Abstract
The role of the mitochondrial calcium uniporter (MCU) gene (
Introduction
The integrative role of mitochondrial Ca2+ uptake and cycling in central neurons has been established on molecular, cellular and system levels.1–3 This role comprises a variety of physiological processes, such as maintenance of intracellular Ca2+ homeostasis, synaptic activity, vesicle exocytosis and energy metabolism.4–10 During neurometabolic coupling, mitochondrial Ca2+ can act as a feed-forward regulator of oxidative phosphorylation and ATP production in mitochondria by affecting the activity of pyruvate dehydrogenase (PDH), several TCA cycle enzymes and F1FO-ATP synthase.1,11,12 Moreover, the detrimental consequences of excessive mitochondrial Ca2+ uptake in neurons have been studied under various pathophysiological conditions.1,13,14 Excessive mitochondrial Ca2+ uptake can trigger several harmful events, including opening of the mitochondrial permeability transition pore and loss of mitochondrial membrane potential, which potentially lead to bioenergetic failure and neuronal death.15–19 Both initiation and cellular consequences of excessive mitochondrial Ca2+ uptake likely differ in ischemia, epilepsy and age-related neurodegenerative diseases, however.20–23
During neuronal activity, cytoplasmic Ca2+ transients are associated with the uptake of Ca2+ into mitochondria and increased mitochondrial matrix Ca2+ concentration.2,6,8,24,25 Importantly, mitochondrial Ca2+ uptake requires the recently identified mitochondrial Ca2+ uniporter (MCU),26,27 which is part of a large protein complex.2,28 Alternative routes for mitochondrial Ca2+ uptake, such as ion exchangers and the rapid uptake mode, have been also described.2,29,30
The role of the MCU in the cellular homeostasis underlying the generation of fast neuronal network rhythms that associate with cortical information processing in vivo is unknown. This role is of particular interest because the awake human brain features
We started to address this by studying gamma oscillations (30–70 Hz) and sharp wave-ripples in hippocampal slice preparations.34–37 These physiological network rhythms require precise synaptic transmission between excitatory pyramidal cells and inhibitory interneurons and occur during wakefulness and sleep.38,39 They support, for example, spike-timing and synaptic plasticity on the cellular level and are thus thought to contribute to the emergence of higher brain functions, such as perception, attention and memory.38–40 We used various knockout (KO) and knockdown (KD) strategies of the
Materials and methods
Mcu-knockout and Mcu-knockdown strategies
All animal experiments conformed to national and institutional guidelines including the Animals [Scientific Procedures Act] 1986 (UK), and the Council Directive 2010/63/EU of the European Parliament, and had full Home Office ethical approval (University of Edinburgh Animal Welfare and Ethical Review Body; University of Heidelberg Animal Welfare Office and Regierungspraesidium Karlsruhe, licenses T46/14 and T96/15). All animals were maintained in pathogen-free and light- (12-h light/12-h dark) and temperature-controlled (22°C ± 2°C) conditions. Food (LasVendi Rod 16 or Rod 18) and water were available ad libitum. Animals were group-housed in conventional cages with ABBEDD LT-E-001 bedding material and were provided with environmental enrichment. Animal welfare was assessed daily by staff of the animal facility. Experiments were performed and reported in accordance with the ARRIVE guidelines. The investigator was blinded for the genotype.
To obtain mice with brain-specific loss of
Recombinant adeno-associated viral vectors for the expression of short hairpin RNAs (shRNAs) contain a ubiquitous U6 promoter for shRNA expression and a CaMKII promoter driving mCherry expression. Note that due to the use of a CaMKII promoter, the fluorescent infection marker is expressed in glutamatergic neurons only, while shRNAs are expressed in all infected cells. To verify that serotype 1/2 efficiently targets parvalbumin-expressing GABAergic basket cells, we infected slice cultures with a rAAV that expresses mCherry under the control of a ubiquitous CMV promoter. Immunostaining for parvalbumin confirmed reliable infection of basket cells (Suppl. Figure 2). The following shRNA target sequences were used in this study (5′–3′): control shRNA targeting LacZ:
Slice preparations
Wild type and
Slice cultures were prepared as described.36,44 In brief, hippocampal slices (400 µm) were cut with a McIlwain tissue chopper (Mickle Laboratory Engineering Company Ltd., Guildford, UK) from 10-day-old male Wistar rats under sterile conditions. Slices with intact hippocampal structures were maintained on Biopore™ membranes (Millicell standing inserts, Merck Millipore, Darmstadt, Germany) between culture medium, which consisted of 50% minimal essential medium, 25% Hank’s balanced salt solution (Sigma-Aldrich, Taufkirchen, Germany), 25% heat-inactivated horse serum (Life Technologies, Darmstadt, Germany), and 2 mM L-glutamine (Life Technologies) at pH 7.3 titrated with Trisbase, and humidified normal atmosphere (5% CO2, 36.5°C) in an incubator (Heracell, Thermoscientific, Dreieich, Germany). Slices were randomly assigned to experimental groups. The culture medium (1 ml) was replaced three times per week. Slice cultures were used after 7–14 days in vitro (DIV), when the tissue had recovered from slice preparation and damaged cut surfaces had been re-organized.1,45
Recording solutions and drugs
Acute slices and slice cultures were stored in a Haas-type interface chamber and constantly supplied with heated (34 ± 1°C) recording solution, i.e. ACSF containing: 129 mM NaCl, 3 mM KCl, 1.25 mM NaH2PO4, 1.8 mM MgSO4, 1.6 mM CaCl2, 26 mM NaHCO3, and 10 mM glucose.36,44 The pH was 7.3 when the recording solution was saturated with 95% O2 and 5% CO2. The gas supply to the interface chamber was 1.5 l/min (95% O2 and 5% CO2).
Gamma oscillations were induced by bath application of acetylcholine (2 µM) and physostigmine (400 nM) in rat slice cultures or the muscarinic receptor agonist carbachol (5 µM) in mouse acute slices.34,36,45,46
Carbachol and physostigmine were purchased from Tocris (R&D Systems GmbH, Wiesbaden-Nordenstadt, Germany). Standard salts and acetylcholine were from Sigma-Aldrich (Taufkirchen, Germany).
Recordings of local field potentials
In rat slice cultures, the local field potential was recorded with glass electrodes (tip diameter 3–5 µm) that were pulled from GB150F-8P borosilicate filaments (Science Products GmbH, Hofheim, Germany) with a PC-10 vertical micropipette puller (Narishige International Ltd., London, UK) and backfilled with ACSF. In mouse acute slices, carbon fibre electrodes (Kation Scientific, Minneapolis, MN, USA) were used. The microelectrodes were positioned in stratum pyramidale of the CA3 region with a mechanical micromanipulator (MX-4, Narishige). Extracellular field potentials were low-pass filtered at 3 kHz, and digitised at 10 kHz using CED 1401 interface and processed with Spike2 software (Cambridge Electronic Design, Cambridge, UK) for offline analysis.
Offline signal analysis of 5-min data segments from local field potential recordings was performed using custom-made scripts in MATLAB 2015a (The MathWorks, Inc., Natick, MA, USA). Recordings of gamma oscillations were low-pass filtered with a digital Butterworth algorithm at 200 Hz cutoff frequency and processed with Welch’s algorithm and fast Fourier transform with a Hamming window size of 32,768 points for calculation of the power spectral density (power) (bin size of 0.3052 Hz). Gamma oscillations were analysed for peak power, frequency and full width at half-maximum (FWHM). Recordings of sharp wave-ripples were band-pass filtered with a Butterworth algorithm between 5 and 60 Hz corner frequencies to detect the transient sharp wave component. Amplitude and incidence of sharp waves were measured and averaged. Ripples superimposed on the sharp wave were analysed using continuous wavelet transformation. Multi-unit activity was assessed by high-pass filtering of the local field potential with a Butterworth algorithm at 700 Hz corner frequency. The threshold for unit detection was set to 4.5 standard deviations of the local field potential during spontaneous asynchronous activity and the correctness of unit detection was checked visually.
Toluidine blue staining and immunohistochemistry
For toluidine blue staining (Sigma-Aldrich), acute hippocampal slices were fixed overnight with 4% paraformaldehyde in phosphate-buffered salt solution (PBS), incubated for 5 h in 30% sucrose (AppliChem GmbH, Darmstadt, Germany) and cut in thin sections (40 µm) with a CM1850 cryostat (Leica Microsystems GmbH, Nussloch, Germany). Sections were mounted on slides, exposed to descending ethanol series, briefly rinsed in double-distilled water and then incubated in 0.1% toluidine blue working solution (pH 2.3) for 1–5 min. Thereafter, the sections were briefly rinsed in double-distilled water; 95% ethanol with traces of glacial acetic acid was used for colour differentiation of the staining. The sections were then exposed to ascending ethanol series, a 1:1 mixture of 100% ethanol and xylene, and finally xylene for 6–10 min. Afterwards the sections were embedded with Entellan®Neu (Merck Millipore, Schwalbach, Germany).
For eGFP, parvalbumin and glial fibrillary acidic protein (GFAP) staining, mice were anaesthetized by intraperitoneal injection of sodium pentobarbital and transcardially perfused with 20 ml of phosphate buffer (PB) followed by 20 ml of ice cold 4% paraformaldehyde. A vibratome (Leica, UK) was used to cut 50 µm thick sections, which were stored free-floating in PB at 4°C until use. Sections were washed in PB, blocked with 5% donkey serum for 1 h and incubated in the following antibodies overnight: anti-GFP conjugated to FITC (1:500 Abcam ab6662), mouse anti-parvalbumin (1:5000, Swant PV235) and rabbit anti-GFAP (1:1000, Dako Z0334). Sections were then washed in PB and incubated in donkey anti-mouse Alexa Fluor 546 antibody (1:500) and donkey anti-rabbit Alexa Fluor 546 antibody (1:500) for 1 h before being washed and mounted onto slides. Sections were imaged using a Leica AF6000LX immunofluorescence microscope.
Western blotting
Wild type,
Gene expression analysis
Wild type and
pPDH analysis
Acute slices were recorded in standard ACSF in a recording chamber, or were treated with carbachol in a recording chamber for 40 min. Slices were then fixed overnight with 4% paraformaldehyde at 4°C. After keeping the slices in 30% sucrose (AppliChem, Darmstadt, Germany) they were cut into 50 µm sections on a cryostat (CM1850, Leica Microsystems, Wetzlar, Germany). Free floating sections were blocked and permeabilised with 5% normal goat serum and 0.3% Triton X-100 for 90 min at RT, then labelled overnight at 4°C with a mixture of primary antibodies (mouse anti-PDH E1-alpha, Life Technologies 456600, lot 456600/G0529, 1:500; rabbit anti-pPDH Ser293, Millipore ABS204, lot 2315725, 1:2000), and then labelled over night at 4°C with a mixture of secondary antibodies (Alexa 594-labelled goat anti-mouse IgG, Thermo Fisher Scientific A-11005, and Alexa 488-labelled donkey anti-rabbit IgG, Thermo Fisher Scientific R37118; both 1:1000). Sections were counterstained with Hoechst 33258 (Sigma-Aldrich, 1 µg/ml), and mounted in Mowiol 4-88 (Calbiochem).
Images were recorded in sequential mode on a Leica TCS SP8 confocal microscope equipped with a Leica HC PL APO CS2 63× NA 1.4 oil-immersion objective (pixel size 240 nm × 240 nm). Images were imported into FIJI 48 and converted to 32-bit; 488 nm and 552 nm excitation channels were split and a threshold was set in both channels to contrast mitochondria. The 488 nm excitation channel (pPDH) was divided by the 552 nm excitation channel (total PDH). The resulting 488 nm/552 nm ratio image was displayed in fire lookup table. The 488 nm/552 nm ratio was measured in stratum pyramidale (as identified in the Hoechst channel) and data were exported to Microsoft Excel. Image acquisition and data analysis were done blinded to genotype.
Calculations and statistics
Electrophysiological data are presented as median with 25th and 75th percentile derived from ‘n' acute slices (cultured slices) from ‘N' mice (rats), unless stated otherwise. Statistical significance,
Results
We used a combined experimental approach to determine the role of the MCU in the generation of fast neuronal network rhythms in the hippocampus of mice and rats.
First, we used acute hippocampal slices from conditional
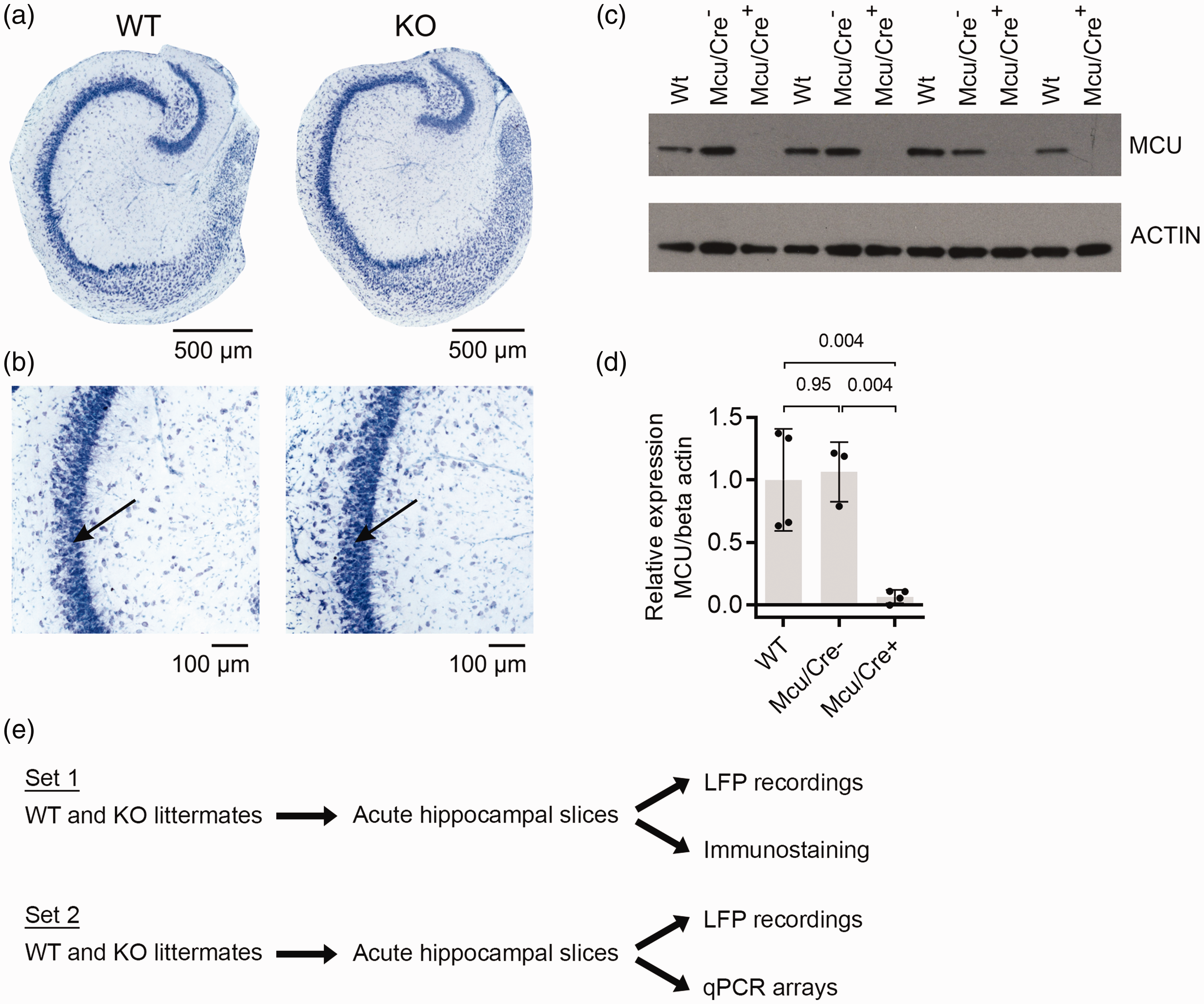
Cytoarchitecture of the hippocampus in wild type (WT) and
As a second approach, we used organotypic hippocamal slice cultures from rats.36,44 In these slice cultures,
In these slice preparations (Figure 1(e)), we performed local field potential recordings, including fast Fourier transform, to characterize the properties of cholinergically induced, persistent gamma oscillations and spontaneously occurring, recurrent sharp wave-ripples, both of which share many features with their counterparts in vivo.36,37,46,51 We focussed on the CA3 region, which is an intrinsic generator of gamma oscillations and sharp wave-ripples in the hippocampus,34,50,51 and shows a higher
Disturbances of gamma oscillations
Gamma oscillations with a frequency of around 32 Hz were present in the CA3 region of wild type and
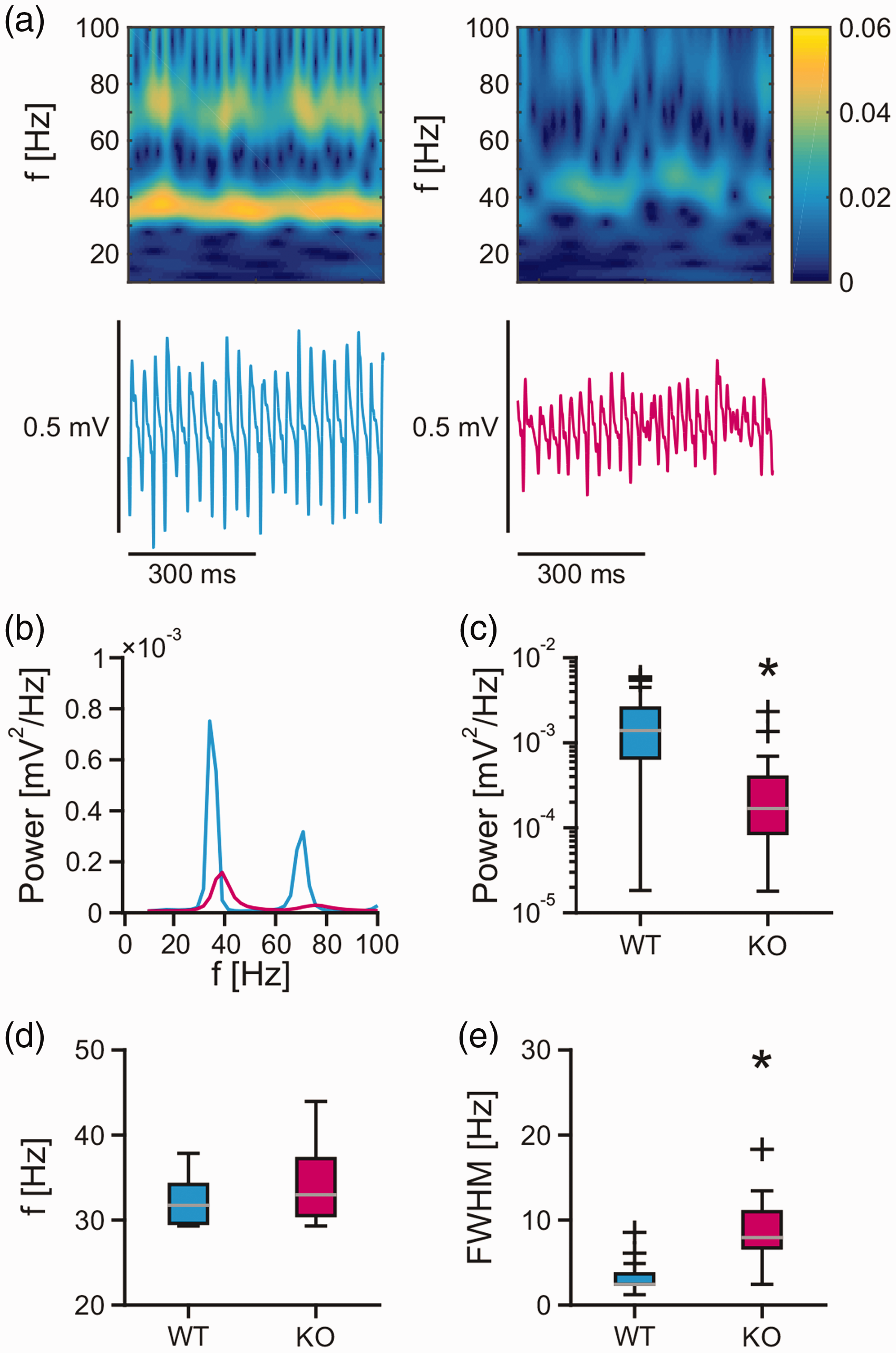
Gamma oscillations in mouse acute slices. Persistent gamma oscillations were recorded in stratum pyramidale of the CA3 region in acute hippocampal slices of the mouse. (a) Sample traces of local field potential recordings and corresponding wavelet transform of gamma oscillations in wild type (blue trace) and
Similar disturbances in the power of gamma oscillations were present in the CA3 region of rat slice cultures expressing shRNAs against
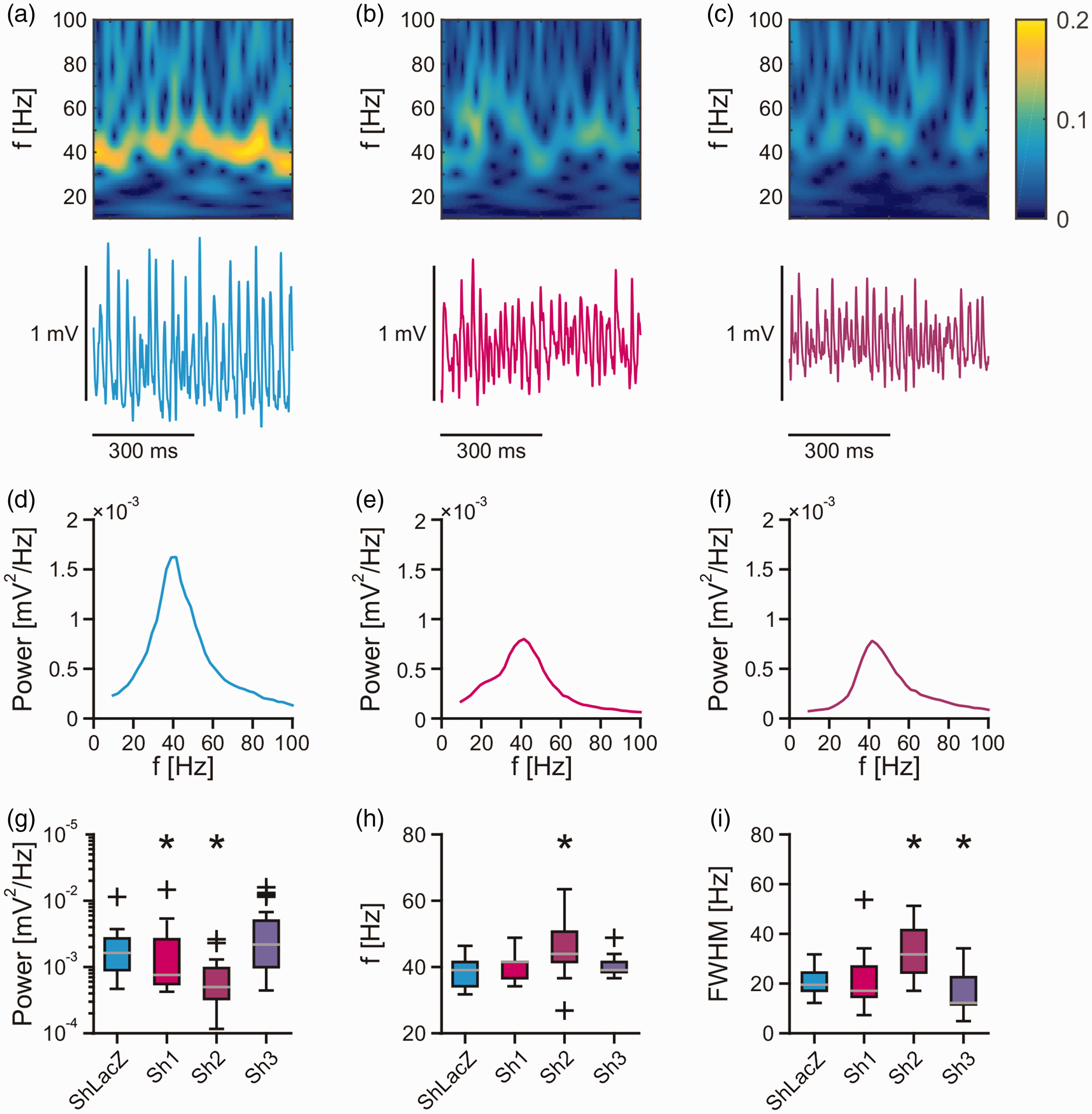
Gamma oscillations in rat slice cultures. Persistent gamma oscillations were recorded in stratum pyramidale of the CA3 region in hippocampal slice cultures of the rat. (a–c) Sample traces of local field potential recordings and corresponding wavelet transform of gamma oscillations in control (a, ShLacZ) and
These findings suggest that the MCU is required for proper generation of gamma oscillations.
Disturbances of neuronal spiking synchronization
We further characterized the synchronization of neuronal action potential generation (‘spiking') during gamma oscillations. For this purpose, multi-unit activity was extracted from local field potential recordings (Figure 4(a)). In wild type mice, there was a second peak of the multi-unit intervals present at 20–30 ms, reflecting synchronized neuronal spiking at the gamma-band rhythm (Figure 4(b)). Notably, this second peak of multi-unit intervals was absent in
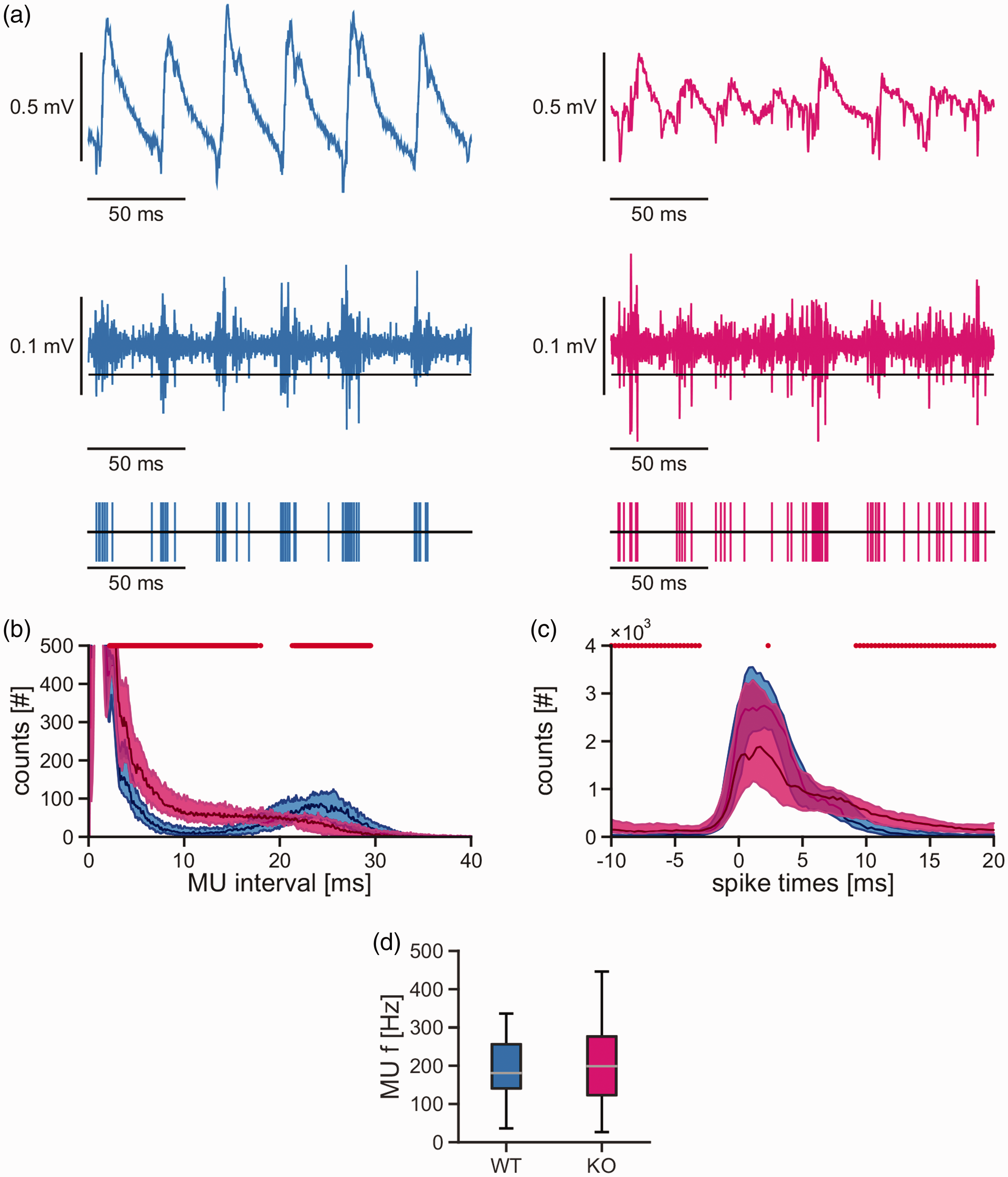
Spiking synchronization during gamma oscillations in mouse acute slices. Persistent gamma oscillations were recorded in stratum pyramidale of the CA3 region in acute hippocampal slices of the mouse. (a) Sample traces of the local field potential (top), with 700 Hz high-pass filter (middle) and extracted multi-unit (‘spiking') activity (bottom) during gamma oscillations in slices from wild type (blue traces) and
These findings suggest that the MCU is required for proper synchronization of neuronal spiking during gamma oscillations.
Disturbances of sharp wave-ripples
To test whether the disturbances on the network and cellular level described above are specific for gamma oscillations, we additionally characterized the properties of sharp wave-ripples. Recurrent sharp wave-ripples were present in the CA3 region of wild type and
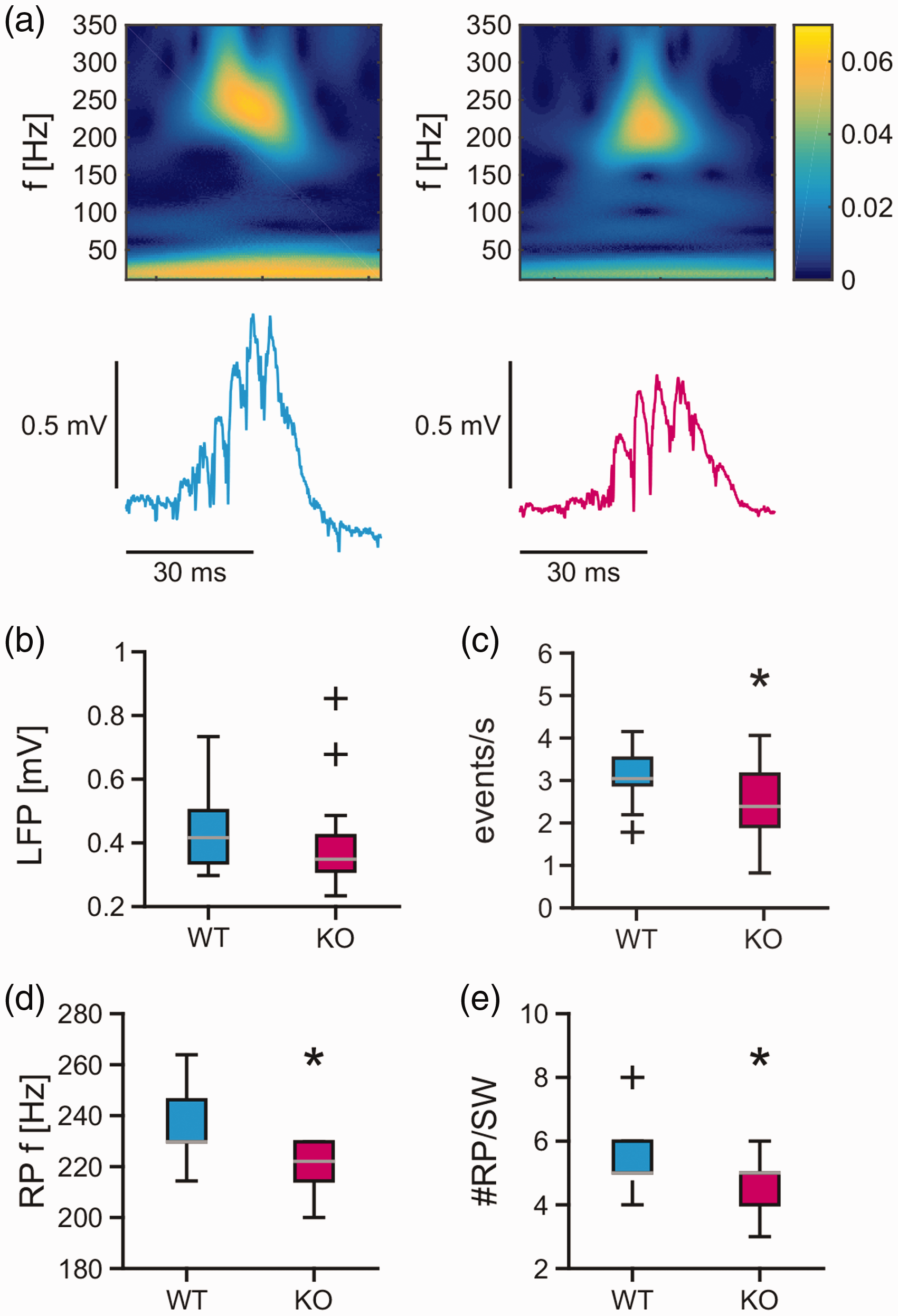
Sharp wave-ripples in mouse acute slices. Spontaneously occurring, recurrent sharp wave-ripples were recorded for 5 min in stratum pyramidale of the CA3 region in acute hippocampal slices of the mouse. (a) Sample traces of local field potential recordings and corresponding wavelet transform of single sharp wave-ripples in wild type (blue trace) and
These findings suggest that the MCU is also required for proper generation of sharp wave-ripples.
Alterations of neurometabolic coupling
Cortical information processing depends on ATP supply by neuronal mitochondria.
22
We therefore reasoned that impaired Ca2+-mediated activation of mitochondrial oxidative metabolism might be one mechanism that underlies the disturbances in neuronal network oscillations in
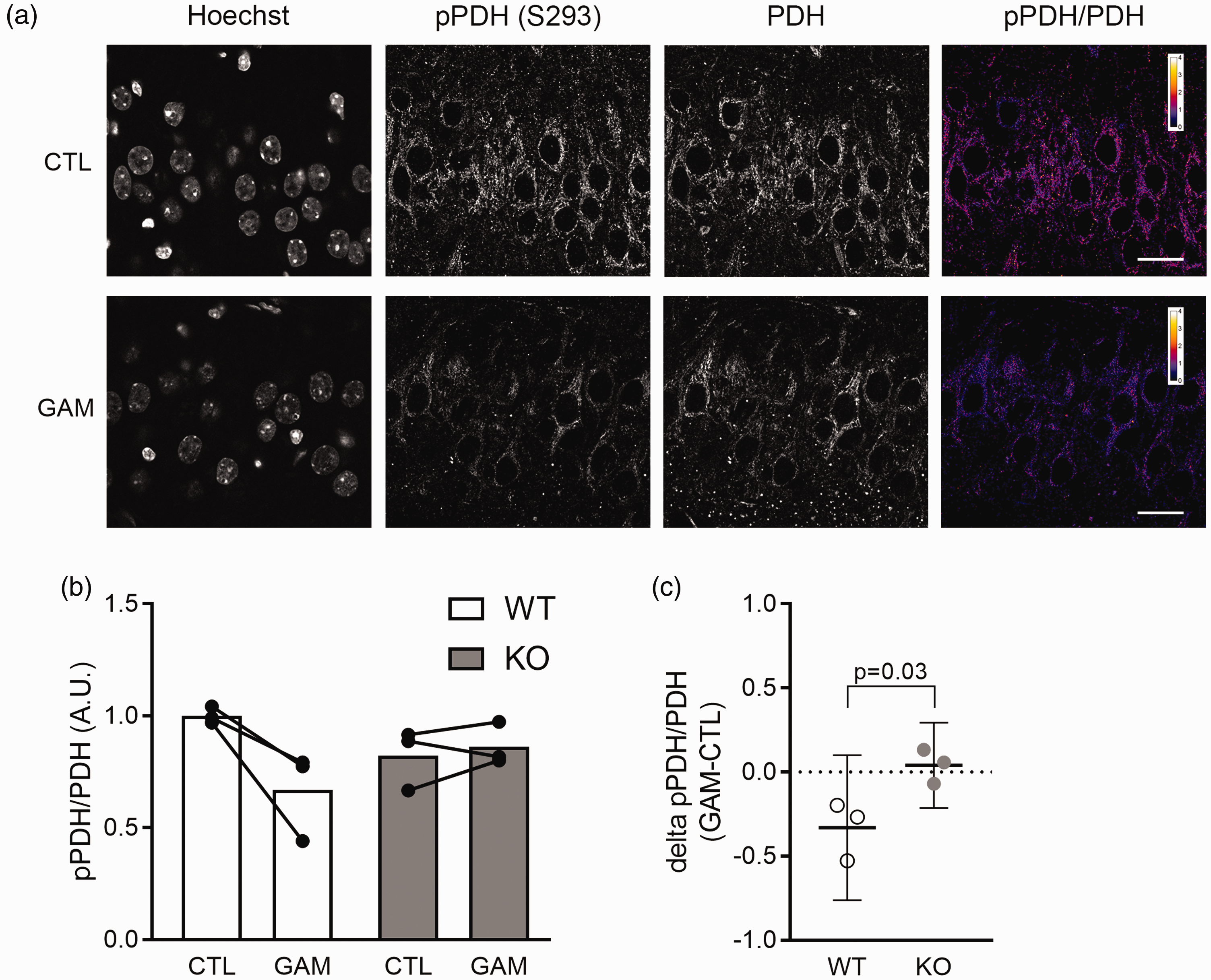
Gamma oscillation-mediated dephosphorylation of PDH in mouse acute slices. (a) Immunofluorescence labelling of acute mouse hippocampal slices. Anti-phospho PDH labelling, anti-total PDH labelling, and a pPDH/PDH ratio image are shown for a control slice (CTL) and for a slice that underwent 40 min of gamma oscillations (GAM). Both slices are from the same wild type mouse. Scale bars represent 20 µm. (b) Quantification of pPDH/PDH ratio in control slices (CTL) and slices that underwent 40 min of gamma oscillations (GAM). Slices from three animals were analysed for both conditions in each genotype. Round symbols represent individual animals; bars represent mean ratio. Values were normalized to average ratio in wild type control slices. (c) Quantification of gamma oscillation-mediated PDH dephosphorylation in wild type and
These findings indicate that during gamma oscillations the Ca2+-dependent stimulation of the mitochondrial TCA cycle is dysfunctional in
Absence of adaptations in gene expression
We next asked if the reduced capability to activate mitochondrial oxidative phosphorylation in
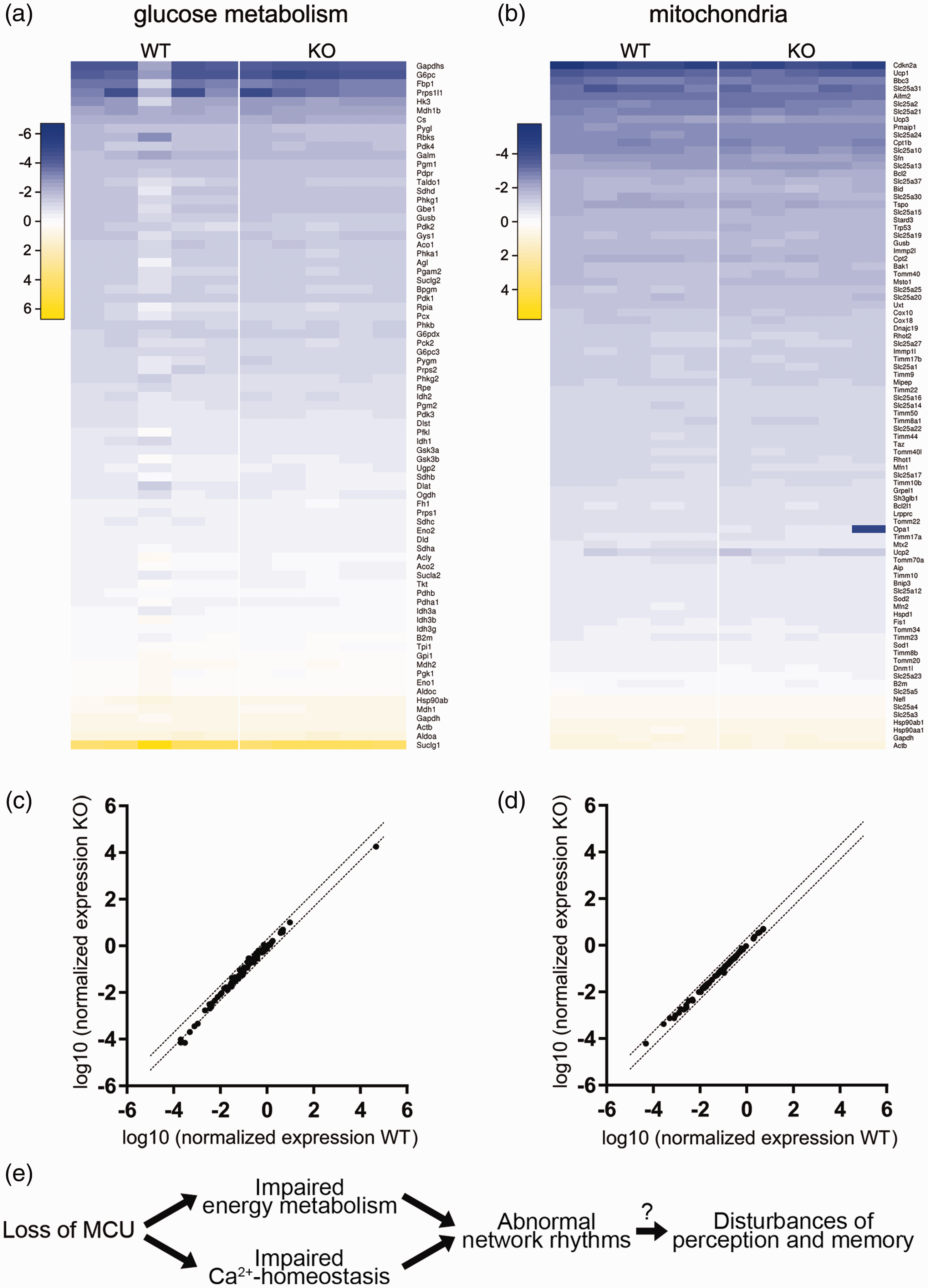
Expression of genes related to glucose metabolism and mitochondria in the mouse hippocampal region CA3 and summary scheme. (a, b) Heatmaps illustrating the results of RT2 Profiler PCR Array gene expression analyses of glucose metabolism-related (a) and mitochondria-related (b) genes. N = 5 mice per genotype. Colour scale represents log10 of normalized expression (2−dCT). (c, d) Comparison of average expression per gene in wild type versus
These findings suggest that under basal conditions there are no compensatory adaptations in energy metabolism in
Discussion
Mcu-KO and Mcu-KD strategies
In this study, we used two complementary approaches to investigate the role of the MCU during fast neuronal network rhythms. First, we generated a brain-specific
Disturbances of gamma oscillations and sharp wave-ripples
The role of the MCU complex in neurometabolic coupling has been addressed in a number of previous experimental studies. These studies, however, focused on widely undefined activation stages of cultured neurons using artificial stimuli 54 or on intense metabolic stress during hypoxic-ischemic brain injury. 55
Here, we used gamma oscillations and sharp wave-ripples as models of physiological neuronal network rhythms. Gamma oscillations emerge in many cortical areas in awake mammals, including humans, and have been associated with sensory perception, attentional selection, motor activity and memory formation.22,39 Sharp wave-ripples arise in the hippocampus during waking immobility, consummatory behaviour and slow-wave sleep. They assist in transferring compressed hippocampal information to distributed neocortical circuits to support memory consolidation.38,39 In addition, sharp wave-ripples have been implicated in erasure of hippocampal memory traces and certain aspects of active spatial navigation.38–40
Gamma oscillations and sharp wave-ripples rely on the precise synaptic transmission between excitatory pyramidal cells and GABAergic interneurons, in particular parvalbumin-positive, fast-spiking interneurons, such as basket cells.34,35,50,51 In generating these patterns of activity, pyramidal cells excite fast-spiking inhibitory interneurons that in turn transiently inhibit the perisomatic region of pyramidal cells through rhythmic GABA release. During gamma oscillations, for example, individual pyramidal cells generate action potentials at 1–3 Hz, whereas fast-spiking inhibitory interneurons show much higher firing rates (>20 Hz).22,34 We note that fast-spiking inhibitory interneurons were not targeted with our
We demonstrate disturbances in the generation of gamma oscillations and the underlying neuronal spiking. In addition, we report the lack of activity-dependent PDH dephosphorylation in
However, we also demonstrate disturbances in the generation of sharp wave-ripples that are thought to involve less energy expenditure compared with gamma oscillations. 37 Therefore, the functions of the MCU in excitatory pyramidal cells may not be restricted to neurometabolic coupling and energy metabolism (Figure 7(e)). Similar to other excitable cells, the MCU also has an important role in Ca2+ homeostasis in neurons.2,60,61 During neuronal activity, mitochondrial Ca2+ uptake significantly shapes cytoplasmic Ca2+ transients and thereby modulates neural excitability9,62,63 and the release of neurotransmitters,4,7,59,64,65 including nitric oxide.45,66,67 Thus, loss or dysfunction of the MCU might affect neuronal signalling on different levels.
Disturbances of gamma oscillations and sharp wave-ripples might affect spike-timing and synaptic plasticity in local cortical networks as well as information transfer between remote cortical networks.38–40
Our data suggest that dysfunction of the MCU might result in cognitive impairment, with putative relevance to several neurological and psychiatric disorders.2,13,22,68,69
Absence of adaptations in gene expression
Similar to previous studies in different MCU loss-of-function models,30,55,56 we found no evidence for impaired cellular function under basal conditions in our newly generated
Conclusions
We provide substantial evidence that the neuronal MCU is crucial for the generation of fast cortical network rhythms, most likely by adapting oxidative phosphorylation, and perhaps by controlling cytoplasmic Ca2+ homeostasis. Our study might contribute an increased understanding of the functions of the MCU during neuronal information processing. Future studies on neuronal Ca2+ homeostasis and synaptic transmission as well as on the behavioural level will further clarify these relationships.
Supplemental Material
JCB887777 Supplementary material - Supplemental material for The mitochondrial calcium uniporter is crucial for the generation of fast cortical network rhythms
Supplemental material, JCB887777 Supplementary material for The mitochondrial calcium uniporter is crucial for the generation of fast cortical network rhythms by Carlos Bas-Orth, Justus Schneider, Andrea Lewen, Jamie McQueen, Kerstin Hasenpusch-Theil, Thomas Theil, Giles E Hardingham, Hilmar Bading and Oliver Kann in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deutsche Forschungsgemeinschaft (Collaborative Research Center 1134, projects B02 and C03; FOR2289, BA1007/9-1 and BA3679/4-2) and also by the UK Medical Research Council, Alzheimer’s Research UK and Alzheimer’s Society.
Acknowledgements
The authors thank Yan-Wei Tan for providing shRNA against LacZ.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Carlos Bas-Orth: Conceptualization, Funding acquisition, Investigation, Formal analysis, Visualization, Writing – original draft, Writing – review and editing
Justus Schneider: Investigation, Formal analysis, Visualization, Writing – review and editing.
Andrea Lewen: Investigation, Formal analysis, Visualization, Writing – review and editing.
Jamie McQueen: Resources, Investigation, Formal analysis, Writing – review and editing.
Kerstin Hasenpusch-Theil: Resources, Interpretation, Writing – review and editing.
Thomas Theil: Resources, Interpretation, Writing – review and editing.
Giles E Hardingham: Conceptualization, Resources, Project administration, Writing – review and editing.
Hilmar Bading: Conceptualization, Funding acquisition, Project administration, Writing – review and editing.
Oliver Kann: Conceptualization, Funding acquisition, Project administration, Writing – original draft, Writing – review and editing.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
