Abstract
Cognitive dysfunction, as a consequence of dementia, is a significant cause of morbidity lacking efficacious treatment. Females comprise at least half of this demographic but have been vastly underrepresented in preclinical studies. The current study addressed this gap by assessing the protective efficacy of physical exercise and cognitive activity on learning and memory outcomes in a rat model of vascular dementia. Forty ovariectomized Sprague-Dawley rats (~6 months old) were exposed to either a diet high in saturated fats and refined sugars or standard laboratory chow and underwent either chronic bilateral carotid occlusion or Sham surgery. Learning and memory abilities were evaluated using standard cognitive outcomes over the ensuing 6 months, followed by histologic analyses of hippocampal CA1 neurons. In Experiment 1, we confirmed hypoperfusion-induced cognitive dysfunction using a 2 × 2 (Surgery × Diet) experimental design, without alterations in hippocampal architecture. In Experiment 2, hypoperfused animals were either exposed to alternating days of physical (wheel running) and cognitive activity (modified Hebb–Williams maze) or sedentary housing. In contrast to males, this combination rehabilitation paradigm did not improve cognition or histopathologic outcomes in hypoperfused animals. These findings, highlighting differences between female and male animals, show the necessity of including both sexes in preclinical experimentation.
INTRODUCTION
Dementia, as a result of pathologic cognitive changes including Alzheimer's disease and vascular dementia (VasD), becomes more prevalent with age. 1 Unfortunately, scientists have yet to identify effective therapeutic interventions to ameliorate these cognitive disorders.
One promising avenue of research in treating cognitive dysfunction involves using a more holistic approach of physical and cognitive exercise. Recently, we showed that a rehabilitation paradigm consisting of a combination of physical and cognitive activity (PA/CA) improved learning and memory abilities in young, naïve, 2 and early middle-aged, chronically hypoperfused 3 male rats. In the latter study, chronic hypoperfusion was used to model VasD4,5 and interestingly, the PA/CA intervention had neurorestorative effects, preventing (or reversing) hippocampal CA1 hypertrophy.
These preclinical data are particularly important given beneficial reports of both PA and CA in the clinical population. For example, exercise has been linked with improvements in executive, spatial and speed tasks as well as cognitive flexibility.6,7 Further, rates of cognitive decline and dementia appear to be reduced in those individuals who were regular exercisers. 8 Importantly, this effect held true in a randomized controlled trial. 9
Similar to the positive effects of PA on brain health, CA has also been linked to improvements in cognition. Activities such as board and card games and reading have an overall positive effect on cognition. 10 This was also noted in the famous Nun study where higher linguistic skills early in life correlated with a lower incidence of dementia in later life. 11 It is notable that the above two studies were conducted using only female participants. Consequently, there appears to be somewhat of a mismatch between these patient demographics and those commonly employed in animal studies where males predominate. Women comprise at least half of the dementia population 1 and as outlined by the STAIR (Stroke Therapy Academic Industry Roundtable), inclusion of both male and female animals in preclinical studies is imperative to the successful development of therapies to treat stroke-associated impairments.12,13
In the current study, and following the STAIR recommendations, the impact of PA/CA on learning and memory outcomes in a female rat model of VasD was examined. We implemented comorbid conditions, also highlighted by the STAIR committee, including older animals (early middle-age) and a diet rich in saturated fats and refined sugars, similar to that consumed in western society.12,13 The current study first sought to validate the chronic hypoperfusion model of VasD in this cohort of female animals and the contribution of dietary manipulation on outcome measures (Experiment 1) and, second, to assess the efficacy of a rehabilitation paradigm that combined PA/CA in reducing cognitive dysfunction associated with VasD (Experiment 2). We predicted that VasD animals would experience significant learning and memory deficits and these deficits would be at least partially attenuated by a combination rehabilitation therapy.
MATERIALS AND METHODS
Subjects
Forty female, Sprague-Dawley rats (Memorial University Vivarium, St John's, Newfoundland and Labrador, Canada) weighing ~430 g (6 to 8 months; Table 1) upon arrival were used in this study. Animals were placed on a 12:12 hour reverse light:dark cycle (lights off at 0600) and before group designation, animals were pair-housed in standardized cages. All procedures were approved by Memorial University's Institutional Animal Care Committee and conformed to the Canadian Council on Animal Care guidelines.
Animals' body weight (g) (means ± SEM)
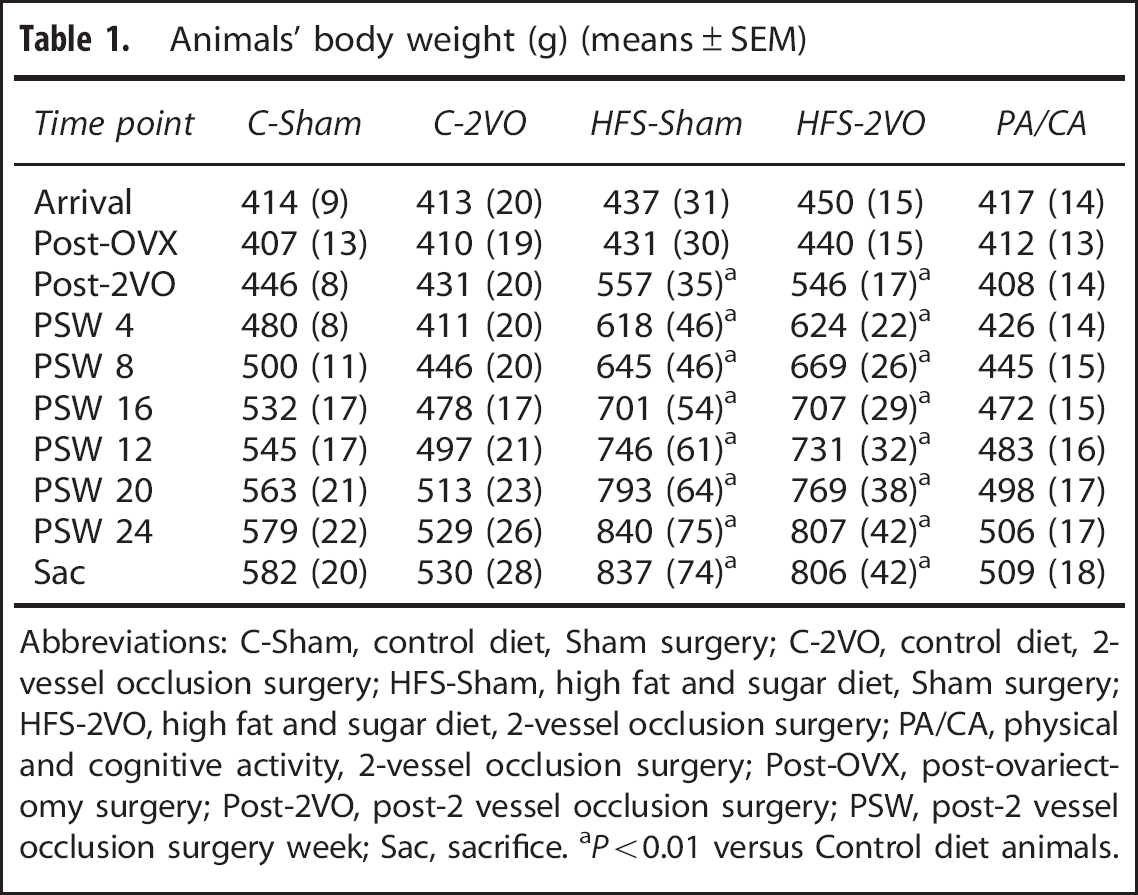
Abbreviations: C-Sham, control diet, Sham surgery; C-2VO, control diet, 2-vessel occlusion surgery; HFS-Sham, high fat and sugar diet, Sham surgery; HFS-2VO, high fat and sugar diet, 2-vessel occlusion surgery; PA/CA, physical and cognitive activity, 2-vessel occlusion surgery; Post-OVX, post-ovariectomy surgery; Post-2VO, post-2 vessel occlusion surgery; PSW, post-2 vessel occlusion surgery week; Sac, sacrifice. aP < 0.01 versus Control diet animals.
Experimental Conditions
Animal pairs were pseudorandomized into one of five conditions: Sham—control diet (n = 5); Sham—high fat, high sugar diet (HFS; n = 6); 2-vessel occlusion—control diet (2VO; n = 8); 2VO-HFS diet (n = 10); and 2VO + PA/CA (n = 11). Animals in the Sham, HSF, 2VO, and 2VO-HSF groups were housed in pairs and a single condition was represented within a cage. The PA/CA condition consisted of a paradigm previously used in our laboratory 3 and was implemented after 2VO surgery (Experiment 2). Animals in this condition were individually exposed to voluntary running wheels, 3 days/week and on alternate days were housed in pairs and exposed to cognitive activity, 3 days/week; the seventh day they were housed in pairs in the absence of PA or CA. Physical activity consisted of exposure to 36 cm diameter wheels for 24 hours a day. Cognitive activity consisted of modified Hebb–Williams maze (100 × 100 × 20 cm3), 14 for 24 hours a day, with the maze configuration changed weekly to increase novelty. Additionally, novel objects and food rewards were scattered throughout the maze and the water bottle and food hopper were separated to encourage exploration. The PA/CA animals were housed in these conditions throughout the study. The PA/CA animals were housed in these conditions throughout the study except during behavioral testing and pair-housed in standard cages on the remaining day each week.
Experimental Diet
Animals were acclimated to the animal care facility for 2 weeks before condition assignment, pair-housed and were given standard laboratory chow and water ad libitum. After ovariectomy, animals were pseudorandomized to either standard laboratory chow (Control diet) or HFS diet (high fat/high sucrose purified diet meal, TestDiet, 57NZ, Richmond, IN, USA) that consisted of protein (21% of energy (kcal)), fat (39%; lard), and carbohydrates (40%; of which 45% was refined sugar (sucrose), and 3% fiber). The effects of dietary manipulation were assessed in Experiment 1.
Surgical Procedures
For all surgical procedures, animals were anesthetized with isoflurane (4.0% induction, 2.5% maintenance) in 70/30% nitrous oxide/oxygen mixture. To model the postmenopausal period, animals underwent bilateral ovariectomy using a dorsal surgical approach. 15 One month after ovariectomy, animals underwent permanent bilateral carotid artery occlusion 5 or Sham surgery. Briefly, a midline neck incision was made and both common carotid arteries were isolated from adjacent nerve bundles. Each artery was doubly ligated (2VO) with 4-0 silk sutures, occlusion ensured, and returned to the neck cavity. Sham surgery consisted of similar procedures (i.e., neck incision and isolation of arteries) but patent arteries were returned to neck cavity without ligation. Animals received topical 2% Xylocaine (AstraZeneca, Mississauga, ON, Canada) on the midline neck incision and were placed in a standard cage on a heating blanket until recovered.
Behavioral Assessments
Figure 1 illustrates the experimental timeline for all behavioral assessments. All functional assessments were conducted during the animals' dark cycle.
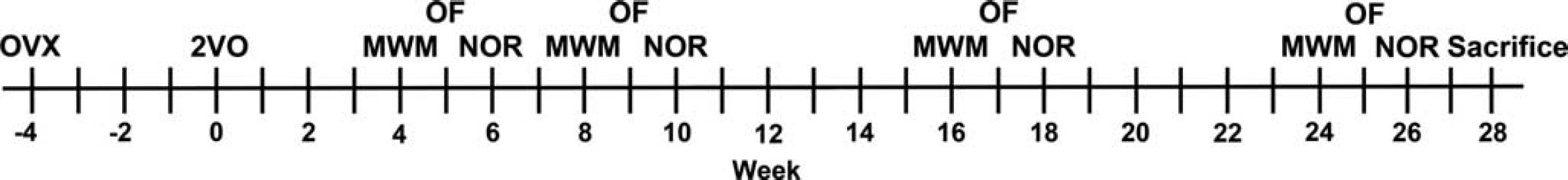
Experimental timeline of behavioral assessments. MWM, Morris Water Maze; NOR, novel object recognition; OF, open field; OVX, ovariectomy; 2VO, 2-vessel occlusion (bilateral carotid artery occlusion).
Morris Water Maze
Animals were tested in the Morris Water Maze (MWM) at 4, 8, 16, and 24 weeks after 2VO surgery. A standard spatial MWM paradigm 16 was implemented that used numerous distal visual cues (e.g., door, computer, and shapes), auditory cue (radio) in a corner of the room and a scented marker located on the wall of one quadrant. The maze consisted of a circular black pool (180 cm diameter) filled with water (~50 cm depth, ~22°C). Animals were required to locate a submerged circular platform (~2.5 cm) that was placed within a designated target quadrant, 25 to 30 cm from the maze wall. The MWM paradigm consisted of 4 trials/day for 4 consecutive days (acquisition), with a maximum latency of 90 seconds, at which an animal was gently guided to the platform. Once on the platform, animals remained there for 10 seconds and were then removed from the maze and placed in a holding cage until the next trial. The intertrial interval was 60 seconds. Animals were pseudorandomly placed, facing the wall, into a quadrant at the beginning of each trial. The platform remained in the same location throughout each test period, but was moved to a new location at the beginning of each new test period. Dependent measures included latency and distance traveled to locate the hidden platform. Twenty-four hours after acquisition, a probe trial (memory) was implemented where the platform was removed. Animals were placed in the maze directly opposite the platform location, facing the maze wall and allowed 60 seconds to freely explore the maze. An additional probe trial was implemented 1 week later, assessing long-term memory retention. The dependent measure in the probe trials was the amount of time spent in the target quadrant.
To exclude the possibility of visual dysfunction caused by 2VO, 4 animals' visual abilities were assessed using a visible platform paradigm 1 day after the first probe at each test period. The circular platform was elevated ~2.5 cm above the water surface and wrapped with brightly colored tape to increase visibility. Animals' latency to locate the visible platform was recorded.
Open Field
One week after the MWM probe session at weeks 5, 9, 17, and 25 after surgery, animals were placed into an open field chamber (72 × 75 × 57 cm) in a soundproof room for 5 minutes and videotaped from above. The dependent measure was the number of grid crosses using a 3 × 3 grid (24 × 25 cm).
Novel Object Recognition
The object recognition test was implemented 1 week after each Open Field test session (i.e., weeks 6, 10, 18, and 26) using the same chamber located in the soundproof room. 17 Rats received three, 5-minute trials during each session, with a 7-minute intertrial interval, and were placed in the home cage during the intertrial period. Novel objects were used at each test period. During the first trial, two identical objects were placed in opposite corners and animals were allowed to freely explore the maze and objects. The second trial consisted of changing the location of one object and the last trial consisted of using the original locations but replacing one of the objects with a new object. Each session was videotaped from above and the dependent measure included time spent investigating an object. Object investigation was counted when the rat was actively sniffing the object or if the nose was within ~2 cm of the object (touching the object with its vibrissae).
Hippocampal CA1 Quantification
On completion of behavioral analyses, animals were deeply anesthetized with isoflurane (4.0% in 70/30% nitrous oxide/oxygen mixture) and transcardially perfused with ice-cold 0.9% heparinized saline and 4.0% paraformaldehyde. Animals were decapitated, brains removed and postfixed overnight in 4.0% paraformaldehyde at 4°C. The following day, brains were placed in a 20% sucrose solution in phosphate-buffered saline and stored at 4°C until saturated, then frozen on dry ice and stored at −20°C until further processing.
Brains were sectioned at 40 μm and slide mounted using a cryostat. Slides were stained with cresyl violet and seven equally spaced sections between bregma areas −2.30 mm and −4.52 mm 18 corresponding to the rostral and caudal dorsal hippocampus, respectively, were analyzed. Dorsal CA1 cell number and size were estimated using unbiased stereological procedures using a Leica DMRXE microscope (Leica Microsystems Inc., Richmond Hill, ON, Canada) and the Optical Fractionator method of Stereo Investigator (MBF Bioscience, Williston, VT, USA). A three-dimensional optical dissector counting frame (x, y, and z dimensions of 35 × 35 × 13 μm, respectively) was applied to systematic random sample sites resulting in an average of 15 to 20 sampling sites superimposed on the circumscribed area of dorsal CA1. 3 The dorsal CA1 hippocampus was outlined using a 5× objective and this allowed for the generation of sampling sites using the Optical Fractionator method (300 μm in the x direction and 100 μm in the y direction). One hippocampus was measured in each brain because there were no anticipated asymmetric differences with this model of VasD. 3 A border guard of 2 μm was set at the top and bottom of each section and CA1 cell number and size (calculated using the Nucleator method of Stereo Investigator) estimates were calculated using a 40× objective and the Serial Section Manager method of Stereo Investigator. On average, ~470 cells were counted/brain with a coefficient of error of 0.09.
Statistical Analyses
All data are presented as mean ± standard error of the mean (SEM) and were analyzed with the Statistical Package for Social Sciences (IBM SPSS Statistics; v. 19.0.0, Armonk, NY, USA). Behavioral and histologic data for Experiment 1 were analyzed using a 2 × 2 (Surgery × Diet) analysis of variance (ANOVA) with or without repeated measures, where appropriate. Performance in the novel object test was analyzed with a 2 × 2 ANOVA as well as a paired t-test to assess exploratory behavior. Post hoc tests were conducted using Tukey's Honestly Difference (HSD) test. In Experiment 2, rehabilitation efficacy was analyzed with a one-way ANOVA, with or without repeated measures where appropriate or an independent t-test. In cases where homogeneity of variance or sphericity assumptions were violated, the Brown–Forsythe or Huynh–Feldt correction was used, respectively. Data were collected and analyzed by experimenters blinded to experimental condition. The alpha level was set at 5% and data were considered as statistically significant at P < 0.05.
RESULTS
Two rats (2VO + Control Diet and 2VO + HFS) were excluded from the analyses because of unilateral hippocampal injury.
Experiment 1: Model Validation
Morris Water Maze
Visible platform. To exclude the possibility of visual defects due to permanent bilateral carotid occlusion, 4 animals' visual abilities were assessed using a visible platform paradigm in the MWM at each test period. There was no effect of Surgery, Diet and no significant interaction in latency to locate the visible platform at postsurgery weeks (PSW) 4, 8, and 16 (P > 0.05). There was, however, a significant effect of Surgery at PSW 24 (F1,15.821 = 10.727, P < 0.01), but there was no effect of Diet and no interaction. Post hoc analysis revealed that 2VO animals required significantly more time to locate the visible platform.
Acquisition: hidden platform. In all cases, 2VO animals required significantly more time and swam longer path lengths to locate the hidden platform than the Sham control animals thus indicating a deficit in learning abilities (Figure 2).
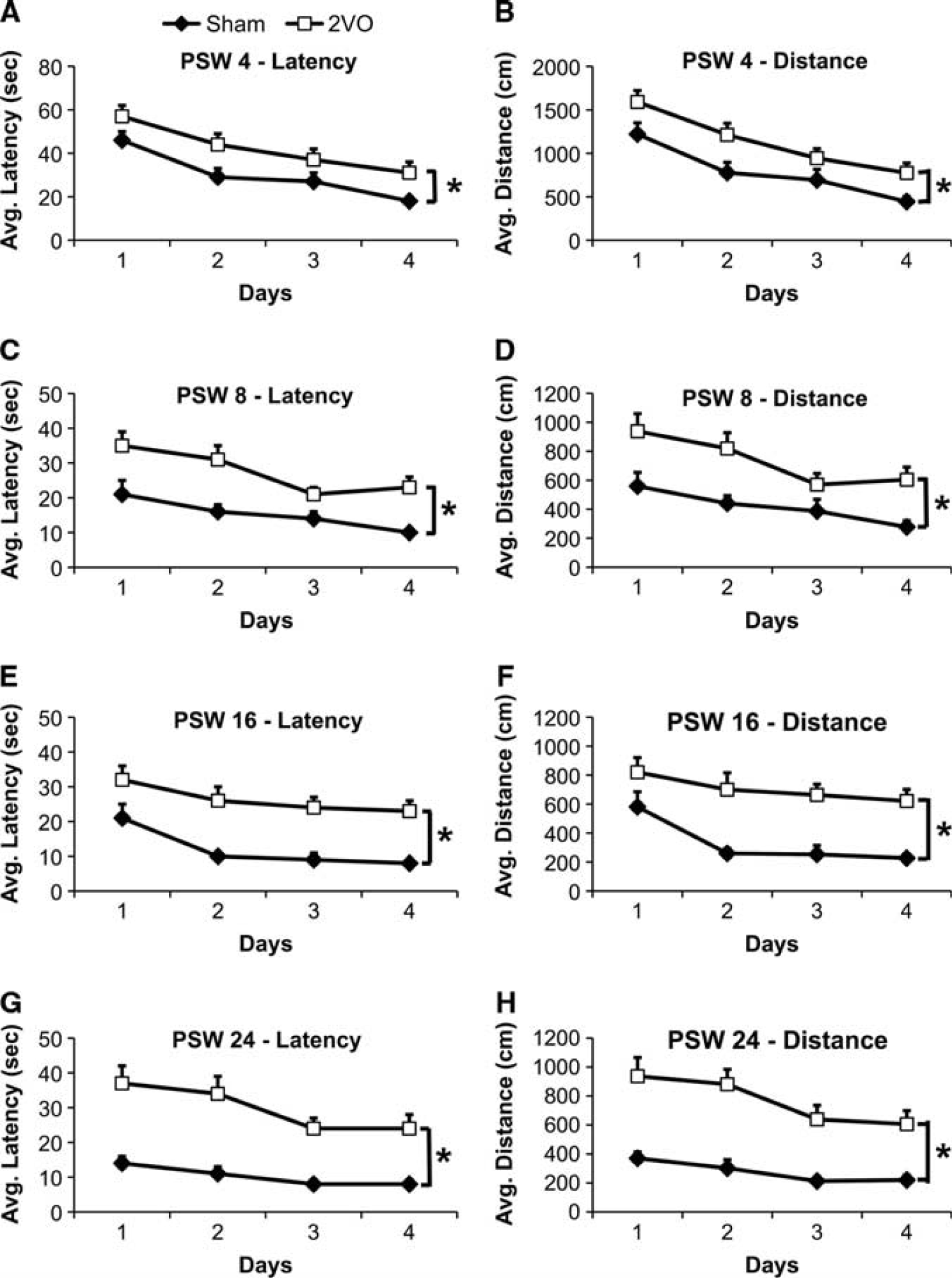
Morris Water Maze average (mean ± SEM) latencies (seconds) and distances (cm), respectively, to locate the hidden platform in Experiment 1 at postsurgery week (PSW) 4 (
Analysis of PSW 4 data (latency and distance, respectively) revealed that there was a significant effect of Time (F3,69 = 14.593, P < 0.01; F3,69 = 17.967, P < 0.01), Surgery (F1,23 = 7.749, P < 0.02; F1,23 = 11.510, P < 0.01), and Diet (latency, NS; F1,23 = 4.638, P < 0.05; data not shown) with respect to latency and distance traveled to locate the hidden platform (Figure 2; Supplementary Figure 1). With respect to the effect of Surgery, 2VO animals required a significantly longer latency and swam significantly longer path lengths to locate the hidden platform (Figures 2A and 2B, respectively). Further, animals exposed to the HFS diet had a decreased path length to locate the hidden platform at this time point only (Supplementary Figure 1).
The remaining test periods revealed similar results with respect to the dependent measures (latency and distance, respectively). At PSW 8, 16, and 24, there was a significant effect of Time (PSW 8: F3,69 = 7.608, P < 0.01; F3,69 = 6.144, P < 0.01) (PSW 16: F3,69 = 7.967, P < 0.01; F3,69 = 6.770, P < 0.01) (PSW 24: F2.445,56.235 = 5.347, P < 0.01; F2.635,60.615 = 4.612, P < 0.01) and Surgery (PSW 8: F1,23 = 13.830, P < 0.01; F1,23 = 12.268, P < 0.01) (PSW 16: F1,23 = 21.879, P < 0.01; F1,23 = 23.918, P < 0.01) (PSW 24: F1,17.317 = 32.322, P < 0.01; F1,18.920 = 41.665, P < 0.01), but no effect of Diet and no significant interactions (Figures 2C to 2H).
Further stratification of the data assessing animals' latency and path length within day 1 for each period revealed a significant effect of Time at PSW 4 (F3,69 = 6.660, P < 0.01; F3,69 = 15.828, P < 0.01) and a significant effect of Surgery at PSW 8 (F1,23 = 5.242, P < 0.04; F1,23 = 5.431, P < 0.03) and PSW 24 (F1,18.785 = 17.560, P < 0.01; F1,18.382 = 17.297, P < 0.01), where expectedly, 2VO animals required significantly more time and traveled longer distances to locate the hidden platform. There was no effect of Diet and no significant interactions at any time point.
With the exception of PSW 4, there was no significant effect of Diet (P > 0.05) and no interactions between independent variables on any of the dependent variables at any time point measured.
24-Hour and 1-week probes. There were no effects of Surgery or Diet and no significant interactions between the independent variables at PSW 4, 16, and 24 with respect to the amount of time spent in the target quadrant in either the 24-hour or 1-week probe trials. There were significant differences among conditions at PSW 8. At PSW 8, Sham animals spent significantly more time in the target quadrant than 2VO animals during the 24-hour probe trial (F1,23 = 6.468, P < 0.02). There was no effect of Diet and no interaction during this trial. During the 1-week probe test, there was no effect of Surgery or Diet but a significant Surgery × Diet interaction. Tukey's post hoc analysis failed to reveal the origin of this interaction.
Open field. Assessment of the number of grid crosses during the 5-minute trials at each PSW 5, 9, 17, and 25 revealed a significant effect of Time (F3,66 = 17.825, P < 0.01) but no effect of Surgery or Diet and no interaction between independent variables (Supplementary Figure 2).
Novel object recognition. A paired t-test at each time point showed that animals spent a similar amount of time exploring each of the objects during Trial 1 (P > 0.05). During Trial 2 (change in object location), paired t-test revealed that animals spent significantly less time exploring the object in the new location at PSW 6 (t25 = 2.503, P < 0.02) but similar amounts of time exploring both objects at PSW 10, 18, and 26 (P > 0.05). Similar analyses of Trial 3 data, however, revealed that animals spent significantly more time exploring the novel than the familiar object at each test period (PSW 6: t25 = 6.556, P < 0.01; PSW 10: t25 = 6.348, P < 0.01; PSW 18: t25 = 6.680, P < 0.01; PSW 26: t25 = 5.512, P < 0.01).
A 2 × 2 ANOVA of the time spent exploring the object in the new location (Trial 2) revealed that there was no effect of Surgery or Diet and no significant interactions at any time point (P > 0.05). Similarly, there was no effect of Surgery or Diet on the amount of time spent exploring the novel object in Trial 3 at any time point. There was, however, a significant Surgery × Diet interaction at PSW 18, but Tukey's post hoc test failed to reveal a significant difference among conditions. There were no other interactions at the other test periods (Supplementary Figure 3).
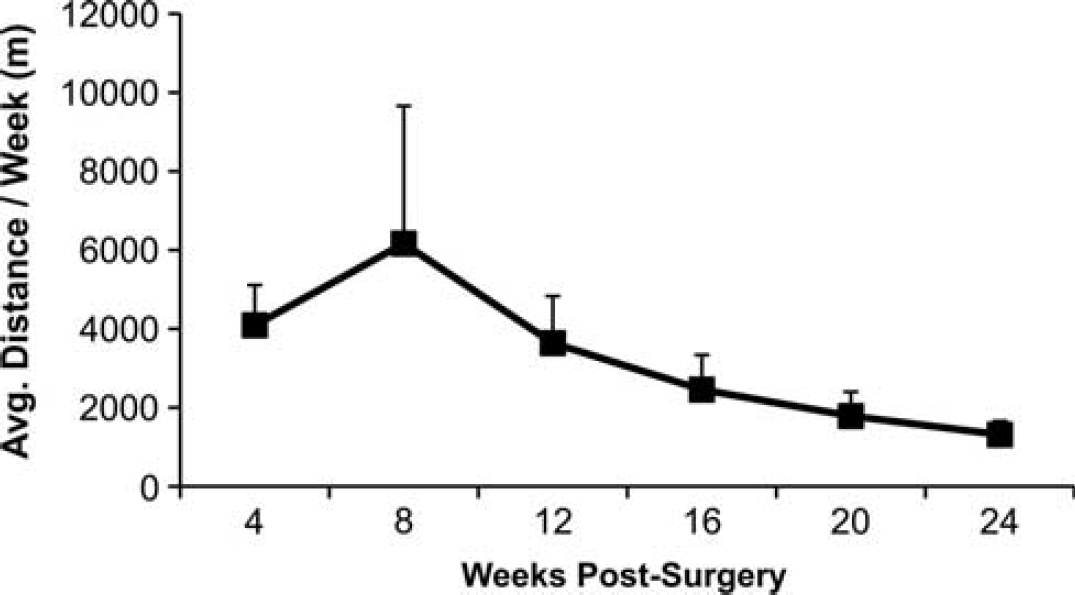
Average running distances in meters (mean ± SEM) of physical and cognitive activity (PA/CA) animals. PA/CA animals were exposed to voluntary running wheels 24 hours/day for 3 days/week. Data represent weekly running totals averaged over each month.
CA1 cell quantification and size estimate. A 2 × 2 ANOVA of hippocampal CA1 cell number estimates, cell soma area and volume failed to reveal an effect of Surgery or Diet and no significant interaction between variables (P > 0.05; Table 2).
Analysis of hippocampal CA1 cell number, area (μm2), and volume (μm3) (mean ± SEM)
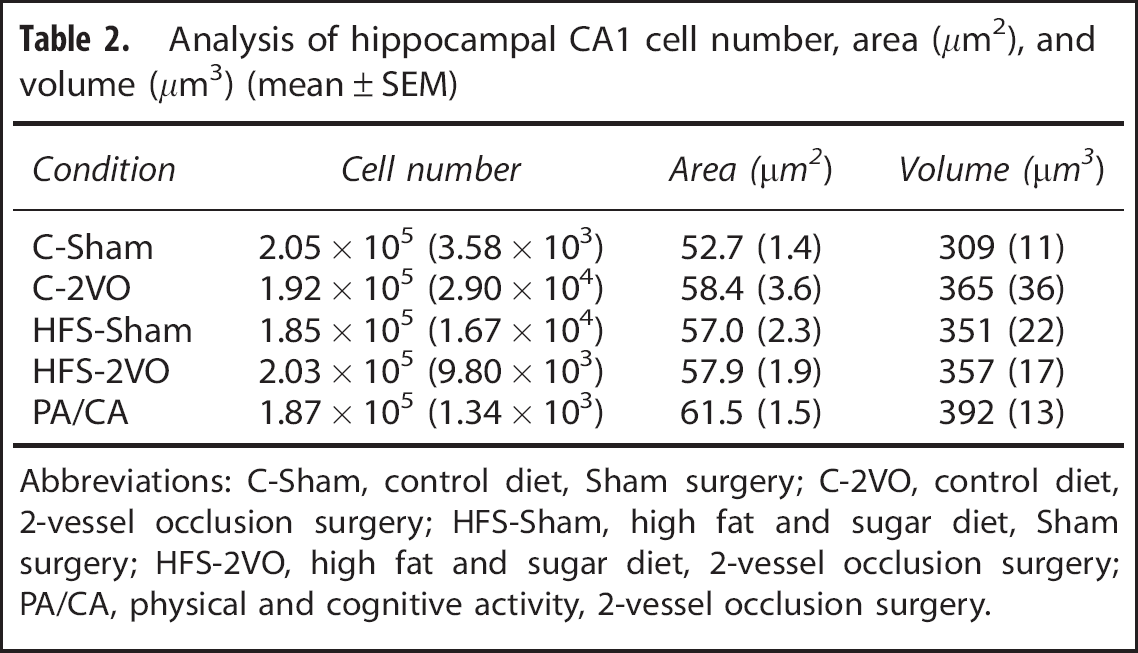
Abbreviations: C-Sham, control diet, Sham surgery; C-2VO, control diet, 2-vessel occlusion surgery; HFS-Sham, high fat and sugar diet, Sham surgery; HFS-2VO, high fat and sugar diet, 2-vessel occlusion surgery; PA/CA, physical and cognitive activity, 2-vessel occlusion surgery.
Experiment 2: Effects of Rehabilitation on Vascular Dementia Outcome
Due to the variable effects of the HFS diet on outcome measures in the behavioral assessments (Experiment 1), comparisons to assess rehabilitation efficacy were made in control diet conditions between 2VO (n = 7; in Experiment 1) and PA/CA + 2VO animals (n = 11). Animals' running data are presented in Figure 3.
Morris Water Maze
Visible platform. There were no significant differences between conditions with respect to latency to locate the visible platform at any of the time points assessed (P > 0.05).
Acquisition: hidden platform. Although 2VO significantly increased latency and path length to locate the hidden platform at all time points compared with Sham surgery (Experiment 1), there was no attenuation of learning and memory deficits in animals exposed to the rehabilitation paradigm. There was a significant effect of Time at each measurement (P < 0.05; indicative of learning) but there were no significant differences between 2VO-sedentary and 2VO + PA/CA animals with respect to latency (P > 0.05) or path length (P > 0.05) (Figure 4), nor was there a significant Time × Rehabilitation interaction to locate the hidden platform during the acquisition trials at any of the time points measured. Additionally, there were no differences between conditions with respect to the amount of time spent in the target quadrant during either the 24-hour or the 1-week probe trials at any of the time points measured (P > 0.05; data not shown).
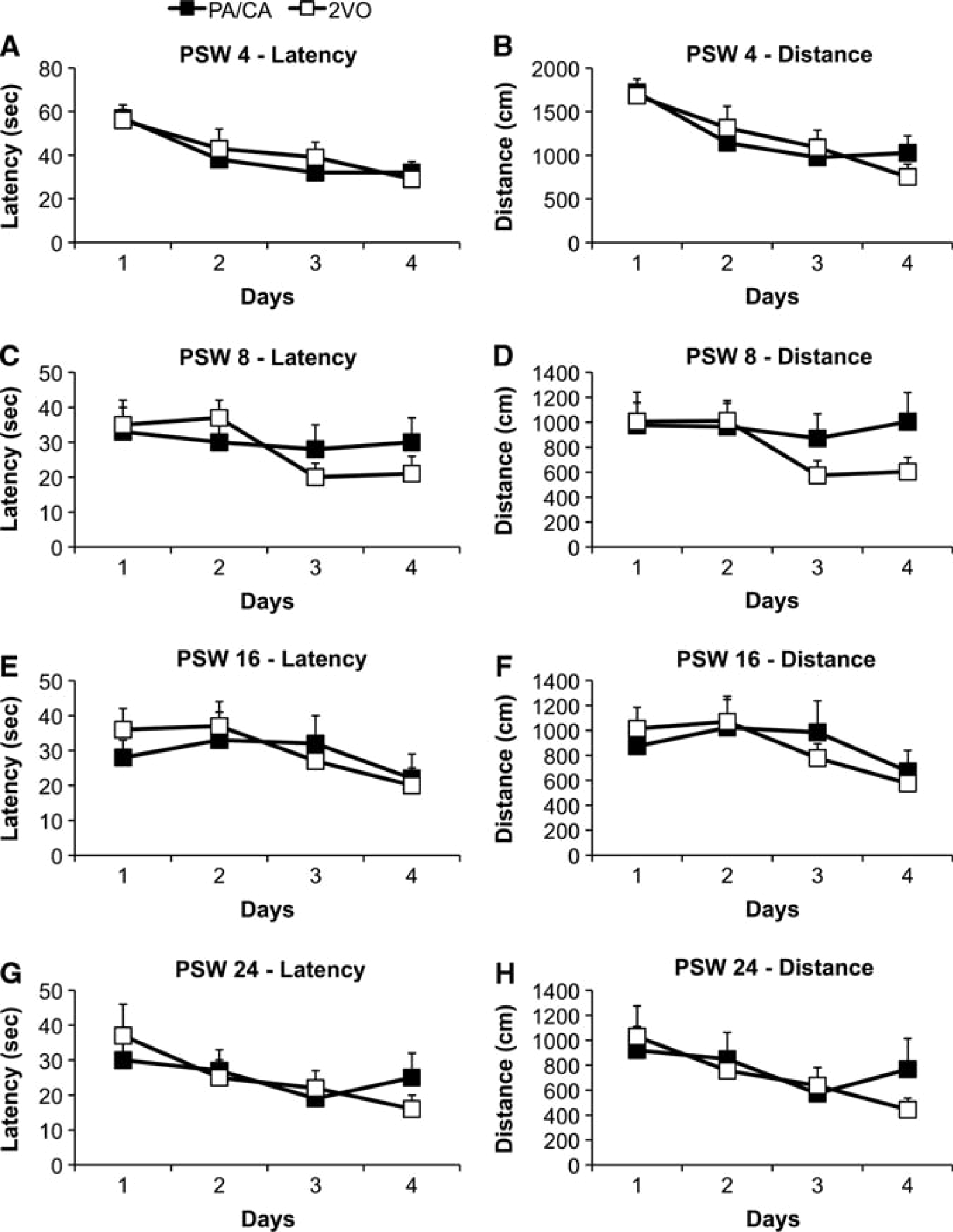
Morris Water Maze average (mean ± SEM) latencies (seconds) and distances (cm), respectively, to locate the hidden platform in Experiment 2 at postsurgery week (PSW) 4 (
Open field. There was a significant effect of Time (F3,48 = 4.115, P < 0.02) and Rehabilitation (F1,16 = 4.650, P < 0.05), but no significant interaction on the number of crosses made during the open field trials. Post hoc analysis revealed that, overall, 2VO + PA/CA animals made significantly more grid crosses than 2VO-sedentary animals.
Novel object recognition. A paired t-test at each time point revealed that there were no preferences for either object (i.e., Trial 1; P > 0.05). During PSW 6, animals spent significantly more time exploring the object in the familiar location than the object in the new location (t17 = 2.282, P < 0.04). There were no other differences in the amount of time exploring either object location at any other time point. During Trial 3 however (novel object), all animals spent significantly more time exploring the novel than familiar object (PSW 6: t17 = 7.093, P < 0.01; PSW 10: t17 = 4.678; PSW 18: t17 = 3.045, P < 0.01; PSW26: t17 = 7.396, P < 0.01).
An independent t-test at each time point revealed that there were no differences (P > 0.05) between conditions with respect to the amount of time exploring the object in the novel location (Trial 2) or the novel object (Trial 3).
CA1 cell quantification and size estimate. Independent t-tests showed that there were no statistical differences between 2VO-sedentary and 2VO + PA/CA animals with respect to the estimated cell number, soma area, and soma volume (P > 0.05; Table 2).
DISCUSSION
The current study assessed the consequences of chronic cerebral hypoperfusion on learning and memory abilities in early middle-aged female rats exposed to a diet rich in saturated fats and refined sugars, and the effects of PA/CA on the restoration of cognitive function after this insult. Our results confirm previous reports from this laboratory 3 and others 4 that older animals exposed to permanent, bilateral carotid occlusion exhibit significant learning and memory impairments. Further, data from the current study extend these findings to include early middle-aged female rats, a cohort of animals not commonly assessed in preclinical experimentation. Interestingly, cognitive rehabilitation, efficacious in a male model of VasD, 3 failed to ameliorate learning and memory impairments in female rats exposed to 2VO in the current study. Additionally, the 2VO model of VasD with female rats did not significantly affect hippocampal CA1 cell number or size as previously reported in male animals.3,19
The learning and memory deficits manifested early (4 weeks) after the vascular perturbation and persisted throughout the assessment period (~6 months) in the MWM. These data support the results from other studies of chronic hypoperfusion using male rats where the onset of cognitive deficits occurred early after surgery. 4 Although not statistically progressive in nature, visual inspection of Figure 2 suggests that cognitive dysfunction continues to worsen over time in hypoperfused animals, as previously proposed, 5 potentially reflecting the clinical pathologic progression of VasD. Further, we cannot definitively conclude that differences observed at 24 weeks after surgery solely reflect cognitive impairments because 2VO animals had significantly increased latencies to locate the visible platform compared with control animals at this time point. Additional mechanisms such as decreased visual acuity, 4 motivation, 20 or a combination of these with cognitive impairments may have influenced escape latency. However, in the study by Pappas et al, 4 visible platform trial deficits were noted to occur early after surgery and were transient in nature, not extending past 28 days after surgery. In contrast, all animals performed similarly in visible platform latencies during test periods at 4, 8, and 16 weeks after surgery and showed similar swim velocities, but 2VO animals had significantly longer latencies and path lengths to locate the hidden platform during acquisition trials. These data are more likely to reflect overt cognitive dysfunction as a result of chronic cerebral hypoperfusion.
It is important to further highlight the effects of cerebral hypoperfusion on learning and memory in the current study. Generally, relatively few preclinical studies use female subjects in models of VasD.21,22 To our knowledge, this is the first study to describe in detail, such robust cognitive deficits in a middle-aged female cohort of rats. Importantly, and in alignment with the STAIR recommendations,12,13 we have extended our results beyond the young, healthy male rat in the current study to include early middle-aged, OVX rats consuming an unhealthy diet and assessed during both the acute and long-term phases of chronic hypoperfusion. These results are particularly relevant because ~50% of the clinical population affected by VasD are women, with rates dramatically increasing with age. 1 Therefore, preclinical therapeutic interventions must show efficacy in both male and female animals to minimize the likelihood of translational failure.
Using the 2VO model of VasD, behavioral deficits were limited to spatial learning and memory as previously noted.3,4 Performance in the open field and novel object recognition was not affected. One explanation for this relatively specific deficit may involve the underlying brain circuitry involved. These tasks appear to require greater input from other hippocampal areas including CA3 and the dentate gyrus. 23 Further, changes in open field have been noted after overt CA1 cell loss,24,25 which was not shown in the current study.
The lack of rehabilitation efficacy observed in the current study is interesting, in light of the many studies reporting the positive effects of both PA9,26 and CA11,27 on brain health in preclinical and clinical studies. Notably, cognitive improvements have been shown in young, intact female mice exposed to voluntary wheel running, 28 young, ischemic, female gerbils exposed to environmental enrichment 29 and postmenopausal women who participated in a diversity of cognitive tasks (e.g., board games, cards, and puzzles). 10 The current rehabilitation paradigm consisted of a combination of PA/CA and was selected because of the synergistic effect showed in young naïve, intact male 2 and middle-aged hypoperfused rats. 3
The reasons why PA/CA failed to improve animals' learning and memory abilities in the current study are presently unclear. However, all animals in the current study were ovariectomized (OVX), which has been shown to significantly reduce endogenous estrogen levels 30 and subsequent running activity. 31 The middle-aged female rats in the current study ran similar distances as the young, healthy male animals used previously 2 that resulted in learning and memory enhancements. More importantly, both OVX 15 and aging 32 significantly reduce brain-derived neurotrophic factor (BDNF), especially throughout the hippocampus, an area integral for spatial learning and memory. Considerable evidence suggests that the beneficial cognitive effects of physical exercise are mediated, at least in part, by BDNF. 33 In adult male and female rodents, dentate gyrus long term potentiation (a biologic model of learning and memory 34 ) is increased by voluntary wheel running.35,36 However, this effect is abolished in adolescent female rats (postnatal day 35), 37 a time approximating vaginal opening and subsequent gonadal maturation onset, 38 thus corresponding to a time at which estrogen levels are thought to be low. Further, in a related study, 39 middle-aged, male mice did not exhibit higher levels of BDNF as did younger animals, when exposed to 1 month of voluntary running, reinforcing the notion of age-associated declines in neuroplasticity. 40 Replacement of estrogen in OVX animals significantly increases hippocampal BDNF levels to normal in several subregions (e.g., CA2 and dentate gyrus). 15 Perhaps hormone replacement therapies have a similar role in restoring the neuroplastic potential in postmenopausal women, especially when initiated early. 41 It is also the case that in rodent studies that used running 28 or environmental enrichment 29 to improve cognitive function, the access to running wheels or enrichment was unlimited (i.e., 7 days per week). Consequently, it may be that with ovariectomized middle-aged female rats it may be necessary to use more intense forms of PA/CA to offset the reduced levels of BDNF. Similarly, a more gradual hormonal reduction as occurs with humans instead of the relatively rapid depletion induced by OVX may also have provided an extended period by which rehabilitation efficacy may have been realized. Regardless, the data from the above studies provide insight into possible mechanisms that potentially mediate the differences in rehabilitation efficacy between male and female rats exposed to chronic hypoperfusion.
The dietary effects shown in the current study are difficult to interpret. There are reports that diets high in saturated fats exacerbate overt ischemic damage, 42 but see. 43 Previously, we reported that a diet high in saturated fats and refined sugars (HFS) did not exacerbate functional or histopathologic deficits/damage in chronic cerebral hypoperfusion in male rats. 3 In the current study, however, the HFS diet appeared to have an equivocal influence on cognition and VasD outcomes. This effect was an unexpected, relatively minor decrease in path length during the acquisition trials at week 4 in animals exposed to the HFS diet. There were several additional interactions, but due to the small effect size, post hoc analyses failed to reveal differences. One might conclude from these data that a diet high in saturated fats and refined sugars may affect cognition, especially when combined with cerebrovascular perturbations in female rats but this possibility requires further study.
Contrary to previous reports,3,19 there were no detectable changes in hippocampal architecture noted in the current study. Because identical surgical, behavioral and histologic measures were employed in the current and our earlier study with male animals, 3 it would appear that there are fundamental differences in female and male rats' response to chronic hypoperfusion. Although chronic 2VO generally does not result in focal damage outside the hippocampal region, 44 changes in white matter connectivity may have contributed to cognitive dysfunction noted in the current study. 45
In conclusion, we report significant cognitive impairments in early middle-aged, OVX rats exposed to chronic cerebral hypoperfusion, a finding consistent with results from other laboratories using male animals.4,5 A rehabilitative intervention, consisting of a combination of PA/CA, failed to attenuate these impairments although the same intervention was effective in male rats. 3 These findings are important, in view of the many preclinical stroke interventions that have failed in the clinical population. Translational roadblocks continue to be problematic and the causes of such problems require that preclinical experimentation capture key elements (e.g., age, diet, and sex) of the clinical population as attempted in the current study.12,13
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors thank Dr John Evans, Garry Chernenko, Kathy McKay, Suzanne Evans, and Andrew Orsborne for their technical assistance.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
