Abstract
Stroke is the second leading cause of death and a significant cause of disability worldwide. Recent advances in DNA sequencing, proteomics, metabolomics, and computational tools are dramatically increasing access to the identification of host–microbiota interactions in systemic diseases. In this review, we describe the accumulating evidence showing how human microbiota plays an essential role in cerebrovascular diseases. We introduce the symbiotic relationships between microbiota and the mucosal immune system, focusing on differences by anatomical sites. Microbiota directly or indirectly contributes to the pathogenesis of traditional vascular risk factors including age, obesity, diabetes mellitus, dyslipidemia, and hypertension. Moreover, recent studies proposed independent effects of the microbiome on the progression of various subtypes of stroke through direct microbial invasion, exotoxins, functional amyloids, inflammation, and microbe-derived metabolites. We propose the critical concept of gene-microbial interaction to elucidate the heterogeneity of stroke and provide possible therapeutic avenues. We suggest ways to resolve the vast inter-individual diversity of cerebrovascular disease and mechanisms for personalized prevention and treatment.
Keywords
Background
Stroke is one of the leading causes of death and a primary cause of disability worldwide. Traditional risk factors contributing to the development of stroke include age, obesity, diabetes mellitus, dyslipidemia, hypertension, smoking, physical inactivity, diet, and alcohol consumption. 1 However, in most cases of stroke, the trigger of the event remains unknown. In fact, not all elderly patients with hypertension, diabetes mellitus, or smoking have strokes. Furthermore, the contribution of these traditional risk factors to stroke differs among ethnicities, 1 suggesting the need for personalized prevention of stroke. Recently, rapid advances in metagenomics, meta-transcriptomics, and meta-proteogenomics have identified novel host–microbiota interactions. 2 Moreover, emerging evidence has demonstrated the role of both infectious diseases and dybiosis (imbalance in the microbiota) in the development of stroke. 3 Increased consideration of the effects of the microbiota will provide us with crucial benefits to deepen our understanding of the pathophysiology of stroke. Here, we comprehensively review the influence of microbiota on risk factors, atherosclerosis, the subtypes of stroke, and vascular cognitive impairment (VCI), while also examining the underlying molecular mechanisms.
Tissue-specific immunity and microbiota
The difference in microbiota between the oral cavity and gut
The human gut is home to several trillions of microbes that form a bidirectional relationship with the human host. 4 Time-series metagenomic analyses using next-generation sequencing 5 and phylogeny-based metric 6 revealed that the human microbiota begins to colonize in early life. 7 Exposure to microbes starts before birth as some bacteria in the mother’s oral cavity can be transmitted through the placenta. 8 The route of delivery, breastfeeding, infant care-associated behavior, diet, and gender altogether contribute to the establishment of microbiota in different body sites, including the skin, oral cavity, and gastrointestinal (GI) tract during infancy. 9 As a consequence of ecological processes, in adults, the microbiota of the oral cavity is distinct from that of the GI tract. 10 A statistical approach using Dirichlet multinomial mixture models, which assigned samples to community subtypes, demonstrated that changes of oral bacterial populations may subsequently give rise to a bacterial community shift in the gut, resulting in dysbiosis. 11
The mucosal immune system of the oral cavity and the gastrointestinal tract
The GI mucosal immune system consists of three significant compartments, the epithelium, lamina propria, and gut-associated lymphoid tissue. The mucosal immune system in the oral cavity, as the gateway to the GI tract, shares functions with the GI tract in the following four points
12
:
Enterocytes and Paneth cells produce antimicrobial peptides (immunoglobulins, defensins, lysozyme, and cathelicidins), and goblet cells produce mucin in the GI tract, which contributes to innate host defense against gut microbiota.
13
In the oral cavity, oral epithelial cells, neutrophils, and salivary glands secrete these antimicrobial peptides.
14
Intraepithelial lymphocytes, especially regulatory T cells, respond to antigens presented with major histocompatibility complex molecules and express toll-like receptors (TLRs); they also eliminate foreign antigens and exhibit surveillance functions in both the GI tract and oral cavity.
12
Dendritic cells (DCs) in the GI tract and oral cavity acquire luminal antigens via extension processes. After taking up the antigen, DCs migrate into the mucosa-associated lymphoid tissue and lymph nodes, initiating the adaptive immune response by inducing T cell proliferation and differentiation.15,16 In the GI tract, microfold cells (M cells) transport luminal antigens to antigen presenting cells, stimulating B cells in follicle to differentiate into plasma cells in Peyer’s patches. Corresponding to M cells in the GI tract, a group of M cell-like cells has been identified in the epithelium of the palatine tonsil crypt in the oral cavity.
17
On the other hand, the gingival crevice in the mouth has unique anatomical and immunological features. The junctional epithelium, where the mucosa meets the tooth at the base of gingival tissue, has few layers of thickness attached to the tooth via hemidesmosomes. The junctional epithelium connection to the tooth is highly permeable, allowing the bidirectional passage of oral microbiota, host-protective factors, and inflammatory cells (Figure 1). 18 Both histological studies and flow cytometry analyses showed that neutrophils, resident terminally differentiated memory subset of CD4+ and CD8+ cells, and Foxp3+ regulatory T cells are more abundant in the gingival crevice than in the GI tract.19,20
The role of microbiota in the host immune system
The Human Microbiome Project showed that the oral and gut microbiota play essential roles in the maturation of the host immune systems (Figure 2)
21
:
The regulation of IgA secretion involves crosstalk between innate immunity and commensal bacteria. Recent approaches assessing the coating of gut microbes with host-derived IgA suggested that microbiota-reactive and polyreactive IgA antibodies arise naturally in all naïve B cell populations, significantly in IgA-secreting plasma cells.
22
Nevertheless, evidence from germ-free mice and fecal microbiota transplantation (FMT) revealed that commensal bacteria increased secretion of IgA and antimicrobial peptides into the gut lumen,
23
which indicates that the orchestra of innate immunity and commensal bacteria is necessary to maintain IgA synthesis in the local mucosal environment. Commensal bacteria induce IgA production through living bacteria and bacterial products,
24
as well as intestinal epithelial cell-derived B-cell stimulating and activating factors.
25
Hematopoiesis and innate immunity. The microbiota can impact stem-cell-derived myeloid cell development, and the size of the bone marrow myeloid cell pool correlates with the complexity of the gut microbiota.
26
Circulating TLR ligands and short-chain fatty acids (SCFAs), which are gut microbial metabolites derived from carbohydrates, promote DC and monocyte trafficking into the bloodstream (reviewed in Thaiss et al.
27
). Moreover, SCFAs play essential roles in the central nervous system, critically mediating the maturation and function of brain microglia and the blood–brain barrier (BBB).
28
Bridge between innate and adaptive immunity. Interleukin (IL)-17-producing effector T helper (Th17) cells are crucial for maintaining tissue physiology. Th17 acts on epithelial cells to enhance anti-microbial defense and epithelial barrier integrity via IL-17 and IL-22.
29
Th17 cells initially have a central role in preventing infection by several species of pathogenic bacteria; however, stimulation with IL-23
30
or high salt diet
31
makes Th17 cells proinflammatory. Furthermore, it is now becoming clear that specific microbes, including segmented filamentous bacteria (SFB),
Traditional risk factors and microbiota
Aging
Aging is one of the most critical risk factors for all subtypes of stroke.
34
The proinflammatory status accompanied by aging, which was termed “inflamm-aging” in 2000 by Claudio Franceschi,
35
is a risk factor for cerebrovascular and neurodegenerative diseases.
36
Among the underlying mechanisms for inflamm-aging, dysbiosis is central due to its crosstalk with dysregulation of metabolism and immune system,
35
along with genomic instability and mitochondrial dysfunctions.
37
In the elderly, the composition of gut microbiota is different from that of younger adults, with a higher proportion of
Metabolic diseases
In randomized controlled trials, lean-donor FMT in subjects with metabolic syndrome improved obesity and insulin sensitivity, suggesting that the microbiota can regulate host metabolism as an “organ.”
43
Accumulating evidence illustrates more detailed mechanisms linking either the presence of specific bacteria or the production of bacteria-derived metabolites with metabolic disorders.
44
Hypertension
Hypertension is the leading cause of stroke and heart diseases. Genetic and environmental factors influence the individual variation of increased blood pressure (BP). Extensive populational genome-wide association studies (GWAS) identified several genetic factors associated with blood pressure control, including the renin-angiotensin-aldosterone system, sympathetic nervous system, atrial natriuretic peptide signaling, and dopaminergic system; however, these genetic factors contribute to only 3% of hypertension in humans.
55
Now, growing evidence supports different roles of the GI tract in BP regulation, not only as an organ for salt absorption, but also controlling autonomic and endocrine functions. The gut microbiome in patients with hypertension is comprised by a higher percentage of bacteria from the genus
Stroke and acute or chronic infectious burden
As mentioned above, microbiota–host interactions are associated with various vascular risk factors of stroke, including aging, metabolic diseases, and hypertension. Recent case-crossover studies suggested acute infection as a possible trigger of stroke.
59
Cumulative exposure to chronic infectious diseases, or “infectious burden,” has been measured using serological profiles against several pathogens and inflammatory biomarkers. Using an analyzed serological profile for
Large artery atherosclerosis and microbiota
Intra- and extracranial large artery atherosclerosis occurs concomitantly with systemic atherosclerosis. Atherosclerosis occurs at sites in the arterial tree where the laminar flow is disrupted and presents multiple pathological processes, including accumulation of lipid and extracellular matrix, migration of inflammatory cells, plaque rupture, and luminal thrombosis.
62
A meta-analysis assessing bacteria in atherosclerotic plaque samples confirmed the abundance of oral commensal bacteria, including
Cardioembolic stroke
Atrial fibrillation (Af) is one of the most important causes of cardioembolic stroke. Cardiac autonomic ganglionated plexuses facilitate electrophysiological modulation of the atrium and arrhythmogenesis. 75 A few studies have revealed the association between Af and microbiota in humans. 76 In a recent cohort study, a higher level of plasma TMAO was a significant independent risk factor of Af. 77 In a canine model of Af, TMAO increased the instability of atrial electrophysiology and aggravated acute atrial remodeling. 78
Cerebral small vessel disease
Cerebral small vessel disease (CSVD), including lacunar infarction, cerebral microbleeds (CMBs), enlarged perivascular spaces, leukoaraiosis, and cortical atrophy, is commonly observed in neuroimaging among elderly persons. CSVD contains two major pathological classifications, hypertensive vasculopathy and cerebral amyloid angiopathy (CAA), and is clinically relevant to stroke, including recent subcortical small infarction and intracerebral hemorrhage. Furthermore, the CSVD burden is highly relevant to cognitive decline.79,80 In a multiethnic cohort study, procalcitonin was associated with subclinical CSVD. 81 Additionally, LPS administration induces CMBs in wild-type mice. 82 Hereafter, we review the specific role of microbiota in the development of each subtype of CSVD.
Hypertensive vasculopathy
Pathological features of hypertensive vasculopathy include lipohyalinosis and fibrinoid necrosis. BBB disruption and inflammation are two crucial etiologies of hypertensive vasculopathy. Nakano et al.,
83
using vessel occlusion model mice, reported that
Cerebral amyloid angiopathy
Even though there is little human data investigating the role of the microbiome in patients with cerebral amyloid angiopathy (CAA), there is accumulating evidence about Alzheimer’s disease (AD), which has shared pathophysiology with CAA. The amyloid cascade hypothesis suggests that vascular amyloid-β (Aβ) accumulation plays a central role in elucidating the pathogenesis of AD.
88
However, repeated failures of anti-amyloidogenic trials prompted us to explore the brain–microbiota axis as an alternative to the amyloid cascade hypothesis, from initiation to aggravation of AD and CAA. We have proposed that functional bacterial amyloid proteins in the gut may cause cross-seeding of Aβ via a prion-like mechanism. In studies of aged rats, we showed that exposure to bacterial amyloid initiates cross-seeding of cerebral alpha synuclein aggregation and neuroinflammation.
89
For instance, curli, a functional amyloid protein expressed by
Future directions
Gene–microbial interaction
The single nucleotide polymorphisms (SNPs) of microbial-derived metabolite receptors and PRRs can contribute to the susceptibility of metabolic diseases and atherosclerosis. Among microbial-derived metabolites, SNPs of GPR modulate the effects of SCFAs on hypertension and type 2 diabetes mellitus, 96 and SNPs of farnesoid X receptors affect lipid and glucose metabolism. 97 Among PRRs, TLR-4 SNPs are associated not only with the differences in LPS responsiveness but also with progression of atherosclerosis or ischemic stroke. 98 Some studies report the association between oligomerization domain-containing protein 1 genotypes and susceptibility to cerebrovascular diseases. 99 However, these links between SNPs and cerebrovascular diseases are controversial for different ethnicities, implying that combination of microbiome and genome-wide association studies, called “mGWAS,” is essential to unravel the complexity of cerebrovascular diseases. 100
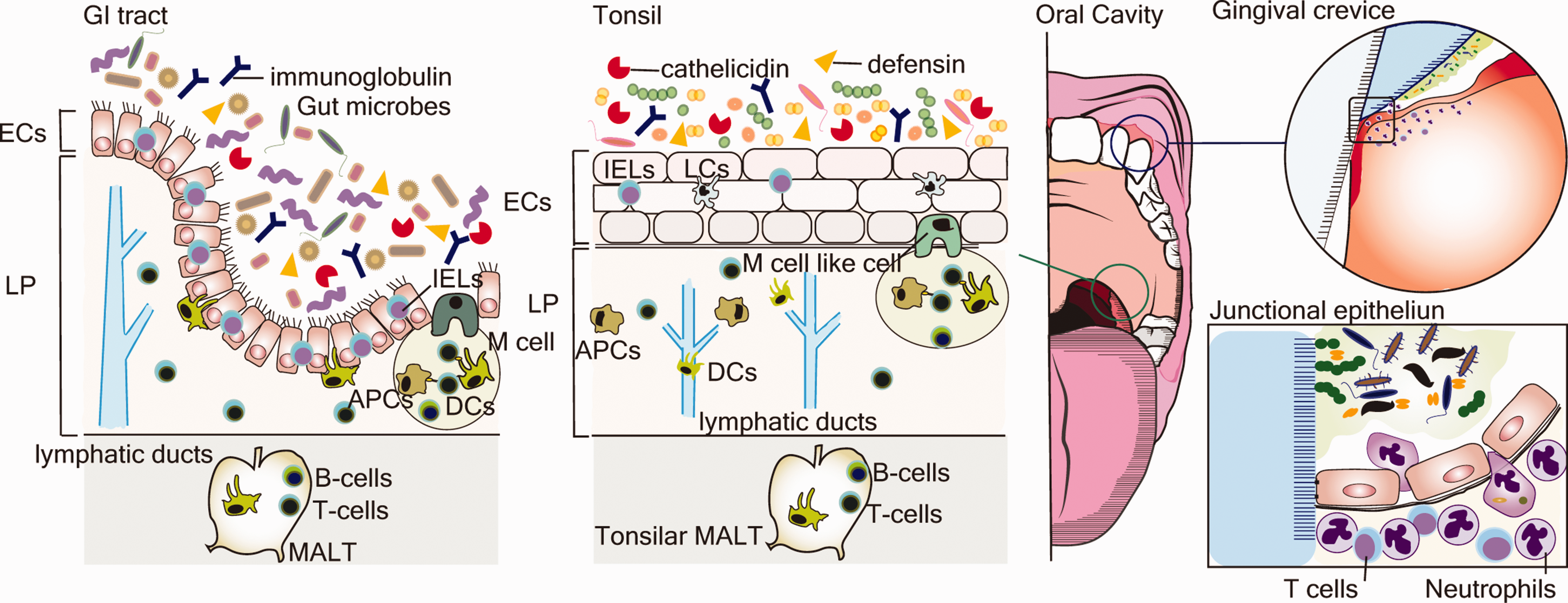
The mucosal immune system of the oral cavity and GI tract. The oral and GI immune systems share similar anatomical compartments, epithelium, LP, and MALT. IELs reside within the epithelial layer. LP-resident DCs directly uptake luminal antigens via extension process and migrate to MALTs, tonsillar MALTs in the oral cavity, and GALTs in the GI tract. M-cells in the epithelium of Peyer’s patches and M-cell-like cells in the epithelium of tonsil crypts pass the antigens to DCs or macrophages. Instead, the immunological interactions in the gingiva are specific to the oral cavity. The gingival crevice, the inner base of the gingiva, is lined with a few layers of non-keratinized highly permeable thin epithelium. In the gingival crevice, neutrophils and oral microbiome constantly transmigrate through JE and mediate inflammation even in steady state. DC: dendritic cell; GALT: gut-associated lymphoid tissue; GI: gastrointestinal; IEL: intraepithelial lymphocyte; JE: junctional epithelium; LP: lamina propria; MALT: mucosa-associated lymphoid tissue; M-cell: microfold cell.
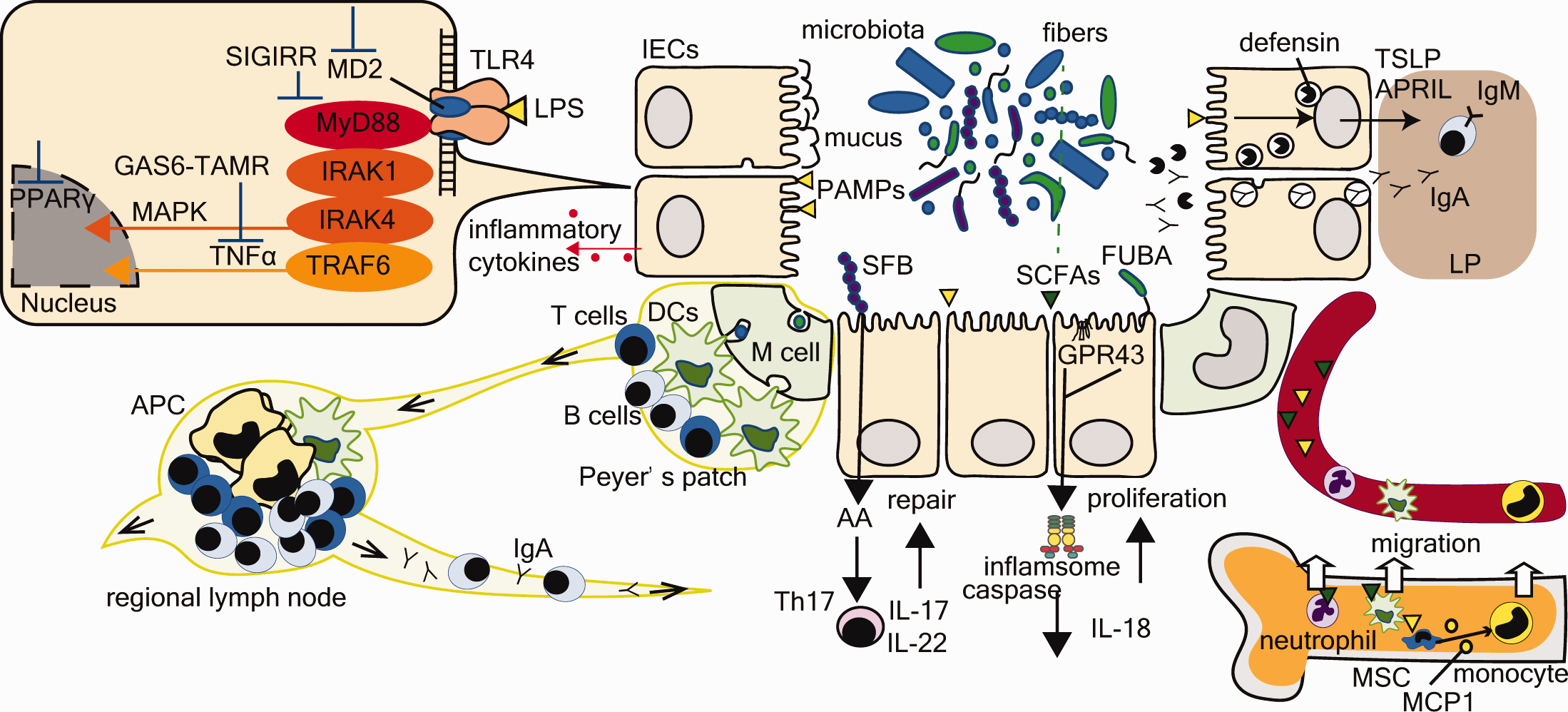
The interactions of the microbiota with the host immune system. Bidirectional interactions between the mucosal immune system and the microbiota. These include (1) the mucus layer, (2) epithelial cells (intercellular tight junction, antimicrobial peptide secretion, recognitions of MAMPs), (3) innate immunity (hematopoiesis, IgA secretion in LP, Inflammasome), and (4) acquired immunity (Th17 cell activation, DCs in LP, M-cells and APC cells in Peyer’s patches, regional lymph nodes). As a recognition of MAMPs in epithelial cells, for instance, TLR4-MD2 complex binding to LPS activates NF-κB and MAPK via the MyD88-dependent signaling pathway. Microbes control inflammatory reaction in epithelial cells activating or inhibiting these pathways (GAS6-TAMR axis, MD2, MyD88, SIGIRR, PPARγ). Metabolites or MAMPs of microbiota have a role in educating the host innate system. In bone marrow, SCFAs promote DC maturation and PAMPs induce MCP1 production in MSC, resulting in monocyte migration. Epithelial cell-derived cytokines TSLP and APRIL promote class-switch recombination and the production of IgA by B cells in intestinal LP, transmitted into mucosal layer. Not only microbiota, but also FUBA or SCFAs influence the inflammasome involving caspase 1 and proinflammatory cytokines. Th17 cell differentiation induced by commensal microbiota, including SBF, produce IL-17 and IL-22, which play a homeostatic role in microbiota–host interactions. The M-cell of Peyer’s patches transfers the luminal antigen to DCs and APCs, which is drained and induces T cell-dependent B cell maturation in MLNs. APC: antigen-presenting cell; APRIL: aproliferation-inducing ligand; DCs: dendritic cells; FUBA: functional bacterial amyloid; GAS6: growth-arrest specific gene 6; G-CSF: granulocyte-colony stimulating factor; GPR43: G-protein coupled receptor 43; IgA: immunoglobulin A; IL: interleukin; LP: lamina propria; LPS: lipopolysaccharide; M cell: microfold cell; MAMPs: microorganism-associated molecular patterns; MAPK: mitogen-activated protein kinase; MCP1: monocyte chemoattractant protein 1; MD2: myeloid differentiation factor 2; MLN: mesenteric lymph node; MSC: mesenchymal stem cell; MyD88: myeloid differentiation factor 88; NF-κB: nuclear factor-κB; PPARγ: peroxisome proliferator activated receptor γ; SBF: segmented filamentous bacterium; SCFAs: short-chain fatty acids; SIGIRR: single immunoglobulin interleukin-1 receptor-related molecule; TAMR: tyro3 axl mer receptor; TLR: Toll-like receptor; TSLP: thymic stromal lymphopoietin.
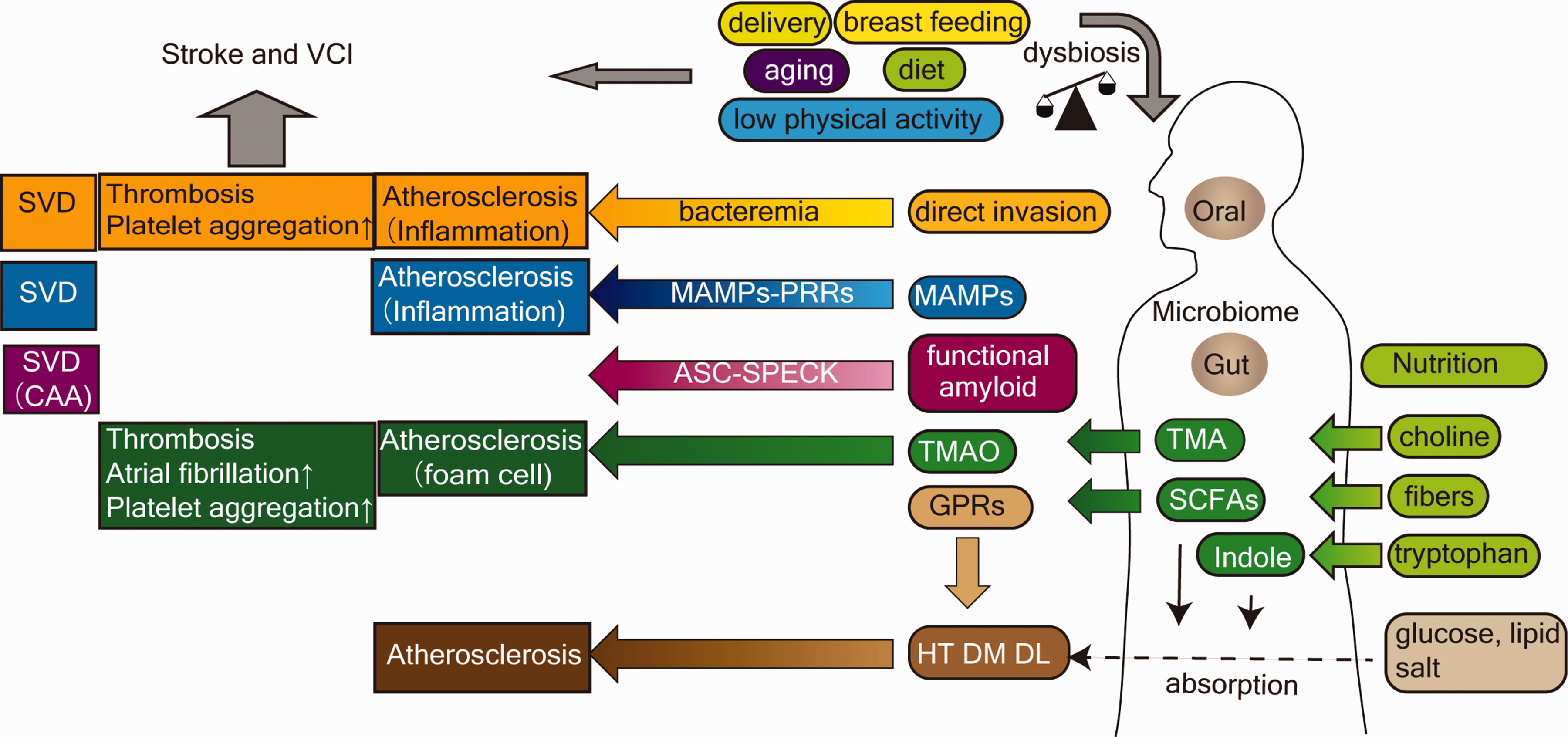
Overview of the interplay between microbiota and stroke or vascular cognitive impairment. Beside the traditional risk factors, including HT, DM, DL, physical activity, and aging, mounting evidence has shown the role of gut microbiota in the development of stroke. Delivery, breastfeeding, aging, diet, and physical activity are bidirectionally related to dysbiosis. There are several possible mechanisms between oral and gut microbiota and the development of stroke and VCI. Microbes in the oral cavity can act directly on the cerebrovascular system via bacteremia. PRRs recognize MAMPs, for example TLRs and LPS, lipoteichoic acid, or peptidoglycan, which subsequently stimulate immune cells and induce the production of inflammatory cytokines. Among bacterial metabolites, TMA and TMAO interact with coagulation and inflammatory cascades in atherosclerotic plaque. SCFAs and indoles affect the absorption rate of nutrients and have hormonal activity in metabolic homeostasis via various types of GPRs. Bacteria-derived functional amyloid induces Aβ aggregation in CAA through inflammasome secretion or the ASC-SPECK system. Aβ: Amyloid β; ASC-SPECK: adaptor protein apoptosis-associated speck-like protein containing a caspase recruitment domain; CAA: cerebral amyloid angiopathy; GPRs: G-protein-coupled receptors; DL dyslipidemia; DM: diabetes mellitus; HT: hypertension; LPS: lipopolysaccharide; MAMPs: microorganism-associated molecular patterns; SCFAs: short-chain fatty acids; SVD: small vessel disease; TLRs: toll-like receptors; TMA: trimethylamine; TMAO: Trimethylamine N-oxide; VCI: vascular cognitive impairment; PRRs: pattern recognition receptors
Gene–microbial interaction also gives us the novel insight that human genetics and microbiota have shared mechanisms in the development of each subtype of stroke, including modulation of HDAC in large artery atherosclerosis, modulation of type IV collagen in hypertensive vasculopathy, and modulation of Aβ-NLPR3-ASC-SPECK signaling in CAA via apolipoprotein E (APOE) and triggering receptor expressed on myeloid cells (TREM2). HDAC is a pivotal epigenetic factor modulating chromatin topology and regulating gene expression. HDAC9 polymorphisms are associated with large artery atherosclerosis and stroke, which is inhibited by butyrate, as previously mentioned.
101
Additionally, COL4A1 and COL4A2 polymorphisms are associated with hemorrhagic stroke, disrupting type IV collagen of the vascular basement protein,
102
where Cnm-positive
Prevention perspectives
The antibiotic eradication of
Bacterial cell-to-cell communication
The quorum-sensing (QS) system of bacterial cell-to-cell communication is a novel therapeutic target. Microbial QS systems regulate the expression of many virulence factors, biofilm synthases, and even clustered regularly interspaced short palindromic repeat (CRISPR)-associated defense system. 106 QS-interfering agents that potentially reduce bacterial virulence and prevent biofilm formation of infectious pathogens are expected to influence metabolic disorders affecting the microbiome. 107 However, most QS-interfering agents are still in preclinical trials. Further research is necessary to assess the side effects toward non-target bacteria and the possibility of resistance development in humans.
Microbiome
Prebiotics are compounds that are digested by bacteria which may be beneficial to the host, depending on the nature of the host’s microbiome. Probiotics, the direct administration of beneficial microbes, and FMT are attractive therapies to shape a healthy microbiome and modify immune and metabolic balance. However, these microbiome-modifying therapies bear many unsolved questions.108,109 Personalized nutrition in combination with beneficial microbes might be an integrated approach to microbiome-modifying therapies.
110
Furthermore, as the proof-of-concept study with
Microbial-derived metabolites
Roberts et al. 112 proposed that small molecules inhibiting microbial production of TMA in the gut lumen suppress thrombosis formation and prevent atherosclerosis. Chambers et al. 113 showed that oral administration of inulin-propionate ester can deliver propionate to colon and ameliorate obesity and glucose homeostasis. This indicates the beneficial role of SCFAs and GPR modulators as novel preventive interventions for metabolic disorders and atherosclerosis. This work illustrates that the new strategies of leveraging the host–microbiota interactions as druggable molecular targets show promise
Immunomodulation
The microbiota interacts with the host immune system through immune cell polarization, stimulating MAMP-PRR signals, Aβ-NLPR3-ASC-SPECK signals, and inducing anti- or pro-inflammatory cytokines. Several studies attempted to investigate new therapeutics modulating these chemical mediators.114,115 Fingolimod, a sphingosine-1-phosphate receptor modulator, salvaged penumbra tissues in patients with acute internal carotid artery or middle cerebral artery occlusion in a clinical pilot trial. 116 Subcutaneous administration of anakinra, an IL-1 receptor antagonist, significantly decreased IL-6 and C-reactive protein, which are associated with worse clinical outcomes after stroke, in a randomized phase 2 clinical trial. 117 mTORC1 is another possible therapeutic target associated with the host immune–microbiota interaction. Recent evidence suggests that rapamycin, mTORC1 inhibitor, induces neuroprotective autophagy and pleiotropic effects on BBB and inflammation. 118 Microbiota and microbial metabolites control mTORC1 expression. 119 Finally, the collagen-binding microbial surface components recognize adhesive matrix molecules, which are common invasive and adhesive factors for various types of virulent Gram-positive cocci, 120 might become possible targets for vaccine design to prevent infectious endocarditis and specific subtypes of CSVDs.
Conclusions
Firstly, it must be emphasized that the microbiota is our most substantial environmental exposure. They represent an enormous potential threat to survival, a threat to which the evolution of both the host and the microbes has adapted over the past several million years. Thus, it is critical that the host tolerates beneficial organisms, and that pathogens do not acquire immune tolerance. It is similarly vital for the microbes that we tolerate them. These inter-relationships are the foundation for the influences of the microbiota on immune systems. We propose several interactions by which the microbiota may impact stroke and VCI in humans. Mechanisms involved in these interactions include innate and adaptive immunity, epigenetic alteration of protein expression, proteostasis, hematopoiesis, coagulation, hormone secretion, autonomic nerve system, and metabolism.
Secondly, in particular, we have shown the differences in the microbiota and host defense systems between the GI tract and oral cavity, suggesting the concept of “Microbiota–Oral–Brain Axis,” which is distinct from the “Microbiota–Gut–Brain Axis.” The role of the “Microbiota–Gut–Brain Axis” in human health is receiving increasing scientific and public attention. However, the influence of “Microbiota–Oral–Brain Axis” has not yet been properly recognized for its importance. According to the U.S. National Library of Medicine, PubMed database, there were 22,349 citations for “microbiota” and “gut,” while there were only 4743 citations for “microbiota” and “oral” (4 February 2020). Bidirectional gut–brain interactions involve several signaling molecules, immune mediators, and gut hormones by the microbiota. Conversely, considering that the nasal and oral cavities are anatomically close to the brain, microbiota and their metabolites can have direct influences through the blood, the cranial nerves and the central nervous system.
Despite promising data in both animal and human studies, it is challenging to integrate these findings into personalized prevention and treatment for stroke. As described above, the microbiota is influenced by genetics, nutrition, and other lifestyle factors. Thus, epidemiological research requires careful consideration of ethnicity, genetics, and lifestyle factors.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grant-in-Aid for Japan Society for Promotion of Science Fellows to S.T (20 J), Grant-in-Aid for Challenging Exploratory Research to M.I. (16K14573, 19K22610), Japan Heart Foundation Research Grant, Smoking Research Foundation, Novartis Research Grants, the University of Louisville and the Jewish Heritage Fund for Excellence, W. Cowan and the family of E. A Ford II.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
