Abstract
Collateral number/density varies widely in brain and other tissues among strains of Mus musculus mice due to differences in genetic background. Recent studies have shown that prolonged exposure to reduced atmospheric oxygen induces additional collaterals to form, suggesting that natural selection may favor increased collaterals in populations native to high-altitude. High-altitude guinea pigs (Cavia) and deer mice (Peromyscus) were compared with lowland species of Peromyscus, Mus and Rattus (9 species/strains examined). Collateral density, diameter and other morphometrics were measured in brain where, importantly, collateral abundance reflects that in other tissues of the same individual. Guinea pigs and high-altitude deer mice had a greater density of pial collaterals than lowlanders. Consistent with this, guinea pigs and highlander mice evidenced complete and 80% protection against stroke, respectively. They also sustained significantly less ischemia in heart and lower extremities after arterial occlusion. Vessels of the circle of Willis, including the communicating collateral arteries, also exhibited unique features in the highland species. Our findings support the hypothesis that species native to high-altitude have undergone genetic selection for abundant collaterals, suggesting that besides providing protection in obstructive disease, collaterals serve a physiological function to optimize oxygen delivery to meet oxygen demand when oxygen is limiting.
Keywords
Introduction
In the setting of acute occlusion or chronic narrowing of the trunk or a branch of an arterial tree, collaterals connecting to an adjacent tree(s) provide retrograde perfusion of the obstructed territory.1–4 The amount of collateral blood flow and how broadly it is distributed depend primarily on the number and diameter of the collaterals that are present (i.e. the extent or abundance of native collaterals). In mice, collateral extent exhibits remarkably wide variation due to differences in genetic background. Cerebral (pial) collateral number varies by 30-fold and diameter by 3-fold among 21 inbred strains, and infarct volume following permanent middle cerebral artery occlusion inversely mirrors these differences. 5 , 6 Similar variation is present in other tissues of the same mouse of a given strain, 7 , 8 indicating that collateral extent in one tissue reflects that in other tissues of the same individual. These differences arise from polymorphisms in genes/genetic elements within the pathway that directs formation of collaterals during development, a process termed collaterogenesis. 6 ,9–12 Similar genetic differences in collateral abundance may also contribute to the wide variation in collateral blood flow in patients with ischemic stroke, myocardial infarction, and atherosclerotic coronary and peripheral artery disease. 2 , 4 ,13–15
It has been hypothesized that collaterals may also serve a physiological function by optimizing oxygen delivery to meet oxygen demand when oxygen availability is limited. 16 , 17 Collateral blood flow at baseline slowly ebbs to-and-fro, averaging zero net flow in the absence of arterial obstruction, owing to the lack of a difference in pressures in adjacent arterial trees. 10 , 18 When an increase in activity/metabolism occurs in a region of a tissue supplied by one of the trees, local vasodilation occurs. This creates a drop in local pressure that increases flow into the activated region which, if the vasodilation is sufficiently robust, increases local oxygen delivery to match the increase in local oxygen demand—a process termed neurovascular coupling in brain and functional hyperemia elsewhere. 19 , 20 The hemodynamic efficiency of this mechanism would be enhanced if the increase in blood flow into the activated region was supplied not only by the arterial tree serving the region but also by recruitment of flow across nearby collaterals, if present, connecting to an adjacent tree(s). An analogous principle underlies the efficient design of interconnected municipal electric power grids, wherein different communities supplied proximally by separate power plants are interconnected at their peripheries, allowing current to be drawn from more than one source when an increase in regional demand occurs.
There is indirect support for the above concept. Patients with chronic obstructive pulmonary artery disease, cyanotic heart disease, and sleep apnea have increased coronary collateral conductance,21–25 as do piglets following prolonged exposure to reduced inspired oxygen 26 and dogs made chronically anemic. 27 , 28 Humans with coronary atherosclerosis have increased collateral number. 29 Sickle cell anemia often leads to formation of moyamoya-like micro-collaterals in brain. 30 , 31 In addition, we recently reported that several weeks of exposure of mice to reduced atmospheric oxygen induced a hypoxia “dose- “and duration-dependent de novo formation of additional new cerebral collaterals and outward remodeling of the native population present at baseline. 17 Notably, neither effect reversed on return to normoxia, resulting in sustained decrease in infarct volume following acute ischemic stroke when examined many weeks later. A similar hypoxemia-induced stimulation of new collateral formation and protection against ischemic injury was also seen in the heart. 32 , 33 The collaterogenesis pathway is driven by oxygen-sensitive genes, 17 , 33 and collateral formation occurs between the crowns of adjacent arterial trees where oxygen levels are lower than elsewhere in the arterial vasculature.10–12 That collaterals exist in this vascular “outpost” may account for the effect of reduced arterial oxygen to re-activate this embryonic signaling pathway in the adult, resulting in an increase in collateral density in brain and other tissues. 17 , 32 , 33 The strength of this physiological adaption may also be subject to genetic selection for gain-of-function variants of genes within the collaterogenesis signaling pathway, leading to a greater abundance of collaterals in highlander species and populations.
A corollary arising from the hypothesis that collaterals optimize oxygen delivery to meet oxygen demand is that the advantage of abundant collaterals in healthy tissues would be minimal when oxygen availability is not limited, for example in species native to low-altitude. Consequently, natural selection for abundant collaterals would not be expected to occur in such species. 17 This could explain why collateral extent exhibits so much variation among Mus musculus mice5–8 and, potentially, among humans (see above). Mus musculus evolved at low altitude, although commensal populations have associated with human settlements at altitudes above 2500 m (8250 ft; “high altitude”) in the past few hundred years.34–36 By contrast, abundant collaterals could provide an adaptive advantage in species indigenous to high altitude, evolving through positive selection on advantageous alleles of genes that direct collaterogenesis. Interestingly, expression of many of the genes thus far identified in the collaterogenesis pathway is increased by low oxygen. 11 , 17 , 32 In support of this hypothesis, collateral blood flow in the domesticated guinea pig, Cavia porcellus, following acute coronary artery ligation was, remarkably, 100% of orthograde flow before ligation and 6-fold higher than the next highest species (dog) among eight species examined. 37 These findings were consistent with an earlier anecdotal report that guinea pigs sustained no infarction after coronary ligation. 38 Neither study commented on why guinea pigs might have such abundant coronary collaterals as suggested by their findings. However, evidence indicates that the immediate ancestor of C. porcellus is the “high montane” guinea pig, C. tschudii, that evolved at ∼4200 m in the Andes mountains beginning 12 million years ago.39–42 Cavia porcellus was domesticated 5000 years ago by humans residing at these altitudes, then brought to Europe at the end of the 16th century and bred by “fanciers”, leading to the breeds currently available for research and pets.39–41 This short time of residence at low altitude makes it unlikely that favorable alleles of genes that promote abundant collaterals would be lost, given their presence would not be envisioned to reduce fitness at low-altitude, their homozygous loss would likely be required, 9 , 12 and that the guinea pig has a relatively long gestation of ∼66 days. Additional support for the above hypothesis comes from the Swiss National Cohort Study which found that mortality from stroke and coronary artery disease was reduced by 12 and 22%, respectively, for every thousand feet of residence above 200 m, with an added benefit seen in those born at higher altitudes. 43 Mortality from stroke was also lower in high-altitude areas of Peru 44 and the United States 45 and 5-fold lower in Tibetan than in Han Chinese populations living in Lhasa (3644 m). 44 , 47 As well, the prevalence of ischemic stroke and heart disease was lower in the Tibetan versus Han populations in Lhasa and in other high- versus low-altitude populations. See literature 43 ,45–49 and references therein.
In the present study, we find that the density of native collaterals at baseline in two high-altitude breeds of Cavia porcellus and two high-altitude subspecies of the North American deer mouse, Peromyscus maniculatus, is greater and ischemic injury markedly diminished compared to that seen in lowlander conspecifics and Mus musculus mice and rats. These findings suggest that, in addition to their bypass function in obstructive disease, collaterals optimize oxygen delivery to meet oxygen demand when oxygen supply is limited and that positive genetic selection may have occurred for abundant collaterals in species native to high-altitude as a fitness advantage for survival in their low-oxygen environment.
Methods
The following animals were maintained at low altitude (89–978 m): (1) Three-to-six months-old C57BL/6J and BALB/cByJ inbred mice (Mus musculus domesticus, lowlanders, Jackson Laboratories, Bar Harbor, ME). Unless indicated otherwise, both sexes of these and the other species listed below were studied. Collateral number/density and diameter and other vascular morphometrics do not demonstrate sexual dimorphism, 50 thus n-sizes were not chosen to allow examination of sex differences. (2) Six-to-seven-months-old male and female outbred Hartley Albino and male American Tricolor guinea pigs (Cavia porcellus, highlander, Elm Hill Labs, Chelmsford, MA). (3) The following 4–10 months-old outbred male and female North American deer mouse species: Peromyscus maniculatus sonoriensis (P.m.sonoriensis, highlander, Peromyscus Genetic Stock Center, PGSC, University of South Carolina, Columbia, SC) originally caught near the summit of White Mountain, CA, 4344 m 51 , 52 ; P.m.rufinus (highlander) male 12–14 months-old F2-generation; the grandparents of these mice were caught near the summit of Mount Evans, CO, 4348 m 53 ; P.m.bairdii originally caught near Ann Arbor, MI (277 m, PGSC), conspecific lowlander to P.m.sonoriensis and P.m.rufinus. 51 , 52 (4) Three-to-six-months-old female Wistar and male and female Long-Evans outbred rats (Rattus norvegicus domesticus, lowlanders, Charles River Labs, Morrisville, NC). The above deer mouse species were obtained from PGSC in 2019 where they have been maintained outbred for many generations. The above ages are below those that are accompanied by a loss/reduction in collateral number and diameter that occurs by 22 and 16 months-age, respectively, in C57BL/6J mice (i.e. age-induced collateral rarefaction). 54 Angiography and morphometry were performed after maximal vasodilation with perfusion of 10−4M nitroprusside in PBS followed by Microfil® (Flow Tech, Carver MA). 17 Brain tissue was counterstained with Evans blue dye or 2,3,5-triphenyltetrazolium chloride (TTC). 17 Vascular filling was confined to the precapillary vessels by adjusting the viscosity and perfusion pressure of the Microfil. 17 Optical tissue clearing was as described. 32 All collaterals between the outer branches of the ACA, MCA and PCA trees of both hemispheres were counted (Figure 1), and lumen diameter of each collateral was obtained at midpoint and averaged for each animal. 17 Density of intra-tree anastomoses was determined in a defined area of the MCA tree (Figure 1). Permanent middle cerebral artery occlusion (pMCAO) was by occlusion just distal to the lenticulostriate branches, and infarct volume was measured 24 h later by TTC. 17 Ligation of the ∼midpoint of the left anterior descending coronary artery (LAD) was followed 24 h later by measurement of infarct volume by TTC. 55 Femoral artery ligation was by ligating immediately proximal and distal to the superior epigastric artery which was also ligated, followed by cross-cutting and separating the cut ends of the vessels by 2 mm (moderate ligation) or by ligating, cross-cutting and separating the ends of the proximal femoral artery just distal to the inguinal canal (severe ligation). 56 Hindlimb perfusion immediately after ligation was measured by laser Doppler perfusion imaging of the plantar surface of the foot. 56
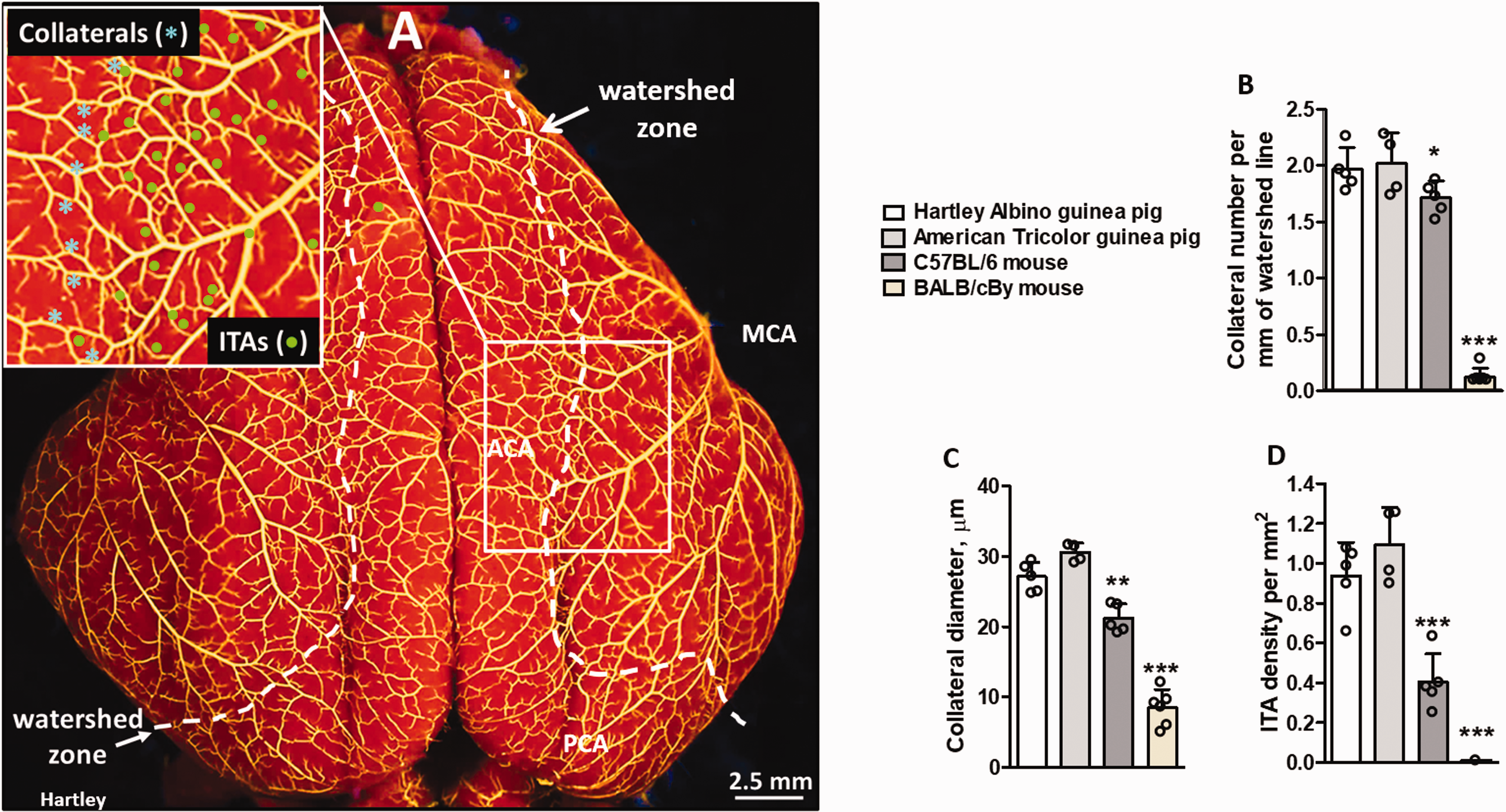
High altitude-evolved guinea pigs (Cavia porcellus) have a higher number/density of pial collaterals than the abundant-collateral C57BL/6 and sparse-collateral BALB/cBy mouse strains (Mus musculus domesticus, lowlander species). (a) The cerebral vasculature was maximally dilated and filled with Microfil whose viscosity and filling pressure were adjusted to limit filling to pre-capillary vessels, followed by tissue counterstaining. MCA, ACA, PCA, middle, anterior and posterior cerebral artery trees separated by a line drawn to delineate their watershed zones. For each animal, all collaterals between the trees were counted (b) and their lumen diameters were determined and averaged (c); number of intra-tree anastomoses (ITAs) in the indicated region was also determined (d, inset in a). Data in this and subsequent figures are mean ± SD for n number of animals per group (here 5,4,5,6 per left-to-right bar in each panel). *,**,***P < 0.05, 0.01, 0.001 by one-sided t-test vs. either guinea pig strain for collateral number and by two-sided t-tests for other parameters; p value symbols are identical for vs. Hartley and vs. Tricolor guinea pigs. Sidedness was pre-specified in this and subsequent figures. Values for the guinea pig breeds did not differ by two-sided t-tests.
Study design and statistical analysis were in accordance with the ARRIVE and STAIR guidelines 57 , 58 : Due to the nature of the experiments, investigators were not blinded to species or strains with the exception of deer mice strains; No data points were identified as outliers and excluded; The discussion and citation of the literature was unbiased; N-sizes (number of animals studied) were based on our previous studies which demonstrated sufficient power to test hypotheses using the variables measured herein. Values are means ± SD, with significance denoting p < 0.05. N-sizes and statistical tests are given in the figure legends and Supplemental Table I. Data from each of the bars presented in the figures and Supplemental Table II that had n > 7, which is required for normality testing (D’Agostino-Pearson test, Graphpad Prism), were evaluated: The data in Figure 2, bars 1,4–6 in each panel, Figure 4(b), bars 4,8, Figure 4(c), both bars, and Supplemental Table II, groups 5–7, were normally distributed (p > 0.05) except for Figure 2(a), bar 6 (p = 0.03). All procedures were approved by the University of North Carolina Institutional Animal Care and Use Committee (IACUC# 18-123.0-A, 04/2019) and were conducted in compliance with the NIH Guide for the Care and Use of Laboratory Animals.
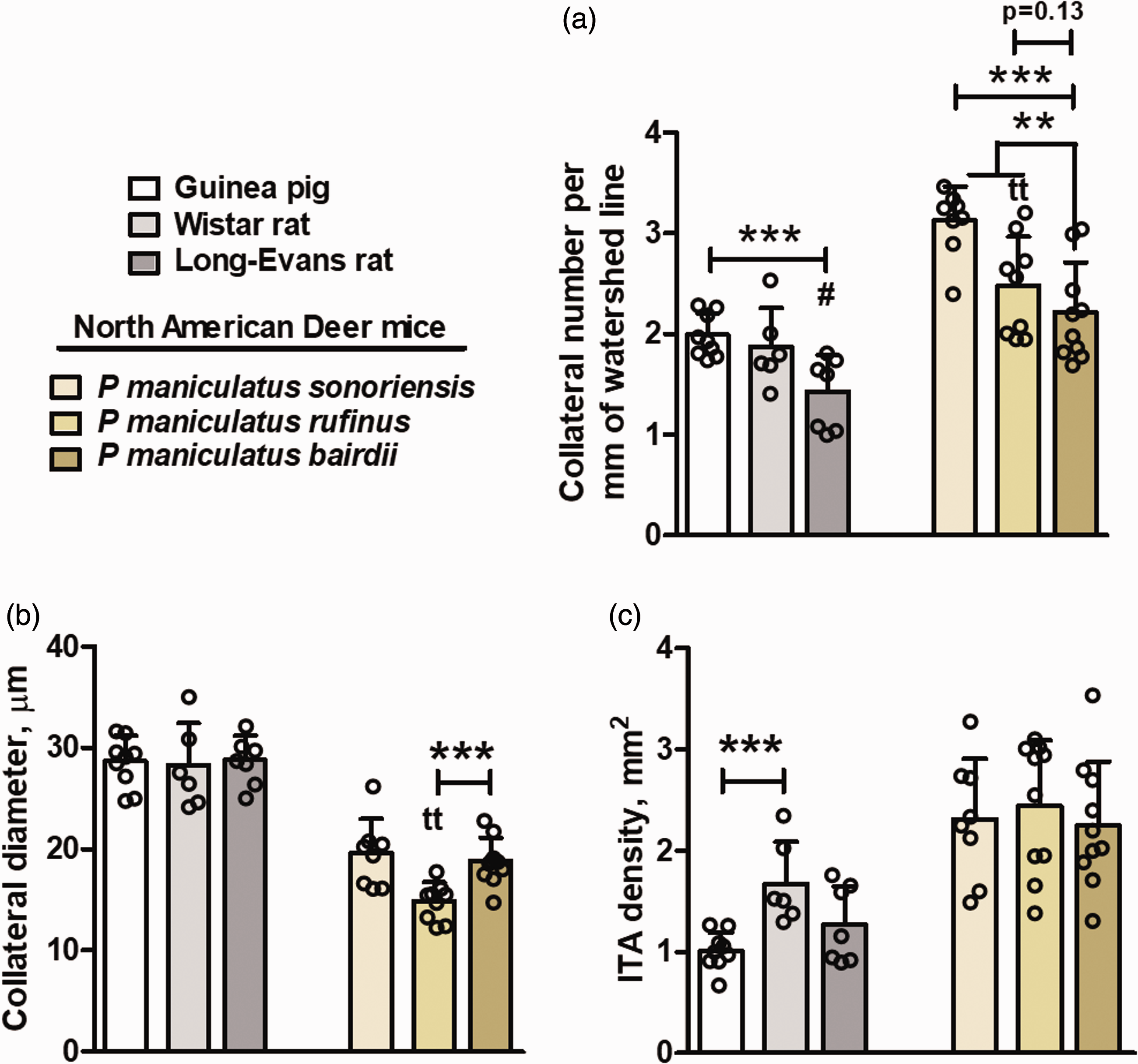
High-altitude evolved guinea pigs and deer mice, Peromyscus maniculatus sonoriensis and P.m.rufinus, have a greater number of pial collaterals than lowlander rats (Rattus norvegicus domesticus) and deer mouse P.m.bairdii, respectively. Data for both breeds of guinea pigs in Figure 1 were combined since they did not differ by two-sided t-tests and are shown for comparison with other species. (a,b) Collateral number and diameter. (c) Rats and deer mice have a greater density of ITAs than guinea pigs irrespective of highlander (guinea pig, P.m.sonoriensis, P.m.rufinus) versus lowlander status (other strains). Collateral diameter and ITA density do not associated with high- vs. lowlander status. N = 9,6,7,8,8,9 per left-to-right bar in each panel. Collateral number for highlanders vs. lowlanders by one-sided t-tests, other parameters by two-sided t-tests. **,***, P < 0.01,001; NS if bracket not shown; bracket end-tics identify the two bars tested. #,###, P < 0.05, 0.001, Long-Evans vs. Wistar. tt, P < 0.01, P.m.rufinus vs. P.m.sonoriensis. Sidedness of t-tests same as defined in legend for Figure 1.
Results
High-altitude guinea pigs and deer mice have greater density of pial collaterals than lowlander species
We first examined two breeds of guinea pigs. Both have a greater density of cerebral pial collaterals, i.e. number per mm length of the line demarcating the watersheds between the MCA, ACA and PCA trees, than the C57BL/6 and BALB/cBy mouse strains (Mus musculus domesticus) (Figure 1). We compared guinea pigs to these strains because: (1) No lowlander conspecific strain of Cavia exists, i.e. none that has resided at low-altitude for thousands of years, (2) Among 21 strains of M.m.domesticus, M.m.musculus. and M.m.castaneus, C57BL/6 have nearly the greatest and BALB/cBy the least number of pial collaterals (the same is true in their other tissues examined), 6 , 8 thus these strains bracket well the naturally occurring variation in collateral abundance in Mus musculus; (3) C57BL/6, BALB/cBy and other M.m. mice are lowlanders, with no populations residing at high-altitude for more than several hundred years.34–36 Guinea pigs also have a higher density of intra-tree anastomoses (ITAs, Figure 1), i.e. vessels that cross-connect adjacent arteriole branches within a given vascular tree, than C57BL/6 and BALB/cBy mice. However, this was not seen in other species regardless of altitude status (see below), which is consistent with the inability of ITAs to recruit blood flow from adjacent arterial trees.
Anatomic diameter of pial collaterals (i.e. when fully dilated) is also greater in guinea pigs than C57BL/6 and BALB/cBy mice (Figure 1). However, guinea pigs weigh ∼30-fold more than mice (Supplemental Table II), and brain weight closely follows body weight in rodents and other species. Therefore, comparing differences in collateral diameter among species is confounded by differences in body weight. However, normalizing to body or brain weight does not provide clarity because collaterals are anastomoses of distal arterioles, and both vessel types are similar in diameter among mammalian species irrespective of body weight (e.g. ∼10–20 µm in mice, ∼30 µm in guinea pigs and rats; see below). Consistent with this, genetic background-dependent differences in collateral diameter varied by 3-fold and collateral number by 30-fold among 21 M.m. strains, and neither parameter varied significantly with body or brain weight despite as much as 2-fold differences in body weight. 5 , 59
We next compared guinea pigs to Wistar and Long-Evans rats (Rattus norvegicus domesticus) based on the same rationale as above for C57BL/6 and BALB/cBy mice, i.e. both strains are lowlanders and no highlander conspecific exists. 60 Both are also known to have abundant collaterals, although to our knowledge, density and diameter have not been quantified. Collateral density is greater in guinea pigs than in Long-Evans rats; however, the difference was not statistically significant for Wistar rats (Figure 2). Density of ITAs is greater in Wistar but not Long-Evans rats. In agreement with diameter in guinea pigs versus mice, differences in collateral diameter between guinea pigs and rats do not follow altitude status regardless of differences in body weight (Supplemental Table II).
We also examined high-altitude deer mouse strains Peromyscus maniculatus sonoriensis and P.m.rufinus with their conspecific lowland strain, P.m.bairdii. Collateral density is greater in P.m.sonoriensis and trends the same for P.m.rufinus (Figure 2). All three species have a high density of ITAs irrespective of highlander versus lowlander. Like the above in guinea pigs versus Mus musculus mice and rats, collateral diameter does not follow altitude status, irrespective of body weight differences (Supplemental Table 1).
Guinea pigs and high-altitude deer mice are protected from cerebral infarction after arterial occlusion
Consistent with their high pial collateral density, guinea pigs, strikingly, had no cerebral infarction after acute ligation of the MCA (Figure 3(a)). In contrast, rats and M.m. sustained infarctions whose volumes, as expected, 5 varied inversely with their collateral densities given in Figures 1 and 2. Similarly, infarct volume in P.m.sonoriensis was significantly smaller than in lowlander P.m.bairdii (Figure 3(b) and (c)); P.m.rufinus was not tested due lack of availability of these wild-caught mice. Interestingly and for reasons that are unclear, unlike guinea pigs, rats, and Mus musculus mice (and humans) in which the MCA tree dominates perfusion of the neocortex (Figures 1 and 3(a)), the ACA tree is dominant in deer mice (Figure 3(b); Supplemental Figure I shows the same for P.m.sonoriensis and P.m.rufinus). Therefore, infarct volume was examined after ligation of the large central-most branch of the ACA just after it exits from under the mid-sagittal sinus. To our knowledge, such an exceptionally dominant ACA tree has not been reported for other mammalian species. Importantly, territory of the ACA was the same among the three strains: P.m.sonoriensis 77.3 ± 1.0 mm2, n = 8; P.m.rufinus 77.6 ± 2.2, n = 10; P.m.bairdii 77.3 ± 1.0, n = 9.
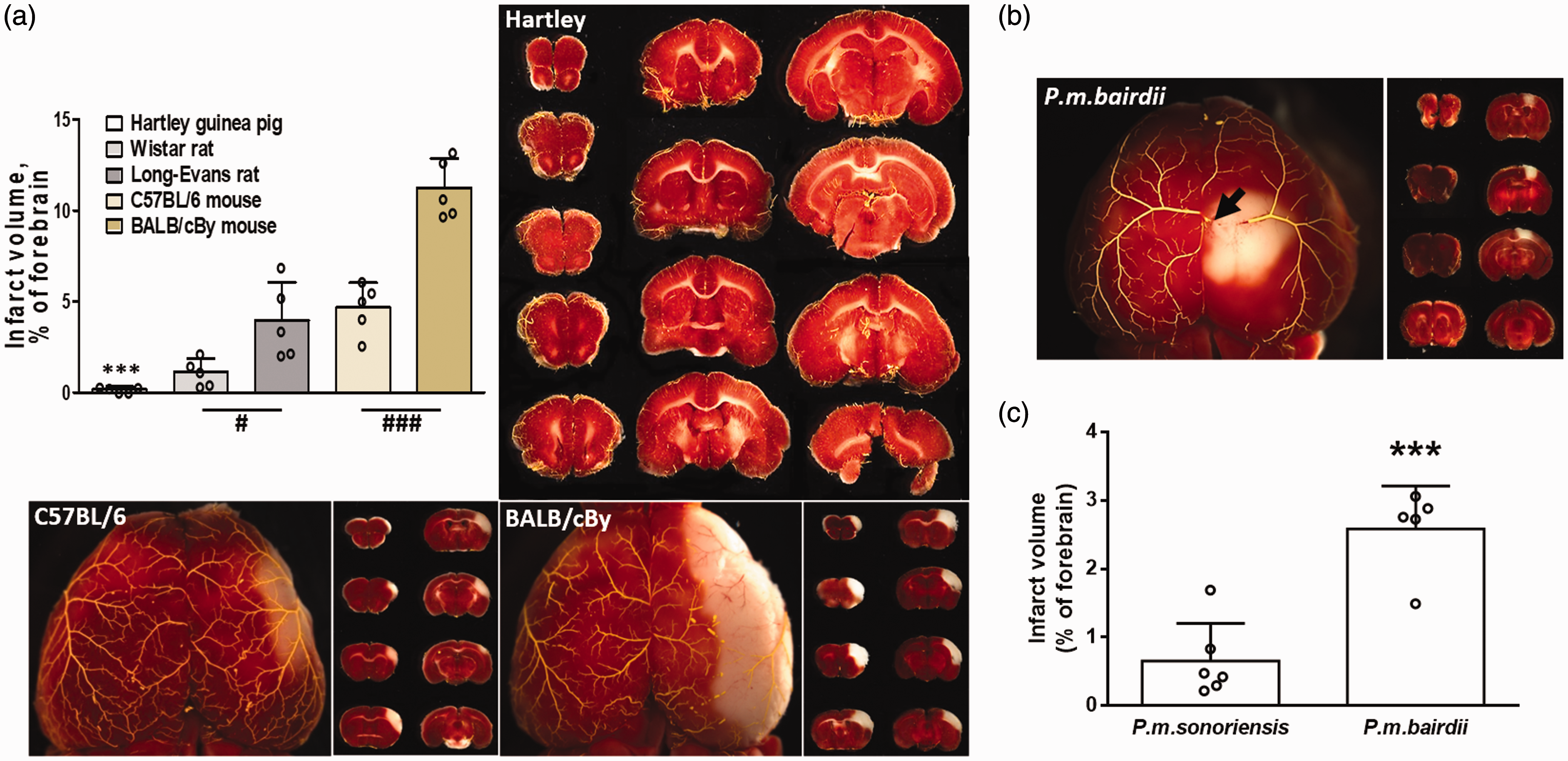
Guinea pigs sustain no cerebral infarction after ligation of the right MCA, and high-altitude deer mouse Peromyscus maniculatus sonoriensis has smaller infarctions after ligation of right ACA than its lowlander conspecifc, P.m.bardii. Differences in infarct volume in the seven rodent species are consistent with their differences in collateral number/density in Figures 1 and 2. Infarct volume by 2,3,5-triphenyltetrazolium chloride staining 24 h after MCA occlusion (representative images in a, b; arrow in b, ligation point). N = 5 per group in panel A, and 6, 5 in panel C. ***, p < 0.001 vs. each bar to the right (panel A) and vs. P.m.sonoriensis (panel C); #,### P < 0.05, 0.001; t-tests pre-specified as one-sided based on differences in collateral number identified in Figures 1 and 2. Unlike guinea pigs, rats, and Mus musculus in which the MCA tree dominates cortical perfusion (a, see also Figure 1), the ACA tree is dominant in deer mice (b, see also Supplemental Figure I).
Guinea pigs and high-altitude deer mice have more extensive collaterals in skeletal muscle than lowlander species
Previous studies of M.m. strains found that similar genetic-dependent and gene-targeted-induced differences in collateral abundance in brain are also seen in skeletal muscle and other tissues of the same individual.7–9, 56 These studies also found that collateral-dependent blood flow in brain and hindlimb measured immediately after arterial ligation and the severity of subsequent tissue injury mirrored the differences in collateral extent in these tissues. Unlike in brain where pial collaterals reside on the surface and are thus readily visualized, collaterals in hindlimb and most other tissues are located deeply and are thus hard to differentiate from similarly sized end-arterioles, making quantification difficult. We therefore examined blood flow in the planter region of the foot immediately after acute femoral artery ligation. Plantar perfusion is a widely used index of collateral-dependent blood flow in the hindlimb. Its value reflects the number of collaterals, primarily in skeletal muscle, that interconnect branches of the iliac and femoral arteries with the popliteal and saphenous arteries below the points of ligation.7–9, 56 In agreement with the above findings for pial collaterals, guinea pigs have more extensive hindlimb collaterals than Wistar and Long-Evans rats and C57BL/6 mice, as evidenced by 3- to 4-fold smaller declines in blood flow after moderate or severe femoral ligation (Figure 4(a) and (b)). Likewise, perfusion fell 2.3-fold less after FAL in the high-altitude deer mouse P.m.sonoriensis compared to the lowlander P.m.bairdii strain (Figure 4(c)).
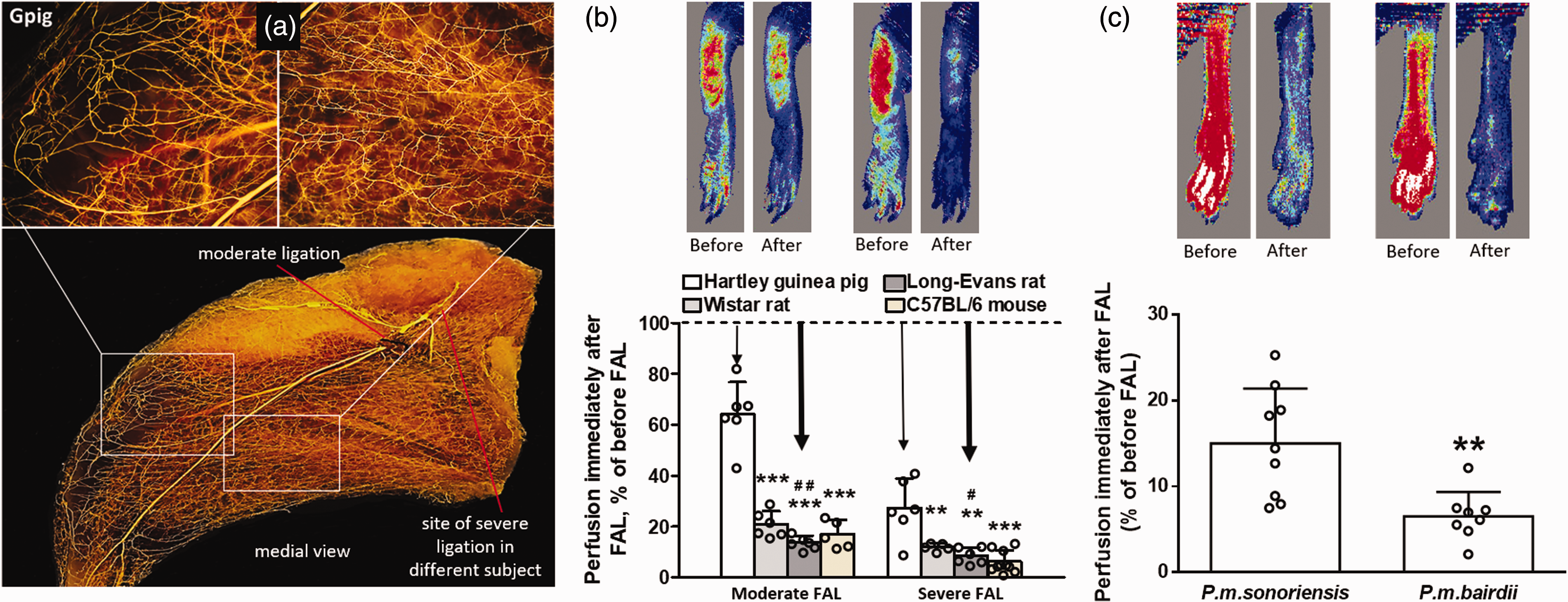
Guinea pigs and high-altitude deer mouse Peromyscus maniculatus sonoriensis have greater collateral-dependent blood flow after femoral artery ligation (FAL) than lowlander rat, C57BL/6 mouse, and conspecific deer mouse, P.m.bardii. (a) Hartley guinea pig (Gpig) arterial vasculature immediately after moderate FAL (black suture; line shows site of severe ligation when performed), maximal dilation, filling with Microfil and optical clearing, showing extensive collaterals resulting in complete retrograde filling of arterial trees distal to the ligation. (b,c) Laser Doppler perfusion imaging of the plantar side of the guinea pig (b) and P.m.sonoriensis hindpaw (c) immediately after FAL (index of collateral-dependent blood flow). (b) Perfusion falls less after distal (moderate) and proximal (severe) FAL in guinea pigs than in rats and C57BL/6 mice. Severe ligation not tested in deer mice. N = 6,6,6,5,6,5,6,10 per left-to-right bars in panel B and 9,8 in panel C. **,***, P < 0.01, 0.001 vs. guinea pig (b) and vs. P.m.sonoriensis (c), one-sided t-test. #,##, P < 0.05, 0.01 vs. Wistar rat, one-sided t-test.
Guinea pigs have extensive collaterals in heart and are protected from myocardial infarction after coronary artery ligation
Consistent with the above in brain and hindlimb, guinea pigs also have extensive coronary collaterals and, remarkably, sustained no myocardial infarction after ligation of the proximal left anterior descending (LAD) artery (Figure 5(a) and (c)). In contrast, M.m. mice and rats, which have no or sparse coronary collaterals, respectively, 32 , 33 , 55 , 61 evidenced large infarctions after ligation of the midpoint of the LAD (Figure 5(b) and (c)). It is not known why coronary collaterals are absent in mice and very sparse in rats, yet are present in substantial number in brain and skeletal muscle of C57BL/6 and other high-collateral strains of M.m.5–8, 32 , 33 , 55 , 56 and rats 61 , 62 (Figures 1 to 4). Unlike the above absence of infarctions that we observed in male and female six to seven-months-old Hartley guinea pigs after proximal LAD ligation, Hardwick et al. 63 reported that young nine weeks-old male Hartley guinea pigs sustain a small 5.6% infarction measured as scar tissue six weeks after ligation just distal to the first diagonal branch; it is unclear whether the infarcts were produced by occlusion of flow to the surrounding tissue caused by the ligature itself. We did not evaluate myocardial infarction in deer mice because preliminary studies showed that, like M.m. 32 , 33 , 55 (Figure 5), deer mice lack coronary collaterals irrespective of highlander or lowlander status (i.e. no retrograde filling of the LAD tree occurred downstream of acute ligation, data not shown).
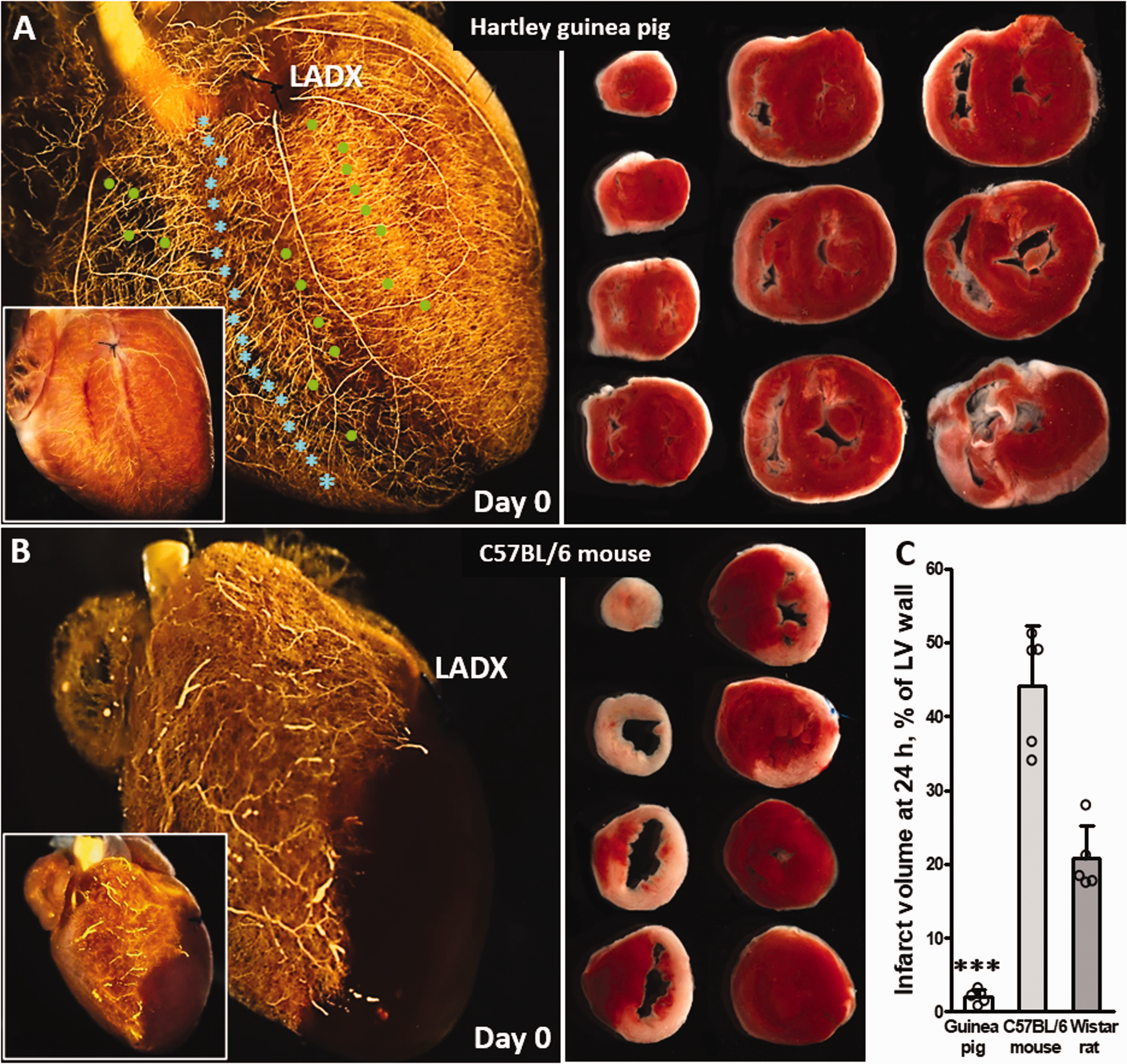
Guinea pigs have extensive coronary collaterals and are protected from myocardial infarction after ligation of the left anterior descending coronary artery (LADX). (a–c) left ventricular (LV) infarct volumes for Hartley guinea pig and other species by 2,3,5-triphenyltetrazolium chloride staining 24 h after LADX. Immediately after LADX (black suture in a, b) the arterial vasculature was maximally dilated and filled with Microfil as described in Figure 1, followed by optical clearing 55 (insets are before optical clearing). (a) Guinea pigs have abundant coronary collaterals and intra-tree anastomoses (green stars and blue circles identify several of each vessel type in the sub-epicardium), thus complete back-filling of the LAD tree occurs distal to the point of ligation. (b) 2% infarct volume in guinea pigs reflects ligature-induced compression of the surrounding tissue. C57BL/6 mice (and other M. musculus strains 32 , 33 , 55 ) lack coronary collaterals, thus no back-filling occurs. Evidence suggests rats have sparse native coronary collaterals. 37 , 64 (c) N = 5 per group; *** P < 0.001 vs. mouse and also versus rat, one-sided t-test. Infarct volume was not determined in deer mice because, like M. musculus, they lack native coronary collaterals (i.e. no retrograde filling of the LAD tree occurred downstream of the acute ligation; data not shown).
Guinea pigs and deer mice have a robust configuration of the circle of Willis
We also examined the architecture of the ventral cerebral arterial circulation, including the collateral arteries of the circle of Willis (CoW, Figure 6). Both breeds of guinea pigs have a complete, symmetric (i.e. diameters are similar on both sides) CoW primarily supplied by the posterior circulation, i.e. consistent presence in all subjects of: an exceptionally large-diameter basilar artery (BA) relative to the internal carotid arteries (ICAs) and bilateral paired posterior cerebral arteries (P1-PCAs)—features that to our knowledge have not been reported to be simultaneously present together in another mammalian species—and large bilateral posterior communicating collateral arteries (PComs) and anterior communicating collateral artery (ACom). Some of these features in the guinea pig have been described or are evident in images from previous studies.64–68 Deer mice also have a complete, symmetric CoW that is primarily supplied by the ICAs, plus consistent presence of bilateral PComs and an azigos artery (functional equivalent to the ACom), irrespective of altitude status (Figure 6). We did not examine M.m. mice and rats because the configuration of their CoWs is known to have large ICAs and, depending on strain, frequently asymmetric, incomplete CoWs (i.e. often lacking bilateral PComs, with PComs and the azigos/ACom having small and varying diameters); other mammals including humans also frequently have variations similar to those described above for M.m. 59 and references therein.64–67
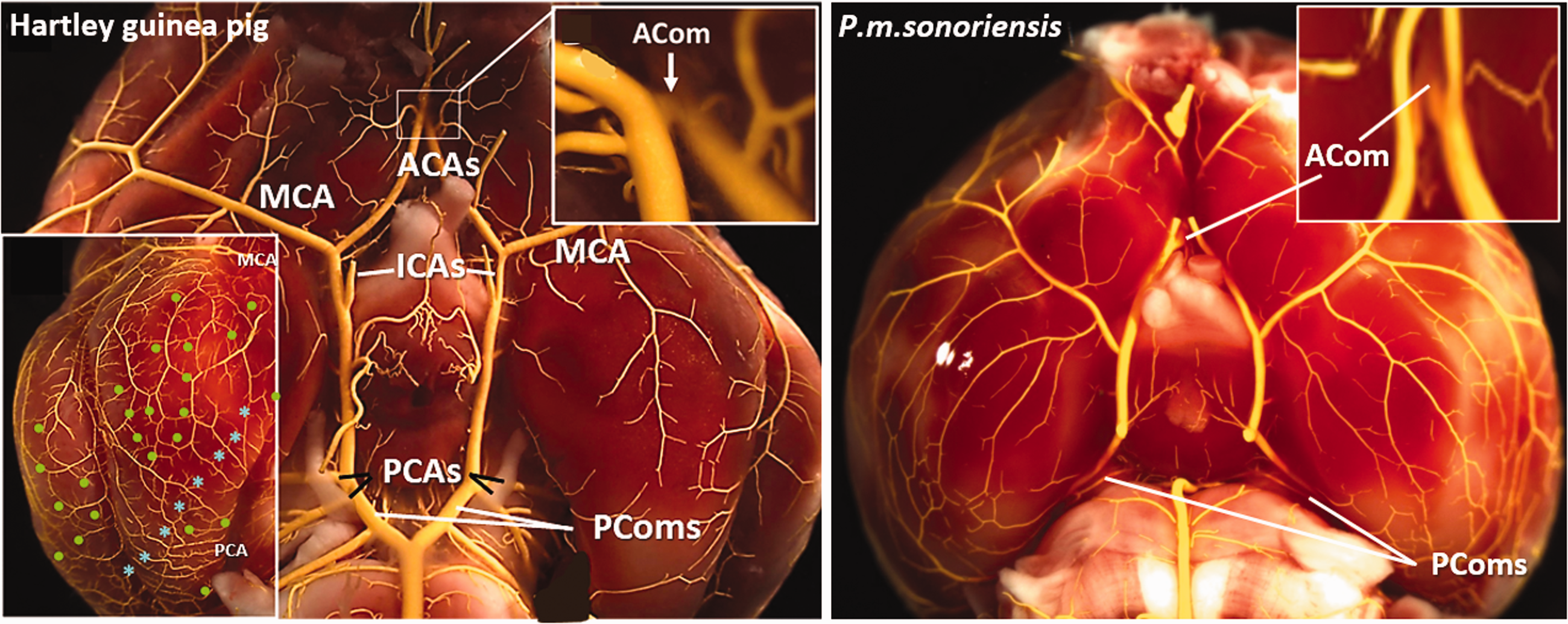
Ventral cerebral arterial vasculature of guinea pigs and deer mice. Images are representative of the two guinea pig breeds and three deer mouse strains examined in previous figures. Left panel, guinea pigs have a complete circle of Willis primarily supplied by the posterior circulation, i.e. consistent presence in all individuals examined of straight-coursing large-diameter: basilar artery (BA), bilateral paired P1-PCAs, anterior communicating artery (ACom), and bilaterally present posterior communicating arteries (PComs) with similar diameters. Internal carotids (ICAs) are small-diameter and located anteriorly (both features are unlike deer mice, Mus musculus, and rats). Right panel, P.m.sonoriensis (and P.m.rufinus and P.m.bairdii, see Supplemental Figure I) deer mice have complete circles and, unlike M.m., rats and humans, do not evidence variation for unilateral or bilateral absence or atresia of the PComs or ACom. See Faber et al. 59 and references therein. Diameter of the ACom is similar among the three deer mouse strains, whereas order of PCom diameter is P.m.bairdii > P.m.rufinus > P.m.sonoriensis. Inset in left panel, like Figure 1, shows abundant MCA-PCA pial collaterals (blue stars) and intra-tree anastomoses (green dots) from a different subject with filling pressure optimized to fill distal precapillary pial vessels.
Discussion
Findings in this study support the novel hypothesis that that—in addition to providing protection in obstructive disease—collaterals may help match oxygen delivery to oxygen demand under conditions of environmental hypoxia, thereby contributing to genetic adaptation in high-altitude species. By increasing the hemodynamic efficiency of regional oxygen delivery, an increase in collateral abundance could help sustain effective cognitive, sensory-motor, skeletal muscle and cardiovascular function in species living at high altitude.
Unlike collaterals, the density of intra-tree anastomoses (ITAs) that interconnect a fraction of the branches within a given arterial tree did not associate with altitude. The density of ITAs in the cerebral circulation of guinea pigs was substantially greater than in C57BL/6 mice, a strain of lowlander Mus musculus that, unlike the BALB/cByJ strain and other low-collateral strains, has a relatively high density of ITAs and collaterals. 5 , 6 , 10 However, ITA density in two strains of lowlander Rattus norvegicus was greater than in guinea pigs and even greater yet in deer mice irrespective of their high- or lowlander ancestry. In M.m. mice ITAs form late during gestation at the same time that pial collaterals form and display the same genetic background-dependent variation as collaterals in the number that form, 10 indicating that formation of both vessel types, and variations in it, are driven by the same or a similar signaling pathway and its polymorphic genetic elements . However, the majority of ITAs that form are pruned away by three weeks after birth, leaving the density present in the adult. 10 Our finding that ITA density does not associate with altitude status suggests that, unlike collaterals, ITA abundance is not an adaptive trait shared by high-altitude natives. Given the inability of ITAs to recruit blood flow from adjacent arterial trees, this contrasting finding vis-à-vis collaterals supports the concept that the particularly abundant collaterals in highlanders are a genetic adaption.
The circle of Willis (CoW) which by way of its PCom and ACom collateral arteries provides alternate routes of perfusion—like pial collaterals, also displayed several unique features in the high-altitude species examined. Guinea pigs had a robust, i.e. complete and symmetric CoW with large, similarly sized bilateral PComs and consistent presence of an ACom. A previous study of juvenile Hartley guinea pigs schematically described features similar to the above. 65 However, it was unclear if the findings were based on replicate analysis, and another study concluded that AComs were absent in guinea pigs. 68 Deer mice also had a robust CoW; however, this was true in both low- and highland strains. We did not examine M.m. mice and rats because the configurations of their CoWs are known, depending on strain, to frequently lack bilateral PComs and when present to be small and vary in diameter. See Faber et al. 59 and references therein. Moreover, these features vary widely among replicates of the same M.m. strain, indicating a high level of stochastic variability. Other low-altitude-evolved mammals including humans also frequently have variations similar to those described above for M.m. and rats, including absence of AComs. See literature 59 ,64–67 and references therein. Interestingly, 91% of Nepalese have a complete CoW compared to 5–45% in lowlander Indian and other ethnicities.69–73 Paired P1-PCAs was either not observed or not commented on in previous studies in humans69–71 or was reportedly present in 2.4%. 72 Considerations that aid interpretation of the presence of wide variation in the CoW in lowlanders versus robust circles in guinea pigs and high-altitude deer mice—but presence too in the latter’s low-altitude conspecific—are discussed below (“Limitations”). However, besides its collateral function, the CoW also dampens pulse pressure and variation in blood flow in the cerebral arteries that arise from variations in blood flow in the ICAs and BA during normal awake behaviors. 69 It is possible that a robust CoW is an adaptive trait selected for not only in high-altitude species like guinea pigs, but also in deer mice irrespective of altitude status to assure optimal cerebral blood flow during cognitive and sensory-motor states that favor survival in certain low-altitude wild non-domesticated environments.
Although measurement of capillary density was not part of our aims, increased capillary density might be an adaptive trait in highland species. However, published data are inconsistent: Capillary density was comparable in intestine, 74 soleus 75 and stria vascularis 76 in guinea pig versus rat. Similarly, density and capillary-to-fiber ratio were comparable in left ventricle of guinea pigs either collected in the Andean mountains, born at sea level and subjected to high altitude postnatally, or born and kept at sea level. 77 In highland P.m.rufinus. and lowland P.m.nebracensis maintained at low altitude, capillary density in left ventricle was comparable 78 ; however, density and capillary-to-fiber ratio were increased in gastrocnemius of P.m.rufinus. 79 , 80 The absence, in general, of increased capillary density in highlanders in the above studies may reflect that (1) capillary blood flow and the density of perfused capillaries in a tissue at a given metabolic rate depend primarily on mechanisms that control smooth muscle tone, and (2) increased capillary density is a well-known rapid and reversible acclimatization response to high-altitude exposure. 81 , 82 Myocardial capillary density in the lizard, Phrynocephalus vlangalii, of the Qinghai-Tibet Plateau, was greater in cohorts collected at high (4564 m) versus low altitude (2750 m). 83 Likewise, in the sternothyroid muscle of Andean dogs native to 4350 m, density was three times greater than in dogs native to low altitude. 84 However, capillary density in heart and several skeletal muscle of the high-altitude Andean coot, Fulica americana peruviana, was comparable to its sea level conspecific. 85 Moreover, residents of mixed ethnicities in La Paz (3800 m) had lower, rather than higher, capillary density and capillary-to-fiber ratios in vastus lateralis muscle than in a comparable lowland population. 86 In contrast, sublingual microvessel density in Sherpas was 30% greater than in Kathmandu lowlanders, 87 with a similar 40% greater difference in capillary density in vastus lateralis muscle. 88 In addition to the caveat stated at the end of the preceding paragraph, the variability of the findings in the above studies may also reflect differences in muscle fiber number, volume and type, which could differ in highland and lowland populations and were not measured concomitantly in most of the above studies.
No previous studies have examined the collateral circulation in species native to high-altitude. However, there is circumstantial evidence consistent with the hypothesis that limits on tissue oxygenation could favor an increase in collaterals, in addition to that cited in the Introduction. Evidence suggests that humans have inhabited the Tibetan Plateau, and the Ethiopian highlands for millenia. 89 High-altitude species have evolved numerous adaptations, and in humans,89–91 a number of HIF pathway genes have been identified as candidates for hypoxia adaptation in Tibetan, Andean and Ethiopian highlanders (e.g., EPAS1, EGLN1/2/3, VEGFA, NOTCH1, CXCR4, NOS1/2/3, FLT1, PIK3CA).89–91 Several have been shown to be involved in collaterogenesis (Egln2, Vegfa, Notch1, Cxcr4). 1 , 11 , 12 , 17 , 32 , 55 An Epas1 polymorphism in deer mice exhibits a striking altitudinal pattern of allele frequency variation, and is associated with differences in cardiovascular function as well as genome-wide transcriptional responses to hypoxia in heart and adrenal gland, including HIF-regulated genes. 92 Future studies could investigate guinea pigs, deer mice and other high-altitude rodents to determine whether genetic variation in the collaterogenesis pathway is associated with differences in collateral abundance. It would also be interesting to investigate directly whether Tibetan, Andean or Ethiopian highlanders have a lower incidence of stroke or better outcomes, as suggested by several previous studies examining Tibetan cohorts.43–48
Limitations
This study has several limitations. First, since no guinea pig population has resided at low altitude for the thousands of years required for natural selection, we compared two breeds of guinea pigs to two strains each of the lowlander species, M.musculus and R.norvegicus—a comparison that is not optimal. Second, only two highlander strains of P.maniculatus that have been studied widely were available for analysis. Our previous studies of collateral extent in M.m. make clear the limitations created by the above constraints: collateral number in 21 strains of M.m. varied by 30-fold, with two-thirds having abundant and one-third having sparse collaterals, like the C57BL/6 and BALB/cBy strains studied herein. 5 , 6 We hypothesize that this reflects a relaxation of stabilizing selection in low-altitude populations, since variation in collateral abundance may have little or no relevance to fitness under normoxic conditions. Accordingly, the conclusions of the present study could be strengthened by studying a larger number of high-altitude strains of P.maniculatus and their lowlander conspecifics, if such were described and available. Our conclusions could also be strengthened by examining additional species, for example, Andean mice, Phyllotis andium and P.xanthopygus, and their lowlander conspecifics, P.amicus and P.limatus. 93 However, our above M.musculus findings indicate once again that more than just these two conspecific pair would have to be studied to reach a firm conclusion. Examining additional high-altitude evolved species such as pikas 94 , 95 would also strengthen our conclusions, although these and many other high-altitude evolved species, like guinea pigs, lack a lowland comparator strain. As well, the above studies would be faced with the logistics of wild capture in remote regions. The observation that collateral abundance varies widely in low-altitude strains of M.musculus, which is also the cases for variation in the CoW59 and may also be true for capillary density (see above) in this and other species including humans, means that significant numbers of high- and low-altitude populations will need to be studied. It also calls into question the conclusion that large differences in collateral blood flow exist among species of mammals, when the conclusion is based on comparing only a single strain or cohort of each species made up of closely related/reared individuals. 37 An additional limitation of this study is that the extent of tissue injury in response to artery ligation is affected by several factors besides collateral blood flow that could vary among the species examined, independent of altitude status. This includes ischemic threshold for cell death, factors that affect resistance in the vascular bed downstream of the collaterals, and differences in arterial oxygen content, hemoglobin affinity and blood pressure.
Whether selection has shaped patterns of collateral abundance in high-altitude humans will require studies of natural variation in collateral extent, for example in cadavers, or collateral score in patients with acute ischemic stroke. These studies will be logistically difficult and will require adjustment for vascular risk factors that can affect collateral abundance. 1 , 54 , 56 It will also be important in these studies to examine SNPs in genes that have been shown to be important for robust collaterogenesis during development and in the adult. 1 , 11 , 12 , 17 , 32 , 55 ,96 Such studies could add collateral extent to the list of physiological mechanisms that contribute to hypoxia adaptation by improving oxygen transport and distribution of blood flow. 52 , 53 ,93–95 Investigation could also be extended to populations affected by sickle cell disease and thalassemia. Identifying genetic factors that determine collateral extent in high-altitude animal and human populations may help clarify differences in susceptibility to ischemic diseases and lead to new therapeutic approaches.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X20942609 - Supplemental material for High-altitude rodents have abundant collaterals that protect against tissue injury after cerebral, coronary and peripheral artery occlusion
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X20942609 for High-altitude rodents have abundant collaterals that protect against tissue injury after cerebral, coronary and peripheral artery occlusion by James E Faber, Jay F Storz, Zachary A Cheviron and Hua Zhang in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institutes of Health’s National Institute of Neurological Diseases and Stroke NS083633 to JEF. National Institutes of Health, National Institute of Heart, Lung, and Blood Diseases HL087216 and National Science Foundation OIA-1736249 to JFS and OIA-1736249 and IOS-1354934 to ZAC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
HZ performed the experiments, morphometry, statistical analysis and constructed the figures; JF conceived and designed the experiments, analysis and figures and wrote the manuscript; JS and ZC provided P.m.rufinus mice, advice and discussion. All authors provided edits to the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
