Abstract
The metalloprotease meprin β (Mep1b) is capable of cleaving cell-adhesion molecules in different tissues (e.g. skin, kidney and intestine) and is dysregulated in several diseases associated with barrier breakdown (Alzheimer´s disease, kidney disruption, inflammatory bowel disease). In this study, we demonstrate that Mep1b is a novel regulator of tight junction (TJ) composition and blood–brain barrier (BBB) integrity in brain endothelium. In Mep1b-transfected mouse brain endothelial cells (bEnd.3), we observed a reduction of the TJ protein claudin-5, decreased transendothelial electrical resistance (TEER) and an elevated permeability to paracellular diffusion marker [14C]-inulin. Analysis of global Mep1b knock-out (Mep1b−/−) mice showed increased TJ protein expression (claudin-5, occludin, ZO-1) in cerebral microvessels and increased TEER in cultivated primary mouse brain endothelial compared to wild-type (wt) mice. Furthermore, we investigated the IgG levels in cerebrospinal fluid (CSF) and the brain water content as additional permeability markers and detected lower IgG levels and reduced brain water content in Mep1b−/− mice compared to wt mice. Showing opposing features in overexpression and knock-out, we conclude that Mep1b plays a role in regulating brain endothelial TJ-proteins and therefore affecting BBB tightness in vitro and in vivo.
Introduction
The metalloprotease Mep1b is a member of the astacin family of zinc endopeptidases and exhibits a unique structure and specificity among all extracellular proteases. Originally found to be highly expressed in intestine and kidney, recently, a much broader expression pattern has been detected implicating functions in several different biological processes such as inflammation and neurodegeneration. 1
Mep1b is predominantly membrane bound but can also be released from the cell surface by proteolytic shedding. 2 The protease has been implicated in connective tissue formation and detachment of the intestinal mucus layer for proper barrier function, but the physiological roles of Mep1b are in many cases still elusive. 3 As an extracellular protease Mep1b is capable of cleaving extracellular matrix components and cell-adhesion molecules in different tissues such as kidney (E-cadherin and tenascin-C) and skin (E-cadherin and desmogleins).1,2,4,5 Previously, we reported elevated Mep1b RNA levels in human brain compared to most other organs, indicating that Mep1b could also be important for cerebral cell–cell adhesion properties. 6 The ability of cleaving cell-adhesion molecules and the elevated transcription levels in brain, let us hypothesize whether Mep1b could have an effect on TJs in brain endothelium and, therefore, also on the integrity of the BBB.
The BBB regulates the molecular exchange between blood and brain and contributes to the maintenance of central nervous system (CNS) homeostasis, which is essential for normal neuronal function. 7 Protecting the CNS from pathogens and toxic agents, the BBB compromises blood vessel endothelial cells, pericytes and astrocytes, termed the neurovascular unit.8,9 The endothelial cells are linked by TJs that are formed by transmembrane adhesion proteins like claudin-5, occludin and their cytoplasmic-associated protein Zonula occludens 1 and 2 (ZO-1, ZO-2). The TJs regulate the paracellular passage of polar solutes and macromolecules across the barrier and are responsible for the high electrical resistance and the low endothelial permeability by closing the paracellular space of adjacent cells.7,8,10
A breakdown of the BBB includes a structural disruption of the TJs and increased paracellular permeability, which is a common feature of several CNS disorders, including acute and chronic neurodegenerative diseases like Alzheimer’s disease (AD), Parkinson disease (PD), Huntington disease (HD), amyotrophic lateral sclerosis (ALS) or multiple sclerosis (MS).7,8,11 This can lead to an extensive influx of hematogenous fluid into the extravascular space, resulting in an increased brain water content and swelling/edema.7,8,11,12 Additionally, immune cells and mediators that are normally separated by the BBB from the CNS gain access to the brain, causing neuroinflammation.7,8,11,13 For many of these neurodegenerative diseases like AD or MS, BBB breakdown has been associated with an increased activity of matrix metalloproteinases such as MMP-9 in CSF or in pericytes and endothelial cells compromising the BBB.14,15
In this study, we examined the function of the metalloprotease Mep1b for the integrity of the BBB in vitro and in vivo.
Material and methods
Mice
Mature congenic global
Cell culture and antibodies
The following antibodies were used: rabbit anti-claudin-5 and mouse anti-occludin (both from Invitrogen, Darmstadt, Germany), rabbit anti-Zo-1 (Santa Cruz Biotechnology, Dallas, TX, USA), rabbit anti-Mep1b (self-made, polyclonal against a peptide of the extracellular domain), rabbit anti-β-actin and secondary HRP-conjugated antibodies against rabbit (both from Sigma-Aldrich, Schnelldorf, Germany) and mouse (Jackson Immuno Research, Cambridgeshire, UK). For a detailed description of all primary and secondary antibodies, see Supplemental Table 1. All cells were cultured at 37°C, 5% CO2 atmosphere and 95% relative humidity. Mouse brain bEnd.3 endothelial cells (ATCC, Manassas, VA, USA) such as the packaging cell line GP-293 (Clontech, Mountain View, CA, USA) were grown in Dulbecco’s Modified Eagle Medium (DMEM) (Gibco, Darmstadt, Germany) high glucose medium containing 10% fetal calf serum (FCS) and 100 U/ml penicillin/streptomycin (Gibco, Darmstadt, Germany).
Generation of stably overexpressing meprin β bEnd.3 cells
To generate a Mep1b-containing retrovirus, the packaging cells GP-293 were transfected by polyethylenimine (PEI) (Sigma-Aldrich, Schnelldorf, Germany) with a pLBCX
Isolation and cultivation of primary mouse and porcine brain capillary endothelial cells
Primary mouse brain capillary endothelial cells (pMBCECs) were isolated from 8- to 12-week-old global
Cells were cultured in the cellZscope device, in which TEER and capacitance were monitored over time. After four days in culture, puromycin was withdrawn and when cells reached confluency and the capacitance was around 1 μF/cm2, culture medium was removed and serum-free DMEM (Gibco, Darmstadt, Germany) medium containing 1 mM l-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin was supplemented; 550 nM hydrocortisone (Sigma-Aldrich, Schnelldorf, Germany) was added to induce high TEER for 24 h.
Primary porcine brain endothelial cells were isolated as described previously. 18 Cells were plated in Earle’s media 199 (FG0615, Biochrom) supplemented with 0.7 mM L-glutamine, 10 mg/ml penicillin/streptomycin and 10% FCS on 60 mm plates coated with 0.4 mg/mL collagen IV and 0.1 mg/mL fibronectin (both from Sigma-Aldrich, Schnelldorf, Germany). Puromycin was withdrawn after four days in culture. Cells were used for studies when they were confluent.
Treatment of cells with soluble Mep1b
Recombinant Mep1b was generated as described elsewhere. 2 After confluence of bENd.3 or primary porcine brain endothelial cells, recombinant Mep1b was added to serum-free media. As a control, cells incubated with recombinant protein in the presence of actinonin (10 μM) or without Mep1b were analyzed. After 24-h incubation, cells were washed and lysed as described below.
Determination of ventricle size
Eight-to-twelve-week-old mice were transcardially perfused with PBS containing 10 U/ml Heparin and brain was harvested and put in Roti®-Histofix (Carl Roth) containing 4% PFA for 24 h. After that, brains were put in 30% sucrose until they sunk and then embedded in cryomedia and frozen on dry ice; 60 µm coronal sections were cut with a cryostat and stained with Nissl. In similar coronal sections, the size of the lateral ventricles and third ventricle was examined under a microscope (Olympus CKX41).
Transendothelial electrical resistance studies
The integrity of bEnd.3 and pMBCEC cell monolayers was analyzed by the measurement of TEER. Cells (15,000 cells/insert) were seeded in collagen-coated 24-transwell inserts (pore size, 0.4 μm; surface area, 33.6 mm2; Greiner Bio-One, Frickenhausen, Germany) and placed into the cellZscope device (nanoAnalytics, Münster, Germany). TEER and capacitance of cells were measured automatically every hour by impedance spectroscopy. When capacitance values were between 1.0 and 0.8
Cerebral vessel isolation
Cerebral microvessels for tight junction protein analysis were isolated from parenchymal brain tissue of 8- to 12-week-old global
CSF isolation
Samples of CSF of 8- to 12-week-old global
Cell extraction and Western blot analysis
BEnd.3 cells were washed with cold PBS and scraped off from culture dishes. Both bEnd.3 and brain vessel extracts were lysed in NP40 lysis buffer supplemented with complete protease inhibitors (Roche, Mannheim, Germany) for 20 min. Extracts were centrifuged at 18,000
Brain water content
Mice (38 to 42 weeks old) were anesthetized, sacrificed by cervical dislocation, and the brain was immediately removed, weighed and then dried overnight at 100°C. The dried brain was re-weighed and the brain water content calculated as (wet weight- dry weight) × 100/wet weight. The net change in brain water in global
Activity assays
To quantify Mep1b proteolytic activity towards the extracellular loops of occludin, we designed two quenched fluorogenic peptides comprising the corresponding amino acid sequences from murine occludin, a fluorophore (7-methyloxycoumarin-4-yl (mca)) and a quencher (K-ε-2,4-dinitrophenyl [dnp]) (peptide 1: mca-GYGGYTDPRAA-K-ε-dnp; peptide 2: mca- GLYVDQYLYHYSVVDPQE-K-ε-dnp; Genosphere Biotechnologies). For in vitro activity measurements, peptides were used in a final concentration of 20 μM incubated with 20 nM or 50 nM recombinant Mep1b 21 in 100 μl total volume in 96-well plates. In order to quantify Mep1b activity in tissue lysates (150 μg in 1% Triton-X/PBS lysis buffer), a highly specific Mep1b fluorogenic peptide substrate (mca-EDEDED-K-ε-dnp; Genosphere Biotechnologies) in a final concentration of 50 μM was used. Fluorescence intensities were detected at 37°C every 30 s for 120 min using a spectrophotometer (Tecan). For data analysis, slope of equal linear activity range was compared.
Statistical analysis
All graphs and statistical analyses were prepared using GraphPad Prism 8 software (GraphPad, La Jolla, CA, USA). Data were analyzed by unpaired
Results
The expression of TJ protein claudin-5 is decreased in Mep1b-overexpressing endothelial bEnd.3 cells
Since Mep1b is able to cleave cell-adhesion molecules in different tissues (e.g. skin, kidney and intestine)1,2,4,5 is dysregulated in various diseases22–24 and shows elevated transcription levels in the brain compared to most other organs,
6
we investigated whether
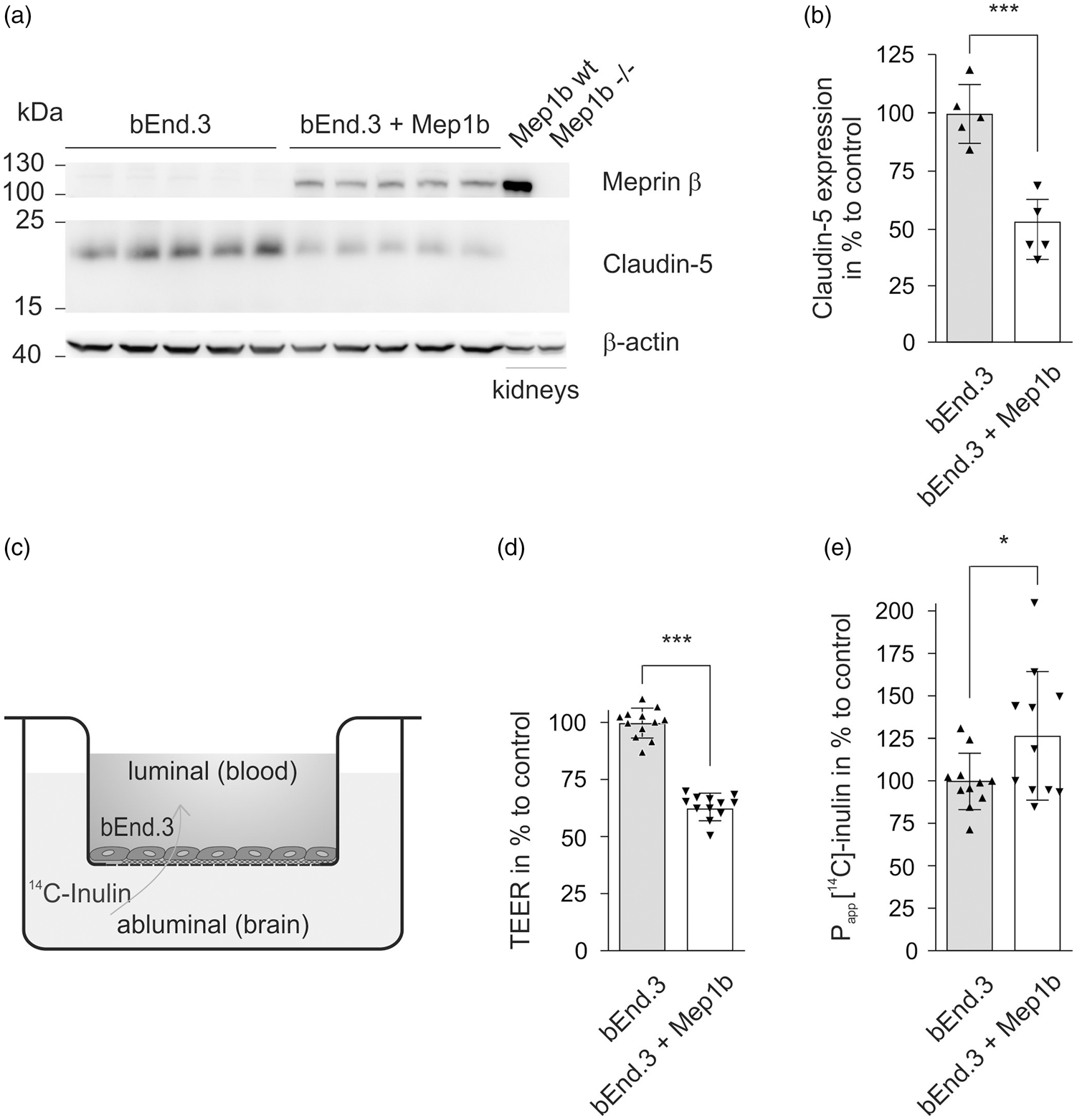
Decreased expression of claudin-5 and impaired barrier integrity in Mep1b-overexpressing bEnd.3 cells. (a) Representative immunostaining of Mep1b and claudin-5 in lysates of control bEnd.3 cells and Mep1b-overexpressing bEnd.3 cells. Anti-β actin is shown as loading control. Kidney lysates of
Overexpression of Mep1b leads to an impaired barrier integrity in bEnd.3 cells
TJs dictate the paracellular permeability of the BBB. 25 As we observed that overexpression of Mep1b results in reduced levels of the TJ protein claudin-5 in vitro, we examined whether these decreased TJ protein levels also affect the barrier integrity. Hence, Mep1b overexpressing bEnd.3 cells and the control mock-transfected bEnd.3 cells were seeded on standard transwell inserts and TEER and capacitance were measured by impedance spectroscopy using a cellZcope device (Figure 1(c)). High TEER values are associated with greater tightness of the endothelium and lower permeability of the barrier, and therefore the TEER value acts as an appropriate indicator for BBB integrity.25,26 When Mep1b-overexpressing and mock transfected bEnd.3 cells showed similar capacitance values, indicating that cells were confluent, we observed a significant reduction of TEER values by approximately 40% in the Mep1b-overexpressing bEnd.3 cells compared to the controls (Figure 1(d)).
To use a second marker for barrier integrity, we analyzed the permeability of radiolabeled [C14]-inulin in Mep1b overexpressing bEnd.3 cells compared to mock-transfected bEnd.3 cells. The small hydrophilic substance [C14]-inulin is a suitable marker for paracellular diffusion, as it is neither a ligand for influx or efflux transporters, endothelial receptors, nor a substrate for endothelial enzymes. 25 The Mep1b overexpressing bEnd.3 cells showed an approximately 30% higher permeability to [C14]-inulin compared to controls (Figure 1(e)). Taken together, these data provide evidence that overexpression of Mep1b in bEnd.3 cells not only results in a decrease of TJ protein claudin-5, but furthermore leads to an impaired barrier integrity in these cells, which is apparent in reduced TEER and elevated permeability to [C14]-inulin.
Increased TJ protein levels in cerebral microvessels of Mep1b−/− mice
To investigate whether the Mep1b-mediated decrease of claudin-5 levels observed in bEnd.3 cells is also of in vivo relevance, we isolated and characterized brain microvessels from global
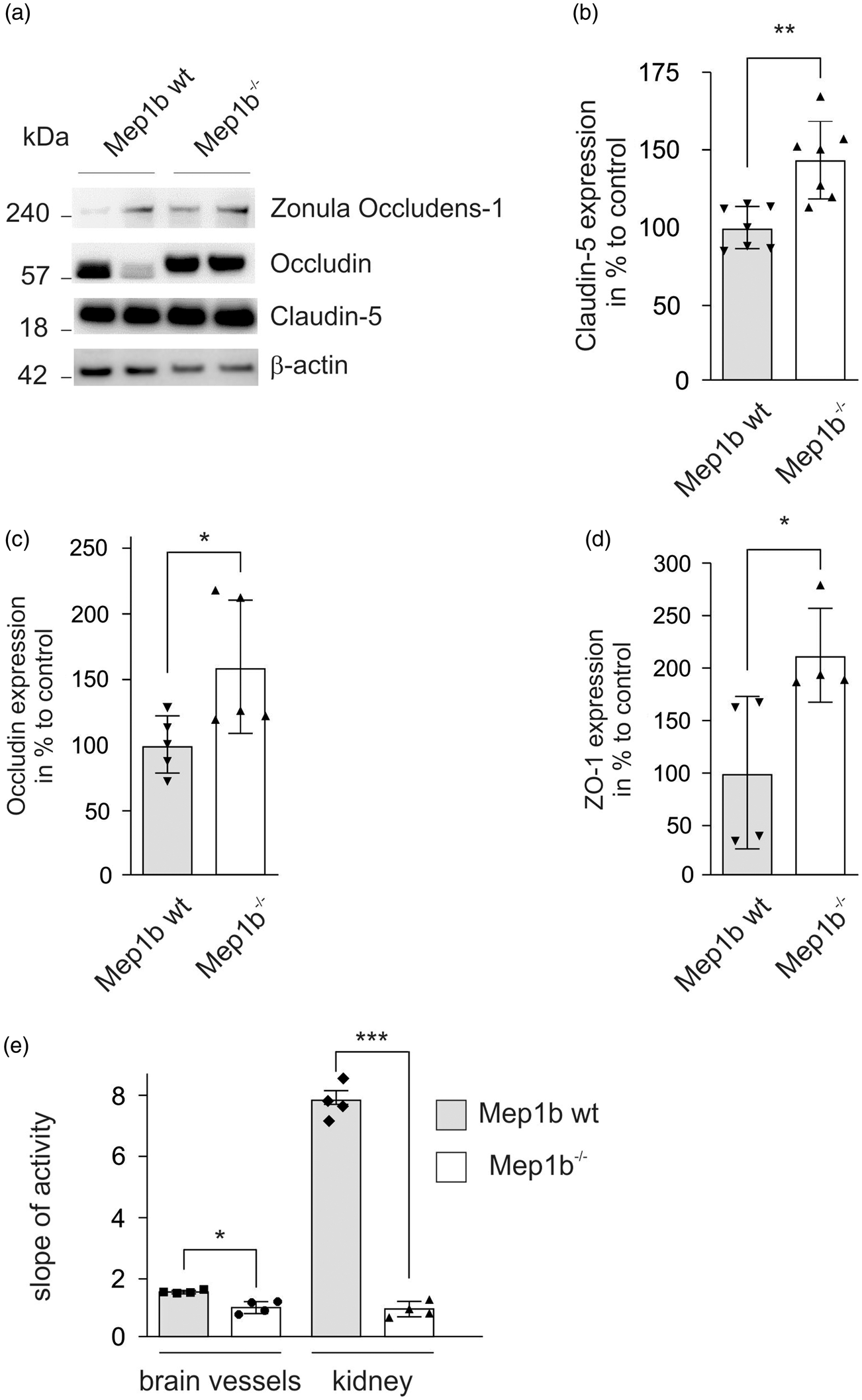
Increased TJ protein levels in cerebral microvessels of
We tried to analyze Mep1b expression in isolated microvessels of global
Of note, in Western Blot analysis the band size detected for occludin was shifted upwards in the
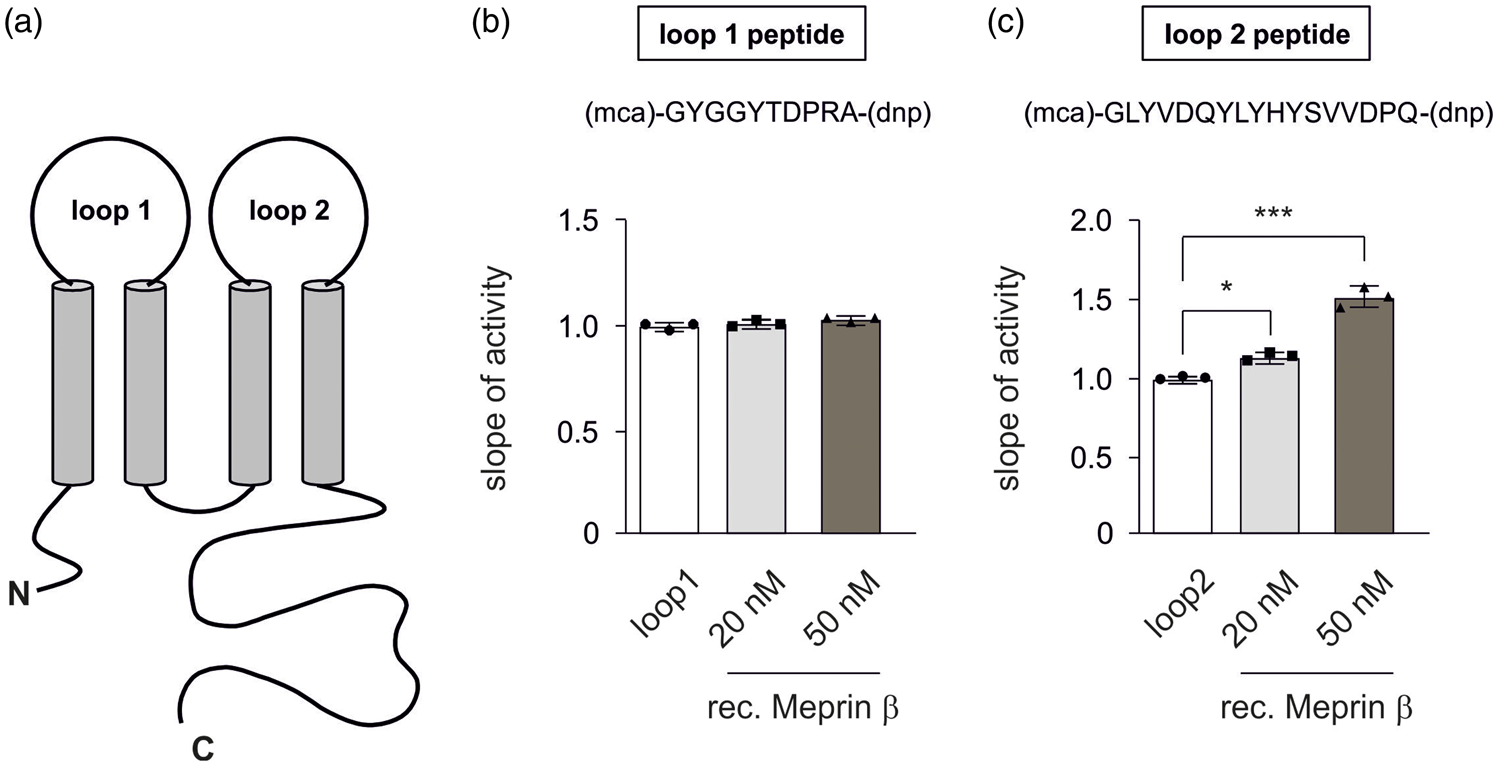
A peptide cleavage assay shows specific cleavage of a peptide corresponding to the second extracellular loop of occludin by proteolytic activity of Mep1b. (a) Schematic representation of occludin. (b, c) Mep1b proteolytic activity assay towards the extracellular loops of occludin. Quenched fluorgenic peptides compromising the amino acid sequence of the first (b) and second (c) extracellular loop of occludin were designed. For in vitro activity measurement, peptides were used in a final concentration of 20 µM incubated with 20 nM or 50 nM recombinant Mep1b in 100 µl total volume in 96-well plates. Fluorescence intensities were detected at 37°C every 30 s for 120 min using a spectrophotometer. Intensities of loop peptides without Mep1b were set as 1.0. For data analysis, slope of equal linear activity range was compared. Data represent dot plots and mean ± standard deviation of three independent experiments. For statistical analyses, one-way ANOVA followed by Tukey’s post hoc test was used. *
Elevated TEER in primary mouse brain endothelial cells from Mep1b−/− mice
In vitro models using monolayers of primary endothelial cells provide significant insight into BBB physiology by allowing the measurement of molecule transport17,18 or barrier integrity.19,28 TEER is an indicator for tightness and integrity of the BBB and is directly dependent on TJ formation. Since, we observed elevated TJ protein levels in cerebral microvessels from global
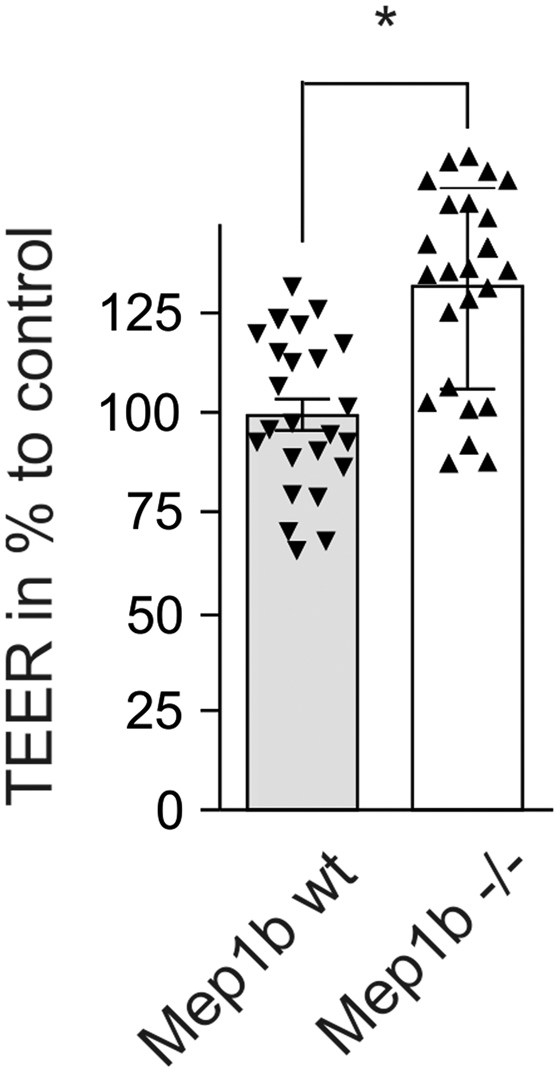
Elevated TEER in primary mouse brain endothelial cells (pMBCECs) from
Soluble Mep1b is potent to regulate claudin-5 in vitro
The effects of the lack of Mep1b on TEER values in primary cultured endothelial cells of
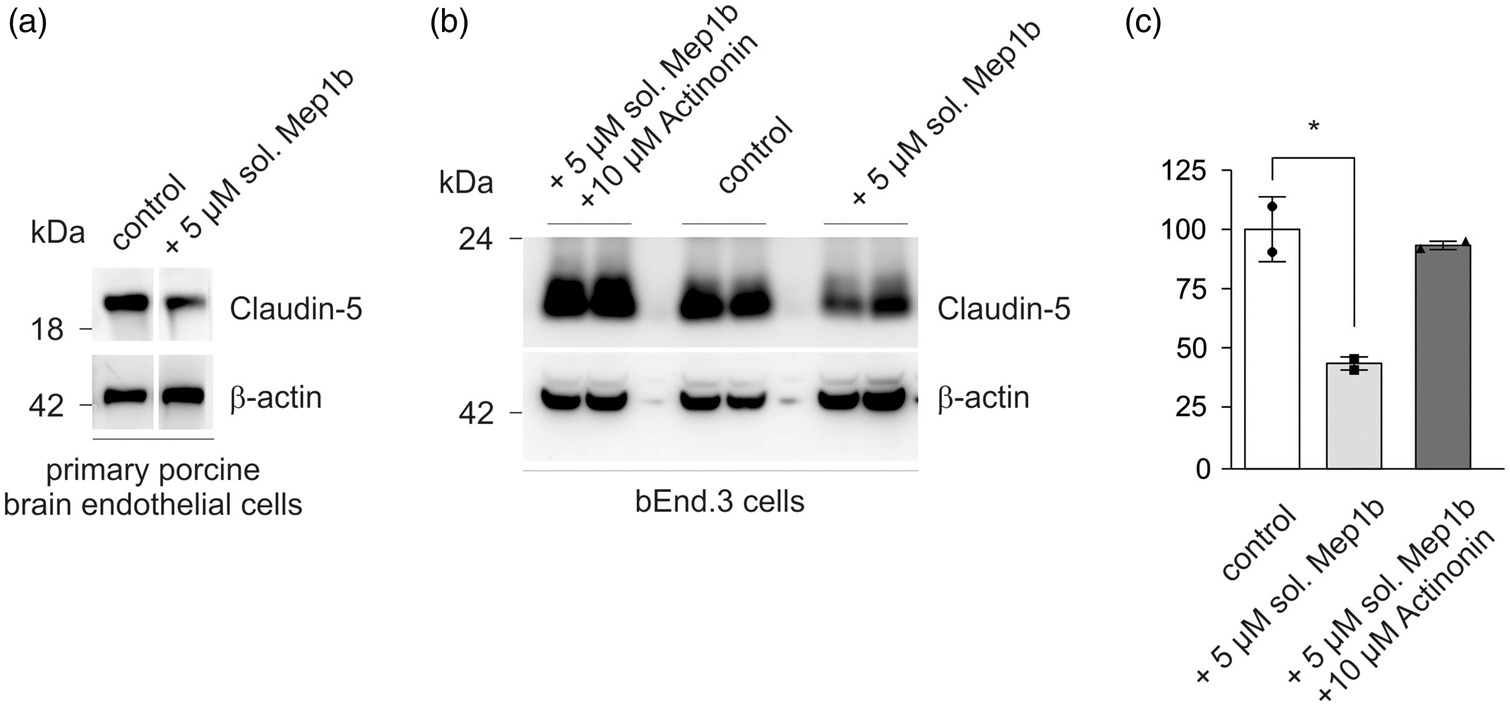
Decrease of claudin-5 expression in soluble Mep1b-treated endothelial cells. (a) Immunostaining of claudin-5 in lysates of 5 µM soluble Mep1b-treated primary porcine brain endothelial cells and control cells. Anti-β actin is shown as loading control. (b) Immunostaining of claudin-5 in lysates of 5 µM soluble Mep1b + 10 µM actinonin-treated bEnd.3 cells, control bEnd.3 cells and 5 µM soluble Mep1b-treated bEnd.3 cells. Anti-β actin is shown as loading control. (c) Claudin-5 levels were quantified by densitometric analysis of Western blots. Intensities of control bEnd.3 cells were set as 100%. Data represent dot plots and mean ± standard deviation of
Similarly, bEnd.3 cells treated with 5 μM recombinant Mep1b showed a significant reduction of full length claudin-5 by approximately 50% compared to untreated bEnd.3 cells. The Mep1b effect on claudin-5 expression could be rescued by simultaneous application of actinonin, a meprin inhibitor (Figure 5(b) and (c)). Therefore, our data suggested that not only endothelial-derived Mep1b but also secreted Mep1b by other cells can influence TJ proteins in brain endothelial cells.
Reduced levels of IgG in CSF of Mep1b−/− mice
As we observed that Mep1b affects both TJ composition and BBB integrity, we wanted to know whether there is also an effect of the Mep1b knock-out on BBB permeability in vivo. Hence, we analyzed the IgG protein levels CSF which we gained by cisterna magna puncture from global
Investigating the CSF of mice lacking Mep1b, we found a significant decrease in IgG levels by approximately 50% compared to wt animals (Figure 6(a) and (b)). These data are in line with the increased TJ protein levels and TEER values, indicating an elevated tightness of BBB in
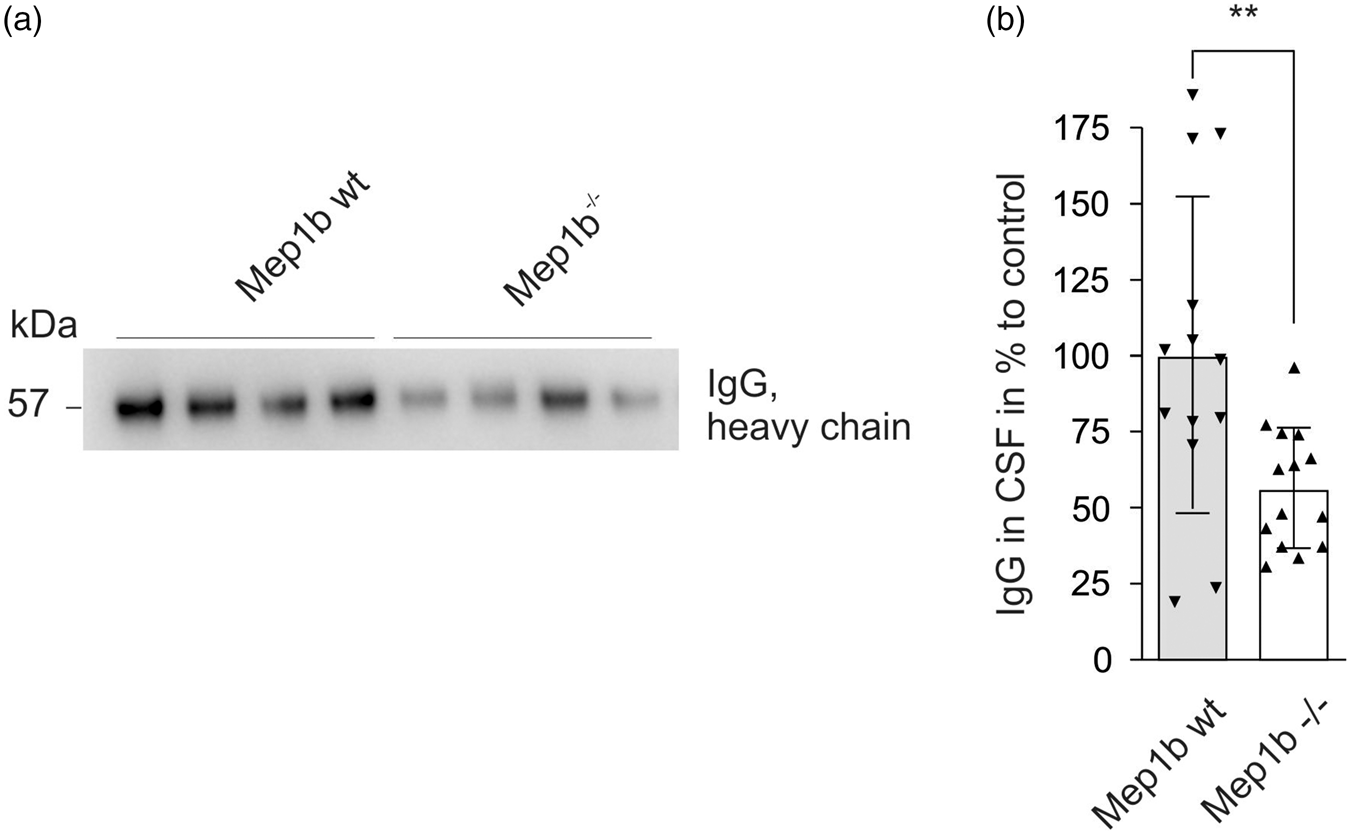
Reduced levels of IgG in CSF of
Brain water content in Mep1b−/− mice is significantly altered
A breakdown of the BBB includes a structural disruption of the TJ and increased paracellular permeability, which is a common feature of several CNS disorders, including acute and chronic neurodegenerative diseases, such as AD, PD, HD, ALS or MS.
8
This can lead to an extensive influx of hematogenous fluid into the extravascular space, resulting in an increased brain water content, which is a marker for brain swelling.
7
Since we observed in
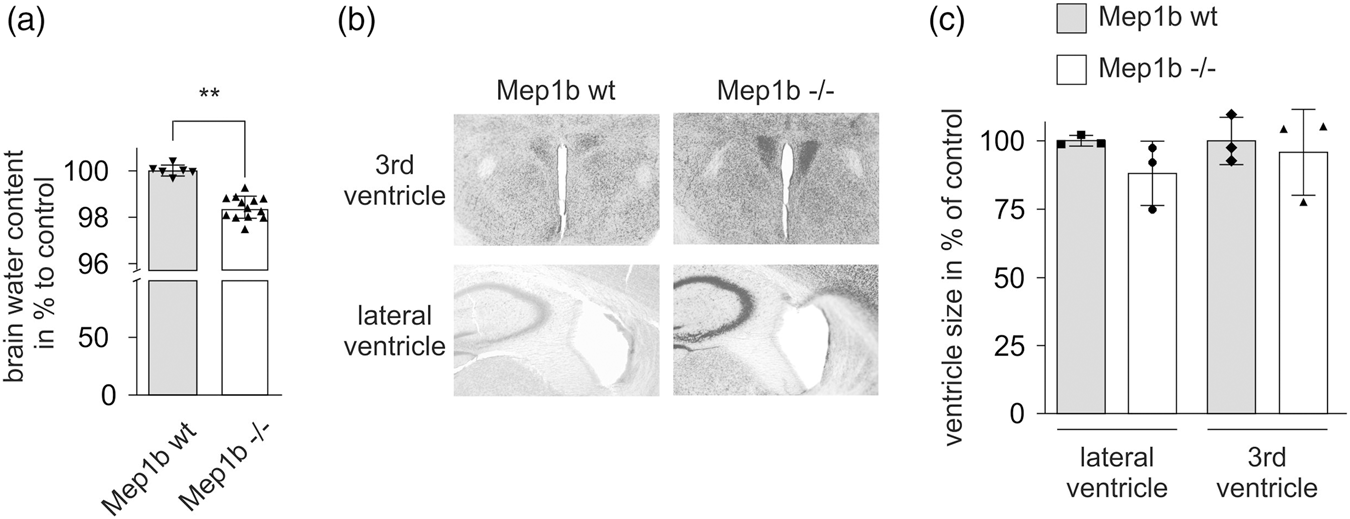
Decreased brain water content in
To investigate whether the reduction in brain water content in
Discussion
Mep1b is a predominantly membrane bound metalloprotease which was originally found to be highly expressed in kidney and intestine. 1 Hence, research focused on those organ specific functions implicating Mep1b in inflammatory bowel disease and acute kidney failure. Over the last years, a much broader expression pattern of Mep1b has been detected.1,22 We reported elevated Mep1b transcription levels in the brain of AD patients, where it may contribute to increased Aβ-peptide generation. 16 It has been shown that Mep1b is capable of cleaving cell-adhesion molecules in different tissues (e.g. skin, kidney, intestine),1,2,4,5 which is in line with dysregulation of Mep1b in diseases associated with barrier breakdown (AD, kidney disruption, inflammatory bowel disease).20–22 Therefore, we investigated the potential influence of Mep1b on TJ function and BBB integrity in brain endothelium in vitro and in vivo.
In the present study, we demonstrate that Mep1b overexpression in mouse brain endothelial cells (bEnd.3) leads to decreased claudin-5 expression levels, which correlated with decreased TEER values and an elevated permeability to [C14]-inulin in an in vitro transwell system, indicating an impairment of the BBB in vitro. Additionally, we observed elevated TJ proteins, increased TEER values, decreased IgG levels in the CSF and a reduced brain water content in mice globally lacking Mep1b, suggesting that a knock-out of Mep1b results in a tighter BBB with a greater integrity. Since Mep1b overexpressing bend.3 cells and
To determine whether Mep1b expression influences TJ function in vitro, we generated stable Mep1b overexpressing endothelial cells (bEnd.3). We observed a 50% reduction in claudin-5 protein expression in the Mep1b overexpressing bEnd.3 cells, indicating a detrimental effect of Mep1b on TJ protein level in vitro accompanied with lower TEER and higher permeability of paracellular tracers. Similar observations have been reported by Chen et al. 34 when they transfected bEnd.3 cells with matrix metalloproteinase-9 (MMP-9), which resulted in decreased TJ protein levels (claudin-5, occludin, ZO-1, ZO-2) and impairment of BBB permeability. MMP-9 is a member of the family of matrix metalloproteinases that has been shown to degrade occludin and claudin-5 in focal cerebral ischemia resulting in BBB opening.35,36 In mice where murine ApoE was replaced with the human isoform of APOE4, a major genetic risk factor for AD, it has been shown that BBB leakage is caused by an upregulation of MMP-9 due to a lack of ApoE4 signaling. Moreover, these effects could be reversed by MMP inhibitor treatment or MMP-9 short interfering (si)RNA administration into the hippocampus of these mice. 29 Whereas MMP function in general and its role in BBB breakdown during stroke (MMP-9, MMP-2) or AD have been intensively studied, the physiological functions of the metalloprotease Mep1b are still in many cases elusive. 1
Since we observed similar effects as reported for other MMPs 36 by Mep1b overexpression on TJ composition in bEnd.3 cells, we suggest that Mep1b may have a comparable influence on BBB integrity. In contrast to MMP-9, which was demonstrated to degrade occludin and claudin-5, 35 we have not deciphered the exact mechanism of action yet how Mep1b influences the TJ expression. One way of action could be a direct mechanism, whereby the Mep1b sheds junctional proteins, therefore compromising BBB integrity. This is supported by Western blot analysis and peptide cleavage assays. Another possibility is that Mep1b acts via an indirect mechanism by activation of other proteases or signaling cascades that in turn influences junctional protein expression including claudin-5.
Interestingly, in brain microvessels, Mep1b expression not only showed effects on TJ proteins sealing the paracellular space (claudin-5, occludin) but also on the intracellular protein ZO-1. As both membrane-bound and soluble Mep1b does not have access to intracellular ZO-1, it might be plausible that Mep1b cleavage of claudin-5 or occludin leads to the internalization of the whole TJ complex. However, further studies will be necessary to understand how Mep1b affects intracellular proteins like ZO-1 or whether other intracellular proteins of the TJ complex are affected as well.
Moreover, it remains to be seen whether the observed in vivo effects of Mep1b on BBB integrity are due to endothelial Mep1b or secreted Mep1b released by other cells. It has been shown that pericytes14,29,30 or the composition of the basement membrane
31
influence BBB integrity. However, also circulating cells in the blood or soluble Mep1b in the body fluids could alter BBB permeability.
37
The effects that we observed on TEER in endothelial cells cultured in the presence of puromycin, which kills non-endothelial cells due to their lack of ABC transporters, suggest that brain endothelial-derived Mep1b is potent to influence TJ protein levels.
38
However, we were not able to detect Mep1b in brain microvessels via Western blot analysis with several different antibodies. In our hands, these antibodies easily detect Mep1b in kidney lysates but so far we have been unable to see specific signals in brain microvessels or whole brain lysates. In the brain, we have reported Mep1b transcription by RT PCR analysis.
6
Another plausible effect for the lack of Mep1b protein detection is its low expression in brain microvessels. Single cell analysis of vascular cells in the brain revealed that only very few of the total amount of analysed endothelial cells expressed Mep1b.39,40 The expression of Mep1b was restricted to specific subtypes of endothelial cells (termed as endothelial cells type 2 or arterial endothelial cells) suggesting a regional heterogeneity of Mep1b in the vasculature of the brain. This is in line with previously published bulk RNA data from another group that showed general low Mep1b expression in brain vasculature and also with the Mep1b activity data that we gathered from isolated whole brain vessels showing a slight but significant elevation of Mep1b activity in brain vessels from Mep1b wt mice compared to
Nevertheless, we also provide evidence that soluble Mep1b in the various body fluids could alter BBB integrity in vivo. Both, Mep1b-treated bEnd.3 cells and primary porcine endothelial cells showed reduced claudin-5 levels compared to untreated control cells. In bEnd.3 cells, the effect of Mep1b on TJ proteins effect could also be rescued by the addition of the Mep1b inhibitor actinonin. These results suggest that soluble Mep1b released by non-endothelial cells in the periphery or brain, present in blood and interstitual fluid or CSF, are potent to influence TJ protein levels in the brain. These findings go along with a report showing that the deletion of the Mep1 gene in leukocytes diminishes the ability of them to disseminate through the extracellular matrix indicating that Mep1b expressed by non-endothelial cells influences barrier-dependent processes like transendothelial migration. 37 Given that there is evidence that Mep1b effects on TJ and BBB integrity can both be attributed to endothelial Mep1b and Mep1b released by other cells, we believe that during disease both sources could contribute to BBB impairment.
Claudin-5 and occludin are two of the major TJ proteins in brain microvessels and are dysregulated in many neurological disorders such as multiple sclerosis, stroke, Alzheimer’s disease, small vessel disease, during CNS invasion of cancers and during infection.42,43 Mice lacking occludin appear to have morphologically unaltered TJs but show a complex phenotype with postnatal growth retardation and brain calcification. 44 In contrast, claudin-5 deficient mice die within 10 h after birth and show a massive BBB impairment along with leakage of high molecular molecules of up to 800 Dalton. 45 Interestingly, double knockdown of claudin-5 and occludin even enhanced the permeation of tracer molecules up to a size of 3000 to 10,000 Da. 46 In our study, both claudin-5 and occludin were affected by Mep1b expression along with higher IgG levels in the CSF suggesting that the loss of TJ proteins enhances brain penetration of IgG from blood.
In vivo BBB integrity was further investigated by analysis of brain water content in
Taken together, Mep1b overexpression in bEnd.3 cells in vitro and
Supplemental Material
JCB905206 Supplemental Material - Supplemental material for Meprin β: A novel regulator of blood–brain barrier integrity
Supplemental material, JCB905206 Supplemental Material for Meprin β: A novel regulator of blood–brain barrier integrity by Markus Gindorf, Steffen E Storck, Anke Ohler, Franka Scharfenberg, Christoph Becker-Pauly and Claus U Pietrzik in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Deutsche Forschungsgemeinschaft (DFG) Grant SFB877, “Proteolysis as a Regulatory Event in Pathophysiology” (A9, A15). MG would like to thank the Integrated Research Training Group (IRTG) of SFB877 for financial support. This work comprises substantial parts of the dissertation of Markus Gindorf.
Acknowledgements
The authors thank Johanna Wesselowski for excellent technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
MG and SES, as the first authors, designed the studies, conducted the experiments, and wrote the manuscript. AO contributed to the experimental design of the study and performed experiments. FS conducted the activity assays. FS, CB and CUP helped write the manuscript. CUP supervised the experimental design and entire work of the manuscript. All authors read and approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
