Abstract
Leukocytes are systematic inflammation indicators related to stroke prognosis and can exhibit large dynamic waves before and after recombinant tissue plasminogen activator (r-tPA) therapy. However, additional evidence is needed to determine the prognostic significance of various leukocytes including both static and dynamic data among patients who underwent r-tPA therapy. A total of 251 patients treated with r-tPA were included; their leukocyte data were collected at two time points, and patients were followed up for three months. Analysis revealed the following findings. (i) Patients with hemorrhagic transformation (HT) and unfavorable outcomes had a higher level of leukocytes after r-tPA therapy (leukocyte count (adjusted OR (aOR) 1.191 for HT and 1.184 for unfavorable outcomes), neutrophil count (aOR 1.215 and 1.214), neutrophil-to-lymphocyte ratio (NLR; aOR 1.084 and 1.091)) and larger dynamic leukocyte changes. (ii)Among all leukocytes, the NLR after r-tPA administration demonstrated the strongest correlation with HT and unfavorable outcomes. (iii) Patients with an NLR ≥ 3.322 had a 3.492-fold increased risk for HT, and those with an NLR ≥ 5.511 had a 3.024-fold increased risk for functional outcomes. Overall, this study shows that leukocytes, especially leukocyte count, neutrophil count and the NLR, are independently associated with HT and functional outcomes in stroke patients.
Introduction
Intravenous thrombolysis with recombinant tissue plasminogen activator (r-tPA) administered within 4.5 hours of stroke onset is the approved and effective method for increasing and improving survival rate and quality of life after acute ischemic stroke (AIS). 1 Nevertheless, higher risks of cerebral hemorrhage 2 and neuroinflammation 3 are associated with intravenous thrombolysis (IVT) treatment due to the toxicity of r-tPA and reperfusion injury, thereby rendering this life-saving treatment a double-edged sword.
To date, evidence has demonstrated that numerous parameters, including leukocyte count, neutrophil count, lymphocyte count, and neutrophil-to-lymphocyte ratio (NLR), may play a crucial role in stroke progression. Elevated neutrophils are positively related to the risk of symptomatic intracerebral hemorrhage (sICH), death, and functional outcomes after r-tPA administration among stroke patients. 4 Similarly, there is growing evidence that an elevated NLR served as a well-documented predictor for hemorrhagic transformation (HT), 5 early neurological outcomes, 6 morbidity and mortality 7 after treatment with mechanical thrombectomy or r-tPA therapy. A number of studies have confirmed that neutrophils can aggravate the impairment of the blood–brain barrier (BBB), be absorbed by endothelial cells and be used in the basement membrane via the release of matrix metalloproteinase–9 (MMP-9),8,9 thus leading to an incremental risk of HT and mortality after r-tPA therapy. 10 Furthermore, endothelial cells can release some proinflammatory mediators (e.g., TNF-α, IL-1 and IL-6), which are inclined to aggravate injuries to the brain tissue. 11 It has been reported that inflammatory mediators initiate the breakdown of the BBB, followed by the invasion of the brain parenchyma through inflammatory cytokines and chemokines.12,13
Thus, elucidating the prognostic role of leukocytes in AIS patients is important for developing more targeted interventions and improving the efficacy of r-tPA. Previous studies explored the relationship between leukocytes and clinical outcomes. Nevertheless, most of them failed to consider the contribution of leukocytes to clinical outcomes in patients treated with r-tPA. Additionally, most previous studies measured leukocytes at a single time point and were unable to examine dynamic changes in explored leukocytes; therefore, these studies did not provide a comprehensive reflection of patient conditions. Furthermore, prior studies did not compare leukocyte subtypes, both static and dynamic, to determine the best predictors of clinical outcomes. Hence, this study was undertaken to elucidate the relationship between leukocytes and clinical outcomes and to identify which leukocyte subtypes, including both static and dynamic data, best predicted HT and functional outcomes in patients who underwent r-tPA treatment.
Materials and methods
Subjects and setting
This study was conducted with data that were prospectively collected from patients treated with IVT at the First Affiliated Hospital of Wenzhou Medical University. From December 2017 to September 2021, a total of 366 patients with IVT treatment (0.9 mg/kg up to a maximum of 90 mg, 10% of the dose as a bolus followed by a 60-minute infusion of the remaining dose) were examined within 4.5 h of stroke. Five patients with intravenous therapy on urokinase, one patient who underwent bridging therapy, six patients who experienced endovascular thrombectomy in addition to intravenous thrombolysis, 57 patients without information on the HT and three-month mRS scores, and 46 patients with a lack of available information regarding leukocytes were excluded. Ultimately, 251 patients were eligible for inclusion in this study, and written informed consent was obtained from all subjects (Supplementary Figure I). The study protocol was approved by the Ethics Committee of the First Affiliated Hospital of Wenzhou Medical University and performed in accordance with the Helsinki Declaration.
Data collection
The following laboratory data gathered included: (1) demographic characteristics (age and sex); (2) clinical characteristics (National Institutes of Health Stroke Scale (NIHSS) score, blood pressure, onset-to-treatment time, stroke subtype, history of smoking and drinking alcohol); (3) medical history (hypertension, diabetes mellitus, dyslipidemia, previous stroke, patent foramen ovale and atrial fibrillation); (4) blood biochemistry, including leukocytes (leukocyte, neutrophil, lymphocyte and NLR), platelets, blood glucose, INR, creatinine and alanine aminotransferase (ALT).
Within the first 4.5 h of stroke onset, patients underwent emergent neuroimaging with carotid ultrasonography and computed tomography (CT)/magnetic resonance imaging (MRI). After IVT treatment, CT and MRI were reassessed to assess the presence of AIS complications. The etiology of AIS was determined based on the TOAST criteria. 14 Additionally, stroke severity was evaluated using the NIHSS score at admission, after treatment with IVT and at discharge. All patients underwent venous blood sampling for leukocyte count, neutrophil count and lymphocyte count before r-tPA therapy (within one h) and after r-tPA therapy (within 24 h). The XE-2100 automated hematology analyzer (Sysmex Corporation) was used to analyze leukocytes. There was a significant difference between leukocyte subtypes pre-thrombolysis and post-thrombolysis. Other blood-related data, such as platelets, blood glucose, INR, creatinine and ALT, was performed at admission (within one h before r-tPA therapy). We calculated ΔNLR (the absolute dynamic variation in NLR levels) as follows: NLR after IVT minus baseline NLR. Additionally, R NLR (the relative dynamic variation in NLR levels) was calculated as follows: ΔNLR/baseline NLR. Other types of leukocytes were calculated using the same formulas.
Clinical assessment
In accordance with the recommendation of the European Cooperative Acute Stroke Study (ECASS), 15 HT can be categorized as parenchymal hematoma (PH) or hemorrhagic infarction (HI). HI is defined as small petechiae along the periphery of the infarct (HI1) or as confluent petechiae within the infarcted area without any space-occupying effect (HI2). PH is defined as bleeding in <30% of the infarcted area with some mild and slight space-occupying effect (PH1) or as bleeding in ≥30% of the infarcted area with abundant space-occupying effect or any hemorrhagic lesion outside the infarcted area (PH2). The modified Rankin Scale (mRS) was utilized to evaluate functional outcomes at three months following symptom onset. 16 An mRS score of 0–2 represented a favorable outcome, whereas an mRS score of 3–6 represented a poor outcome.
All patients underwent a minimum of three-month follow-up via telephone, email, questionnaires and outpatient reviews collected by certified assessors. Trained physicians were blinded to the clinical data and performed assessments as part of their clinical duties.
Statistical analysis
The normality of the data was assessed using the Shapiro–Wilk normality test. The equality of variance between two datasets with normal distributions was examined by the F test. Normally distributed continuous variables were expressed as the mean ± standard deviation (mean ± SD) and compared using the T test. Nonnormally distributed continuous variables were expressed as median and quartile ranges (median, IQR) and compared using the Mann–Whitney U test. Categorical variables were expressed as frequencies and percentages (N, %) and compared using the chi-square test. The independent effect of leukocytes and their variations on clinical outcomes was investigated using logistic regression modeling, adjusted for potential confounding variables (including age, sex, atrial fibrillation, hypertension, diabetes mellitus, blood glucose, platelets, NIHSS scores and onset-to-treatment time at admission). The receiver operating characteristic (ROC) curve was utilized to compare the predictive power of each covariate for HT and three-month mRS and to determine the optimal cutoff values at which the specificity and sensitivity were the highest. Spearman’s ρ, a measure of the correlation between a set of predictors and specific outcomes, was also used to test the predictive value of markers. The Integrated Discrimination Improvement (IDI) and the Net Reclassification Index (NRI) were calculated to evaluate the value of adding the NLR after r-tPA treatment to the conventional model. Restricted cubic spline curves with three knots (defined at the 10th, 50th, and 90th percentiles) for HT and five knots (defined at the 5th, 35th, 65th, and 95th percentiles) for three-month mRS were further plotted to provide more precise estimates and examine the linear and nonlinear relationships. Subgroup analysis was also conduced to further validate the reliability and practicability of the results, and the multiplicative interaction was tested with interaction terms by the likelihood ratio test. A two-tailed p < 0.05 was deemed to be statistically significant. All statistical analyses were conducted and all plots were developed using SPSS version 25.0 software (SPSS Inc., Chicago, IL, United States) and R version 4.1.0 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline characteristics and study outcomes
The clinical characteristics of individuals with r-tPA treatment are detailed in Table 1. Among all patients with IVT, HT occurred in 45 (17.9%) participants, and 83 (33.1%) patients presented an unfavorable outcome (mRS > 2) at three months. The HT rate observed herein was higher than that of other large r-tPA trials, which reflected the real-world setting of our study and the characteristics of the local area in China. In individuals attributable to HT, the mean age was 69 years, ranging from 60 to 79 years, and 59.5% of the patients were male. Compared with patients without HT, patients with HT had higher NIHSS scores, blood glucose levels, INR, proportion of atrial fibrillation as well as elevated leukocyte count, neutrophil count and NLR after IVT therapy, whereas patients with HT had lower levels of platelets at admission, lower lymphocyte count after IVT and a lower likelihood of smoking (all p < 0.05). No significant difference in leukocytes at admission was found in individuals with regard to HT. Apart from atrial fibrillation, previous stroke and NIHSS scores, the majority of the demographic characteristics between the included and excluded individuals were not different (Supplementary Table I). The distribution of leukocyte, neutrophil, lymphocyte and NLR counts at admission and after IVT is shown in Figure 1. The leukocyte count was unable to differ between pre-thrombolysis and post-thrombolysis (p = 0.249). The neutrophil count (p = 0.003) and NLR (p < 0.001) were higher after r-tPA treatment than at admission, whereas the lymphocyte count (p < 0.001) was lower after r-tPA than at admission.
Demographic and clinical information stratified by the presence of HT.
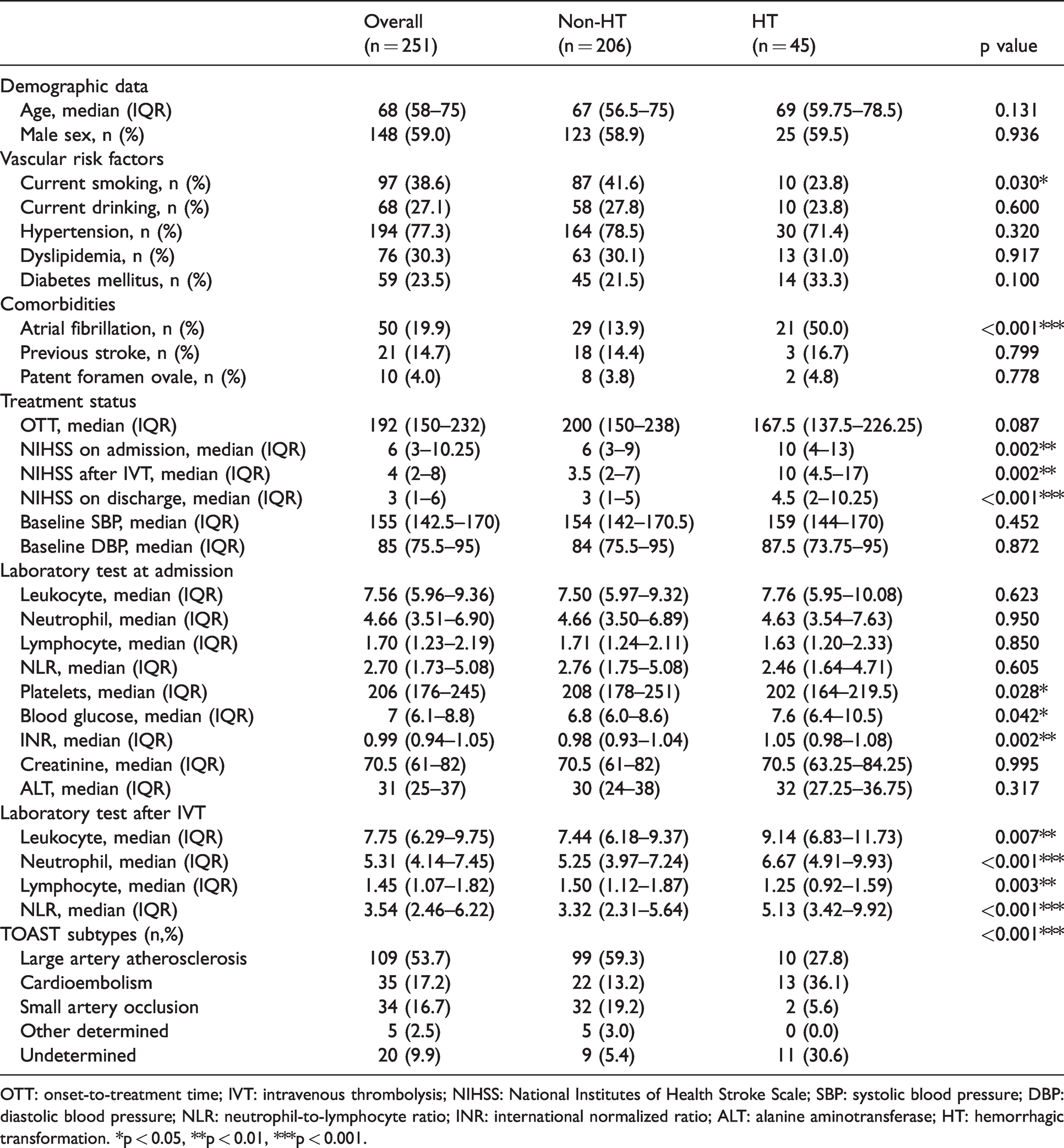
OTT: onset-to-treatment time; IVT: intravenous thrombolysis; NIHSS: National Institutes of Health Stroke Scale; SBP: systolic blood pressure; DBP: diastolic blood pressure; NLR: neutrophil-to-lymphocyte ratio; INR: international normalized ratio; ALT: alanine aminotransferase; HT: hemorrhagic transformation. *p < 0.05, **p < 0.01, ***p < 0.001.
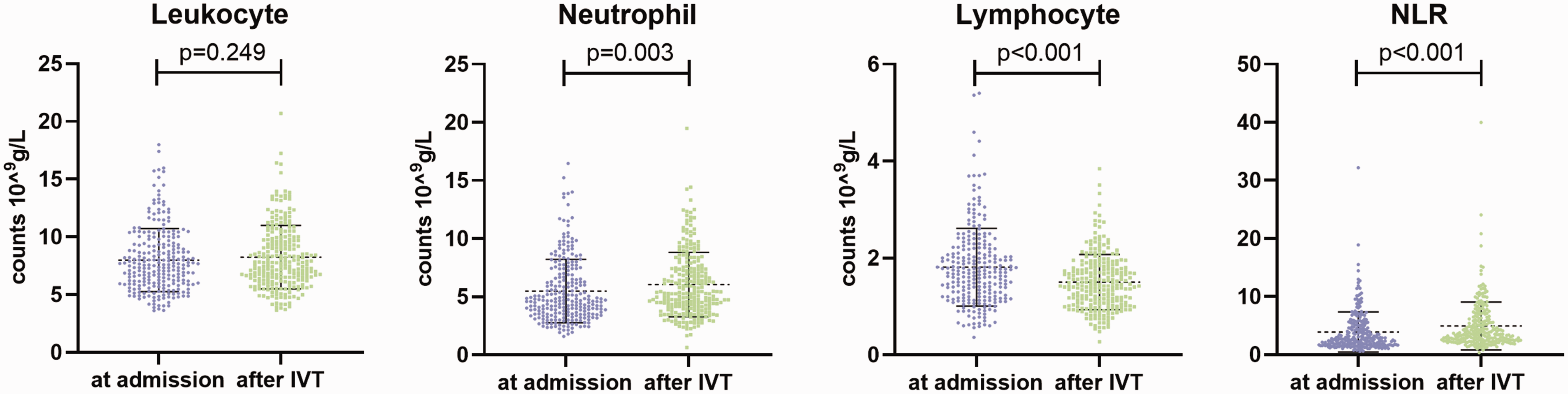
The distribution of leukocyte, neutrophil, lymphocyte and NLR counts at admission and after IVT. NLR: neutrophil-to-lymphocyte ratio; IVT: intravenous thrombolysis.
Leukocytes and hemorrhagic transformation
Patients who developed PH were prone to suffering higher leukocyte count (p < 0.001), neutrophil count (p < 0.001) and NLR (p < 0.001) as well as lower lymphocyte count (p = 0.003) after rt-PA treatment than non-HT individuals (Figure 2). However, there was no difference between those with HI and non-HT individuals. Similar results were found with respect to the absolute dynamic changes of leukocytes. The logistic regression for the relationship between leukocytes and HT is shown in Table 2. Apart from leukocytes at admission, the other parameters were strongly associated with the incidence of HT in univariate logistic regression analysis. The leukocyte count (adjusted OR (aOR), 1.191 (95% CI, 1.041–1.363), p = 0.011), neutrophil count (aOR, 1.215 (95% CI, 1.060–1.392), p = 0.005) and NLR (aOR, 1.084 (95% CI, 1.002–1.174), p = 0.046) after IVT, along with potential confounders, were independently correlated with the incremental odds of HT in the multivariate analysis. Notably, dynamic changes in absolute leukocytes, including neutrophil count, lymphocyte count and the NLR, were also identified as independent predictors for the incidence of HT (aOR, 1.171 (95% CI, 1.020–1.344), p = 0.025; aOR, 0.564 (95% CI, 0.349–0.912), p = 0.019; aOR, 1.100 (95% CI, 1.005–1.205), p = 0.038, respectively). Similarly, consistent results could be discovered with regard to relative changes in leukocytes. The OR values of leukocytes with the occurrence of PH remained remarkable; however, in regard to HI, the results were jointly insignificant, manifesting that leukocytes were not related to the risk of HI (Supplementary Table II).
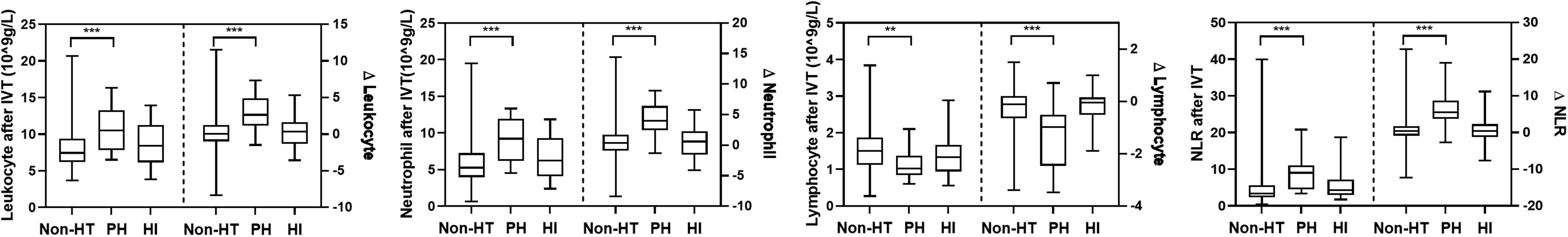
Comparison of patients without HT and with different types of HT among leukocytes parameters. NLR: neutrophil-to-lymphocyte ratio; HT: hemorrhagic transformation; PH: parenchymal hemorrhage; HI: hemorrhagic infarct. ***p < 0.001, **p < 0.01, *p < 0.05.
Clinical characteristics of patients, stratified by the presence of clinical outcomes.
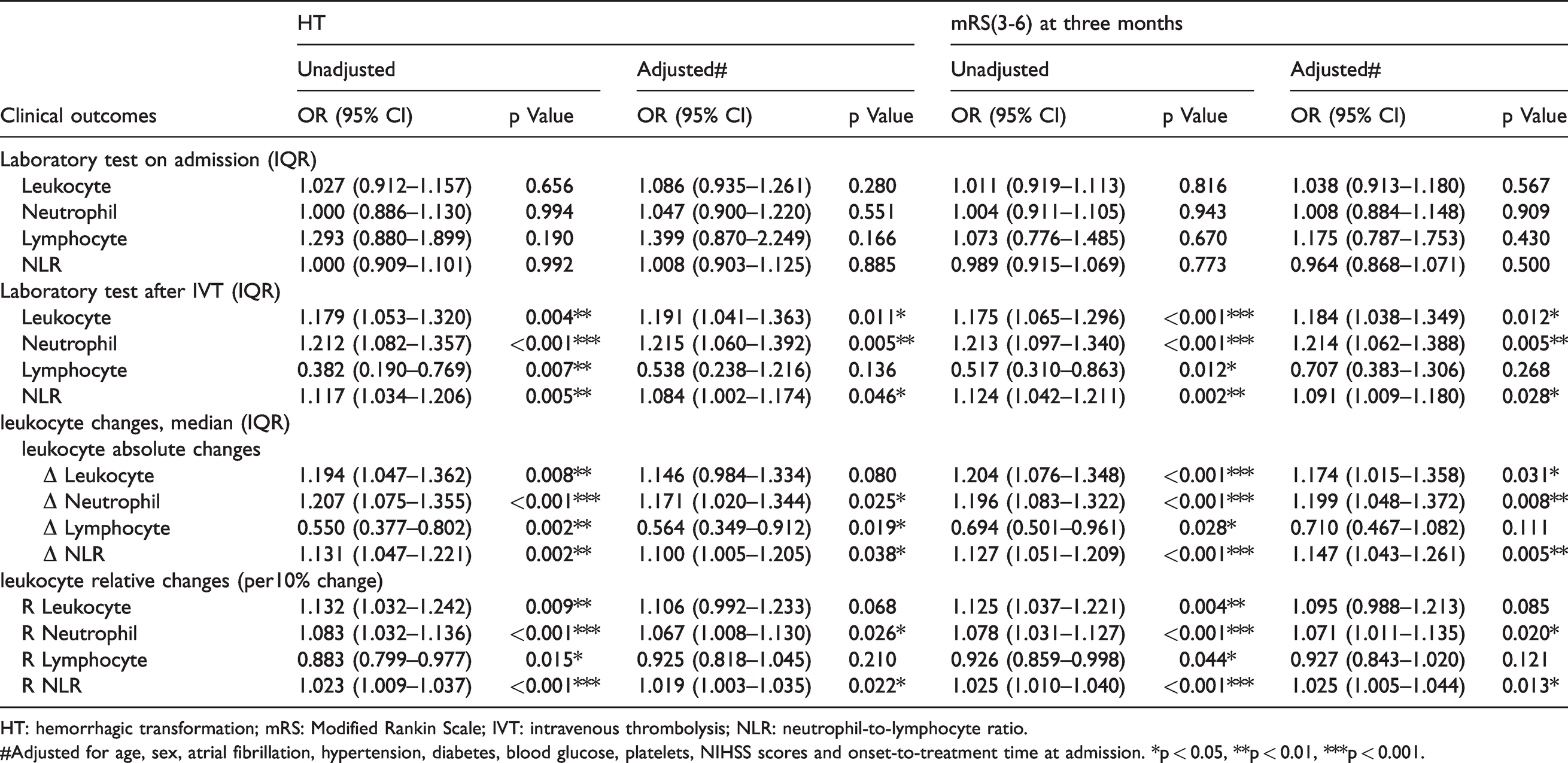
HT: hemorrhagic transformation; mRS: Modified Rankin Scale; IVT: intravenous thrombolysis; NLR: neutrophil-to-lymphocyte ratio.
#Adjusted for age, sex, atrial fibrillation, hypertension, diabetes, blood glucose, platelets, NIHSS scores and onset-to-treatment time at admission. *p < 0.05, **p < 0.01, ***p < 0.001.
Leukocytes and 3-month functional outcomes
There existed no difference between leukocytes at admission and functional outcomes. The leukocyte count (p = 0.014), neutrophil count (p = 0.003) and NLR (p < 0.001) after IVT were higher in patients with a three-month mRS score of 3–6, whereas a decrease in lymphocyte count (p = 0.009) after r-tPA therapy was observed in patients who developed unfavorable functional outcomes (Supplementary Figure II). Furthermore, the same results could be found in the dynamically changed leukocytes. Interestingly, the increases in the leukocyte count, neutrophil count and NLR after IVT were significantly related to unfavorable outcomes in both the unadjusted and adjusted models (Table 2; aOR, 1.184 (95% CI, 1.038–1.349), p = 0.012; aOR, 1.214 (95% CI, 1.062–1.388), p = 0.005; aOR, 1.091 (95% CI, 1.009–1.180), p = 0.028, respectively). The absolute variations in the leukocyte count (aOR, 1.174 (95% CI, 1.015–1.358), p = 0.031), neutrophil count (aOR, 1.199 (95% CI, 1.048–1.372), p = 0.008) and NLR (aOR, 1.147 (95% CI, 1.043–1.261), p = 0.005) were discovered to predict an increased risk of mRS score of 3–6. Similar results could also be found for the relative changes in leukocytes.
Predictive values of leukocytes for the clinical outcomes
Among all potential parameters examined herein, the NLR after r-tPA treatment had the highest predictive value, with an area under the curve (AUC) value of 0.691 and a Spearman rho of 0.247 for the occurrence of HT. The AUC and Spearman rho values for the three-month mRS score of 3–6 were 0.649 and 0.243, respectively (Supplementary Figure III; Supplementary Table III). Additionally, the optimal cutoff values for the NLR after IVT to predict HT and poor outcomes were 3.322 (with a sensitivity of 81.0% and a specificity of 50.2%) and 5.511 (with a sensitivity of 49.4% and a specificity of 79.8%), respectively. When NLR was classified as a categorical variable, it remained independently correlated with the occurrence of HT (aOR, 3.492 (95% CI, 1.371-8.890), p = 0.009) and unfavorable outcomes (aOR, 3.024 (95% CI, 1.493–6.125), p = 0.002) after adjusting for potential confounders. Furthermore, the new model, the addition of NLR after r-tPA treatment based on the conventional model (including age, sex, the history of atrial fibrillation, hypertension and diabetes mellitus, blood glucose, platelets, NIHSS scores and onset-to-treatment time at admission), was found to statistically ascend the discriminatory power and the risk reclassification for HT, as observed by the category-free NRI (p = 0.020) of 39.3% and IDI (p = 0.039) of 2.1% (Table 3). The ROC curves for the conventional model and the new model were also constructed (Supplementary Figure IV) with the AUCs values of 0.7967 and 0.8270 for the HT respectively, which indicated a slightly improved discrimination ability. What’s more, the AUCs values of two models to predict functional outcomes were 0.7322 and 0.7623, respectively.
Reclassification and discrimination statistics for outcomes by leukocytes after IVT.
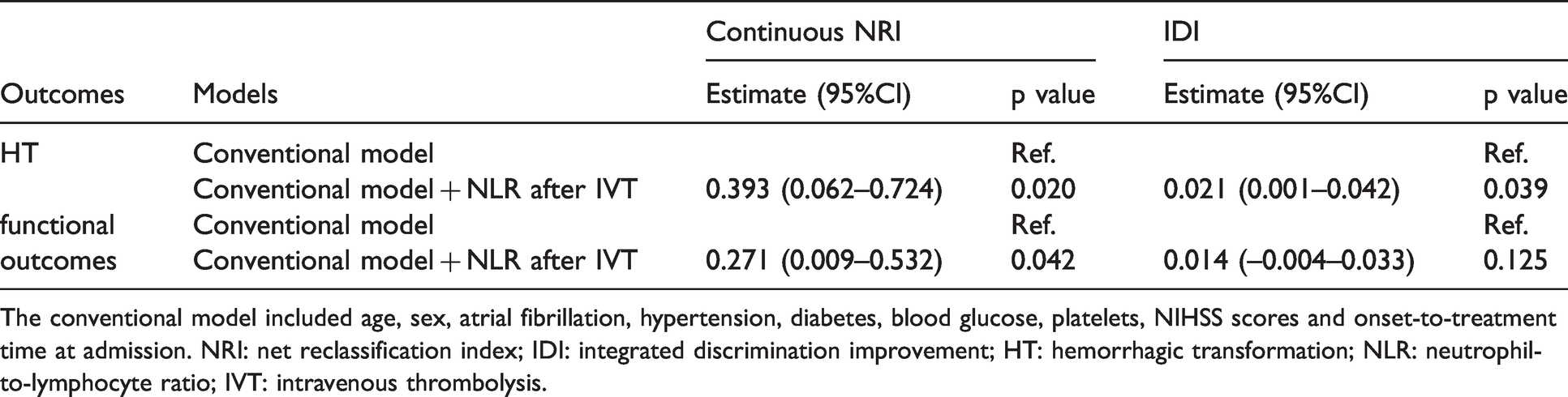
The conventional model included age, sex, atrial fibrillation, hypertension, diabetes, blood glucose, platelets, NIHSS scores and onset-to-treatment time at admission. NRI: net reclassification index; IDI: integrated discrimination improvement; HT: hemorrhagic transformation; NLR: neutrophil-to-lymphocyte ratio; IVT: intravenous thrombolysis.
Nonlinear correlation and subgroup analysis for the clinical outcomes
The dose–response relationships between an elevated NLR after IVT and the higher risk of HT as well as poor functional outcomes were further manifested with restricted cubic spline regressions (Figure 3; p for linearity = 0.024 for HT; p for linearity = 0.015 for poor outcomes; p for nonlinearity = 0.050 for both clinical outcomes). The results of subgroup analyses stratified by age (<60 or ≥60 years), sex (male or female), NIHSS scores at admission (<4 or ≥4), cigarette smoking and alcohol consumption, history of atrial fibrillation and diabetes mellitus are outlined in Figure 4. Strikingly, an increased NLR level after IVT was discovered to be associated with an increased risk of HT as well as unfavorable outcomes, especially in patients with no history of alcohol use (both p = 0.003) and an NIHSS score of four or greater (p = 0.002 for HT; p < 0.001 for poor outcomes).
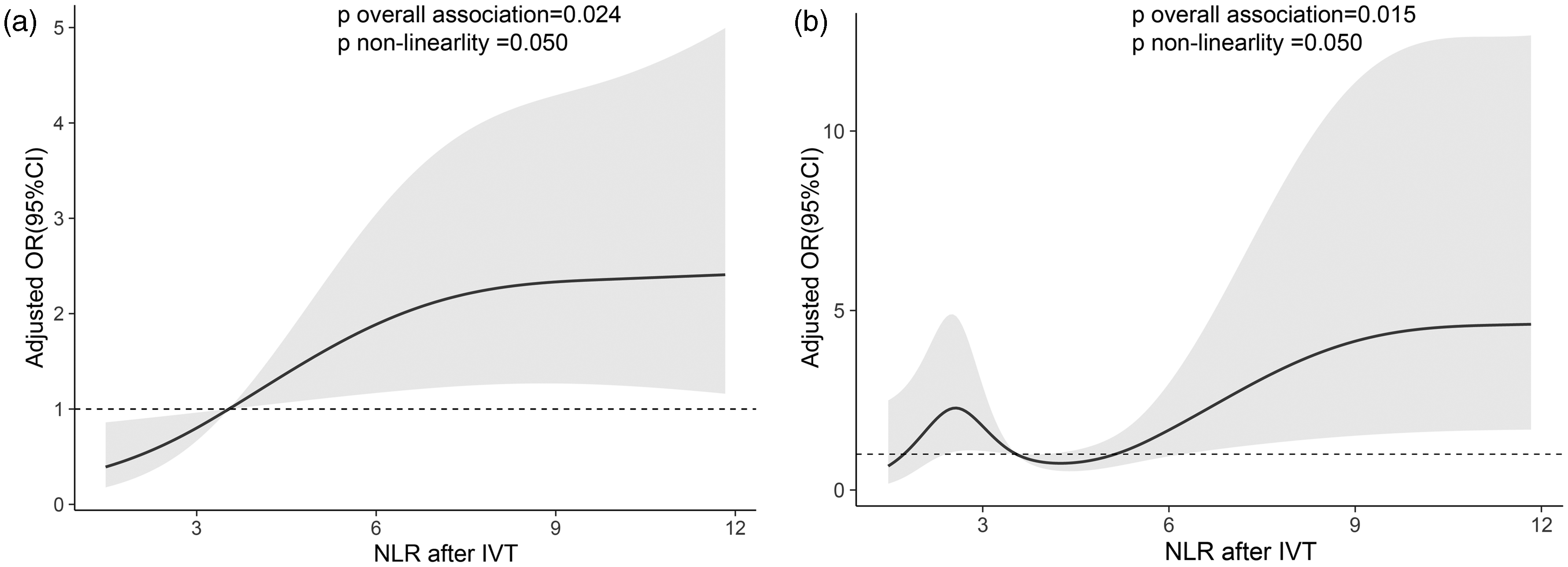
Adjusted association of NLR after IVT with (a) HT and (b) poor functional outcomes. Multiple spline regression analyze with three knots is used in HT and five knots is utilized in unfavorable functional outcomes. The solid line indicates odds ratio, while the shadow indicates 95% CI. Data is adjusted for age, sex, atrial fibrillation, hypertension, diabetes, blood glucose, platelets, NIHSS scores and onset-to-treatment time at admission. HT: hemorrhagic transformation; NLR: neutrophil-to-lymphocyte ratio; IVT: intravenous thrombolysis.
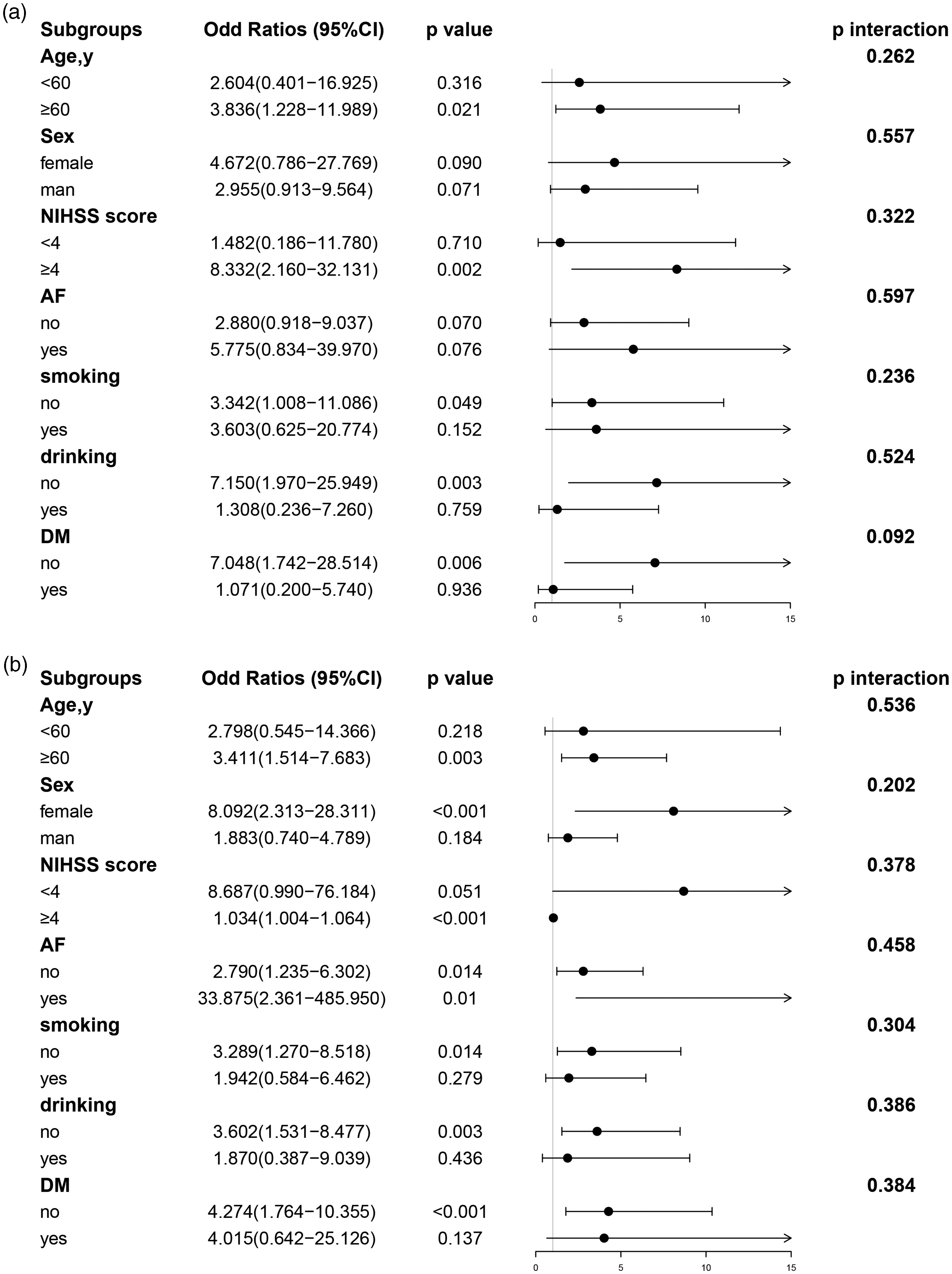
Subgroup analyses for the risk of (a) HT and (b) poor outcomes by NLR after IVT. The above model is adjusted for age, sex, atrial fibrillation, hypertension, diabetes, blood glucose, platelets, NIHSS score and onset-to-treatment time at admission. In each case, the model is not adjusted for the stratification variable. NLR: neutrophil-to-lymphocyte ratio; IVT: intravenous thrombolysis; HT: hemorrhagic transformation; NIHSS: National Institutes of Health Stroke Scale; DM: diabetes mellitus; AF: atrial fibrillation.
Discussion
In light of our findings, we systemically and comprehensively explored the following. (1) The increase in leukocyte count, neutrophil count, the NLR after r-tPA therapy and their absolute and relative variations were associated with increased risks of HT and three-month functional outcomes. (2) There was no correlation between leukocytes at admission and clinical outcomes. Additionally, lymphocyte count was not linked to the incidence of HT and functional outcomes. (3) The best values of the NLR after r-tPA treatment for discriminating the occurrence of HT and three-month functional outcomes were 3.322 or more and 5.511 or more, respectively; these values were correlated with a 3.492-fold increased risk of HT and a 3.024-fold increased risk of three-month functional outcomes. In general, leukocyte subtypes may predict the presence of HT and unfavorable outcomes.
In this single-center retrospective study, the incidence of HT was 17.9%, which was higher than that reported in other large r-tPA trials. 17 There is some evidence that the propensity of HT in Asian populations was obviously higher than that in Western populations.18,19 This higher result may be attributed to the higher NIHSS scores, which has been confirmed as an independent risk factor for the incidence of HT after IVT. 20 A study that compared races and ethnicities demonstrated that, Asian individuals had the highest NIHSS scores for stroke, compared with White and Black individuals. 18 Additionally, this result may also be due to the older age. Stroke occurs mostly in elderly individuals with more comorbidities, which may increase the risk of HT.
Profound explorations on the underlying mechanisms of leukocytes, which are essential components of immunity and inflammatory reactions, in the pathogenesis and progression of stroke has become widespread. The incidence of ischemic stroke leads to the interruption of blood flow, thereby initiating the inflammatory cascade and increasing permeability of the BBB, especially in the acute phase of stroke (6–48 h after stroke onset). 21 Damage-associated molecular patterns (DAMPs), which are produced by injured and dying neurons, can activate pattern recognition receptors on both microglia and astrocytes. 12 Proinflammatory mediators and DAMPs enter the systemic circulation through the broken BBB and the cerebrospinal fluid drainage system, and then induce an immune response in primary and secondary lymphoid organs, thereby triggering an enhanced systemic inflammatory response syndrome (SIRS). SIRS can transitorily promote the increased release of inflammatory mediators (e.g., neutrophils, TNF, IL-2 and C-C motif chemokine ligand 2). 12 MMP-9, a major extracellular matrix digesting enzyme released by neutrophils, may weaken brain microvessels and lead to the breakdown of the BBB by degrading the basement membrane and extracellular matrix components, such as laminins, fibronectin and type IV collagen.8,9 It is reported that MMP-9 is increased in the vicinity of infarct and hemorrhagic transformation areas, thus confirming the destructive effects of MMP-9 on the brain. 22 Additionally, neutrophils can mediate reactive oxygen species (ROS), thereby destroying the BBB and neurovascular units through the regulation of endothelial cells, smooth muscle cells and perivascular cells.23,24 The disruption of the BBB promotes the infiltration of peripheral immune cells into the infarct tissue and aggravates the impairment of endothelial cells through an incremental local inflammatory response. Consequently, this process has the potential to aggravate the complications of stroke, especially cerebral edema and HT. 11 The occurrence of HT is a response to endothelial cell destruction and is correlated with the infiltration of leukocytes in the brain. 25 Accordingly, these pathological findings may unveil the potential mechanisms and provide a novel approach for therapeutic targets of stroke.
In line with our study, Olivier Pétrault et al. revealed that the reduction in neutrophil count induced by vinblastine and RP3-anti-rat neutrophil monoclonal antibodies was associated with the prevention of endothelial dysfunction and smooth muscle injury after experimental cerebral ischemia. 7 Similarly, a multitude of empirical studies have investigated whether a higher neutrophil count is positively linked to intracerebral hemorrhage (ICH) and unfavorable outcomes, regardless of r-tPA infusion.4,26 Nevertheless, most previous studies measured leukocytes at a single time point and ignored leukocytes in different processes of stroke. Additionally, these previous studies did not compare leukocyte subtypes, including both static and dynamic levels, to assess their prognostic values for clinical outcomes. In our study, leukocyte subtypes pre-thrombolysis and post-thrombolysis were examined and their absolute and relative dynamic changes were calculated.
The NLR, a predictor of stroke that can be determined rapidly and inexpensively, jointly reflects the balance between neutrophil and lymphocyte levels. Consistent with our research, it is intriguing to note that in patients treated with r-tPA, NLR had a positive association with increased risk of HT and poor outcomes.4,27 The potential mechanisms for the occurrence of HT may result from the breakdown of BBB and the increment of permeability through inflammatory response. A single-center retrospective study conducted at Christian Doppler Medical Center and translated the NLR findings on AIS to the development of ICH treated with endovascular thrombectomy, and revealed that the NLR at admission was related to the occurrence of ICH in AIS patients. 28 The pathological mechanism for ICH may be as follows: hematoma components initiate inflammatory signals by activating microglia and release chemokines and pro-inflammatory cytokines, which elicits the recruitment of inflammatory cells (e.g., neutrophils) in the brain.29,30 Furthermore, a multitude of studies have investigated that the NLR significantly increases with the incidence of the complications of stroke, such as early neurological deterioration, 6 cerebral edema 31 and poststroke delirium. 32
In our study, superior to other leukocyte subtypes and their dynamic variations, NLR after IVT treatment was the best marker for the presence of HT and mRS scores of 3–6 at three months. The reasons are as follows.
First, among all the leukocytes, the NLR after IVT therapy had the highest AUC value and strongest Spearman correlation for HT and functional outcomes. Second, the NLR comprehensively reflects the inflammatory action of neutrophils and the regulatory and immune actions of lymphocytes. 33 The release of inflammatory cytokines mediated by neutrophils is likely to trigger lymphocyte apoptosis. 34 Due to the overactivation of neutrophils, the gap between neutrophils and lymphocytes increases, causing obvious changes in the NLR. Third, leukocytes that are changed dynamically do not appropriately account for leukocyte counts at pre-thrombolysis or post-thrombolysis. For instance, the implication of a ΔNLR of three varies dramatically for a patient with an NLR of two versus an NLR of eight. Therefore, the NLR after IVT is a more appropriate parameter than other leukocyte subtypes and their variations.
Several limitations need deep considerations. First, leukocytes were examined only at two time points: at admission and after r-tPA therapy. However, the distribution of leukocytes is constantly changing during the whole thrombolytic process. Second, we concentrated on leukocytes, but some inflammatory biomarkers, such as CRP and LDL levels, were unavailable in our research.35,36 Hence, in future research, data on these inflammatory indicators should be collected prospectively. Finally, in view of the fact that the samples were collected from only one city in China, the results may not be generalizable. Further large-scale prospective studies and multicenter collaborative studies are needed to validate our conclusions.
In total, as neuroprotective prognostic biomarkers, higher leukocyte count, neutrophil count, and NLR after IVT, as well as their absolute and relative dynamic changes, may be independently related to the increased risk of clinical outcomes. Hence, the monitoring of leukocytes in AIS patients treated with r-tPA may provide insights into therapeutic targets and merit attention in future studies.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221142694 - Supplemental material for Leukocyte indicators and variations predict worse outcomes after intravenous thrombolysis in patients with acute ischemic stroke
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221142694 for Leukocyte indicators and variations predict worse outcomes after intravenous thrombolysis in patients with acute ischemic stroke by Jiali Xie, Chunyang Pang, Huan Yu, Wanli Zhang, Chuancheng Ren and Binbin Deng in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (grant number 81901273) and Science Technology Department of Zhejiang Province (grant number Q21H090076).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The Ethical Decision Committee of the Research Administration at First Affiliated Hospital of Wenzhou Medical University approved the study (2021-R077 and KY2022-R008). And all patients agreed to participate in the study, and use their clinic data and information for research purpose.
Authors’ contributions
Jiali Xie and Chunyang Pang contributed to data statistics and writing paper. Huan Yu, Wanli Zhang and Chuancheng Ren were responsible for data collection. Binbin Deng provided resources and designed the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
