Abstract
Novel therapeutic intervention that aims to enhance the endogenous recovery potential of the brain during the subacute phase of stroke has produced promising results. The paradigm shift in treatment approaches presents new challenges to preclinical and clinical researchers alike, especially in the functional endpoints domain. Shortcomings of the “neuroprotection” era of stroke research are yet to be fully addressed. Proportional recovery observed in clinics, and potentially in animal models, requires a thorough reevaluation of the methods used to assess recovery. To this end, this review aims to give a detailed evaluation of functional outcome measures used in clinics and preclinical studies. Impairments observed in clinics and animal models will be discussed from a functional testing perspective. Approaches needed to bridge the gap between clinical and preclinical research, along with potential means to measure the moving target recovery, will be discussed. Concepts such as true recovery of function and compensation and methods that are suitable for distinguishing the two are examined. Often-neglected outcomes of stroke, such as emotional disturbances, are discussed to draw attention to the need for further research in this area.
Introduction
Over the past three decades, research efforts in developing viable stroke therapies have focused on neuroprotective mechanisms. A plethora of studies utilizing the neuroprotective approach have produced very promising results in animal studies. Yet several hundreds of potential therapeutic agents that have been tried at Phase II or III clinical trials showed very little or no efficacy.1,2
The apparent failure in bench-to-bedside translation was recognized as early as 1999, and recommendations to improve preclinical research design and standards were published by The Stroke Therapy Academic Industry Roundtable (STAIR). 3 Almost 20 years after the publication of numerous guidelines,4,5 literature indicates that the STAIR and STEPS initiatives have made significant contributions in increasing the quality of research in the field. 6 While certain crucial recommendations of these initiatives are yet to be adequately implemented, 7 the scientific rigor in stroke studies has been ameliorated drastically. 6 Despite improvements, preclinical research is still affected by quality issues, including publication bias, false positives, lack of randomization and blinding, suboptimal sample size and inadequate long-term functional assessments. 8
The failures in translation were not due solely to shortcomings in preclinical research. In fact, several clinical studies were undertaken with no or inadequate preclinical data, 9 and in many cases the amount of evidence supporting the efficacy of the selected drug was not greater than that of other experimental interventions. 10 It has also been proposed that some clinical trial results may be false negatives 8 and many clinical trial designs may be suboptimal.11,12 Accordingly, STAIR has published guidelines to improve numerous aspects of clinical trials, including, but not limited to, patient selection criteria, pharmacokinetic evaluation, characterization of the dose response, and functional outcome assessments. 13
As our understanding of the extent and physiology of recovery has improved, research focus has shifted to enhancing the endogenous recovery potential of the brain. 14 It is now well established that a substantial amount of functional recovery occurs after stroke due to plastic remodeling. 15 Recent studies employing neurorestorative therapies have primarily relied upon potentiating plasticity-related processes such as angiogenesis, neurogenesis, oligondendrogenesis and white matter remodeling. 16 Various intervention modalities, including cell-based and pharmacological therapies, brain stimulation, and environmental enrichment,16–18 have produced very promising results in animal models and small clinical trials, 19 thereby rejuvenating stroke research. Moreover, combination therapies – such as treatment with potential neuroprotective agents in conjunction with successful recanalization – may lead to a renaissance of neuroprotective and other experimental therapies that have previously been deemed unsuccessful when used alone in clinical trials. 20
While the shortcomings of the “neuroprotection era” are yet to be fully addressed, the ongoing shift in research efforts towards enhancing recovery presents new challenges to preclinical and clinical researchers alike. The scientific community has already acknowledged the need to take action and a set of recommendations from Stem cell Therapeutics as an Emerging Paradigm for Stroke (STEPS) initiative and Stroke Recovery and Rehabilitation Roundtable (SRRR) have been published.21–23 Both STEPS and SRRR recommendations underlined the need to improve sensory motor evaluations and provided directions. 24
To expand upon STEPS and SRRR recommendations, this review aims to give a detailed evaluation of functional outcome measures used in clinics and preclinical studies. Post stroke impairments and the timeline of recovery in human patients and preclinical models will be discussed from a functional testing perspective. Often-neglected outcomes of stroke – emotional disturbances, particularly post-stroke depression and anxiety – are discussed in a separate section. “Proportional functional recovery” is a critical observation in stroke patients, whereby the majority of patients recover about 70% of maximum possible improvement. We will question whether proportional recovery is present in animal models and what steps should be taken to ensure accordance between clinical and preclinical observations. Finally, the distinction between real recovery and compensation will be discussed, along with their effects on functional endpoint selection.
Post-stroke impairments
Impairments in stroke patients
Human stroke is highly variable and outcomes depend on location of ischemia, severity, duration, age, gender and comorbid conditions. Typically, a single infarction causes impairments in numerous domains of neurological functioning. Stroke patients display a myriad of impairments that include sensorimotor, emotional, and cognitive deficits. 25 More than 50% of stroke survivors are left with physical disabilities.26,27 Notable motor impairments include loss of control of the face, arm, or leg of one side of the body and affects approximately 80% of stroke patients. 27 Muscle weakness is a cardinal feature of stroke that affects balance, posture and movement. 28 Loss of somatosensation is present in one in two people after stroke and may manifest as difficulty in sensing touch, pressure, and temperature, perceiving limb position and recognition of objects by touch, 29 as well as altered or reduced pain sensation. 30 Upper limb motor impairment, seen in 77% of stroke patients, is commonly experienced in conjunction with somatosensory impairments. 31 Cognitive decline is frequent in stroke patients and particularly affects learning 32 memory, attention, concentration, and alertness. 27 Speech impairments are also common, with up to 30% of patients exhibiting aphasia and dysarthria. 33
Impairments observed in preclinical models
To mimic the heterogeneity of human stroke, numerous animal models of focal ischemia have been developed (see Fluri et al. 34 for a review). The deficits observed depend on the model used, intensity of ischemia (i.e. time and extent of occlusion) and the affected regions. Among experimental models, MCAo via the intraluminal filament method has been the most widely used method (∼47%) and produces the largest lesions (on average ∼32% of the hemisphere). 35 Post-MCAo animals are lethargic with paresis on one side and display mild to severe circling behavior towards ipsilesional side. Animals lose a significant amount of body weight as the lesion develops; the extent of body weight loss is correlated with the lesion size and reaches maximum by days 3 to 5. The initial circling and apparent paresis resolve rapidly and animals begin gaining weight by day 5, gradually reaching baseline body weight.
MCAo induces postural reflex anomalies, 36 sensory deficits in proprioception, 37 coordination problems, 38 muscle weakness, and reduced endurance39,40 and leads to impairments in gross motor function, 41 walking, 42 gait39,43 and posture.44,45 Fine motor function deficits39,40 on hind46,47 and front limbs, 48 along with deficits in skilled reaching 49 are frequently observed. Tactile sensation is also impaired.50,51 Stroked animals perform poorly in learning and memory tasks,52,53 as MCAo impairs episodic and spatial memory, 54 working memory, 55 procedural learning, 56 contextual fear learning 57 and strategy switching. 58 Abnormalities in ultrasonic vocalizations59,60 used in various social interactions are also observed. 61 Targeted stroke models such as endothelin-1 (ET-1) injection or photothrombosis can also induce similar impairments in a less diverse and isolated fashion.62,63 Studies using stereotactic injection of the potent vasoconstrictor ET-1 to produce focal infarctions in either or both the forelimb motor cortex (homologous to the human primary motor cortex) and dorsolateral striatum report persistent reproducible deficits in limb use, skilled reaching, grasping and fore and hind limb coordination. 34 Photothrombotic occlusions in the motor cortex also produce similar lesions and impairments. 64 Distal MCAo models primarily affect the whisker/vibrissal cortex and cause somatosensory deficits. 65 Similar lesions are rare in clinical populations. 66 Microembolic stroke models lead to distributed lesions, which represents a minority of stroke cases in humans, and induces deficits in attention and memory. 67
Aligning preclinical lesion characteristics and impairments with clinical populations
Animal models can replicate practically all significant functional deficits of human strokes. In rodents, moderate to severe MCAo induces almost all of the impairments observed in stroke patients. 39 An ideal model, however, should not only mimic the clinical impairments but ensure that behavioral deficits and lesion characteristics simultaneously match those of stroke patients. Due to the heterogeneity in lesions and relevant impairments in stroke patients, there is no “silver bullet” model. Preclinical researchers should be well-versed about the lesion characteristics seen in clinical studies and select or adjust their stroke models accordingly.
In this sense, there are discrepancies between a significant portion of clinical and preclinical studies. Widely used severe MCAo models (>60 min, permanent) typically lead to large malignant infarctions that cause arterial compression, progressive edema and infarct expansion. In comparison, most human stroke lesions are small in size, encompassing 4.5% to 14% of the ipsilateral hemisphere. Only 10% of human strokes are malignant infarctions (lesion size >39% of hemisphere). 68 Such patients have high mortality (∼80%) and respond very poorly to therapy. 69 As a result, they are rarely included in clinical studies. In addition, a significant portion of clinical rehabilitation and long-term recovery studies select patients with mild to moderate hemiparesis.70,71 Edwardson et al. have compared preclinical studies of upper limb recovery to patients from the Interdisciplinary Comprehensive Arm Rehabilitation Evaluation (ICARE) study. Their findings indicate that within the patient population, less than 1% had lesions resembling surface vessel occlusion or severe MCAo models used in preclinical studies evaluating limb recovery. Only 16% of patients had lesions similar to cortical photocoagulation and endothelin-1 injection models. Capsular hematoma and focal ischemia models leading to white matter lesions were more representative. 72 It should be noted that these findings do not argue against the usefulness of MCAo model, but rather underline a mismatch between the preclinical and clinical studies of forelimb recovery. Indeed, authors state that “Short occlusion times for temporary MCAo may best match the relative lesion volumes from the ICARE sample”. 72 Lesion sizes in widely used ET-1 models are also small and representative of clinical populations. Recent studies of forelimb recovery in rodents using the ET-1 model reported lesion sizes comparable to clinical populations. 73 Regardless, there is an apparent gap in preclinical literature regarding pure white matter stroke studies and further studies are warranted.
Animal models of stroke have been met with criticism and assertions that there are serious differences between human and animal brains and that preclinical models do not mimic clinical conditions. However, with proper model selection, lesion characteristics and post-stroke deficits observed in human patients can be mimicked with utmost fidelity for long-term recovery studies. Preclinical researchers should be meticulous in terms of matching lesion characteristics and functional deficits to clinical patient populations. This can only be achieved by detailed reporting of lesion and impairment characteristics in clinical trial populations. Unfortunately, the broad inclusion criteria and insufficient patient stratification in earlier clinical trials of neuroprotection, which has been criticized as a factor contributing to translational failures, have stagnated the field in this regard as well.
Tools used for functional assessments
Clinical assessments
Historically, mortality and event recurrence have been used in stroke clinical trials. Since these clinical endpoints do not reflect the full spectrum of the disabling conditions of stroke, assessment of functional ability has been adopted. 74 The “World Health Organization International Classification of Function” (ICF) characterizes function in terms of body functions, activity and participation. Various scales are used to evaluate the above mentioned levels of functional outcomes in stroke trials as primary or as secondary endpoints. 75 There is no consensus and considerable heterogeneity in the functional outcome measures with more than 47 different scales used in clinical trials by 2009. 75 In addition, statistical analysis methods were not uniform when identical scales were used. 76 Nevertheless, a few of these scales have been widely used in clinical trials.
The Barthel Index
The Barthel Index (BI) was developed to measure independence and is used to score improvement in rehabilitation and assist patient discharge. It is a 10-item scale that measures self-care and mobility-related basic activities of daily living such as feeding, bathing, dressing. With its ease of use, familiarity among clinicians and satisfactory construct and face validity,77,78 it has been the most frequently used scale in acute clinical trials of stroke until 1998. 79 The BI is scored via observation and interviewing of the patient by a trained individual using the items on the scale. The reliability and responsiveness of the scale are reasonable. However, the BI suffers from a number of shortcomings including “Floor” and “Ceiling” effects. Importantly, it does not address key aspects of independence such as vision, cognition and pain. 80
The modified Rankin scale
Adapted from the original Rankin scale from 1957, the modified Rankin scale (mRS) evaluates recovery from stroke and functional independence. The mRS is a six-point scale in which 0 indicates no deficit, 4 indicates moderately severe disability and need for help in performing daily activities, 5 indicates severe disability and 6 marks death. Like the BI, the mRS is also scored via observation and interviewing of the patient by a trained individual using the items on the scale. However, compared to the BI, the mRS offers rapid and easy evaluation in clinical practice and is a better tool for distinguishing between changes in mild to moderated disability. Consequently, the mRS has replaced the BI as the primary functional outcome measure in clinical trials. 75 The scale offers excellent reliability and validity. However, due to the basic 6-point assessment, its overall responsiveness (sensitivity to change) is poor. 81
The National Institutes of Health Stroke Scale
The National Institutes of Health Stroke Scale (NIHSS) has been developed as an impairment scale utilized in acute stroke practices and clinical trials. 82 The NIHSS is a 15-item scale that aims to standardize and quantify a basic neurological examination. Through observation and interaction with the patient, the NIHSS assesses the following domains: level of consciousness, eye movements, visual field integrity, facial movements, leg and arm muscle strength, sensation, coordination, speech, language and neglect. The test is simple to perform and takes approximately 6 min. Impairments are scored on a scale ranging from 0 to 2, 0 to 3, or 0 to 4. The sum total of the item scores ranges from 0 to 42, and scores above 21 are generally considered as severe. 83 The NIHSS offers excellent reliability and adequate validity, but also suffers from several weaknesses: it does not include a detailed assessment of cranial nerves, leading to relatively lower scores in patients with cerebellar or brainstem lesions. 80 Its validity in non-dominant hemisphere strokes has also been questioned, 84 and a ceiling effect was observed at six months in 20% of patients. 85
The Fugl-Meyer assessment
Often considered to be the most comprehensive quantitative measure of stroke, the Fugl-Meyer assessment (FMA) was developed as the first disease-specific impairment index for the assessment of recovery in hemiplegic patients. 86 The FMA scale is a 226-point scale that is divided into five categories: motor function, sensory function, balance, range of motion in joints, and joint pain. Each of these categories is scored on a 3-point ordinal scale (0 = unable to perform, 1 = partially performs, 2 = fully performs). The motor domain measures movement, coordination and reflex actions at several joints and is scored from 0 to 100 points. The other categories also follow a similar point system, and the entire assessment takes about 30 min to perform. 87 Despite its relative complexity, the FMA is widely used in rehabilitation-related clinical studies and increasingly used in others. A current search in the clinical trials database of NIH (clinicaltrials.gov) shows 245 active clinical trials are utilizing Fugl-Meyer as a primary or secondary outcome measure. The FMA displays excellent face, predictive, 88 content 89 and construct 90 validity both for acute and chronic strokes. In chronic stroke, its responsiveness is moderate to large. 91 Shortcomings of the FMA include a ceiling effect with the sensation score 92 and a floor effect in the modified balance score 93 and hand and lower extremity items. 87
Modality specific functional tests
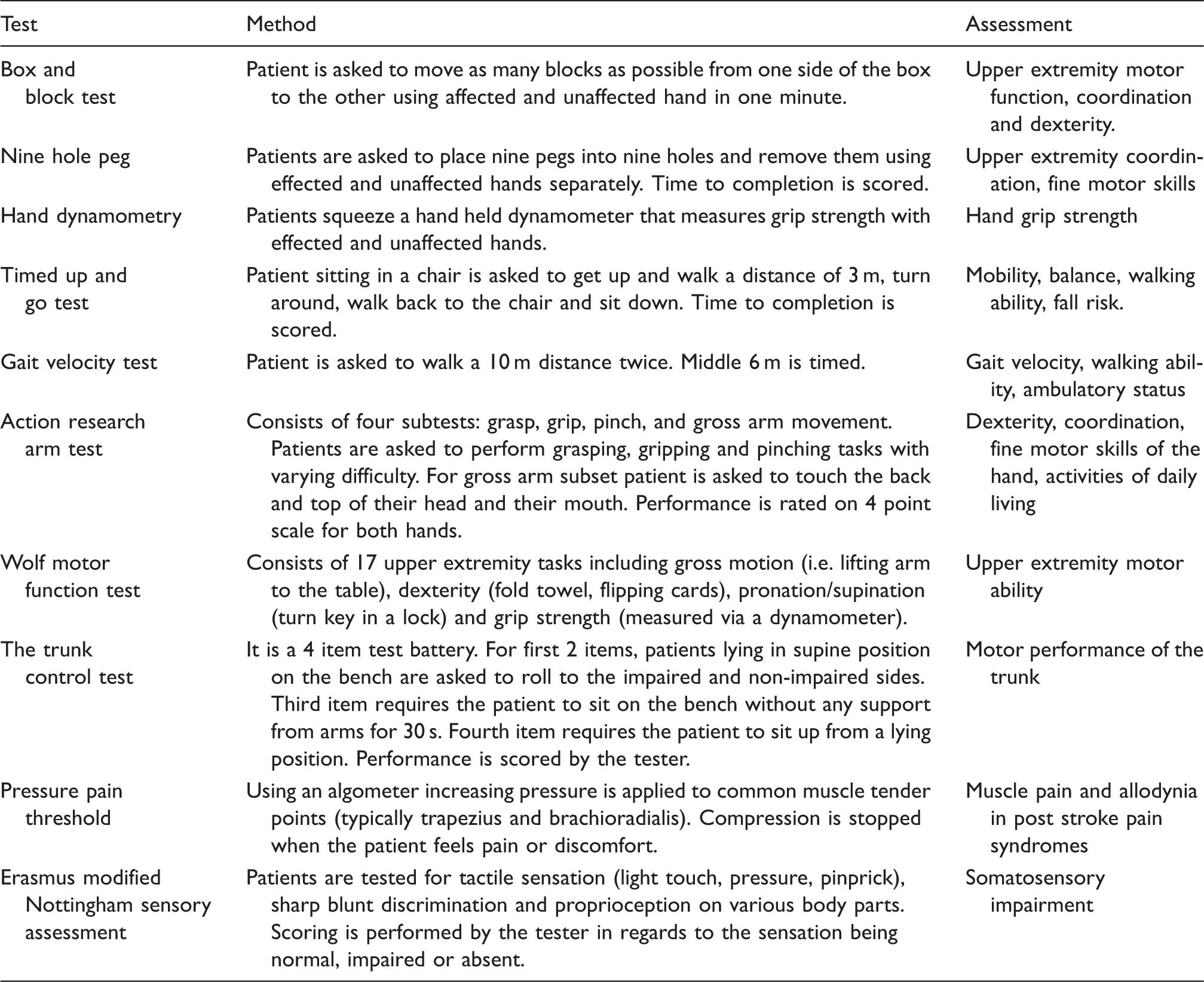
Clinical tests of sensorimotor function.
Preclinical assessments
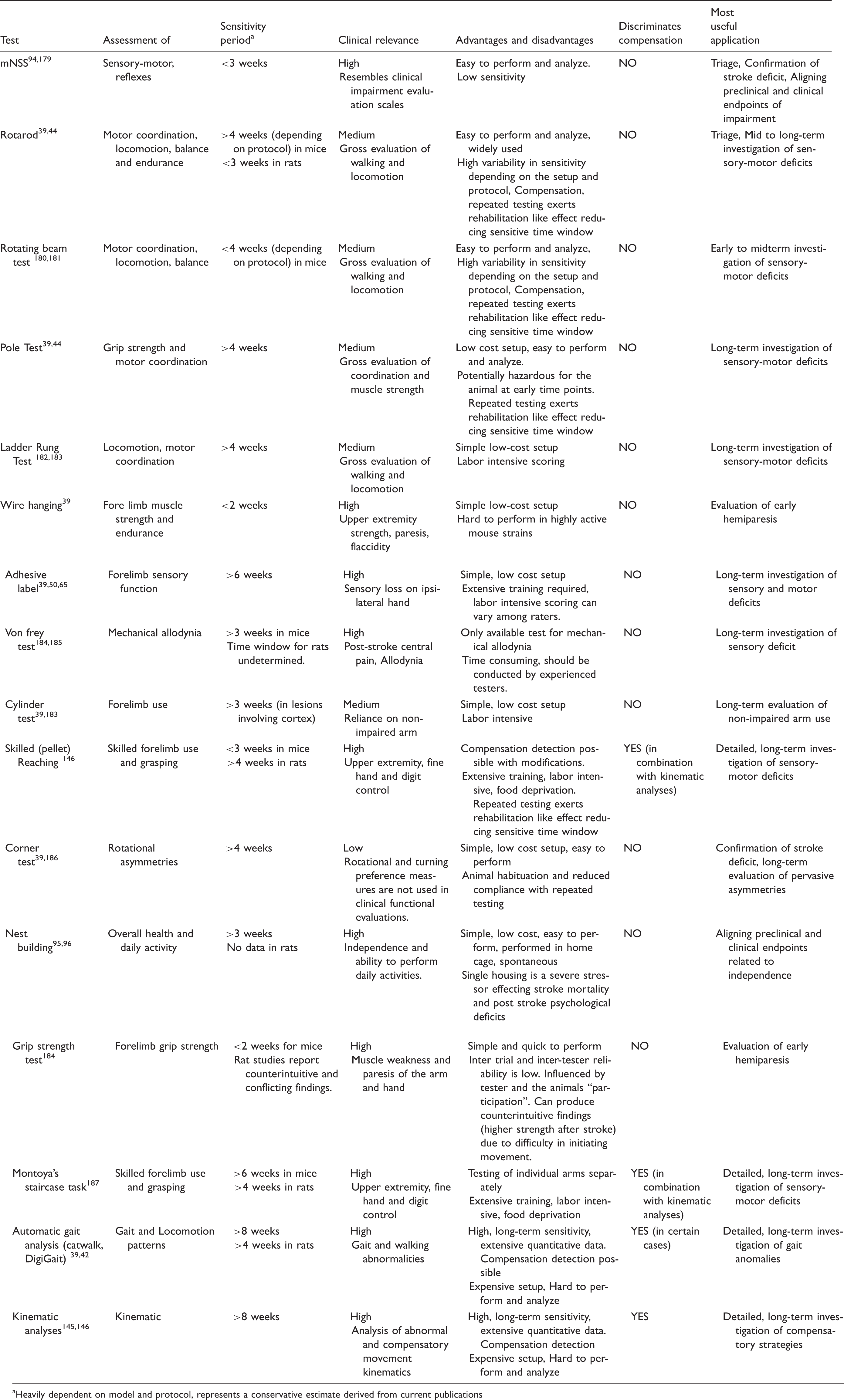
Behavioral tests used in rodent models of focal ischemia.
Heavily dependent on model and protocol, represents a conservative estimate derived from current publications
Functional recovery
Recovery in stroke patients
Patients surviving the initial high mortality phase of stroke experience some degree of spontaneous recovery within weeks. A significant portion of this recovery takes place within the first month and can continue up to six months post stroke. 101 Voluntary movements are observed in hemiplegic patients within the first week to month after stroke. 102 A significant amount of upper extremity motor recovery occurs in the first four weeks and reaches plateau levels at three months after stroke. 103 Depending on stroke severity, dexterity is regained in 41% to 78% of patients within six weeks. 104 However, spontaneous recovery is not homogeneous in terms of degree and rate for every type of neurological function. 105 Fine motor skills, such as picking up objects between the fingers and thumb, that involve smaller movements performed via wrists, hands, feet, fingers and toes take longer to recover compared to gross motor skills, such as walking or jumping, that involve large muscles of torso, arms and legs. Accordingly, proximal upper extremity recovery takes longer than upper extremity recovery.106,107 Language deficits continue to improve years after stroke. 108 Improvements in motor impertinence can be observed up to 12 months 109 and urinary incontinence can resolve as late as five months after stroke onset.
A similar heterogeneity is also observed in the percentage of patients that show improvement. Near full recovery is observed in 25 to 50% of patients depending on the assessment method used. 101 However, more conservative analyses reveal that only 15% of stroke survivors with initial paresis had complete recovery of both the upper and lower extremities. A majority of studies report no noteworthy improvement in approximately a quarter of patients.110,111 Interestingly, among the patients that experience recovery, the amount of improvement appears to be highly predictable within a given time frame. 112
Proportional recovery
“The proportional recovery rule,” which is derived from clinical studies, asserts that a stroke patient should recover 70% of his/her potential function within three months. For example, a patient that scores 36 out of the maximum 66 in a Fugl-Meyer scale for upper extremity function will recover to ((66 − 36) × 0.7)) + 36 = 57 points.
112
This rule has been validated for visuospatial neglect,
113
aphasia,
114
lower
115
and upper limb
116
motor recovery, as well as upper limb somatosensory processing recovery.
117
Interestingly, while patients with mild to moderate hemiparesis recover in line with proportional recovery rule (fitters), patients with severe hemiparesis (FMA score <20) do not fit the rule (non-fitters) and show little recovery (see Figure 1).
118
Absence of motor-evoked potentials
119
and significant damage to the corticospinal tract tissue predicts the non-fitter status.119–121 Proportional recovery observations imply that recovery is mainly a spontaneous biological process driven by remaining healthy tissue and current rehabilitation regimens have no significant effect on functional gains.
Fugl-Meyer scores of fitter and non-fitter patients at admission and three months. Fitter (blue) patient receives an FMA score of 36 at admission and from the maximum possible recovery score (black dashed line) of 30 recovers 70% (red dashed line) reaching a final score of 57. Non-fitter patient receives 19 at admission but does not recover according to the proportional recovery rule with minimal increase in FMA scores. Fitter and non-fitter patients differ in lesion size, corticospinal tract integrity and motor-evoked potential responses.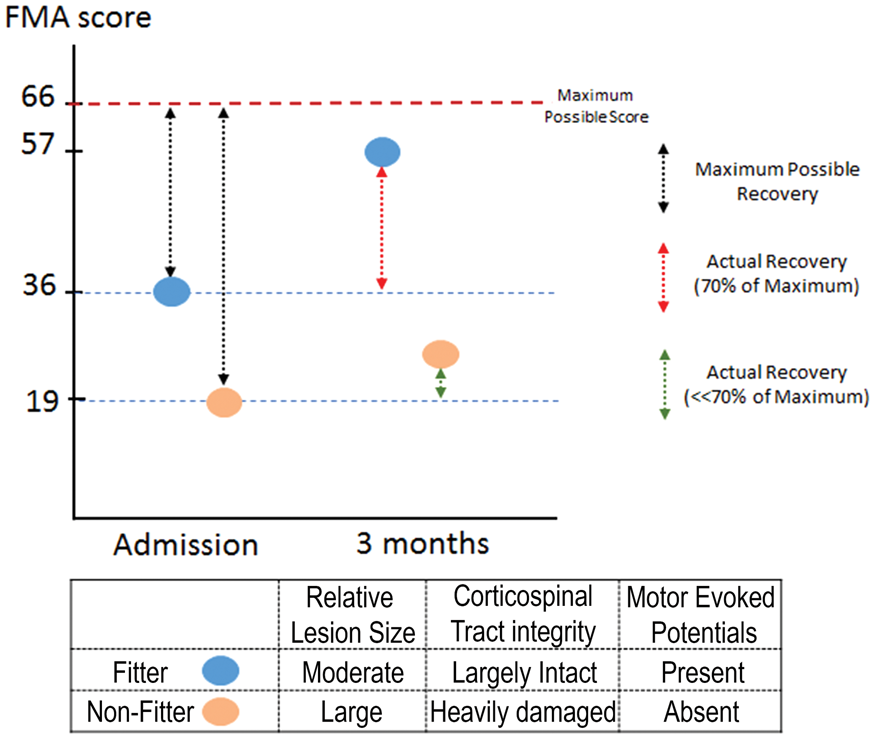
Recovery in preclinical models
Preclinical behavioral test for depression and anxiety.

The fact that a significant portion of the behavioral tests fail to distinguish between stroke and control within weeks suggests the possibility that the proportional recovery rule does not hold for animal models of stroke. Full recovery may simply be due to the limited sensitivity of the test. Indeed, a clear ceiling effect is apparent in tests such as the Rotarod. In high activity strains (e.g. C57), naive animals can easily achieve max score with brief training. Stroked animals with apparent postural anomalies also can achieve a max score in Rotarod, indicating a high susceptibility of the test to compensation. 126 Alternatively, for some tests, the deficit in lab animals might have been compensated by the untapped redundant capacity. Lab animals live in a constant state of deprivation in impoverished, confined cages that offer little stimuli. Compared to their wild cousins, lab rodents display extremely low motor and cognitive skills. While selective inbreeding is certainly a main contributor, constant deprivation might be lowering baseline skills in lab animals, which in turn is rapidly compensated for post stroke. Experiments utilizing “enriched” environments are needed to test whether baseline scores would improve in behavioral tests and the percentage of animals showing full recovery would be altered accordingly.
A number of behavioral tests, however, retain their sensitivity over long periods of time. Despite rapid early recovery, animals reach plateau levels below baseline in tests of skilled reaching, forelimb sensory function and rotational preference up to months after stroke. Whether this sub-baseline improvement fits the proportional recovery rule remains to be determined. Animal experiments investigating proportional recovery in stroke models are extremely scarce. In a series of recent papers, Jeffers and Corbett have investigated proportional recovery and relevant biomarkers. Using a retrospective analysis of data from 593 rats, they have demonstrated that proportional recovery occurs in approximately 30% of rats (fitters) with a similar ratio (62–70%) seen in human patients. 73 Approximately 62% of rats were non-fitters to the rule. Contrary to human observations, remaining animals declined over time. Infarct volume and initial impairment were predictive of recovery as well as fitter, non-fitter and decliner status. 127
Aligning clinical and preclinical functional evaluation methods for long-term recovery studies
Putting rehabilitation studies aside, there is a striking discrepancy between clinical and preclinical functional endpoints. Composite scores (such as BI, mRS) that combine multiple aspects of neurological functioning suffer from numerous shortcomings, including floor and ceiling effects and low responsiveness. As a result, they have been criticized as a contributing factor to translational failures. 66 The multimodal nature of stroke impairments and the fact that recovery occurs at different rates and to varying degrees further indicates that such scales are simply inadequate. Particularly, with the description of the proportional recovery rule in studies using the FMA, current trends in clinical studies favors its use as a primary functional outcome measure. Furthermore, it has been proposed that the proportional recovery rule should be utilized as a benchmark against which the efficacy of neurorestorative interventions can be tested. 112 Nevertheless, the multimodal nature of stroke and the promise of impairment-specific drug development potential will inevitably increase the adoption of impairment-specific tests in clinical trials. 128
How should preclinical research adjust to meet the upcoming changes in clinical evaluations? In terms of impairment-specific testing, the preclinical behavior arsenal is rich with sensitive tests. Clinically relevant deficits, such as gait abnormalities and forelimb, skilled reaching, and hind limb impairments should be the main focus of behavioral testing. Though it may sound controversial, we believe that “revisiting” and refining neuroscores to create a preclinical counterpart of the FMA is a worthwhile endeavor. Behavioral testing should be performed at baseline and post-stroke at multiple time points at least up to one month post-stroke. Post stroke behavioral data should be normalized to the baseline, and change against baseline, rather than the absolute numbers, should be used for statistics. An imminent need in the field is to determine whether proportional recovery is observed in animal models. Accordingly, preferred behavioral tests should ideally show chronic impairments that spontaneously recover and reach plateau levels below pre-stroke baseline and are compatible with the proportional recovery rule. 66
True recovery versus compensation
The concept of recovery and the role of compensation in stroke often leads to confusion. In most cases, the term “recovery” is used to define the restitution of damaged tissue and clinical improvements simultaneously. 129 This terminological ambiguity impairs the proper interpretation of clinical and preclinical results alike and impedes dissemination of knowledge among professionals from different disciplines. Recovery and compensation can occur at different levels. The proposed definitions by Levin et al. state that recovery at the neuronal level would represent “restoring function in neuronal tissue that was lost initially lost after injury” and compensation occurs when “neuronal tissue acquires a function that it did not have prior to injury.” In contrast, at the performance level, recovery is “restoring the ability to perform a movement in the same manner as it was performed before injury” and compensation is “performing an old movement in a new manner”. 129
Clinical findings
A considerable portion of the functional recovery observed in stroke patients is a result of compensatory behavioral strategies. It has been documented that more than half of the patients with severe upper extremity paresis achieve some degree of recovery only through compensation. 130 Similarly, gains in locomotor function are achieved through a combination of recovery and compensatory behaviors. 131 Compensation also accounts for a significant portion of rehabilitative training-induced functional improvements. 132 Interestingly, compensatory strategies at the cognitive level are also present and utilized by patients to make up for the lost capacity.133,134
Muscle weakness, reduced muscle activation and impaired muscle co-activation in stroke patients disrupts movement coordination. 135 As a result, coupling of elbow and shoulder movements is disturbed, and wrist stability, grasp control and grasp release are impaired, leading to decreased hand dexterity. 136 Thus, stroke patients rely on the non-paretic hand when executing unimanual tasks. During skilled reaching with the paretic hand, the cumulative effects of these impairments are observed as less direct trajectories, with slower and more variable movements.137,138 To compensate for instability, stroke patients fix hand and elbow positions and use trunk and scapula movements to aim for the target object. 136
Clinical scales and tests measure functional improvement by assessments of independence or task accomplishment with little regard as to how the task is performed. They are therefore unable to distinguish compensation. 129 Novel methods of gait analysis139,140 and kinematic analysis of upper141,142 and lower extremity131,143 enable precise identification of compensatory action patterns. However, these methods generally require expensive and sophisticated equipment, limiting their use in clinical studies.
Preclinical findings
Animals and humans display significant homologies in skilled reaching and grasping movement patterns. Post-stroke compensatory strategies employed are quite similar in humans and animals as well. Rodents, similar to human patients, rely on the unimpaired limb when performing unimanual tasks. 144 When paretic arm is used, rodents execute slower, aberrant movements with high variability.145,146 Compensatory movements of the trunk and head are used to control paw position and bring the head to the paw. 136
Behavioral evaluations in a great majority of preclinical studies evaluate recovery by utilizing gross latency scores or task completion success, making a distinction between recovery and compensation impossible. Nevertheless, kinematic analysis of locomotion and skilled reaching behavior in preclinical stroke models is feasible. Such methods not only enhance long-term sensitive analysis of function but enable the characterization of compensatory patterns, even in mild stroke models, both qualitatively and quantitatively.126,147 For example, utilizing a model of small phototrombotic cortical lesion, Lai et al. 146 reported persistent (up to 30 days) kinematic modifications in a skilled reaching task. A very important finding of the study was that when reaching success is taken as the endpoint, there was no statistical difference between sham and stroke animals by week 2. In a similar study, the beneficial effects of somatic cell therapy were demonstrated in a MCAo model in rats. While both showed some degree of recovery, improvements in control animals were a result of compensatory adjustments. Treated groups had improved more and “kinematics more closely resembled pre-stroke movement patterns”. 145 In a mouse model of MCAo, Qin et al. demonstrated enhanced motor recovery in animals carrying val66met polymorphism in the BDNF gene. Gait analysis of locomotion patterns revealed that enhanced motor “recovery” was a result of compensation in the ipsilateral hind limb. 42 Such examples demonstrate the power of automated gait and kinematic analyses in distinguishing true functional recovery and compensation.
Aiming for the optimal recovery
Improving functional independence of stroke patients is a primary goal of clinical practice. To this end, it has been argued that compensatory behaviors leading to altered movement patterns should be considered adaptive as long as there is functional improvement. 148 In addition, in rehabilitation settings, compensatory behavior in patients with severe disabilities is encouraged and reinforced. However, in certain conditions, compensatory behavior is maladaptive and leads to distorted joint positions. 149 Moreover, maladaptive compensation and learned misuse may hinder long-term recovery of healthy movement patters. Further clinical and preclinical research is needed to identify optimal time windows and means to improve true recovery over compensation. Gait and kinematic analyses of locomotion and skilled reaching offer long-term sensitivity and enable detection of compensatory patterns. While dedicated setups for such analyses exist, kinematic analyses can also be achieved by low cost means and should gain further adoption.
Neuroprotective interventions with recanalisation
Intravenous thrombolysis via tissue plasminogen activator (tPA) and endovascular thrombectomy are two potent treatment options for acute stroke that can significantly reduce stroke damage though recanalization (reopening of the occluded principal artery) and reperfusion (microvascular blood flow restoration). In clinical trials of neuroprotection, documentation of successful arterial canalization as an enrollment criteria has not been obligatory. 150 Typically, 10% of stroke patients are considered suitable for recanalization therapies. 151 Considering that only one-third of MCA occlusions and 10% of distal ICA occlusions recanalize, 152 certain comorbid conditions such as diabetes and carotid artery disease further reduce canalization rates, 153 and 25% percent of recanalizations do not lead to reperfusion, 154 it is safe to conclude that, particularly in earlier clinical trials, the number of patients that achieved successful reperfusion is quite limited. The low number of patients with reperfusion in clinical studies puts into question whether drugs reached adequate concentration in potentially salvageable tissue and indicates a drastic disparity between clinical trials and preclinical studies, in which a majority of the neuroprotective agents were primarily tested in transient focal ischemia models with complete reperfusion. It is important to note that numerous neuroprotective agents that show significant efficacy in transient models fail to show any effect in preclinical studies employing permanent occlusions. 155 As such, the differences between preclinical and clinical studies in reperfusion profiles are potentially a primary contributor to translational failures.
Earlier and wider access to tPA treatment, development of more potent thrombolytic agents and advances in endovascular treatment of acute stroke increased the success rate and number of patients receiving recanalization. This raises the possibility that when combined with successful recanalization, neuroprotective agents may exert their true potential observed in animal models and become a feasible treatment option.154,155 Particularly, intra-arterial delivery of putative neuroprotectants post-thrombectomy when and where they are most needed is a promising treatment strategy.
In this context, Savitz et al. proposed that preclinical research will need to address several lines of research questions: (1) Can a neuroprotectant extend the time window for thrombolysis or thrombectomy? (2) Does its efficacy differ with or without reperfusion? (3) Does the route of delivery alter its efficacy? (4) Does it ameliorate reperfusion injury? Sufficiently addressing such research questions will create considerable challenges in model selection, experimental design and especially behavioral evaluation.
It has to be expected that in such experimental studies, the effect size in any given behavioral evaluation would be considerably lower compared to a typical neuroprotection study. Researchers should utilize conservative effect size estimates while calculating their sample sizes, which will increase the number of animals required but significantly increase the sensitivity of their evaluations. Behavioral test selections should include separate tests for motor, sensory and cognitive function to screen for improvements in all of these domains. Kinematic analyses that offer superior responsiveness both at acute and chronic phases should be employed.
Post-stroke neuropsychiatric disturbances
Neuropsychiatric disturbances in stroke patients
Cerebrovascular lesions can lead to emotional disturbances including depression, anxiety, emotional incontinence, fatigue, mania, and anger proneness.156–158 Post-stroke depression (PSD) is the most frequent and debilitating mood disorder observed in stroke survivors and has profound effects on patient recovery and well-being. PSD increases mortality by up to 10 times, slows recovery and predicts worse functional and cognitive outcome. 159 Depressed mood increases limitations on daily activity and rehabilitation efforts. 160 In addition, depression after stroke increases hospitalization time and healthcare costs. 161 Interestingly, depressive symptoms tend to increase in the chronic phase of recovery162,163 and persist for over three years in 29% of elderly patients. 164 Taken together, PSD is a chronic, frequent and severe complication with significant negative effects on the patient and healthcare efforts.
In clinical settings, post stroke depression diagnosis and follow-up evaluations are performed using structured clinical interviews using depression scales. There are a number of scales validated for depression diagnosis and clinical studies vary in terms of the preferred method. Scales commonly used in stroke clinical trials include the Hamilton depression and anxiety scales, Beck depression index, patient health questionnaire-9, and Montgomery–Asberg Depression Rating Scale. 165 Due to differences in study settings and criteria reported, PSD prevalence rates vary widely and range between 5% and 79%. A detailed examination of the literature indicates that the mean prevalence rate was 24.0% for major depression and 23.9% for minor depression after stroke. 166 Two recent meta-analysis studies reported pooled prevalence rates of 29% 167 and 31% 168 at any time up to five years post stroke. The percentage of patients who had one or more depressive episodes within five years following stroke ranged from 39% to 52%. 167 In addition, approximately 30% of stroke patients display anxiety, and a high percentage of these patients show comorbidity with PSD.
Neuropsychiatric disturbances in animal models of stroke
Studies modeling PSD and other mood disorders are scarce. While global ischemia models induce robust PSD and anxiety-like behaviors,169,170 findings have been inconsistent in focal ischemia models. Various reports describe the “spontaneous” evolution of anxious or depressive phenotypes after MCAo in rodents. An early study by Winter et al. has reported anxiety after left MCAo. Interestingly, no sign of anxiety was observed in animals with right MCAo. 171 The same group reported anhedonia, depressive-like behavior and anxiety 13 weeks after left strokes with the right MCAo group exhibiting hyperactivity as the only behavioral alteration. 172 Conversely, using the sucrose consumption test, an anhedonic phenotype in animals with right MCAo at two weeks after stroke was reported by Craft et al. 173 In a rat model of permanent MCAo, Ifergane et al. have reported PSD-like phenotype in a wide battery of tests, including the two-way shuttle avoidance task. Importantly, using cluster analysis, they demonstrated that about 56% of stroked animals developed significant behavioral disruptions associated with PSD. 174 Others have demonstrated depressive phenotypes in the forced swim test can be further exacerbated by chronic mild stress exposure, which was reported in animals with left MCAo. 175 In line with that study, numerous groups have employed long-term stress exposure after stroke to ensure robust and replicable PSD phenotypes. 176 Housing conditions also seem to affect PSD development. Animals with right MCAo that displayed reduced immobility in the forced swim test showed higher immobility as compared to controls when housed in isolation. 177 Age can be a predicting factor for PSD. Buga et al. 178 reported that MCAo leads to depressive behaviors only in aged rats.
PSD as a major focus of preclinical research
Despite growing interest in the debilitating consequences of PSD in the last two decades, PSD and its pathophysiology have been grossly understudied in stroke research. Clinical studies are narrow and animal models require further refinement. Our understanding of PSD pathophysiology is thus limited and complicated by conflicting findings from clinical and experimental studies. Importantly, PSD assessments are rarely included in clinical studies of long-term functional recovery. Given the reliability and ease of administering and depression scales, this represents a missed opportunity. We believe that PSD should be one of the primary targets of therapy in clinical and preclinical neurorestorative studies.
Closing remarks
Closing the gap between clinical and preclinical studies requires adjustments in study design on both sides. Early clinical studies have been heavily criticized for very broad inclusion criteria, lack of patient stratification and use of imprecise global outcome measures. Stroke symptoms are multimodal and there is considerable difference between different neurological functions in terms of degree and rate of recovery. As such, composite scales of global outcome measures are not able to offer enough sensitivity and do not address the broad spectrum of post-stroke impairments and their variations in recovery. 128 Notably, motor-weighted composite scales are likely to miss improvements in neglect, aphasia, cognitive function and emotional disturbances. The clinical test arsenal has well-established modality-specific tests that have been validated and successfully used, particularly in rehabilitation studies. Clinical studies of neurorestorative interventions should employ such assessments – including cognitive and emotional – to align with preclinical studies and achieve greater sensitivity.
The preclinical behavior models can mimic all human functional deficits. There are, however, inconsistencies between the lesion and functional deficit characteristics between preclinical and clinical studies. Preclinical researchers should therefore try to match lesion and impairment characteristics of the clinical population. This requires detailed descriptions of the patient data from clinical studies. Behavioral tests utilized in preclinical models offer enough sensitivity for long-term analysis. Test selection should be done by taking the relevant clinical impairments into account, and testing should be performed repeatedly at least up to a month after stroke. Proportional recovery observation has far reaching implications on rehabilitation and recovery studies. Addressing whether proportional recovery is consistently present in animal models among various functional domains is of significant importance. In studies combining recanalization with neuroprotective treatments, utilizing conservative effect size estimates and having large experimental group sizes are of utmost importance for demonstrating potential individual and synergistic beneficial effects of these treatments. Finally, PSD affects a large number of stroke survivors, but the number of studies focusing on the pathophysiology of PSD is very limited. The stroke field can benefit greatly from studies focusing on establishing reliable animals models of PSD.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
