Abstract
We sought to investigate whether carotid endarterectomy (CEA) can achieve long-term cerebral hemodynamic improvement and reduce recurrence of cerebral ischemic events in symptomatic and asymptomatic patients with severe (> 70%) carotid artery stenosis contralateral to carotid occlusion (CO). Thirty-nine patients with severe carotid lesion contralateral to CO were studied before (1 day) and after CEA (at 7 days, 1, 3 and 6 months, and then yearly thereafter). Collateral flow and cerebral vasomotor reactivity (VMR) were assessed by transcranial Doppler sonography (TCD). A total of 32 unoperated patients with severe carotid lesion contralateral to CO, who were comparable with respect to age and sex, served as a control group. The average period of TCD follow-up was 10 years and was obtained in all patients; during this period, major clinical events (stroke, acute myocardial infarction and death) were also recorded. The proportion of patients with collateral flow via the anterior communicating artery increased significantly from 61.5% before to 89.7% after CEA (P = 0.01). Cerebral VMR ipsilateral to CO improved in 85.7% of patients (30 of 35) within 30 days of CEA, and in all patients within 90 days. No significant spontaneous VMR recovery was recorded in the control group. After the initial recovery, no significant change in VMR was observed in the surgical group or the control group during the follow-up. In conclusion, in patients with severe carotid stenosis, CEA contralateral to symptomatic and asymptomatic CO determines a durable cerebral hemodynamic improvement not only on the side of the CEA but also on the contralateral side, with no difference between symptomatic and asymptomatic patients.
Introduction
Cerebral hemodynamics might be compromised in patients with an occluded carotid artery (Powers, 1991). A concurrent contralateral severe (> 70%) carotid stenosis (CS) may further alter blood flow (BF) to the brain and increase the risk of low-flow infarcts (Klijn et al, 1997; Weiller et al, 1991) and embolic ischemic events (Georgiadis et al, 1993).
Carotid endarterectomy (CEA) of the contralateral severe CS is performed to remove atherosclerotic plaque, which is a possible source of emboli, and to improve perfusion of the hemisphere ipsilateral to carotid occlusion (CO) (AbuRahma et al, 2000; Markus et al, 1993; Mattos et al, 1992).
Though many authors have investigated the cerebral hemodynamic effect of CEA (Cikrit et al, 1992; Kluytmans et al, 1998; Markus et al, 1993; Rutgers et al, 2001; Soinne et al, 2003; Vernieri et al, 2001; Visser et al, 1997; Widder et al, 1994), only a few have reported a benefit. Some showed an improvement only in the hemisphere ipsilateral to the side of the CEA, while others also on the side of the CO (Rutgers et al, 2001; Visser et al, 1997). Some detected a cerebral vasomotor reactivity (VMR) recovery in all patients, regardless of clinical presentation, others only in subjects with symptomatic CO (Rutgers et al, 2001; Soinne et al, 2003), or, they found differences in symptomatic and asymptomatic patients (Soinne et al, 2003).
The aim of this prospective research study was to analyze the natural course of cerebral hemodynamics in patients with severe CS contralateral to symptomatic and asymptomatic CO, and to investigate whether CEA of the severe CS could determine a long-term hemodynamic improvement in the two hemispheres. Baseline cerebral hemodynamic parameters and VMR in patients with severe CS contralateral to CO who did and did not undergo CEA were evaluated by transcranial Doppler ultrasonography (TCD) over an average period of 10 years.
Materials and methods
Between January 1992 and December 1994, the participants in this study were prospectively enrolled from among consecutive patients referred to the Neurosonology Laboratory of our Department of Neurology. All subjects had to be independent in daily life (modified Rankin Scale score < 2) and had a symptomatic or asymptomatic severe CS contralateral to a CO on duplex ultrasound scanning (DUS). Duplex ultrasound scanning was performed on an ATL 3000 HDI System (Advanced Technology Laboratory, Inc., Bothell, Washington, DC, USA) according to the criteria established by our vascular laboratory and validated yearly (Ballotta et al, 1999a). Findings were confirmed by angiography of the supraaortic trunks with biplanar extracranial and intracranial views. The degree of CS was calculated as the percentage of diameter reduction on the preoperative arteriograms, in compliance with the North American Symptomatic Carotid Endarterectomy Trial (NASCET) method (NASCET collaborators, 1991). Exclusion criteria were: a history of previous CEA or radiotherapy, a very recent (< 3 months) cerebral or retinal ischemic event, a potential cardiogenic origin of emboli, a poor insonation of the transtemporal window, and occlusion or severe stenosis of intracranial vessels. Of the 76 subjects enrolled, five were excluded from the study (three for poor insonation of the transtemporal window, one for atrial fibrillation, and one for a severe intracranial stenosis); they underwent uneventful CEA, however. Of the 71 patients included, 39 underwent CEA and constituted the surgical group, while the other 32 considered the risk of the surgical procedure unacceptable and refused CEA (all patients were informed about the pros and cons of surgery by the same physician): they were comparable in terms of age and sex, and served as a control group. Of the 39 patients in the surgical group, their CO was the sole cause of symptoms in 10 cases, symptomatic associated with contralateral CS symptoms in 12, and asymptomatic in 17; their CS was the sole cause of symptoms in 20 cases, symptomatic associated with contralateral CO symptoms in 12, and asymptomatic in 7. As for the 32 patients in the control group, their CO was the sole cause of symptoms in 10 cases, symptomatic associated with contralateral CS symptoms in 9, and asymptomatic in 13; their CS was the sole cause of symptoms in 13 cases, symptomatic associated with contralateral CO symptoms in 9, and asymptomatic in 10. Patients in the surgical group provided a thorough clinical history and underwent careful neurological and cardiological assessments, complete blood chemistry tests, electrocardiogram (ECG), echocardiography, and cerebral computed tomography (CT) scan. They were studied by TCD 1 day before CEA, while control patients were evaluated at referral. All patients and control subjects gave their written informed consent to take part in the study, which was approved by the local ethics committee.
Transcranial Doppler Ultrasonography
Transcranial Doppler sonography was performed by three experienced neuro-sonographers (C Baracchini, G Meneghetti, and R Manara), with the patient lying in a comfortable supine position, with no visual or acoustic stimulation, in a quiet room. Recordings were made using commercially available equipment (EME TC 2000/S) with the 2-MHz pulsed-wave transducer, as described by Aaslid et al (1982). The baseline assessment comprised a bilateral transtemporal insonation of the middle cerebral arteries (MCAs), anterior cerebral arteries (ACAs), and posterior cerebral arteries (PCAs), together with a suboccipital insonation of the vertebral arteries (VAs) and the basilar artery (BA). Gosling's pulsatility index (PI) was determined as the difference between the peak systolic and end-diastolic velocities divided by the mean BF velocity (mBFV) in the MCA (Michel and Zernikow, 1998).
Intracranial collateral vessel evaluation was performed without compression manoeuvres to avoid mobilizing any carotid plaque debris. Collateralization was assumed to be: (1) through the anterior communicating artery (AcoA) when the ACA flow ipsilateral to CO was reversed; (2) through the external carotid artery when the ophthalmic artery (OphA) flow ipsilateral to CO was reversed; (3) through the posterior communicating artery (PcoA) when a 100% increase in mBFV was detected in the PCA ipsilateral to CO; lastly, vertebro-basilar (V-B) activation was identified by a 100% increase in mBFV in the BA.
Cerebral VMR was determined by acetazolamide challenge (Ringelstein et al, 1992): 1 g of acetazolamide was injected intravenously and a simultaneous bilateral recording of mBFV of the MCA was conducted for 30 mins. The upper limits of the confidence intervals (5% of the distribution in the right tail) for the reference sample of normal subjects were used as threshold values to establish a normal response to acetazolamide. Vasomotor reactivity was calculated as the maximal percentage increase in mBFV after pharmacological stimulation compared with mBFV at rest. Vasomotor reactivity was considered ‘normal’ whenever an increase in mBFV of at least 30% and a contralateral asymmetry < 20% were recorded. When the values of these two variables were not normal, VMR was said to be 'compromised'. In the follow-up, we arbitrarily adopted descriptors to define different degrees of VMR recovery as follows: 'full', if there was an increase in mBFV of at least 30% after pharmacological stimulation with respect to the previous examination; ‘marked', if the increase was between 20% and 30%; ‘slight', if it was between 10% and 20%; ‘absent', if it was < 10%.
Surgical Procedure
All CEAs were performed by the same surgeon (E Ballotta) with the patient under deep general anesthesia and cerebral protection involving continuous perioperative electroencephalographic (EEG) monitoring for selective shunting. All perioperative EEGs were visually analysed by a neurologist with extensive experience in the interpretation of studies during sleep. Shunting criteria were based exclusively on EEG changes suggestive of cerebral ischemia. The technical details of the CEA procedure have been described elsewhere (Ballotta et al, 1999b).
Surveillance Protocol
Long-term follow-up ranged between 6 and 150 months, with a mean of 10 years and a median 130 months, and was obtained in all patients. Patients were followed up clinically by a neurologist. In the surgical group, DUS and TCD studies were performed before discharge and repeated regularly at 1, 3, and 6 months, and every year thereafter, while in the control group they were performed yearly. Study end points were as follows: (1) perioperative (30-day) complications, and late neurological events and death in operated and unoperated patients; and (2) immediate and long-term cerebral hemodynamic changes in the two groups.
Statistical Analysis
To compare baseline characteristics between the two groups, Student's t-test (two-tailed) for continuous data and χ2 or Fisher exact test (two-tailed), as appropriate, for frequencies and categorical data were used. Differences in prevalence of collateral flow were analyzed with the McNemar test for paired proportions. Cumulative life-table analyses (Kaplan-Meier) were performed to assess stroke-free survival rates. A P-value < 0.05 was considered statistically significant.
Results
The baseline characteristics of both groups are shown in Table 1. There were no statistical differences between the two groups in terms of risk factors, side and clinical presentation of CO, or degree of contralateral CS. The intercurrent medication—namely antiplatelet/anticoagulant agents, ACE inhibitors, and statins—was also comparable between the two groups. Although the incidence of retinal symptoms related to CS was higher in the surgical group (5 of 39 versus 0, with a trend towards statistical significance, P = 0.06), the patients with the said symptoms were comparable between the two groups.
Demographic and clinical data
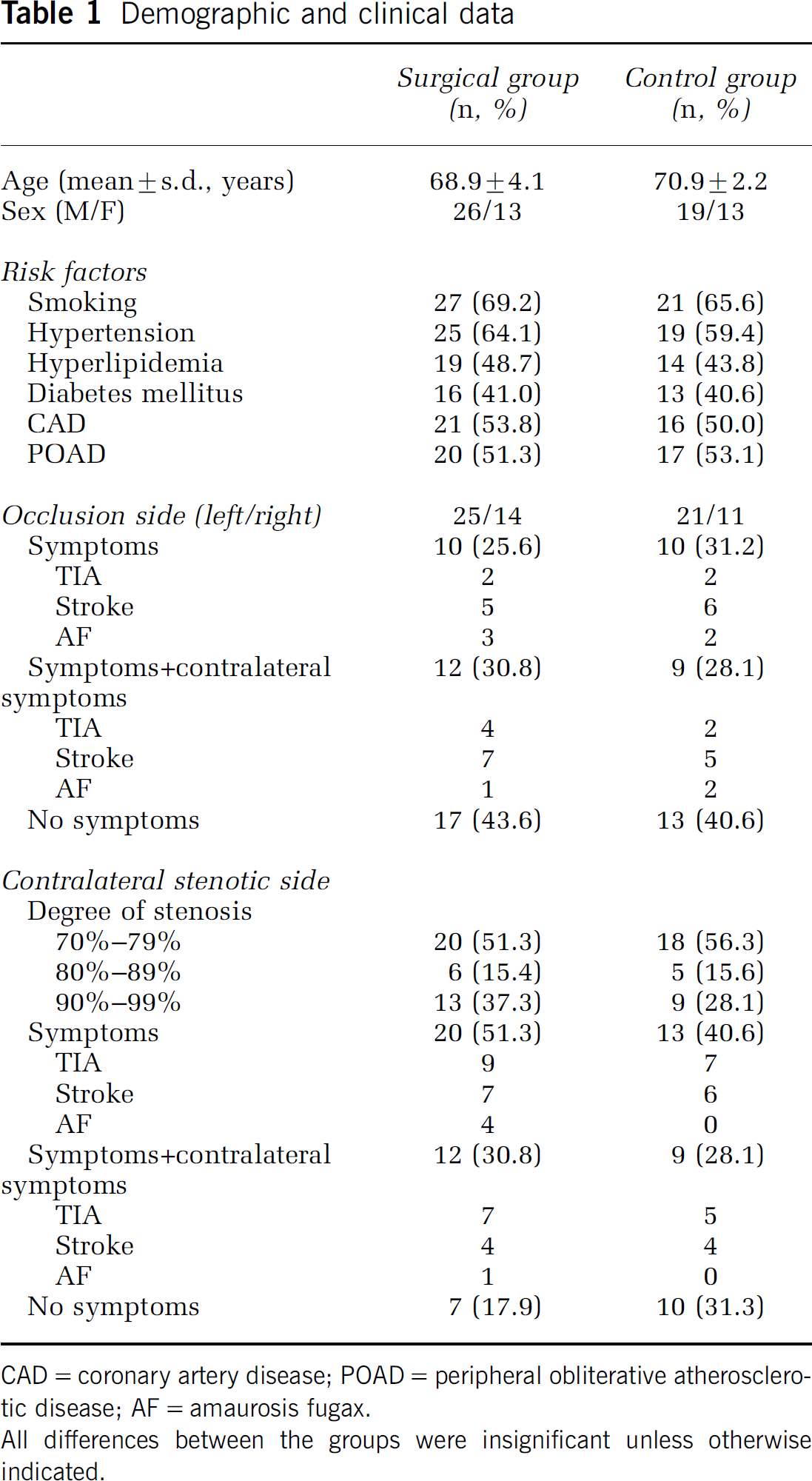
CAD = coronary artery disease; POAD = peripheral obliterative atherosclerotic disease; AF = amaurosis fugax.
All differences between the groups were insignificant unless otherwise indicated.
There were no perioperative (30-day) deaths or strokes. Two patients, one of them with symptomatic CS, experienced a hemispheric TIA ipsilateral to CO within the first 7 days after CEA. Postoperative DUS investigations showed no occlusion or restenosis of the operated vessel.
Although the incidence of late strokes was higher in the control group with respect to the surgical group (18.8%, 6/32 versus 7.7%, 3/39, P = 0.28), the difference was not statistically significant (Table 2). Of the three strokes that occurred in the surgical group, one was lacunar and ipsilateral to a symptomatic CO, and two were atheroembolic and ipsilateral to the operated vessel (one symptomatic and the other asymptomatic carotid lesion). In the control group, one stroke was hemodynamic and ipsilateral to a symptomatic CO, four (one of them fatal) were atheroembolic and ipsilateral to CS (two symptomatic and two asymptomatic carotid lesions), and one was due to occlusion of a symptomatic CS and proved fatal (Table 2). Kaplan-Meier analysis showed that freedom from stroke at 1, 3, 5, and 10 years was 100%, 100%, 97%, and 90% in the surgical group and 97%, 91%, 87%, and 78% in the control group (P=0.16). Overall, there were 11 late deaths. The mortality rate was significantly higher in the control group (28.1%, 9/32 versus 5.1%, 2/39, P = 0.02), but there were two stroke-related deaths (both strokes occurring in the hemisphere ipsilateral to a symptomatic CS) in the control group. The other causes of death were myocardial infarction (n = 7) and malignancy (n = 2). The two deaths that occurred in the surgical group were due to myocardial infarction.
Long-term (1-150 months; mean, 10 years) stroke and mortality rates
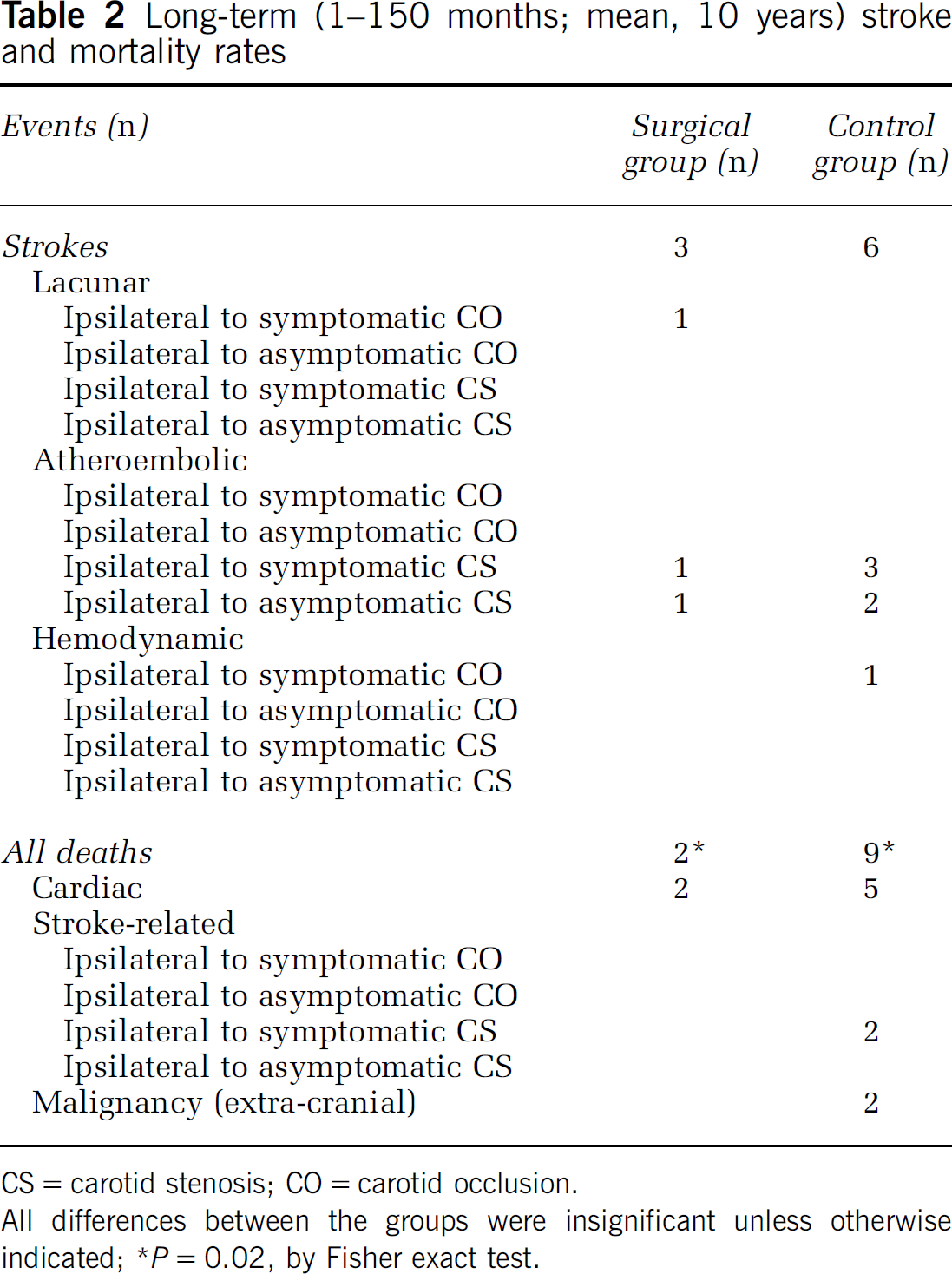
CS = carotid stenosis; CO = carotid occlusion.
All differences between the groups were insignificant unless otherwise indicated; *P = 0.02, by Fisher exact test.
No statistical difference was observed between the two groups with respect to the degree of CS when the PI was considered. The PI was comparable at the baseline, although it was lower on the side of CO in symptomatic patients, with a trend towards significance (P = 0.08). After CEA, the PI improved on the operated side, with no significant difference between symptomatic and asymptomatic patients, and remained stable on the contralateral side (Table 3).
TCD characteristics: PI

All differences between the groups were insignificant unless otherwise indicated; *P = 0.08, by Student's t-test.
There was no significant difference in the percentage of patients with intracranial collaterals between the surgical group before CEA and the control group (Table 4). The proportion of patients with collateral flow via the AcoA increased significantly from 61.5% before to 89.7% after CEA (P = 0.01), while the percentage of patients with collateral flow via the PcoA or with V-B activation decreased from 51.3% before to 43.6% and 41% after CEA, respectively. Moreover, there was no significant change in the proportion of patients with retrograde flow via the OphA on the side of CO (from 61.5% before to 58.9% after CEA). In the control group, no variations were observed in the proportion of patients with collateral flow via AcoA, PcoA, and OphA (Table 4).
Intracranial collaterals
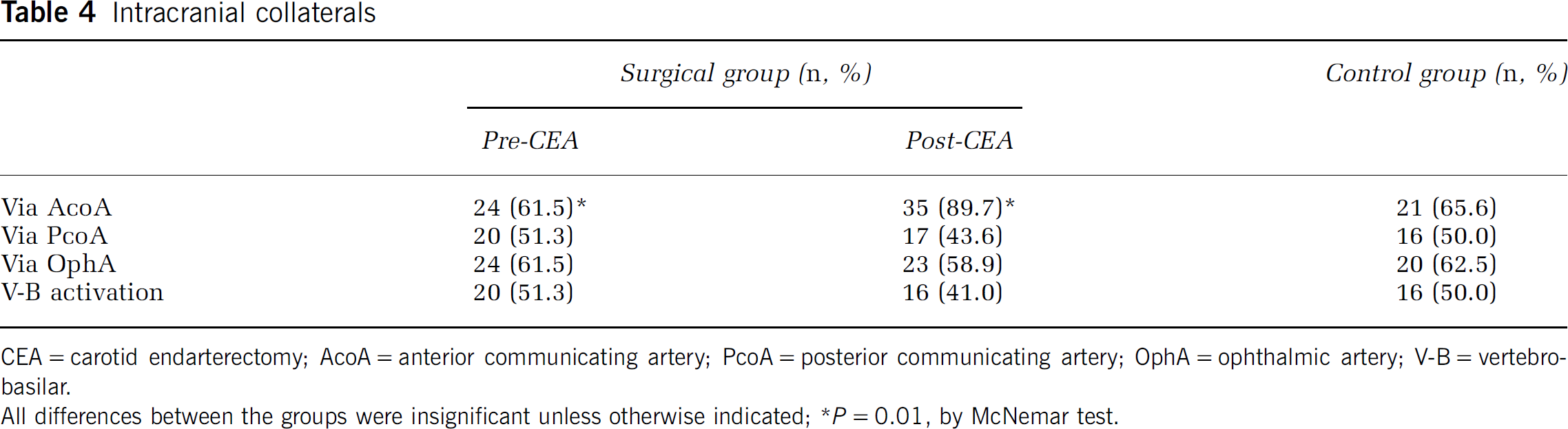
CEA = carotid endarterectomy; AcoA = anterior communicating artery; PcoA = posterior communicating artery; OphA = ophthalmic artery; V-B = vertebro-basilar.
All differences between the groups were insignificant unless otherwise indicated; *P = 0.01, by McNemar test.
Cerebral VMR at the baseline was compromised in most surgical and control patients (89.8% and 87.5%) on the side of CO, with no significant difference between symptomatic and asymptomatic patients (Table 5). Within 1 month after CEA, the improvement was present in 85.7% of patients (30 of 35): in particular, the recovery was full in 57.1% of patients (20 of 35), it was marked in 28.6% (10 of 35), and present, but slight, in the remaining 14.3% (5 of 35). In each of these three subgroups, there was no significant difference between symptomatic and asymptomatic patients. At 3 months, all operated patients showed an improved VMR. During the follow-up, this improvement persisted in 87.9% of the operated patients (29 of 33), while in 12.1% (4 of 33) it deteriorated, probably due to the onset of a severe cardiac dysfunction.
Cerebral VMR
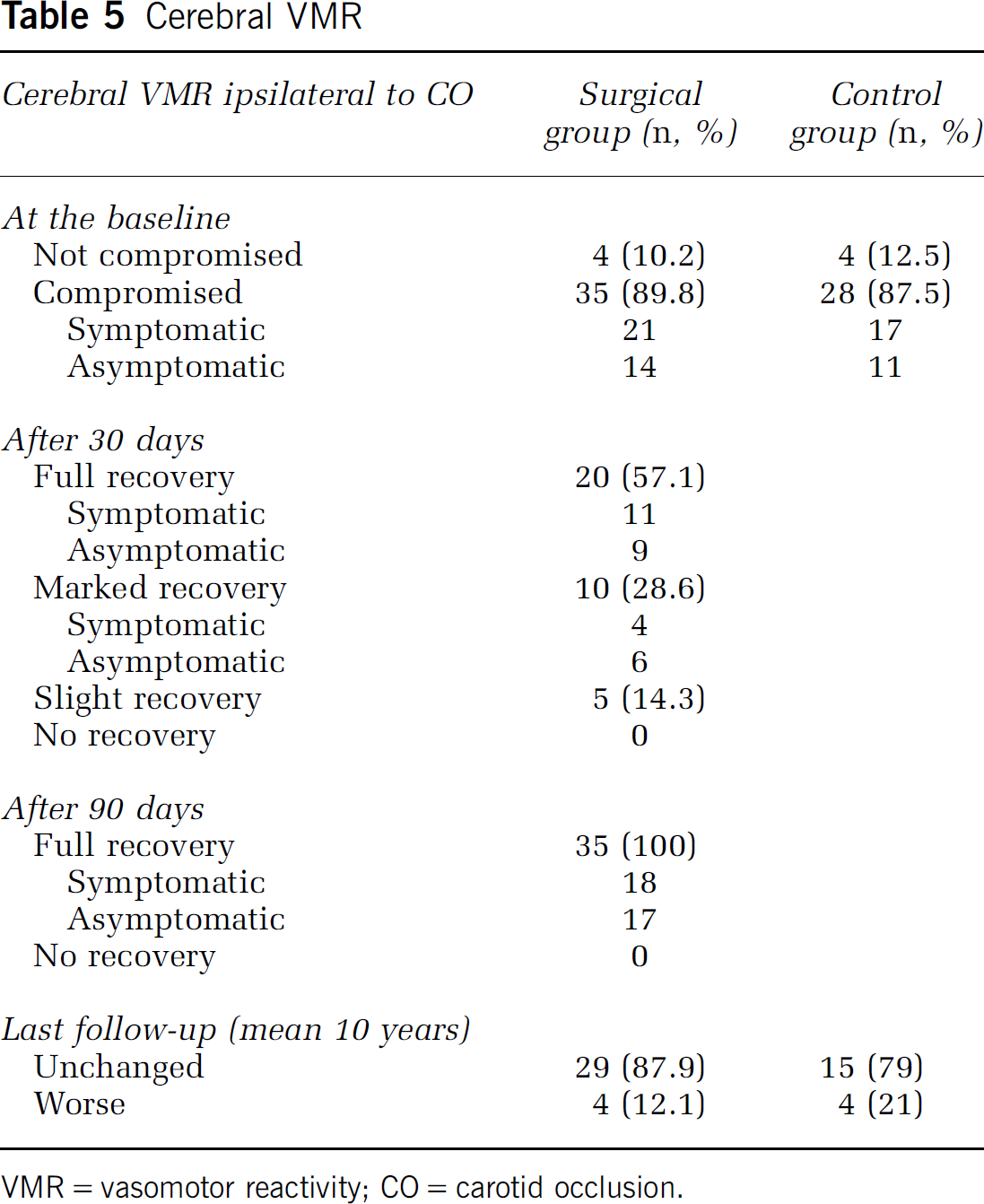
VMR = vasomotor reactivity; CO = carotid occlusion.
No spontaneous VMR improvement was recorded in the control group; in 79% of patients (15 of 19) it remained compromised, and in 21% (4 of 19) it became worse due to a progression of CS.
Discussion
The most important findings of this study are as follows: (1) cerebral hemodynamics and VMR are seriously impaired in patients with CO and severe contralateral CS, irrespective of the clinical presentation, and they do not recover spontaneously with time; (2) a significant cerebral hemodynamic improvement is observed in almost all patients within 1 month after CEA and in all patients within 3 months, especially on the contralateral side, regardless of any prior presence of symptoms; (3) during an average 10-year follow-up, this improvement remains unchanged; (4) despite this lasting recovery, the beneficial effect of CEA is not translated into a prolonged survival advantage.
By improving the BF in the operated vessel, CEA contralateral to CO increases the cerebral perfusion pressure and determines a redistribution of the blood supply to the brain, as shown by the subsequent increase in collateral flow via the AcoA to the side of CO and a decrease in the collateral flow via the BA. There was no decrease in the prevalence of retrograde flow in the OphA on the side of CO. This would suggest that, despite the improved collateral flow via AcoA and the increased cerebral perfusion pressure, BF to the hemisphere ipsilateral to CO might still be suboptimal after CEA. These findings closely parallel those reported by Rutgers et al (2001) in a similar, recently published study.
Severe CS and CO are known to decrease PI by reducing inflow, and the retained pulsatility is thought to be an indicator of lower impedance of collateral vessels. The preoperative difference in PI between asymptomatic and symptomatic CO groups is in accord with previous findings (Schneider et al, 1988). We also confirm the increase of PI after CEA on the operated side. In previous reports (Soinne et al, 2003), there was a greater improvement of PI in symptomatic CS, supporting the hypothesis of a more severe initial hemodynamic impairment in symptomatic CS. In our study we found, however, no preoperative difference and a similar post-CEA improvement in both symptomatic and asymptomatic CS; these results are not surprising at all if one thinks that in the majority of symptomatic CS we deal with an embolic cerebral event and not a hemodynamic stroke.
Cerebral VMR on the side of the CO was compromised in the preoperative assessment of surgical patients and in the first assessment of unoperated patients. As cerebral arteries and arterioles distal to CO may dilate to maintain an adequate BF (Derdeyn et al, 1999; Rutgers et al, 2001), the low VMR in response to acetazolamide administration showed that these vessels have a limited reserve capacity for dilation. Vasomotor reactivity improved significantly in operated patients on the side of CO, however, showing that BF to the ipsilateral hemisphere increased after CEA, in line with PI, velocity, and collateral flow measurements. The effect of contralateral CEA on VMR was unrelated to the clinical presentation with respect to CO.
Cerebral VMR recovery after CEA occurred in different ways. Individual anatomical and functional characteristics could account for this staged response to the increased BF after CEA. The improvement persisted during the follow-up period. However, in contrast with the report from Widder et al (1994), who suggested the possibility of a spontaneous improvement of an initially impaired cerebral VMR in most of patients with CO, no such spontaneous improvement was recorded during a mean follow-up of 10 years in unoperated patients.
Although CEA contralateral to CO has been associated with a relatively high perioperative stroke risk rate (Gasecki et al, 1995), recent studies have shown that CEA might be performed in these ‘high-risk’ patients with perioperative results comparable with those recorded in patients with no contralateral CO (Ballotta et al, 2004b; Mozes et al, 2004). In our study, albeit on a small sample, there was a very low perioperative complication rate and no mortality. However, according to recent guidelines, for CEA to be worthwhile, the long-term benefit must outweigh the perioperative risk (Biller et al, 1998). The outcome of this study (no restenosis, no late occlusion) correlates closely with the findings of a recently published large series, demonstrating that CEA is a safe, effective and durable procedure (Ballotta et al, 2004a). The long-term follow-up showed that, although the stroke rate was twice as high in unoperated patients as in operated patients, the difference was not statistically significant; moreover, there were only two stroke-related deaths in the control group, both strokes occurring ipsilaterally to CS. Carotid endarterectomy contralateral to CO significantly and persistently improves cerebral hemodynamics in both hemispheres but, because the late mortality in both groups was related more to cardiac than cerebrovascular events, a longer freedom from stroke in operated patients was not translated into a longer survival.
The present study has the inherent drawbacks of a nonrandomized protocol; although the data were collected prospectively, the nature of the analysis was retrospective. Moreover, although the two groups were ideal for the purposes of a valid comparison and the follow-up was long enough to draw to a definite conclusion, the sample was small. The absence of late stroke-related deaths in the surgical group and the low incidence of late major neurological events in both groups explain the study's relative lack of power, preventing any final consideration on the potentially beneficial effects on mortality and morbidity of CEA contralateral to CO.
In conclusion, within 3 months after surgery, CEA contralateral to symptomatic and asymptomatic CO in patients with severe CS determines a durable cerebral hemodynamic improvement not only on the side of the CEA but also on the contralateral side, with no difference between symptomatic and asymptomatic patients. This lasting recovery nonetheless does not seem to be able to offer any beneficial effects on late mortality and morbidity in this patient subgroup.
