Abstract
Low cerebral levels of brain-derived neurotrophic factor (BDNF), which plays a critical role in many brain functions, have been implicated in neurodegenerative, neurological and psychiatric diseases. Thus, increasing BDNF levels in the brain is considered an attractive possibility for the prevention/treatment of various brain diseases. To date, BDNF-based therapies have largely focused on neurons. However, given the cross-talk between endothelial cells and neurons and recent evidence that BDNF expressed by the cerebral endothelium largely accounts for BDNF levels present in the brain, it is likely that BDNF-based therapies would be most effective if they also targeted the cerebral endothelium. In this review, we summarize the available knowledge about the biology and actions of BDNF derived from endothelial cells of the cerebral microvasculature and we emphasize the remaining gaps and shortcomings.
Keywords
Introduction
In the brain, the synaptically released neurotrophin brain-derived neurotrophic factor (BDNF) has emerged as a real mediator of synaptic plasticity and synaptic communication. Therefore, neuronal-derived BDNF and its cognate TrkB (tropomyosin-related kinase B) receptors have been considered promising targets for the treatment of many neurological, neurodegenerative and psychiatric diseases. 1 However, in contrast to the traditional thinking that BDNF present in the adult brain is mostly neuronal BDNF, 2 the in situ removal of the endothelium from the cerebral microvasculature was reported to result in a marked decrease in brain BDNF levels. 3 Endothelium removal was achieved by a brain perfusion (by transcardial route) with a 0.2% CHAPS (3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulphonate) solution. Importantly, as BDNF is also present in blood, CHAPS perfusion was preceded by saline perfusion in order to avoid blood contamination of the brain as a confounding factor. More precisely, BDNF levels analyzed by Western blotting were twice lower in brain without endothelial cells than in brain with intact endothelium. Even though Western blotting is only a semi-quantitative method, these data suggest that ∼50% of BDNF levels measured in brain homogenates correspond to BDNF expressed by endothelial cells. This paradigm shift in our understanding of the cellular source of brain BDNF combined with evidence that cultured cerebral endothelial cells (CECs) secrete BDNF in a bioactive form4–6 supports the exciting hypothesis that normal brain functioning might be dependent on BDNF synthesis and secretion by the cerebral endothelium and that endothelial BDNF might be the missing link between endothelial function and cognition. Unfortunately, research on cerebrovascular BDNF is still in its infancy as compared with the major progress in our understanding of neuronal BDNF synthesis, secretion and functions. In the present review, after a brief overview of neuronal-derived BDNF, we will present the specificities of the endothelium of the cerebral microvasculature. Then, after a presentation of the methodological considerations regarding the measurement/detection of endothelial BDNF, we will present the available knowledge on secretion, regulation and functions of BDNF derived from endothelial cells of the cerebral microvasculature. Finally, the key shortcomings to resolve in future research will be summarized.
Neuronal-derived BDNF
Neuronal synthesis, storage and release of BDNF have been reviewed in detail elsewhere.7,8 This paragraph only summarizes what is known on neuronal-derived BDNF with the objective to show readers that research on cerebrovascular BDNF is still in the early stage as compared with research on neuronal BDNF.
Synthesis and secretion
BDNF was first discovered in the porcine brain. 9 This probably explains why almost all of what is known about BDNF synthesis and secretion concerns neuronal BDNF. Like all peptides, BDNF synthesis starts from the transcription of the BDNF gene into BDNF mRNAs which are then processed into pre-proBDNF, proBDNF and mature BDNF (mBDNF) via the Golgi apparatus and the trans-Golgi network. The secretion of mBDNF by neurons is both constitutive and regulated, the latter being preferential and activity-dependent. 10 The fact that mBDNF secretion is under the control of neuronal activity-dependent mechanisms is important as it explains how mBDNF modulates the synaptic plasticity in response to experience and environment. BDNF exerts its action primarily in an anterograde fashion,11–14 i.e. BDNF is released by axons and binds to presynaptic and postsynaptic receptors. As the widespread diffusion of mBDNF is prevented by its high molecular weight (28 kDa for the mBDNF homodimer) and its positive charge under physiological conditions (isoelectric point close to 1015), BDNF secreted by axons only acts locally. For instance, it has been shown that the neuronal source of BDNF has to be within a distance of 4.5 µm to induce dendritic growth in the recipient neurons. 16 However, there is substantial evidence that BDNF can also participate in retrograde local or axonal signaling. Here, BDNF is released from postsynaptic neurons and binds to presynaptic receptors. According to local retrograde signaling, BDNF elicits a rapid change in presynaptic activity. 17 By contrast, according to axonal retrograde signaling, BDNF induces delayed changes in transcription processes as a result of the transport of BDNF-TrkB complex from the distal terminal to the nucleus as previously described for nerve-growth factor. 18 However, whether or not dendrites secrete BDNF is a matter of debate. While BDNF secretion by dendrites was reported to occur in cultured neurons,19,20 immunoreactivity experiments on mice hippocampus failed to detect the presence of granules containing BDNF in dendrites. 14
Although controversial, 21 cultured neurons were reported to secrete proBDNF22,23 that is subsequently cleaved into mBDNF in the extracellular space by plasmin. 24 Plasmin, which is generally expressed in neurons as an inactive plasminogen, must be activated by proteolytic cleavage by tissue plasminogen activator (t-PA). Both proBDNF and t-PA secretion by neurons seems to be activity-dependent 25 but only high-frequency neuronal activity induces t-PA secretion. 23 Of interest, mBDNF stimulates the expression of t-PA in primary cultures of cortical neurons in a time- and concentration-dependent manner.26,27
Neuronal receptors activated by mBDNF
mBDNF binds to two kinds of plasma membrane receptors, TrkB receptors and pan75 neurotrophin receptor (p75NTR), the affinity of mBDNF being much higher for the former type (dissociation constant ∼10−11 M vs. 10−9 M). In fact, p75NTR is the preferential receptor for proBDNF, and activation of p75NTR/sortilin complex often leads to the activation of apoptotic pathways and death.28–30 TrkB receptors include full-length (FL) receptors, which are linked to a tyrosine kinase domain, and truncated (T1, T2, T3) receptors, which are devoid of a tyrosine kinase domain.31–33 TrkB-FL and -T receptors share the same extracellular domain and consequently bind mBDNF with the same affinity. The binding of mBDNF to membrane TrkB-FL receptors induces trans-phosphorylation of TrkB cytoplasmic domain tyrosine residues, initiating recruitment of signaling adapter proteins that foster signaling by ras/ERK1/2, PI3 kinase/Akt STAT and phospholipase Cγ pathways. 34 This pathway has proven to be of elementary importance in the ability of mBDNF to promote neurogenesis, neuroplasticity and neuronal survival. Then, the complex BDNF/TrkB-FL is internalized by endosomes from which the receptor is either degraded by lysosomes or recycled back to the cell surface or delivered to the neuronal soma (axonal retrograde transport) in order to permit control of nuclear transactivation of genes. Interestingly, evidence that regulation of TrkB transcription, TrkB mRNA trafficking, TrkB insertion at the neuronal surface and BDNF-TrkB complex endocytosis are activity-dependent 35 may explain why the actions of BDNF are restricted to active synapses. The other major TrkB receptor expressed by neurons is TrkB-T1. In cells that co-express TrkB-FL and TrkB-T1, the binding of mBDNF to TrkB-T1 results in the inhibition of TrkB-FL signaling. However, in addition to this role of a dominant negative receptor, TrkB-T1 might induce its own signaling. 33 Finally, the formation of heteromeric complexes between TrkB isoforms and p75NTR, 36 which are frequently coexpressed in the same cells, contributes to the complexity of the neurotrophic response. Another source of complexity is the activation of TrkB-FL in the absence of mBDNF.37,38
The human BDNF variant Val66Met
In humans, a Val66Met polymorphism has been identified in the BDNF gene. Approximately 30% of people worldwide are heterozygous (Val/Met) for the methionine substitution at codon 66 in the prodomain of the BDNF gene and this percentage varies, depending on both the geographical region and ethnicity. 39 Studies in neurosecretory cells and primary cultured neurons have shown that the Met substitution interferes with intracellular trafficking of proBDNF to the secretory pathway, thus resulting in reduced activity-dependent secretion of mBDNF.40,41 A number of clinical studies have reported lower cognitive performance,40–42 increased susceptibility to depression and anxiety,43,44 greater errors in short-term motor learning 45 and reduced neuroplastic response to physical exercise and post-stroke rehabilitation46,47 in Met allele carriers. However, other studies failed to report a major impact of this polymorphism on brain functioning.48,49
Endothelium of the cerebral microvasculature
This paragraph provides a short anatomical and functional description of the endothelium of the cerebral microvasculature. This knowledge represents a basis for discussing the potential fate of endothelial-derived BDNF and its potential recipient cells in vivo.
Localization
The endothelium of the cerebral microvasculature corresponds to the monolayer of endothelial cells lining intraparenchymal brain vessels from arterioles to venules with endothelial cells anchored to the endothelial basement membrane. A particularity of the cerebral microvasculature is that astrocyte end-feet completely covers the wall of vessels present in the brain neuropil
50
even though capillaries were found to have substantially more processes contacting them per unit surface area than arterioles and venules.
51
For precapillary arterioles, capillaries and postcapillary venules, endothelial cells are surrounded by pericytes (30% coverage), which are encased in the endothelial basement membrane (Figure 1) and display heterogeneity in terms of morphology and contractile protein expression as a function of position along the capillary bed.
52
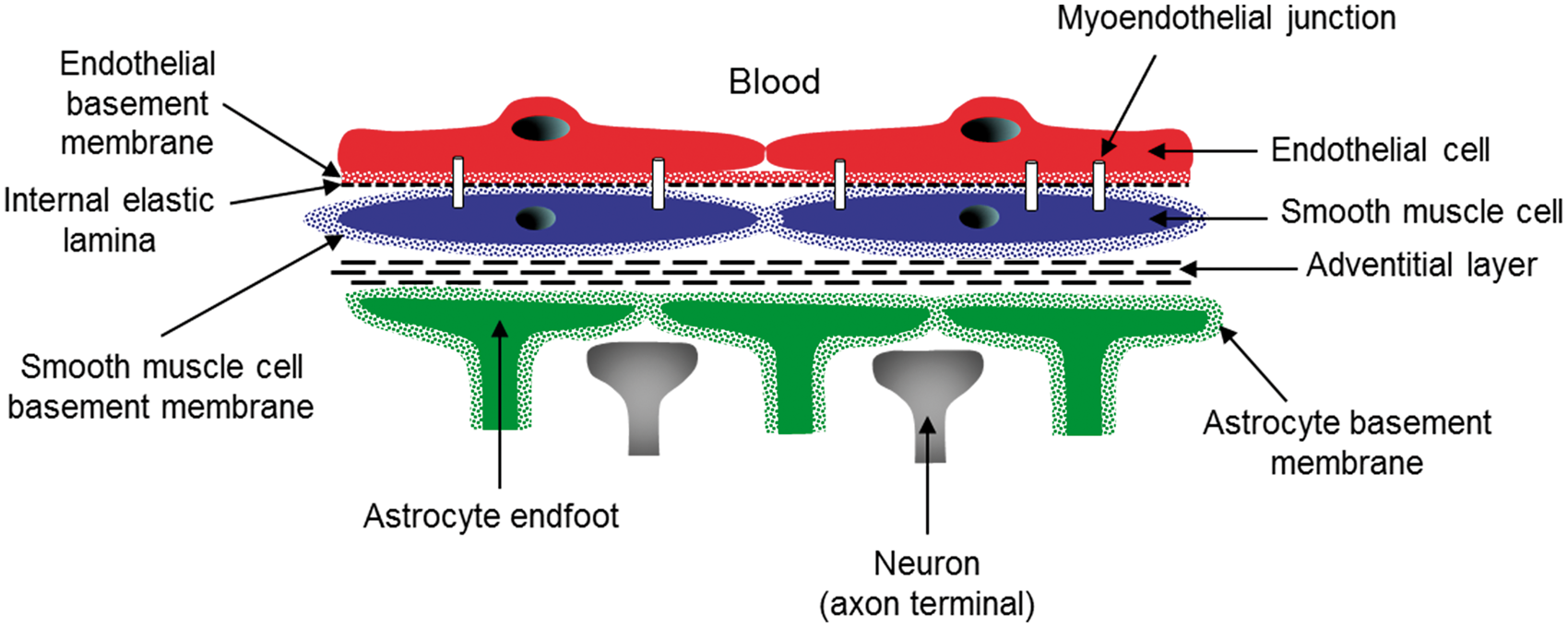
For arterioles and venules, the vascular wall includes the intima layer (endothelial cells separated from an internal elastic lamina by the endothelial basement membrane), the media layer, which comprises a single layer of smooth muscle cells (SMCs) surrounded by a basement membrane and the adventitial layer (fibroblasts not surrounded by a basement membrane and collagen fibers) (Figure 2). In these vessels, endothelial cells are separated from SMC by the endothelial basement membrane, which adheres to the basal surface of endothelial cells, and from the internal elastic lamina and the SMC basement membrane, which adheres to the surface of SMC. However, endothelial cells form extensions that project through fenestrations in the internal elastic lamina and basement membranes to contact SMC directly.
53
These extensions (termed myoendothelial projections) provide direct contact between endothelial cells and SMC through gap junctions but do not allow the diffusion of mBDNF. In contrast, direct cell–cell contact has not been observed between astrocytic end-feet and SMC. Finally, the notion of neurogliovascular units (NGVUs) emphasizes the interplay between endothelial cells of capillaries and neighboring pericytes, astrocytes and neurons. Importantly, this interplay probably involves a paracrine pathway. Indeed, it is unlikely that a heterotypic gap junction between these different cell types can be established as endothelial cells, pericytes and astrocytes are physically separated from each other by a basal lamina.
50
Schematic representation of a transversal section of a cerebral capillary and its neighboring cells. The wall of a cerebral capillary includes a layer of endothelial cells (red) and the basement membrane (red points). This wall is covered by pericytes (purple). Astrocytic end-feet (green) is separated from endothelial cells by the endothelial (red points) and astrocytic (green points) basement membranes. Both astrocytes and pericytes are innervated by local neurons (grey). Note that the drawing is not to true scale. Schematic representation of a longitudinal section of a cerebral arteriole. The wall of cerebral arterioles includes endothelial cells (red), the basement membrane of endothelial cells (red points), the internal elastic lamina (black fine dotted line), a single layer of smooth muscle cells (blue) surrounded by their basement membrane (blue points) and an adventitial layer (large dotted lines) with fibroblasts (not shown). The arterial wall is covered by astrocytic end-feet (green) surrounded by their basement membrane (green points) and is innervated by local neurons (grey). Note the presence of myoendothelial junctions. Note that the drawing is not to true scale.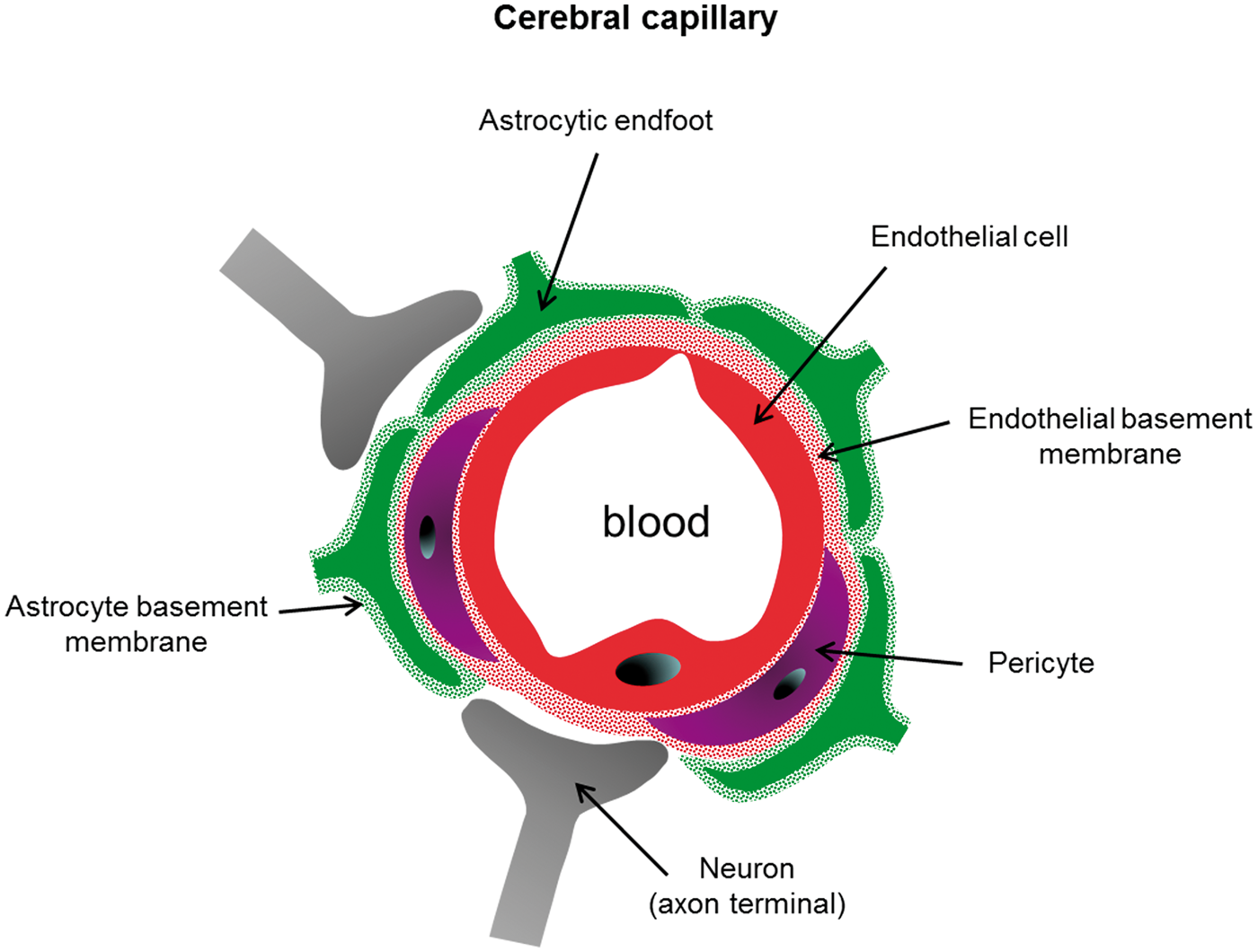
Features and functions of brain endothelial cells
Brain endothelial cells are phenotypically unique and different from endothelial cells in the periphery. They have apical tight junction complexes that prevent paracellular diffusion but express specific transporters to actively transport nutrients from the blood into the brain. Endothelial cells also basolaterally express transporters to remove toxic substances from the brain into the blood. Other unique features of the cerebral endothelium are a lack of fenestrations, a very low rate of pinocytosis, which limits transcellular transport, and a high number of mitochondria. 54 Endothelial cells are the most important cellular component of the blood–brain barrier and barrier properties seem to be more expressed in capillaries than in larger vessels 55 even though the expression of markers of the BBB varies greatly among the endothelial cells of capillaries. 56 An unresolved point concerns the permeability of the BBB to BDNF57–59 even though certain studies60,61 but not all62,63 reported positive correlations between blood and regional brain BDNF levels. Besides being implicated in barrier properties, the endothelium of the cerebral microcirculation is also involved in numerous processes, including the regulation of inflammatory and immune responses, 64 thrombosis, 65 neuroplasticity, 66 angiogenesis 67 and cerebral blood flow.68,69 Endothelial cells of the neural stem niches were also reported to stimulate self-renewal and expand neurogenesis. 70
Methods to investigate endothelial BDNF synthesis and secretion
BDNF synthesis by endothelial cells of the cerebral microvasculature can be investigated by measuring levels of BDNF mRNA, proBDNF or mBDNF protein in cell lysates prepared from primary or immortalized CECs or cerebral microvessel-enriched fractions. The contamination of these preparations with either pericytes or astrocytes, which have been reported to express BDNF,71–74 may be a confounding factor. The secretion of BDNF (proBDNF or mBDNF) by endothelial cells can be assessed from changes in levels of these compounds in CEC culture medium. Importantly, the measurement of phosphorylated TrkB-FL receptor levels in brain homogenates, which is traditionally used to gather information on mBDNF secretion in vivo, fails to identify the cellular source of mBDNF (neuronal vs. non-neuronal cells). It must also be kept in mind that levels of proBDNF (35 kDa) and mBDNF (14 kDa) protein can be measured separately using Western blotting analysis only. Indeed, while commercial ELISA kits are available for BDNF measurement, they use antibodies that are not specifically directed against mBDNF and the affinity of antibodies for mBDNF and proBDNF differs from one kit to another. 75 The same concern is valid for antibodies used to localize mBDNF by immunoreactivity experiments. In the following paragraphs, the term “mBDNF” will be used when data have been provided by Western blotting analysis. When data come from the measurement of BDNF by ELISA kits or from immunoreactivity experiments, the term “BDNF” will be used.
Expression and secretion of BDNF by CECs in baseline conditions
To the best of knowledge, Leventhal et al. 4 published the first paper on endothelial BDNF. The authors showed that primary human CEC and human umbilical vein endothelial cells (HUVEC) produced BDNF mRNA and constitutively released the bioactive form of BDNF. Later, BDNF mRNA levels were reported to be five times lower in HUVEC than in human CEC 76 suggesting that transcription processes are less activated, or alternatively that BDNF mRNA processing is more rapid in HUVEC than in human CEC. An interesting but often neglected finding is that the rate of BDNF production by mouse CEC cultured in static conditions was estimated to be 50 times greater than that by cultured (unstimulated) neurons. 5 Evidence that cerebral microvessel-enriched fractions (isolated from the rat cortex) express BDNF mRNA and mBDNF in baseline conditions is more recent3,77 and clearly indicates that the cerebral microvasculature synthesizes and secretes mBDNF in vivo. However, BDNF in CECs might also originate from the fusion of BDNF-containing exosomes with endothelial cells. Indeed, more than 90% of blood BDNF is stored in platelets 78 and platelet BDNF is present not only in cytoplasmic but also in a vesicle (alpha granules) pool.79,80
Cultured CEC secrete a bioactive form of BDNF as evidenced by the fact that the neuroprotective and angiogenic effect of conditioned media from cultured CEC is inhibited by the removal of BDNF or the application of TrkB receptor antagonists (see below). Cultured human CEC have revealed that BDNF secretion is both constitutive and regulated and that BDNF secretion in the medium decreased with the number of cell passages. 81 One question that remains to be investigated is whether cultured CEC secrete proBDNF, and if so, whether t-PA secreted by endothelial cells (a major source of t-PA) is involved in the extracellular maturation of the peptide. Another point that deserves further investigation is whether the BDNF Val66Met polymorphism affects BDNF processing and subsequent mBDNF release by the cerebral endothelium. Strikingly, a genetic knock-in mouse carrying the human BDNF Val66Met polymorphism displays a hypercoagulable state, 82 reduced endothelial cell proliferation and vessel density in response to stroke. 83 In addition, in patients with reversible cerebral vasoconstriction syndrome, vasoconstriction scores were higher in patients with the Val66Met polymorphism. 84 Collectively, these data support the notion of an interaction between vascular biology and the Val66Met polymorphism. However, whether this polymorphism results in the decreased secretion of mBDNF by endothelial cells is not known.
Modulation of endothelial BDNF synthesis/secretion
Control of endothelial BDNF synthesis/secretion by CECs has been poorly investigated and available studies have been mainly restricted to the role of nitric oxide (NO), the best characterized endothelium-derived factor, even though control by pro-inflammatory cytokines and hypoxia was also explored.
Modulation by NO
The role of NO in endothelial BDNF synthesis/secretion was first investigated using the exposure of cultured mouse CEC to a NO donor. The NO donor, NOC-18, was first reported to induce BDNF overexpression by immortalized mouse CEC. 85 By contrast, exposure of mouse CEC to the NO donor sodium nitroprusside (SNP) decreased BDNF levels in the medium. 5 These apparently discrepant results may be explained by differences in the chemical nature of NO donors and resulting changes in intracellular NO concentrations. Indeed, unlike SNP, which rapidly increases NO concentration, NOC-18 is a slow-releasing NO donor and consequently delivers NO more progressively. Consistent with this hypothesis, the exposure of cerebral microvessel-enriched fractions (isolated from the rat cortex) to glyceryl trinitrate (GTN), which is another slow-releasing NO donor, also increased mBDNF levels. 3 As NOC-18 and GTN better mimic endogenous production of NO by endothelial cells, it is tempting to suggest that NO positively controls endothelial BDNF synthesis via an autocrine pathway. One argument for this hypothesis is the strong correlation that we found in rat cerebral microvessel-enriched fractions between eNOS phosphorylated at serine 1177 (peNOSser1177 as an index of sustained NO production 86 ) and mBDNF levels after modulation of peNOSser1177 by hypertension (decrease) and physical training (increase). 87 This association between endothelial NO production and mBDNF expression is in line with the stimulating effect of shear stress elevation on mBDNF synthesis and secretion by HUVEC 88 since shear stress is the most potent stimulus to induce phosphorylation of eNOS at serine 1177 in HUVEC. 89 A positive control of endothelial mBDNF synthesis by NO is consistent with studies that reported downregulation of BDNF production by cultured human brain microvascular endothelial cells exposed to the eNOS inhibitor asymmetric dimethyl arginine (ADMA) 90 or to advanced glycation end products, 77 which act as NO scavengers. 91 More recently, using adjuvant-induced arthritis (AIA) in rats as a model of endothelial dysfunction (i.e. decreased endothelial NO bioactivity), 92 we reported lower BDNF levels in cerebral microvessel-enriched fractions in AIA than in control rats as well as the efficacy of GTN to prevent the vascular BDNF changes induced by AIA. 93 The fact that decreased cerebrovascular mBDNF levels and endothelial dysfunction are found in arthritis, diabetes and hypertension suggests that decreased mBDNF synthesis by CEC could be a useful marker of endothelial function at the cerebral level. However, whether changes in endothelial mBDNF expression is an epiphenomenon or contributes to endothelial dysfunction is unknown. Finally, as NO positively controls t-PA release by human endothelial cells 94 and as proBDNF could be secreted by endothelial cells, it is conceivable that NO, and by extension endothelial function, may control the maturation of secreted proBDNF in the extracellular space.
Modulation by other factors
Although neuroinflammation is an important component in the pathogenesis of neurological (stroke, multiple sclerosis) and neurodegenerative (Parkinson and Alzheimer) diseases, the link between inflammation and BDNF synthesis by the cerebral endothelium has been poorly explored. Long-lasting (72 h) but not short-lasting (24 h) exposure of cultured human CEC to TNFα (10 ng/mL) was reported to increase BDNF mRNA in cell lysates and BDNF protein in the medium. 95 In addition, rat endothelial cells display increased BDNF immunoreactivity in the acute post-stroke period71,96 even though hypoxia rather than neuroinflammation may be involved. Indeed, hypoxia was reported to increase mBDNF levels in lysates of rat CEC and BDNF immunoreactivity in the cerebral microvasculature. 6 It is noteworthy that the stimulating effect of hypoxia on BDNF synthesis and secretion by mouse CEC is higher for intermittent hypoxia (IH) than for continuous hypoxia, the stronger effect of IH being related to oxidative stress and calcium mobilization from internal stores. 97
Fate of BDNF secreted by the brain endothelium
According to the histology of the cerebral vasculature, the potential recipient cells of BDNF derived from endothelial cells of the cerebral microvasculature include endothelial cells themselves, pericytes, astrocytes, neurons and SMC providing that these cells express TrkB receptors. Unfortunately, TrkB expression by cells other than neurons is poorly documented. TrkB receptor protein was detected in cultured rat CEC,
6
while TrkB mRNA was not found in human CEC collected from neurosurgical preparations.
98
We recently detected TrkB-FL and TrkB-T receptors in rat cerebral microvessels-enriched fractions.
93
Rodent astrocytes express TrkB, but only the TrkB-T1 isoform.99,100 Human pericytes do not express TrkB.
101
Finally, to the best of our knowledge, studies aiming to explore TrkB expression by SMC of the cerebral microvasculature are lacking, while rats cultured SMC express TrkB receptor mRNA.
102
Figure 3 summarizes the cellular localization of TrkB and P75NTR receptors in the brain.
Localization of receptors recognized by mBDNF and proBDNF. Except for pericytes (purple), all the remaining cells of the neurogliovascular unit including endothelial cells (red), astrocytes (green) and neurons (grey) express TrkB-FL (yellow diamond), p75NTR (blue diamond) and/or TrkB-T1 (pink diamond) receptors. See the tripartite synapse at the top of the figure. Note that the drawing is not to true scale.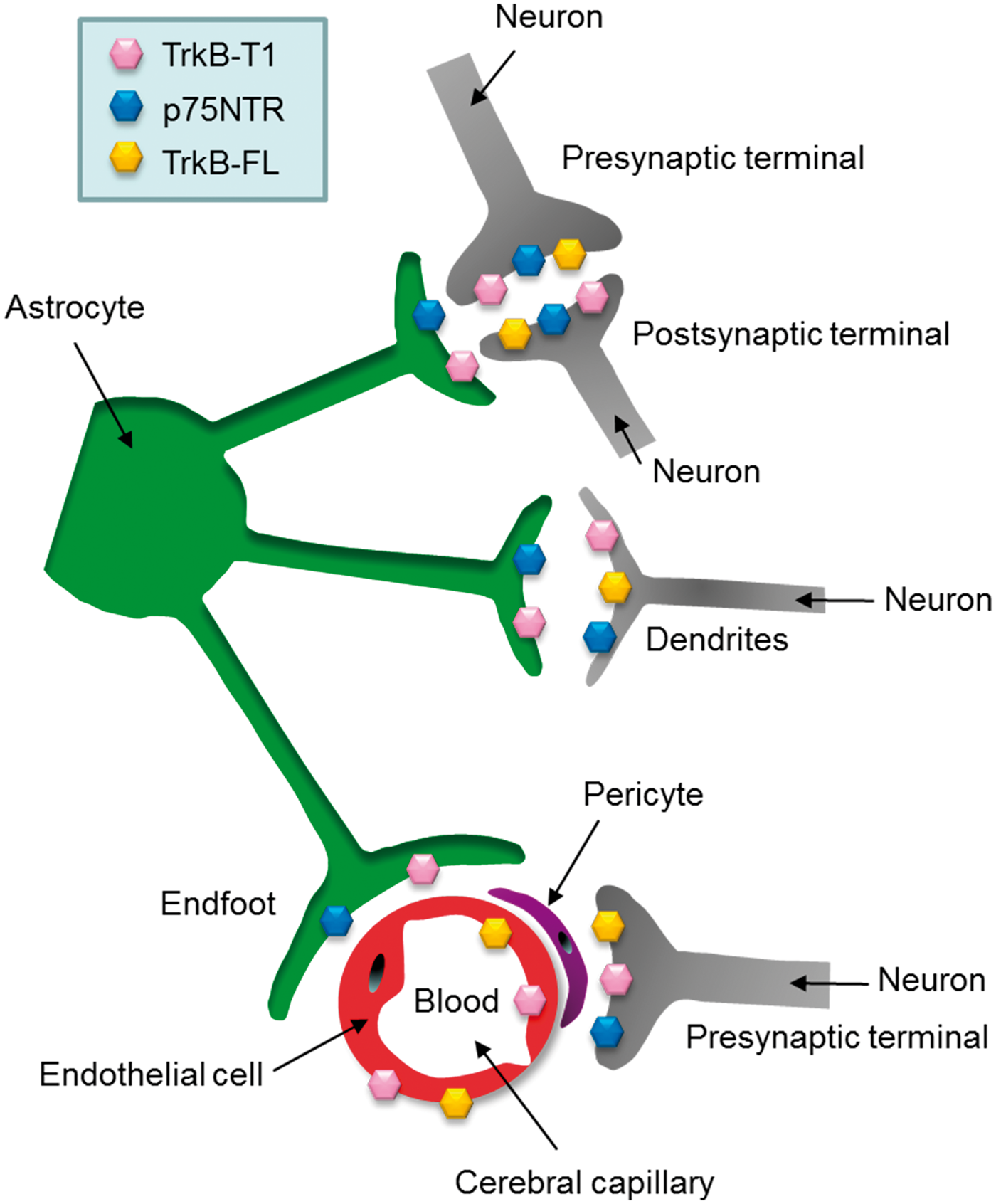
Both the luminal (facing the blood) and basal (facing the brain) membrane of CEC can in theory secrete mBDNF (Figure 4). When released by the luminal membrane into the blood, it is unlikely that mBDNF exerts an endocrine effect as plasma mBDNF is probably rapidly taken up by platelets.
78
Consistent with this hypothesis, the plasma half-life of mBDNF does not exceed 2 min.
103
In contrast, an autocrine effect of BDNF released by the luminal membrane of cerebral endothelial is possible as suggested by the strong association between BDNF and phosphorylated TrkB receptors levels in rat cerebral microvessel-enriched fractions.
93
If released by the basal membrane, mBDNF may again act as an autocrine factor and, in addition, as a paracrine factor. In the latter case, mBDNF derived from rat endothelial cells needs to diffuse through the basement membranes, which may hamper its diffusion as a result of its negative charge and its thickness of ∼50 nm.
104
Once mBDNF has diffused through the basement membranes, the nature of potential recipient cells differs depending on the localization of endothelial cells, inside versus outside the NGVU. At the NGVU level, endothelial-derived BDNF can be recognized by TrkB receptors expressed by astrocytes (TrkB-T1) and/or neurons (TrkB-FL and TrkB-T). As regards mBDNF released by endothelial cells outside the NGVU (endothelial cells of arterioles and venules), its unique recipient cells are SMC of the media layer, the limited extracellular diffusion of BDNF being incompatible here with any action on neurons or astrocytes.
Fate of mBDNF secreted by endothelial cells of cerebral capillaries. mBDNF might be secreted into the blood by the luminal membrane or into the cerebral interstitial fluid by the basal membrane of endothelial cells (red). Once secreted into the cerebral interstitial fluid, mBDNF can exert a paracrine action on astrocytes (green) or neurons (grey) as well as an autocrine action.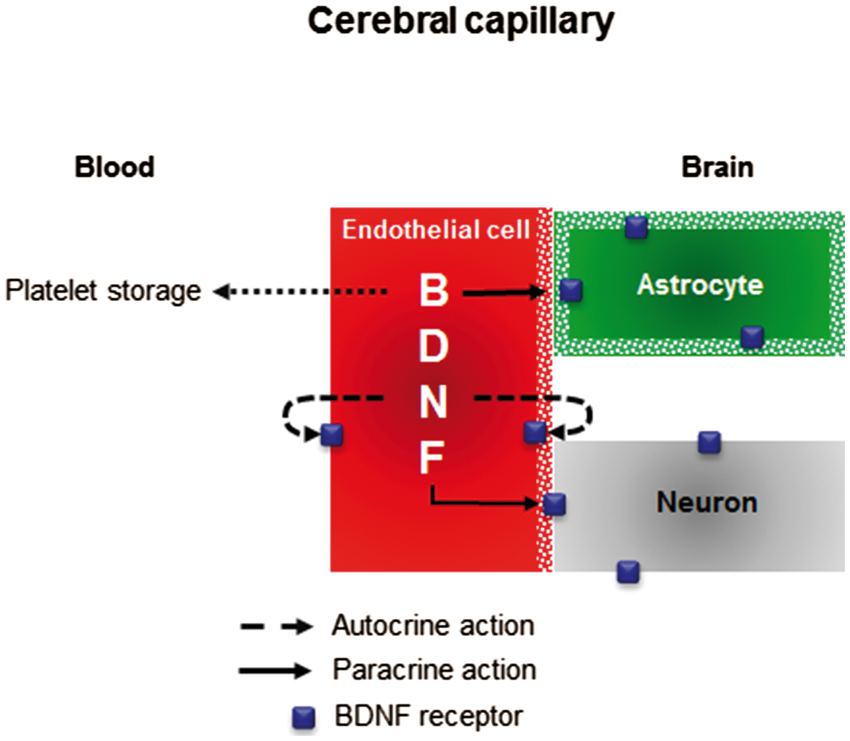
Physiological role of cerebral endothelial BDNF
While the link between cardiovascular risk and endothelial dysfunction has been known for more than two decades, 105 the link between endothelial dysfunction and impaired cognition has been identified more recently. For instance, cognition is impaired in patients with hypertension,106–108 diabetes109–111 and rheumatoid arthritis.112–114 It is noteworthy that cerebral endothelial BDNF expression is decreased in animal models of hypertension, diabetes and rheumatoid arthritis, supporting the notion that low endothelial BDNF expression might be a new marker of endothelial dysfunction. However, whether altered BDNF expression is a pathogenic event in or a consequence of endothelial dysfunction remains to be explored. An additional point that deserves further attention concerns the place of endothelial BDNF in the link between endothelial function and cognition. Indeed, available studies suggest that cognition might be dependent on BDNF derived from endothelium of the cerebral microvasculature. However, these studies were mainly performed on cell culture models which do not reproduce the in vivo environment of CEC and the limited capacity of BDNF diffusion. These studies as well as the potential role of endothelial BDNF in neuroplasticity are presented in the following section.
Endothelial BDNF and neurogenesis
Neurogenesis is a process of generating functional neurons from neural precursors. In the adult brain, neural progenitors are present in the subventricular zone (SVZ), where they are located adjacent to a layer of ependymal cells in the lateral ventricles. They are also present in the dentate gyrus (DG) near the hilus in the subgranular zone. An additional germinal zone of brain is the subependymal layer of the posterior periventricular (PPv) area surrounding the hippocampus. SVZ neurogenesis seems to replace neurons that die in the olfactory bulb after migration along the rostral migratory stream, while DG and PPv neurogenesis serves to replenish hippocampal neurons. Interestingly, neurogenesis in both SVZ and DG is stimulated by brain ischemia 115 and physical training.116,117 In support of the notion that BDNF derived from endothelial cells of the neural stem niches plays a role in neurogenesis, explants of the subependymal layer of the adult rat raised on human CEC generated more neurons, which survived longer, than did explants raised on astrocytes, fibroblasts and laminin. 4 mBDNF secreted by mouse CEC has also been shown to guide neuronal precursor cell migration from the SVZ to the olfactory bulb. 118 More recently, migration of newly generated neurons from SVZ towards the ischemic striatum was reported to be dependent on mBDNF produced by mouse vascular cells. 119 This finding, combined with the possibility that endothelial BDNF synthesis is controlled by eNOS-derived NO, resonates with previous studies that identified NO formed by eNOS as a promotor of stroke-induced neurogenesis 120 but contrasts with the study of Nygren et al., 121 which reported greater neurogenesis in BDNF heterozygous knockout mice than in wild type mice following brain ischemia. Last but not the least, while decreased neurogenesis is a hallmark of animal models of Alzheimer disease,122,123 no studies have investigated whether BDNF synthesis by CEC is impaired in this disease, even though the peptide amyloid Aβ was reported to decrease mBDNF production by mouse CEC. 5
Endothelial BDNF and neuroprotection
Studies have suggested that mBDNF secreted by cerebral capillaries plays a neuroprotective role, as BDNF secreted by cultured CEC increased resistance of cultured neurons against hypoxia, oxygen-glucose deprivation, oxidative damage-related endoplasmic reticulum stress and Aβ-induced death of primary neurons.5,77,124
Endothelial BDNF and cerebral angiogenesis
Angiogenesis that occurs by the processes of intussusception and sprouting 125 refers to the formation of new capillaries from existing capillaries. The exposure of immortalized rat CEC grown on matrigel to exogenous mBDNF stimulated tube formation, thus supporting a proangiogenic effect of cerebral endothelium-derived BDNF.6,85 In addition, hypoxia, stroke and physical training, which are all associated with angiogenesis,126–128 are also recognized as being able to stimulate BDNF synthesis by CECs (see above).
Endothelial BDNF and cerebral circulation
While exogenous mBDNF was recently reported to induce endothelium-dependent relaxation of precontracted peripheral isolated vessels,88,129,130 only one study investigated the effect of mBDNF on cerebral circulation.
131
In this study, a cisternal injection of replication-incompetent adenovirus encoding rat BDNF was reported to increase acetylcholine-induced relaxation of basilar arteries. This vasomotor effect of BDNF was related to mBDNF-induced prostacyclin synthesis and apparently independent of NO production by endothelial cells. However, exposure of cultured human pulmonary endothelial cells to exogenous mBDNF induced a rapid release of NO.
129
Preliminary and unpublished data from our laboratory showed that exogenous mBDNF induced relaxation of preconstricted rat isolated middle cerebral artery (Figure 5), a resistance vessel of the cerebral circulation. Collectively, these data support the premise that activation of endothelial TrkB receptors by endothelium-derived BDNF induces the release of vasorelaxant factors and, by extension, that cerebral blood flow is dependent on endothelial BDNF/TrkB signaling.
Vasorelaxant effect of BDNF on cerebral circulation. Isolated rat middle cerebral arteries were preconstricted with serotonin (10−6 M) and then exposed to human recombinant mBDNF (10−8 M).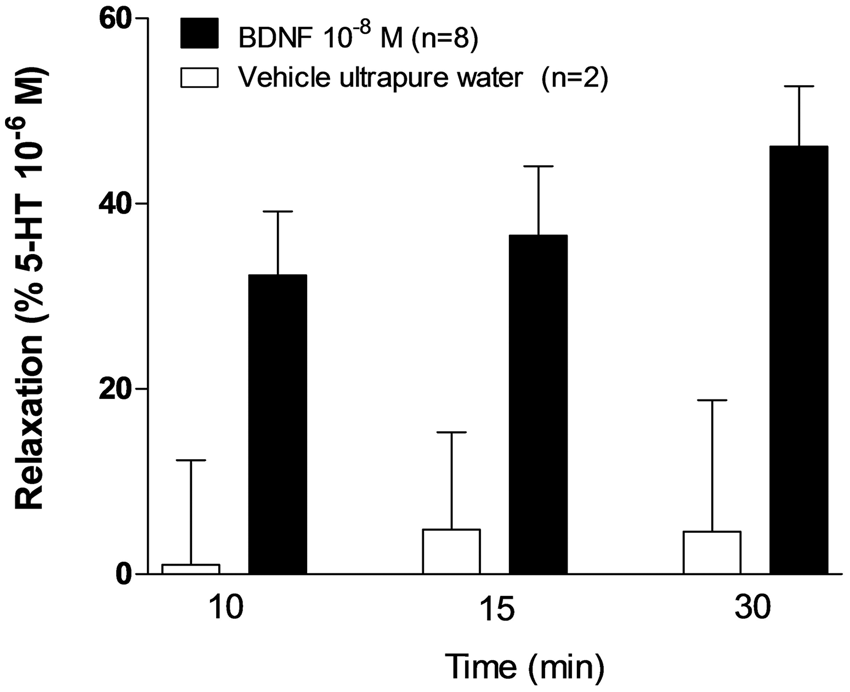
Endothelial BDNF and neuroplasticity
Neuroplasticity, which occurs during development in response to the environment, in support of learning, in response to disease, or in relation to therapy
132
is defined as the modulation of synapse number and strength. Neuroplasticity largely relates to activation of presynaptic and postsynaptic TrkB-FL receptors by mBDNF, which is traditionally thought to be secreted by neurons in an activity-dependent manner. However, synapses are surrounded by astrocytic end-feet
133
and astrocytes also play an important role in the regulation of synaptic behavior.134,135 At the capillary level, astrocytes can contact not only endothelial cells but also synapses (tripartite synapses) (Figure 3). Thus, according to the concept of NGVU, mBDNF derived from endothelial cells might be involved in neuroplasticity according to two distinct scenarios based on the involvement or not of astrocytes (Figure 6). Endothelial mBDNF might bind to neuronal TrkB receptors directly, the distance between capillaries and synapses being compatible with such a scenario in theory. Alternatively, endothelial-derived mBDNF might control neuroplasticity through astrocyte-dependent mechanisms. According to such a mechanism, endothelial-derived mBDNF would be first sequestered by astrocytes through internalization of mBDNF-TrkB-T1 complex and then released back into the extracellular space before binding to neuronal TrkB-FL receptors
136
even though mBDNF binding to astrocytic TrkB-T1 receptors might also stimulate signaling pathways.100,137 Endothelial mBDNF might also interact with neuroplasticity through mechanisms independent on neuronal TrkB receptors. Consistent with this hypothesis, activation of endothelial TrkB receptors by mBDNF would result in sustained NO production,
129
then NO signals originating from endothelial cells would induce long-term potentiation,
138
which is believed to be a neuronal correlate of learning and memory.
139
Lastly, if we hypothesize that CEC also secrete proBDNF, the question arises as to the role of proBDNF in neuroplasticity. Astrocytes express p75NTR,
140
which binds proBDNF with high affinity. In addition, internalization of the proBDNF-p75NTR complex by astrocytes is followed by intracellular maturation of proBDNF into mBDNF and subsequent release as the mature protein.141,142 In these conditions, endothelial proBDNF has to be considered a potential astrocyte-dependent modulator of synaptic behavior. A recent study also suggested that astrocytes play an additional role in the link between endothelial proBDNF and neuroplasticity. This role is related to the ability of astrocytes to regulate the balance between the formation and elimination of plasmin in the brain parenchyma
143
and consequently maturation of BDNF in the extracellular space. The putative scenarios by which mBDNF or proBDNF derived from the cerebral endothelium can affect neuroplasticity is summarized in Figure 6. Demonstrating that synaptic behavior is dependent not only on neuronal-derived BDNF but also on endothelium-derived BDNF will cast doubt on the dogma that neuroplasticity is primarily triggered by neuronal activity.
Putative scenarios by which mBDNF or proBDNF derived from endothelium of cerebral capillaries might induce neuroplasticity. Once released by endothelial cells, mBDNF might activate neuronal TrkB-FL receptors either directly or indirectly via astrocytes. Alternatively, endothelium-derived mBDNF might induce neuroplasticity via nitric oxide (NO). According to this scenario, mBDNF activates endothelial TrkB-FL receptors, thus resulting in NO overproduction and subsequent diffusion of NO into neurons. ProBDNF, if released by endothelial cells might induce neuroplasticity via BDNF recycling by astrocytes or via a plasmin-dependent maturation into mBDNF in the extracellular space.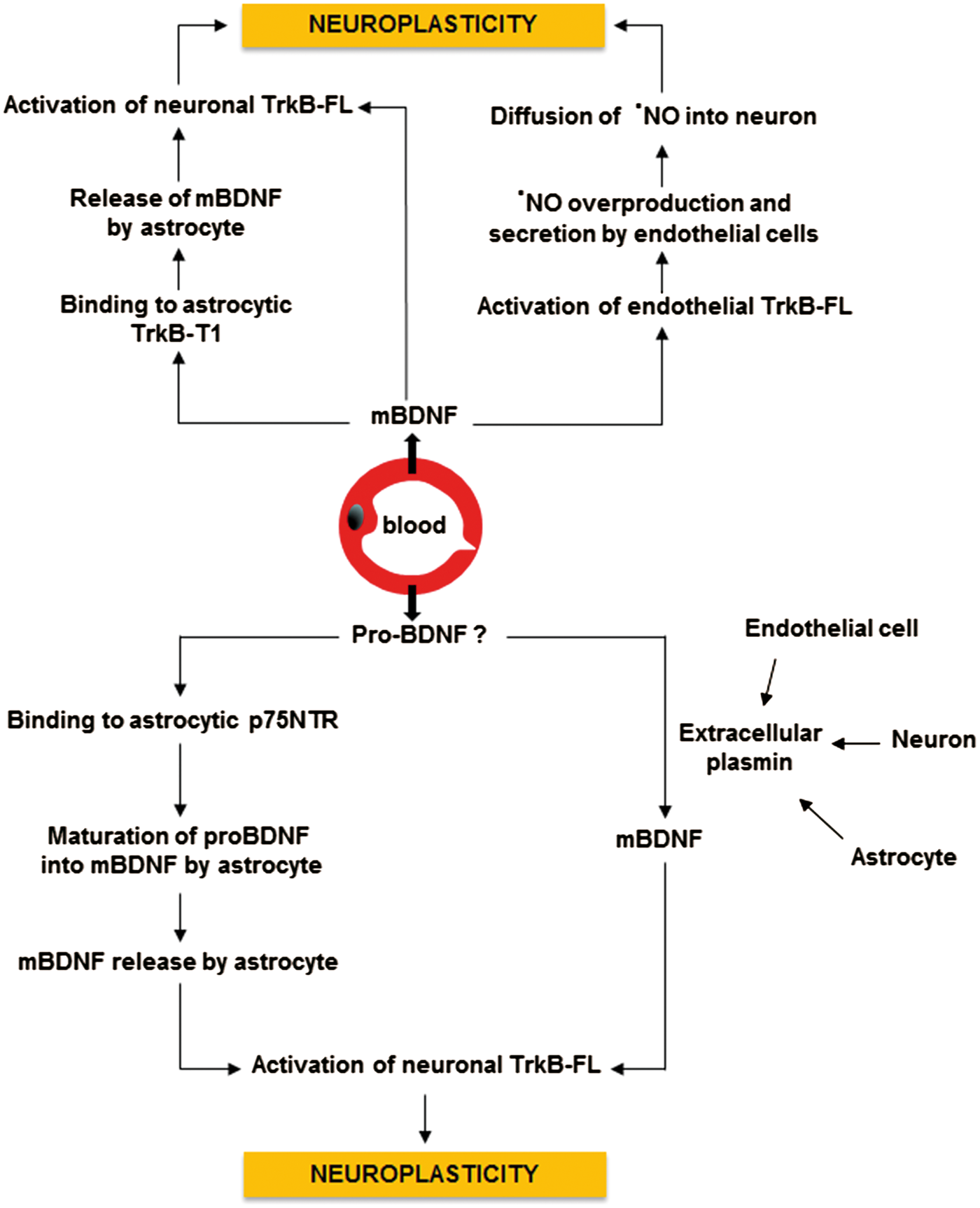
Conclusion
It has been estimated that the human brain contains equal numbers of neurons and non-neuronal cells 144 and that endothelial cells represent 17% of the cell population at least in the rat cortex, with neurons accounting for 47%. 145 Given the density of endothelial cells and their ability to synthesize and secrete large quantities of mBDNF, we must rethink and reinterpret some of the published data on changes in regional brain BDNF levels consecutive to disease or drug administration and consider endothelial mBDNF as an attractive linker connecting brain health with endothelial health. The discovery that mBDNF is secreted by CEC has led to optimism that brain health may be improved by enhancing BDNF signaling using strategies that target CECs rather than neurons. Such an approach would avoid the often insurmountable problem of drug delivery across the blood–brain barrier and potential side-effect of drugs on neurotransmission. However, before this approach can be implemented, it is essential to gain better understanding of the mechanisms involved in endothelial BDNF production and the central effect of mBDNF derived from endothelial cells of the cerebral microvasculature. Several important questions remain to be explored. Are neuronal TrkB receptors a target of BDNF secreted by cerebral capillaries? Does endothelium of the cerebral microvasculature secrete proBDNF? Are TrkB receptors expressed by both the luminal and antiluminal membrane of endothelial cells? Do human and rodent endothelial cells differ in terms of regulation of BDNF secretion and TrkB receptors expression? What are the consequences of the val66met polymorphism on endothelial BDNF synthesis and secretion? Is altered synthesis of endothelial BDNF a pathogenic event in endothelial dysfunction or only an epiphenomena? Establishment of quadruple cell co-culture (neurons, CEC, astrocytes and pericytes) or mouse models with specific endothelial deletion of BDNF or TrkB-FL receptors may be helpful to determine whether BDNF is a new endothelium-derived relaxing factor (EDRF) involved in the control of cerebral hemodynamics and/or a new endothelium-derived enhancer of neuroplasticity (EDEN).
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
