Abstract
The Lutheran (LU) glycoprotein was shown to be a specific marker of brain capillary endothelium, which forms the blood-brain barrier (BBB) in vivo. A 1.5 kb partial cDNA encoding the bovine LU was isolated from a bovine brain capillary cDNA library. Sequence analysis showed that the bovine and human LU had a 75% and 79% identity in the amino acid and nucleotide sequences, respectively. Northern blot analysis demonstrated a very high level of gene expression of the LU transcript in freshly isolated bovine brain capillaries, but no measurable LU mRNA in whole bovine brain. The high level of LU gene expression was maintained when bovine brain capillary endothelium was grown in tissue culture. Because many BBB specific genes are downregulated in tissue culture and in brain tumors, the expression of the LU mRNA and immunoactive LU protein was investigated in primary and metastatic human brain tumors. Immunocytochemistry of fresh frozen human brain and human brain tumors showed abundant immunostaining of brain capillary endothelium. Northern blot analysis showed the presence of LU transcripts in a panel of primary and metastatic human brain tumors. These studies demonstrated that the LU glycoprotein was a novel new marker of the BBB, and unlike other BBB specific genes, there was a persistent gene expression of the LU glycoprotein both in brain capillary endothelial cells grown in culture and in the endothelium of capillaries perfusing human brain cancer.
The Lutheran (LU) glycoprotein is an 85 kDa glycoprotein that was originally detected in human erythrocytes and subsequently shown to be a basolateral antigen in diverse epithelial cells (Parsons et al., 1995). The LU glycoprotein is related to the human basal cell adhesion molecule (B-CAM), which is a cell surface glycoprotein of epithelial cancers (Campbell et al., 1994). The LU glycoprotein and the B-CAM protein arise from mRNA molecules that are spliceoforms of the same transcript (Rahuel et al., 1996). The B-CAM protein has a truncated carboxyl terminus and is translated from a 4.0 kb transcript, whereas the LU mRNA is 2.5 kb. Northern tissue surveys have demonstrated the presence of the 2.5 kb LU transcript, but not 4.0 kb B-CAM mRNA, in human brain (Rahuel et al., 1996).
The cellular origin of the LU glycoprotein in brain is not known. Apart from the choroid plexus epithelium, the other cell in brain that has epithelial-like characteristics is the capillary endothelium, which forms the blood-brain barrier (BBB) in vivo. Like epithelial barriers in peripheral tissues, the capillary endothelia in vertebrate brain are fused together by epithelial-like, high resistance, tight junctions to form the BBB in vivo (Pardridge and Boado, 1993). The current studies report that a partial cDNA of the bovine homologue of the LU glycoprotein was, in fact, identified in screening a bovine brain capillary cDNA library. The current studies described the cloning and sequencing of this partial cDNA and the high degree of homology to the human LU glycoprotein. Bovine LU cDNA was used in Northern blotting to demonstrate the selective expression of the LU gene in brain at the capillary endothelium. Unlike many BBB specific genes, which are downregulated in tissue culture (Black et al., 1990), the high LU gene expression at the BBB persisted in cultured brain capillary endothelium. Because many BBB specific genes are also downregulated in capillaries of brain tumors (Black et al., 1990; Guerin et al., 1990, 1992), the current studies examined the expression of the LU mRNA and protein in human brain tumors. Lutheran gene expression was identified in primary brain tumors, and immunocytochemistry showed that the LU protein was selectively expressed at the tumor capillary endothelium. These results indicated that the LU glycoprotein was a novel marker of brain capillary endothelia, which form the BBB in vivo, and that the high LU gene expression at the BBB persists in cell culture and in primary brain tumors.
MATERIALS AND METHODS
32P-dCTP (3000 Ci/mmol), 35S-dATP (1000 Ci/mmol) and GenScreen Plus membranes were purchased from NEN Life Science Products (Boston, MA, U.S.A.). Megaprimer DNA labeling kit and Sequenase 2.0 DNA sequencing kit were obtained from Amersham Life Science (Cleveland, OH, U.S.A.). Restriction endonucleases were obtained from Promega Life Science (Madison, WI, U.S.A.). Oligodeoxynucleotides were custom synthesized at Biosource International (Camarillo, CA, U.S.A.). The BK-actin plasmid was prepared as previously described in this laboratory (Boado and Pardridge, 1993). Total and size-fractionated bovine brain microvessels cDNA libraries were prepared in λgt11 and pSPORT vectors, respectively, and characterized as previously reported in this laboratory (Boado and Pardridge, 1990; Boado et al., 1999). Human primary and metastatic brain tumors were obtained fresh frozen from the UCLA Department of Surgery/Neurosurgery and Department of Pathology/Neuropathology as described previously (Boado et al., 1992). C6 rat glioma cells were obtained from the American Type Culture Collection (Rockville, MD, U.S.A.). BcI-10 cells, or ECL cells, are a line of bovine brain capillary endothelial cells described previously (Boado and Pardridge, 1993).
Isolation of bovine LU cDNA
A partial cDNA encoding the bovine LU glycoprotein was identified in the process of isolating cDNA for the bovine large neutral amino acid transporter type 1 (LAT1), as described recently (Boado et al., 1999). The bovine LAT and the LU cDNA were isolated from a bovine brain capillary λgt11 cDNA library, which has been characterized in previous studies (Boado and Pardridge, 1990). Approximately 2.2 × 105 recombinants of this library were screened with the 32P- clone LAT1-6, which was a pCR2.1 plasmid encoding an approximate 0.2 kb fragment produced by the polymerase chain reaction and corresponding to a fragment of the rat LAT1. This screening produced a clone that released a 2.0 kb cDNA, which was subcloned in the Bluescript plasmid and named LAT IV-5 (Boado et al., 1999). DNA sequencing analysis of this clone revealed that it was composed of the bovine BBB LAT1 cDNA (accession # AF174615, nt 838-2244) artifactually fused to a partial cDNA that was 86% similar to nt 1520–1797 of the human LU (accession # X83425). Restriction endonuclease analysis revealed that a single BstXI site separated the LU and LAT1 cDNA in addition to the BstXI site in the multiple cloning region of the plasmid. Therefore, the 200 bp LU fragment was released by BstXI and the ~200 bp BstXI LU 32P-cDNA was used for screening of a size-fractionated bovine brain microvessel cDNA library in the pSPORT vector, which has been characterized previously (Boado et al., 1999). After screening of 2.4 × 105 recombinants, 37 clones were positive for bovine LU, and 4 of them were isolated for further characterization. cDNA inserts were released by double digestion with NotI and SalI and characterized by ethidium bromide staining of samples separated by agarose gel electrophoresis and by Southern blot analysis, which was performed under high stringency conditions with 32P-labeled bovine LU cDNA released by BstXI digestion of clone LAT IV-5 (Boado et al., 1999). The bovine LU clone with the highest size, approximately 1.5 kb, was selected for DNA sequencing.
DNA sequencing
DNA sequencing of isolated clones was performed in both directions at the Keck Biotech Resource Laboratory (DNA Sequencing Core Facility, Yale University, CT, U.S.A.) and at the Biotech Core (Palo Alto, CA, U.S.A.), and confirmed by manual DNA sequencing as described previously (Boado et al., 1999). Similarities with related cDNA deposited in GenBank were investigated using the BLAST program (NCBI, NIH), and the phylogenetic analysis of the LAT sequence was performed with the LALIGN program available through the FASTA package at the University of Virginia (Pearson and Lipman, 1988).
Isolation of poly A+ RNA and Northern blot analysis
Poly A+ RNA was isolated from bovine brain capillaries, rat organs, cultured bovine brain capillary endothelium, C6 rat glioma cells, and primary and metastatic human brain tumors using a single step method previously described (Boado and Pardridge, 1991); 2 to 10 μg of poly A+ RNA were resolved in 1.5% agarose:2.2 mol/L formaldehyde gels, and blotted onto GeneScreen Plus membranes. Northern blots were hybridized with [32P]-labeled LU or β-actin cDNA probes as described previously (Boado et al., 1999). cDNA were labeled with [32P]-dCTP by the random priming technique using the Megaprimer DNA labeling kit (Boado and Pardridge, 1991). Blots were quantified by optical scanning densitometry of autoradiograms using the National Institutes of Health Image 1.54 program on a Power Macintosh 7100/66 AV microcomputer (Boado et al., 1999).
Immunocytochemistry
Immunocytochemical detection of LU was performed in 10-μm frozen sections of normal human brain and human glioblastoma mutiforme using the avidin-peroxidase method of immunocytochemistry (Hsu et al., 1981). Sections were fixed with 100% methanol (20 minutes, −20°C), washed with ice-cold PBS, and air-dried. Samples were incubated with anti-LU hybridoma conditioned medium or mouse IgG control (1μg/mL), and the immune reaction was developed using biotinylated horse anti-mouse IgG (Vector Labs, Burlingame, CA, U.S.A.) and the ABC Elite kit (Vector Labs) (Hsu et al., 1981). Slides were counterstained with Mayer's hematoxylin. The antihuman LU hybridoma (LM342/767) conditioned media was kindly provided by Dr. Robin Fraser of the Glasgow and West Scotland Blood Transfusion Service (Lanarkshire, Scotland); this antibody recognizes the first immunoglobulin superfamily domain of the LU glycoprotein (Zen et al., 1999).
RESULTS
The nucleotide and predicted amino acid sequence of the partial bovine LU cDNA are shown in Fig. 1. The 1540 nucleotide cDNA encodes the entire 3′-untranslated region including a 36-mer poly A tail preceded by an ATAAA polyadenylation consensus site. The partial cDNA begins at Gln243 which corresponds to the Pro243 in the human LU glycoprotein (Parsons et al., 1995). There was a single 20 amino acid transmembrane region near the carboxyl terminus of the LU glycoprotein, and this region was highly conserved between human and bovine with only two conservative substitutions. There was a 75% identity in the amino acid sequence of the bovine and human LU protein. There was a 79% identity between the corresponding human and bovine LU nucleotide sequence and an 85% nucleotide identity over the 378 nucleotides comprising the 3′-untranslated region.

Nucleotide and predicted amino acid sequence for the bovine LU partial cDNA. The amino acid sequence begins with Gln243 which corresponds to Pro243 of the human LU protein (Parsons et al., 1995). The bovine and human LU glycoprotein are predicted to terminate at Cys597. The 20 amino acid single transmembrane domain is underlined. Within the bovine LU sequence beginning with Gln243, there are 10 cysteine residues, which are 100% conserved in the bovine and human sequences and are also underlined. The Pro557 residue, which corresponds to the carboxyl terminus of the B-CAM isoform, is underlined. Three different consensus N-linked glycosylation sites are in bold italics. The 378 nucleotide 3′-untranslated region includes a 36-mer poly-A tail proceeded by an ATAAA polyadenylation consensus sequence which is underlined.
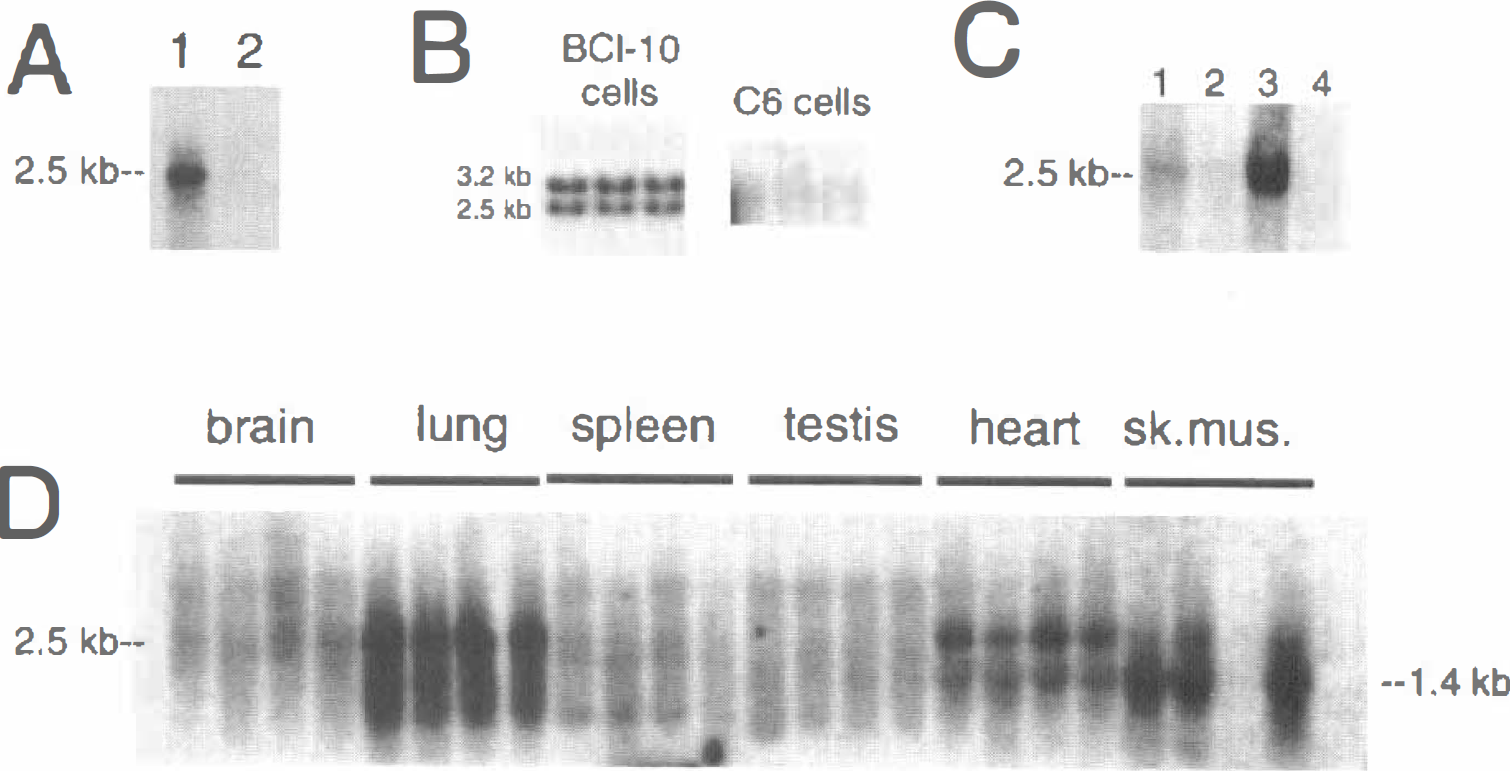
Northern blot analysis with the bovine LU cDNA showed that the LU mRNA was highly abundant in freshly isolated bovine brain capillaries, as the 2.5 kb bovine BBB LU mRNA was detected after a 16-hour exposure of the x-ray film (Fig. 2A, lane 1). In contrast, no LU transcript was detected in total bovine brain after this exposure (Fig. 2A, lane 2). High levels of gene expression of the LU transcript in bovine brain capillary endothelial cells persisted in cell culture as shown by the Northern blot in Fig. 2B, which was exposed at room temperature for only 4 hours. In contrast to the BBB in vivo (Fig. 2A), the cultured bovine brain capillary endothelial cells produced 2 LU transcripts of 3.2 and 2.5 kb (Fig. 2B). The LU gene expression in C6 rat glioma cells was negligible because no measurable LU transcript was detected after a 9-day exposure of the Northern blot at −70°C (Fig. 2B). The bovine LU cDNA cross-hybridized with rat LU transcript as Northern blotting of 2 μg per lane of poly A+ RNA from rat organs demonstrated expression of the LU gene in rat lung (Fig. 2C). When larger amounts of the rat organ RNA were analyzed (10 μg poly A+ RNA per lane), it was possible to visualize the rat LU transcript in rat heart and skeletal muscle, in addition to rat lung. No LU transcript was detected in rat brain, spleen, or testes (Fig. 2D). A smaller sized transcript, 1.4 kb, was found in lung, heart, and skeletal muscle (Fig. 2D).
The Northern blotting results indicated that the LU glycoprotein gene expression in brain was restricted to the BBB. This was confirmed by immunocytochemistry using a monoclonal antibody to the human LU glycoprotein. The studies with control human brain are shown in Fig. 3. No detectable immunoreactive LU glycoprotein was observed in brain parenchyma. In contrast, abundant immunoreactive LU glycoprotein was detected at the brain microvasculature and the continuous immune staining of the microvessels indicated an endothelial origin of the immunoreactive LU glycoprotein in human brain.
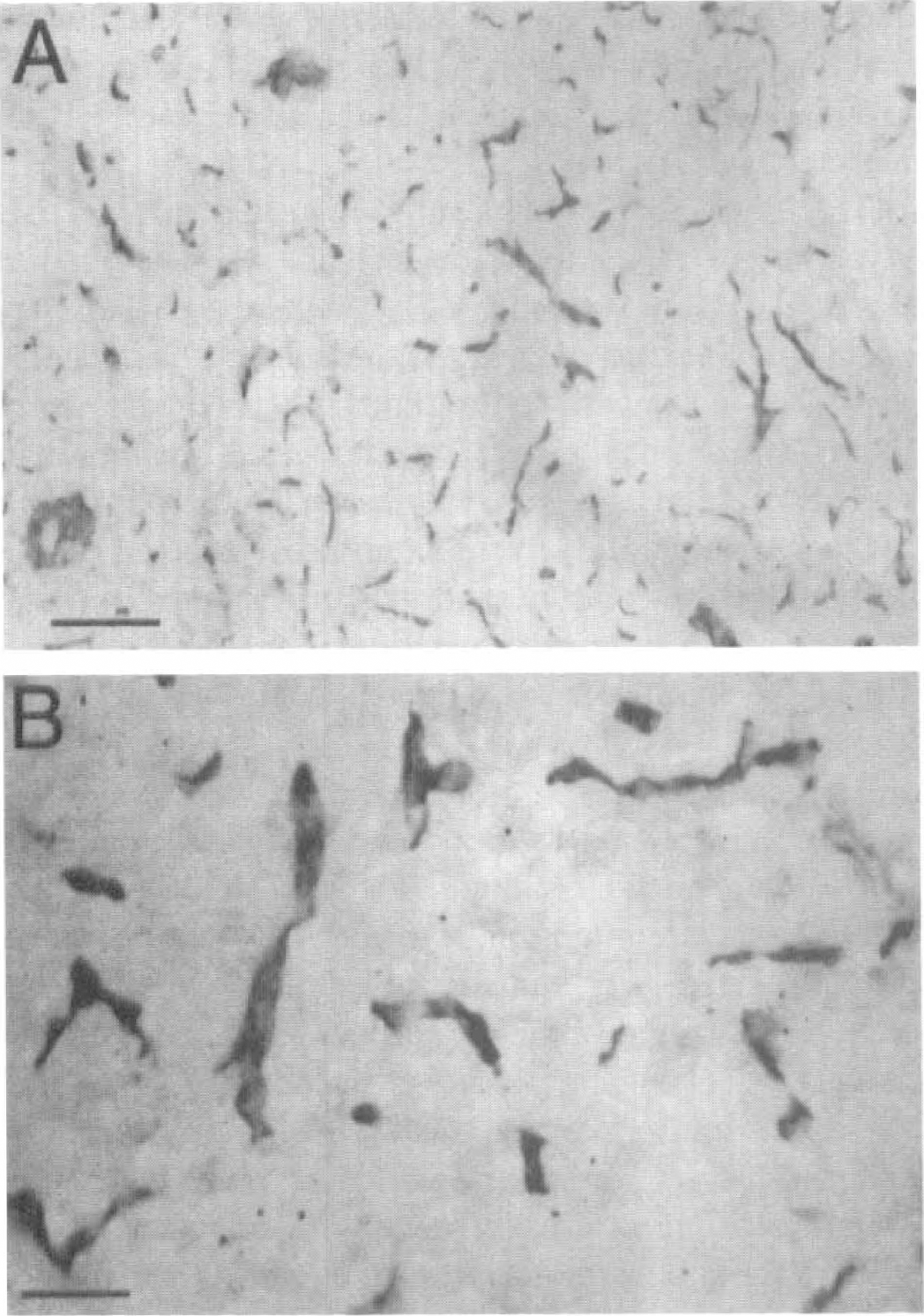
Immunocytochemistry of autopsy fresh frozen normal human brain at low
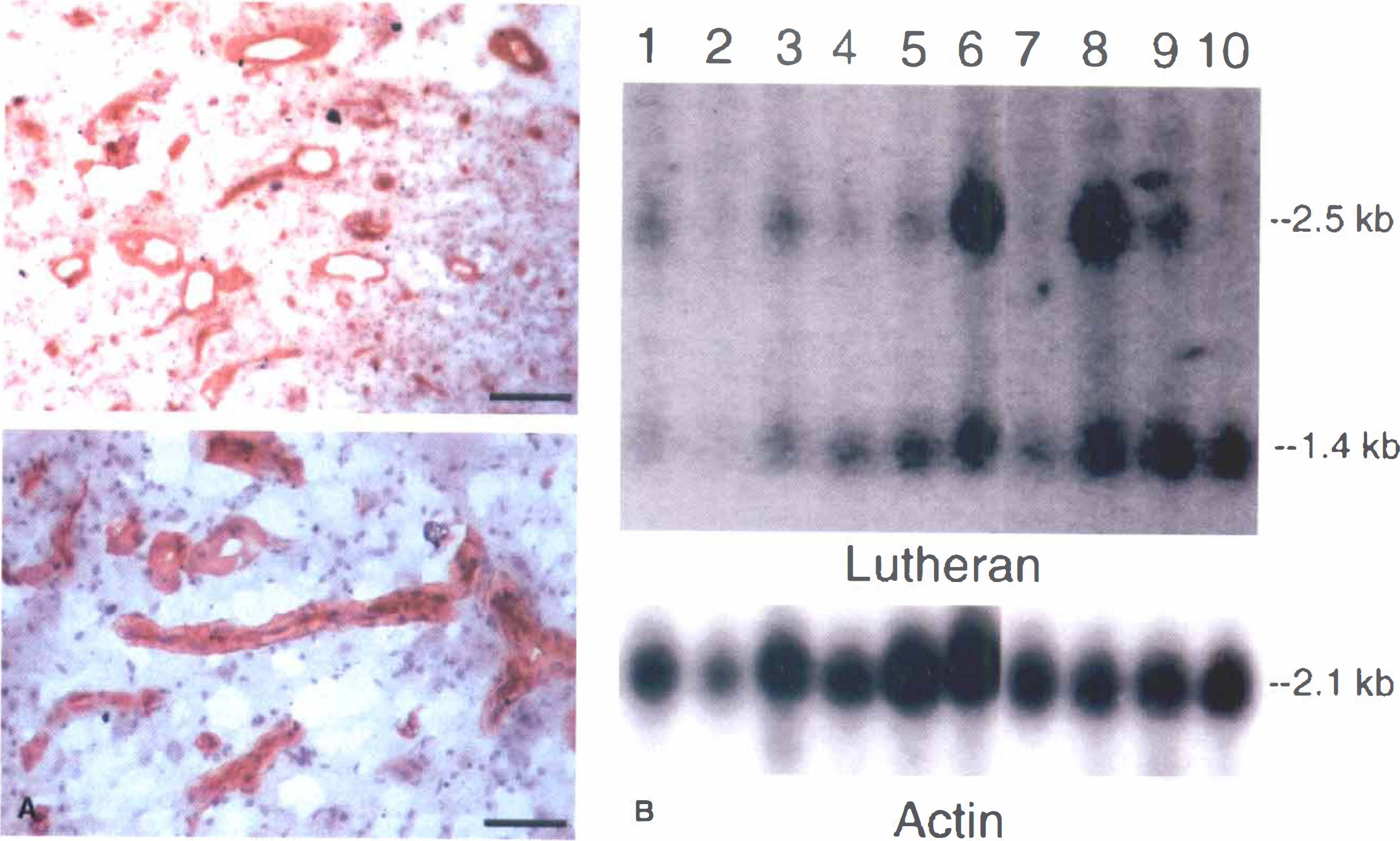
Because the LU glycoprotein gene expression was maintained at a high level in brain capillary endothelial cells grown in tissue culture (Fig. 2B), it was hypothesized that there might be persistent gene expression in capillaries perfusing human brain tumors. This hypothesis was confirmed by both Northern blot analysis and immunocytochemistry as shown in Fig. 4. The 2.5 and 1.4 kb LU transcripts were detected in gliomas, meningiomas, and metastatic tumors to the brain (Fig. 4B). Immunostaining of frozen sections of human glioblastoma multiforme demonstrated immunoreactive LU glycoprotein in tumor capillaries (Fig. 4A).
DISCUSSION
The results of these studies agreed with the following conclusions. First, the LU glycoprotein is expressed at the bovine and human BBB in normal brain, similar to epithelial barriers in peripheral tissues. Second, unlike many BBB specific gene products (Black et al., 1990; Guerin et al., 1990, 1992; Pardridge and Boado, 1993), there is no downregulation of the LU glycoprotein gene in brain capillary endothelium grown in tissue culture (Fig. 2). Third, there is persistent gene expression of the LU glycoprotein at the human BBB perfusing both primary and metastatic human brain tumors (Fig. 4).
The high level of gene expression of the LU glycoprotein at the BBB was demonstrated in normal human brain by immunocytochemistry (Fig. 3) and in control bovine brain by Northern blot analysis (Fig. 2). However, there was no detectable LU mRNA in whole bovine brain (Fig. 2A), or in C6 rat glioma cells even after prolonged exposure of the filter to x-ray film (Fig. 2B). The copy number of the LU glycoprotein mRNA at the BBB was extremely high as the transcript was detected with only a 16-hour exposure of the x-ray film using 2 μg of poly A+ mRNA per lane (Fig. 2A). Therefore, the copy number of the LU mRNA is nearly comparable to that of the LAT1 mRNA at the bovine BBB (Boado et al., 1999), but is much greater than the abundance of the Gluti glucose transporter mRNA at the BBB (Pardridge and Boado, 1993). There is a downregulation of many BBB specific genes when brain capillary endothelial cells are grown in cell culture (Black et al., 1990; Guerin et al., 1990, 1992; Pardridge and Boado, 1993), For example, there is a 100-fold downregulation in Gluti glucose transporter mRNA in bovine brain capillary endothelial cells grown in cell culture relative to freshly isolated bovine brain capillaries (Boado and Pardridge, 1990, 1993). In contrast, there was no downregulation of the LU glycoprotein mRNA when bovine brain capillary cells were grown in cell culture as shown by the Northern blotting studies in Fig. 2B. There was differential processing of the LU glycoprotein mRNA in cell culture as there was a doublet 3.2/2.5 kb LU glycoprotein transcript in bovine brain capillary endothelial cells grown in tissue culture (Fig. 2B). The 3.2 kb LU mRNA had not been reported previously and might be the product of differential mRNA processing in cultured cells.
The persistent expression of the LU glycoprotein gene in brain capillary endothelial cells grown in tissue culture paralleled the persistent high level of expression of this gene at the BBB in human brain tumors (Fig. 4). The anti-LU antibody illuminated capillaries in human high grade gliomas (Fig. 4A), similar to normal human brain (Fig. 3). These immunocytochemistry results were the first demonstration of LU glycoprotein expression at the microvascular endothelium. Previous studies have reported the expression of immunoreactive LU glycoprotein in the arterial walls of human liver, kidney, and placenta (Parsons et al., 1995). Northern blotting showed 2.5 kb and 1.4 kb LU transcripts in primary and metastatic brain tumors (Fig. 4B). The smaller 1.4 kb LU mRNA was also observed in rat lung, heart, and skeletal muscle (Fig. 2D). The 1.4 kb LU mRNA has not been reported previously (Campbell et al., 1994; Rahuel et al, 1996), and the structure and function of this truncated transcript is not known. The relatively low level of LU glycoprotein mRNA in high grade gliomas was likely because this gene was expressed only at the tumor microvasculature (Fig. 4A) and the dilution of the tumor microvascular-derived LU mRNA in the whole tumor extract used to prepare the mRNA samples (Fig. 4B).
Many BBB specific gene products that are downregulated in cell culture are also down regulated in capillaries perfusing human brain tumors (Black et al., 1990; Guerin et al., 1990, 1992). The persistent high gene expression of the LU glycoprotein at the BBB in either cell culture or human brain tumors might relate to the functional significance of this gene product at the BBB. The LU glycoprotein appeared to be an important cell adhesion molecule that might function in pathophysiologic states. The LU glycoprotein in erythrocytes from patients with sickle cell anemia is a receptor for the laminin protein (Nemer et al., 1998; Udani et al., 1998). Laminin constitutes a significant part of the brain microvascular extracellular matrix and basement membrane (Sobel et al., 1998). Whether the BBB LU glycoprotein is a laminin receptor under normal or pathophysiologic conditions is not known at the present time. The persistent expression of the LU gene in brain tumor capillaries may enable tumor capillary endothelium to adhere to basement membrane components such as laminin, and enable angiogenesis. Recent studies have demonstrated that the Leu577-Leu578 motif directs the LU glycoprotein to the basolateral membrane in cultured epithelium (Nemer et al., 1999). The selective localization of the LU glycoprotein to the antiluminal membrane of the brain capillary endothelium, which is the functional equivalent of the epithelial basolateral membrane, would enable LU binding to components of the extracellular matrix such as laminin.
There was a high degree of structural similarity between the nucleotide and amino acid sequence of the bovine and human LU glycoprotein (Fig. 1). All 10 of the cysteine residues in the bovine LU sequence, beginning with residue 243, were 100% conserved compared with the human sequence, including the carboxyl terminal cysteine residue. The single transmembrane region of this glycoprotein was strongly conserved with only 2 conservative substitutions between the human and bovine sequences (Parsons et al., 1995, Fig. 1). Between amino acid Gln243 and the transmembrane region, there were several consensus sequences for N-linked glycosylation in the human and bovine LU (Fig. 1). The Pro557 residue, which forms the carboxyl terminus of the B-CAM isoform was also conserved, as was the src homology 3 (SH3) region between the Pro557 and Pro562 (Parsons et al., 1995).
In summary, these studies demonstrated that the LU glycoprotein was a novel new marker of BBB function in normal and pathophysiologic states such as brain tumors. The LU glycoprotein might be a preferred BBB marker owing to the high level of gene expression of the LU glycoprotein at the BBB and owing to the persistent expression of the LU glycoprotein gene at the BBB in cell culture and in human brain tumors (Fig. 4).
Footnotes
Acknowledgments
Margarita Tayag provided expert technical assistance. Daniel Jeong and Melissa Lara skillfully prepared the manuscript. The authors are indebted to Dr. Harry Vinters, UCLA Department of Pathology/Neuropathology, for review of the histologic specimens.
