Abstract
Mutations of the β-glucuronidase protein α-Klotho have been associated with premature aging, and altered cognitive function. Although highly expressed in specific areas of the brain, Klotho functions in the central nervous system remain unknown. Here, we show that cultured hippocampal neurons respond to insulin and glutamate stimulation by elevating Klotho protein levels. Conversely, AMPA and NMDA antagonism suppress neuronal Klotho expression. We also provide evidence that soluble Klotho enhances astrocytic aerobic glycolysis by hindering pyruvate metabolism through the mitochondria, and stimulating its processing by lactate dehydrogenase. Pharmacological inhibition of FGFR1, Erk phosphorylation, and monocarboxylic acid transporters prevents Klotho-induced lactate release from astrocytes. Taken together, these data suggest Klotho is a potential new player in the metabolic coupling between neurons and astrocytes. Neuronal glutamatergic activity and insulin modulation elicit Klotho release, which in turn stimulates astrocytic lactate formation and release. Lactate can then be used by neurons and other cells types as a metabolic substrate.
Introduction
Since its serendipitous discovery, Klotho expression has been associated with normal aging, and age-related health declines both in rodents and humans.1,2 In mice, Klotho hypomorphism results in growth retardation and an accelerated aging phenotype characterized by sarcopenia, osteopenia, cognitive dysfunction and shortened lifespan.1,3 Conversely, Klotho overexpression extends rodent life span.
4
In humans, a polymorphism of the
The
Despite its widespread biological effects, Klotho expression pattern is confined to the kidneys and the CNS. 1 In the brain, elevated levels of Klotho can be found in the apical membrane of the ependymal cells of the choroid plexus,10,20 and with lower expression in neurons and oligodendrocytes in the cortical layers, the hippocampal formation, 21 and cerebellar Purkinje cells. 22 The possible physiological functions of Klotho in the brain are just beginning to be elucidated. The experimental evidence gathered so far from rodent models suggests it may exert neuroprotective functions,23–25 as well as regulate neuronal synaptic plasticity and long-term potentiation promoting increased learning and memory.5,26 Hypomorphic Klotho mice display hypomyelination, 27 degeneration of mesencephalic dopaminergic 28 and hippocampal 29 neurons, as well as impaired memory retention. 3 Conversely, mice overexpressing Klotho have enhanced spatial learning and memory, 5 and are partially protected against cognitive decline in transgenic Alzheimer’s disease mouse models. 30 In humans, circulating Klotho levels decrease with age, a decline further exacerbated in the CSF of Alzheimer’s patients. 31 Human polymorphisms that increase the secretion of Klotho have been correlated with greater cortical volumes, 32 network connectivity, 33 and enhanced executive function, visual processing and learning. 5 In older adults, age-associated cognitive decline is inversely correlated with the levels of circulating Klotho. 34
Despite the growing evidence of beneficial influence of secreted Klotho on healthy brain aging, the observations from mice and humans lack a mechanistic explanation. Impaired brain metabolism plays a cardinal role in both normal age-related cognitive decline, as well as in neurodegenerative disorders. 35 Given the role played by Klotho in peripheral metabolism, and the importance of metabolic coupling between astrocytes and neurons,36–38 we hypothesized that secreted Klotho could influence energy metabolism and the dynamic cooperation between these two cell types.
Materials and methods
Animals and primary cell culture
Newborn C57BL/6J mice (postnatal days 0–5) were used for primary cortical astrocyte cell culture. Briefly, cortices from newborn pups were dissected in ice-cold Hanks’ balanced salt solution (HBSS) under a microscope and their meninges removed. Cortices were then cut into small pieces and incubated in a 0.05% trypsin solution (GIBCO) for 20 min in a 37℃, 5% CO2 incubator. Cortical tissue was then transferred to a conical tube containing DMEM (GIBCO) + 10% serum (Hyclone FetalClone III Serum, GE Healthcare), and underwent mechanical trituration with a glass pipette. One million viable cells were plated in T75 flasks and allowed to grow in DMEM + 10% serum until approximately 90% confluent. To obtain astrocyte-pure cell cultures (>98%), flasks were incubated in an orbital shaker at 37℃, 180 r/min, for 15 h.
Primary culture of hippocampal neurons was performed using hippocampi collected from E18 Sprague-Dawley rat or E16 C57BL/6J mouse embryos. Hippocampi were cut into small pieces and incubated in trypsin solution (2 mg/mL) for 20 min in a 37℃, 5% CO2 incubator. After removal of the trypsin solution, the tissue was washed twice with HBSS, incubated in a trypsin inhibitor solution (2 mg/mL) for 1 min, and rinsed with HBSS. Hippocampal tissue was dissociated in HBSS containing 0.1 mg/mL DNAse by mechanical trituration with a glass pipette. Cells were counted and plated (0.1 × 106 cells/cm2) in polyethyleneimine (Sigma-Aldrich) pre-coated dishes. Neurons were maintained for two weeks in Neurobasal medium (GIBCO) supplemented with B27 (GIBCO), 2 mM glutamine, 100 U/mL penicillin, 100 µg/mL streptomycin and 0.25 µg/mL amphotericin B. This research was approved by the National Institute on Aging Animal Care and Use Committee and was performed according to guidelines in the NIH Guide for the Care and Use of Laboratory Animals.
Transfection and FRET imaging
Mouse astrocytes plated on glass bottom dishes (MatTek Corporation) were transfected with the FRET probe for lactic acid Laconic (a gift from Dr. Barros; Adgene plasmid #44238) 39 using Fugene 6. After 24 h, cells were incubated in artificial cerebrospinal fluid (aCSF: 112 mM NaCl, 3 mM KCl, 1.25 mM CaCl2, 1.25 mM MgCl2, 2 mM glucose, 10 mM HEPES, and 24 mM NaHCO3, pH 7.4) and images of mTFP (∼ 480 nm) and Venus (∼ 530 nm) fluorescence were acquired using an LSM 510 confocal microscope. The ratio of mTFP/Venus in presence of 1.0 µM monocarboxilic acid transporter inhibitor AR-C155858 (Tocris Biosciences) and either aCSF or 1.0 nM recombinant mouse Klotho (R&D Systems) was monitored for 10 min to determine the rate of intracellular lactate increase. All FRET ratios (mTFP/Venus) are expressed relative to baseline equal to 1. The initial slope of lactate accumulation was determined by linear regression within the same range of ratio values.
Lactate release measurement
For evaluation of lactate release, astrocytes were plated in 12-well plates (1 × 105 astrocytes/well) and allowed to grow until confluent. Cells were then washed twice with warm HBSS and treated in DMEM without serum. Experiments assessing Klotho-evoked lactate release under different glucose concentrations were performed using high (4.5 g/L) or low (1 g/L) glucose DMEM with or without 1.0 nM recombinant mouse Klotho. Involvement of MCTs, Erk, and FGFR1 was assessed by incubation of cells with 1 µM AR-C155858, 10 µM PD 98059 (Cayman Chemical Company) and 500 nM PD 166866 (Sigma-Aldrich), respectively, for 5–30 min prior and throughout Klotho treatment. After 1 h, supernatant aliquots were collected and 50 µL used for lactate evaluation using a lactate assay kit (Sigma-Aldrich).
Glucose uptake
Glucose uptake was measured using the fluorescent analog 2-(N-(7-Nitrobenz-2-oxa-1,3-diazol-4-yl)Amino)-2-Deoxyglucose (2-NBDG). Briefly 10,000 astrocytes/well were plated in a 96-well black with clear bottom plate. On the day of the assay, the growing media was removed, cells were washed twice with warm HBSS then incubated at 37℃ in glucose deficient DMEM supplemented with 50 µM 2-NBDG and either vehicle or 1 nM Klotho. After 3 h incubation, the cells were washed twice with warm HBSS, and the intracellular content of 2-NBDG measured as fluorescence intensity at 465 nm/540 nm excitation/emission.
Western blotting
Cells treated as indicated were collected at different time points, pelleted and homogenized in RIPA lysis buffer containing protease and phosphatase inhibitors (20 mM Tris-HCl, 150 mM NaCl, 1 mM Na2EDTA, 1 mM EGTA, 1% NP-40, 1% sodium deoxycholate, 2.5 mM sodium pyrophosphate, 1 mM β-glycerophosphate, 1 mM sodium orthovanadate and mini protease inhibitor cocktail). Protein concentration was determined with Bradford’s colorimetric assay. Equal amounts of proteins were separated in 4–12% bis-tris NuPAGE gels (Invitrogen) and transferred to a nitrocellulose membrane, which was then incubated with a 5% bovine serum albumin (BSA) solution in Tris-buffered saline with tween (TBST) (10 mM Tris, pH 8.0, 150 mM NaCl, 0.5% Tween 20) for 2 h. Membranes were incubated in primary antibody solution (TBST, 1% BSA) with antibodies against Akt (1:1000; Cell Signaling #9272), p-Akt Ser473 (1:1000; Cell Signaling # 9271), Erk 1/2 (1:1000; Cell Signaling # 4695), p-Erk (1:1000; Cell Signaling #9106) or actin (1:5000; Sigma A2066) at 4℃ overnight. Membranes were then washed three times with TBST and incubated with secondary antibody solution (TBST, 1% BSA) containing horseradish peroxidase conjugated antibodies anti-rabbit or anti-mouse IgG-Fc (1:4000) for 2 h. After three washes with TBST, membranes were developed using an ECL system (Pierce ECL Western Blotting Substrate, Thermo Scientific).
Oxygen consumption and extracellular acidification rates evaluation
Astrocytic cellular oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) were measured in a Seahorse XF-96 extracellular flux analyzer (Seahorse Biosciences-Agilent). Ten thousand astrocytes/well were plated in a 96-well Seahorse XF96 microplate and the assay was performed 48 h later. Cartridges (Seahorse Biosciences-Agilent) were allowed to hydrate overnight in 200 µl/well of XF Calibrant (Seahorse Biosciences-Agilent), in a non-CO2 incubator at 37℃. At the day of the experiment, cells were washed twice with a non-buffered DMEM pH 7.4 containing 25 mM glucose, 2 mM glutamine, with or without 1 mM pyruvate, as noted. Cells were maintained in this media with or without 1 nM Klotho for 1 h in a non-CO2 incubator at 37℃ prior to evaluation at the Seahorse Analyzer. Respiration was measured in three consecutive cycles of 3 min mixing plus 3 min reading. Following determination of the basal respiration rate, the coupling efficiency was evaluated by injection of 1 µM oligomycin, maximal respiration by addition of 2 µM carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone (FCCP), and non-mitochondrial respiration by injection of 0.5 µM rotenone/antimycin A.
Klotho quantification
Rat hippocampal neuronal cultures were used for experiments assessing intracellular concentrations of total Klotho protein utilizing a Rat KL/Klotho ELISA Kit (LSBio). Cells were harvested at day in vitro (DIV) 5, 10 and 15. Alternatively, at DIV14, 24-h treatments were performed by replacing the media with fresh Neurobasal plus B27 with (4 µg/mL) or without insulin, glutamate (20 µM), or 20 µM α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor antagonist 2,3-dihydroxy-6-nitro-7-sulfamoyl-benzo[f]quinoxaline-2,3-dione (NBQX) and N-methyl-D-aspartate receptor (NMDA) antagonist (2
Gene expression microarray analysis
RNA extraction from control and treated cell cultures was performed using Qiagen RNeasy Mini Kit (Qiagen, Inc., Valencia, CA, USA). The RNA concentration and quality were evaluated by Nanodrop (ThermoFisher, Waltham, MA) and Agilent Bioanalyzer RNA 6000 Chip (Agilent, Santa Clara, CA). Equal amounts of RNA were labeled using Agilent Low-Input QuickAmp Labeling Kit amplification kit, and hybridized overnight to Agilent SurePrint G3 8 × 60 K mouse oligo microarray (G4852A). Following posthybridization rinses, arrays were scanned using an Agilent SureScan microarray Scanner at 3 µm resolution, and hybridization intensity data extracted from the scanned images using Agilent’s Feature Extraction Software. Raw microarray data were first analyzed by scatter plot, principal component and gene sample z-scores-based hierarchy clustering to exclude possible outliers. Microarray data were analyzed using DIANE 6.09, a spreadsheet-based microarray analysis program based on SAM JMP 7.0 system. Genes were determined to be differentially expressed after calculation of
Real-time PCR
RNA was isolated and purified with an RNA Micro Kit (Qiagen, Valencia, CA). Following treatment with DNase I, RNA was quantified and equal amounts were reverse-transcribed using the SuperScript First Strand Synthesis System (Invitrogen Life Technologies). Real-time PCR analysis was performed with CFX96 RealTime System (BioRad, Hercules,CA), and Sybr® Green PCR Master Mix according to the manufacturer’s instructions (Applied Biosystems, Foster City, CA). The comparative ΔΔCt method was used to determine the normalized changes of the target gene relative to a calibrator reference (
Statistical analysis
Statistical analysis was performed by Student’s
Results
Klotho expression in neurons is modulated by glutamatergic and insulin signaling
Characterization of Klotho expression in the brain has shown that it is expressed in neurons, ependymal cells and oligodendrocytes.1,21 To gain a better understanding of the cellular mechanism exerted by Klotho, we first evaluated its expression in mouse hippocampal neuronal cultures at different days in vitro (DIV) (Figure 1(a)). We found that the total cellular content of Klotho was significantly increased with the maturation of the neurons, remaining stable after DIV10 (Figure 1(a)) (DIV5: 3.10 ± 0.18; DIV10: 4.83 ± 0.41; DIV15: 5.82 ± 0.30 pg/ µg protein). Because neuron maturation in vitro is characterized by increased network connectivity and electrical activity,41,42 we tested the impact of glutamate receptor activation on Klotho expression (Figure 1(b)). Neurons at DIV14 were treated for 24 h in complete media with vehicle, a sub toxic concentration of glutamate (20 µM), or a mixture of the AMPA and NMDA receptor antagonists NBQX and APV (20 µM). Compared to vehicle-treated cells, Klotho content in neurons was significantly increased by glutamate receptor stimulation and lowered by glutamate receptor inhibition (Figure 1(b)) (Vehicle: 5.58 ± 0.15; glutamate: 8.48 ± 0.24; NBQX + APV: 3.83 ± 0.16). In kidney, it was shown that insulin can increase Klotho secretion.
7
We wondered if insulin can exert a similar effect in neurons. We incubated for 24 h DIV14 hippocampal cultures in fresh media supplemented with insulin-depleted or normal B27. The results showed that neurons in media with insulin had higher levels of Klotho than those switched to media without insulin (Figure 1(c)) (Insulin [+]: 8.72 ± 0.39; insulin [−]: 5.77 ± 0.26), suggesting a role for insulin in maintaining/stimulating Klotho protein expression.
Klotho expression in primary hippocampal neuronal cultures is modified by maturation status, and by glutamatergic and insulin stimulation. (a) Protein levels of Klotho were assessed by ELISA in primary hippocampal neuronal cultures at the indicated days in vitro (DIV). **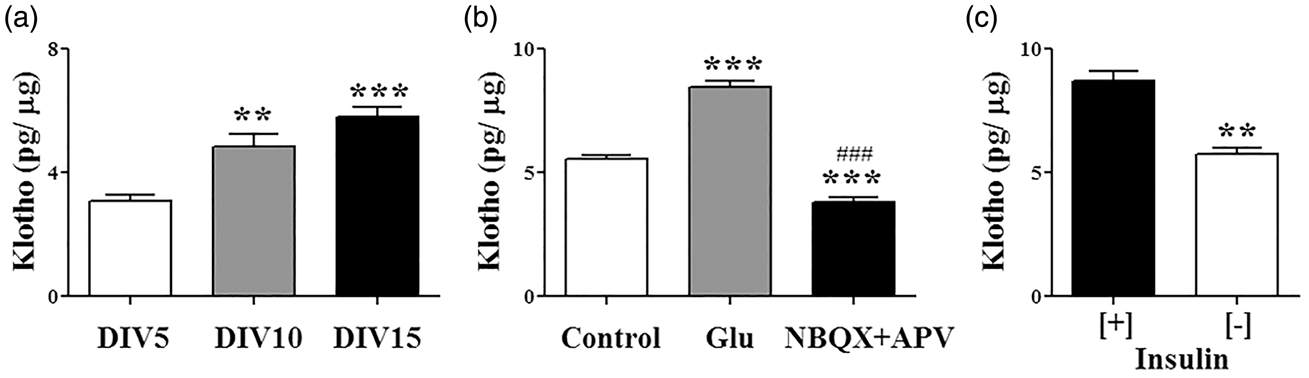
Secreted Klotho modulates astrocyte metabolism
The impact of secreted Klotho on oligodendrocyte physiology has been recently reported.
27
It is however unclear if secreted Klotho plays any role in neuronal and astrocytic functions. We analyzed the expression profile of 14 DIV mouse neurons and astrocytes treated for 24 h with 1 nM recombinant secreted Klotho. We found a greater response to secreted Klotho treatment in astrocytes compared to neurons. Compared to vehicle-treated cells, 96 genes were significantly changed by secreted Klotho treatment in astrocytes (Table S2), while only 17 genes were modified in neurons (Table S3). To more systematically determine the metabolic and signaling pathways elicited by secreted Klotho in neurons and astrocytes, we used the ingenuity pathway analysis, focusing on KEGG classification. We observed 43 enriched pathways in astrocytes (Table S4), but only 19 in neurons (Table S5). Consistent with a predominant role for astrocytes in energy metabolism and amino acid synthesis, the most upregulated pathways were carbohydrate, glycan, lipid and amino acid metabolism (Table S4). Conversely, the greatest inhibitory effect of Klotho treatment was of immune-related signaling pathways (Table S4), substantiating previous results supporting an anti-inflammatory role for Klotho.43,44 In neurons, Klotho treatment decreased lipid metabolism and enhanced proteasomal degradation (Table S5). We decided to focus on the effect of Klotho on the astrocyte transcriptome, and selected several genes that were up- or down-regulated for validation, and included a non-modified gene as an internal control. The analysis was performed on the RNA used for the array hybridization, as well as RNA from additional independent experiments. All the genes selected for real-time PCR displayed the same expression trend seen in the gene array analysis (Figure 2). Amongst the most upregulated genes were C-X-C chemokine receptor type 4 ( Real-time PCR analysis of a subset of the array genes modified by Klotho treatment in astrocytes. We verified the gene array changes performing real-time PCR on the indicated genes selected amongst those were modified by soluble Klotho treatment in astrocytes. The relative changes in RNA transcripts were identical to those found in the array analysis. *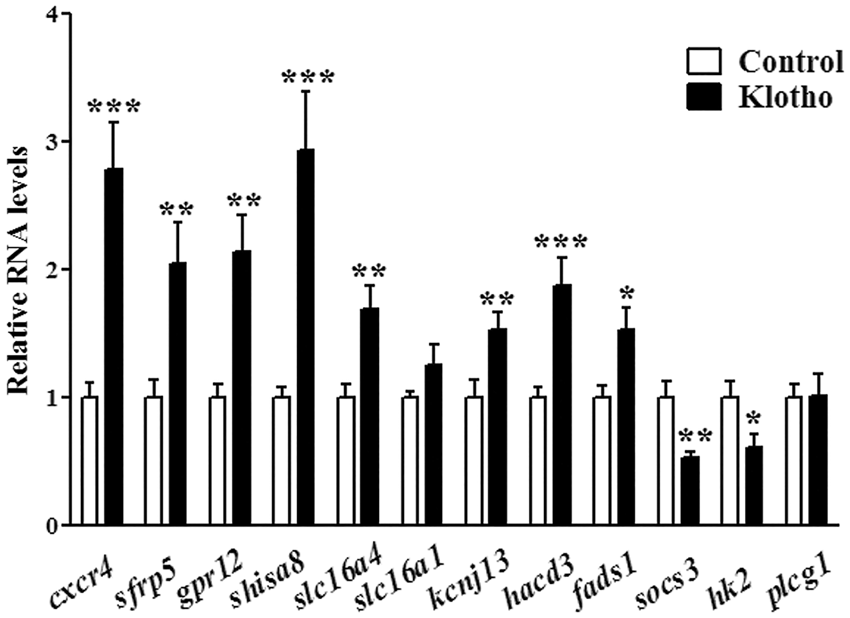
Klotho stimulates a rapid increase of astrocytic aerobic glycolysis and lactate release
Astrocytes importance in brain metabolism and the striking effects of Klotho on their transcriptome prompt us to investigate how Klotho impacts astrocytes’ cellular respiration. Astrocytes’ oxygen consumption and extracellular acidification rates were measured using a Seahorse XF-96 flux analyzer. Exposure of astrocytes to secreted Klotho for 1 h was sufficient to cause a significant increase in the rate of extracellular acidification under basal conditions (Figure 3(d) and (f)) (Control: 0.97 ± 0.05; Klotho: 1.97 ± 0.04), and a decrease in maximal respiratory capacity (Figure 3(a) and (c)) (Control: 1.84 ± 0.08; Klotho: 0.77 ± 0.02). When the same experiments were performed in the absence of pyruvate, a significant decrease in the maximal respiratory capacity was seen in control-treated cells but not in Klotho-treated astrocytes (Figure 3(a) to (c)) (Control no pyruvate: 1.38 ± 0.04; Klotho no pyruvate: 0.67 ± 0.03). Klotho-induced extracellular acidification persisted in the absence of pyruvate (Figure 3(e) and (f)). Overall these results suggest that under Klotho stimulation only a limited portion of the pyruvate is used to generate acetyl-CoA and ATP via oxidative phosphorylation, allowing a higher production of lactic acid. In neurons, consistent with the modest changes observed in gene expression, we did not observe any effect of Klotho on cellular respiration (Figure 3(g)).
Klotho enhances astrocyte aerobic respiration. Cellular respiration was measured in astrocytes treated with vehicle (Control) or 1 nM Klotho for 1 h in presence (a) or absence (b) of extracellular pyruvate. Klotho treatment caused a significant decrease of the maximal oxygen consumption rate (OCR) independently of the presence of pyruvate (c). The extracellular acidification rate (ECAR) was significantly increased by Klotho treatment regardless of the presence of pyruvate in the media (d–f), indicating a shift toward aerobic glycolysis. (g) Klotho treatment had no effect on cellular respiration in hippocampal neurons. O: oligomycin; FCCP: carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone; R + A: rotenone plus antimycin A. Data are mean and SEM of six independent experiments. ***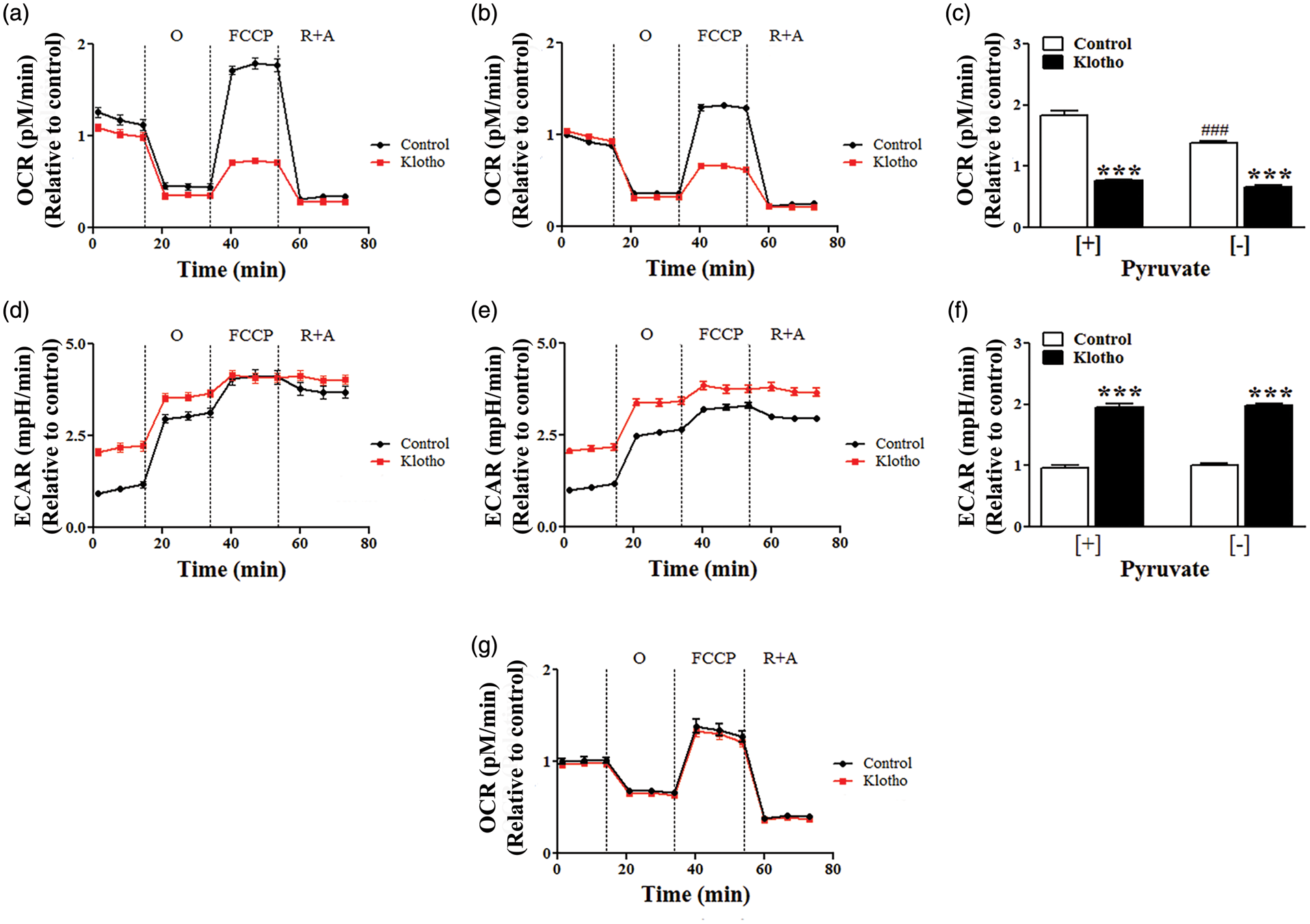
The astrocyte-neuron lactate shuttle hypothesis suggests that neurons uptake lactate released by astrocytes, and utilize the lactate as an energy substrate.
36
This mechanism is particularly important during high energy demanding tasks such as action potential firing.
36
We therefore determined whether Klotho affects lactate production in astrocytes. Astrocytes transfected with the lactate FRET probe Laconic
39
were treated with the monocarboxylate transport inhibitor AR-C155858 to prevent export, and the accumulation of intracellular lactate was evaluated in astrocytes exposed to vehicle or Klotho. As suggested by the Seahorse results, the rates of lactate production were significantly accelerated by the addition of recombinant Klotho (Figure 4(a)). We next investigated if changes in glucose uptake were required for the observed increase in lactate production using the glucose fluorescent analog 2-( Klotho effects on glucose metabolism in astrocytes. Astrocytes transfected with the lactate sensor Laconic were incubated in artificial cerebrospinal fluid and the increase in intracellular lactate monitored after addition of the MCTs inhibitor AR-C155885 plus vehicle (Control) (PBS) or recombinant Klotho. (a) Representative images of mTFP intensity at baseline (0′) and after 10 min (10′) of recording, as well as the relative rate of FRET changes (Δ mTFP/Venus per minute) and time lapse lactate accumulation are shown. The straight lines represent slopes of lactate accumulation fitted by linear regression during the exposure. Scale bar is 20 µm. Data are the mean and SEM of 14–19 transfected astrocytes from three separate cultures. (b) Astrocytes were treated with vehicle (Control) or 1 nM secreted Klotho in glucose deficient media containing 50 µM 2-NBDG. The cellular uptake of 2-NBDG measured after 3 h showed no difference between the two treatments. (c) Treatment of astrocytes with Klotho-induced lactate release in the culture media, independently of the extracellular glucose concentrations (low: 1 g/L; High: 4.5 g/L). (d) Inhibition of MCTs with 1µM AR-C155858 completely abolished Klotho-induced lactate release. ** 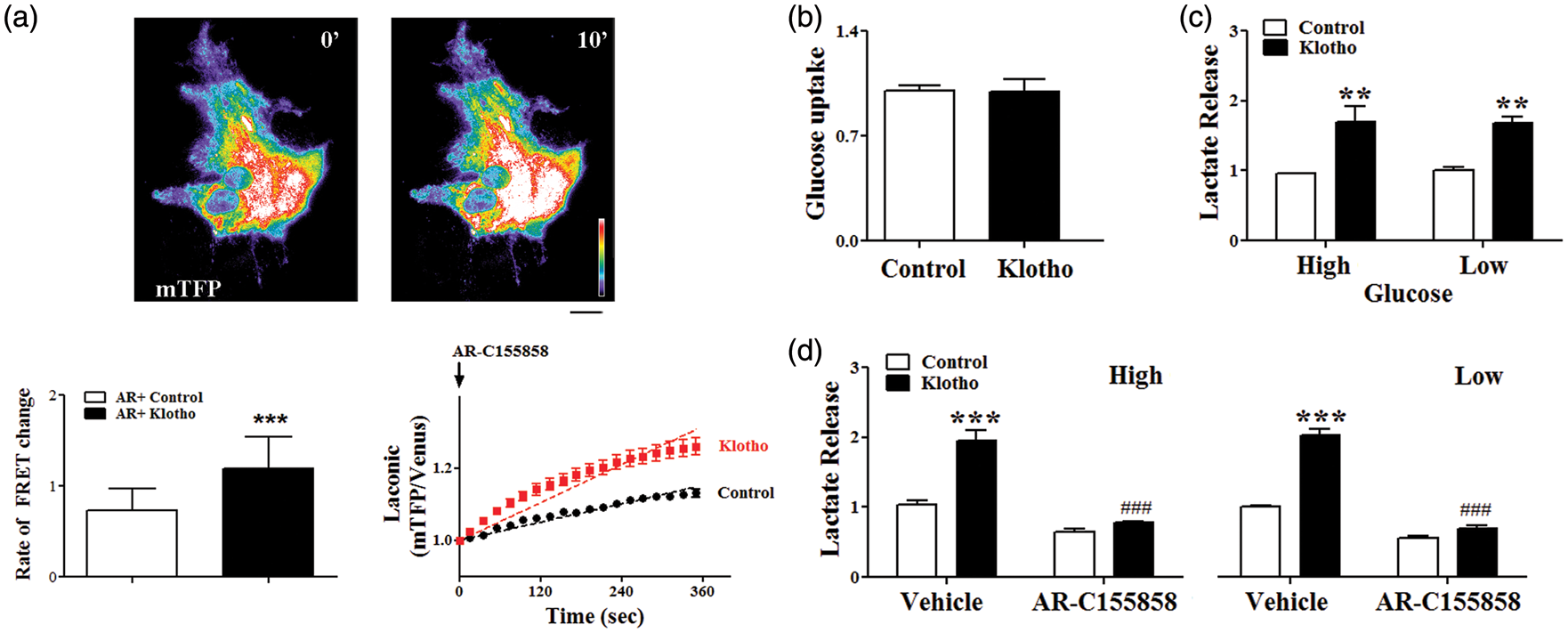
FGFR1 and Erk activation are required for Klotho-induced lactate release
Klotho has been shown to interact with several fibroblast growth factor receptors (FGFR) in various cell types.
51
Signaling through FGFR1 has been linked to increased aerobic glycolysis in cancer cells.
52
Notably, the trophic effect exerted by astrocytes on neurons is impaired in FGFR1 mutant astrocytes both in vitro and in vivo.
53
We analyzed the impact of secreted Klotho on two FGFR signaling pathways known to positively modulate glycolysis, the PI3K/Akt pathway
54
and the MEKK/MAPK/Erk1/2 pathway.
55
Treatment of astrocytes with secreted Klotho resulted in time-dependent decrease of Akt serine phosphorylation (Figure 5(a)), and an increase of threonine/tyrosine phosphorylation of extracellular signal-regulated kinase 1/2 (Erk1/2) (Figure 5(b)). Co-treatment with the MEK1 inhibitor PD98059 efficiently prevented Klotho-dependent Erk1/2 phosphorylation (Figure 5(c)), as well as lactate release into the media (Figure 5(d)) (Control: 1.00 ± 0.01; Klotho: 2.01 ± 0.08; Klotho +PD98059: 0.89 ± 0.02). Antagonism of FGFR1 with PD166866 was also able to significantly decrease Klotho-induced Erk1/2 phosphorylation (Figure 5(e)) and lactate release (Figure 5(f)) (Control: 1.00 ± 0.05; Klotho; 1.85 ± 0.10; Klotho + PD166866: 1.23 ± 0.07). These results indicate that in astrocytes, secreted Klotho agonism of FGFR1 results in activation of the MEKK/MAPK/Erk1/2 pathway leading to increased aerobic glycolysis and lactate release.
Klotho-induced lactate release is mediated by FGFR1 and Erk signaling pathways. Astrocytes were treated with vehicle (Control) or secreted Klotho for the indicated periods of time and phosphorylation of Akt (Ser 473) (a) and Erk1 (Thr202/Tyr204) and Erk2 (Thr185/Tyr187) (b) assessed by western blotting. **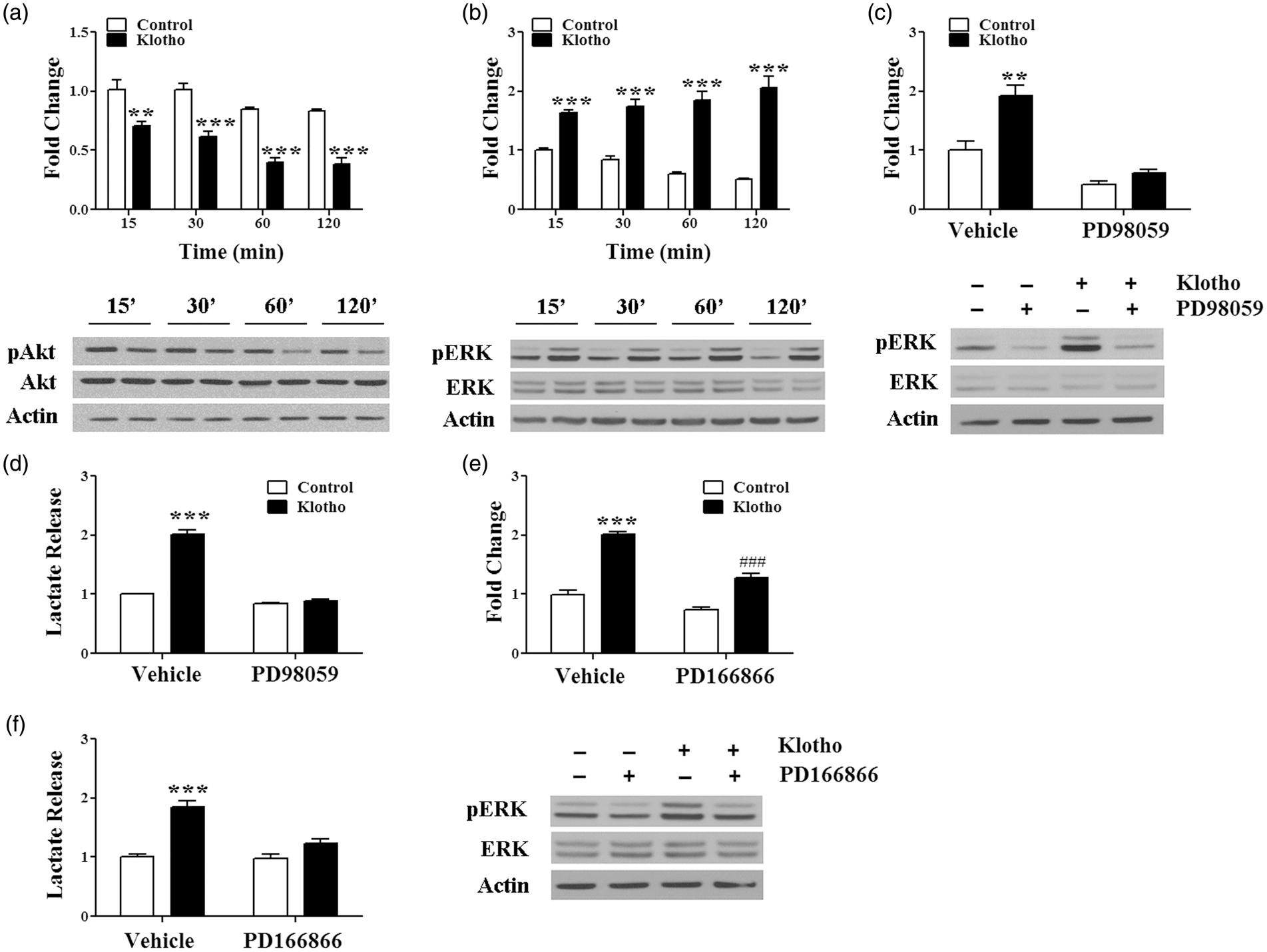
Discussion
In the present study, we provide evidence that Klotho is produced by neurons in response to insulin, as well as glutamatergic stimulation. We also show that secreted Klotho influences astrocytic metabolism stimulating aerobic glycolysis and lactate release by a mechanism requiring FGFR1 and Erk1/2 activation. The ability of Klotho to modulate cellular energy metabolism via FGFR signaling in astrocytes is consistent with results in other cell types.
51
In cancer cells, FGFR1 activation decreases mitochondrial pyruvate utilization and promotes aerobic glycolysis via tyrosine phosphorylation of lactate dehydrogenase A,
56
pyruvate kinase isoform M2,
57
and pyruvate dehydrogenase kinase 1 (PDHK1).
52
Similarly, the MAPK/Erk pathway has been shown to stimulate the synthesis of lactate in activated macrophages.
58
Also, sustained aerobic glycolysis due to ADAM17-dependent growth factor shedding and constitutive MAPK/Erk pathway activation have been reported in polycystic kidney disease.
59
With respect to the brain, neuronal activity can stimulate ADAM10 and ADAM17 activities, facilitating the ectodomain shedding of various transmembrane proteins such as nectin-1,
60
signal regulatory protein a,
61
and neuregulin-1 type II precursor.
62
In neurons, transmembrane Klotho is expressed both at the pre- and post-synaptic compartments.
26
Given the role played by ADAM10 and ADAM17 in cleavage of Klotho in other tissues,
7
it is possible that the activity-dependent increase in Klotho levels in neurons we observed is mediated by both enhanced synthesis and shedding. It is well established that neurons and astrocytes are functionally coupled. For example, astrocytes play a role in neuronal excitability controlling the extracellular concentration of potassium and removing neurotransmitters such as glutamate and gamma-aminobutyric acid from the synaptic cleft.
63
Astrocytes bridge the vasculature and neurons by removing essential nutrients from the blood stream and transferring them to neurons.
35
Lactate produced by astrocytes has been shown to be an important metabolic substrate for neurons.
36
The recognition of the importance of the astrocyte-neuron lactate shuttle (ANLS) in brain metabolism under physiological and pathological conditions has steadily increased over the past decades.64,65 For instance, lactate transport through MCTs at excitatory synapses constitutively supports synaptic transmission
66
and is required for the establishment of long-term memory formation.
38
While the ANLS hypothesis assumes that lactate is exclusively used for energy metabolism by replacing glucose as substrate, it should be noted that other neuronal metabolic needs can be fulfilled by lactate, either directly or by “sparing” glucose.
67
For example, the pyruvate generated from lactate can be used by neurons to indirectly support the tricarboxylic cycle by coupling its transamination to alanine with the conversion of glutamate to α-ketoglutarate, the rate limiting substrate in the cycle (Figure 6). Alternatively, neurons can divert part of the generated pyruvate toward the synthesis of lipids necessary to maintain or create new synaptic connections (Figure 6). By engaging the recently discovered hydroxycarboxylic acid receptor-1, lactate may act as a signaling molecule rather than a metabolite.
68
Hypomorphic Klotho mice display myelin abnormalities and dysregulation of oligodendrocytes maturation and functions.
27
Satellite oligodendrocytes are closely associated with neurons and astrocytes,
69
and avidly uptake lactate for energy purposes and lipid synthesis.
70
It is thus possible that neuronal produced Klotho acts on astrocytes to stimulate aerobic glycolysis and lactate release, and on oligodendrocytes to stimulate lipid synthesis via lactate uptake (Figure 6). Our findings suggest Klotho may play a key role in coordinating the metabolic and functional interplays among various cell types in the brain. Furthermore, they provide insight into the recent observation that neuronal activity modifies astrocytes gene expression to up-regulate their metabolism and promote lactate export.
71
Given their high energy and metabolic demands, it is reasonable that neurons would be able to rapidly modify astrocyte metabolism in order to obtain substrates for their functional needs. Klotho could be the signaling molecule utilized by neurons to drive such metabolic changes (Figure 6). Interestingly, the trophic effect exerted by astrocytes on neurons is impaired in FGFR1 mutant astrocytes both in vitro and in vivo.
53
The finding that, similarly to what occurs in hepatocytes,
7
neuronal Klotho production is increased in presence of insulin might explain, in part, how insulin impacts brain energy metabolism. It is well established that insulin receptors are widely expressed in the brain, and that insulin crosses the blood–brain barrier and is also produced in brain cells.
72
The higher expression of insulin receptors in neurons compared to glia
73
is likely the reason why insulin functions in the brain are primarily neurotrophic.
74
Impaired insulin receptor signaling has been shown to decrease experience-dependent plasticity,
75
and insulin receptor substrate 2 knock out mice display impaired LTP.
76
While insulin does not modify glucose uptake in neurons nor astrocytes,
77
a rapid increase in local glycolysis has been observed in hippocampus after direct intracerebral insulin administration.
78
It is possible that in response to insulin signaling, neurons produce more Klotho that can then stimulate local astrocytic glycolysis (Figure 6). Aerobic glycolysis in the human brain has been shown to be particularly higher in the areas associated with the default mode network, and with the dorsolateral prefrontal and parietal cortex associated with task control processing.
79
Notably, the areas showing high aerobic glycolysis are those with higher numbers of non-neuronal cells, and are nearly identical with those affected by amyloid deposition and altered metabolism in Alzheimer’s disease.
80
Glucose hypometabolism is a salient feature of normal aging and a preclinical finding in many neurodegenerative disorders.
35
In humans, activity-dependent astrocytic ANLS genes have been shown to decline with age.
71
Klotho levels in the central nervous system are also lower in older versus younger adults, and are further decreased in Alzheimer’s disease patients versus age-matched healthy individuals.
31
In view of our findings, it is tempting to speculate that changes in the ability of neurons to produce sufficient Klotho can be involved in altered age- and/or pathology-dependent brain metabolism. Reduced Klotho production would result in inability of astrocytes to rapidly modify their metabolic flux, and increase their lactate release to support the metabolic/functional needs of adjacent active neurons and oligodendrocytes (Figure 6).
Neuronal Klotho stimulates astrocytic aerobic glycolysis and lactate release. Klotho expression in neurons is positively regulated by glutamatergic neurotransmission and insulin stimulation. Neuronal secreted Klotho (sKlotho) causes a rapid upregulation of astrocytic aerobic glycolysis and lactate release through a mechanism requiring fibroblast growth factor receptor 1 (FGFR1) and extracellular signal-regulated kinase 1/2 (Erk1/2) activation. The lactate released by astrocytes in the extracellular space can be directly utilized by neurons to support their energy requirement during times of high activity. Alternatively lactate can indirectly support neuronal oxidative phosphorylation by coupling the transamination of pyruvate to alanine (Ala) with the conversion of glutamate to α-ketoglutarate (αKG). Furthermore, lactate can be used as a substrate for lipid synthesis contributing to the establishment and maintenance of the dendritic arbor and to synaptic plasticity. Satellite oligodendrocytes are closely associated with neurons and astrocytes and can also utilize the released lactate for energy purposes, as well as lipid synthesis. During aging or pathological conditions such as Alzheimer’s disease (AD), reduced production of sKlotho will impair the neuron-astrocyte coupling resulting in insufficient lactate generation and neuronal dysfunction. Abbreviations are as follow: ADAM: a disintegrin and metalloproteinase domain containing protein; ECT: electron transport chain; F6P: fructose-6-phosphate; F1,6P: fructose-1,6-dyphosphate; G6P: glucose-6-phosphate; G3P: glyceraldehyde-3-phosphate; GLUT1: glucose transporter 1; HCAR1: hydroxycarboxylic acid receptor 1; IR: insulin receptor; LDH: lactate dehydrogenase; MCT: monocarboxylic acid transporter; PEP: phosphoenolpyruvate; PKF1: phosphofructokinase 1; PKM2: pyruvate kinase M2; TCA: tricarboxylic acid.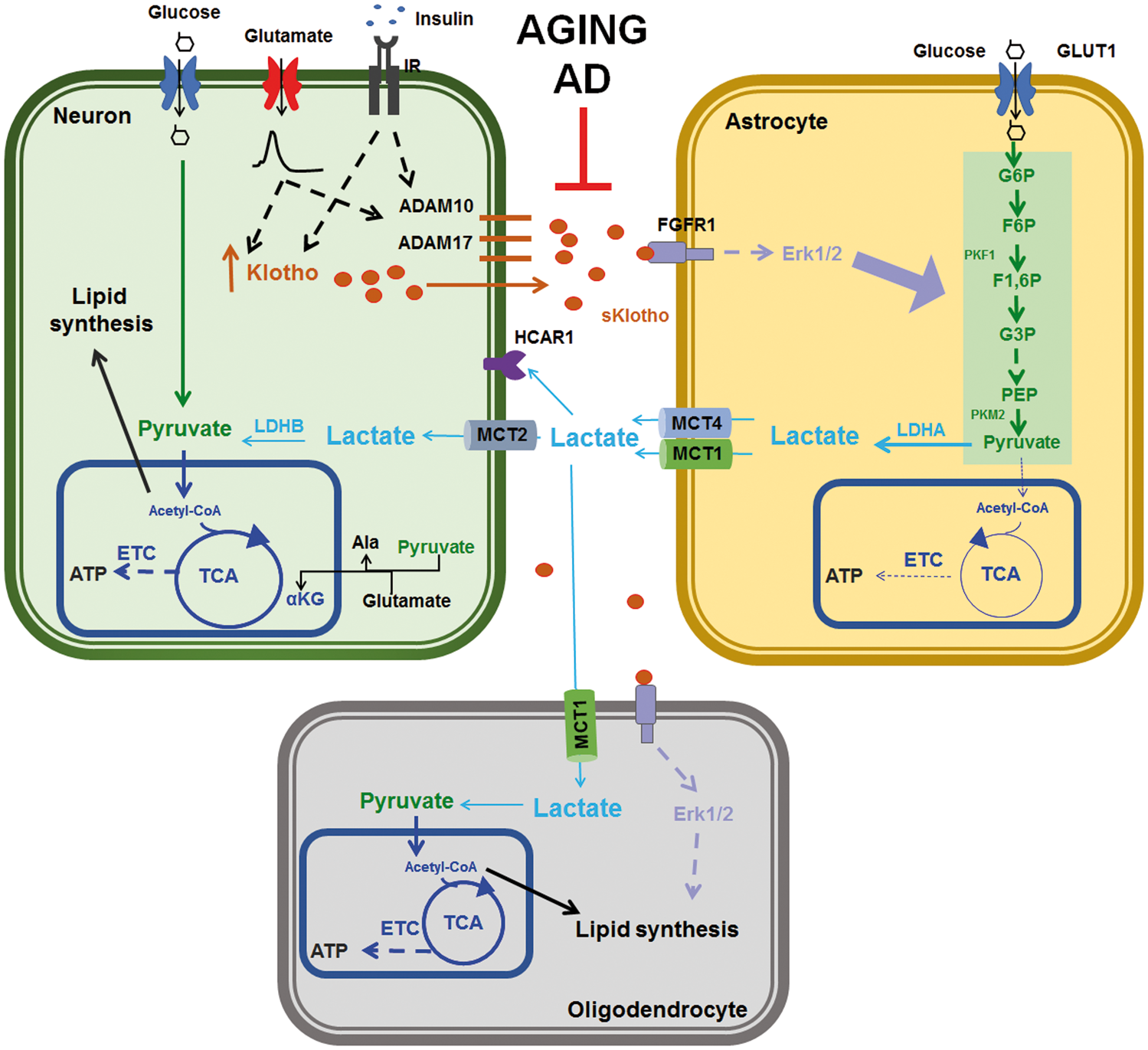
In summary, our findings suggest that Klotho plays a significant role in brain metabolism, modulating neuron-astrocyte coupling and substrate availability to neurons.
Supplemental Material
Supplemental material for Activity-dependent neuronal Klotho enhances astrocytic aerobic glycolysis
Supplemental material for Activity-dependent neuronal Klotho enhances astrocytic aerobic glycolysis by Caio H Mazucanti, Elisa M Kawamoto, Mark P Mattson, Cristoforo Scavone and Simonetta Camandola in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Intramural Research Program of the National Institute on Aging. CHM was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP 2016/07427-8; 2016/09971-7). CS is a research fellow from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Acknowledgments
We would like to thank the Gene Expression & Genomic Unit directed by Dr. Kevin Becker, Drs. Elin Lehrmann and Yongqing Zhang for microarray assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
CHM and SC performed experiments, analyzed the data and wrote the manuscript. EMK, MPM, and CS contributed to the writing of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
