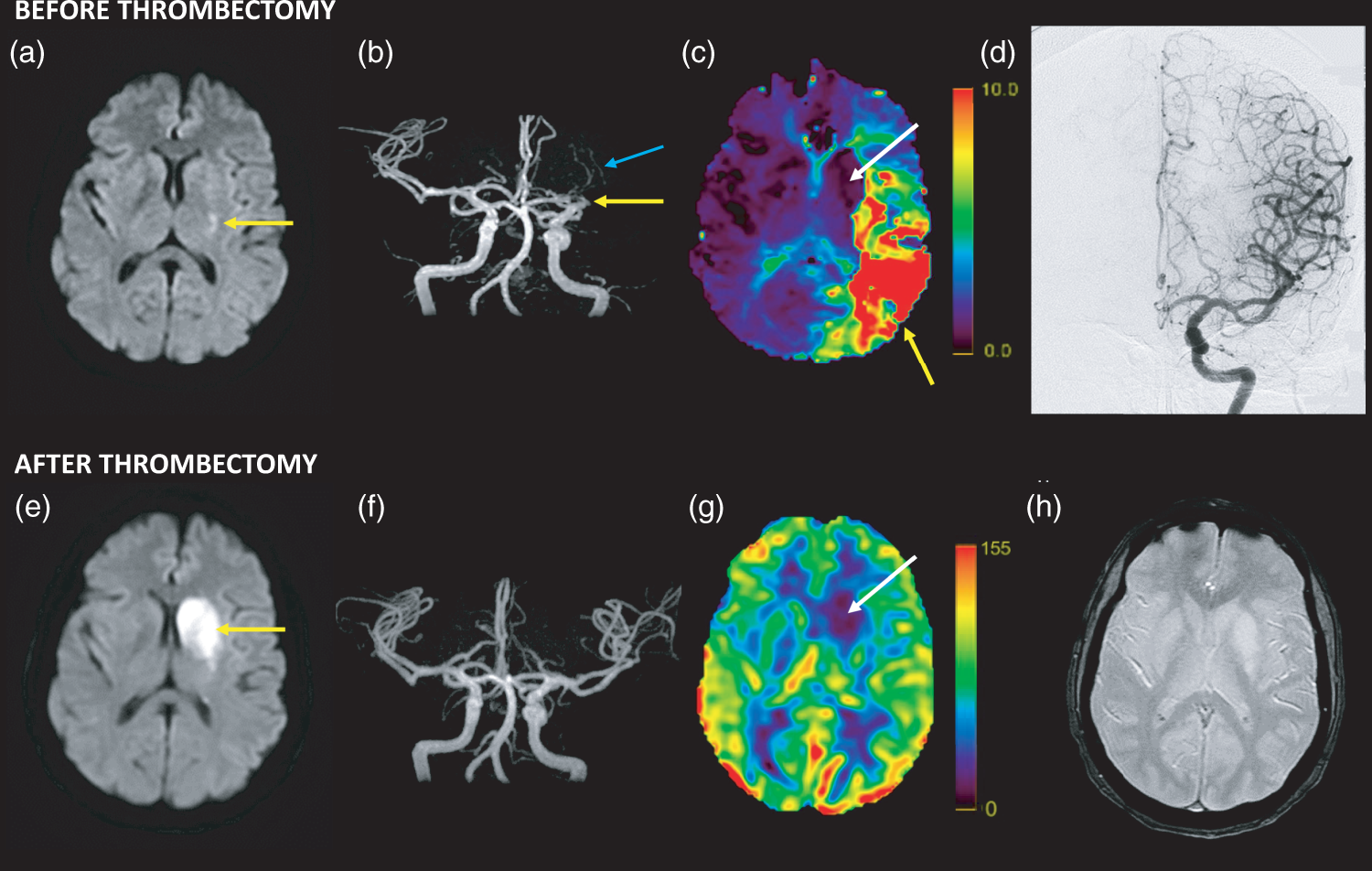
Co-registered imaging data for the single patient in whom no-reflow was identified. The patient had sudden-onset severe hemiparesis (three levels) and global aphasia. On admission to our hospital, the neurological deficit had partly resolved and the patient had mild three-level hemiparesis and non-fluent dysphasia (NIHSS = 4). She was treated with thrombectomy 160 min after symptom onset. (a–c) MRI-1 obtained 84 min after symptom onset, showing a small, faint DWI lesion involving the posterior-most lentiform nucleus (a, arrow), an MCA-M1 occlusion on time-of-flight angiogram (b, yellow arrow) distal to the lenticulo-striate arteries (blue arrow), and on PWI (Tmax; the pseudo-color scale ranges from 0 to 10 s) an extensive area of severe hypoperfusion covering a large part of the left cortical MCA territory (yellow arrow), but sparing the deep MCA territory (white arrow) (c). (d) Digital subtraction arteriography obtained immediately following thrombectomy, showing mTICI 3 recanalization. (e–h) Twenty-four-hour MRI-2 showing extension of the DWI lesion to the entire caudate and lentiform nuclei (e), without re-occlusion on time-of-flight MRA (f). These newly infarcted areas are severely hypoperfused on 24 h ASL-CBF maps (g; the pseudo-color scale is in milliliters/100 g/min), fulfilling the operational criteria for no-reflow for both regions (CBF relative to mirror region: –63% and –55%, respectively). No hemorrhage was present across the MCA territory on T2* MRI-2 (h). (a, c, e, g, and h) Images co-registered using GE Volume Viewer (see Methods). Stroke etiology was atrial fibrillation. This patient enjoyed early neurological recovery (24-h NIHSS = 1) and excellent outcome (three-month mRS = 0).
Automatic analysis
Consistent with the visual analysis, no ASPECTS ROI exhibited severe hypoperfusion in any patient save for the patient already identified with the visual analysis, involving the caudate nucleus (CBF = –73%) but not reaching the prescribed CBF threshold for the lentiform ROI (–22%).
Mild hypoperfusions
Given the rather unexpected finding that no-reflow, as operationally defined here, was present in one patient only in our cohort, as compared to the widely cited prevalence of ∼25%, 19 we carried out a post hoc analysis assessing milder hypoperfusions. Using the same visual analysis procedure as described above, each ASPECTS region in the affected hemispheres (saved for the two severely hypoperfused regions just described) was classified by the two independent observers into four categories, according to the presence or absence of (i) hypoperfusion not fulfilling the criteria for severe hypoperfusion and (ii) infarction on concomitant DWI. The inter-observer reproducibility was again very good. Of 33 patients, 11 had at least one mildly hypoperfused ASPECTS region (Supplemental Table 2). Across the whole sample, 43/320 ASPECTS regions displayed mild hypoperfusion, of which two only were infarcted on MRI-2, both involving the basal ganglia in a single patient (Figure 3). Of note, however, both regions already showed definite diffusion abnormality at baseline. The remaining 41 mildly hypoperfused but non-infarcted ASPECTS regions were all cortical, except one involving the caudate.
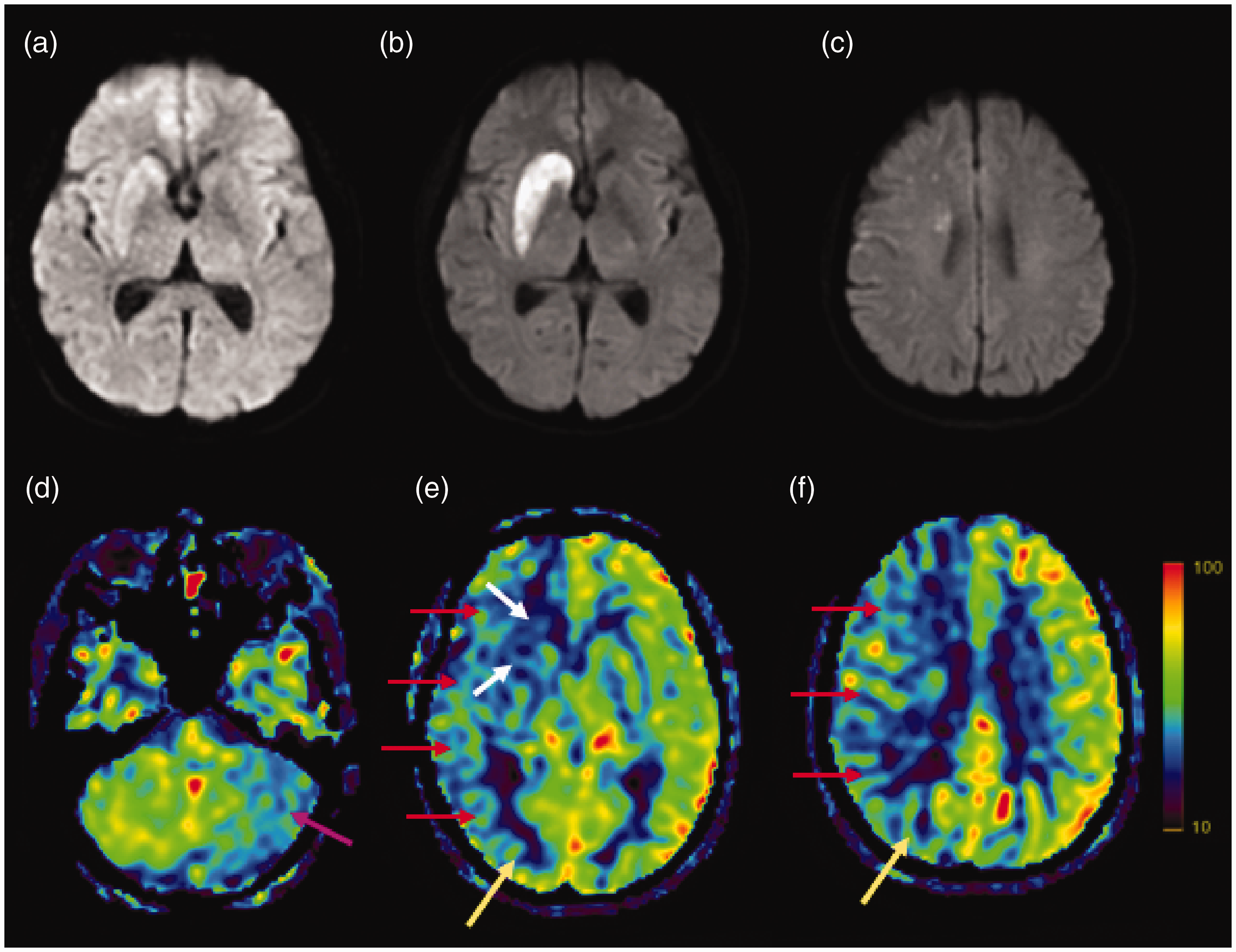
Illustrative image dataset of the patient with mild hypoperfusion associated with infarction. This 66-year-old patient presented with acute-onset moderate deficit (NIHSS score = 11) and was treated with IV thrombolysis followed by thrombectomy for right terminal ICA occlusion, with mTICI 2c recanalization obtained 215 min after symptom onset. Perfusion MRI was carried out prior to thrombolysis in this patient but unfortunately the data were not interpretable due to marked head motion and technical errors. (a–c) Illustrative DWI b = 1000 s/mm2 sequences obtained (i) on admission (68 min after symptom onset) (a), and at 24 h follow-up (b and c), showing a stable DWI lesion in the striato-capsular area (i.e. deep MCA territory); (d–f) 24 h follow-up ASL-CBF maps, showing mild hypoperfusion within the DWI lesion (15% and 17% reduction in the caudate and lenticular nuclei, respectively, relative to contralateral ASPECTS regions; white arrows), but also cortically at distance from the DWI lesion (20–35% reduction relative to contralateral ASPECTS regions; red arrows), spreading beyond the ICA territory (i.e. posterior circulation, yellow arrows; note that in this patient both posterior cerebral arteries originated from the basilar artery on MRA, data not shown). The left cerebellar hemisphere, i.e. contralateral to the stroke, also shows mild hypoperfusion compared to the right cerebellum (purple arrow), reflecting crossed cerebellar diaschisis. The patient made a good recovery (24 h NIHSS score = 1, three-month mRS = 1).
Discussion
Consistently with both the visual and automatic analyses, there was evidence of no-reflow, as operationally defined in the present study, in one out of 33 patients only. This somewhat unexpected finding suggests that no-reflow may be a rare phenomenon, unlikely to substantially contribute to “futile” recanalizations.
Supporting this interpretation, the only patient who fulfilled the no-reflow criteria in our cohort had excellent outcome (mRS = 0), while none of the 11 patients with poor outcome (mRS > 2) did. Conversely, there was a strong association between unfavorable outcome and larger baseline DWI lesion volume, consistent with extensive previous literature.41–43 Thus, large baseline core, rather than no-reflow, may be the main factor accounting for futile recanalizations.
Interestingly, in the patient fulfilling the no-reflow operational criteria, the affected basal ganglia was not initially hypoperfused and only exhibited a small faint diffusion abnormality in the lentiform’s posterior-most part, consistent with the fact that the lenticulo-striate arteries were patent on admission MRA (Figure 3). In retrospect, the clinical events prior to admission suggest initially more proximal M1 occlusion followed by spontaneous distal clot migration, resulting in clinical improvement and spontaneous reperfusion of the basal ganglia preventing extensive damage. 44 Although lack of baseline hypoperfusion is unexpected for no-reflow, which is thought to develop in ischemic tissue, 19 the initial ischemic insult might have triggered delayed no-reflow. Two alternative mechanisms may account for basal ganglia delayed hypoperfusion and infarction in this patient, namely inadvertent occlusion of lenticulo-striate arteries during, and cardio-embolic recurrence after the procedure, both implying another ischemic event rather than no-reflow.
The discrepancy between the visual and automatic analyses regarding this patient’s lenticular nucleus can be explained by the fact that severe hypoperfusion on 24 h ASL spared its postero-lateral part (Figure 2), diluting the average CBF in the lentiform ASPECTS ROI used in the automatic analysis. Although the visual analysis of ASL maps has been validated against the template-based analysis, 32 our data suggest that it is more sensitive to focal hypoperfusion not affecting entire ASPECTS ROIs.
How do our findings compare to published literature? The characteristics and findings of all 11 previously published articles that investigated impaired reperfusion despite recanalization as a surrogate for no-reflow5–7,25,45–51 are presented in Table 3. The key observation is that all the studies had significant methodological shortcomings (see Introduction), calling their findings into question. Thus, two studies only used DSA to assess recanalization,45,47 while the rest used techniques such as transcranial Doppler, MRA, or CTA, which can miss distal occlusions. Importantly, those two studies that used DSA to assess recanalization45,47 unfortunately had major limitations: one defined recanalization as either partial or complete, 45 and in the other DSA was used to assess reperfusion. 47 Nine studies reported no-reflow rates of 21–40%, which due to these limitations may be overestimated. However, the two remaining studies, though affected by similar drawbacks, reported no instance of hypoperfusion following recanalization. In one study, 25 hypoperfusion on 24 h ASL was present in 41/100 patients (all receiving medical treatment only), however none of these patients had complete recanalization on 24 h MRA, suggesting absence of no-reflow—a finding not discussed by the authors. Another study 7 reported no instance of no-reflow in 13 patients with full recanalization on follow-up MRA, but most initially had distal (M2) occlusions, where no-reflow might be difficult to detect, while reperfusion was assessed ≤3 h of admission MRI, which might be too early to reliably assess no-reflow. Despite these caveats, these two studies are consistent with our findings, i.e. they suggest that no-reflow is infrequent. Also favoring a low prevalence of no-reflow, another study (not listed in Table 3 as its focus was not on tissue reperfusion) quoted in its Methods section a rate of incomplete reperfusion in mTICI3 thrombectomized patients of 7% only. 52 The data reported in the EXTEND-IA 53 and DEFUSE-3 54 thrombectomy trials also appear to be consistent with a low prevalence of no-reflow: although in both reports the actual data on reperfusion after complete recanalization are missing and the latter included TICI 2b, the reported reperfusion rates were 89% and 79%, while recanalization rates were 86% and 76%, respectively.
Previously published articles (listed in chronological order) on cerebral hypoperfusion in recanalized stroke patients, and present study.
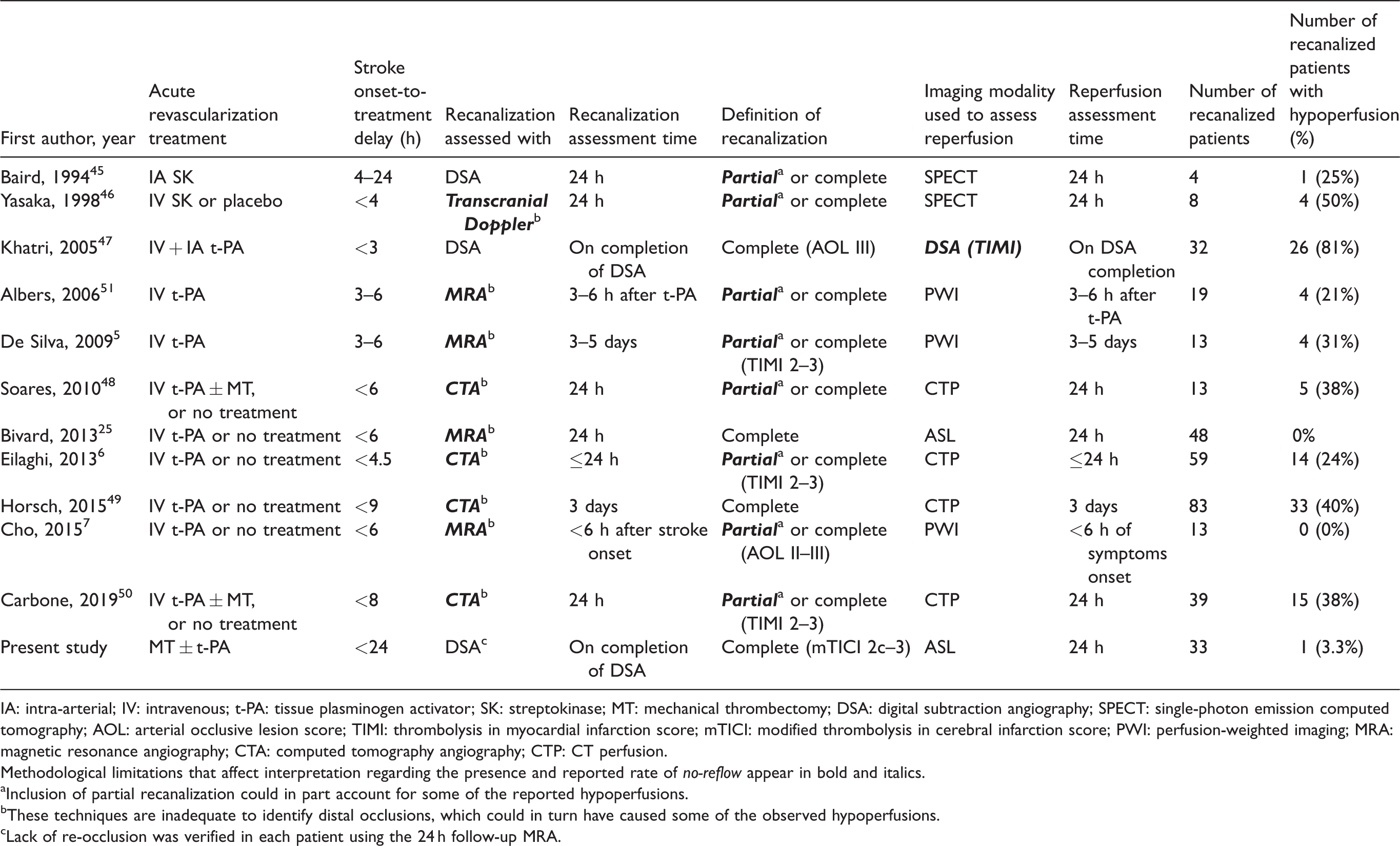
IA: intra-arterial; IV: intravenous; t-PA: tissue plasminogen activator; SK: streptokinase; MT: mechanical thrombectomy; DSA: digital subtraction angiography; SPECT: single-photon emission computed tomography; AOL: arterial occlusive lesion score; TIMI: thrombolysis in myocardial infarction score; mTICI: modified thrombolysis in cerebral infarction score; PWI: perfusion-weighted imaging; MRA: magnetic resonance angiography; CTA: computed tomography angiography; CTP: CT perfusion.
Methodological limitations that affect interpretation regarding the presence and reported rate of no-reflow appear in bold and italics.
aInclusion of partial recanalization could in part account for some of the reported hypoperfusions.
bThese techniques are inadequate to identify distal occlusions, which could in turn have caused some of the observed hypoperfusions.
cLack of re-occlusion was verified in each patient using the 24 h follow-up MRA.
Another piece of evidence mitigating against a high prevalence of significant no-reflow in our population was the negligible infarct volume growth from MRI-1 to MRI-2 (median = 2 mL; Table 2), which is consistent with a recent thrombectomy study. 55 Although a portion of the acute DWI lesion may revert on 24 h follow-up MRI in some patients, it is now widely acknowledged that the latter provides a valid metric for final infarct. 56
In the present study, we considered complete recanalization as a key inclusion criterion in order to reliably assess no-reflow. Accordingly, only patients with TICI 2c–3 arteriographic reperfusion grades were considered eligible. As a note of caution, TICI 2c definition is “near-complete perfusion except for slow flow in a few distal cortical vessels or presence of small distal cortical emboli”, as opposed to TICI 3 which is normal angiogram.21,22 If one considered only TICI 3 as true complete recanalization, the incidence of no-reflow would be one in 11 patients, i.e. <10%. However, the lack of no-reflow in the 22 patients with TICI 2c would be unexpected given this arteriographic grade implies poorer distal contrast clearance. Interestingly, recent studies point to no significant difference in clinical outcome between the TICI 3 and TICI 2c grades. 22
An entirely different hypothesis to account for our observation would posit that no-reflow may exist mainly with longer occlusion times. In primates, no-reflow was prominent only after 3–4-h MCA occlusion,10,11,13,57 and it was absent following 2-h occlusion in a rat study. 58 Conversely, however, some pre-clinical studies have observed no-reflow as early as 1 h after occlusion.14,18 Our patients received thrombectomy as per the mothership paradigm, and accordingly the median delay between last-time-seen-well and recanalization was relatively short, namely 208 min, although the range was wide (112–750 min, with one outlier at 1325 min). Nonetheless, no-reflow was rare in our standard mothership population and in turn would not account for the poor outcomes that affected one-third of our patients despite complete recanalization (Table 2).
As almost two-thirds of our patients received thrombolysis (Table 1), could t-PA have prevented no-reflow? To our knowledge, this issue has not been addressed experimentally, but one clinical study reported a 22% no-reflow incidence despite 68% of the sample receiving thrombolysis. 49 Alternatively, could no-reflow be common only in situation of incomplete recanalization? In a thrombectomy study by Arsava et al., 59 iodine contrast was injected into the distal site of occlusive thrombi prior to stent-retriever deployment, which revealed lack of capillary blush—taken to indicate microcirculatory obstructions—in 7/34 patients, all of whom subsequently had incomplete recanalization, 59 suggesting that impaired microcirculatory flow might impact recanalization per se. However, post-thrombectomy perfusion status was not assessed in this study, and the above pattern was not seen in patients who fully recanalized, making these observations of unclear relevance to the no-reflow phenomenon.
In the present study, presence of PH was a cause of exclusion as it would markedly hinder interpretation of 24 h perfusion maps. This is unlikely to have affected our finding, as no-reflow by definition prevents tissue reperfusion, and accordingly no-reflow was not associated with PH in primate models,10–13 while one rodent study only reported associated micro-hemorrhages on pathology. 16 In the above-mentioned Arsava et al.’s study, 59 the sole PH-2 case was a patient without evidence of microcirculatory obstruction. Likewise, we excluded a priori all ASPECTS regions with hemorrhagic infarction type-2, which might per se affect regional perfusion. However, this concerned only three regions, none of which displayed significant hypoperfusion on post hoc assessment, consistent with the established notion that hemorrhagic transformation is associated with post-recanalization hyperperfusion. 60
Taken together, therefore, our findings suggest that no-reflow, as operationally defined here, may be at best infrequent in human stroke. How can this be reconciled with the pre-clinical evidence? Given the animal, including primate, literature,9,10,12,13,19 where no-reflow is defined as markedly impaired or absent capillary perfusion, we considered severe hypoperfusion a key operational criterion to define no-reflow and used as cut-off the value of 40% previously validated in earlier ASL studies as a surrogate to Tmax > 6 s, the validated penumbra threshold.23,26 Using this definition, one patient only out of 33 with complete recanalization showed possible no-reflow, which prompted us to revisit post hoc our dataset to assess milder hypoperfusions. Assessing milder hypoperfusions would also address the seminal observation made by Ames et al. following complete cerebral circulation arrest that lack of capillary filling involved large confluent areas interspersed with “mottled” areas. 9 If mottled no-reflow also existed in human stroke, and considering the limited spatial resolution of ASL, it would translate as milder hypoperfusion. The results of the post hoc analysis revealed that mild hypoperfusions were relatively common, with 11/33 patients showing at least one mildly hypoperfused ASPECTS region, for a total of 43 regions across the cohort. However, two such regions only were infarcted on MRI-2, both involving the basal ganglia in a single patient. Although these two regions might represent a milder form of no-reflow, it is important to note that they were already infarcted on admission DWI (Figure 3). This kind of mild no-reflow developing in already infarcted brain tissue would arguably have little clinical relevance.
Could mottled no-reflow exist without frank tissue infarction? One might raise the possibility that the penumbra rescued from infarction might be the seat of microvascular impairment as a result of either or both pre-reperfusion ischemic endothelial and pericyte damage17,61 and reperfusion injury, 62 potentially causing selective neuronal loss (SNL), which has no counterpart on conventional MRI. 63 Occurrence of SNL in the salvaged penumbra is well established histopathologically in rodent models of transient MCA occlusion 63 and is strongly supported by 11C-flumazenil PET in stroke patients with early reperfusion.64–66 However, although in rodents SNL slows down sensori-motor recovery,67,68 no clinical counterpart has been documented so far in human stroke.65,69 Further studies of no-reflow combining 24-h ASL and delayed 11C-flumazenil PET would be important to address this hypothesis.
What additional mechanisms could underly the mild hypoperfusions observed on 24 h ASL maps, knowing that patients with persistent significant arterial stenosis/occlusion on 24 h MRA were a priori excluded from the present study? One likely mechanism is ipsilesional diaschisis, i.e. functional suppression in areas remote from but connected to the damaged area—a well-established, widely prevalent phenomenon after stroke, which frequently involves the cerebral cortex overlying deep MCA territory infarcts, so-called intra-hemispheric diaschisis.70–72 Interestingly, all 11 patients with cortical hypoperfusion in our sample had deep MCA infarcts, supporting this idea (Supplemental Table 2). To further address this hypothesis, we assessed for the presence of crossed cerebellar diaschisis (CCD), a type of diaschisis known to frequently associate with cortical diaschisis.73,74 CCD was present in 6/28 patients and was significantly more frequent in those with, as compared to those without, mildly hypoperfused cortical regions (p = 0.038, Fisher). These findings are illustrated in Figure 3; please note also in this patient the extension of the cortical mild hypoperfusion beyond the MCA territory, which would also favor diaschisis. Intra-hemispheric diaschisis therefore likely accounts for a substantial fraction of the mild cortical hypoperfusions observed in our cohort.
Our mechanistic study has limitations. Given the relatively small sample and retrospective design, its findings warrant confirmation in prospective cohorts. However, our cohort was extracted from a large prospective database of consecutively thrombectomized patients (Figure 1), using stringent criteria required to match our operational definition, notably both TICI 2c–3 recanalization and availability of 24 h ASL. As a result of our choice to replicate the only previously validated method to assess post-thrombectomy ASL maps, 32 severe hypoperfusion was evaluated in ASPECTS regions only. To address this potential limitation, a post hoc visual analysis was carried out assessing all axial sections across the whole sample, which did not identify any additional severely hypoperfused region (data not shown). Finally, our results are applicable only to the population studied, namely mothership patients with proximal occlusion.
Summary
In our cohort of patients with proximal occlusion who enjoyed complete and stable post-thrombectomy recanalization, severe focal hypoperfusion in the affected vascular territory on 24 h follow-up imaging associated with newly developed infarction was a rare occurrence. This unexpected finding may suggest that in human stroke no-reflow is in fact an infrequent phenomenon that may not substantially contribute to “futile” recanalizations or stand as an important therapeutic target. Prospective studies are however warranted to confirm our findings.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X20954929 - Supplemental material for Tissue no-reflow despite full recanalization following thrombectomy for anterior circulation stroke with proximal occlusion: A clinical study
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X20954929 for Tissue no-reflow despite full recanalization following thrombectomy for anterior circulation stroke with proximal occlusion: A clinical study by Adrien ter Schiphorst, Sylvain Charron, Wagih Ben Hassen, Corentin Provost, Olivier Naggara, Joseph Benzakoun, Pierre Seners, Guillaume Turc, Jean-Claude Baron and Catherine Oppenheim in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AtS received a scholarship from the Société Française Neurovasculaire (SFNV) to carry out this project as part of his MSc.
Acknowledgements
The authors are indebted to Ms Stéphanie Lion for expert technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
AtS: conception and design of the study; acquisition and analysis of data; drafting of the manuscript and figures; SC: analysis of data; drafting a significant portion of the manuscript; WBH and CP: data analysis; ON and JB: acquisition and analysis of data; PS and GT: data acquisition; J-CB: conception and design of the study; drafting of the manuscript and figures; CO: conception and design of the study; data acquisition; drafting a significant portion of the manuscript and figures.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
