Abstract
The effect of blood pressure (BP) reduction on cerebral blood flow (CBF) in acute ischemic stroke is unknown. We measured regional CBF with perfusion-weighted MRI before and after BP treatment in a three-armed non-randomized prospective controlled trial. Treatment arm assignment was based on acute mean arterial pressure (MAP). Patients with (MAP) >120 mmHg (n = 14) were treated with intravenous labetalol and sublingual (SL) nitroglycerin (labetalol group). Those with MAP 100–120 mmHg (n = 17) were treated with SL nitroglycerin (0.3 mg) (‘NTG Group’) and those with baseline MAP<100 mmHg (n = 18) were not treated with antihypertensive drugs (untreated group). Forty-nine patients (18 female, mean age 65.3 ± 12.9 years) were serially imaged. Labetalol reduced MAP by 12.5 (5.7–17.7) mmHg, p = 0.0002. MAP remained stable in the NTG (6.0 (0.4–16, p = 0.3) mmHg and untreated groups (−0.3 (−2.3–7.0, p = 0.2) mmHg. The volume of total hypoperfused tissue (CBF<18 ml/100 g/min) did not increase after labetalol (−1.1 ((−6.5)–(−0.2)) ml, p = 0.1), NTG (0 ((−1.5)–4.5) ml, p = 0.72), or no treatment 0.25 ((−10.1)–4.5) ml, p = 0.87). Antihypertensive therapy, based on presenting BP, in acute stroke patients was not associated with an increased volume of total hypoperfused tissue.
Introduction
Elevated systolic blood pressure (BP) is present in 70–80% of patients with acute ischemic stroke.1,2 Acute hypertension is associated with an increased rate of hemorrhagic transformation, poor functional outcome, and early mortality.3–6 Indeed, symptomatic intracranial haemorrhage (SICH) rates are linearly correlated with BP. 7 Furthermore, observational data from thrombolysis trials suggest pharmacological lowering of BP reduces hemorrhagic transformation. 8
The relationship between acute BP and outcome is complex, however, as lower pressures are also associated with elevated mortality and stroke morbidity. 9 Decreases in mean arterial pressure (MAP) (≥10%) following conscious sedation in patients with large vessel occlusion undergoing endovascular therapy have been associated with poor functional outcomes. 10
There is evidence that cerebral autoregulation is impaired in experimental ischemic stroke.11,12 Under ischemic conditions, regional cerebral blood flow (CBF) may be directly proportional to MAP. 13 Thus, ischemic tissue may be vulnerable to acute BP reductions, potentially leading to infarct growth and poor functional outcome. The precise relationship between BP treatment and CBF in acute ischemic stroke patients has not been studied systemically. Given observational data showing a U-shaped relationship between baseline MAP and outcome measures, a single target pressure does not appear justified. It is possible that treatment with antihypertensive agents has differential risks and benefits, depending on the baseline MAP.
The aim of this study was to elucidate the relationship between BP reduction in acute ischemic stroke and CBF. We tested the hypothesis that the volume of hypoperfused tissue will not increase significantly with BP reduction in acute ischemic stroke patients. We tested this hypothesis with a three-group non-randomized prospective controlled study of CBF measurements before and after BP reduction.
Materials and methods
Patients
The perfusion and antihypertensive therapy in acute ischemic stroke study
The perfusion and antihypertensive therapy in acute ischemic stroke (PATIS) study was a prospective three-group non-randomized serial controlled study of CBF and BP treatment in acute ischemic stroke patients (ClinicalTrials.gov Identifier: NCT02327793). The study was approved by Human Research Ethics Office, University of Alberta, Canada (Pro00001847). The ethical guideline governing the conduct of study was the Tri-Council Policy Statement: Ethical Conduct for Research Involving Human, Canada. Written Informed consent was obtained from all patients or legally authorized representatives (surrogate consent).
Inclusion criteria
Eligible patients were ≥18 years of age, with acute ischemic stroke presenting within 72 h of symptom onset. All patients had evidence of cerebral infarction on diffusion-weighted MRI (DWI) and had a measurable perfusion deficit. Partial and total anterior circulation infarcts (PACI and TACI; Oxfordshire Stroke Classification Project; OSCP), as well as posterior circulation infarcts (POCIs) with evidence of cortical infarction in the territory of the posterior cerebral artery or its branches, including the occipital and mesial temporal lobes were all eligible. 14 Patients treated with tissue plasminogen activator (tPA) were eligible.
Exclusion criteria
Patients were excluded if they had a contraindication (i.e. known extracranial/intracranial arterial stenosis, high-grade stenotic valvular heart disease, or severe renal failure) or definite indication (i.e. hypertensive encephalopathy, or aortic dissection) for BP reduction. Other exclusion criteria were contraindications to magnetic resonance imaging (MRI), allergy to gadolinium contrast, requirement for oxygen therapy (>4 l/min to keep Sp02 ≥ 92% by nasal cannulae), suspected hemodynamic stroke mechanism, evidence of significant mass effect secondary to acute infarction, known allergic reaction to antihypertensive agents or use of phosphodiesterase inhibitors such as sildenafil or tadalafil within 12 h of the baseline MRI.
Procedures
BP management protocols
Patients were stratified into three groups based on the baseline MAP (≥2 readings ≥ 5 min apart). MAP was calculated as = [(2 × diastolic)+systolic]/3. Patients with MAP > 120 mmHg (‘labetalol group’) were treated with intravenous labetalol (5–20 mg boluses) and sublingual (SL) nitroglycerin (NTG) (0.3 mg) immediately after the first MRI, aiming for a target MAP of 110–120 mmHg. Patients with baseline MAP between 100 and 120 mmHg were treated with SL NTG (0.3 mg) alone immediately after the first MRI (‘NTG Group’). Patients with MAP < 100 mmHg were not treated with any antihypertensive therapy (‘Untreated Group’). MRI was repeated 30 min after baseline MRI or once target BP was achieved (labetalol group). BP was measured every 5 min while in the MRI scanner.
Clinical assessments
Demographic profile, medical history including antihypertensive medications and Glasgow Coma Scale were recorded in a case record form. National Institute of Health Stroke Scale (NIHSS) scores were assessed at baseline, immediately after the second MRI, at 24 h, 30 days and 90 days. Functional assessment (modified Rankin scale) was performed at 24 h 30 and 90 days.
Imaging procedures
MRI data acquisition occurred in a single session, with repeat sequences before and after BP treatment. Patients did not leave the MRI table during treatment. Baseline images were acquired within 72 h of stroke onset. Based on MAP at the time of the first MRI, patients were treated with BP management protocols described above while in the scanner. Thirty minutes later, the second MRI was performed.
Patients were imaged using an 8-channel phased array radiofrequency head coil (MRI Devices, Waukesha, WI) on a 1.5-T whole-body Siemens Sonata MRI scanner (Siemens Medical Systems, Erlangen, Germany). The imaging protocol consisted of a T1-weighted sagittal localizer, time-of-flight magnetic resonance angiogram (MRA), gradient recalled echo (GRE), diffusion-weighted (DWI), fluid-attenuated inversion recovery (FLAIR), and perfusion-weighted images (PWIs). DWI acquisition parameters were: repetition time (TR) of 3 s, spin echo time (TE) of 86 ms, 8 averages, 128 × 128 matrix base resolution zero filled to 256 × 256, 22 cm FOV and slice thickness of 5 mm with a 1.5 mm inter-slice gap. Isotropic DWI images were obtained by averaging the b = 1000 signal from all orthogonal directions. Raw perfusion images were acquired with the administration of intravenous gadolinium (0.1 mmol/kg; 5 ml/s power injection, followed by 20 ml saline flush 5 ml/s, via an 18 g angiocatheter in an antecubital vein) with EPI gradient-echo (T2*) images acquired every 1.4 s for 84 s (16 axial slices, 60 time points). The second MRI acquisition consisted of PWI as well as a post-contrast FLAIR, sequence. DWI and PWI images were obtained at the same time, with identical slice acquisition parameters minimizing the need for additional post-processing co-registration.
Image analysis
Measurement of infarct core, hypoperfused tissue and hemispheric CBF in a patient with an acute right middle cerebral artery territory infarct. The infarct core region (red) of interest (ROI) was drawn on baseline diffusion-weighted images (DWI) and co-registered with cerebral blood flow (CBF) maps at baseline and 30 min later to assess mean absolute CBF in infarct core. Relative perfusion measures were calculated using homologous contralateral ROIs (green). The hypoperfused region ROI included voxels with delayed contrast arrival (delay time (DT)+2 s) and CBF < 19 mL/100 g/min. The perfusion-weighted imaging (PWI) base image was used to draw hemispheric ROIs.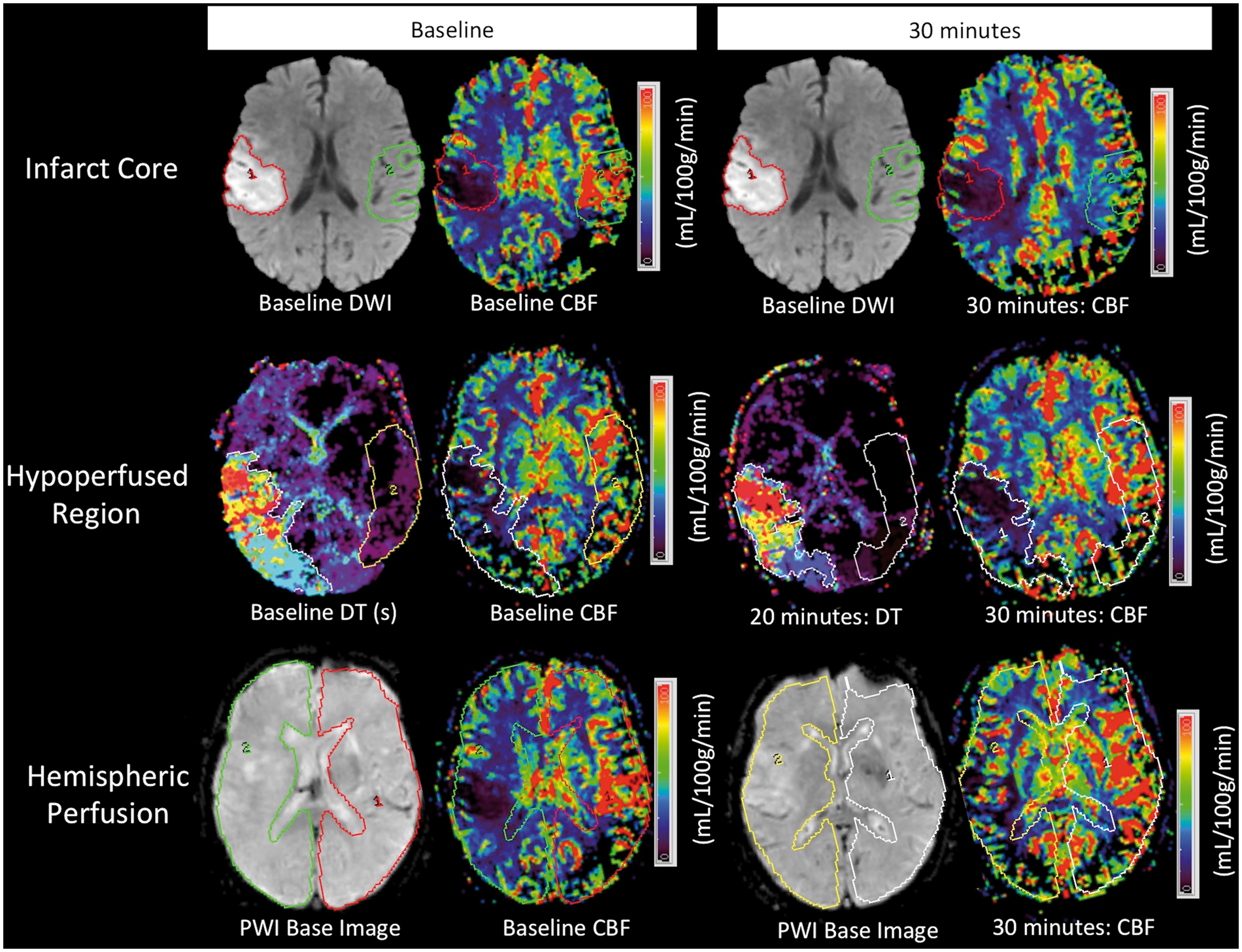
Study outcomes
The primary outcome was change in volume of the total hypoperfused tissue between baseline and repeat PWI scans 30 min later (Post-treatment hypoperfused tissue volume – Pre-treatment volume = Change in hypoperfused tissue volume). Secondary imaging outcome measures included relative mean total hypoperfused tissue, penumbral tissue, infarct core and hemispheric perfusion indices before and after BP reduction. Clinical outcome measures included MAP, NIHSS score change post-BP reduction and modified Rankin scale at 90 days.
Statistical analysis
Statistical analyses were completed with SPSS Statistics version 24. Total hypoperfused tissue volumes and penumbral tissue volumes in the three treatment groups were non-normally distributed (Shapiro–Wilk test, p < 0.0001). A Wilcoxon signed-rank test was used to assess differences in median hypoperfused tissue volumes baseline and 30 min after BP treatment. One-way ANOVAs were used to assess differences between groups in mean absolute CBF, rCBF, rCBV, rMTT, rDT in the infarct core, total hypoperfused tissue, penumbral tissue and ipsilateral/contralateral hemisphere. Paired t-tests were used to assess differences pre and post BP treatment in mean absolute CBF, rCBF, rCBV, rMTT, rDT in the infarct core, total hypoperfused tissue, penumbral tissue and ipsilateral/contralateral hemisphere. An ANOVA on ranks (Kruskal–Wallis) test was used to assess between group differences in the total hypoperfused tissue volume, penumbral tissue volume and infarct core, followed by pairwise comparisons. A p value of <0.05 was considered significant.
Results
Baseline characteristics
Baseline and post-treatment characteristics of patients.
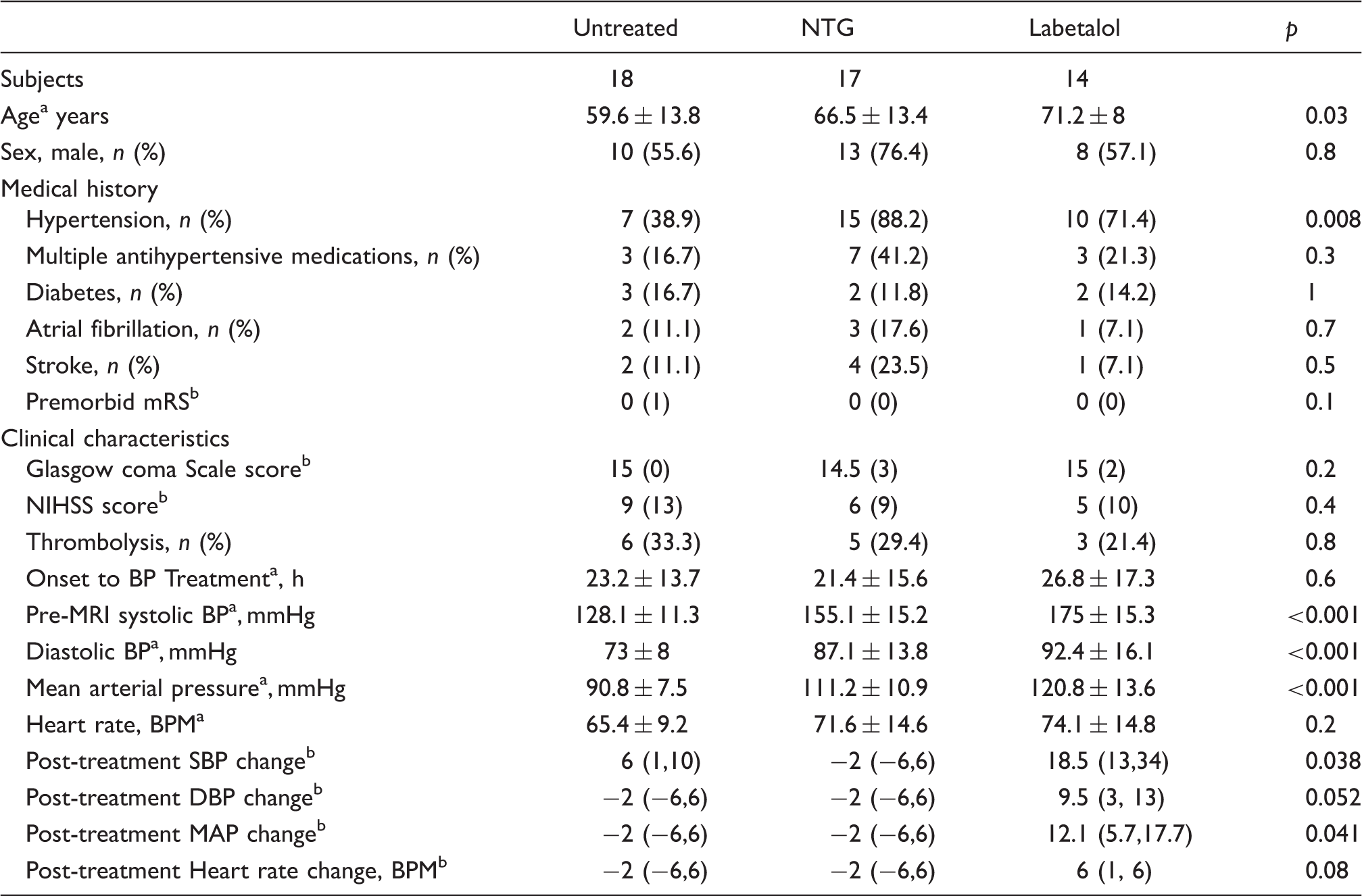
Mean±SD.
Median (interquartile range: 25th, 75th percentile).
NTG: nitroglycerin; NIHSS: National Institute of Health Stroke Scale; BP: blood pressure.
Baseline cerebral perfusion
Total hypoperfused tissue and penumbral tissue volumes.
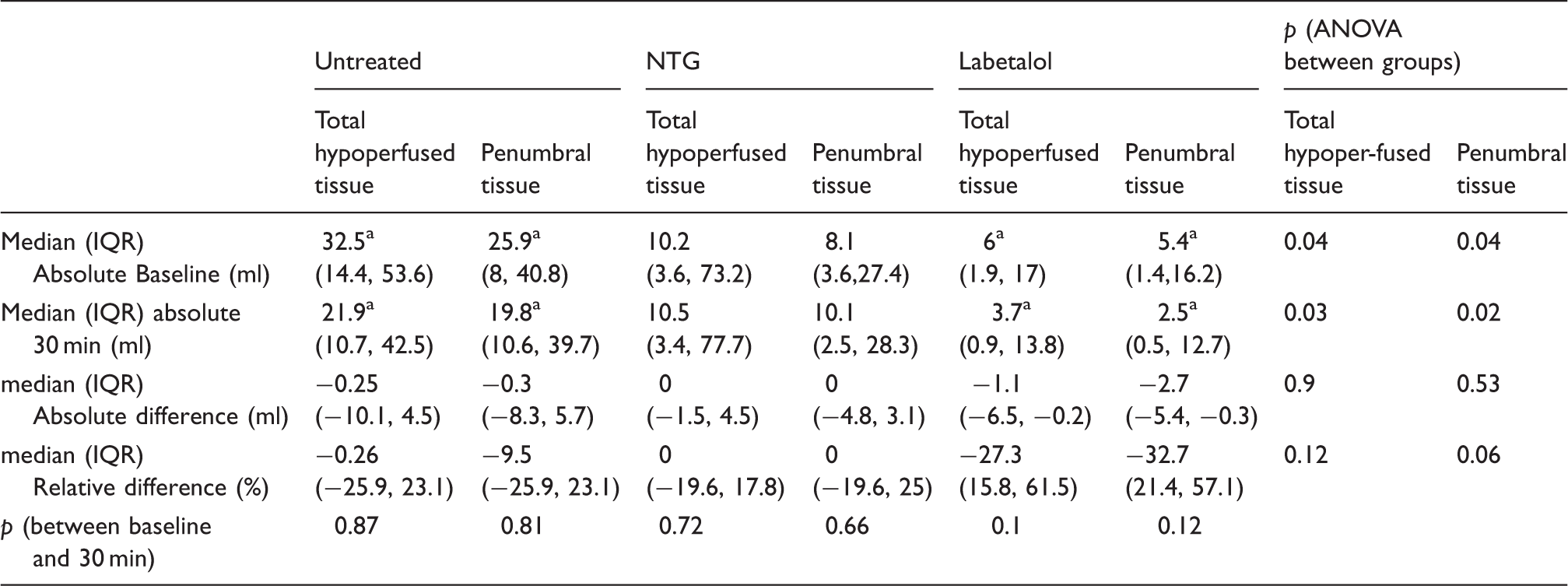
Post hoc analysis suggestive of a significant difference between untreated and labetalol groups.
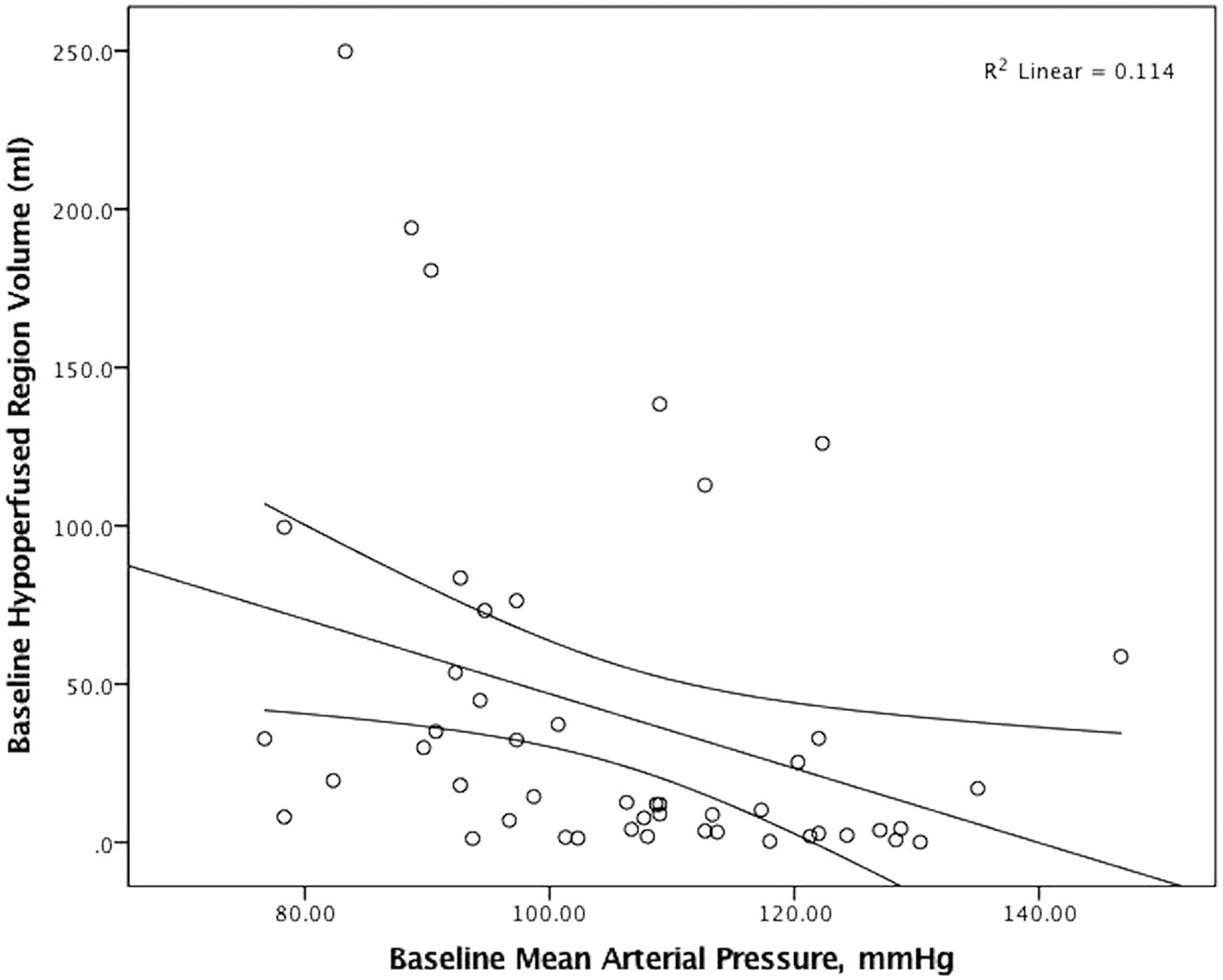
Linear regression and 95% confidence interval between baseline mean arterial pressure and baseline hypoperfused tissue volume in all patients. There was a trend to larger volumes of hypoperfused tissue in patients with lower pressures, but this was not significant.
Regional and relative cerebral perfusion changes by treatment group.
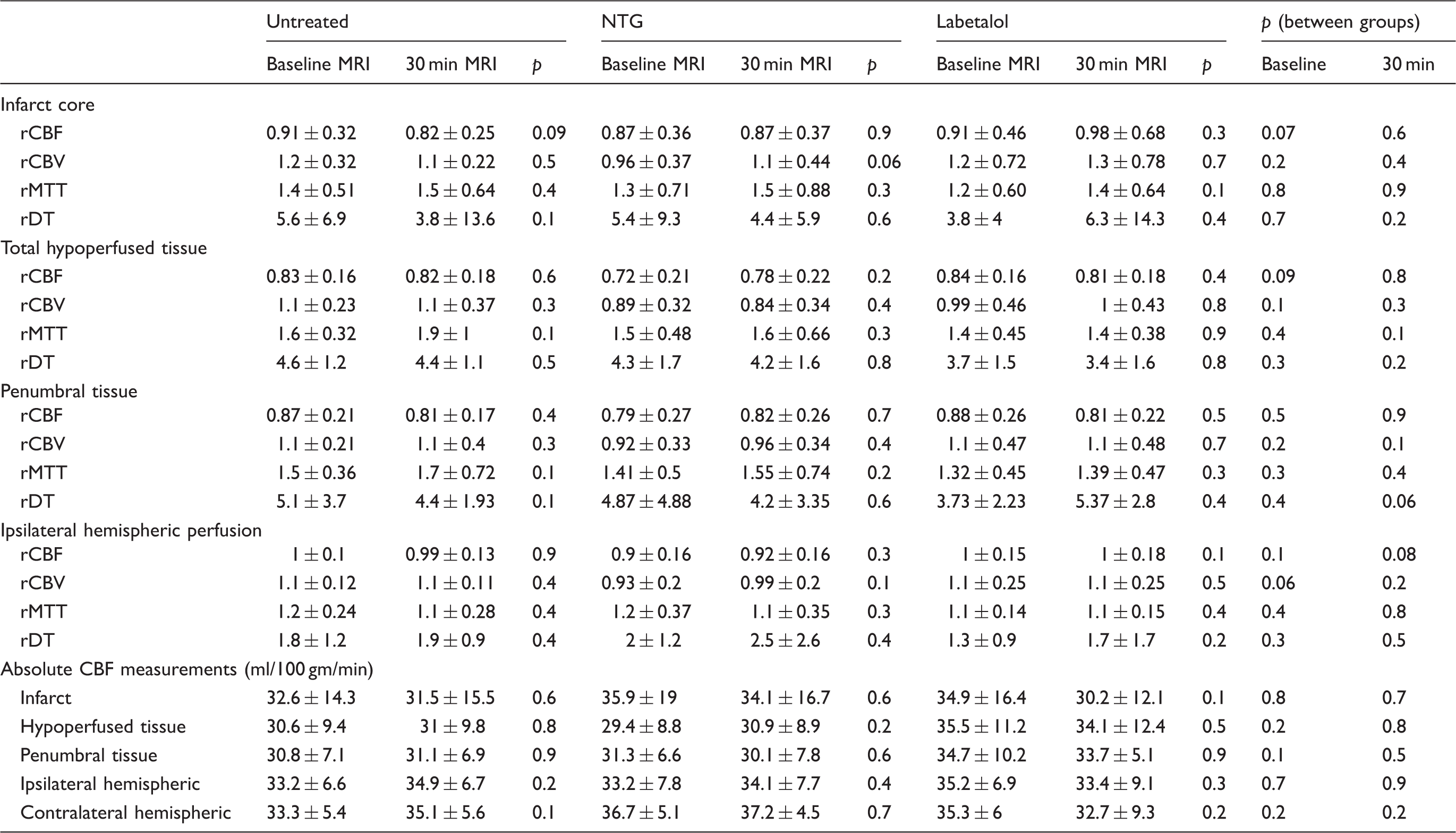
NTG: nitroglycerin; rCBF: relative cerebral blood flow; rCBV: relative cerebral blood volume; rMTT: relative mean transit time; rDT: relative delay time.
BP management
Patients in the MAP > 120 group received a median (IQR) labetalol dose of 15(10–20) mg in addition to SL NTG 0.3 mg. All patients with MAP 100–120 group received SL NTG 0.3 mg. Labetalol reduced MAP by 12.5(5.7–17.7) mmHg, p = 0.0002. MAP remained stable in the NTG (6.0 (0.4–16, p = 0.3) mmHg and untreated groups (−0.3 (−2.3–7.0, p = 0.2) mmHg (Table 1).
Outcome measures
Total hypoperfused and penumbral tissue volumes
The mean time between pre and post-treatment PWI acquisition was 26.3 ± 9.8 min. The absolute hypoperfused tissue and penumbral volumes remained stable between the two PWI acquisitions in all three groups (Table 2). There were also no between-group differences in the median relative hypoperfused tissue and penumbral tissue volume changes following BP reduction (Table 2).
Absolute and relative perfusion changes
There were no significant changes in any absolute or relative perfusion measure following BP reduction (Table 3). Linear regression indicated no relationship between the change in hypoperfused tissue volume and the decrease in MAP across groups (β = 0.101, 95% CI (−0.22) – 0.45, p = 0.5). There was no difference in penumbral rCBF at baseline and 30 min after in the untreated group (p = 0.4), NTG group (p = 0.7) and labetalol group (p = 0.5).
Clinical assessments
No patients' experienced clinical worsening following BP reduction. Two patients died during the study period due to severe systemic sepsis (days 45 and 90). There were no differences in 30-day NIHSS score (labetalol group: 1.5 (0, 3), NTG group: 2 (0.5, 6), untreated group: 2 (2, 11), p = 0.6) and 30 day mRS score (labetalol group: 2 (1, 3), NTG group: 3(1.5, 4), untreated group: 3 (2, 4), p = 0.4). Similarly there were no differences in 90 day NIHSS score (labetalol group: 2 (0, 3.5), NTG group: 3 (0,5.5), untreated group: 2 (1, 8.5), p = 0.7) and 90 day MRS score (labetalol group: 2 (1, 3), NTG group: 3 (1, 4), untreated group: 3 (1.5, 4, p = 0.3).
Discussion
In this three-group non-randomized prospective controlled study we found, cerebral perfusion was not affected by pharmacological reduction in BP in patients with subacute ischemic stroke. All absolute and relative measures of BP remained stable, irrespective of the degree of BP reduction, in our study. Blood flow was unaffected in the infarct core, total hypoperfused region (penumbra and core), penumbral tissue or the hemisphere ipsilateral to the stroke. Collectively, we found no evidence that acute BP treatment in hypertensive patients reduced perfusion in any region.
There are limited studies of cerebral perfusion following BP reduction in acute ischemic stroke. One investigation assessed effect of transdermal NTG (5 mg) on CBF (global, hemispheric, arterial territory and lesion) using xenon computed tomography and BP (peripheral and central) within 5 days of acute ischemic stroke and intracerebral hemorrhage (12 patients in the treatment arm and 6 patients in the control arm). CBF was measured before and 1 h after treatment initiation. A 14% decrease in systolic BP at 1 h was not associated with any reduction in CBF. 21 In the present study, we did not observe a decrease in MAP at 30 min despite using a higher dose of NTG, delivered more rapidly (0.3 mg of SL). In two recent clinical trials conducted in acute (<48 h from symptom onset 22 ) and hyper-acute (<4 hours from symptom onset 23 ) ischemic stroke patients, systolic BP was 7 mmHg (24 h) and 18 mmHg (2 h) lower in NTG treated than untreated patients. The lack of hypotensive effect of NTG observed in our study at 30 min may reflect a delayed onset of action. 24
A cerebral autoregulation study in acute ischemic stroke patients 1–11 days after stroke (n = 9) assessed CBF using PET at baseline and 1 h after IV nicardipine infusion (2.5 mg/h, up to maximum of 15 mg/h). 25 There was no decrease in CBF in infarct, peri-infarct or the ipsilateral hemisphere despite an 11% decrease in MAP. One patient developed clinical worsening associated with a 7 mmHg decline in MAP and a 30% decrease in the global CBF. The authors attributed this to a rightward shift of the cerebral autoregulatory curve due to chronic hypertension. They hypothesized that BP reduction in patients with chronic hypertension may lead to global, rather than focal, hypoperfusion. We did not observe clinical worsening in any of the patients in the current study, even in the labetalol group where a 10.2% decrease in MAP was achieved. In our study, 80% (n = 25/31) of the treated patients had a known history of hypertension, 40% of whom were on two or more antihypertensive agents. It is possible that patients in the PET study had more severe or uncontrolled hypertension, or alternatively their findings were an effect specific to nicardepine.26,27
In fact, a systematic review of 11 studies (313 stroke patients) assessing the effect of antihypertensive therapy (including angiotensin converting enzyme inhibitors, angiotensin-receptor antagonist, calcium channel blockers, diuretic and nitrates) on CBF or CBF velocity in acute ischemic stroke indicated no reduction in any perfusion measure after BP lowering. 28 Thus, global hypoperfusion as result of modest BP reduction may be the exception rather than the rule.
Recent systolic BP reduction trials in acute ischemic stroke have demonstrated no clinical benefit 23,29 but do provide some support for a tiered BP management approach in acute ischemic stroke patients and so we also used a stratified approach to BP treatment. Although the causal relationship between BP and mortality is unknown, it is possible that both hypertension and hypotension have detrimental effects in acute ischemic stroke. 9 Patients presenting with higher pressures may therefore tolerate some BP reduction. The evidence that this may be beneficial comes primarily from stroke thrombolysis studies indicating that elevated BP is associated with increased rates of hemorrhagic transformation.6,7,30 The BP arm of the ongoing Enhanced Control of Hypertension and Thrombolysis in stroke Study (ENCHANTED) is testing whether intensive lowering of BP (130–140 mmHg) improves clinical outcomes and decreases the risk of hemorrhagic transformation. 31 The efficacy of nitric oxide in stroke (ENOS) study results also suggest early BP reduction (<6 h) reduces post-stroke disability. 23 Thus, lowering BP in selected patients may be beneficial. Other studies have suggested that NTG may improve CBF, potentially via its vasodilatory effect, 32 although we did not observe any change in flow in our study.
Our study has several limitations. It was relatively small and patients were not randomized, although outcomes were collected in blinded manner. In addition, post-treatment cerebral perfusion was measured after a mean of 26 min. Although we did not observe rapid changes in blood flow after antihypertensive therapy, longer duration therapy may reduce cerebral perfusion. 33 The majority of patients treated with labetalol had small infarcts and hypoperfused tissue volumes at baseline. These results may therefore not be generalizable, particularly in patients with large infarcts. Most patients had documented chronic hypertension before their stroke and were taking oral antihypertensive drugs, which may have affected outcomes. We examined beta-blockers and nitrates only and cannot make any assertions about other drug classes. Data are also not generalizable to the hyperacute phase of acute stroke since our patients were treated >24 h after onset.
Conclusion
BP reduction in patients with elevated MAP (>120 mmHg) was not associated with differences in relative or absolute cerebral perfusion severity or volume in the infarct core, penumbral tissue or total hypoperfused region. Whether or not BP lowering in stroke patients with high initial BP may benefit from more aggressive treatments remains unknown.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This trial was funded by grant-in-aid support from Canadian Institutes of Health Research (G11816079). KB holds a Canada Research Chair in Cerebrovascular Disease, a Heart and Stroke Foundation of Alberta (HSFA) Professorship in Stroke Medicine and a New Investigator Award from Alberta Innovates Health Solutions (AIHS). MK and LG were supported by the Clinician Fellowship, Alberta Innovates Health Solutions, Alberta, Canada. SRM holds the Endowed Chair in Patient Health Management supported by the Faculties of Medicine and Dentistry and Pharmacy and Pharmaceutical Sciences at the University of Alberta.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors' contributions
MK – Acquisition of data, analysis, interpretation, drafting article, final approval. NG – Acquisition of data, revising, final approval, LG – Acquisition of data, analysis, revising, final approval, BB – Acquisition of data, revising, final approval, SM – Concept design, revising, final approval, TJ – Acquisition of data, revising, final approval, AS – Acquisition of data, revising, final approval, DE – Acquisition of data, revising, final approval, CB – Concept design, revising, final approval KB – Concept design, Acquisition of data, revising, final approval.
