Abstract
Rodents display “empathy” defined as perceived physical pain or psychological stress by cagemates when co-experiencing socially distinct traumatic events. The present study tested the hypothesis that empathy occurs in adult rats subjected to an experimental neurological disorder, by allowing co-experience of stroke with cagemates. Psychological stress was measured by general locomotor activity, Rat Grimace Scale (RGS), and plasma corticosterone. Physiological correlates were measured by Western blot analysis of advanced glycation endproducts (AGE)-related proteins in the thymus. General locomotor activity was impaired in stroke animals and in non-stroke rats housed with stroke rats suggesting transfer of behavioral manifestation of psychological stress from an injured animal to a non-injured animal leading to social inhibition. RGS was higher in stroke rats regardless of social settings. Plasma corticosterone levels at day 3 after stroke were significantly higher in stroke animals housed with stroke rats, but not with non-stroke rats, indicating that empathy upregulated physiological stress level. The expression of five proteins related to AGE in the thymus reflected the observed pattern of general locomotor activity, RGS, and plasma corticosterone levels. These results indicate that stroke-induced psychological stress manifested on both the behavioral and physiological levels and appeared to be affected by empathy-associated social settings.
Introduction
Rodents display a level of “empathy,” defined as a modulation of perceived pain or physiological stress in response to social environment. Mice show hyperalgesia when they are injected with 0.9% acetic acid intraperitoneally together with an injected cagemate, but not in isolation or with a non-injected cagemate. 1 Restrained stress measured by plasma corticosterone is lower when restrained cagemates are housed together, but higher when non-restrained cagemates are housed together in comparison with a control condition in which subjects are restrained alone. 2 Mice develop freezing behavior when observing other mice (demonstrators) receiving repetitive foot shocks. Such fear response is higher when demonstrators are socially related, such as siblings or mating partners. 3 This empathy phenomenon has also been reported in rats. Observer rats having previously experienced foot shocks display vicarious freezing behavior upon witnessing a cagemate experience footshocks. 4 Freely moving rats in an arena with restrained cagemates intentionally and quickly open the restraining chamber and free cagemates. Even when another restraining chamber containing chocolate is put in the arena, freely moving rats open both chambers and share the chocolate with cagemates. 5
In the context of experimental models of neurological disorders, which can be considered equally as stressful as electrical shock or the restraint paradigm, social environment has been shown to be an exacerbating or improving factor in experimental stroke. Mice repeatedly exposed to a large, aggressive, stranger mouse exhibit larger infarction after middle cerebral artery occlusion (MCAo), a model of ischemic stroke. 6 On the other hand, mice housed in pairs showed decreased infarct size and improved contralateral paw use after MCAo in comparison with single housed mice. 7 Co-habituation with partitioned cagemates does not have such neuroprotective effects, suggesting that physical contact is an important mediator of social influences on experimental stroke outcome. 8 The social influence on ischemic damage closely involves a neuroinflammatory response, whereby brain interleukin-6 (IL-6) signaling is down-regulated in isolated stroke mice relative to socially housed stroke mice. 9 Taken together with the rodent “empathy” studies described earlier, not only these simple social settings (e.g. presence/absence of a stranger/cagemates), but also more complex social conditions involving an empathetic situation, can be incorporated in rodent disease models in an effort to better manage and treat patients with brain disorders, particularly stroke.
Psychological and physical stress factors have been implicated to increase glucocorticoids, including corticosterone. Increased levels of glucocorticoids induce various physiological and molecular responses in metabolic and immune systems. A key stress response under ischemic condition is gluconeogenesis as evidenced by the high proportion of patients with acute stroke developing hyperglycemia, including those without pre-existing diabetes.10–13 Interestingly, gluconeogenesis may be mediated by the advanced glycation end products (AGEs)-signaling cascade,14–16 with the aberrant inflammation in response to stroke mediated via the receptor of the AGE pathway.17,18 Thus, we hypothesized that MCAo-induced stress would increase corticosterone levels, including gluconeogenesis, likely via upregulation of the AGE pathway altogether accompanying empathy-like social behavior. The expression of the receptor for AGE on brain capillary endothelial cells may facilitate the transport of oxytocin enhancing appropriate expression of maternal behavior. 19 Since oxytocin mediates social behavior/recognition including empathy, 20 the AGE pathway may serve as a good candidate signaling mechanism in investigating the effects of social settings on experimentally induced stroke.
The present study aimed to elucidate relationships between MCAo and its pathological “stress” symptoms including physical and biological alterations in a rat stroke model, in order to better understand the occurrence of empathy after stroke. We hypothesized that (1) MCAo would cause stress, including pain which damages the thymus, and that (2) these adverse biological effects of stroke could be modulated by social setting. Here, we utilized the RGS, which has been recently reported as a novel pain scale for mice 21 and rats, 22 to measure subjective pain level due to MCAo. Before appearance of the RGS, pain scales for animals were difficult to utilize generally because it had specific algesic stimulus – response relationships. For example, the tail flick test 23 gives heat stimulus to a tail and measures latency to flick a tail; The hotplate test 24 puts a subject on a heated hotplate and measures latency to licking of paws, rearing, and jumping; The formalin test 25 injects 5% formalin into a paw, then observes behaviors such as licking, shaking, and lifting of the injected paw; and so on. In contrast to those classic procedures, the RGS enabled us to measure pain level from facial expressions of rodents, including changes in eyes, nose, cheeks, ears, and whiskers, in a drug-free setting. The present study was the first application of the RGS to a neurological disease model.
In an effort to study the molecular basis of stress, we also investigated AGEs-related proteins in the thymus, an important organ of the immune system known to be strongly impaired after receiving MCAo. 26 AGEs are products of non-enzymatic glycation and oxidation of proteins, and accumulate in long-lived proteins through aging. AGEs activate signaling mechanisms that cause cell stress. Target organs are damaged by AGEs-induced cellular dysfunction, leading to complication. 27 Receptor for advance glycation end products (RAGE) expresses in the lesions. Oxidative stress and protein glycation induce the expression of the RAGE in the kidney. 28 The RAGE dependent down-regulation of glyoxalase I (GLO1), the key enzyme for preventing formation of AGEs precursor, would perpetuate further RAGE up-regulation. 29 RAGE stimulation up-regulates NF-κB, which is implicated in inflammatory responses,30,31 and in turn, NF-κB regulates RAGE expression so that it further up-regulates and sustains cellular dysfunction. 32 Myeloid differentiating factor 88 (MyD88) is essential for cell survival through NF-κB regulation, although it opposes the RAGE pathway. 33 Diaphanous-1 is involved in RAGE signaling transduction 34 and appears to regulate the various pathways of AGE-RAGE signal transduction. 35
Materials and methods
Subjects
Sixty-four eight-week-old male Sprague-Dawley rats (Harlan Sprague Dawley, Indianapolis, IN) were served as subjects for this study. They were kept two individuals per cage and allowed to habituate for a week. The experimenter daily handled them for 5 min. Their weights at nine-week-old, the beginning of the experiment, were around 250 g. They had access to food pellets and water ad lib. Light/dark cycle of the colony room was 12 L:12D. The experiment was conducted on the first half of the light period. All experiments were conducted under a protocol approved by the Institutional Animal Care and Use Committee (IACUC) of the University of South Florida. All studies were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and the United States Public Health Service’s Policy on Humane Care and Use of Laboratory Animals, and in compliance with Animal Research: Reporting in Vivo Experiments (ARRIVE) guidelines. Animals were tested with RGS at different time points (Figure 1(a)).
(a) Timeline of experimental protocols. Animals were perfused on Day 3. Behavior measurements, blood, and thymus tissues were collected from all animals. MCAo: middle cerebral artery occlusion; RGS: rat grimace scale; ELISA: enzyme-linked immunosorbent assay. (b) Schematic illustration of caging conditions. (a) Stroke with stroke (SS): both rats kept in one cage received MCAo. (b) Stroke with naïve (SN). Rat that received MCAo is referred to as SN. (c) Naïve with stroke (NS): One of two rats received MCAo. Naïve rat is referred to as NS. (d) Naïve with naïve (NN): both rats did not receive MCAo. Card boards were inserted between cages to prevent visual access to rats other than the cagemate. Hereafter, the terms for caging conditions are as follows: (a) SS, (b) SN, (c) NS, and (d) NN. Gray: MCAo rats; White: Naïve rats.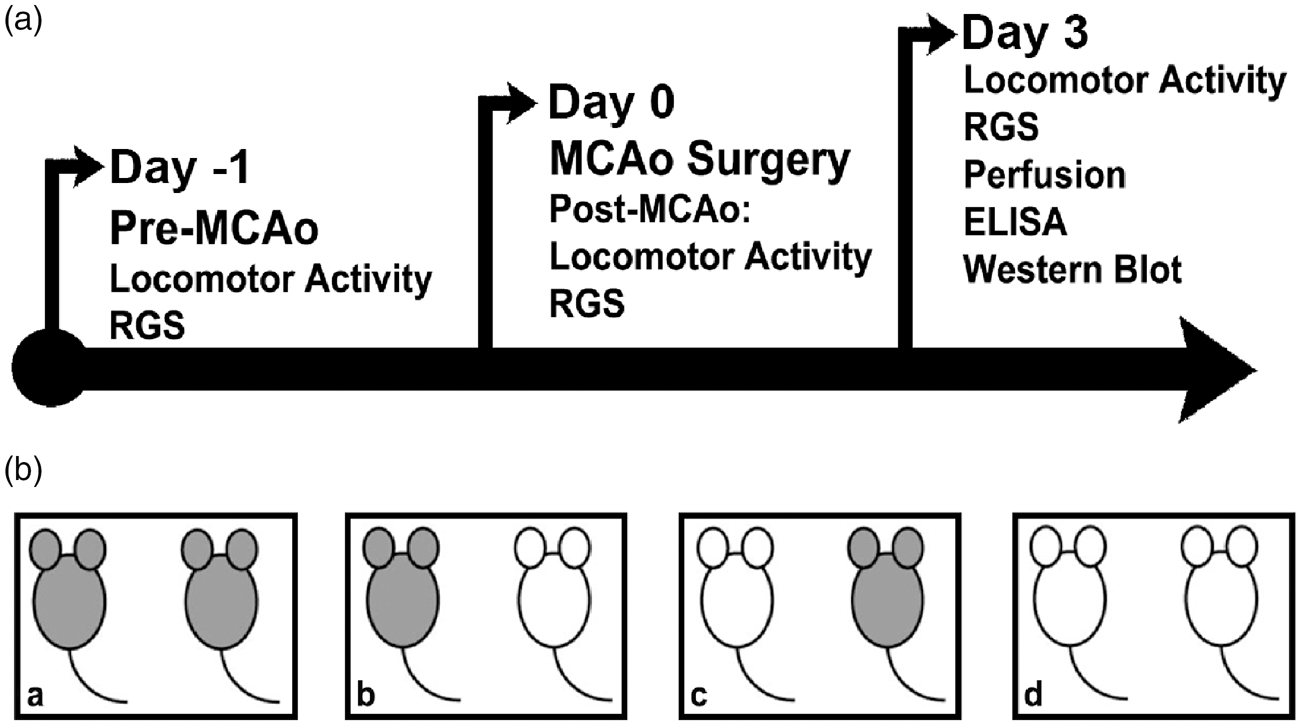
Social conditions
Subjects were randomly assigned to one of four conditions: (1) surgery with surgery, referred as Stroke with Stroke, both rats in each cage received MCAo surgery, (2) and (3) surgery with naive (and naive with surgery vice versa), referred to as Stroke with Naive and Naive with Stroke, only one of two rats in each cage received MCAo surgery, (4) Naive with Naive, referred as Naive with Naive, neither rat in each cage received MCAo surgery (Figure 1(b) for a schematic illustration). To prevent visual access to neighboring cages, a white cardboard panel was inserted between cages during the experimental period. If one of the two rats in a cage died during the experimental period, another rat was excluded from the experiment to sustain the social conditions described above.
Procedure
MCAo and tissue collections
Subjects were randomly assigned to the stroke condition in groups 1, 2, 3, and 4, and received MCAo surgery in accordance with a well-established and standard protocol. 36 Briefly, subjects were anesthetized with isoflurane and a small incision was made on the neck to expose the junction of the right external carotid artery (ECA) and internal carotid artery (ICA). A coated nylon suture was inserted from a junction of the ECA to the origin of the MCA through the ICA. Reperfusion was induced 1 h after occlusion by removing the suture while subjects were under anesthesia. Thirty-four subjects received MCAo surgery, then eight died after surgery (mortality rate =23.5%). These dead animals and their cagemates were excluded from the experiment, and thus 48 subjects were used for the analysis. Subjects were sacrificed at day 3 after MCAo. Two rats in a cage were simultaneously exposed to CO2, and then blood samples were immediately collected in heparinized tubes transcardially for plasma corticosterone and MG assay. Then the subjects were perfused with 0.9% saline and 4% paraformaldehyde. The thymuses were harvested, post-fixed with 4% paraformaldehyde, frozen in liquid nitrogen, and stored at −80° C until use. Thymus tissues were collected from all animals.
Activity score
To confirm the behavioral effect of MCAo, subjects’ general locomotor activity a day before and, day(s) 0 and 3 after MCAo surgery was rated. We used videos which were recorded for making still images of rats’ faces for the modified Rat Grimace Scale (see below). For video recording, a home cage was brought from a colony room to an experimental room during the first half of the light period. A video camera (HDR-XR520V, Sony) was set in front of the cage. The distance from the cage to the camera was around 30 cm. An experimenter was positioned about 1.5 m from the cage and stood still during recording. Under these settings, freely moving rats in the home cage were recorded for 15 min. It was found by pre-observation that subjects were more active in the first 5 min than the remainder of the 10 min, probably due to exploratory behavior in response to changing rooms. Therefore, the first 5 min of each video was used for analysis. For each video, the experimenter rated subjects’ activity as 0 (completely static), 1 (moving without locomotion; e.g. sniffing and grooming), or 2 (locomotion, rearing, and play fighting) for 5 min with instantaneous sampling with a time interval of 1 min. Scores were averaged for each subject for each day, so that the maximum possible activity score was 2. Scoring locomotor activity at a certain interval is a commonly used method in behavioral studies.37,38
Modified Rat Grimace Scale
Subjects’ pain related to MCAo was evaluated in accordance with the Rat Grimace Scale (RGS), 22 which is a rat version of the Mouse Grimace Scale 21 with a slight modification. Subjects in a home cage were videotaped for 15 min a day before, and 0, 1, 2, and 3 day(s) after, MCAo. A mesh ceiling containing food pellets and a water bottle were removed during recording. After the recording, five still images of each subject were taken from the video with approximate intervals of 3 min. All images were taken from left side of subject’s face to avoid bias caused by expressions of the right side of the face due to right-sided MCAo. Still images were cropped using PhotoShop CS2 (Adobe) to exclude the subject’s body. After taking all of the face pictures, a PowerPoint slideshow was created to allow each picture to be rated individually. The order of rating was randomized by a PowerPoint macro (http://www.tushar-mehta.com/powerpoint/randomslideshow/index.htm) to make a rater blind to experimental conditions. The rater evaluated subject’s facial expressions which included changes in four action units, namely, eyes, nose/cheeks, ears, and whiskers. Each action unit was scored from 0 (no pain) to 2 (high pain). The rater evaluated all pictures three times, each in a different, random order. High coefficient of concordance between three ratings was confirmed (Kendall’s coefficient of concordance W = 0.84, p < 0.0001). The scores were then averaged for each subject and used for subsequent analysis. All animals underwent behavioral testing.
Enzyme-linked immunosorbent assay for plasma corticosterone and methylglyoxal
Blood samples were collected in heparinized tubes. The samples were centrifuged at 3000 r/min for 10 min at 4℃ in order to collect the plasma. The collected plasma was stored for analysis at −80℃. The plasma concentration of coriticosterone was analyzed with a corticosterone ELISA kit (ab108821, abcam). Also, the plasma concentration of methylglyoxal (MG), which is a precursor of AGEs, was analyzed with an ELISA kit (ab238543, abcam). Blood was collected from all animals.
Western immunoblotting
Thymic tissues were frozen in liquid nitrogen immediately after harvesting. The frozen tissues were sonicated in ice-cold buffer (2% SDS, 100 nM dithiothreitol, and 50 mM Tris, pH 7.4). The sonicated solutions were boiled for 5 min in a hot water bath, and then centrifuged at 300 g for 15 min at room temperature. The supernatants including 5, 10, or 20 µg of protein/lane, depending on amount of target protein, were subjected to SDS/glycerol PAGE (10% acrylamide/10% glycerol gels). Then, the separated proteins were transferred onto nitrocellulose membranes (BIO-RAD, USA). After electrophoretic transfer, the nitrocellulose membranes were incubated with a blocking buffer (0.1% Tween-20, 20% non-fat milk, and 50 mM Tris, pH 7.4) for 2 h at room temperature, then incubated with the primary antibodies, either anti-RAGE (ab37647, abcam), anti-NF-kB (ab7970, abcam), anti-GLO1 (ab137098, abcam), anti-MyD88 (ab2064, abcam), anti-DIAPH1 (ab129167, abcam), or anti-GAPDH (ab8245, abcam) antibody diluted to 1:5000, 1:1500, 1:3000, 1:1000, 1:1000, and 1:5000, respectively, overnight at 4℃. After washing, the nitrocellulose membranes were incubated with a secondary antibody (IRDye 800CW, either goat anti-mouse IgG or goat anti-rabbit IgG, LI-COR) for 90 min at room temperature. The membranes were scanned with the Odyssey infrared imaging system (LI-COR) immediately after washing.
Statistical analysis
Data were analyzed with a one-way or two-way analysis of variance (ANOVA) with a significance level of 5%, followed by multiple comparisons with Sidak correction. As necessary, the Huynh-Feldt ɛ correction was used to evaluate F ratios for repeated measures involving more than one degree of freedom. All of the analyses were performed on IBM SPSS version 20.
Results
Stroke with stroke (SS) and Stroke with Naïve (SN) rats exhibited a significantly sustained reduction of general locomotor activity after surgery in comparison with those of pre-surgery (Figure 2, Stroke with Stroke: day 0, p < 0.001; day 3, p = 0.001, Stroke with Naive: day 0 and 3, p < 0.001, multiple comparisons with Sidak correction). These results indicated that MCAo surgery successfully impaired subjects’ motor function. Interestingly, the Naive with Stroke (NS) rats also showed significant reduction of general locomotor activity compared to pre-surgery levels (day 0, p < 0.001; day 3, p = 0.003), although Naive with Naive (NN) rats did not show such reduction. Thus, the locomotor activity of the naive rats, which did not receive surgery, was socially suppressed by cohabiting with the stroke rats.
General locomotor activity reduction after MCAo. SS, SN, and NS but not NN expressed significantly reduced locomotor activity after MCAo (pre- vs. post-MCAo, p < .001 for three groups, pre-MCAo vs. Day 3, SS, p = .001, SN, p < .001, NS, p = .003, repeated measures ANOVA with Sidak correction, N = 14 for SS, 11 for SN and NS, 12 for NN). Filled circle with solid line: SS, filled square with solid line: SN, open square with dashed line: NS, open circle with dashed line: NN. Mean ± standard deviation.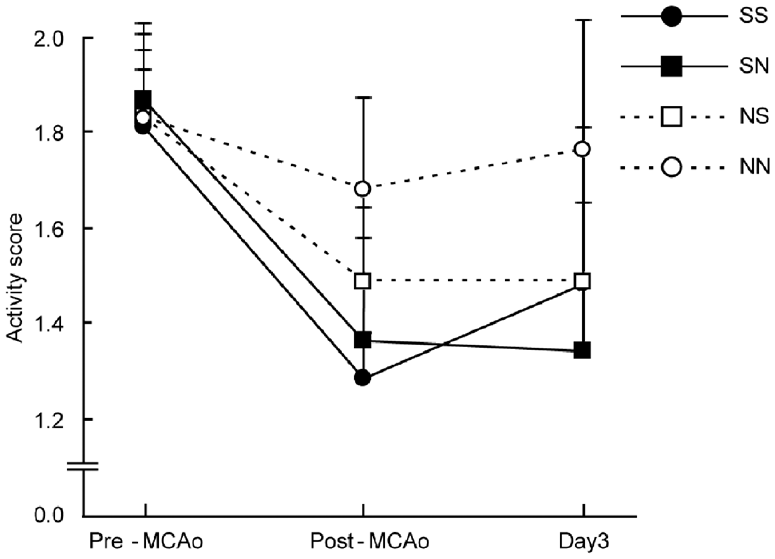
The Rat Grimace Scale
22
was originally used to determine the pain level in rats. Examples of pictures frame-grabbed from the video are shown in Figure 3(a) (a–c, examples from high-scored pictures; d–f, examples from 0 points pictures). The right eye typically narrowed following right-sided MCAo. Therefore, to avoid potential bias in determination of the score, all pictures were taken from the left-side of the face. The Stroke with Stroke group exhibited significantly higher scores in day 0 compared to pre-surgery (Figure 3(b), p < 0.001, multiple comparisons with Sidak correction). In day 3, pain scores were still marginally higher than pre-surgery levels (p = 0.085) although they showed marked reduction from day 0. The stroke rats that cohabitated with naive rats showed results similar to the Stroke with Stroke rats. They exhibited a significant increase in scores on day 0 (p < 0.001) which then decreased but remained higher than the pre-surgery scores (p = 0.029). In contrast to those groups, Naive with Stroke and Naive with Naive rats did not show any significant changes in scores.
(a) Representative examples for the rat grimace scores (RGS). Face pictures were grabbed from video. Left side face was always used for analysis in order to avoid influence of MCAo-induced narrowing eye on right side. (a–c), high scoring pictures. (d–f), 0 points pictures. (b) Increasing RGS score after MCAo. SS and SN significantly increased RGS score after MCAo (SS, p < .001, SN, p < .001). Both groups showed reduction of scores on Day 3 but still significantly higher or marginally higher than pre-MCAo (SS, p = .085, SN, p = .029). NS and NN did not increase scores through the experiment. Repeated measures ANOVA with Sidak correction, N = 13 for SS, 11 for SN and NS, 12 for NN. Filled circle with solid line: SS, filled square with solid line: SN, open square with dashed line: NS, open circle with dashed line: NN. Mean ± standard deviation. (c) Higher plasma corticosterone level in SS. SS exhibited significantly higher plasma corticosterone level in comparison with other groups on Day 3 after MCAo (vs. SN, p = .035, vs. NS, p = .001, vs. NN, p = .019, ANOVA with Sidak correction, N = 16 for SS, 12 for SN, NS, and NN). Mean ± standard deviation. (d) Higher plasma MG level in SS and SN. SS and SN exhibited significantly/marginally higher plasma MG level compared to NN on Day 3 after MCAo (vs. SS, p = .045, vs. SN, p = .060, vs. NS, p = 1.00, ANOVA with Sidak correction, N = 5 for SS, 4 for SN, 3 for NS and NN). Mean ± standard deviation.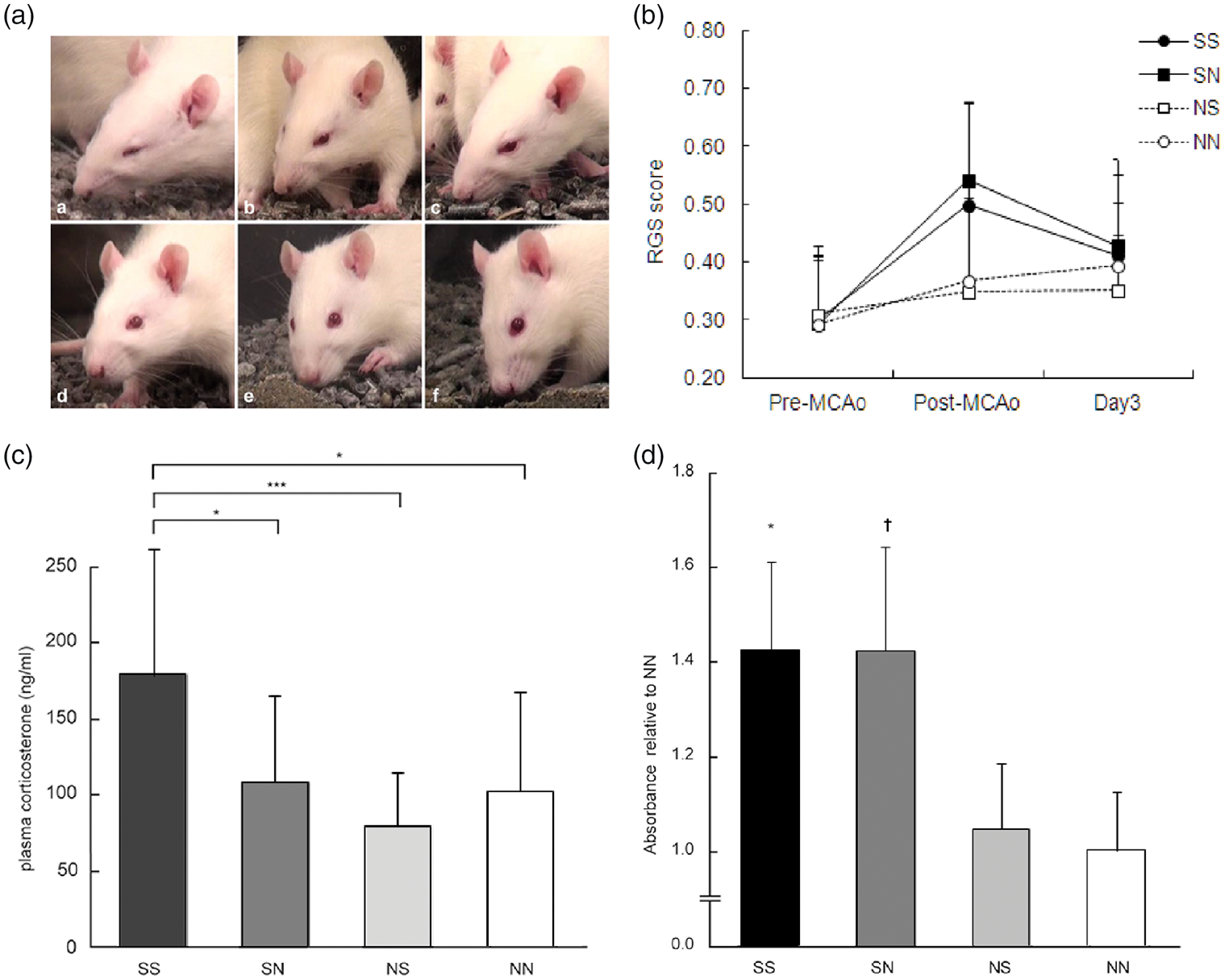
To determine subjects’ stress level, ELISA for plasma corticosterone was performed at Day 3 after MCAo (Figure 3(c)). The Stroke with Stroke group showed significantly higher plasma corticosterone levels in comparison with the other three groups (p = 0.035 vs. the Stroke with Naive, p = 0.001 vs. the Naive with Stroke, and p = 0.019 vs. the Naive with Naive, multiple comparisons with Sidak correction). In contrast, Stroke with Naive rats did not differ from Naive with Stroke and Naive with Naive rats (p > 0.05). Thus, the direct experience of MCAo stroke surgery coupled with cohabitation with another Stroke rat was required to display higher stress at this time point. Of note, the stress associated with the surgical experience of stroke could be suppressed by cohabitation with Naive rats.
Plasma MG concentration was examined with an ELISA assay (Figure 3(d)). Stroke with Stroke and Stroke with Naive rats, but not Naive with Stroke rats, showed significantly/marginally higher MG concentration, respectively, when compared to Naive with Naive rats (p = 0.045 for the Stroke with Stroke, p = 0.060 for the Stroke with Naive, and p = 1.000 for the Naive with Stroke, multiple comparisons with Sidak correction).
Next, we investigated the molecular basis of MCAo response to social settings by focusing on the thymus, which is known to be stress sensitive and an important organ for T-cell maturation. Western blotting analysis was conducted to examine expression levels of AGE-related proteins in the thymus (Figure 4). Expression of the AGE receptor (RAGE) was higher in Stroke with Stroke, Stroke with Naive, and Naive with Stroke rats than in Naive with Naive rats. This pattern was also seen in NF-κB expression (Figure 4(b)). GLO1 exhibited the opposite pattern: lower expression was seen in Stroke with Stroke, Stroke with Naive, and Naive with Stroke than in Naive with Naive. Moreover, comparisons between SS, SN, and NS groups demonstrated no significant differences in RAGE, NF-κB, and GLO1 expression. Myd88 showed lower expression in Stroke with Stroke and Stroke with Naive than Naive with Stroke and Naive with Naive. There were no significant differences between the SS and SN groups, and between the NS and NN groups. DIA-1 expression was reduced only in Stroke with Stroke. Additionally, no significant differences were found between the SN, NS, and NN groups. Thus, Stroke with Stroke shows a completely opposite pattern of expressions when compared to Naive with Naive, suggesting strong activation of the stress-related pathological cascade due to co-experience of stroke, although Stroke with Naive and Naive with Stroke show an intermediate pattern of expressions.
Western immunoblotting analysis of thymus tissue for RAGE cascade. Immunoblots are shown in Panel A and quantified in Panel B. SS, SN, and NS exhibited higher expression of RAGE and NF-kB than NN. In contrast, GLO1 expression showed opposite pattern. Myd88 expression was lower in SS and SN than NS and NN. DIA-1 showed lower expression in SS than the others.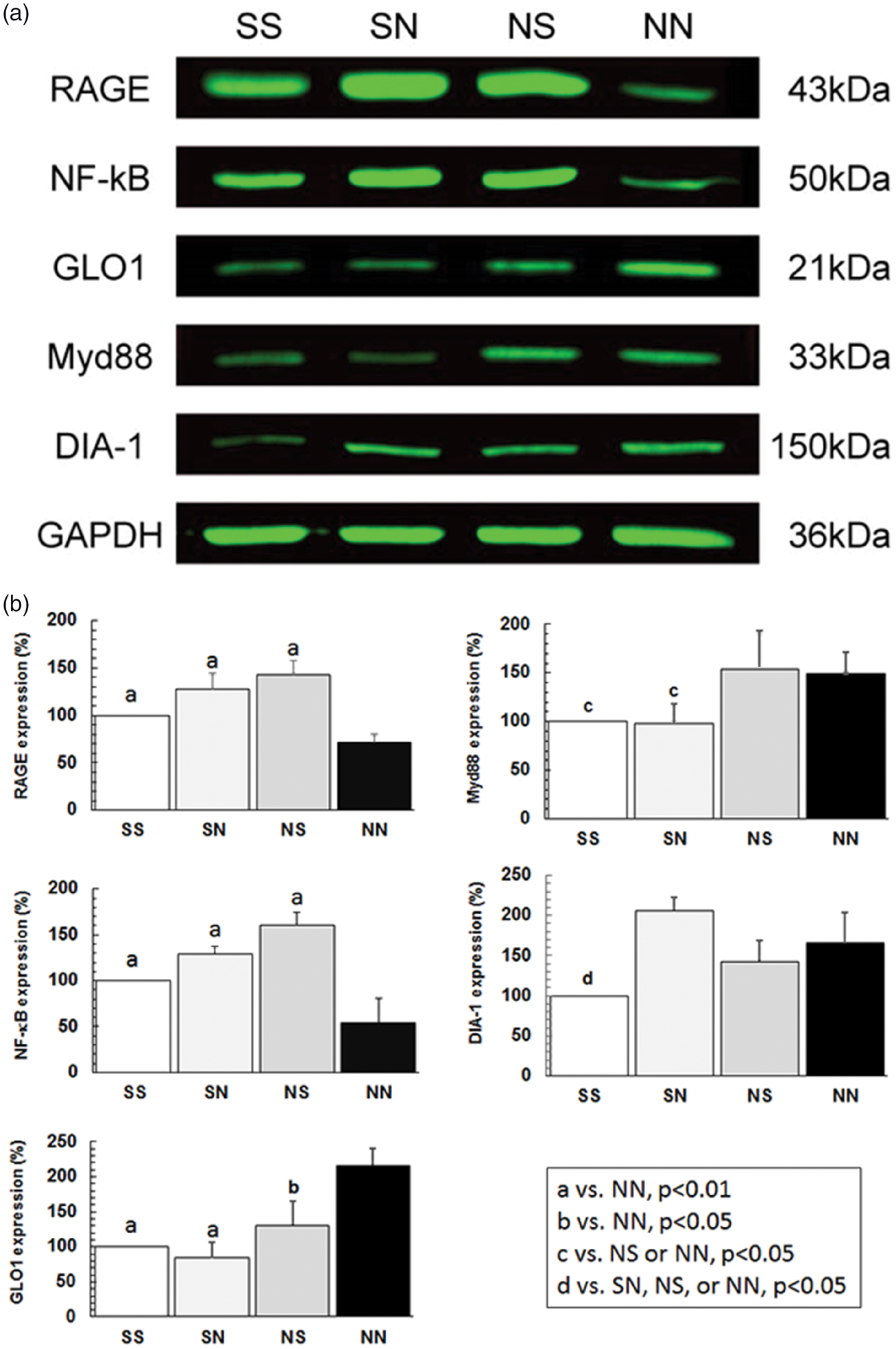
Discussion
The present study demonstrated behavioral, physiological, and molecular changes following experimental stroke, in particular advancing the utilization of the Rat Grimace Scale to evaluate pain levels after stroke. This is the first report of the application of this scale in neuropathological disease condition. Highest level of pain was observed on the first day following MCAo. Pain then markedly decreased on day 3, supporting the original studies which demonstrated the MGS/RGS can detect moderate durations of pain.21,22 Around 8% of stroke patients are estimated to have central post-stroke pain. 39 A few studies have demonstrated a lowering of pain thresholds, measured with traditional tests, after ischemic or hemorrhagic insult in rats.40,41 However, in contrast to measuring such hypersensitivity, measuring spontaneous pain in non-human animals was obviously a difficult task. The MGS/RGS could become powerful tools for the study of treatments of post stroke pain in animal models of stroke. We showed that an ischemic injury elevated corticosterone levels and gluconeogenesis concomitant with upregulated AGE proteins, suggesting a key role for AGE-signaling pathway as a potential mechanism underlying empathy-like behaviors in ischemic stroke animals.
Our present observations revealed various aspects of MCAo-induced changes were affected by social settings. Simple social manipulation, namely, paired- or isolated-housing, has been known to affect MCAo-induced symptoms.7–9 The present results revealed that complex social settings, namely co-experience or single experience of stroke in pair-housed rats could differentially influence pain and stress outcomes after stroke. Plasma corticosterone was significantly higher only in Stroke with Stroke rats than any other groups on day 3, indicating that the direct experience of the stroke surgery plus the cohabitation of another stroke rat induced sustained stress. This is inconsistent with a previous report demonstrating that rodents subjected to acute restrain stress together with cagemates experience lower corticosterone levels than when the stressful event is experienced with non-restrained and non-stressed cagemates. However, these differences may be attributed to the fact that the prior study utilized an acute, temporary stressful event, while the rats in the present study were subjected to a lasting stroke injury. 2 A Stroke rat co-habitated with a Naive rat displayed lower stress levels compared to Stroke with Stroke rats. In contrast, the RGS score did not differ between the Stroke with Stroke and Stroke with Naive groups. We originally expected to observe different pain levels between Stroke with Stroke and Stroke with Naive rats because pain perception is known to be facilitated by co-experience of painful stimulus with a cage-mate. 1 As previously reported, the RGS can effectively detect moderate durations of pain. 22 Thus, while the Stroke with Stroke rats exhibited significantly higher level of physiological stress than Stroke with Naive rats, the behavioral pain as revealed by RGS could not detect any significant difference between the two conditions likely due to sustained, instead of moderate, pain duration in both settings. Moreover, the level of locomotor activity in naïve rodents housed alone is not significantly different from the level of locomotor activity in naïve rodents housed in pairs. 42 This suggests that the decreased locomotor activity of NS rats in the current study was due to social suppression rather than the lack of stimulation due to decreased activity of the stroke-afflicted cagemate. Social facilitation or inhibition induced by co-experience of an event is a type of phenomenon which has been referred to as “emotional contagion” or “empathy”. The present study demonstrated a neuropathological condition also elicited empathy-like effects through co-experience of the painful stroke surgery and cohabitation with another stroke animal.
Western immunoblotting analysis found changes in expressions of RAGE-related proteins in the thymus tissue after MCAo. RAGE interacts with AGEs and is likely to accelerate the aging process through the maintenance of inflammatory reactions. Stimulation of RAGE induces cytokine production and adhesion molecule expression in a manner dependent on JNK signaling. 43 It also up-regulates NF-κB and early growth response-I (Egr-1) which are transcription factors implicated in inflammatory responses.30,31 Significant increases in RAGE and NF-κB expression in the NS group compared to the NN group and no significant differences between the SS, SN, and NS groups suggest that housing Naïve rats with Stroke rats enabled the Naïve rats to also experience RAGE- and NF-κB-dependent stress cascades and an immune response, and did not abate these physiological changes in Stroke rats. GLO-I (glyoxalase I) is the key enzyme to prevent AGEs precursor formation. It down-regulates RAGE dependently, and then serves to further up-regulate RAGE expression. It increases methylglyoxal (MG) and glyoxal, leading to increased AGE formation. 29 Furthermore, the significant reduction in GLO1 expression in the NS group relative to the NN group and no significant differences between the SS, SN, and NS groups imply that placing Naïve and Stroke rats in the same cage did not prevent the decrease in GLO1 levels in Stroke rats and enabled Naïve rats to acquire this physiological change in GLO1 from their stroke-afflicted cagemates, which likely induced a higher state of stress and inflammation in the Naïve rats. The induction of physiological changes in RAGE, NF-κB, and GLO1 expression corresponding to stress and immune responses in Naïve rats is consistent with past reports indicating that rodents not subjected to experimental stressors manifest physiological symptoms of stress when housed with cagemates subjected to experimental stressors. 44 Myeloid differentiating factor 88 (MyD88) is one of RAGE adaptor proteins. It is also implicated in the inflammatory response and cell damage signaling, although it opposes the RAGE signaling pathway. 33 MyD88 is thoroughly essential for cell survival via regulation of NF-κB, therefore the activation of the RAGE signaling pathway antagonizes survival mechanisms. 33 As MyD88 expression was significantly different between the SN and NN, and between the SN and NS groups, but not between the SS and SN, and between the NS and NN groups, it is likely that an actual stroke injury is necessary to generate the physiological decrease in MyD88, and housing Stroke and Naïve rats together were not sufficient to induce a decrease in MyD88 in the Naïve rat nor an increase in MyD88 in the Stroke rat. Diaphanous-1 is another adaptor protein involved in RAGE signal transduction. It is a member of the formin family which mediates the effects of the small GTPase of the Rho family. 34 The SS group was significantly different from the SN, NS, and NN groups, and there were no significant differences between the SN, NS, and NN groups, indicating that housing Stroke rats with Naïve rats enabled diaphanous-1 expression to increase in Stroke rats without a decrease in Naïve rats. Changes in MyD88 and diaphanous-1 expressions are known to regulate various pathways of AGE-RAGE signal transduction. 35 Those changes are sustained by positive feedbacks. In contrast to typical receptors which are down-regulated by its ligands, RAGE expression is up-regulated by RAGE ligands.32,45 Since RAGE itself is regulated by NF-κB, this will further increase, amplify, and sustain cellular dysfunction. 32
Taken together, our findings indicated that MCAo caused activation of RAGE-dependent cascades in the thymus tissue possibly via the corticosterone pathway. The thymus is a glucocorticoid sensitive organ, and known to drastically shrink when animals receive stress. The activation of the hypothalamic-pituitary-adrenal (HPA) axis leads to a rise in glucocorticoids and an acute systemic proinflammatory cytokine cascade, the combination of which culminates in involution, where CD4 + CD8 + thymocytes undergo apoptosis.46,47 There are various cytokine cascades that are triggered by HPA axis activation, including tumor necrosis factor (TNF), interleukin-1 (IL-1), interleukin 6 (IL-6), and nerve growth factor (NGF).46,48 These cytokine cascades are proinflammatory in that the resulting inflammation activates NF-κB, which is also important in the immune response. 49 Thus, MCAo-induced stress could activate RAGE-dependent cascades in the thymus via NF-κB activation. This hypothesis could be supported by our results which showed that significantly higher corticosterone levels in Stroke with Stroke rats paralleled strong activation of RAGE-related cascades.
There are technical and conceptual limitations in the present study. We focused on the acute phase of stroke, and as seen in Figure 2, stroke rats (both SS and SN) and Naive with Stroke rats (NS) did not show significant recovery of locomotor activity on Day 3 after MCAo surgery. Thus, it is difficult to extrapolate the effect of empathy on stroke recovery over time. In contrast, the RGS (Figure 3) showed significant reduction of score in SS and SN rats on Day 3, indicating, in this context, recovery over the acute time was seen. However, under this setting, NS rats did not show empathy-like response on Day 0, indicating variability in empathy levels even in the early phase of stroke. Additionally, the assessment of expression of AGE- and NF-κB-related molecules was limited to the thymus, one of the primary lymphoid organs. Other immune organs such as born marrow and spleen, and also brain could have similar changes. Experiments designed to remove the thymus before co-housing may provide gross effects of the thymus; however, we did not detect overt differences in thymus size and weight in stroke and non-stroke rats across treatment conditions (data not shown), suggesting that the differences in thymic response due to stroke and social settings may be captured more accurately by investigating cellular components and molecular signaling pathways, as partly seen with AGE- and NF-κB molecular expression. Future studies will be designed to further probe these cellular and molecular signature profiles of empathy in stroke that may reveal specific therapeutic targets for enhancing post-stroke recovery. Another limitation of the present study relates to lack of direct interrogation of the AGE pathway via agonistic and antagonistic agents, but previously glucocorticoids have been associated with stress-induced thymic involution. 50 In parallel, we envision that manipulating the levels of corticosterone, by modifying the empathy-like social settings, may also differentially affect the thymic AGE pathway, which will be an equally powerful mechanistic study.
Translational and clinical implications can be derived from the present findings. Preclinical stroke animal modeling should take into account the possibility that empathy-like phenomenon may influence neurological assessments since caging condition can amplify or dilute the data on behavioral recovery. For example, caging effects can confound neurological assessments in animal studies, in that if there is one animal that does not recover well, then this may cause problems with other cage-mates. Thus, it is preferable that studies using animal model describe caging conditions in more detail, such as number of animals cohabitated, and number of animals in a cage exposed to stroke or similar stress-associated pathological condition. Similarly during recovery at home, stroke patients may be “housed together” with non-stroke family members who may have varying levels of stress that may either aid or impair the recovery process in the stroke patients. Appropriate set-up of the social environment may be critical to facilitate post-stroke recovery. As plasma corticosterone level lowered in Stroke with Naive rats, compared to Stroke with Stroke rats, sharing of the housing environment with, or frequent visits to, a patient’s room by healthy persons may reduce stress of stroke patients. Lowering the corticosterone levels via social settings may confer beneficial effects for recovery because corticosterone may exacerbate ischemic stroke in an animal model. 51 Such minimally invasive and cost-free post-stroke social setting, with special attention to promoting empathy-like positive outcomes, may be easily incorporated in stroke patient management.
The microbiome may represent as an equally robust environmental factor that may affect social empathy in stroke. Indeed, dysbiosis is seen in gut microbiota after stroke, 52 suggesting that the behavioral and physiological changes in naive rats that co-housed with stroke rats may be due to the transfer of gut microbiota from stroke rats to naive rats. The present study cannot eliminate this possibility and will be a topic of future investigations.
In conclusion, we provided evidence that stroke-induced physiological and behavioral symptoms are socially modulated. The direct experience of stroke surgery together with the cohabitation with a stroke cagemate significantly increased plasma stress levels and produced behavioral pain manifestation as detected by RGS. Moreover, this combined painful experience of surgical and emotional stroke activated RAGE-dependent stress cascades in the thymus which potentially caused functional impairments of the immune system (Figure 5). These results highlight the importance of an appropriate social setting for stroke patients in order to limit the complications triggered by environmental stress contributing to an already massive pathophysiological consequence of an ischemic insult.
Transfer of stroke-induced stress. Stroke-induced physiological and behavioral symptoms are socially modulated. Housing a stroke rat with a naïve rat significantly reduced plasma corticosterone levels in the stroke rat and locomotion in the naïve rat. However, RGS scores were not affected. Additionally, RAGE- and NF-kB-dependent stress cascades are upregulated in the thymus, suggesting transfer of immune response to the non-stroke cagemate when housed with a stroke rat. Black dot: no significant difference.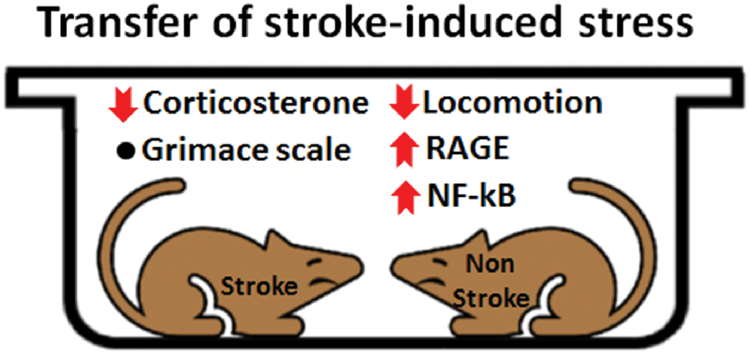
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (NIH) [1R21NS089851] and [5R01NS071956].
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
KS and CVB designed the studies and wrote the manuscript. KS, NT, HI, JPT, JYL, SZ, SC, YK, and CVB analyzed all data. KS, NT, HI, JPT, JYL, PRS, YK, and CVB participated in discussion of results. KS, NT, HI, YK, and CVB performed surgeries, behavioral testing, histology, ELISA, and Western immunoblotting. All authors reviewed the manuscript.
