Abstract
Cerebral microbleeds (CMBs) could contribute to cognitive impairment in the general population and in patients with dementia. We designed a study to (i) develop a murine model of CMBs, (ii) assess whether CMBs affect cognition in this model and (iii) assess whether this model is sensitive to pharmacological modulation. Male C57Bl6/J mice were stereotactically administered collagenase to induce cortical lesion analysed by MRI at 24 h. CMB-mice were assessed at six weeks post-lesion for cognitive performances (Barnes maze and Touchscreen automated paired-associated learning (PAL) task) and for cerebral metabolism (in vivo PET/CT with fluorodeoxyglucose (FDG)). CMB-model sensitivity to pharmacological modulation was assessed by administering atorvastatin (5 mg/kg/day) over the follow-up period. CMB mice were compared to naïve littermates. Collagenase at 0.8 µU/µl appeared suitable to induce reproducible and reliable CMBs. At six weeks, a decline in learning, spatial and visuospatial memory was significantly observed in CMB-mice. Brain metabolism was impaired in all cortex, striatum and the ipsilateral dentate gyrus. A significant improvement in cognition performances was depicted under atorvastatin. In this novel murine model of CMBs, we validated that CMBs lowered cognitive performances and affected regional metabolism. We also proved that this CMB-model is sensitive to pharmacological modulation.
Introduction
Cerebral microbleeds (CMBs) are punctiform hemosiderin deposits surrounding cerebral small vessels, easily visualized via gradient-echo T2*-weighted magnetic resonance imaging (MRI). CMBs have been reported in 5% of the general population, 1 35%–85% of patients with vascular dementia, 2 and 16%–32% of patients with Alzheimer’s disease (AD). 1 Although their clinical significance remains unclear, it has been suggested that CMBs are a sign of vascular damage and amyloid pathologies such as lipohyalinosis (particularly induced by hypertension) and cerebral amyloid angiopathy (CAA). 3
The pathological processes that underlie microbleeds vary from one brain location to another. CAA is the most notable correlate of exclusively lobar microbleeds (mostly in the occipital and posterior temporoparietal regions), with amyloid deposition on vessel walls adjacent to the microbleeds. 4 In contrast, the “hypertensive arteriopathy” affecting deep perforating vessels (e.g. lipohyalinosis) is predominantly associated with deep microbleeds. 4
Lately, CMBs have been thought of as a contributing factor for cognitive impairment as a high CMBs count has been associated with an elevated risk of cognitive deterioration and dementia in the general population. 5 In patients with vascular dementia or vascular cognitive impairment, a large number of CMBs has been linked to a significantly worse performance in cognitive tests of psychomotor speed and executive function. 2 In AD patients, a great CMBs burden was reported to be associated with more severe cognitive impairment and lower levels of Aβ 42 in the cerebrospinal fluid. 6 In a cohort of elder adults followed over three years, the annual incidence of lobar CMBs was greater in subjects with AD (especially those with important amyloid burden) than in healthy subjects. 7 Nevertheless, other clinical studies have failed to evidence an impact of CMBs on cognition or on the rate of cognitive deterioration in AD. 8 In healthy subjects, the CMBs impact on cognition still remains debatable. 9
Furthermore, it is not clear whether CMBs may be affected by concomitant medication – especially treatments with antithrombotic, lipid-lowering or antihypertensive medications – mainly because of the heterogeneous pathophysiological mechanisms involved. 10 Although many studies of lipid-lowering agents (such as statins) effects on cognition have yielded contradictory results, the pleiotropic biological effects of statins may be of interest to affect CMBs and subsequently cognition. 11
In view of these sometimes contradictory clinical findings, the development of an animal model of cortical CMBs may be of value. 12 The model must be reliable and reproducible for studying the impact and the physiopathological mechanisms of cortical CMBs on cognition independently of the pathophysiological process underlying CMB occurrence, as well as the putative effects of pharmacological modulation. Consequently, our study was designed to (i) develop a murine model of surgically reproducible collagenase-induced cortical CMB, (ii) assess whether CMB affect cognition in this model and (iii) assess whether this model is sensitive to pharmacological modulation by statin.
Materials and methods
Animals
Adult male C57Bl6/J mice (Elevage Janvier, Le Genest Saint Isle, France; weight range 28–32 g) were used to develop and validate the model. The animals were housed in transparent cages (five or less per cage) with nesting material and maintained in a climate-controlled room (temperature 19–24℃; relative humidity: 45–65%) with a 12-h low/dark cycle (lights on at 7.00 am), and food and water ad libitum.
Surgical procedures
Cortical CMB was induced by a stereotaxic injection of the proteolytic enzyme type VII bacterial collagenase (COL C2399, Sigma-Aldrich, St. Louis, MO, USA); 10- to 12-weeks-old mice (25–30 g) were anesthetized with isoflurane (induction: 3%; maintenance: 2–2.5%) and body temperature was kept constant at 36 to 36.5℃ via a heating blanket connected to an intrarectal thermometer. Once positioned in a stereotaxic frame (Knopf Instruments, Tujunga, CA, USA), a midline incision was made to retract the scalp. A 1 mm-hole was drilled through the skull at 1 mm (posterior) and −2 mm (lateral) from bregma. A single volume of collagenase or saline was injected 1 mm below the surface of the frontoparietal cortex using a Hamilton syringe (Reno, NV, USA) placed in a micro-syringe pump (Micro 4® Microsyringe Pump Controller – World precisions instruments, Sarusota, Florida). One microliter of a range of collagenase doses (0.2, 0.8 and 2 µU/µl for the COL0.2, COL0.8 and COL2 groups, respectively) or saline (for the sham group) was injected over a period of 5 min. To minimize pathway reflux and bleeding during needle removal, the needle was left in place for 5 min after the end of the injection and then slowly withdrawn at a rate of 0.5 mm/min. Once the scalp was sutured, the mouse was placed under a red warm light to recover.
In vivo imaging
During the MRI and PET imaging, mice were anesthetized with isoflurane, placed on a heating blanket, and monitored for the respiratory rate. MRI and PET imaging were assessed by specialists of small animal imaging (F.A. and L.R.), who were blinded to the mice groups.
MRI
Twenty-four hours after surgery, all mice underwent small animal MR scanner (7-Tesla; BioSpec 70/20, Bruker, Ettlingen, Germany) with a T2 sequence to visualize edema (TR/TE: 5000/33 ms; field of view (FOV): 2 cm), and a T2* sequence to visualize bleeding (TR/TE; 1500/24.5 ms; FOV: 2 cm). Image processing was performed on 0.5 mm-thick-coronal slices to characterize acute-phase lesions: (i) the presence or absence of CMB, (ii) the volume of the lesion, and (iii) the ipsilateral/contralateral relaxation time ratio. Given the very little volumes, each lesion was studied as a single sphere, which volume was calculated as 4/3 × (D/2) × π, where D was the measured diameter. To estimate the relaxation time ratio, we defined a region of interest (ROI) in the visible lesion and then duplicated it onto the contralateral cortex. The relaxation time in the T2* sequence was noted for each ROI (ipsilateral and contralateral) and the ratio was calculated.
Fluorodeoxyglucose – Positron emission tomography
Six weeks after surgery, the cerebral metabolism was studied by performing a microPET scan (Inveon, Siemens) with intraperitoneal injection of fluorinated analog of the glucose, the fluorodeoxyglucose ([18F]FDG) (13±5 MBq; 250–500 µl in volume, IBA-CisBio, Saclay, France). A 15-min PET scan was initiated 60 min after radiotracer injection.
To assess changes in metabolism, ROIs were determined by using a template derived from the MR images. Data from the scanner were formatted into three frames of 300 s, OSEM2D reconstructed and corrected for scatter and attenuation. Counts detected by the scanner were converted into percentage of injected dose/g by use of IRW software (Inveon Research Workflow, version 3.0, Siemens). This software enables PET-CT coregistration and allows imaging realignment with T2-weighted brain MRI where ROIs were accurately hand-drawn and then transferred to PET images. The following areas (A) were selected to be studied as volumes of interest (VoIs): pons nucleus (PN); septal nucleus (SN); striatum (St); Cortex (Cx); denta gyrus (DG); Hippocampus (Hi); Hypothalamus (Ht); Inferior culliculus (IC); Midbrain (MB); Striatum (St); Superior culliculus (SC); Thalamus (Th). A background ROI was also drawn outside the brain region. The apparent activity in this background ROI reflected the contributions from random coincidence events and from scatter and was found to be very small throughout the course of the studies. To avoid bias due to inter-individual differences in the uptake of [18F]FDG, the uptake in each ROI was calculated relative to the uptake in a reference zone (the cerebellum).
Histochemistry
After intraperitoneally injection with a lethal dose of pentobarbital, mice were perfused transcardially with saline. To visualize the collagenase-induced hemorrhage on fresh tissue, the collected brains were left in isopentane (−20℃) for 20 min and then coronal sections were performed using a cryostat. For the hematoxylin eosin (H&E) staining, the collected brains were fixed in methacarn for 24 h, dehydrated, embedded in paraffin and cut into 5 -µm thick slices mounted on glass slides. 13
Behavioral assessment
Six weeks after surgery, three behavioral tests were administered in the following order: the open field test, the elevated plus maze (EPM), the Barnes maze with an interval of at least one day between the test. Other groups of mice underwent concomitantly the touchscreen automated paired-associates learning (PAL) task because of the duration of the test. The mice were tested blinded to treatment between 9.00 a.m. and 7.00 p.m. For the open field test, the EPM and the Barnes maze, the equipment was cleaned with 70% ethanol after each trial so that olfactory cues did not bias the results. For the PAL task, the equipment was cleaned with distilled water.
The open field test
Spontaneous locomotor activity was assessed in an open field test, using an infrared actimeter (Bioseb, USA). The apparatus consisted of a square arena (45 × 45 cm) with a black polymethyl methacrylate floor and transparent 34-cm high polymethyl methacrylate walls. Mice were placed in the center of the arena and allowed to explore freely for 10 min. Activity was recorded by two rows of infrared photocell sensors and processed with Actitack software (Bioseb). The total distance covered (in cm) and the average speed (in cm/s) were calculated.
The EPM
The EPM was used to assess the mice’s anxiety behavior. The equipment consisted of four arms (36.5 × 6 cm) forming an elevated cross (positioned 50 cm above the floor). Two arms were open, and the other two were enclosed by 14.5-cm high walls. The mouse was placed in the middle of the maze (always facing the same arm) and allowed to explore the maze for 10 min. A video tracking system (Ethovision XT 7.0, Noldus, Wageningen, The Netherlands) recorded the movements to calculate the percentage of time spent in the open arms, used as an index of anxiety behavior.
The Barnes maze
The Barnes maze test of learning and spatial memory was performed essentially as described. 14 The apparatus consisted of a circular white PVC platform (diameter: 122 cm) positioned 80 cm above the ground. Forty holes (diameter: 5 cm) were located at regular intervals of 4 cm from the edge (angle between holes from the center: 9°). Spatial cues were placed on the walls of the testing room and were never moved during the study. An aversive bright light (800 Lux) was used as a stimulus to motivate the mice to find a single black escape box (22.9 × 5.3 × 8.6 cm) located beneath one of the holes. One escape hole (i.e. the target hole) was randomly assigned to each mouse for all five days of the experiment.
During a habituation step, the animal initially contained in an opaque PVC cylinder was released at the center of the platform after 10 s, and gently guided towards the escape box, where it stayed for 2 min. During the acquisition phase (4 consecutive days of 4 daily trials with a 15-min break between trials), the mouse was placed at the center of the maze in the opaque cylinder for 10 s and then left to freely explore the maze and search for the escape box over 3 min. Once entered in the escape box, the mouse was left in there for 60 s. On the fifth day (for the probe trial), the maze underwent a 45° rotation and the escape box was removed. The mouse was allowed to move freely for 90 s. A video camera and a computer running Ethovision XT 11.5 software (Noldus, Wageningen, The Netherlands) were used to tracke and analyze the animal’s movements during the session. The primary latency (i.e. time spent before first visit to target hole) and primary errors (i.e. number of holes visited before first visit to target hole) were recorded as indices of learning during acquisition phase. Spatial reference memory was calculated as the percentage of time spent in the target quadrant. (i.e. the quadrant centered on the target hole) during the probe trial.
The touchscreen automated paired-associated learning task
Visuospatial memory was evaluated in the paired-associated learning (PAL) task, which is known to be hippocampus-dependent in rodents.
15
Using touchscreen operant chambers (Campden Instruments Ltd, UK), mice were tested for their ability to associate objects and spatial locations on the screen (six possible types of trial; two of them are represented in Figure 1). Prior to touchscreen testing, appetitive behavior toward sweet fluids was assessed in a sucrose preference test. To ensure that the mice were sufficiently motivated to perform the task, they were food-deprived until they had lost 10–15% of their baseline bodyweight.
The touchscreen automated dPAL task: illustration with two possible trial types. For each trial type, only one visual stimulus (the flower, the airplane or the spider) was presented in its correct location (denoted ‘+’); the second visual stimulus was presented in one of its two incorrect locations (denoted ‘−’), and the third location remained blank.
Before the PAL task, mice were pre-trained during several phases of instrumental operant conditioning. In the “habituation” phase, mice were trained to collect free rewards (7 µl of chocolate-flavor milk, accompanied by illumination of the tray where the reward was and a 1-second sound tone) during 20 and 40-min sessions. In the “initial touch” and “must touch” PAL training phases, mice were trained to touch the screen and then to specifically touch a white square stimulus (presented randomly in one of the three response windows) to obtain a reward. In the “punish incorrect” phase (the final stage of pre-training), a nose-poke of the white square stimulus (i.e. a correct response) was rewarded. A nose-poke of one of the two other blank windows (i.e. an incorrect response) resulted in house light illumination for a 5-s time-out period (no reward). When all the mice achieved a correct response rate of more than 75% over two consecutive sessions, they were moved onto “different PAL” (dPAL) training.
For the dPAL task, one of six possible trial types was randomly presented on the screen. For each trial type, only one visual stimulus (a flower, an airplane or a spider) was presented in its correct location (+); a second visual stimulus was presented in one of the two incorrect locations (−), and the third location remained blank. 16 Each trial type was presented an equal number of times and was not repeated in more than two consecutive trials. Mice were tested for their ability to nose-poke the correct location in each trial. An incorrect choice was followed by an additional correction trial with the previous pair of stimuli in the same spatial configuration, until the mouse made the correct choice. A correct choice was rewarded. An incorrect choice was followed by house light illumination for a 5-s time-out period. Mice performed 36 trials per session in less than 60 min, for 30 successive sessions (one per day). Correction trials were not counted for the total number of trials. In view of potential day-to-day variations in the mice’s performance levels, visuospatial memory was measured as the mean percentage of correct responses over a three-day period.
Pharmacological modulation by atorvastatin
Atorvastatin was administered by mixing a tablet into the mice’s standard chow at a dose of 5 mg/kg/day. Mice were randomized to atorvastatin treatment. Atorvastatin administration was initiated immediately after surgery in the COL0.8 group and continued throughout the follow-up period.
Statistical analysis
Data are quoted as the mean ± standard deviation (SD). The normality of the data distribution was assessed graphically and in a Shapiro–Wilk test. The MRI data in the four groups (COL0.2, COL0.8, COL2, and sham) at 24 h were compared in a Kruskal–Wallis test, followed by Bonferroni correction for multiple comparisons. The PET data at six weeks for the sham and CMB groups were compared in a Mann–Whitney U test. Differences between the sham and CMB groups in the behavioral tests were probed with Student’s t-test or an analysis of variance with repeated measures. ANOVA with or without repeated measures, followed by multiple comparisons with Bonferroni correction were performed on the behavioral tests to compare the three groups ‘sham’, ‘CMB’ and ‘CMB-AT’. All statistical analyses were performed with SAS software (version 9.3, SAS Institute Inc., Cary, NC, USA). The threshold for statistical significance was set to p < 0.05.
Ethical aspects
All animal procedures were approved by the national Ethical Committee in Animal Experimentation (CEEA, comité d’éthique en experimentation animale), from the french Ministry for Education and Research (agreement number: 00279.03) and were performed in strict compliance with the European Union Directive 2010/63/EU. The experiments are reported in accordance with the ARRIVE guidelines for reporting experiments involving animals.
Results
Description of the model
Thirty-eight male mice underwent surgery and were randomized to the COL0.2 (n = 7), COL0.8 (n = 10), COL2 (n = 12) and sham groups (n = 9). Surgery was very well tolerated by all mice but one in the COL2 (high-dose) group that died during anesthesia. There were no late deaths during the follow-up period.
No bleeding related to Hamilton syringe insertion was detectable on brain MRI at 24 h after surgery for the sham group. A proportional dose effect was observed between collagenase concentration and the lesion volume estimated on a T2* MRI sequence (Figure 2(a)). The lesion induced by 0.2 µU was not detectable, the one induced by 0.8 µU ranged from 50 to 100 µm and the concentration of 2 µU induced extensive hemorrhage (Figure 2(b)), also characterized by a significantly elevated hemosiderin level (i.e. a lesioned-to-healthy zone low ratio in a T2* sequence) (Figure 2(c)). In the sham group and the COL0.2 group, the H&E staining only revealed the mark of syringe insertion in the brain tissue without hemorrhagic lesion. In the COL0.8 group, a well delimited hemorrhage was observed. The COL2 group displayed an extensive hemorrhagic lesion along with cerebral tissue disorganization (Figure 2(a)).
(a) Coronal view of a) brain MRI, b) fresh tissue, c) tissue stained with H&E; magnification = ×5, and d) tissue stained with H&E; magnification = ×10, for the different groups (sham, COL 0.2, COL 0.8 and COL 2) 24 h after stereotaxic surgery. Arrows represent the site of injection. (b) Lesion volume (expressed in mm3) in the sham, COL 0.2, COL 0.8 and COL 2 groups. The volume was significantly different in each group when compared to COL 2 group * p < 0.05; **p < 0.01; (c) The T2* lesioned zone/healthy zone ratio for the sham, COL 0.2, COL 0.8 and COL 2 groups; a lower ratio corresponds to a higher hemosiderin level. The ratio was significantly different between the sham and COL 2 groups * p < 0.05. Data are expressed as means ± SD.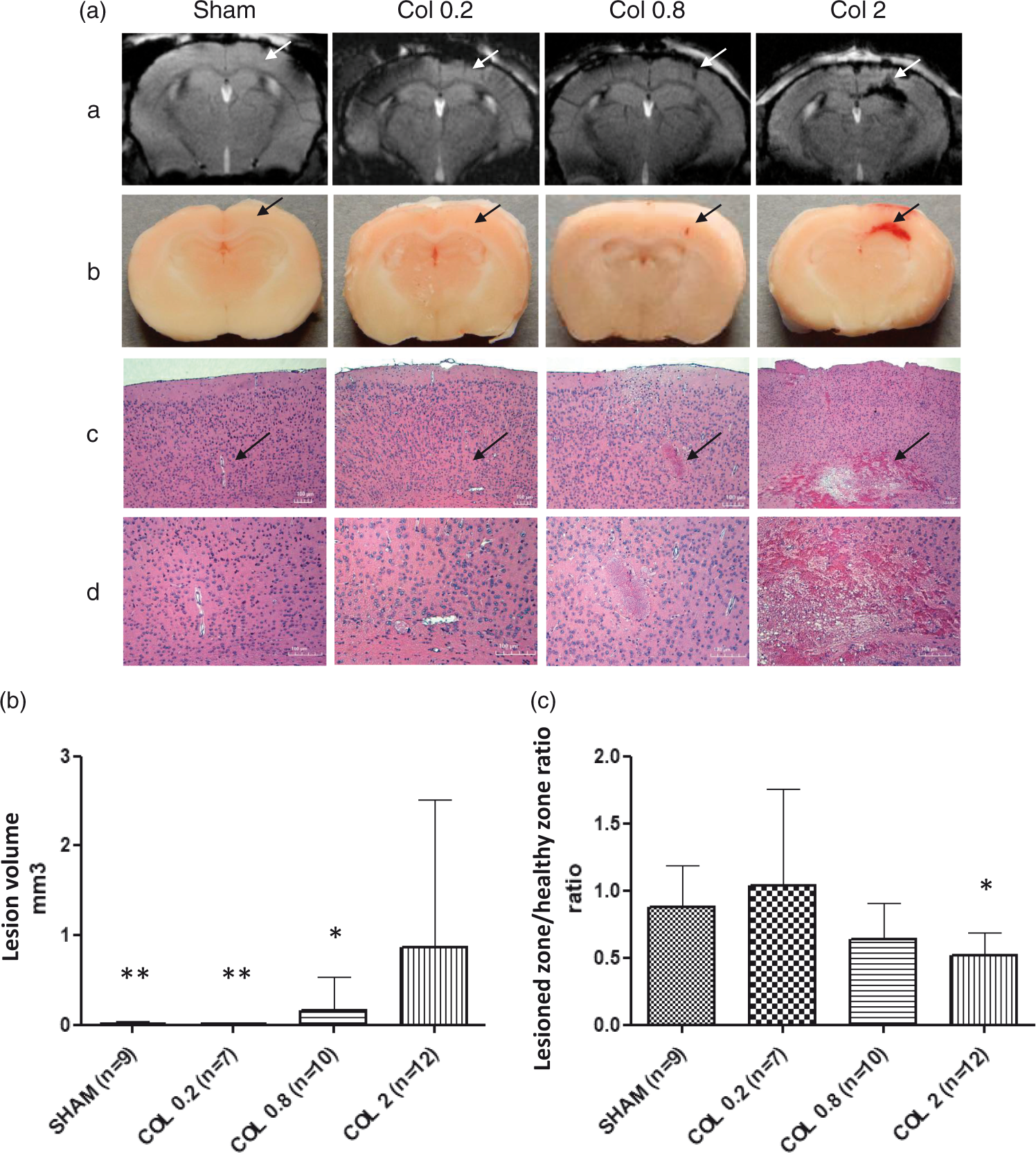
We conclude that a concentration of 0.8 µU collagenase was appropriate to induce a lesion corresponding to CMB.
Assessment of behavior, cerebral metabolism and pharmacological modulation
For the behavioral assessment, a total of 62 mice were tested: 14 mice in Sham group, 14 mice in CMB group, and 9 mice in CMB+AT group for non cognitive tests and the Barnes maze; and independently, 7 mice in Sham group, 9 mice in CMB group, and 9 mice in CMB+AT group for the dPAL test. For the [18F]FDG PET, we randomly selected eight mice in Sham group and six mice in CMB group.
Behavioral and cognitive functions
Locomotor
activity and level of anxiety
The open field test evidenced no difference in motor activity between the sham group and the CMB group (Figure 3(a)) as opposed to the EPM test (Figure 3(b)) where CMB mice spent a significant greater amount of time in the open arms, suggesting a lower level of anxiety.
Non cognitive Tests, General locomotor activity (a), level of anxiety (b) and cognitive tests, spatial learning (acquisition) and reference memory (probe trial) in the Barnes maze (c) and visuo-spatial memory in the dPAL task (d). Comparison of sham (n = 14 in a, b, c and n = 7 in d), CMB (n = 14 in a, b, c and n = 9 in d) and CMB treated by atorvastatin (n = 9 in a and b) groups at six weeks post-surgery. Dotted lines (c and d) represent values expected by chance. Data are expressed as means ± SD. *p < 0.05, CMB vs. sham. §§p < 0.01, D4 vs. D1 (c). **p < 0.01, CMB vs. sham. §§p < 0.01, CMB vs. CMB + At (d).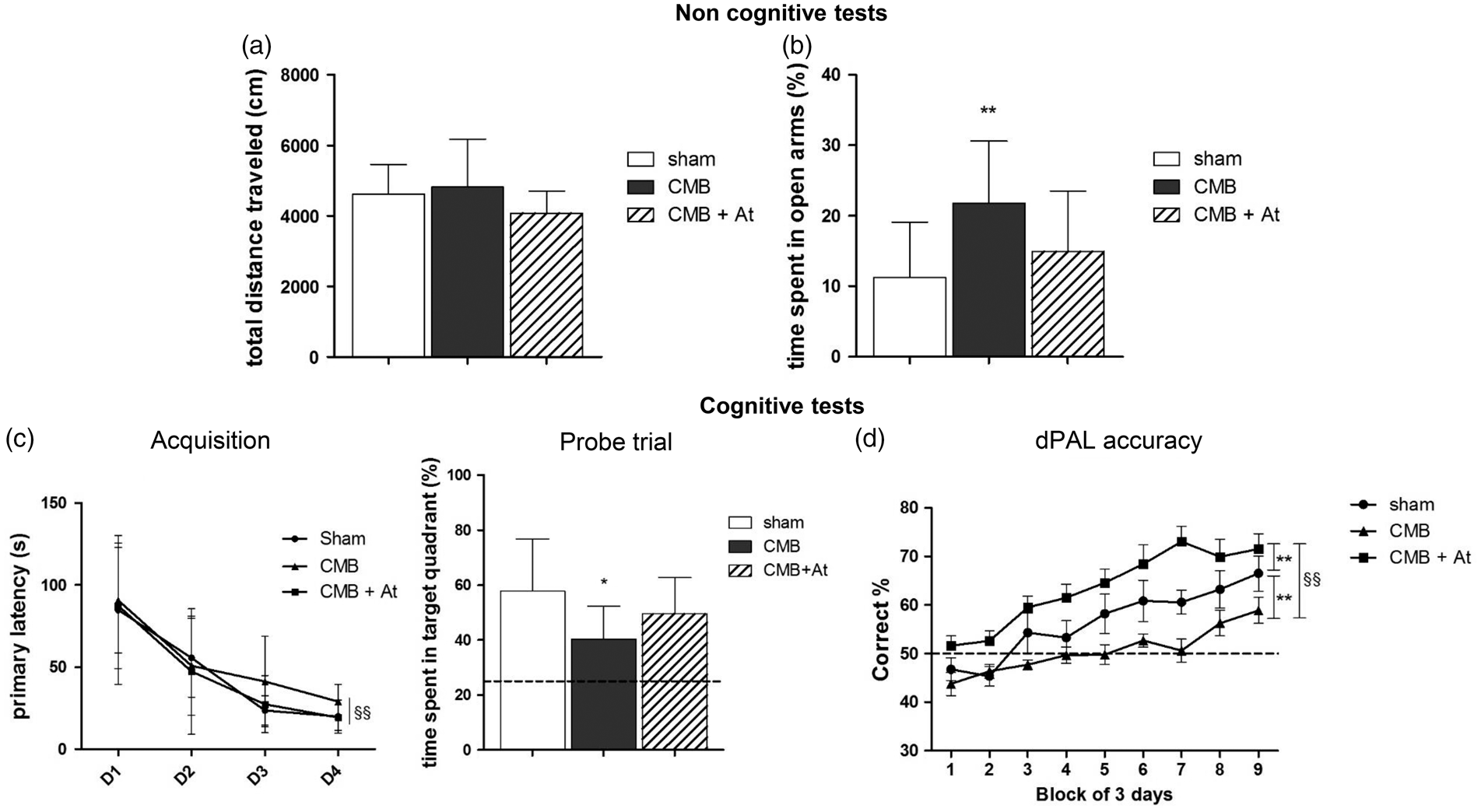
Learning and spatial reference memory
The primary latencies significantly decreased from D1 to D4 (acquisition training period), denoting that all mice learned to find the escape box from day 4 onwards (Figure 3(c)). In the probe trial, although both groups stayed in the target quadrant longer than expected by chance (25%), the CMB group spent significantly less time than the sham (time spent in target quadrant (%), mean ± SD: 40.3 ± 12 vs. 57.8 ± 19 respectively, p = 0.0126), suggesting an altered learning and spatial memory (Figure 3(c)).
Visuospatial memory
Learning process was supported by the increasing percentage of correct response along with test duration from both groups. However, a delay in this learning process was observed among the CMB mice characterized by significantly impaired visuospatial memory from day 12 of training onwards. The intergroup difference remained statistically significant until the end of the assessment period (Figure 3(d)).
Cerebral metabolism
When compared to the sham group, the radiotracer uptake was lower in the CMB group, showing hypometabolism in the ipsilateral dentate gyrus (mean ± SD: 0.81 ± 0.04 vs. 0.92 ± 0.11; p = 0.0169), in the cortex (ipsilateral cortex 0.85 ± 0.05 vs. 0.98 ± 0.10; p = 0.0118 and contralateral cortex 0.86 ± 0.04 vs. 0.97 ± 0.09; p = 0.0118), and in the striatum (ipsilateral striatum 0.87 ± 0.06 vs. 1.03 ± 0.13; p = 0.0239 and contralateral striatum 0.86 ± 0.05 vs. 1.00 ± 0.11; p = 0.0169) (Figure 4).
In vivo [18F]FDG-PET cerebral metabolic measurement six weeks after surgery. (a) Bars represent the mean of uptake of [18F]FDG in each region. The regional cerebral glucose metabolism (rCGM) was calculated relative to the uptake in a reference zone (cerebellum). p values were calculated with non-parametric Mann–Whitney’s test. Significant differences between groups are represented by: *p < 0.05. Data are expressed as means ± SD. Cx_L: left cortex; Cx_R: right cortex; DG_L: left denta gyrus; DG_R: right denta gyrus; Hi_L: left hippocampus; Hi_R: right hippocampus; Hy: hypothalamus; IC: inferior culliculus; MB: midbrain; PN: pons nucleus; SN: septal nucleus; St_L: left striatum; St_R: right striatum; SC: superior culliculus; Th: thalamus. (b) Axial views of fused [18F]FDG PET-MR images of mouse brain Right: CMB Group; Left: Sham group.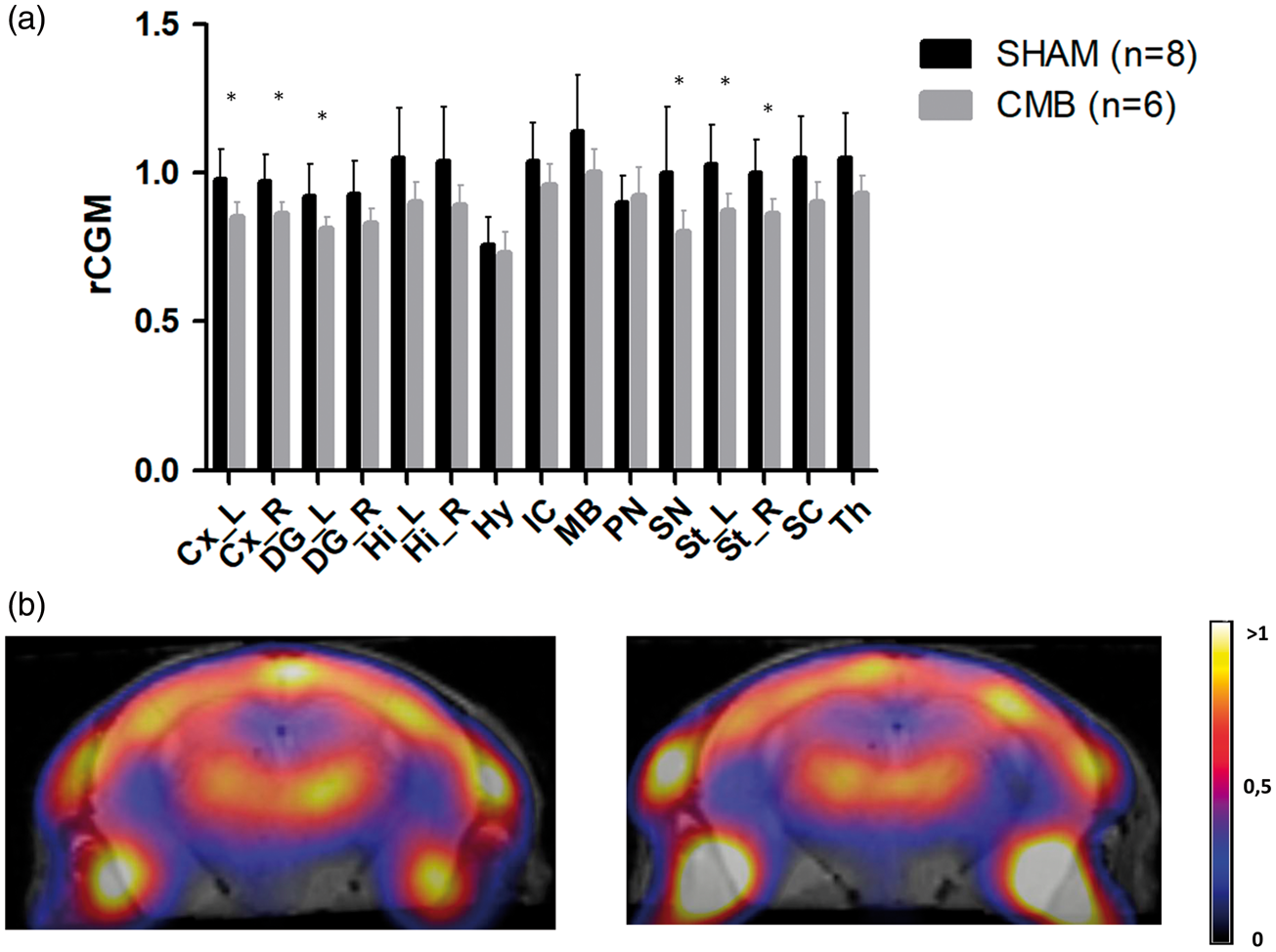
Pharmacological modulation by atorvastatin
At six weeks, there were no significant differences for the motor activity nor for the anxiety among atorvastatin-treated CMB-mice (“CMB-AT”), CMB and sham groups (Figure 3(a), (b)).
For cognitive functions, atorvastatin treatment seemed to improve the CMB-induced impairment in spatial reference memory (time spent in target quadrant (%), mean ± SD: 40.3 ± 12 for CMB group and 49.5 ± 13.2 for CMB-AT group, p = 0.392) (Figure 3(c))
Pharmacological modulation had a significant, beneficial effect on visuospatial memory; an improvement in the CMB-AT group was first seen at the start of the training period and was maintained throughout the experiment. The CMB-AT group achieved a correct response rate even higher than in the sham group (Figure 3(d)).
Discussion
Our present results (i) confirmed the reliability and reproducibility of a murine model of CMB (produced by the cortical injection of 0.8 µU/µL collagenase) (ii) demonstrated significant cognitive impairments six weeks after CMB induction, together with significant hypometabolism in the hippocampus; and (iii) indicated that the CMB model can be pharmacologically modulated by atorvastatin.
Although several methods for inducing brain hemorrhage in mice have been described in the literature, very few models focused on the induction and impact of cortical CMBs. The few reports on CMBs mainly relate to transgenic mice that spontaneously develop microbleeds at many different sites in the brain.17,18 In wild-type mice, lipopolysaccharide has been used to induce cortical and non-cortical CMBs. 19 The dietary induction of hyperhomocysteinemia results in widespread microbleeds in the entorhinal, parietal and frontal cortices and in the hippocampal regions. 13 Recently, a two-photon excitation microscopy study evidenced the induction of a large number of cortical or deep microbleeds after the rupture of a specific targeted penetrating arteriole.20,21 Furthermore, we noted publications on three preclinical models (in rabbit, mouse and rat) of intracerebral hemorrhage (ICH) induction, in which the size of the lesion corresponded to “macrohemorrhages”; ICH was variously induced by injection of autologous blood or bacterial collagenase 22 or the insertion of a microballoon. 23 Manaenko et al. 24 compared these models in the rodent by taking account of their technical aspects and pathophysiological characteristics. The insertion of a microballon mimics the mass effect but not the mechanisms that accompany hemorrhage, such as the release of toxic substances in the blood or damage to the blood–brain barrier (BBB). The injections of autologous blood and bacterial collagenase are quite similar in technical terms, and induce a hemorrhage in a relatively simple, reproducible manner with good control over the lesion size; the latter aspect is particularly relevant when seeking to induce microbleeds. Moreover, it allows to carefully limit the boundaries of the cortical lesion site. When compared with the injection of autologous blood, the ICH induced by collagenase might lead to much the same histological modifications as observed in the human brain after spontaneous brain hemorrhage: edema, neuronal loss, marked rupture of the BBB, slower resolution of hematoma, marked, persistent neurological impairments, and the absence of spontaneous recovery. These are interesting parameters for long-term follow-up studies, explaining the choice of collagenase in our model.
Microbleeds and macrohemorrhages are distinct entities. 25 For the purpose of our study, it was important to obtain small lesions (<100 µm), and to locate and qualify CMBs in T2* MRI sequence. The T2* sequence used with the present model is the same as that used to detect CMBs in the clinic. Considering those constraints, the dose of collagenase providing the best compromise between lesion size and T2* MRI detection 24 h after surgery was 0.8 µU/µL. It produced a lesion volume of about 0.03 mm3 (diameter: 50 to 100 µm). Proportionally, this lesion corresponds to a hemorrhagic burden of 0.006% of the mouse brain’s total volume. This may be compared with CMBs measured in clinical practice, which account for about 0.005% of total brain volume. 1 These CMB volumes are also similar in size to the spontaneous microbleeds observed in transgenic mouse models of amyloid angiopathy. 18
The functional assessment of the present model confirmed that cortical CMBs are directly associated with behavioral and cognitive impairments. Although the CMB and sham groups showed similar levels of spontaneous locomotor activity six weeks after induction of the lesion, we observed significant intergroup differences in other tests. The mice in the CMB group displayed (i) a lower level of anxiety; (ii) an impairment in spatial reference memory, and (iii) an impairment in the visuospatial memory. The significant cognitive impairments observed six weeks after surgery cannot be ascribed to a lack of motor activity or a very high level of anxiety. There are few data on behavioral and cognitive tests in models of ICH; most studies have focused on motor impairments in the context of striatal macrohemorrhages. These models display motor impairments one to two days after the induction of lesions; the impairments then resolve progressively over the following month.26–28 The lesions were not associated with changes in the level of anxiety. 26 On the cognitive level, MacLellan et al. did not observe any impairment one month after lesion induction. Hartman et al. 29 noted learning impairments two weeks after lesion induction (which had disappeared eight weeks afterwards) but did not find an impairment in spatial memory (according to the Morris water maze). It was shown in a murine model of vascular dementia in which CMB-inducing hyperhomocysteinemia was produced by administration of a specific diet for 14 weeks, that the surviving mice presented a significant impairment in spatial memory. 13 However, it must be noted that the model featured many microbleeds in several different regions of the brain (primarily the entorhinal and parietal cortex but also the frontal cortex and the hippocampus). No significant intergroup differences in motor function were noted. 13
Our study highlights the relevance of the touchscreen test in animal models. The PAL test is part of the CANTAB battery in humans, and is known to be a very sensitive tool for detecting the onset and progression of cognitive disorders. 30 Moreover, the touchscreen enables the evaluation of several cognitive functions: attention, flexibility and visuospatial memory. 31 We chose to use the dPAL version of the touchscreen PAL test because it probes memory related to the dorsal part of the hippocampus in rats 32 and mice, 15 which is often insufficiently assessed by conventional tests. As well as enabling a translational approach, the touchscreen PAL test decreases the level of stress induced by the investigator (relative to standard behavioral tests) because the animals are confined in an acoustically insulated, odor-free box. The dPAL test results were coherent with those of the Barnes maze, and may evidence the hippocampal dysfunction induced by CMBs.
The present behavioral data are also in line with the reduced radiotracer uptake depicted through the in vivo FDG-PET scans at six weeks’ post-lesion. A hypometabolic pattern was observed in the cortex, the ipsilateral and contralateral striatum, and the ipsilateral dentate gyrus as opposed to the radiotracer biodistribution featuring in the sham group. To the best of our knowledge, the present study is the first to report the simultaneous cognitive and metabolic impact of cortical CMBs at six weeks post-lesion. In daily clinic, CMBs (and particularly CAA-related CMBs) lead to gray matter atrophy and glucose hypometabolism in patients with AD. 33 In elder adults with normal cognition, cortical CMBs are associated with extensive reductions in resting-state cerebral blood flow but with no changes in brain metabolism. 9 In mice, sustained cerebral hypoperfusion due to microbleeds might produce gliovascular changes and cognitive impairments. 34 Furthermore, impaired glucose metabolism is thought to lead to neuronal/synaptic dysfunction and cognitive decline. 35 The hypometabolism observed in our model (particularly in the hippocampus) is a clear marker of the CMBs’ impact, and is corroborated by the data from cognitive tests.
By adopting a cognitive approach, we designed our model as a suitable tool to assess pharmacological modulation. To this end, lipid-lowering agents (such as statins, with their pleiotropic biological effects) seemed to be good candidates for testing. Although statins are widely prescribed for the treatment of hypercholesterolemia and the secondary prevention of cardiovascular and cerebrovascular disease, they also have many other cholesterol-independent effects 36 : improved endothelial function in atherosclerotic vessels, antithrombotic and anti-inflammatory effects, and a possible reduction in apoptosis and cell death. This effect could be related to peroxisome proliferator-activated receptors (PPARs) modulation that we have previously suggested in vascular and inflammatory process in cerebral ischemia. 37 The results in our model indicate that statins may be of value for reducing the impact of CMBs on cognition. Administered alone or in late life, statins’ effects on cognition in AD and all-cause dementia seem to be slight and remain subject to debate. 38 A recent review suggests the use of statins, in association with other pharmacological and non-pharmacological interventions as disease-modifying therapies. 39
The mechanisms by which CMBs induce impairment of cognitive function may involve both focal brain damage, with inflammation, BBB dysfunction, microglia activation, and a dysfunction of the structural networks in the brain.20,40 These mechanisms are directly related to CMB occurrence, rather than other pathophysiological processes underlying CMB occurrence, opening a way for a direct pharmacological modulation as observed with atorvastatin. These multiple mechanisms will be studied in a specific study, with a long-term follow-up of the mice to precisely describe the impact of CMB over time.
In conclusion, we developed a novel murine model of CMB and used it to assess the animals’ cognitive and metabolic characteristics six weeks after CMB induction. Our results support that this model is sensitive to pharmacological modulation, foreseeing interesting research perspectives. The present model was described in wild-type mice but could also be applied to APP transgenic mice – thus enabling an assessment of the relationships between amyloid and vascular mechanisms in cognitive impairment. Our model also opens up perspectives for long-term follow-up studies, to describe functional, metabolic and mechanistic impact of CMBs as well as to investigate the latter’s impact in APP transgenic mice with spontaneous cognitive impairments. This approach might provide information on the putative interactions between amyloid pathologies and cerebrovascular lesions. Lastly, the model’s sensitivity to pharmacological modulation may facilitate the development of CMB treatment strategies. Such model could contribute to complete assessment of existing or new drugs in vascular cognitive impairment.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgements
We thank the in vivo imaging core facility for assistance, the Neurophysiological and behavioral exploration platform for rodent (Federation of Neurosciences, University of Lille) and Marie-Hélène Gevaert for her technical assistance in brain tissue preparation and staining (Department of Histology, Medical faculty of Lille).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Sandrine Bergeron: study concept and design, acquisition, analysis and interpretation of data, literature search, and draft of manuscript.
Yaohua Chen: study concept and design, acquisition, analysis and interpretation of data, literature search, and draft of manuscript.
Florent Auger: acquisition, analysis and interpretation of imaging data, critical revision of manuscript.
Julie Deguil: critical revision of manuscript.
Nicolas Durieux: acquisition of imaging data.
Emilie Skrobala: analysis of data.
Romain Barus: acquisition of functional and histochemical data.
Camille Potey: study concept and design, literature search.
Charlotte Cordonnier: study concept and design.
Florence Pasquier: study concept and design.
Laura Ravasi: study concept and design, acquisition, analysis and interpretation of imaging data, critical revision of manuscript.
Régis Bordet: study concept and design, critical revision and final approval of manuscript.
Sophie Gautier: study concept and design, literature search, critical revision and final approval of manuscript.
