Abstract
The ischemic penumbra is both a concept in understanding the evolution of cerebral tissue injury outcome of focal ischemia and a potential therapeutic target for ischemic stroke. In this review, we examine the evidence that angiogenesis can contribute to beneficial outcomes following focal ischemia in model systems. Several studies have shown that, following cerebral ischemia, endothelial proliferation and subsequent angiogenesis can be detected beginning four days after cerebral ischemia in the border of the ischemic core, or in the ischemic periphery, in rodent and non-human primate models, although initial signals appear within hours of ischemia onset. Components of the neurovascular unit, its participation in new vessel formation, and the nature of the core and penumbra responses to experimental focal cerebral ischemia, are considered here. The potential co-localization of vascular remodeling and axonal outgrowth following focal cerebral ischemia based on the definition of tissue remodeling and the processes that follow ischemic stroke are also considered. The region of angiogenesis in the ischemic core and its surrounding tissue (ischemic periphery) may be a novel target for treatment. We summarize issues that are relevant to model studies of focal cerebral ischemia looking ahead to potential treatments.
Introduction
The vascular events occurring within the ischemic penumbra following focal cerebral ischemia (ischemic stroke) have not been well worked out, yet their participation holds promise for new treatment approaches that could preserve cerebral tissue function. Among these events are potential contribution(s) of changes in the microvasculature, including focal “no-reflow,” new vessel formation, and vascular remodeling to tissue recovery. Here we address the development of new vessels in the setting of focal ischemia. Angiogenesis after ischemia could be either a pathological process contributing to the post-ischemic injury to the neuropil, or an attempt to limit injury, or both, or perhaps to recover tissue function. It may also have little benefit.
In this review, we will examine the recent evidence that angiogenesis can contribute to beneficial outcomes following focal ischemia in model systems. We also describe the co-localization of vascular remodeling and axonal outgrowth following focal cerebral ischemia based on the definition of tissue remodeling and the processes that follow ischemic stroke (the “maturation phenomenon,” reviewed by Ito et al.).1,2 We also discuss evidence supporting the notion that post-ischemic angiogenesis might contribute to cerebral tissue recovery.
Hypotheses
It has been proposed, based on observations in brain development,3,4 that angiogenesis is necessary for functional recovery after cerebral ischemia. However, the purpose and outcomes of angiogenesis may be different in the ischemic core and the penumbra. Hypotheses central to this notion that could be tested are that (1) the contributions of angiogenesis to tissue and functional preservation might differ with the region and degree of ischemic injury, (2) while apparently coincident, angiogenesis and axonal recovery may indeed be discoordinated, (3) angiogenesis may appear to occur in the ischemic core, but in fact is associated with mini-penumbras within these core regions, and/or (4) axonal recovery may occur independent of angiogenesis along established microvessel supply routes.
Ischemic core and penumbra
The ischemic penumbra was first defined by Astrup et al. 5 as a zone of metabolically compromised tissue around the more densely affected ischemic center or core, with limited neuronal damage if the regional cerebral blood flow (rCBF) was restored by rapid therapeutic intervention (in that case, restitution of blood volume). This concept originated from electrophysiological studies in non-human primates with induced graded reduction in rCBF by partial exsanguination. 6 That maneuver revealed CBF thresholds distinguishing: (i) a lower threshold, due to ion-pump failure, that was associated with tissue infarction, and (ii) an upper threshold, denoted by electrical failure, that was associated with preserved tissue structures. 6 The zones of complete electrical failure and K+ release with functional inactivation, but not yet cell death, define the ischemic penumbra in the neocortex. Clinically, the diffusion/perfusion mismatch using magnetic resonance (MR) imaging or functional impairment, biochemical disturbances, tissue damage, and the duration of critical perfusion by positron emission tomography (PET) provide evidence of the putative ischemic penumbra.7–9
More recently, both experimental and clinical settings of focal cerebral ischemia have demonstrated that early after onset, the ischemic core and penumbra appear heterogeneous, each containing islands of “mini-cores” and “mini-penumbras.” 10 Although not fully characterized, it is a fair assessment that these may represent varying spatiotemporal susceptibilities to injury and cell death. 10
Definitions of the ischemic penumbra
Definitions of ischemic penumbra in use.

The term “ischemic penumbra” has also been used for a region consisting of stratified layers or zones, some of which exhibit high levels of specific molecules of interest that might contribute to injury. These include the zone of selective cell death with alterations in heat shock protein (HSP)-70 expression or hypoxia inducible factor (HIF) related to the zone of spreading depression. 12 In the HIF zone, for instance, HIF stimulates vascular endothelial growth factor (VEGF), inducible nitric oxide synthase (iNOS), and erythropoietin (EPO). VEGF and EPO have been associated with vascular remodeling in vitro and in vivo.13,14 In model studies in rodents, the “ischemic penumbra” has been taken by some investigators as regions peripheral to the core, whether in the cortex or in subcortical tissues (e.g. the basal ganglia), without a clear reference to recovery. However, care must be taken here as (i) the structures do not receive a comparable cerebral vascular supply to that of humans or non-human primates and (ii) often no proof is given that the peripheral regions have metabolic features of the “penumbra” as defined by Astrup et al.
Definitions of the ischemic core
Although the ischemic core is defined as a region or regions below perfusion threshold that correlate with characteristic electrophysiological changes, for practical reasons, in animal models, the core has also generally been defined as regions lacking microtubule-associated protein (MAP)-2 (MAP-2-negative region),15,16 regions displaying deoxyuridine triphosphate (dUTP)-positive cells or lesions, 17 or terminal deoxyribonucleotidyl transferase-mediated dUTP-digoxigenin nick end labeling (TUNEL)-positive lesions 18 depending upon the setting and model system used. Furthermore, several groups have demonstrated that the MAP-2-negative core or dUTP/TUNEL-positive ischemic core is not a homogeneous entity.19,20 Evidence of new vessel formation (called “angiogenesis” by some) has been observed in MAP-2-negative or dUTP+ regions.18,20–24
Abumiya et al. 19 were among the first to demonstrate that markers associated with angiogenesis can appear within hours of the onset of focal ischemia in the ischemic core and periphery, in the awake non-human primate, and are highly significantly associated with activated microvessels, independent of time, after focal ischemia.
Periphery versus core
Experimental data from modeling studies, as well as patient imaging work, indicate that the core and penumbra are dynamic. 10 The definitions of the ischemic core and penumbra, in particular of the ischemic penumbra, may be different for each study. The border of the ischemic core may involve the core itself, the penumbra, or both. The definition of the “border” of the ischemic core can overlap with the ischemic penumbra depending upon the experimental setting. Without metabolic features of the “penumbra,” the “ischemic periphery” is assumed to be a suitable expression that refers to tissue surrounding the ischemic core. However, importantly, with this understanding, the notion of “mini-cores” and “mini-penumbras” may be lost.
The neurovascular unit
The “neurovascular unit” is a conceptual anatomic framework that connects microvessels (endothelium, the basal lamina matrix, astrocyte end-feet, and pericytes) via intervening astrocytes to neurons and their axons, as supported by other cells (e.g. microglia, oligodendroglia).25–35 These are arrayed in networks. Efferent aspects of this arrangement are encompassed by “neurovascular coupling,” while the afferent activities have so far best been displayed by the consequences of restricted nutrient delivery, increased microvessel permeability, matrix protease expression, adhesion receptor change, and the consequences of vascular injury.30,36 Regional differences in the arrangements of neurovascular units in the CNS are likely, that could reflect endothelial cell function, matrix composition, and neuron vulnerability. Although, there is still little normative information about this in three-dimensions among various species.
It has been hypothesized that acute or chronic disruption of microvessel flow and structure can have irreversible effects on astrocyte and neuron function and survival. 37 In the core, for instance, neurovascular units can be irreversibly damaged due to flow cessation and its consequences, while in the penumbra, a proportion of neurovascular units seem viable. 10
Heterogeneity of the microvascular responses
The responses of the microvasculature in the territory-at-risk (e.g. in the striatum) to focal ischemia are very rapid, nearly as rapid as those of the neurons they supply. Within the core regions, in the first minutes after the onset of ischemia, changes in endothelial cell receptor presentation, matrix degradation, and detachment of astrocyte end-feet occur heterogeneously in pockets (“mini-cores”).10,38 Somewhat later, within 4 h after ischemia onset and large artery recanalization, microvessel obstruction can occur. 39 The initial microvessel responses are neither simultaneous nor homogeneous. This suggests that in the early moments following arterial occlusion, the entire “core” is studded with pockets of “penumbra,” and that these “mini-cores” and their “mini-penumbras” evolve dynamically and heterogeneously depending upon local differences of microvessel perfusion. 10 This implies that if not arrested, these mini-cores can grow into their respective mini-penumbras and consolidate to encompass a larger region of injury. In this evolution, signals are generated early that are consistent with new microvessel formation. 19
Vascular remodeling
New blood vessels form by three related mechanisms: (i) angiogenesis, which refers to a combination of the sprouting of new vessels from pre-existing capillaries during a reactive phase, and longitudinal extension of pre-existing vessels in a process termed intussusception,40–42 (ii) vasculogenesis, which refers to initial events of vascular growth in which endothelial cell precursors (angioblasts) differentiate and assemble into primitive vessels during embryogenesis, 43 or (iii) arteriogenesis, which refers to the formation of mature arteries from pre-existent interconnecting arterioles after an arterial occlusion. 44 These forms of new vessel formation have been reviewed elsewhere.45,46 During embryology, vasculogenesis is essential for organ development. Leptomeningeal arteriogenesis may be inducible after cerebral ischemia, 47 although anatomical features of the baseline collateral circulation are a potentially important consideration in the response to focal ischemia.48,49 Angiogenesis and arteriogenesis have been observed after ischemic stroke. Therefore, these two processes can be considered reactive to the signal ischemic event.
Angiogenesis and its quantitation
Studies conducted with animal models, as well as human patients, have been undertaken with the notion that angiogenesis following focal cerebral ischemia could facilitate functional recovery.
To evaluate angiogenesis, some investigators have measured the density of microvessels (microvessel number/unit area) in territories of interest. Microvessel density increases by 14 days after cerebral ischemia in the border of the ischemic core and has been reported to be associated with increased numbers of macrophages.50,51 The presence of a higher density of new vessels in the ischemic periphery and its persistence in the ischemic brain from 4 to 28 days following arterial occlusion in rat and mouse models have been observed with functional recovery following ischemic stroke upon administration of VEGF.52,53 Although the definition of the peri-infarct region is not clear in many studies, increased microvessel density in the peri-infarct region has been reported and has been attributed to or correlated with longer survival times in ischemic stroke patients.54–56
Another approach to the quantitation of new vessel formation following focal ischemia has employed the demonstration of immunoreactive collagen IV, as a measure of microvessel formation and/or presence, and the association with cell-specific BrdU incorporation. 57
Cell responses to cerebral ischemia within the neurovascular unit.
Responses of microvessel integrin expression to ischemia or hypoxia.

Angiogenesis: Receptors and stimulators
Angiogenesis involves the coordinated remodeling of the basal lamina matrix with extension of endothelial cells behind tip cells, 63 in conjunction with the investiture by pericytes, to generate new blood vessels. 35 A coordinated set of stimuli and receptors are involved.
VEGF has been implicated in endothelial cell proliferation, permeability, and angiogenesis (reviewed by Adams and Alitalo).64–66 In mammals, the VEGF family comprises five members: VEGF-A, VEGF-B, VEGF-C, VEGF-D, and placenta growth factor. These members have different functions. While VEGF-A (VEGF) participates in endothelial cell proliferation, permeability, and angiogenesis, 66 VEGF-B mediates embryonic angiogenesis in myocardial tissue, and VEGF-C predominantly mediates lymphangiogenesis. 66 VEGF plays roles in angiogenesis in cerebral ischemia.43,67 In vitro studies have demonstrated that after oxygen–glucose deprivation, VEGF derives from endothelial cells, 68 neurons, 69 astrocytes,20,70 and microglia. 20 Thus, after cerebral ischemia, both VEGF and the VEGF receptor (VEGFR) are found on endothelial cells,20,21,61,71 neurons,20,61,69,71 astrocytes,20,61 and microglia.20,61,72 The role of pericytes in this respect is fully unknown. Binding of VEGF to the receptor tyrosine kinase VEGFR-2 (also known as KDR or Flk1) promotes endothelial proliferation and angiogenesis.73,74 This function is counteracted by the receptor VEGFR-1 (also known as Flt1), which has higher affinity for VEGF, but weak tyrosine-kinase activity.73–75 VEGFR-1 also exists in a secreted, catalytically inactive and, therefore, inhibitory isoform.74,75 The interactions of VEGF with VEGFR-2 induce angiogenic sprouting by guiding filopodial extension from endothelial cells at the tips of the vascular sprouts in vitro. 76 Integrin αvβ3 binds directly to VEGFR-2 for receptor activation and downstream signaling of angiogenesis in the presence of VEGF. 77 VEGF also mediates increased vascular permeability and blood–brain barrier disruption via increased intracellular calcium, 78 src activation, 79 and unknown mechanisms.52,80 Incidentally, the participation of matrix metalloproteinase-9 up-regulation has been suggested 61 ; however, the exact involvement is still unclear. Hence, VEGF may be a double-edged sword after cerebral ischemia.
During development, VEGFR expression precedes the appearance of angiopoietin receptors on nascent blood vessels. 81 Here the tyrosine kinase receptors Tie-1 and Tie-2 are expressed in relation to the ligands angiopoietin-1 (Ang-1) and Ang-2.45,82 Tie-1 and Tie-2 are expressed on endothelial cells. In the CNS, capillary integrity is maintained by the constitutive interaction of Ang-1 from astrocytes with the Tie-2 receptor on endothelial cells. 83 Null preparations for Tie-2 and Ang-1 fail to develop perivascular cells, although vasculogenesis occurs during development. 84 Tie-1 is required for vascular network formation. Murine Tie-2−/− constructs fail to establish the integrity of the developing endothelium resulting in edema and hemorrhage, 85 consistent with the premise that Ang-1 and Tie-2 regulate capillary tubule formation during vasculogenesis. 86
Reports have described that by two days after cerebral ischemia, both VEGFRs are induced in microvessels in the peri-infarct regions with strong expression at three days post-ischemia. 21 By seven days after cerebral ischemia, expression levels of VEGFR-1 and -2 return to normal. A separate group also demonstrated that both VEGFR-1 and -2 are induced in endothelial cells at the border of the ischemic core and peripheral regions at one and three days post-ischemia. 87 Moreover, increasing levels of Ang-1 mRNA were detected at 24 h of ischemia and persisted for 28 days. Ang-2 mRNA was not detected until 24 h after ischemia onset, and the increased Ang-2 mRNA persisted for 28 days after ischemia. 88 Ang-2 has been shown to increase blood–brain barrier compromise, vascular permeability, and injury volume. 89 Tie-1 and Tie-2 were expressed in the ischemic cortex, with Tie-2 appearing in the outer cortical layers and Tie-1 detected in layers II-IV on capillary-like structures. 90 As with brain development, angiogenesis is a response to cerebral hypoxia and contributes to the pathology of injury development.
Notable also are the signaling responses of microvessel pericytes to ischemia and innate inflammation.35,91 Pluripotent cells embedded in the basal lamina matrix of cerebral capillaries, pre-capillary arterioles and post-capillary venules contribute to the integrity of the blood–brain barrier; these cells undergo transformation and migrate in response to injury.92–94 Pericyte migration and proliferation are required for the branching and generation of new microvessels during angiogenesis. 63 This is also stimulated during hypoxia.
Integrin receptors and changes in the setting of new vessel formation
It is noteworthy that in the awake non-human primate model, by 2 h after middle cerebral artery (MCA) occlusion in the basal ganglia, where microvessel-associated β1-integrin antigen decreased rapidly in the core and significantly, β1-integrin transcripts and αv integrin subunits were up-regulated within the boundaries between individual ischemic minicores.19,95 Loss of VLA-1, -3, and -6 occurred acutely relative to the loss of matrix protein immunoreactivity from the microvessel basal lamina within hours after ischemia onset. 95 In contrast, the fibronectin receptor integrin αvβ3 expression in cerebral microvessels that was minimal under normoxia, increased significantly in the early hours following MCA occclusion.19,96
α5β1-integrin expression under conditions of ischemia
18
and under hypoxia97–99 has been studied. The α5-integrin subunit, which is paired with the β1-integrin subunit in vascular remodeling, is expressed by capillary endothelial cells in dUTP+-positive tissue following cerebral ischemia in the mouse seven days after ischemia onset.
18
Increased α5-integrin subunit expression after cerebral ischemia was closely correlated with the number of Ki67-positive endothelial cells, in the border of the ischemic core.
18
Integrin α5β1 participates in angiogenesis in the border of the ischemic core following the onset of focal cerebral ischemia,
18
while integrin-α5β1 is required for angiogenesis during embryogenesis.97,100 The integrin α5β1 (i) appears transiently during embryogenesis of the brain vasculature, and (ii) underlies the vasculogenesis associated with cerebral tissues exposed to chronic hypoxia.
97
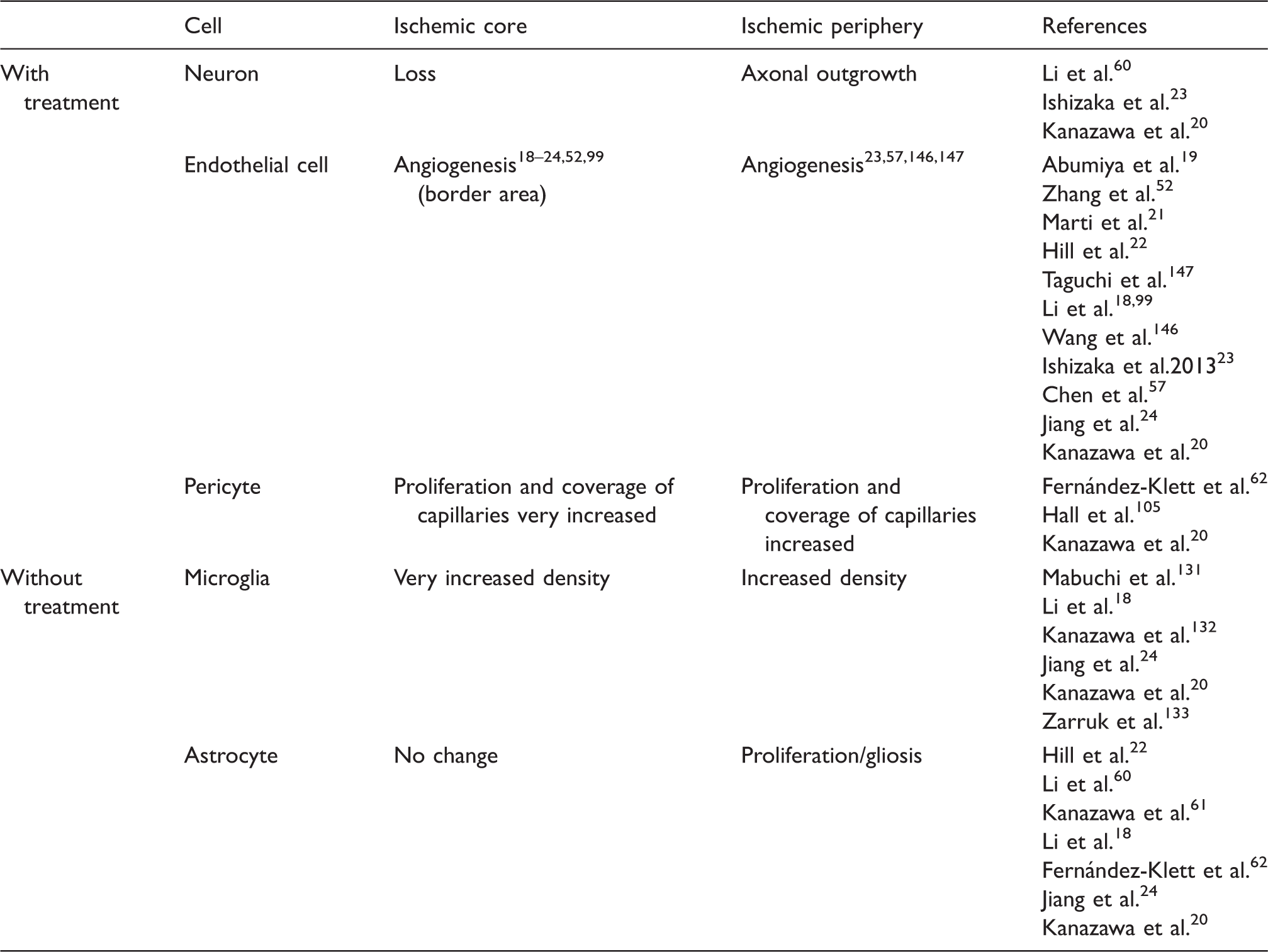
Angiogenesis induced by tumor necrosis factor-α (TNF-α) and VEGF could occur via up-regulation of α5-integrin and binding between integrin α5β1 and Ang-2 in endothelial cells.98,101 Finally, endothelial cell/microvessel proliferation and subsequent angiogenesis are detected in the border of the MAP-2-negative ischemic core, but not in the ischemic periphery, following cerebral ischemia alone (i.e. without therapeutic intervention) (Figure 1(a)).18–21
A scheme for angiogenic responses following focal cerebral ischemia (modified from reference Kanazawa et al.
20
). (a) A proposed scheme depicting the location of angiogenesis after cerebral ischemia. The ischemic core can be defined as the deoxyuridine triphosphate (dUTP)-positive or microtubule-associated protein 2 (MAP-2)-negative ischemic tissue. Angiogenesis (red lines) occurs at the border of the ischemic core (orange) and the peripheral regions. Endothelial sprouting occurs prior to pericyte coverage of capillaries (purple lines). Angiogenic endothelial cells following cerebral ischemia express integrin α5β1 (green columns) in capillaries. Thus, the dUTP-positive or MAP-2-negative ischemic core consists of the angiogenesis-positive ischemic core and angiogenesis-negative ischemic core (black) which can proceed to irreversible change. (b) A proposed depiction of angiogenesis and axonal outgrowth (green cells: neurons and axons) in the setting of treatment interventions after cerebral ischemia. Interventions have been proposed to promote angiogenesis (red lines), with integrin α5β1 expression (green columns) and pericyte coverage of capillaries (purple lines) in angiogenesis vessels in the border of ischemic core. Axonal outgrowth by neuronal cells (green) in association with interventions could be found in the ischemic periphery.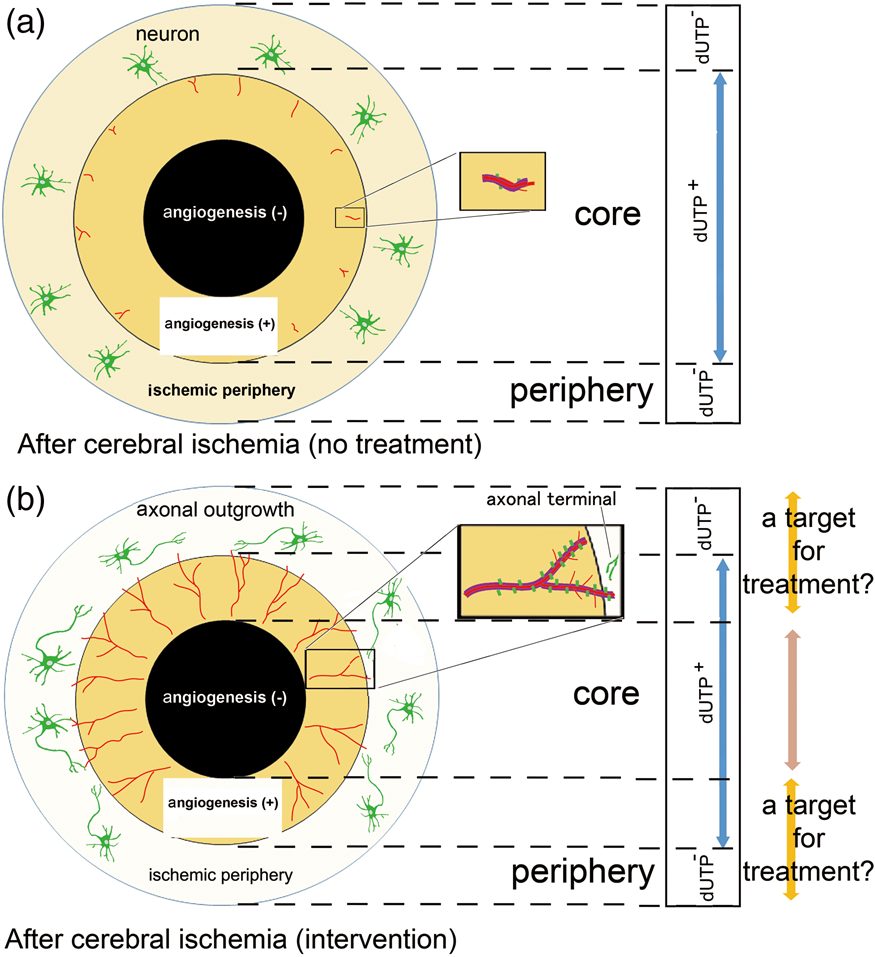
In addition, up-regulation of integrin α5β1 has been associated with the up-regulation of the tight junction proteins claudin-5 and ZO-1, and is associated with Ang-1 in vascular remodeling. 102 Fibronectin, α5β1-integrin, and αvβ3-integrin contribute to endothelial sprouting that is distinct from VEGF-mediated angiogenesis, yet important for the growth of tumors in the cancer setting. 103
Angiogenesis in response to focal cerebral ischemia
New blood vessels that form after focal ischemia appear by days 4 to 7 in peri-infarct regions. 102 Tsutsumi et al.40,41 have demonstrated capillary bud formation by seven days in the cerebral ischemic region in a canine model. Endothelial sprouting and increased capillary density 10 days after cerebral ischemia have been detected prior to the coverage of endothelial cells by pericytes. 104 At the same time, there is a highly significantly coordinated increased expression of VEGF and αv-integrin subunits in the same microvessels that also display proliferative signals, as indicated by the presence of PCNA. 19 The significance of these interactions is emphasized by the fact that they are independent of time from MCA occlusion, occurring from 2 h through seven days post-ischemia. These events occur in 5 to 20-µm diameter microvessels, primarily in the ischemic core and much less in the peripheral regions. Based on the diameter, it seems that the site of angiogenesis is most consistently located in the capillaries. 105 Hence, elements known to be associated with angiogenesis/new vessel formation, including VEGF and integrin subunit αv, appear early following occlusion of the MCA in the awake non-human primate. 19
Interactions with oligodendrocytes
As the concept of neurovascular unit suggests, oligodendrocytes do not stand alone. Rather they actively interact with neighboring cells to allow the integrated control of white-matter function as demonstrated in subcortical ischemia 106 and traumatic brain injury. 107 In addition, oligodendrocyte precursor cell (OPC) migration appears to occur along vascular tracks, and does not require the participation of pericytes during vasculogenesis in the developing CNS. 108 Crosstalk among axons, microvessels, and oligodendrocytes may be important for remodeling and plasticity in post-ischemic injury zones. Proof of cross-cellular communication requires the further teasing apart of complex inter-relationships. This may be accomplished with high-quality ex vivo tissue slice and in vivo preparations.
Return of axons
Axonal regeneration stands in contrast to “axonal sprouting,” which is axonal outgrowth from uninjured neurons in response to injury.109,110 Regeneration of axons following injury in the CNS is limited.110–113 Methodologically, distinguishing between regenerating and sprouting axons is important, because while regeneration failure is the norm in the adult mammalian CNS, axonal sprouting might explain the partial spontaneous recovery of function that is observed following an ischemic event.114,115
Pioneering work by David and Aguayo and Benfey and Aguayo et al.116,117 demonstrated that adult mammalian CNS neurons, which normally do not regenerate, are able to grow for long distances into the permissive environment of a peripheral nerve graft. More recently, Izumi et al. 118 demonstrated that, in vitro, dopaminergic neurites can extend along striatal neurons in the paired-cultures of mesencephalic cells with striatal cells, and that these extensions appear to depend on integrin α5β1.
Therefore, the term “axonal outgrowth” may be suitable for recovery in ischemic settings. Axonal outgrowth in the absence of intervention does not appear until 14 to 28 days after cerebral ischemia even in the ischemic periphery (Table 2).20,119 Most studies have evaluated axonal outgrowth by limited immunohistological means and not functional responses. As the ischemic core is a MAP-2-negative region in experimental settings, axonal outgrowth within the core would not be expected to be observed. However, when angiogenic vessels are in the core, it would be proposed that these could occur in mini-penumbras. This possibility has not yet been tested. Studies generally have not simultaneously evaluated microvessel responses, and the relationship between angiogenesis and axonal outgrowth.
Angiogenesis and axonal outgrowth/neurogenesis
It might be expected that angiogenesis would be essential for ischemic brain repair as this event allows increased blood flow and metabolic nutrients to the stricken regions. Angiogenesis has been purported to be associated with neurogenesis,59,120,121 and the assertion that neurogenesis is dependent on new vessel formation for long-term survival. 122 However, it is speculative whether angiogenesis and axonal outgrowth/neurogenesis could occur together and be coordinated in space and time in vivo.
Experimental systems
Using ex vivo co-culture systems, possible interactions between angiogenesis and neurogenesis have been examined.123,124 For instance, recently, some characteristics of a three-dimensional culture model, including neurons, endothelial cells, and extracellular matrix, have been described. 124 In three dimensions in vivo, features of angiogenesis and neurogenesis appear to be coupled. 59 Furthermore, recently it has been reported that oligodendrocyte precursor cells (OPCs) require vasculature for their extensive migration through the brain and spinal cord during development in whole tissue preparations. 108 After ischemia, VEGF from vessel components might prompt axonal outgrowth.68,125,126 Recently, Lei et al. 127 and Tan et al. 128 reported that axons bound to laminin expressed a β1-integrin, and that laminin/β1-integrin signaling might contribute to axon development and promote axonal outgrowth in vitro. However, it is not known whether β1-integrins behave in this way in brain tissue. It is known for endothelial cells that integrin expression is matrix-dependent in vivo and in vitro.129,130 Angiogenesis in the ischemic core may be associated with axonal outgrowth via laminin/β1-integrin from the periphery. It raises the possibility that angiogenesis and neurogenesis are coupled by VEGF and laminin/β1-integrin signaling, although proof that this is relevant in vivo has not appeared. In typical immunohistochemical preparations, neurons do not display β1-integrins.
Microglia, axons, and angiogenesis
The effects of discrete interventions on neuronal/axonal and microvessel components of the neurovascular unit after focal cerebral ischemia.
Ischemic core versus periphery in vitro and in vivo
If one defines the ischemic core as a region or regions below adequate tissue perfusion threshold and the ischemic periphery as surrounding the ischemic core tissue, it is technically difficult to distinguish between ischemic core and periphery in in vitro models. However, the levels of O2 deprivation and/or nutrient supply can have differential effects on cell survival in vitro that could mimic aspects of the core and penumbra. To date, focal cerebral ischemic models in vivo would be the appropriate setting for this evaluation. However, the study of molecular and microcirculatory communication between the ischemic core and periphery is very relevant and may be facilitated by in vitro modeling.
Angiogenesis and recovery in the subacute to chronic phase of cerebral ischemia
It has been demonstrated that angiogenesis is a necessary early process for recovery after cardiac ischemia 138 or peripheral artery disease. 139 This raises the possibility that enhancement of angiogenesis could be a potential strategy to facilitate functional recovery after ischemic stroke.120,121
Theoretically, the vasculature adjacent to the injury zones may play roles in new vessel formation. The brain employs a low pressure/high flow vascular system in which the established Circle of Willis and pial collateral vasculature provide protection from significant focal ischemic injury.140,141 The volume of ischemic injury is in part determined by collateral protection from arterial segments and venous outflow separate from the arterial supply to the territory-at-risk. 142 While the pial anastomoses that provide potential collateral protection of the cortex are under genetic control, 49 the vascular network for the protection of the striatum in humans and higher mammals is currently unknown. Furthermore, in small animal species, it has been shown that anesthesia can increase the cerebral blood volume (CBV) by 10 to 36% from latent channels.143,144
Important to the processes of recovery and limitation of injury is capillary density. In a rat model of focal cerebral ischemia, arteriolar collateral growth and new capillaries restored perfusion in the borders of the ischemic regions 30 days after ligation of the MCA branches. 145 Additionally, in a model of focal cerebral ischemia in the male Sprague-Dawley rat, increased capillary density in the cortical peri-infarct region was associated with partial functional recovery by 7 and 14 days post-MCA occlusion. 146
Interventions
A number of reports using rodent models of focal ischemia have described the effects of pharmacological and cell-based treatments on the ischemic brain tissue of animals that appeared to increase angiogenesis in the border of the ischemic core. VEGF and/or cell-based interventions appeared to induce angiogenesis at the border of the ischemic core, and axonal outgrowth in the ischemic periphery (Figure 2(b)).20,126 Several groups have demonstrated that in ischemic brain tissue of animals receiving cell-based or pharmacological treatments, mediators might be associated with a 130–400% improvement in functional outcome compared to controls (Table 4).13,20,23,52,57137,146–149 However, the direct causal relationships between angiogenesis and functional recovery have not yet been proven.
Roles of VEGF in angiogenesis of cerebral microvessels. (a) After focal ischemia, vascular endothelial growth factor (VEGF) is secreted from endothelial cells, astrocytes, neurons, and/or microglia. However, there is no direct evidence whether VEGF from neurons enhances angiogenesis because axonal outgrowth and angiogenesis can differ in the location after cerebral ischemia in vivo. (b) VEGF participates in angiogenesis in the brain following focal ischemia. The integrin αvβ3 is expressed in coordination with VEGF. Capillary integrity is maintained by the constitutive interaction of angiopoietin-1 (Ang-1) from pericytes with the Tie-1 and Tie-2 receptor on endothelial cells.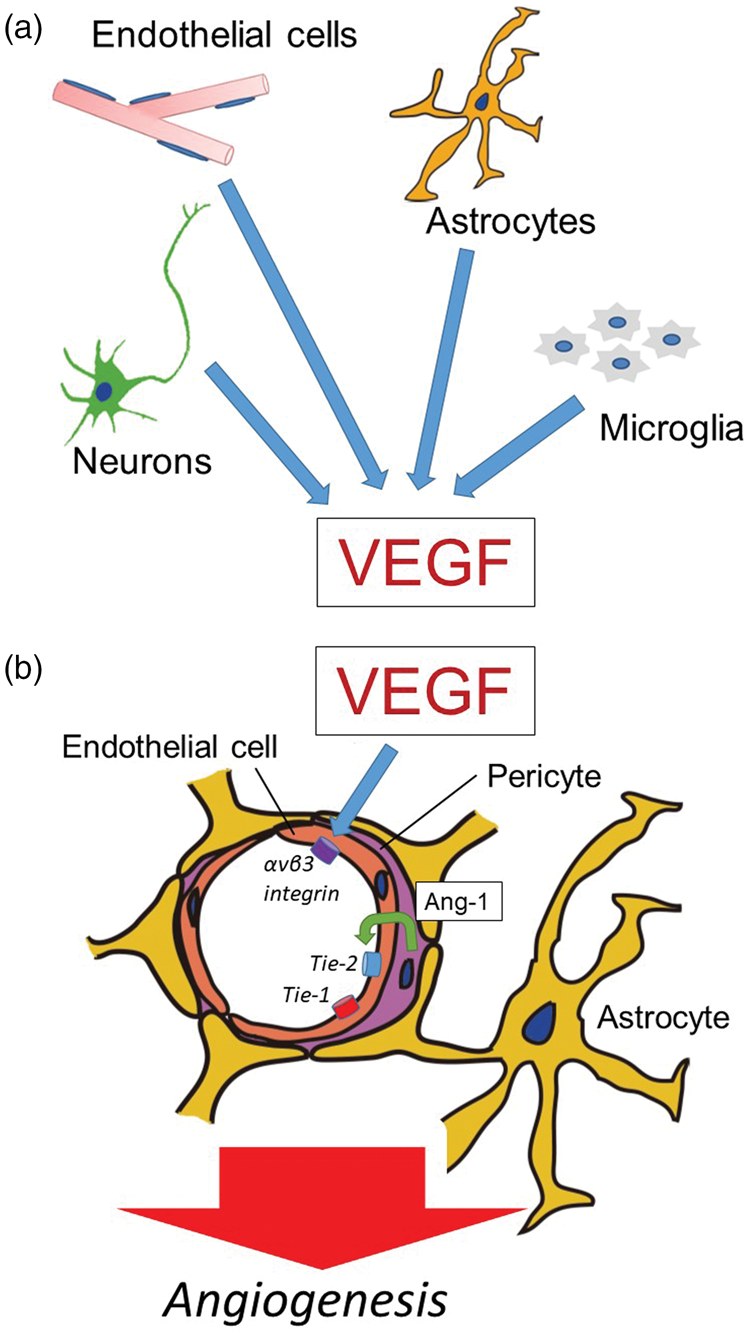
Intra-arterial injection of cultured mesenchymal stem cells appeared to enhance angiogenesis and associated axonal outgrowth in the peri-infarct regions during the chronic post-ischemic period following MCA occlusion in rats (Table 4). 23 In a separate study, systemic administration of human cord blood-derived CD34+ cells or neural progenitor cells following ischemic stroke also enhanced angiogenesis in the ischemic border and suggested a favorable environment for axonal outgrowth.147,148 Axonal outgrowth was, in turn, observed to have effects around angiogenesis, although a causal relationship was not identified.
Systemic administration of valproate (VPA), a histone deacetylase (HDAC) inhibitor commonly used to treat seizures and bipolar disorder, was observed to further increase capillary density at 14 days post-MCA occlusion and to improve functional recovery in a rat model. 146 No other times were examined. In contrast, 2-methoxyestradiol (2ME2), a natural metabolite of estradiol, was shown to inhibit hypoxia-dependent HIF-1 stabilization and appeared to inhibit the effects of VPA on capillary density and on rotarod performance. 146 Separately, it has also been reported that new vessel formation, defined as collagen IV structures with BrdU+ endothelial cells, was significantly increased in peri-infarct regions by apelin-13, and was associated with the appearance of VEGF 14 days after ischemic stroke. 57 A Sonic Hedgehog agonist delivered post-stroke enhanced angiogenesis in the border of the ischemic core and also increased functional recovery in a mouse focal cerebral ischemia model. 149 The observation that increased vascular density can improve functional recovery following several unrelated interventions after cerebral ischemia have not yet proven causality.
Direct interactions between angiogenesis in the ischemic core and axonal outgrowth in the ischemic periphery have not been studied. It is unknown whether angiogenesis in the ischemic periphery could aid in shrinking the ischemic core. However, angiogenesis might provide a suitable environment that triggers axonal outgrowth, although this is not conclusively known. In separate studies, axonal outgrowth with functional recovery was observed after spinal cord injury by delivered chondroitinase ABC 111 and grafted Schwann cells 112 or human neural stem cells. 113 Those observations raise the possibility that agents and/or cell-therapies that prompt angiogenesis at the border of the ischemic core, and axonal outgrowth in the ischemic periphery, may offer opportunities for treatment although the precise mechanisms have yet to be clearly identified (Figure 1(b)).
Quality of the evidence so far
A significant limitation of these studies so far is the lack of clear evidence for a direct link mechanistically among angiogenesis, axonal outgrowth, and functional recovery. To date, the majority of reports that have examined the interaction of an intervention with the development of additional angiogenesis in association with cortical focal ischemic lesions, have been correlational or associational, and have not shown causality with regard to outcome improvement.
After focal cerebral ischemia, specific interventions including the administration of cell-based treatments or VEGF appear to (i) induce axonal outgrowth following angiogenesis, or (ii) have been accompanied by proliferation of pericytes that cover the endothelial cells on the border of the ischemic core, for instance (Figure 1(b)).18,20,23,98 Several mediators seem to prompt both angiogenesis and axonal outgrowth and communicate with the cells within affected neurovascular units after cerebral ischemia. These include VEGF19,20,69–70,126 (Figure 2(a) and (b)), TGF-β,20,150 Ang-1151,152 (Figure 2(b)), platelet-derived growth factor-B (PDGF-B), 153 and brain-derived neurotrophic factor (BDNF). 154 In other words, each cell in the neurovascular unit could communicate with the other cells by several mediators. 28 This is very similar to the situation displayed by innate inflammatory responses within the neurovascular unit.28,155–157
Other processes
A process termed endothelial-mesenchymal transition (EndMT) has received attention in recent years. During EndMT, the stable interactions of endothelial cells and mesenchymal cells (e.g. pericytes, smooth muscle cells, fibroblasts) can be disturbed when claudin-5, occludin, and VE-cadherin within the junctional complex disaggregate or are lost thereby increasing endothelial permeability.158–160 These events are similar to the changes accompanying increased microvessel permeability during focal ischemia 52 and are seen during the formation of cerebral cavernous malformation. EndMT involves β1-integrins, oxygen-free radicals, angiopoietin-2, TGF-β, and VEGF.161–163 Although contributions of EndMT to microvascular changes after cerebral ischemia have not yet been investigated, elements of EndMT may be seen in post-ischemic angiogenesis as currently described.
Some questions that remain
A number of issues involving the development, presence, and contributions of angiogenesis in the CNS following focal cerebral ischemia, in a number of models and in humans, for (i) injury evolution, (ii) clearance of tissue debris, and (iii) processes leading to functional improvement, have not been rigorously explored or described yet. These include
(1) the location of the core versus peripheral regions, and the timing of changes in these locations/regions for each model and for the ischemic stroke patient, (2) differences in mechanism and outcome between the ischemic core and the penumbra when angiogenesis has been augmented, (3) the sequence and interaction of molecular and cellular mechanisms of angiogenesis after cerebral ischemia, (4) how angiogenesis might work for functional recovery after cerebral ischemia, (5) constructive interactions between angiogenesis and axonal outgrowth after cerebral ischemia, (6) how cell–cell interactions that modulate angiogenesis, axonal outgrowth, and functional outcomes might proceed in response to injury or be inhibited in the normoxic condition, (7) vascular cell-extracellular matrix and neuron interactions that modulate these processes, (8) how the components of the neurovascular unit contribute to angiogenesis, and how they are altered, and (9) whether the new cerebral vessels and axons are functional.
Conclusion
Angiogenesis appears to be stimulated in the border of the ischemic core in rodent and non-human primate models acutely following the onset of focal ischemia. New capillaries are observed 4 to 14 days after cerebral ischemia in these model systems. Furthermore, axonal outgrowth is observed in the ischemic periphery surrounding the angiogenesis. Several interventions appear to enhance angiogenesis and axonal outgrowth and might result in functional recovery; however, causality and specific mechanisms have not yet been described. The regions of angiogenesis in the ischemic core and its surrounding tissue (ischemic periphery) may be novel targets for treatment. However, further discrete focused work to develop clear mechanisms for functional recovery based on angiogenesis is required.
Supplemental Material
Supplemental material for Angiogenesis in the ischemic core: A potential treatment target?
Supplemental Material for Angiogenesis in the ischemic core: A potential treatment target? by Masato Kanazawa, Tetsuya Takahashi, Masanori Ishikawa, Osamu Onodera, Takayoshi Shimohata and Gregory J del Zoppo in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Scientific Research (Research Project Number: 15K19478 and 18K07493), a grant from Takeda Science Foundation, the Bayer Scholarship for Cardiovascular Research, Japan Cardiovascular Research Foundation, Astellas Foundation for Research on Metabolic Disorders, and Young Investigator Okamoto Award, and Medical Research Encouragement Prize of the Japan Medical Association (MK).This work has been supported by parts of the grants NS 053716, NS 038710, and NS 036945 from the National Institutes of Health (GJdZ).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
M Kanazawa: None.
T Takahashi: None.
M Ishikawa: None.
O Onodera: None.
T Shimohata: An academic adviser of the ShimoJani LLC biotech company.
G J del Zoppo: None.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
