Abstract
The decision to perform decompressive hemicraniectomy (DHC) by default in malignant hemispheric stroke (MHS) remains controversial. Even under ideal conditions, DHC usually results in moderate to severe disability. The present study for the first time uses neuroimaging to identify independent outcome predictors in a prospective cohort of 96 MHS patients undergoing DHC. The primary outcome was functional status according to the modified Rankin Scale (mRS) at 12 months and categorized as favorable (mRS 0–3) or unfavorable (mRS 4–6). At 12 months, 19 patients (20%) reached favorable and 77 patients (80%) unfavorable outcome. The overall mean infarct volume was 328 ± 114 ml. Multivariable logistic regression identified age per year (OR 1.14, 95% CI 1.04–1.24; p = 0.005), infarct volume per cm3 (OR 1.012, 95% CI 1.003–1.022; p = 0.013), thalamic involvement (OR 8.65, 95% CI 1.04–72.15; p = 0.046) and postoperative pneumonia (OR 5.52, 95% CI 1.03–29.57; p = 0.046) as independent outcome predictors, which was confirmed by multivariable ordinal regression for age (p = 0.004) and infarct volume (p = 0.015). The infarct volume threshold for reasonable prediction of unfavorable outcome in our patients was 270 cm3, which in the future may help prognostication and development of clinical trials on DHC and outcome in MHS.
Introduction
The natural course of malignant hemispheric stroke (MHS) or malignant middle cerebral artery (MCA) infarction is associated with a mortality rate of up to 80%.1,2 Decompressive hemicraniectomy (DHC) decreases mortality by lowering the risk of transtentorial herniation. Depending on the age of the patient, early DHC was shown to significantly reduce one-year mortality by 33% to 50% without increasing severe disability in European randomized controlled trials (RCTs).3,4 However, even under ideal conditions, only 43% have the chance to reach clinical outcome with only mild-to-moderate disability.3 In patients beyond 60 years of age, this proportion is reduced even further to merely 6%. 4 Since recent surveys among patients who underwent DHC for treatment of MHS confirm that survival at the cost of disability may be acceptable to some but not to others,5,6 the identification of outcome-associated variables could facilitate clinical decision making in regard whether or not to initiate surgical treatment based on the patient’s living will. Therefore, the aim of the present study was to investigate the prognostic association between clinical, demographic and for the first time also neuroimaging variables and 12-month outcome in a prospective cohort of MHS patients undergoing DHC.
Subjects and methods
Patient management
The study was approved by the ethics committee of the Charité-Universitätsmedizin Berlin, Germany (EA1/156/14) and conducted in accordance with the Declaration of Helsinki. Informed consent was waived due to the retrospective nature of the study. Ninety-six patients that underwent DHC between January 2006 and June 2012 for treatment of space-occupying MCA infarction with the risk of malignant edema development were included. ‘Malignant’ MCA infarction (=MHS) was defined according to the uniform inclusion criteria of the three RCTs on hemicraniectomy, DECIMAL, DESTINY and HAMLET, and included a severe MCA syndrome with dense hemiplegia, head and eye deviation, hemineglect and aphasia when the dominant hemisphere was involved next to an impaired and progressively deteriorating level of consciousness over the first 24–48 h and definite infarction of two-thirds of the MCA territory with or without additional infarction of the ipsilateral anterior or posterior cerebral artery territory. Exclusion criteria, such as pre-morbid mRS > 1, pre-morbid Barthel-Index <95, other concomitant severe disease that would confound with treatment, or life expectancy <3 years were not applied. All patients were treated according to the guidelines of the German Society of Neurosurgery. Patients were in regular medical consultation during the first year after stroke for scheduling cranioplasty and monitoring treatment complications. For our analysis, follow-up data at 12 months were obtained by retrospective review of patient charts.
Surgery and management
DHC was performed in a standardized fashion and is described elsewhere in detail.4,7–9 Anesthesia was maintained with propofol and remifentanil. Mean arterial blood pressure was targeted between 70 and 80 mmHg. After surgery, patients were transferred to the intensive care unit. Intracranial pressure (ICP) was monitored and patients remained intubated and sedated until ICP was within normal ranges. A critical ICP threshold was defined as an ICP >20 mmHg for a period longer than 10 min and treated according to national and international guidelines with cerebrospinal fluid drainage, osmotic therapy and deep sedation. Blood gases, electrolytes and glucose were controlled every 4 h. A routine postoperative computerized tomography (CT) or magnetic resonance imaging (MRI) scan was performed within 24 h after surgery to rule out procedure related complications.
Neuroimaging analysis
A specialist blinded towards outcome performed volumetry and neuroimaging analysis. Neuroimaging characteristics shown in Table 2 were determined directly from postoperative CT scans or matched diffusion-weighted imaging (DWI) and fluid-attenuated inversion recovery (FLAIR) sequences of the postoperative MRI scans performed within 24 h after the procedure. MRI scans were available for 79 patients, whereas 17 patients (18%) received an early CT scan due to logistical reasons or postoperative ICP crisis that restricted prolonged supine positioning within the MRI scanner. Volumetric analysis was performed with the help of iPlan® Cranial software (BrainLAB, Feldkirchen, Germany) and VISAGE7 (Visage Imaging GmbH, Berlin, Germany). Volume gain was calculated as ipsilateral volume minus contralateral volume. Swelling was determined as volume gain divided by contralateral volume. Volume of infarction corrected for hemispheric swelling was calculated as volume of infarction divided by swelling plus 1.
Clinical outcome analysis
The primary outcome measure was functional status according to the modified Rankin Scale (mRS) score at 12 months and documented by an independent physician. Clinical outcome was categorized as favorable (survival with mild to moderate disability; mRS 0–3) or unfavorable (survival with severe disability or death; mRS 4–6) based on previous definitions with most patients5,6 and physicians 10 considering an mRS score of 3 as the cut-off between favorable and unfavorable.
Statistical analysis
Statistics were calculated for all variables using the SPSS 21.0 software package (SPSS Inc., Chicago, Illinois, USA). For univariate analysis, the χ2-test, Mann–Whitney U-test or Student’s t-test were used as appropriate to test covariates predictive of unfavorable (mRS 4–6) outcome and identify covariates associated with higher mRS scores at 12 months. Variables with p values less than 0.05 were entered into multivariable logistic and ordinal regression models with stepwise backward variable selection leaving only variables with significant coefficients in the model. Standard measures of diagnostic test validity for varying infarct volume threshold levels were calculated by areas under the curve (AUC) with 95% confidence intervals using receiver operating characteristics (ROC) analysis. The optimal threshold for predicting unfavorable outcome (mRS 4–6) was selected as the situation maximizing the Youden-Index (sensitivity + 1-specificity). 11 Statistical significance was set at p < 0.05. All tests were two-tailed. Because of the retrospective and exploratory nature of this study, no correction of α levels for multiple testing was used.
Results
Univariate analysis of demographic and clinical characteristics.
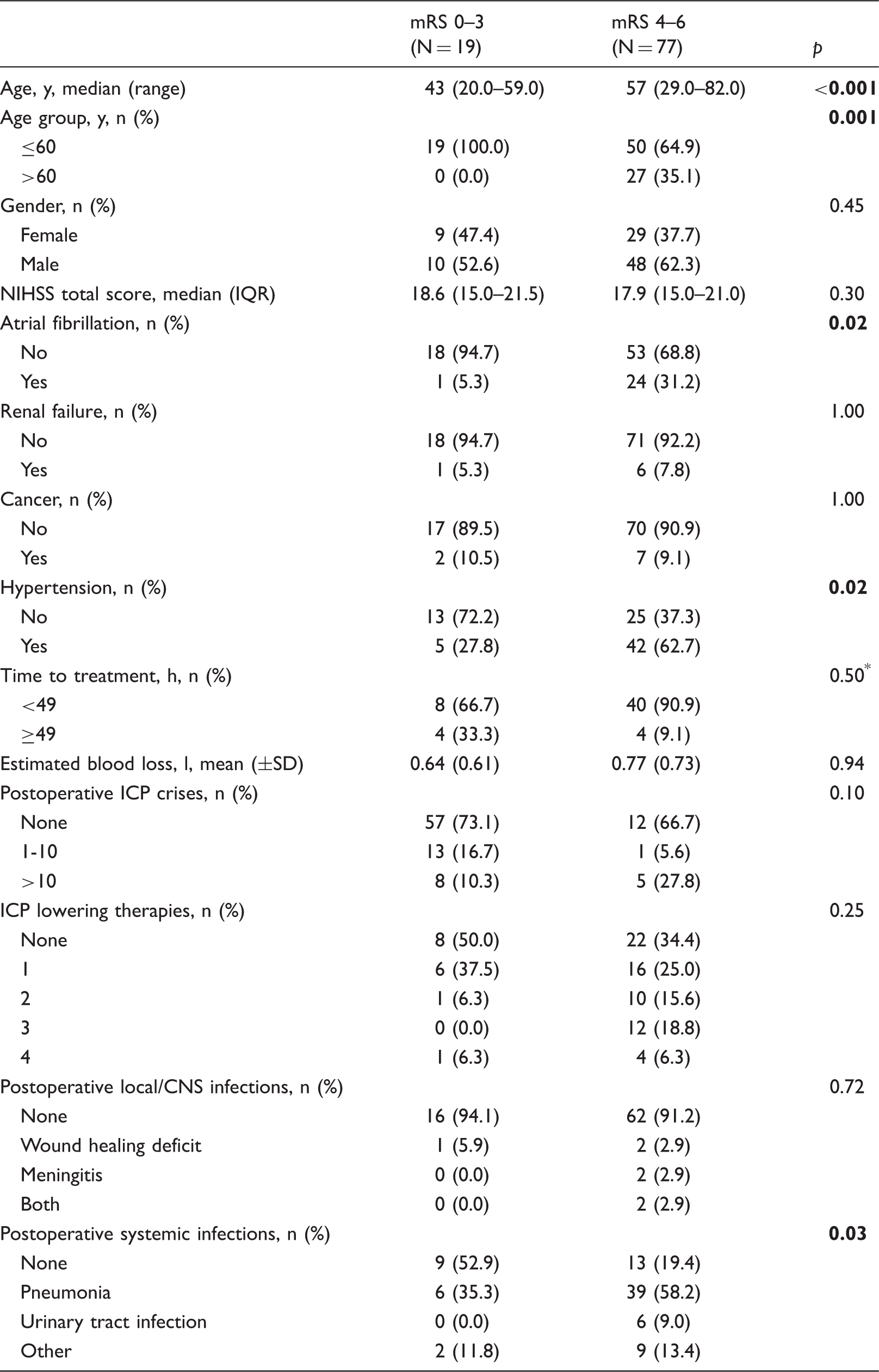
IQR: interquartile range; SD: standard deviation; NIHSS: National Institute of Health Stroke Scale; ICP: intracranial pressure; CNS: central nervous system.
Mann–Whitney-test, all others t-test or Chi-square test as appropriate.
Univariate analysis of neuroimaging characteristics.
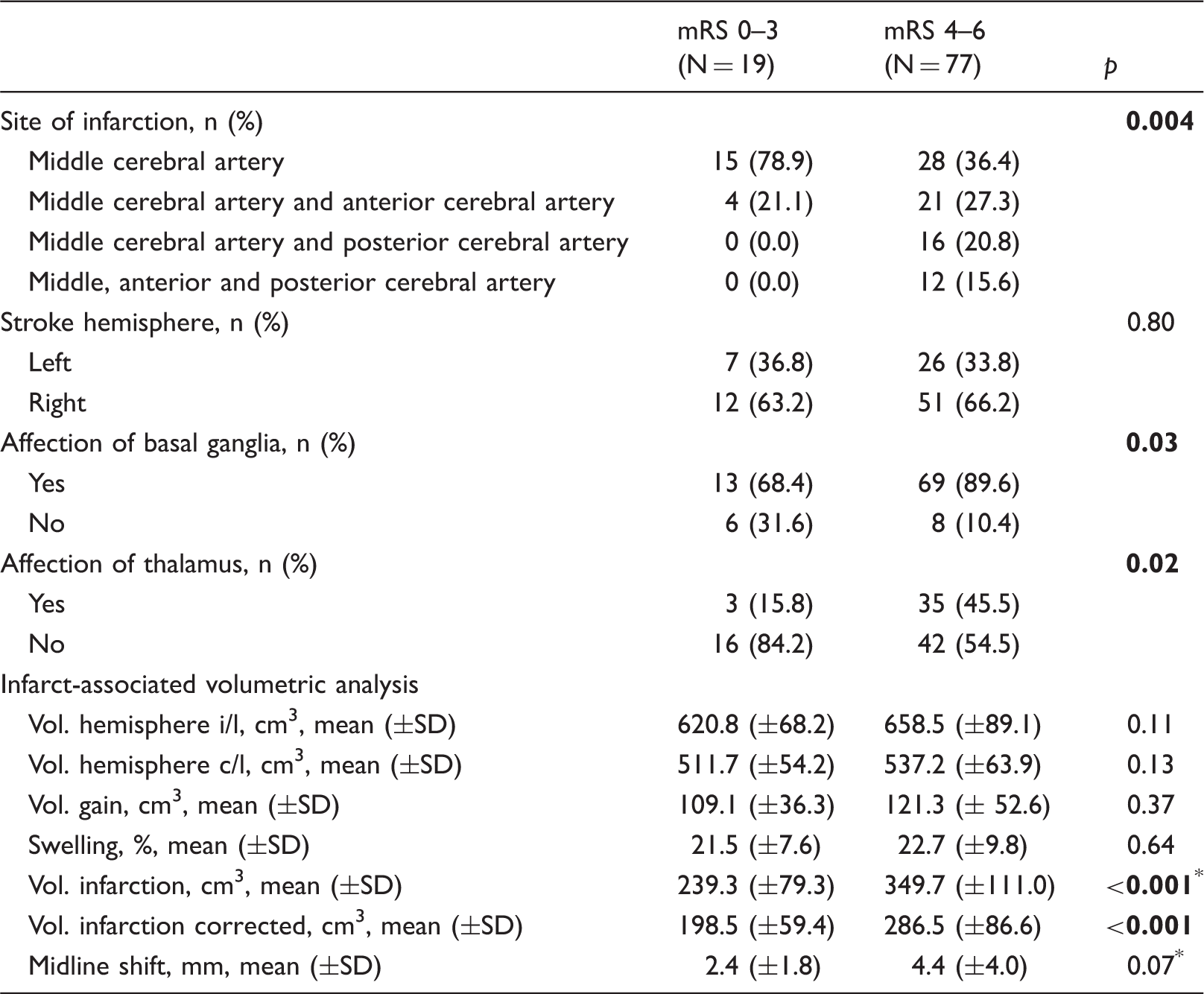
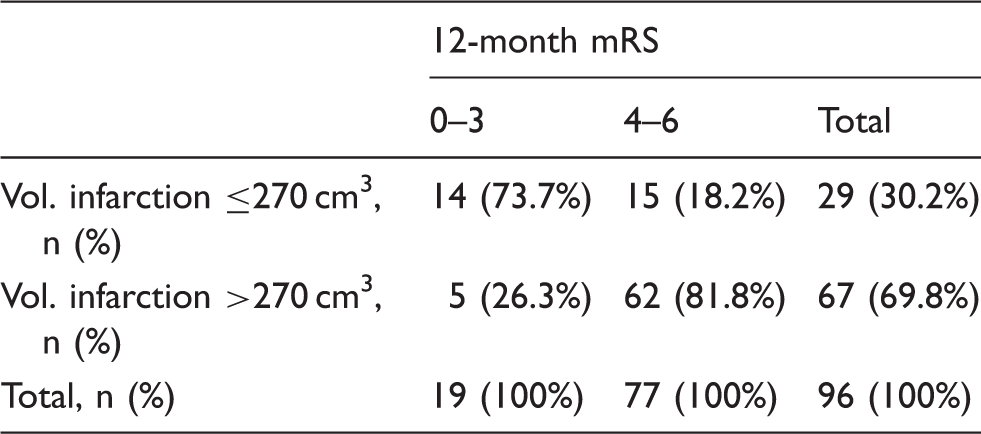
SD: standard deviation; mRS: modified Rankin Scale; Vol.: volume; i: ipsilateral; c: contralateral; Vol. infarction: ischemic brain tissue and edema; Vol. infarction corrected: ischemic brain tissue only; Vol. gain: volume of the ipsilateral hemisphere minus volume of the contralateral hemisphere corresponding to volume of swelling.
Mann–Whitney-test, all others t-test or Chi-square test as appropriate.
Dichotomized outcome analysis
Multivariable logistic regression exploring factors associated with favorable outcome.
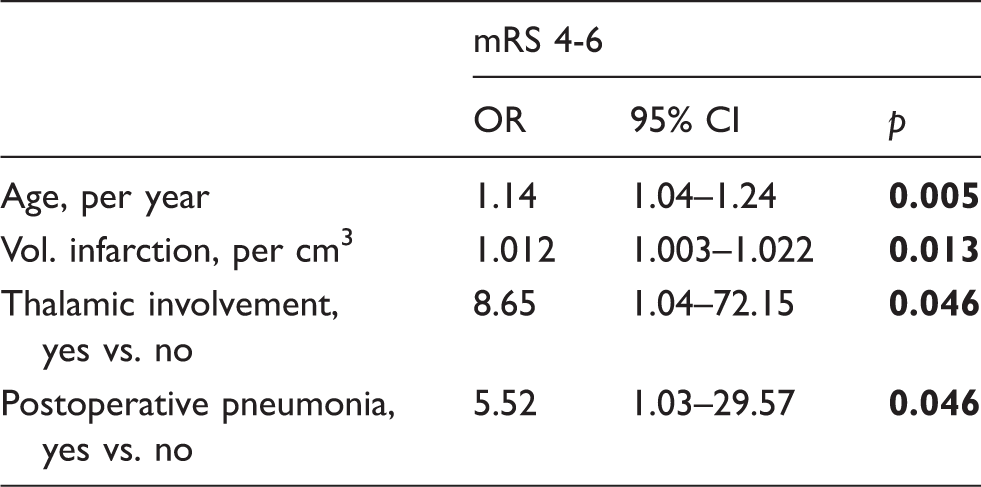
OR: odds ratio; CI: confidence interval; mRS: modified Rankin Scale; Vol.: volume; Vol. infarction: ischemic brain tissue and edema; vs.: versus.
Association of infarct volume and outcome
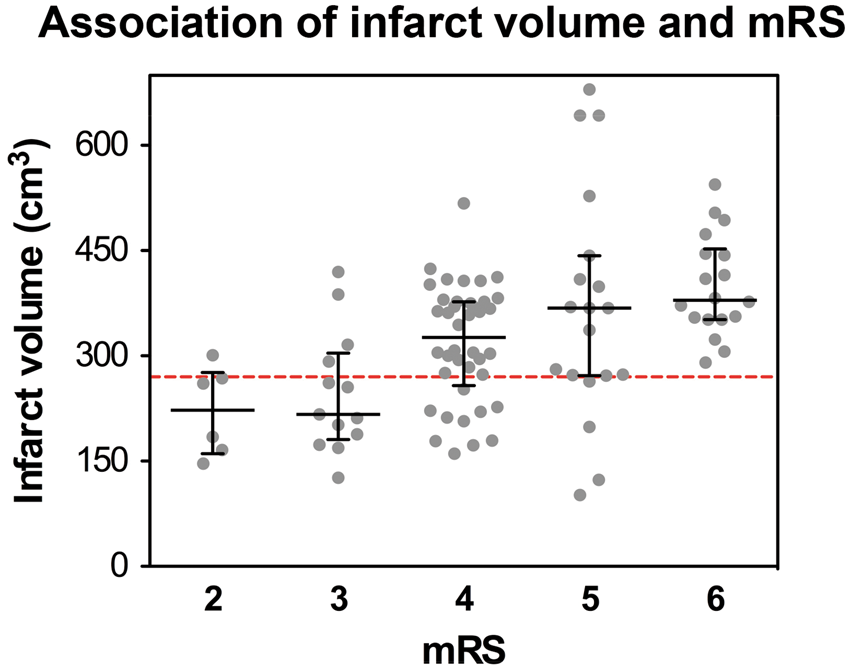
The ROC analysis on infarct volume yielded a good classification result (AUC = 0.80, 95% CI 0.68–0.91; Figure 1) to separate favorable (mRS 0–3) from unfavorable (mRS 4–6) outcome with a sensitivity of 81% and a specificity of 74% at a threshold level of 270 cm3 (Table 4 and Supplemental Tables 2 to 5; http://jcbfm.sagepub.com/content/by/supplemental-data). We then looked at the association of covariates with an mRS shift at 12 months. Here, univariate analysis showed that age (p < 0.001), infarct volume (p < 0.001), involvement of additional vascular territories (p = 0.023), previously diagnosed atrial fibrillation (p = 0.023) and renal insufficiency (p = 0.043) were associated with a higher degree of disability. Multivariable ordinal regression confirmed higher age (OR 1.059 per year, 95% CI 1.019–1.101, p = 0.004) and larger infarct volume (OR 1.005 per cm3, 95% CI 1.001–1.010, p = 0.015) as independent predictors of higher mRS scores at 12 months. The detailed distribution of patients according to their mRS score at 12 months and the infarct volume is illustrated in Figure 2.
ROC analysis of the infarct volume and unfavorable outcome defined as mRS 4–6 yielded a good classification result with an area under the curve (AUC) of 0.80. The Youden-Index at the vertex of the curve identified an infarct volume threshold of 270 cm3 for outcome prediction with the highest sensitivity and specificity in our study population (sensitivity 81.8%, specificity 73.7%; Supplemental Table 2; http://jcbfm.sagepub.com/content/by/supplemental-data). The dashed line represents the identity line. Outcome distribution based on infarct volume threshold. mRS: modified Rankin Scale; Vol.: volume; Vol. infarction: ischemic brain tissue and edema. Illustration of the infarct volume distribution in each patient according to his or her 12-month mRS score. The dashed line represents the threshold infarct volume of 270 cm3, determined at the Youden-Index of the ROC analysis presented in Figure 1.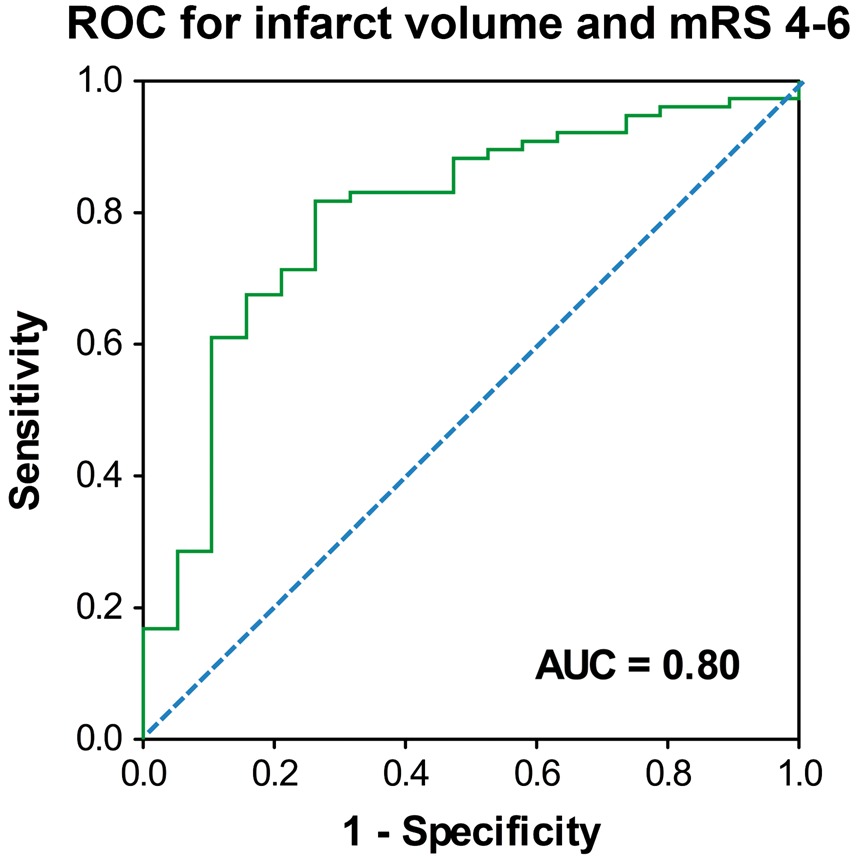
Discussion
With this study, we provide first evidence that the volume of a space-occupying infarct may serve as a clinical variable for estimation of long-term outcome in unselected patients undergoing decompressive surgery. An infarct volume of more than 270 cm3 was associated with unfavorable outcome (mRS 4–6) in more than 80% of all cases with reasonable sensitivity and specificity. While the volume of the infarct should not be the only factor to drive the decision towards DHC, our data support the informed consent discussion by providing probabilities to the most likely outcome.
Outcome after decompressive surgery for treatment of MHS
So far, only few recent reports exist regarding outcome after DHC for treatment of MHS and most have rather focused on early or general mortality and not on long-term neurological disability as a primary outcome measure.7,12–14 Due to the far-reaching consequences of MHS, however, we believe that the decision to perform DHC should not just be based on the patient’s chance to merely survive, but rather on the chance to reach an individually acceptable long-term outcome. 15 Since the purpose of our present study was not to confirm or reject the general indication for surgery like in the RCTs on MHS but rather to retrospectively identify non-modifiable risk factors for a higher degree of long-term neurological disability following DHC, we selected an mRS cut-off between 3 and 4, which according to recent surveys, most patients and physicians would consider acceptable.5,6,10 Although the overall mortality that we noted at 12 months is in agreement with the mortality rates reported in the previous multicenter trials on MHS,3,4 favorable outcome according to our definition was reached less frequently than in DESTINY, DECIMAL and HAMLET.3 Most likely, this is due to the fact that our analysis also included a large number of patients beyond 60 years of age (28%), in whom unfavorable outcome most frequently occurred. The fact that high age was identified as an independent predictor of unfavorable outcome appears in line with this hypothesis. The low number of only six patients (6.3%) suffering death due to transtentorial herniation could reflect our early time point of DHC before the peak development of cerebral swelling1,16 compared to other studies, where DHC was performed up to 72 to 168 hours after MHS onset12,14,17,18 with preoperative signs of herniation already present in 20% to 59% of all patients.14,17 This could explain why mid-line shift as described by others was not an independent predictor of outcome in our cohort, where surgery was generally performed sooner. In fact, the low degree of mid-line shift in both outcome groups even suggests that long-term neurological outcome in patients surviving MHS and DHC may rather depend on factors besides the immediate space-occupying effect of the infarction, such as infections, as one of the main modifiable variables that contribute to poor outcome following acute stroke.19,20 Similarly, pneumonia during the course of hospitalization was also an independent predictor of unfavorable outcome in our analysis.
Association between infarct volume and neurological outcome following DHC
Apart from such modifiable variables, the general severity of an ischemic infarct is classically defined by criteria that relate non-modifiable factors that predict adverse outcome.21–23 In MHS, for example, the initial NIHSS score24,25 and size of the infarction26–28 have been identified as prognostic variables regarding the development of space-occupying edema. Naturally, one would therefore expect that the size of an MHS would also influence long-term outcome in patients undergoing DHC, since the amount of volume created through the bone flap appears to be directly associated with the risk of transtentorial herniation due to the space-occupying effect of edema. 7 However, the link between the volume of an MHS and clinical outcome following DHC has not yet been investigated. Interestingly, we found that only thalamic involvement by itself and not categorical involvement of additional vascular territories was an independent predictor of unfavorable outcome, which is most likely explained by the decreased level of consciousness and marked motor dysfunction of interhemispheric connectivity and dementia reported in thalamic stroke.29–33 Importantly, our finding that only the volume of the infarction but not involvement of additional vascular territories was identified as an independent predictor of long-term outcome supports the idea that the involvement of additional vascular territories does not influence outcome per se but perhaps rather by contributing to a higher overall volume of the infarct. This independent relevance of the infarct volume is backed by the results of our ordinal regression, where a higher degree of disability at 12 months was also significantly associated with a higher volume of the infarct. The usefulness of the infarct volume as a reasonably sensitive and specific clinical variable for outcome prediction in our cohort was also confirmed by the results of our sensitivity analysis shown in Supplemental Table 1, which highlight that the infarct volume is not the only but rather the most consistent variable across different degrees of disability.
Limitations
Naturally, the retrospective, single-center and uncontrolled design of our study bears well-known limitations. First, due to the lack of a uniform definition of MHS, the generalizability of our findings may be somewhat limited because the eligibility for DHC was primarily based on the DESTINY inclusion criteria, which included larger and more severe infarcts compared to DECIMAL and HAMLET in order to increase sensitivity and positive predictive value for life threatening edema and decrease false positive inclusion in the original trial. Second, although our analysis included all consecutive cases undergoing DHC for treatment of MHS that we were able to detect in our hospital records between 2006 and 2012, we cannot completely exclude some degree of selection bias, since the decision to perform DHC was left at the discretion of the patient and/or caregivers together with the treating team of physicians. Third, although outcome at 12 months was per se assessed and documented by an independent physician, patient charts were reviewed by members of the treating physicians’ team. Fourth, there may also be information bias because of missing data as outlined in the tables, which may underestimate potential confounders. Also, the use of postoperative imaging may have introduced some degree of bias, since the volume of the initial infarction may have changed due to early hemodynamic fluctuations or the effect of surgery between the time-point of stroke onset and image acquisition and particularly swelling may be difficult to interpret following DHC, due to the additional space provided by the decompression that may result in larger volumes being recorded. Finally, definite pathophysiological explanations for the relevance of our observations remain speculative, as we did not obtain measurements of cerebral hemodynamics, cerebral microvascular reactivity or cerebral metabolic activity.
Conclusion
Although our study is retrospective and uncontrolled, we believe that our analysis has yielded insights for more detailed prospective studies on outcome prediction for MHS in the future and consideration of the infarct volume of an MHS on neuroimaging may help identify patients, who may benefit more from DHC than others. Despite the predictive value of the infarct volume threshold that we determined, notably, 20% of our patients with infarct volumes beyond this threshold did in fact reach a favorable outcome, which shows that the decision for or against DHC should not be based on a mere threshold value alone but rather on the patients’ individual expectation toward the benefit of surgery, for example with the help of clinical decision algorithms incorporating the patients’ individual goal of care. Most importantly, the infarct volume threshold that we determined does not represent a clear-cut limit, upon which life-saving treatment decisions are to be based but should rather be regarded as reasonable proof that the volume of an MHS may serve as an independent outcome predictor in future prospective studies on DHC.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Deutsche Forschungsgemeinschaft (DFG-WO 1704/1-1), the Bundesministerium für Bildung und Forschung (Center for Stroke Research Berlin, 01 EO 0801) and the Kompetenznetz Schlaganfall.
Declaration of conflicting interests
J. Woitzik and N. Hecht worked as consultants for Integra. P. Vajkoczy has worked as a consultant and served on the scientific advisory board for Aesculap.
Authors’ contributions
JW, EJ and PV conceived, designed and supervised the trial. NH, HN, AP, IF and JW obtained and analyzed the data. NH wrote the first draft of the manuscript. All authors were involved in interpretation of the data and critical revision of the manuscript and approved the final report.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
