Abstract
The majority of stroke patients develop post-stroke fatigue, a symptom which impairs motivation and diminishes the success of rehabilitative interventions. We show that large cortical strokes acutely reduce activity levels in rats for 1–2 weeks as a physiological response paralleled by signs of systemic inflammation. Rats were exposed early (1–2 weeks) or late (3–4 weeks after stroke) to an individually monitored enriched environment to stimulate self-controlled high-intensity sensorimotor training. A group of animals received Anti-Nogo antibodies for the first two weeks after stroke, a neuronal growth promoting immunotherapy already in clinical trials. Early exposure to the enriched environment resulted in poor outcome: Training intensity was correlated to enhanced systemic inflammation and functional impairment. In contrast, animals starting intense sensorimotor training two weeks after stroke preceded by the immunotherapy revealed better recovery with functional outcome positively correlated to the training intensity and the extent of re-innervation of the stroke denervated cervical hemi-cord. Our results suggest stroke-induced fatigue as a biological purposeful reaction of the organism during neuronal remodeling, enabling new circuit formation which will then be stabilized or pruned in the subsequent rehabilitative training phase. However, intense training too early may lead to wrong connections and is thus less effective.
Introduction
For more than 90% of stroke patients, rehabilitative training is the only treatment option to improve lost motor functions. However, the neurobiological mechanisms underlying internal physiological repair processes that are influenced by external rehabilitative stimulation are not well understood. In particular, there is no good scientific basis to causally link type, intensity, and timing of different rehabilitative approaches to functional recovery both in clinical and experimental rehabilitation trials. Individual rehabilitation courses very often still appear as ‘black box experience’. The very divergent outcome of individuals with the same rehabilitative treatment remains unclear. In particular, little is known about the influence of spontaneous ‘self-training’ during daily life as it is often not controlled or recorded and may be also neglected due to frustration.
Traditionally, rehabilitation starts a few weeks after the stroke, when patients move from neurology or internal medicine departments to a specialized rehab clinic. Early on, mild rehabilitation including early mobilization within days after stroke is thought to contribute to the benefit of stroke-unit care 1 and is recommended in many guidelines. However, when to start with which kind of training and which intensity is uncertain as addressed in the literature.2–4 Recently, in a big multicenter randomized controlled clinical trial, early mobilization within 24 h after stroke was compared to a conventional care group. 5 Fewer patients in the early mobilization group had a favorable outcome than those with standard care. Very early mobilization also increased mortality rates for those stroke patients.
Here, we used a Natural Habitat (NH) cage equipped with multimodal training and monitoring devices to study individual courses of rehabilitation and outcome under well-defined conditions in adult rats with large cortical strokes. We examined five different rehabilitative groups with a focus on the timing when the animals were exposed to the enriched environment (EE) – either immediately or starting two weeks post-insult. In addition, animals also received antibodies – either control or neuronal growth promoting Anti-Nogo antibodies. Anti-Nogo-A antibodies have been shown to enhance neuronal fiber growth and improve impaired motor function in various animal stroke models and are part of on-going clinical trials.6–8 A fifth group of rats was exposed to the NH cage for four weeks continuously after the stroke without additional treatment.
We analyzed the anatomical reorganization of the corticospinal tract (CST) system in correlation to the training activity recorded in the NH cage and the detailed characterization of motor recovery as well as blood markers to identify critical time windows to optimize sensorimotor recovery after stroke.
Materials and methods
Animals
A total of 78 adult female Long-Evans rats (200–250 g, 12–16 weeks old, Janvier, France) were included in the present study. Except during the rehabilitation period in the NH cage (see below), animals were housed in groups of two to four in open airflow cages under a 12-h dark/light cycle. Food and water were delivered ad libitum except for restricted periods during the single pellet grasping and staircase training and testing phases. All experimental procedures were approved by the Veterinary Office of the Canton of Zurich, Switzerland. They are in accordance with the Stroke Therapy Academic Industry Roundtable criteria 9 for preclinical stroke investigations.
Experimental set-up
The objective of this study was to examine individual rehabilitative courses and to define optimized rehabilitative protocols. Briefly, rats were trained in the single pellet grasping task, acclimatized to the NH cage (Supplementary Figure 1A) followed by a large photothrombotic stroke destroying a major part of one hemisphere and in particular targeting the entire sensorimotor cortex corresponding to their paw-preference in the grasping task. Immediately after stroke, we subcutaneously implanted osmotic mini pumps with intrathecal catheters delivering either IgG control or Anti-Nogo antibodies or PBS according to the randomization of animals due to their last performance in the single pellet task pre-stroke. All pumps were removed two weeks after stroke. Two days after the stroke, animals were re-tested in the single pellet grasping task and again randomized according to their lesion deficit into early and delayed cohorts by an independent person (not involved in the behavioral or anatomical experiments): animals in the ‘Anti-Nogo parallel’ group received anti-Nogo-A antibodies for the first two weeks after stroke and were housed in the NH cage as a form of EE at the same time. The ‘Control parallel’ group was also housed in the NH cage, but got a control (IgG) antibody instead of the Anti-Nogo-A immunotherapy. Animals in the ‘Anti-Nogo sequential’ group or ‘Control sequential’ group were first treated with the antibodies (Anti-Nogo antibodies or IgG control antibodies, respectively) for two weeks while housed in conventional cages, followed by pump removal and two weeks of housing in the NH cage. In summary, all four groups received antibody treatment for two weeks and were being housed in the EE for two weeks – for two groups (‘Early EE’) both treatments were concurrently delivered, while in the two ‘Delayed EE’ groups antibodies were first applied for two weeks after the stroke and animals were then exposed to the EE in the NH cage. In a fifth group, animals were implanted with pumps delivering PBS and were housed in the NH cage for four weeks starting immediately after the stroke (‘Continuous EE’). This group was included to examine the sole influence of EE for regaining impaired sensorimotor functions.
Anti-Nogo-A and control antibody rats in the ‘Early EE’ and the ‘Delayed EE’ cohort were coded by random numbers. Rats of each cohort were housed together in the NH cage and randomly distributed in the conventional cages. Experimenters were blind for the content of the pumps and the treatments of the animals throughout the experiment.
To test for the degree of recovery of lost forelimb function, animals were retested in the single pellet grasping task 14, 21, and 28–42 days after stroke. At the end of the rehabilitative schedule, the animals were exposed to novel (non-trained) tasks (staircase, narrow beam, plantar heat task), testing their overall functional recovery. Finally, the CST of all animals was traced anterogradely with biotinylated dextran amine (BDA). Rats were perfused three weeks later.
We applied this experimental set-up to N = 4 independent experiments with 14–23 animals per cohort. For the last two experiments, we took blood samples before stroke (baseline), in an acute (7 days), an intermediate (14 days), and in a chronic stage (28–42 days) after stroke to determine changes in blood parameters indicating inflammatory and immune responses. Only animals with a similar, large lesion size and location destroying the sensorimotor cortex as determined by ex vivo magnetic resonance (MR) imaging after perfusion were included (n = 55 of 78 rats).
As there was no statistically significant difference in the outcome of lesions, behavior, and anatomy, the data shown here were pooled from all four experiments. To exclude any investigator driven bias of a study only the person implanting the pumps with either control or Anti-Nogo antibody or PBS randomized animals to the different treatment groups based on the behavioral data provided by the people responsible for behavioral training and assessment. This un-blinded investigator responsible for the ‘distribution code’ was not involved in any further analysis and informed the other blinded investigators at the end of the behavioral, anatomical, and biochemical analysis of the allocation of the animals in the different treatment groups. The single pellet training and testing sessions were video-taped and the video material was analyzed by computer vision specialists who were also blinded to the treatment groups and used un-supervised computer algorithms to quantify the grasping kinematics. This study followed the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines (www.nc3rs.org.uk/arrive-guidelines).
The NH cage
The NH cage was designed to provide a form of EE which allows 24-h activity recording in a three-floor space enabling the close monitoring of individual activity and training profiles of animals. 10 The single floors (each 79 cm × 58.5 cm × 52 cm (high)) were linked through ladders, presenting equipment to train fore and hind limb motor function. Such training equipment, distributed over the whole area, was only accessible through magnetic coil antennas, detecting, and counting the passages of each animal. Supplementary Figure 1A depicts the arrangement in the NH cage: The topmost floor was equipped with a running wheel and a ladder for the ascent to additional platforms connected through wooden beams. In the largest of these platforms, the ‘Penthouse’, food was placed to motivate animals to cross the beams and also train food handling. The first floor contained the training stations for grasping function: the grasping boxes, filled with sugar pellets, and the grasping grid. The elevated grasping grid was reachable through a vertical rope and filled with additional food rewards. An animal house, placed diagonal to the grasping boxes, served as a hiding place. The drinking possibilities (two 1 l bottles) were placed next to the standard food on the ground floor in order to ensure that injured animals which were less willing to move would not be water or food deprived. A raised platform served as basis ladder connecting the ground floor with the first floor and as a gate to the standard cage. During the whole experiment, the cage was located in a standard animal housing facility subjected to a 12 h/12 h light–dark cycle.
Tracking of individual activity profiles in the NH cage
RatTrack – the activity recording system – was designed in collaboration with NewBehavior AG and FBI Science GmbH as described. 10 Activity of rats was detected with RFID transponders (ISO Standard 11784; T-IS 8010 FDX-B, Datamars, Switzerland), eight antennas, and a customized software (OLCUS v2, FBI Science GmbH, Germany). Antennas recognized the specific code of a passaging transponder. They were placed in front of the wheel (‘Wheel tracking’), the two floor entrances (‘Ladder tracking’), the house, the penthouse (‘Penthouse tracking’), the grasping box (‘Grasping box tracking’), and the grasping grid (‘Grasping grid tracking’). The house tracking (a resting and hiding place for the animals) was excluded from the analysis except for the sum of all trackings (‘Total tracking records’).
The OLCUS software recorded the corresponding transponder code and antenna number as well as the time of passage. RFID transponders were implanted above the tail and in the neck region of each rat in the NH cage, allowing discrimination between whole and partial passages. One record (Tracking unit) was defined as detection of both, neck and tail transponders in sequence (first: neck, second: tail). For the implantation of two RFID transponders, animals were shortly anesthetized with isoflurane (3–5%). Transponders were injected subcutaneously in the neck and above the tail. Implantation was done before determination of baseline tracking performance prior to stroke surgery (see experimental set-up, Figure 1(a)).
Strokes induce post-stroke fatigue in rats. (a) Experimental timeline for the five experimental groups (‘Anti-Nogo parallel’, n = 10; ‘Control parallel’, n = 14; ‘Anti-Nogo sequential’, n = 12; ‘Control sequential’, n = 11; ‘Continuous EE’, n = 8, EE = enriched environment rehabilitation in Natural Habitat (NH) cage). (b) Left: Representative coronal MR image depicting the photothrombotic stroke lesion in a rat brain eight weeks after stroke, scale bar = 1000 µm. Right: Stroke lesion size in all five treatment groups. (c) Left: Tracking records of overall activity in animals of the ‘Continuous EE’ group up to four weeks after stroke. Right: Activity in the NH Cage in the first two weeks after stroke for the ‘Anti-Nogo parallel’ and ‘Control parallel’ group compared to activity records for the ‘Anti-Nogo sequential’ and ‘Control sequential’ animals in the third and fourth week after stroke versus baseline. (d) Left: Skilled forelimb training in the grasping boxes pre- and post-stroke. Right: Circadian post-stroke activity normalized to the pre-stroke baseline for all four rehabilitation groups. Data are presented as means ± s.e.m.; statistical evaluation was carried out with one-way (b, c left graph) and two-way ANOVA (c right graph, d) repeated measure followed by Bonferroni post hoc; asterisks indicate significances: *P < 0.05, **P < 0.01, ***P < 0.001.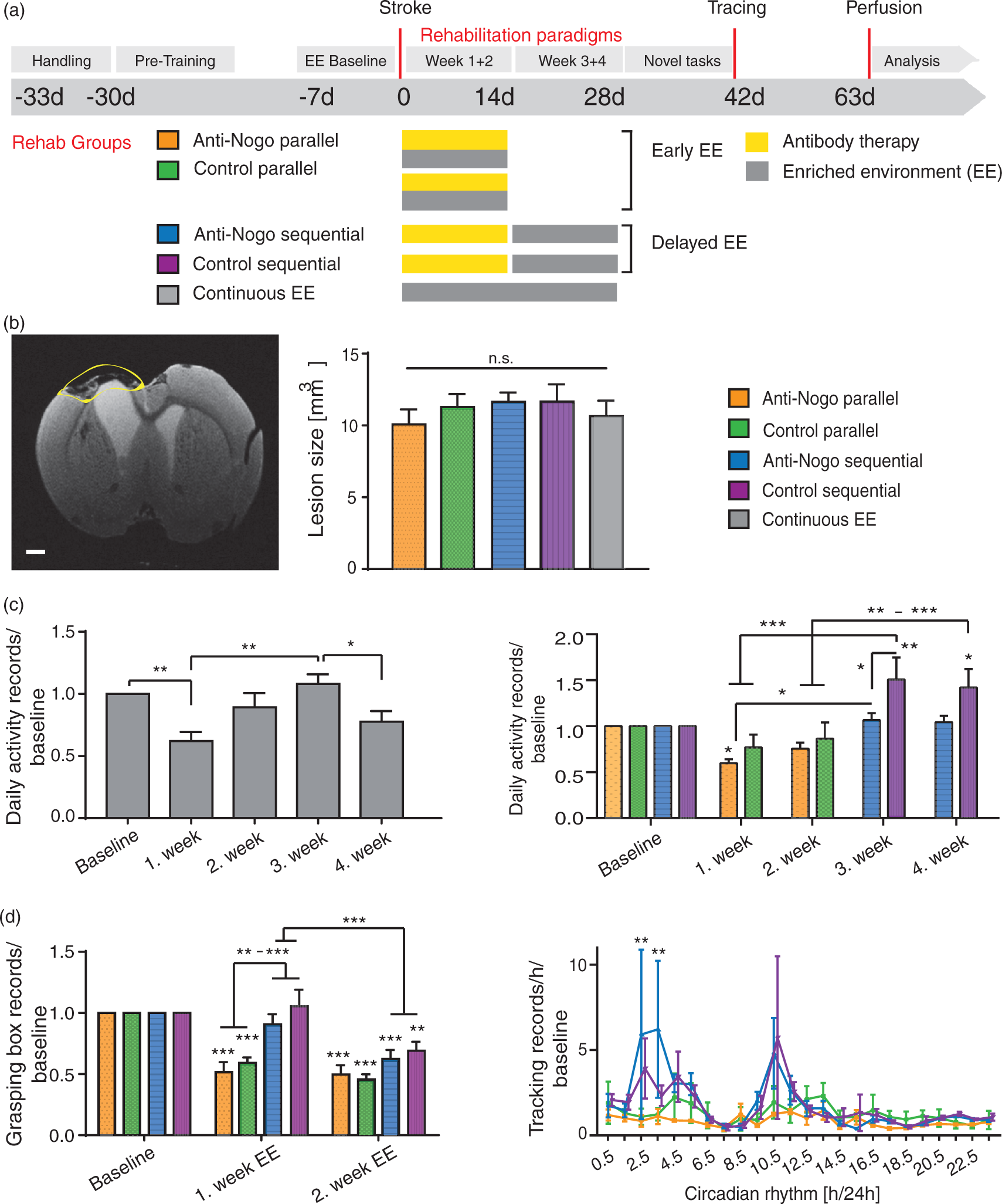
Baseline tracking values of animals with an extreme inactive behavior (Tracking value below 1000; Average baseline tracking value: 5102 ± 1521) during baseline recordings as well as the entire records of animals with an accidental too long or too short period in the NH cage were excluded from the analysis (2/55 animals). The determination of the circadian rhythm was based on tracking records per hour over the whole baseline and rehabilitation time in the NH cage. We excluded days of blood sampling and behavior assessment for single pellet grasping as well as the first and the last day of recordings, when animals were only part-time in the NH cage to avoid confounding effects of these additional stressors in the final analysis for circadian activity.
Besides the activity tracking with the RatTrack system, animals were filmed 24 h/day using infrared cameras (TVCCD 118 B/W camera, Monacor Switzerland AG, Risch, Switzerland) and infrared illumination (IR-12/65LED, Monacor Switzerland AG). Cameras were directed towards the grasping boxes, the grasping grid, the wheel, and the penthouse. Analysis of the videos was conducted with VLC media player (2.1.5 Rincewind, VideoLan organization).
Photothrombotic stroke and antibody treatment
All animals received a unilateral phototrombotic stroke to the sensorimotor cortex. The side of the stroke was chosen according to the handedness of the rat identified during the single pellet grasping training (for details, please see ‘Photothrombotic stroke’, Online-Only Data Supplement). Pumps for the constant administration of the function blocking IgG1 mouse monoclonal Anti-Nogo-A antibody 11C7 or the monoclonal control antibody anti-cyclosporine were implanted right after stroke surgery as described in the Online-Only Data Supplement, ‘Pump implantation for intrathecal delivery of antibodies’.
Training and testing sensorimotor function
As a primary readout, the single pellet grasping task was used. After the completion of the rehabilitative schedules (4 weeks after stroke) animals were exposed to novel tasks of forelimb sensorimotor function and overall locomotion such as the horizontal ladder test, the Montoya staircase, the narrow beam task and the plantar heat task in order to determine their non-task specific levels of recovery for lost or impaired motor functions (Online-Only Data Supplement).
Assessment of lesion by MR imaging
Lesion size and location were determined with a 7 T small animal MR system (Bruker BioSpin GmbH, Ettlingen, Germany; Online-Only Data Supplement).
A detailed description for anterograde tracing of the intact corticospinal tract, blood sampling, body weight measurement is provided in the Online-Only Data Supplement.
Kinematics
To analyze the grasping capabilities of animals at different states of recovery and under different rehabilitative protocols, we utilized computer vision to investigate the skilled motor function of the paw by evaluating the capabilities of the animal to target the sugar pellet in the task of single pellet grasping. We performed tracking of the paw by sequential re-identification, since direct tracking suffers from occlusion by the other paw, nose, the cage, etc. Thus, we have trained a classifier 11 to distinguish clutter from actual paws. We then searched in a local neighborhood around the detection of a previous frame after shifting this by the previous motion vector. To estimate the targeting capabilities of the animal, for each grasp we measured how close the paw got towards the pellet and recorded the two-dimensional location of this closest point in the side view. By averaging over all grasps of all animals in a cohort, we obtained the spatial distribution of the targeting (Figure 3(b, c)), indicating the targeting accuracy.
Statistical analysis
The whole statistical evaluation was performed with Graph Pad Prism (GraphPad Software Inc.; Version 6.05). All data are expressed as means ± s.e.m. Group-dependent data with several time points were analyzed using two-way repeated measures ANOVA and Bonferroni’s post hoc test, e.g. comparing different activity records in the NH cage for the five different rehabilitation groups at different time points (Figure 1) or blood markers for the different groups at different time points (Figure 2) or CST fiber sprouting at different laminae (Figure 4(b)) and different spinal cord levels for the rehabilitation groups (Figure 4(d)). We chose a Bonferroni post hoc test due to the slightly unbalanced data set (slightly different group size among the different rehabilitation groups). An one-way ANOVA repeated measures plus Bonferroni’s multiple comparison test was applied in the case of time-independent group values (lesion size analysis among groups in Figure 1, total CST fiber sprouting in the spinal cord Figure 4(c)) or comparing activity records at different time points within one group (Figure 1(c)). The sample size for the different rehabilitation groups was estimated by means and variance of measured data means in related work7,8,12 predicted to be sufficient to detect a statistically significant result in ANOVA with α = 0.05 and power > 0.8. Correlations were demonstrated carrying out the two-tailed Spearman analysis. The level of significance was set at *P < 0.05, **P < 0.01, ***P < 0.001.**
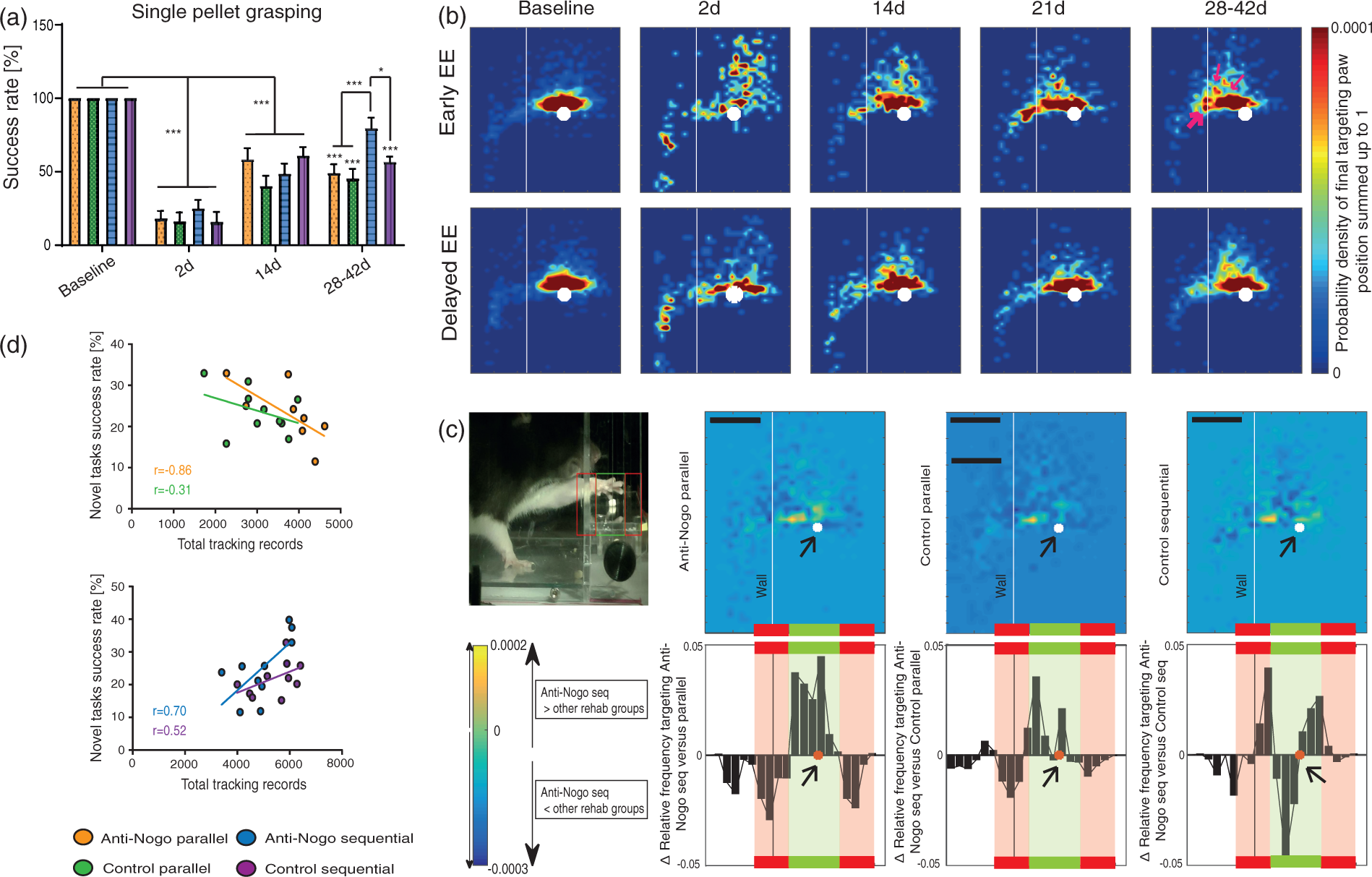
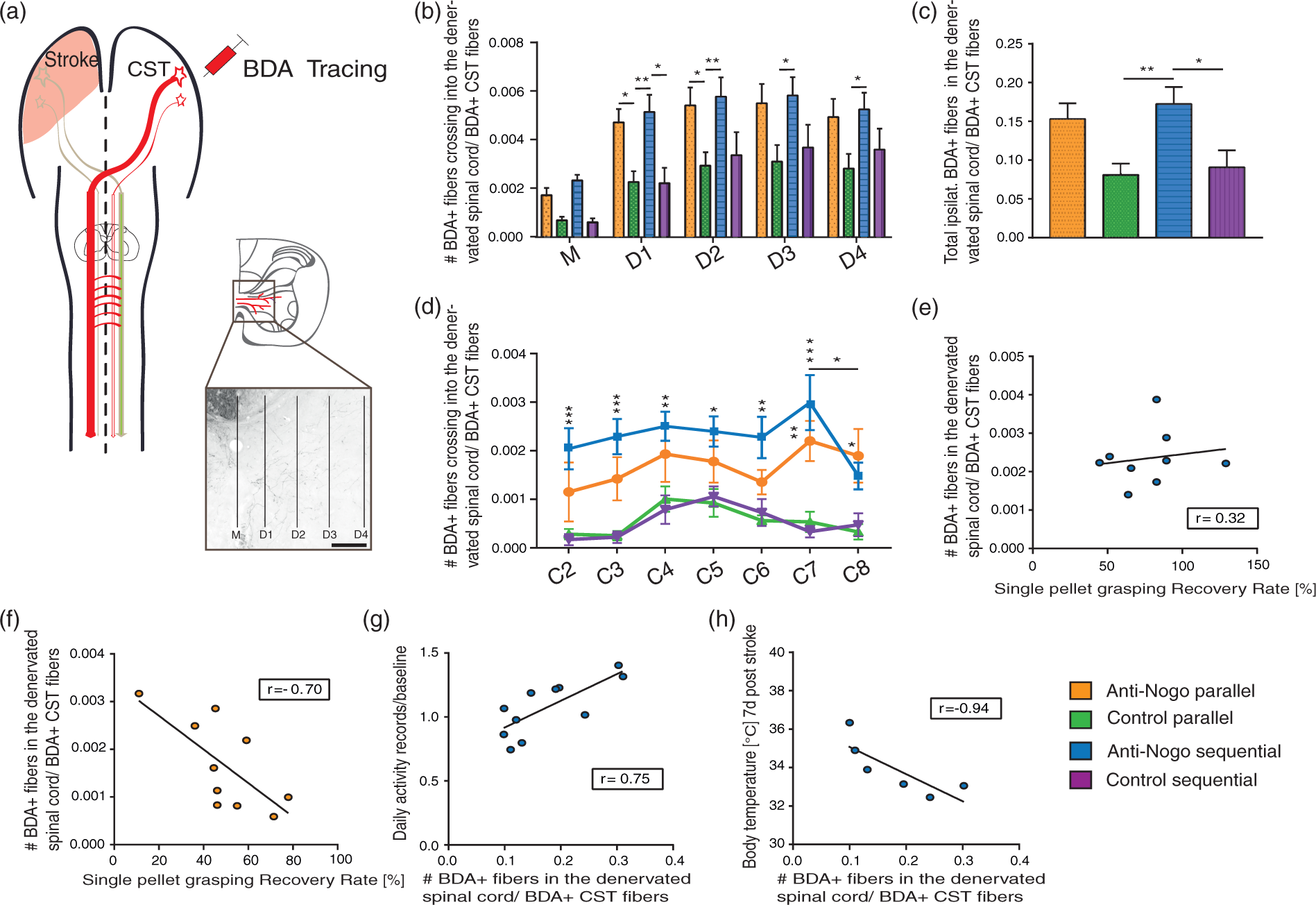
Inflammatory parameters are increased after stroke and potentiated by early intense training in the enriched environment. (a) Blood levels of the acute-phase protein alpha2-macroglobulin in animals with ‘No treatment’ or being housed in the NH cage immediately as ‘Early EE’ cohort, or delayed as ‘Delayed EE’. (b) Body temperature modifications after stroke in the three compared treatment groups. (c) A positive correlation between body temperature of the ‘Early EE’ group and individual total activity tracking records in the NH cage was found within the first week after stroke (r = 0.77, Spearman correlation). (d) Levels of leucocytes, their subtypes and thrombocytes in the blood of stroke rats of the three treatment groups before and after stroke. Data are presented as means ± s.e.m.; statistical evaluation was carried out with two-way ANOVA repeated measure followed by Bonferroni post hoc (for a, b, d–i), asterisks indicate significances: *P < 0.05, **P < 0.01, ***P < 0.001. High-intense self-motivated training starting two weeks after stroke results in significantly higher recovery levels than rehabilitation initiated immediately after stroke. (a) Success rates in the single-pellet grasping task (‘Anti-Nogo parallel’, n = 10; ‘Control parallel’, n = 14; ‘Anti-Nogo sequential’, n = 12; ‘Control sequential’, n = 11). (b) Analysis of grasping kinematics of the impaired forepaw at baseline (naïve) and during the rehabilitative course for the ‘Early EE’ and ‘Delayed EE’ cohort (for details refer to methods section, ‘Kinematic analysis’): Animals in the ‘Delayed EE’ group reveal full recovery of the targeting capability within 4–6 weeks after stroke while animals in the ‘Early EE’ group often gasp too short (thin white arrows) or hit the cage wall (thick white arrow). (c) Detailed analysis of targeting among single groups: The more yellow (in the heat maps) or more positive (in the diagram) the values, the higher the frequency distribution of final paw positions of ‘Anti-Nogo sequential’ animals to that specific location relative to the sugar pellet ( = white dot, scale bar = 1.25 cm). Precise targeting within the ‘green zone’ around the sugar pellet was in particular pronounced in ‘Anti-Nogo sequential’ animals compared to the ‘Anti-Nogo parallel’ and ‘Control parallel’ groups. (d) While there was a negative correlation between early high-intensity self-motivated training and outcome in the novel tasks for the early cohorts (r = −0.86 for ‘Anti-Nogo parallel, r = −0.31 for ‘Control parallel’, Spearman correlation, upper panel), delayed rehabilitative training 2 weeks after stroke in the EE (‘Delayed EE’ cohort) was positively correlated to final success rates in the novel tasks (r = 0.70 for ‘Anti-Nogo sequential, r = 0.52 for ‘Control sequential’, Spearman correlation, lower panel). For (a) data are presented as means ± s.e.m.; statistical evaluation was carried out with two-way ANOVA repeated measure followed by Bonferroni post hoc, asterisks indicate significances: *P < 0.05, ***P < 0.001. Corticospinal fiber sprouting into the denervated cervical hemi-cord. (a) BDA tracer injection into the contralesional pre- and primary motor cortex labels the corticospinal tract and its fibers sprouting across the spinal cord midline (M) into the stroke denervated cervical hemi spinal cord over various distances (D1–D4) nine weeks after the stroke (scale bar = 200 µm). (b) Midline-crossing fiber values (average over segments C2–C8, related to the total number of BDA-positive fibers in the intact CST) were significantly higher in the ‘Anti-Nogo sequential’ group for all distances, and in the ‘anti-Nogo parallel’ group for the inner half of the grey matter compared to both control antibody groups. (c) Averaged relative number of total CST fibers (C2–C8) in the denervated grey matter was significantly higher for the ‘Anti-Nogo sequential’ animals compared to the control antibody groups. (d) Segment specific analysis of midline-crossing CST fiber numbers. While fiber growth was significantly higher for late trained ‘Anti-Nogo sequential’ animals throughout the cervical spinal cord, increased fiber sprouting in the early trained ‘Anti-Nogo parallel’ rats was only significant in the distal cervical segments. (e) While fiber growth is positively correlated to the recovery of impaired grasping function in animals with Anti-Nogo immunotherapy followed by rehabilitative training two weeks after stroke (r = 0.32, Spearman correlation), the reciprocal effect was found in animals with immediate rehabilitative training (f, r = −0.70, Spearman correlation). (g) For the ‘Anti-Nogo sequential’ group, the rehabilitation group with excellent outcome, CST fiber growth was positively correlated with training in the enriched environment starting two weeks after stroke (r = 0.75, Spearman correlation), while increased body temperature within the first week after stroke was a negative predictor for the number of sprouting CST fibers (h, r = −0.94, Spearman correlation). For (b–d) data are presented as means ± s.e.m.; statistical evaluation was carried out with two-way ANOVA repeated measure followed by Bonferroni post hoc, asterisks indicate significances: *P < 0.05, **P < 0.01, ***P < 0.001.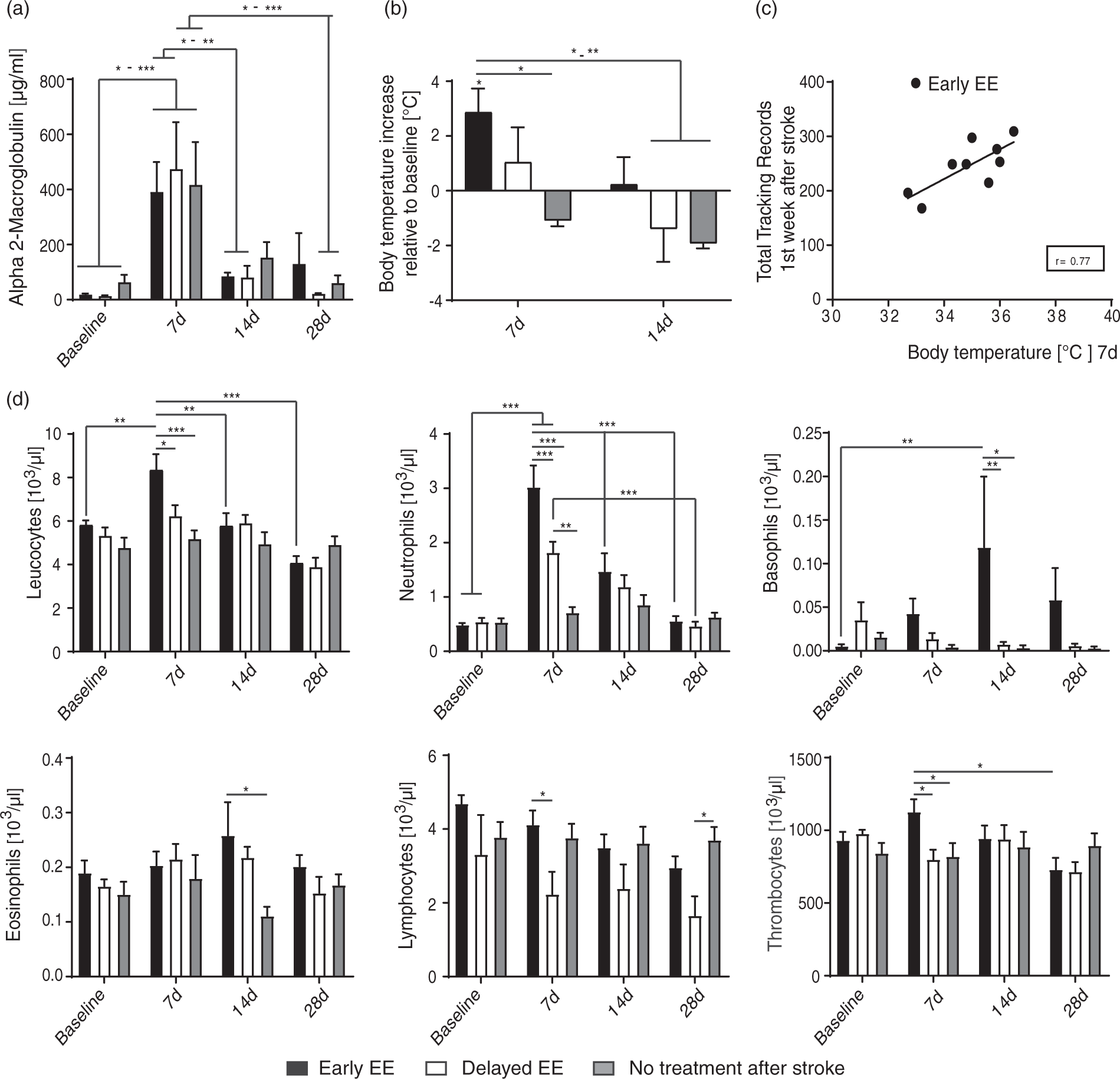
Results
Stroke induces a fatigue syndrome
Rats housed in EEs or NH cages are much more active and spontaneously train a variety of tasks. 10 We used an NH cage consisting of three floors providing training devices for fore- and hind-limb functions for up to 20 animals (Supplementary Figure 1A). Subcutaneous transponders in each animal allowed individual tracking over time providing 24 h/7 days information about training devices used, training device preferences, training intensity, and overall activity of each animal. 10 Additional infrared cameras recorded self-initiated training in the EE throughout a 24-h cycle including the active phase of the animals (6:30 PM to 6:30 AM).
Anti-Nogo antibodies have been shown in various animal models to stimulate neuronal outgrowth after stroke and spinal cord injury.6,8,13,14 However, a recent study 7 demonstrated the importance of the timing when Anti-Nogo treatment was combined with rehabilitative training. Too early forced training concurrently to the Anti-Nogo immunotherapy had resulted in a detrimental outcome. Here, we thus aimed at examining the effect of Anti-Nogo antibodies if combined with self-initiated training in the EE where the intensity of training could be chosen by the animals themselves.
We compared five different schedules applied after a large photothrombotic stroke including the sensorimotor cortex (Figure 1(a)). Only animals with the same lesion size and location as determined by ex vivo MRI at the end of the experiment were included in the final analysis (Figure 1(b), Supplementary Figure 2).
Recording the daily activity in the EE of animals without antibody treatment during four weeks after stroke (‘Continuous EE’ cohort) revealed a significant 38% ± 7.3% reduction of the activity level in the first week after the insult (repeated measures one-way ANOVA with post hoc Bonferroni, P < 0.001, F-value 12.1, DF = 3, Figure 1(c) left graph), which recovered to 108.0% ± 7.8 in the third week in the EE (first versus third week, P = 0.0005). We then examined activity levels post-stroke relative to pre-stroke activity in the NH cage in animals which were exposed to the EE immediately after insult and received either control IgG (‘Control parallel’) or Anti-Nogo immunotherapy (‘Anti-Nogo parallel’) concurrently. A period of post-stroke reduction in overall activity was also found for these ‘Early EE’ groups (‘Anti-Nogo parallel’ 59.7% ± 4.0 of baseline activity first week post stroke, ‘Control parallel’ 76.8% ± 14.0 of baseline activity, Figure 1(c) right graph), however being only significant for the Nogo-antibody treated group (‘Anti-Nogo parallel’, Figure 1(c) right graph). This was in sharp contrast to the behavior of animals for which the self-initiated training in the NH cage started in the third week post insult (‘Delayed EE’ cohort consisting of ‘Anti-Nogo sequential’ and ‘Control sequential’ animals, Figure 1(c) right graph): These animals showed either baseline (‘Anti-Nogo sequential’ group) or increased activity levels (‘Control sequential’ group, 150.7% ± 24.1 and 142.2% ± 19.8 of baseline activity in the third and fourth week after stroke). Remarkably, in the group comparison of the Delayed EE cohort, rats treated with Anti-Nogo-A antibodies showed lower activity levels than their counterparts treated with control antibodies. In addition, animals, which started their self-motivated training two weeks after stroke (‘Delayed EE’ groups) used significantly more the grasping boxes in the NH cage to train fine motor forelimb tasks (Figure 1(d) left graph, P > 0.01, F-value = 6.643, DF = 3, two-way repeated measures ANOVA with post hoc Bonferroni) while animals with early exposure to the EE (‘Anti-Nogo parallel’ and ‘Control parallel’ cohorts) were less willing to train specific fine motor tasks compared to the ‘Delayed EE’ groups (Supplementary Figure 1B).
We also monitored individual circadian activity profiles during the course of rehabilitation in the NH cage, measured as tracking records/h for 24 h. We found that the circadian rhythm characterized by its typical activity peaks at night (in our experiment 6:30 PM to 6:30 AM) and low activity levels during the day (6:30 AM to 6:30 PM) was disturbed in particular within the first two weeks after stroke, showing a marked depression of the activity peaks for the ‘Early EE’ cohort (Supplementary Figure 3). In contrast, the Delayed EE cohort showed enhanced peak activities in the NH cage compared to their pre-stroke baseline. ‘Anti-Nogo sequential’ and ‘Control sequential’ animals had a more than five times higher activity level at certain time points in their active phase compared to the two groups housed immediately after stroke in the NH cage (Figure 1(d), right graph, P < 0.01 for time point ‘2.5’ and ‘3.0’ AM, two-way repeated measures ANOVA with post hoc Bonferroni, F-value = 3.82, DF = 3).
Inflammatory parameters are increased after stroke and potentiated by early intense training in the EE
As fever and elevated white blood counts are often reported in stroke patients,15,16 we hypothesized that the reduced behavioral activity found in the ‘Early EE’ cohort was an inflammation-related effect. When we monitored body temperature, the acute-phase protein alpha-2 macroglobulin, 17 a marker for systemic inflammation, and white blood counts before and at early (7, 14 d) and late (28 d) time points after the stroke, no differences between Anti-Nogo and control antibody treated animals were found at any time point (‘Anti-Nogo parallel’ versus ‘Control parallel’ group and ‘Anti-Nogo sequential’ versus ‘Control sequential’ group; data not shown). We then compared the ‘Early EE’ and ‘Delayed EE’ cohorts to stroke rats without any treatment. For animals of all three cohorts a significant increase of alpha-macroglobulin was measured seven days after stroke (Figure 2(a), P < 0.05 7 d versus baseline for all cohorts, F-value = 14.8, DF = 3, two-way repeated measures ANOVA with post hoc Bonferroni). Animals exposed to the EE early (‘Early EE’ consisting of the ‘Anti-Nogo parallel’ and ‘Control parallel’ group) showed a pronounced, transitory increase in body temperature at 7 d. (Figure 2(b), P < 0.05, F-value = 2.1, DF = 2, two-way repeated measures ANOVA with post hoc Bonferroni). Moreover, there was a positive correlation of activity levels in the rats with increased body temperature for the ‘Early EE’ cohort (Figure 2(c), r = 0.77, Spearman correlation). In the ‘Early EE’ cohort leucocytes, neutrophils and basophils were significantly elevated 7–14 d post insult compared to the healthy baseline condition (Figure 2(d), two-way repeated measures ANOVA with post hoc Bonferroni). White blood counts (neutrophils, basophiles, lymphocytes) and thrombocytes were also in particularly increased in the ‘Early EE’ cohort compared to animals where self-motivated training in the NH cage was started delayed (‘Delayed EE’, Figure 2(d)). Blood cell counts for animals without antibody treatment did not differ from baseline. Cell counts of antibody-treated animals with delayed training did only differ for neutrophils and lymphocytes (Figure 2(d)) pointing to a slight inflammatory reaction to the combination of stroke, catheter and pump implantation and perhaps antibody application also in the ‘Delayed EE’ cohort.
A time window for high-intensity training to improve sensorimotor recovery
Retrieval of small sugar pellets through a window in the Plexiglas testing box requires precise targeting of the forelimb. 18 Success rate of pellet retrieval dropped to 19.2% ± 2.8 of baseline levels in all rehabilitation groups two days after stroke (Figure 3(a)). All groups recovered to a success rate of 40–60% at 14 days, irrespectively of the treatment (Figure 3(a)). While rats in both ‘Early EE’ groups (‘Anti-Nogo parallel’ and ‘Control parallel’) remained on the same poor outcome level at 28–42 d post insult (Figure 3(a)), starting self-motivated motor training two weeks after stroke (‘Anti-Nogo sequential’ and ‘Control sequential’ groups) resulted in a significant further increase in pellet retrieval in the Anti-Nogo-A rats, but not in the control antibody group (‘Anti-Nogo sequential’ 79.7% ± 7.0 of baseline levels, ‘Control sequential’ 56.8% ± 3.4, P < 0.001, F-value = 3.96, DF = 3, two-way ANOVA with post hoc Bonferroni, Figure 3(a)). We then specifically assessed the targeting precision of the reaching movements of the rats (Figure 3(b)): ‘Early EE’ rats often hit the box wall, missing the window. Their grasps through the window were also often too short (Figure 3(b)). In contrast, ‘Anti-Nogo sequential’ rats trained in the NH cage from 14 to 28 d post-stroke, performed with highly precise, targeted arm and paw movements: Figure 3(c) shows the difference of frequency distribution of final paw positions between the ‘Anti-Nogo sequential’ and the other rehabilitation groups: ‘Anti-Nogo sequential’ animals target the sugar pellet more precisely (‘green zone’, Figure 3(c)) than animals of the ‘Anti-Nogo parallel’ and ‘Control parallel’ group which grasp either too short or too long (‘red zone’, Figure 3(c)). Differences in targeting for the ‘Control sequential’ and the ‘Anti-Nogo sequential’ group were more subtle.
Furthermore, the more animals trained early in the NH cage (‘Anti-Nogo parallel’ and ‘Control parallel’ groups), the worse was their final performance in the novel tasks (Figure 3(d), upper panel). The reverse was true for the late training groups: the more ‘Anti-Nogo sequential’ and ‘Control sequential’ animals trained starting 2 weeks after stroke, the more they succeeded in the novel tasks (Figure 3(d), lower panel). For both Anti-Nogo treated groups the correlation between outcome in the novel tasks and overall activity in the NH cage was significantly more pronounced than in the corresponding control antibody groups (for ‘Anti-Nogo parallel’ animals r = −0.86, for ‘Anti-Nogo sequential’ animals r = 0.70, P < 0.05, Spearman correlation). Rats with early exposure to the NH cage (‘Early EE’) were also slightly less sensitive to noxious heat stimuli over time and less precise in the Montoya staircase (Supplementary Figure 4A, B). There was no difference for the narrow beam and horizontal ladder task among cohorts (Supplementary Figure 4C, D). A stable body weight two days after insult was positively correlated to the recovery of grasping function (Supplementary Figure 5, r = 0.33, Spearman correlation).
Corticospinal fibers reinnervating the denervated hemicord are influenced by Anti-Nogo and self-initiated high-intensity training
Previously, we have shown that highly repetitive, forced training of the stroke-impaired forelimb immediately after stroke in combination with Anti-Nogo immunotherapy lead to exuberant and chaotic corticospinal fiber sprouting from the intact-side CST across the midline of the spinal cord, innervating the stroke denervated cervical hemicord. 7 In the present study, animals could themselves initiate and allot the intensity of their rehabilitative training on different tasks in the NH cage. To search for neuroanatomical correlates of motor recovery, we labeled the intact, contralesional CST with the anterograde tracer BDA nine weeks after stroke (Figure 4(a)). Anti-Nogo-A treated rats had higher numbers of midline crossing CST fibers than control antibody rats (Figure 4(b)), but in strong contrast to the previous study 7 animals with concurrent Anti-Nogo immunotherapy and exposure to the EE revealed only enhanced innervation of the inner parts of the ventral horn (CST fiber sprouting at D1 and D2 compared to control groups, P < 0.05, F-value = 4.80, DF = 3, two-way repeated measures ANOVA with post hoc Bonferroni, Figure 4(b)) and the lower cervical spinal cord levels (C7/C8, Figure 4(d)). Sequential Anti-Nogo application followed by exposure to the NH cage resulted in a high proportion of corticospinal fiber sprouting innervating all cervical spinal cord segments (Figure 4(c), right panel, P < 0.01, F-value = 5.59, DF = 3, one-way ANOVA with post hoc Bonferroni). CST fiber sprouting in these ‘Anti-Nogo sequential’ animals was positively correlated to the recovery of skilled forelimb function (Figure 4(e), r = 0.32, Spearman correlation) and activity in the EE (Figure 4(g), r = 0.75, Spearman correlation). In contrast, fiber growth and final outcome was negatively correlated for the early trained ‘Anti-Nogo parallel’ animals (Figure 4(f), r = −0.7, Spearman correlation), suggesting a negative effect even of the reduced early training on the growth of CST sprouts in the denervated spinal cord. A strong negative relationship between increased body temperature within the first week after stroke and the amount of CST fibers in the denervated cervical spinal cord was detected for the ‘Anti-Nogo sequential’ animals (Figure 4(h), r = −0.94, Spearman correlation).
Discussion
We found that large cortical strokes induce an early fatigue syndrome with reduced activity levels during the first two weeks after insult in our rat stroke model. Exposing animals within this early phase to an EE resulted in an increased immune response with elevated body temperature and poorer outcome. Suppression of the neurite growth inhibitory protein Nogo-A during the first two weeks after stroke enhanced sprouting of intact-side CST fibers across the spinal cord midline and re-innervation of the CST-denervated grey matter. This fiber density was positively correlated to the recovery of precision forelimb movements in the Anti-Nogo-A rats exposed to the NH during week 3 and 4 after the stroke. These animals showed high daily activity and grasp training scores and reached an eminent recovery level of sensorimotor function. In contrast, in the ‘Early EE’ Anti-Nogo-A rats which poorly recovered, the amount of CST fiber density was negatively correlated to the outcome, suggesting an absence of refinement for the connections of the newly grown fibers.
Post-stroke objective or subjective fatigue is reported for around 50% of patients; it often appears early after stroke, can last for more than a year after the event and hampers rehabilitation and training. It is usually characterized by a different quality from usual fatigue and reduced response to rest. 19 Affliction of deep brain structures has been associated with severe post-stroke fatigue. It is noteworthy, therefore, that we see this type of early post-stroke fatigue in our rats which have pure cortical strokes.
Notably, there is a coincidence for the time window of early post-stroke fatigue and the critical period of heightened neuroplasticity2,20 inducing fiber sprouting and synaptic remodeling. A potential link between the clinical symptom of fatigue and the reorganizational processes taking place in the CNS after stroke might be the inflammatory/immune response, which induces fatigue through cytokine effects in brain areas involved in interoception and homeostasis. Furthermore, a systemic immune response could support different repair processes: In animal models and human stroke patients, inflammatory signaling has been described as being instrumental at all stages of the ischemic cascade. 21 In animals with early exposure to the EE, the physiological immune response was exaggerated in the first two weeks after stroke when compared to animals with standard housing. That early high-intensity training can activate inflammatory signaling which is then associated with impaired cognition and hippocampal synaptic plasticity has been demonstrated recently. 22
Our study supports also recent clinical findings where early mobilization of stroke patients within 24 h after stroke resulted in a less favorable outcome than usual care. 5 Our data suggest a physiological, homeostatic, protective role of early post-stroke fatigue with the goal to reduce activity levels of the CNS during the post-injury vulnerable time span where the brain and whole organism concentrate on repair mechanisms.
Important structural components of the recovered forelimb movement ability are the sprouts of the CST that grow from the intact side of the spinal cord over the midline and innervate the denervated grey matter. Silencing these fibers by chemogenetics leads to an immediate, reversible collapse of the skilled forelimb reaching.7,23 Single animal analysis revealed a positive correlation for the intensity of the training, the final functional recovery level and the amount of CST fiber growth in the ‘Anti-Nogo sequential’ group while in the ‘Early EE’ ‘Anti-Nogo parallel’ group CST fiber density was negatively correlated to the final outcome, suggesting an insufficient or dysfunctional role of these newly formed corticospinal connections. Both these results resemble those recently obtained. 7 The present results based on self-motivated activity in the NH underline the concept that CNS injuries induce a plasticity and repair response during the initial (probably growth-) phase in which neuronal network activity should be not further exaggerated by excessive unspecific external stimulation.7,24,25 Fatigue may be one way by which the organism ensures this reduced level of activity, thereby protecting the reorganizing CNS. The plastic phase is then followed by a phase of activity driven selection, stabilization or pruning of the new connections leading to functional recovery.
The EE provided by our NH cage enabled spontaneous task-specific training of fore- and hind-limb tasks and also general locomotion. We find a significantly better outcome for animals which were delayed exposed to the NH cage in the single pellet grasping task. This ‘Delayed EE’ cohort also showed a tendency to perform better in the novel tasks – staircase and plantar heat test (although this was not statistically significant)–, while there was no difference in success rates for the more general locomotion tasks (horizontal ladder and beam) between ‘Delayed’ and ‘Early EE’ cohorts. A reason for these observations may be that only a limited number of newly formed or rewired connections exist which become part of a neuronal circuit specialized for a certain motor behavior. Another possibility would be that non-task specific training may induce non-directed fibre sprouting and fails to establish stabilized connections for distinct fine motor skills.7,25
The results of the present study have direct clinical implications: Very early intense rehabilitative training was found to be contraindicated in a recent multicentric stroke trial. 5 Antibodies against Nogo-A or Nogo receptor components are currently in clinical trials for spinal cord injury, multiple sclerosis and amyotrophic lateral sclerosis (www.clinicaltrials.gov). The concept of a physiological, protective role of the early post-stroke fatigue provides a basis to optimize current and future rehabilitative schedules and interventions for stroke patients.
Supplemental Material
Supplemental material for Early reduced behavioral activity induced by large strokes affects the efficiency of enriched environment in rats
Supplemental material for Early reduced behavioral activity induced by large strokes affects the efficiency of enriched environment in rats by Anna-Sophia Wahl, Eva Erlebach, Biagio Brattoli, Uta Büchler, Julia Kaiser, Benjamin V Ineichen, Alice C Mosberger, Shirin Schneeberger, Stefan Imobersteg, Martin Wieckhorst, Martina Stirn, Aileen Schroeter, Bjoern Ommer and Martin E Schwab in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the ERC advanced Grant NOGORISE 294115 (to MES), Swiss National Science Foundation Grant #31003A-149315-1, ETH Zurich Research Grant, and the Spinal Cord Consortium of the Christopher and Dana Reeve Foundation.
Acknowledgments
The authors thank Anna Jeske, Hansjörg Kasper, Stefan Giger, and Marco Tedaldi for technical support.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The University of Zurich holds joint patents with Novartis Pharma for antibodies against Nogo-A and their use in neurological diseases. MES is a board member of the spin-off company NovaGo Therapeutics Inc. Otherwise the authors have no patents pending or financial conflicts to disclose.
Authors’ contributions
ASW and MES designed the study. ASW, EE, SS, and SI carried out experiments. ASW, JK, EE, and ACM performed data analysis. BO, UB, and BB developed Computer and Machine Learning algorithm tools for behavior analysis. MW developed and provided technical tools. BVI performed pump implantations. AS performed MR imaging. MS examined and analyzed blood samplings. ASW and MES prepared figures and wrote the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
