Abstract
Extensive research over the last two decades has advanced our understanding of the pathophysiology of ischemic stroke. However, current pharmacologic therapies are still limited to rapid reperfusion using thrombolytic agents, and neuroprotective approaches that can reduce the consequences of ischemic and reperfusion injury, are still not available. To bridge this gap, we have evaluated the long-term efficacy and therapeutic time window of a novel peptide-based neuroprotectant TAT-STEP, derived from the brain-enriched and neuron-specific tyrosine phosphatase STEP. Using a rat model of transient middle cerebral artery occlusion (90 min), we show that a single intravenous administration of the peptide at the onset of reperfusion (early) or 6 h after the onset of the insult (delayed) reduces mortality rate. In the surviving rats, MRI scans of the brain at days 1, 14 and 28 after the insult show significant reduction in infarct size and improvement of structural integrity within the infarcted area following peptide treatment, regardless of the time of administration. Behavioral assessments show significant improvement in normal gait, motor coordination, sensory motor function and spatial memory following early or delayed peptide treatment. The study establishes for the first time the therapeutic potential of a tyrosine phosphatase in ischemic brain injury.
Introduction
Cerebral stroke is the fourth largest cause of death and a leading cause of disability in developed countries. 1 About 87% of all strokes are ischemic that occurs as a result of a thrombotic or embolic occlusion of a cerebral artery causing brain infarction, if not resolved early. 2 For more than two decades, extensive research efforts using both transient and permanent ischemic stroke models have shed light on important mechanisms of ischemic brain damage and identified multiple potential targets for stroke therapy. Despite these tremendous efforts in understanding the pathophysiology of ischemic stroke, successful treatment remains a major challenge in clinical medicine. Reperfusion with recombinant tissue plasminogen activator remains the only pharmacologic therapy.3,4 In a small number of cases, surgical recanalization is also possible.5,6 However, rapid reperfusion, although necessary for restoration of brain metabolic activity, is also associated with additional risks.7–10 The development of neuroprotection strategies to protect neurons from both ischemia and reperfusion injury, and to amplify the time window for thrombolytic treatment therefore remains a high priority. 11
Emerging evidence indicates that an ischemic insult not only triggers multiple cytotoxic pathways, but also triggers some endogenous protective responses capable of limiting injury. 12 We have identified the striatal-enriched phosphatase, STEP (encoded by the Ptpn5 gene) as one such protein, which is a component of the N-methyl-D-aspartate (NMDA) receptor-mediated excitotoxic signaling pathway that is known to play a central role in the progression of ischemic brain damage.13–17 Our earlier findings have shown that rapid activation of STEP following a transient ischemic insult (middle cerebral artery occlusion; MCAO 90 min) provides initial neuroprotection, while degradation of active STEP over time leads to loss of its neuroprotective effects, thus allowing the activation of deleterious processes.18,19 Consistent with these findings, studies in STEP knockout mice have also shown that deletion of the STEP gene exacerbates ischemic brain injury and neurological deficits following a mild ischemic insult. 19 Based on these observations, we developed a cell-permeable STEP-derived peptide that is resistant to degradation and established that it can cross the blood–brain barrier. 19 We further demonstrated that intravenous administration of the peptide even after the onset of reperfusion is effective in limiting the acute stroke injury at 24 h in both rat and mice models of MCAO. 19 The efficacy of the peptide in the post-stroke treatment paradigm suggests that this approach may be clinically relevant.
The apparent failure of a large number of therapeutic agents in clinical trials has now led to the development of the Stroke Therapy Academic Industrial Roundtable (STAIR) guidelines for evaluation of potential candidate drugs.20–22 One of the recommendations of the STAIR emphasizes on long-term evaluation of infarct size and functional outcome to ensure that early treatment effects are not transient. STAIR also recommends evaluating the most effective intervention time window for understanding the potential benefit of this approach in clinical trials. With these recommendations in mind, the current study sought to evaluate the effect of delayed intervention with the STEP-derived peptide in reducing infarct size and functional deficits over an extended post-stroke period (up to 28 days after MCAO). Infarct size was evaluated at repeated intervals to assess early and late manifestations of ischemic brain injury using the non-invasive magnetic resonance imaging (MRI) approach that is regularly used for evaluating clinical outcome in stroke patients. Functional recovery over time was assessed using a battery of motor, sensory and cognitive tests to determine how the stroke lesions evaluated by MRI compares with behavioral deficits. Validating the efficacy of this potential neuroprotectant through simultaneous and longitudinal evaluation of infarct size and functional outcome could help overcome the translational disparity between preclinical and clinical outcomes.
Materials and methods
Construction and purification of the STEP-derived peptide TAT-STEP-myc
A recombinant DNA construct for TAT-STEP-myc peptide was generated using a bacterial expression vector, expressed in E. coli and purified as described previously. 17 A brief summary of the method has been provided in the “supplementary methods” section.
MCAO and peptide treatment
Male Wistar rats (290–295 gm, eight to nine weeks) were obtained from Envigo (Placentia, CA). The animal studies were approved by the Institutional Animal Care and Use Committee and conducted in the AAALAC accredited animal facility of University of New Mexico, Health Sciences Center. The studies were conducted in accordance with the National Institutes of Health’s Public Health Service Policy. The ARRIVE guidelines were followed for the preparation of the manuscript. Focal cerebral ischemia was induced in all the animals by MCAO using the intraluminal method,19,23 as described in details in the “supplementary methods” section.
Magnetic resonance imaging
MRI scans were performed on 1, 14 and 28 days after MCAO using a 4.7-Tesla, 40 cm bore actively shielded Bruker biospec magnet system (Bruker Biospin, Billercia, MA, USA), equipped with 660 mT/m (rise time within 120 µs) gradient set for small animal imaging. 24 To obtain good signal-to-noise ratio, a small-bore linear radio frequency volume coil (inner diameter = 72 mm) was used for signal excitation, and a 2.5 cm single tuned surface coil (RAPID Biomedical, Rimpar, Germany) was used for signal detection. For the MRI scans, rats anesthetized with isoflurane (2%) in oxygen were placed in a belly down position in a secured cradle positioned in the isocenter of the magnet. During MRI, the rats were maintained under anesthesia, body temperature was maintained at 37.0 ± 0.5℃, and respiration and heart rate were monitored continuously. Initial localizer images were acquired using a two-dimensional (2D) fast low-angle shot sequence with repetition time/echo time 100 ms/6 ms, field of view 8 cm × 8 cm, matrix 128 × 128, and one slice per orientation. To assess brain anatomy, a fast spin echo T2-weighted imaging (T2WI) sequence was then employed using the following parameters: 2D rapid acquisition with relaxation enhancement, repetition time/echo time 5000 ms/56 ms, field of view 4 cm × 4 cm, slice thickness 1 mm, inter-slice gap 1.1 mm, number of slices 12 spanning coronal brain sections from olfactory bulb through hippocampus, matrix 256 × 256, number of averages 1. To assess diffusion abnormalities, T2WI was followed by acquisition of diffusion-weighted echo planar imaging using the following parameters: repetition time/echo time 3800 ms/38 ms, b-values 600 and 1900 s/mm2 in 30 directions, field of view 4 cm × 4 cm, slice thickness 1 mm, number of slices 12, matrix 256 × 256, number of averages 3. The MRI raw data were processed to generate T2 maps from T2-weighted images and apparent diffusion coefficient (ADC) maps from the diffusion tensor images. All MRI scans were performed at the preclinical MRI core located in the Brain & Behavioral Health Institute of UNM Health Sciences Center. The MRI scans were used to calculate ischemic lesion volume as well as ADC and FA values as described in details in the “supplementary methods” section.
Functional assessments
Male Wistar rats treated with vehicle or peptide were subjected to neurological assessment at 24 h after MCAO and a battery of behavioral tests (CatWalk, cylinder, rotarod, adhesive and Morris water maze test) on 7, 14, 21 or 28 days after MCAO to evaluate normal gait, motor-coordination, sensory motor and cognitive functions. On the days where MRI and behavioral studies overlapped, MRI scans were performed after completion of behavioral studies. Habituation to the testing environment and baseline training was performed one week before surgery. An observer blinded to the study groups and treatment conditions evaluated all behavioral parameters after surgery. A detailed description of the behavioral methods has been included in the “supplementary methods” section.
Histology
After repeated MRI and behavioral tests rats were rapidly anesthetized on day 28 after MCAO, perfused intracardially with ice-cold 4% paraformaldehyhde in 0.01 M phosphate-buffered saline (PBS) and the brains were processed for cresyl violet staining. A brief summary of the method has been described in the “supplementary methods” section.
Statistical analysis
Statistical differences between multiple groups were assessed using repeated measures analysis of variance (ANOVA) with vehicle versus peptide-treated groups as between-group factor and days post-stroke as repeated factor. Bonferroni method was used to correct for multiple comparisons (SPSS 24.0 software). A priori power calculation determined the minimum number of rats per group required to achieve >80% power assuming an effect size of 0.75 between two groups in the repeated measures ANOVA was determined to be at least six (G*power 3.1.9.2). The a priori minimum sample size was calculated based on the estimated variance of adhesive test (early peptide treatment) as this test shows the largest variance. Analysis of simple two-group comparison for normally distributed data was done using the Student’s t-test. All t-test data in the text and figures are expressed as mean ± SD (standard deviation). Mean differences between two groups were considered statistically significant when p < 0.05. Pearson’s r was used for effect size calculation for all t-test data. 25 Neurological severity score data that were not normally distributed were analyzed using non-parametric Mann–Whitney test and reported as median with interquartile range. In this exploratory study, all statistical analysis used two-sided tests with no adjustment for multiple comparisons.
Results
Early treatment with the STEP-derived peptide at the onset of reperfusion promotes survival and reduces ischemic brain damage
To evaluate the effect of the STEP-derived peptide in attenuating ischemic brain damage, in initial studies, male Wistar rats were subjected to 90 min MCAO and treated with a single intravenous injection of vehicle or peptide at the onset of reperfusion (early treatment) as outlined in Figure 1(a). At 24 h post-MCAO, development of brain lesion was assessed from T2 maps acquired from MRI scans. The representative T2 maps from both vehicle- and peptide-treated rats show distinctly demarcated regions of increased T2 signal intensity (hyperintense area) indicating ischemic lesions, which are larger in size on images acquired from vehicle-treated rats (Figure1(b)). The scatter plot data illustrating the extent of lesion in individual rats as well as the quantitative measurement of the mean lesion volume (Figure1(c)) from T2 maps show significantly smaller lesion size in peptide-treated rats compared with vehicle-treated controls (vehicle 44.6 ± 6.4% vs. peptide 26.5 ± 14.6%, p < 0.001, r = 0.7). Assessment of neurological function from the neurological severity score using Mann–Whitney U test (Figure1(d)) shows significantly lower neurological impairment in peptide-treated group when compared to the vehicle-treated controls (U = 44, p = 0.029).
Effect of early peptide treatment on ischemic brain injury at 24 h post-occlusion evaluated from T2 maps. (a) Schematic representation of the timeline of MCAO, reperfusion, administration of the TAT-STEP-myc peptide, MRI scans and behavioral assessments. (b) Representative T2 maps acquired from vehicle- and peptide-treated rats illustrating the spatial distribution of brain infarct at 24 h after the onset of ischemia. (c) Ischemic lesion volume in vehicle- and peptide-treated rats represented as mean ± SD with individual values in a scatter plot (vehicle: n = 11, peptide: n = 9). (d) Post-ischemic neurological severity score in vehicle- and peptide-treated rats was assessed 24 h after MCAO on a 5-point scale, and represented as individual data points along with median value and limits of interquartile range. *p < 0.05 and **p < 0.001 for vehicle- versus peptide-treated group.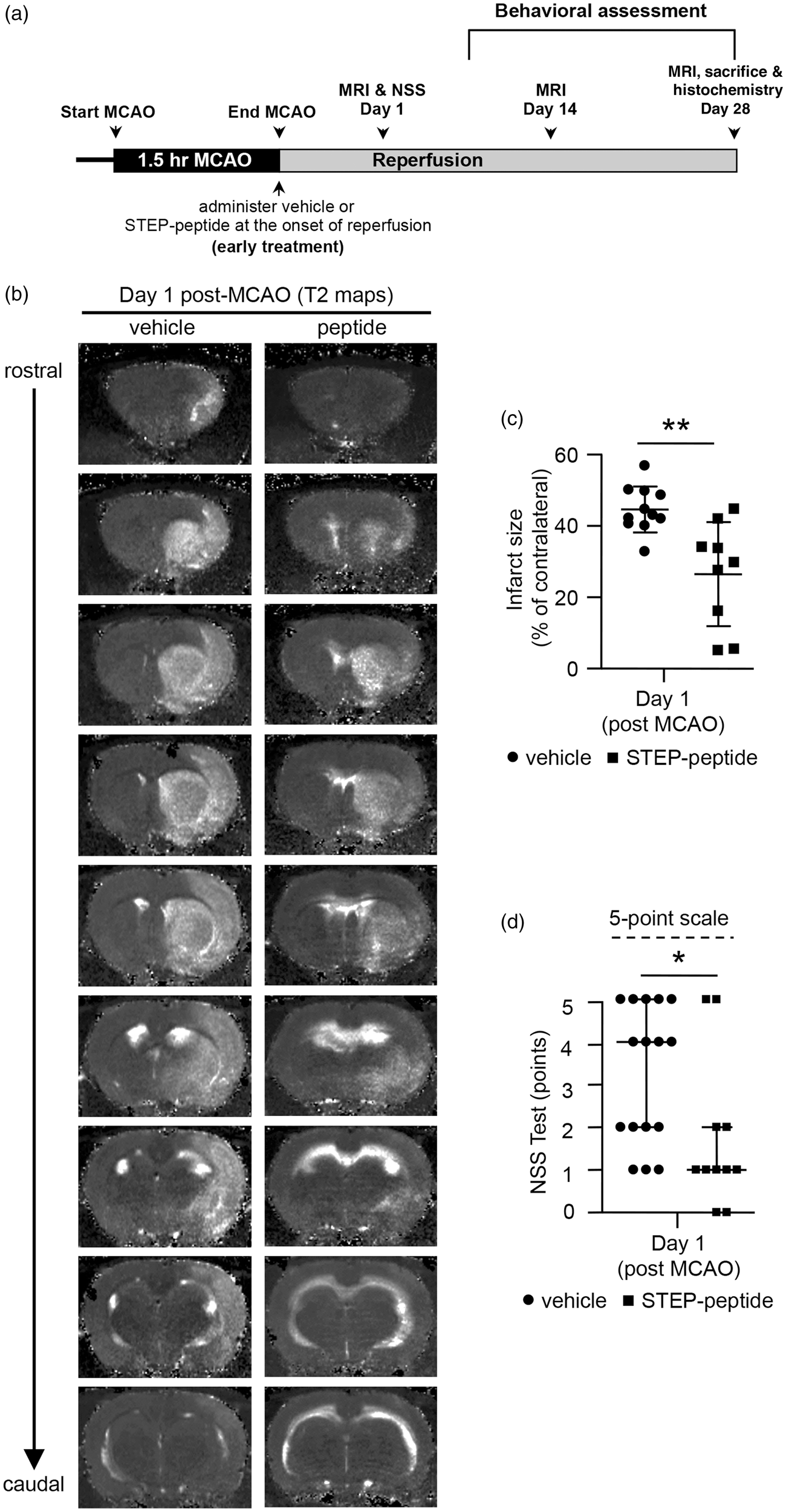
Longitudinal evaluation of the ischemic lesion volume up to 28 days post-MCAO shows significant group effect [F (1, 17) = 12.446, p = 0.003] and day effect [F (1.523, 25.89) = 49.447, p < 0.001] between vehicle- and peptide-treated groups (Greenhouse-Geisser correction was applied as Maulchy’s sphericity test failed). The representative T2 maps and quantitative measurement of lesion volume from the T2 maps (Figure 2(a) and (b)) show significantly smaller lesion volume in the peptide-treated group at both 14-day (vehicle 33 ± 6.9% vs. peptide 18.6 ± 13.8%, p < 0.001, r = 0.6) and 28-day (vehicle 31.5% ± 6.6 vs. peptide 16.2% ± 10.5, p = 0.001, r = 0.7) post-MCAO. Evaluation of stroke-induced mortality in the two groups shows that overall mortality within the first two days after stroke is higher in the vehicle-treated group (n = 6 out of 16) as compared to the peptide-treated group (n = 2 out of 11). However, after the first two days, none of the animals at risk died during the rest of the study period in either the vehicle- or peptide-treated groups, as evident from the long-term survival curve (Figure 2(c)). Thus, it appears that the trend of larger mortality in the vehicle-treated group is not consistent across the time period of the study (log rank test p = 0.295). However, a hazard ratio of 2.3 ([95% C.I.], 0.48–11.2) indicate that the estimated relative risk of mortality in the vehicle-treated group is 2.9 times higher in the first two days after stroke, when compared to peptide-treated group. For histopathological evaluation of infarct size, brain slices from both vehicle- and peptide-treated rats were processed for cresyl violet (Nissl stain) staining at 28 days post-MCAO, following the completion of the MRI study. The representative photomicrographs presented in Figure 2(d) show a unilateral hypertrophy in the cresyl violet stained brain sections and the area of histopathological damage corresponds to the area of injury detectable on T2 maps on day 28 post-MCAO (Figure 2(a)).
Effect of early peptide treatment on the temporal evolution of ischemic brain injury and tissue integrity evaluated from T2, ADC and FA maps. (a) Representative T2 maps acquired from vehicle- and peptide-treated rats on day 1, 14 and 28 post-MCAO showing time-dependent changes in ischemic lesion size from rostral to caudal regions of the brain. (b) Quantitative analysis of lesion volume on day 1, 14 and 28 after MCAO from vehicle- and peptide-treated rats, represented as mean ± SD (on day 1 – vehicle: n = 11, peptide: n = 9; on days 14 and 28 – vehicle: n = 10, peptide: n = 9) with individual values in a scatter plot. (c) Kaplan–Meier graph of cumulative mortality rate in vehicle- and peptide-treated groups followed for 28 days. The numbers below the figures denote the number of animals at risk in each group at the given time point (V = vehicle-treated group; P = peptide-treated group). (d) Representative cresyl violet stained images of rostral to caudal regions of the brain from vehicle- and peptide-treated rats acquired 28 days after MCAO. (e) Representative maps of apparent diffusion coefficient (ADC) acquired from vehicle- and peptide-treated rats at day 28 post-MCAO, featuring hyperintense areas that co-localize with the lesion area observed in the T2 maps at day 28 post-MCAO. Corresponding scatter plot showing quantitative analysis of the ADC values in the lesion area, expressed as mean ± SD (vehicle: n = 10, peptide: n = 9). (f) Representative fractional anisotropy (FA) maps acquired from the same slices as ADC and T2 maps at 28 days post-MCAO. Scatter plot data and quantitative analysis of the FA values expressed as mean ± SD (vehicle: n = 10, peptide: n = 9). *p < 0.05 and **p < 0.01 and ***p < 0.001 for vehicle- versus peptide-treated group.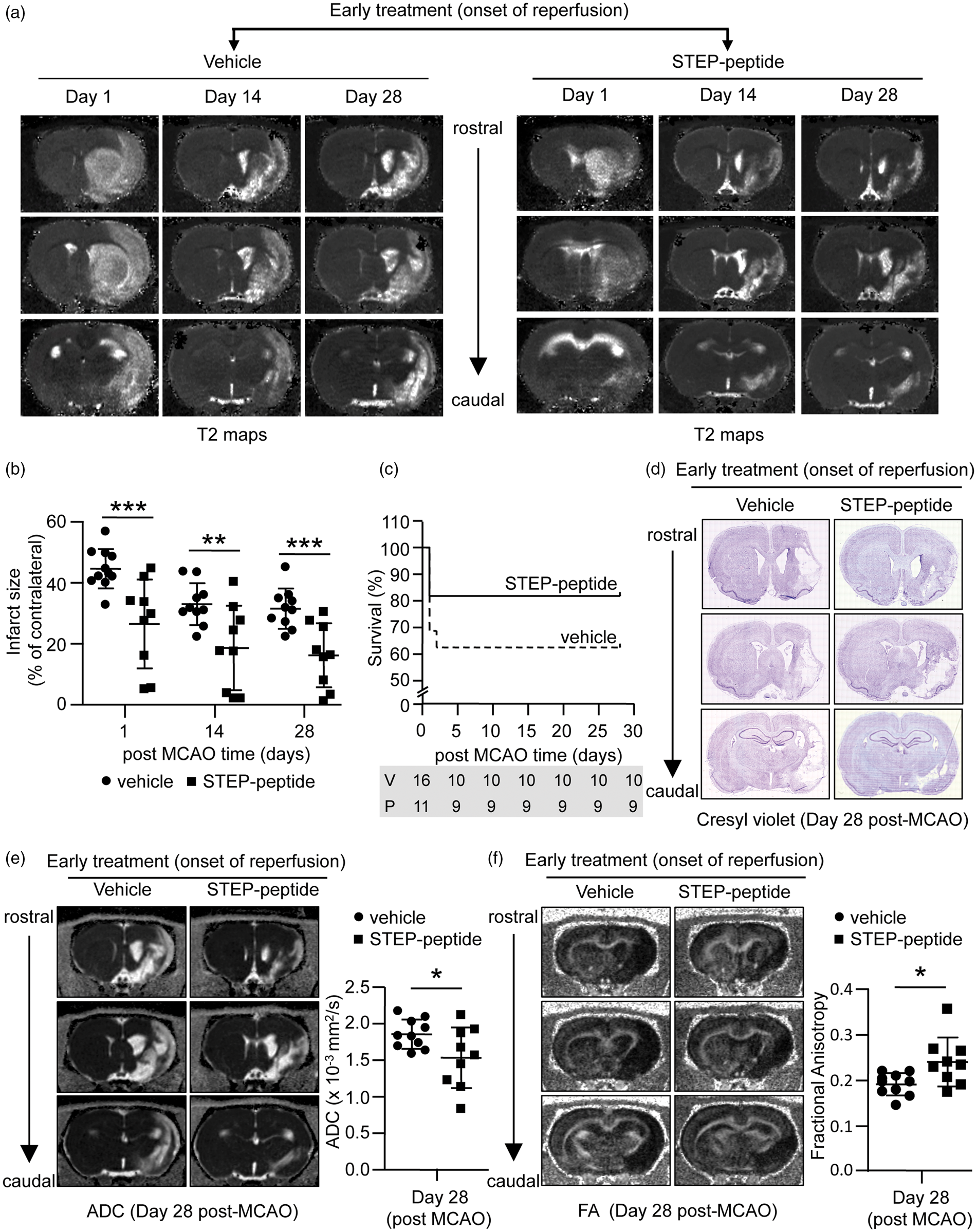
To further evaluate whether treatment with the STEP-derived peptide has an effect on MCAO-induced loss of structural integrity in the damaged area of the ischemic brain, we performed diffusion tensor MRI (DTI) on both vehicle- and peptide-treated groups. Changes in DTI parameters ADC and FA in the damaged area of the brain were measured 28 days after MCAO. The representative ADC maps and voxel-based analysis show that ADC values are significantly lower in the peptide-treated group compared with the vehicle-treated group (vehicle 1.9 × 10−3 ± 0.2 × 10−3 mm2/s vs. peptide 1.5 × 10−3 ± 0.41 × 10−3 mm2/s; p = 0.04, r = 0.5), indicating a decrease in mean diffusion of water following peptide treatment (Figure 2(e)). On the other hand, the significantly higher FA value (vehicle 0.19 ± 0.02 vs. peptide 0.24 ± 0.05; p = 0.02, r = 0.5) observed in the peptide-treated group as compared to the vehicle-treated group (Figure 2(f)) reflects increased directional water diffusion.
Delayed treatment with the STEP-derived peptide 6 h after the onset of the insult promotes survival and attenuates ischemic brain injury
The efficacy of the STEP-derived peptide in a post-stroke treatment paradigm and long-term recovery raises the possibility that STEP is a potential new target for therapeutic intervention. While designing such novel neuroprotective strategies, it is important to consider the effectiveness of intervention when administered at later time points. To determine whether delayed treatment with the peptide could facilitate long-term reduction in ischemia-induced brain damage, in subsequent experiments, rats were subjected to 90 min MCAO followed by reperfusion. A single intravenous injection of vehicle or peptide was administered 6 h after the onset of the insult (Figure 3(a)). The representative T2 maps, scatter plot data of the infarct size in individual rats and quantitative analysis of the mean lesion volume show that lesion volume is significantly lower in peptide-treated group when compared with the vehicle-treated group (vehicle 43.9 ± 10.8% vs. peptide 19.9 ± 9.3%, p < 0.001, r = 0.8) at 24 h post-MCAO (Figure 3(b) and (c)). A significantly lower neurological severity score is also observed in the peptide-treated group at 24 h post-MCAO (U = 20; p < 0.001; Figure 3(d)), indicating improved overall neurological function.
Effect of delayed peptide treatment on ischemic lesion size at 24 h post-occlusion evaluated from T2 maps. (a) Schematic representation of the timeline of MCAO, reperfusion, administration of the TAT-STEP-myc peptide, MRI scans and behavioral assessments. (b) Representative T2 maps acquired from vehicle- and peptide-treated rats illustrating the spatial distribution of brain infarct 24 h after the onset of ischemia. (c) Ischemic lesion volume in vehicle- and peptide-treated rats represented as mean ± SD with individual values in a scatter plot (vehicle: n = 10, peptide: n = 10). (d) Post-ischemic neurological severity score assessed at 24 h after MCAO and represented as individual data points along with median value and limits of interquartile range. *p < 0.001 for vehicle-versus peptide-treated group.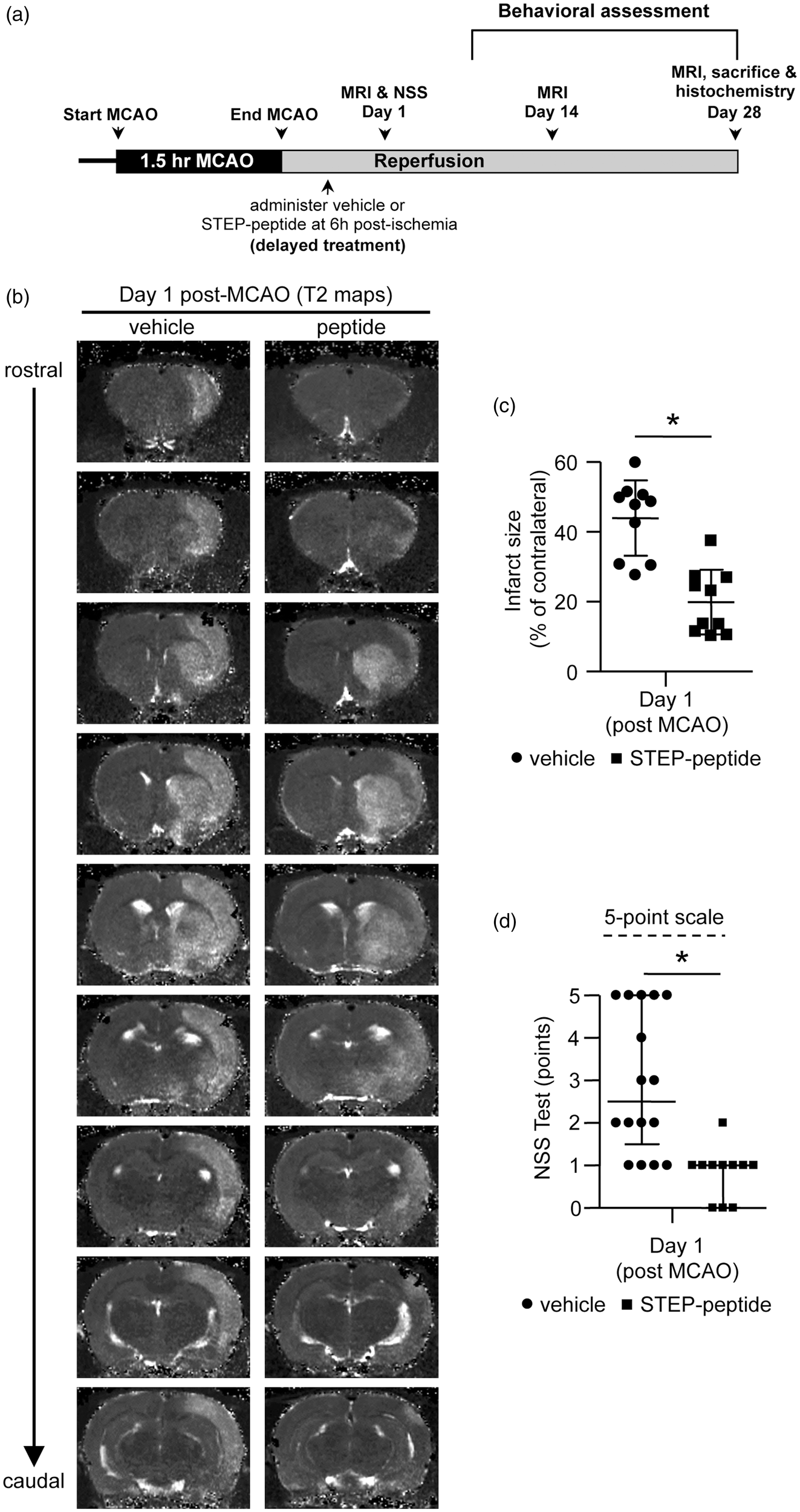
Longitudinal evaluation of brain infarct size up to 28 days post-MCAO further shows significant group difference [F (1, 16) = 14.61, p = 0.002] and day effect [F (2,32) = 44.68, p < 0.001] between the vehicle- and peptide-treated groups (Figure 4(b)). The representative T2 maps and post hoc analysis of the lesion volumes from T2 maps further show that peptide-treated group exhibit significantly lower infarct size at both 14-day (vehicle 31.0 ± 7.7% vs. peptide 14.1 ± 10.1%, p < 0.001, r = 0.7) and 28-day (vehicle 26.3 ± 11.2% vs. peptide 13.1 ± 11.5%, p = 0.025, r = 0.5) post-MCAO, when compared to vehicle-treated controls (Figure 4(a) and (b)). Delayed peptide treatment also results in lower mortality rate in the first two days after stroke when compared to vehicle-treated control group (vehicle; n = 6/16 versus peptide; n = 1/11). However, none of the animals at risk in the two groups died during the rest of the study period (Figure 4(c); a hazard ratio of 3.8 [95% C.I.], 0.78–18.8; log rank test p = 0.099). Cresyl violet staining of brain sections at 28-day post-MCAO further confirms that the area of the histopathological damage correlates with the area of the injury detected by MRI T2 maps acquired 28 days after MCAO (Figure 4(d)). Evaluation of the structural integrity of the brain tissue in the infarcted area by DTI further shows a significantly lower ADC value (vehicle 1.9 × 10−3 ± 0.43 × 10−3 mm2/s vs. peptide 1.4 × 10−3 ± 0.23 × 10−3 mm2/s; p = 0.013, r = 0.6) and higher FA value (vehicle 0.21 ± 0.02 vs. peptide 0.28 ± 0.05; p = 0.002, r = 0.7) in the peptide-treated group when compared with the vehicle-treated group (Figure 4(e) and (f)), suggesting a decrease in the magnitude of water diffusion but an increase in the directional diffusion of water.
Effect of delayed peptide treatment on the temporal evolution of ischemic brain injury and tissue integrity evaluated from T2, ADC and FA maps. (a) Representative T2 maps acquired from vehicle- and peptide-treated rats on day 1, 14 and 28 after MCAO showing time-dependent changes in ischemic lesion size from rostral to caudal regions of the brain. (b) Quantitative analysis of changes in lesion volume in vehicle- and peptide-treated rats on day 1, 14 and 28 after MCAO, represented as mean ± SD (on day 1 – vehicle: n = 10, peptide: n = 10; on days14 and 28 – vehicle: n = 9, peptide: n = 9). (c) Kaplan–Meier graph of cumulative mortality rate in vehicle- and peptide-treated groups followed for 28 days. The numbers below the figures denotes the number of animals at risk in each group at the given time point (V = vehicle-treated group; P = peptide-treated group). (d) Representative cresyl violet stained images of rostral to caudal regions of the brain acquired from vehicle- and peptide-treated rats 28 days after MCAO. (e) Representative ADC maps acquired from vehicle- and peptide-treated rats at day 28 post-MCAO, featuring hyperintense areas that co-localize with the lesion area observed in the T2 maps at day 28 post-MCAO. Corresponding scatter plot showing quantitative analysis of the ADC values in the lesion area, expressed as mean ± SD (vehicle: n = 9, peptide: n = 9). (F) Representative FA maps acquired from the same slices as ADC and T2 maps at 28 days post-MCAO. Scatter plot data and quantitative analysis of FA values expressed as mean ± SD (vehicle: n = 9, peptide: n = 9). *p < 0.05, **p < 0.01 and ***p < 0.001 for vehicle- versus peptide-treated group.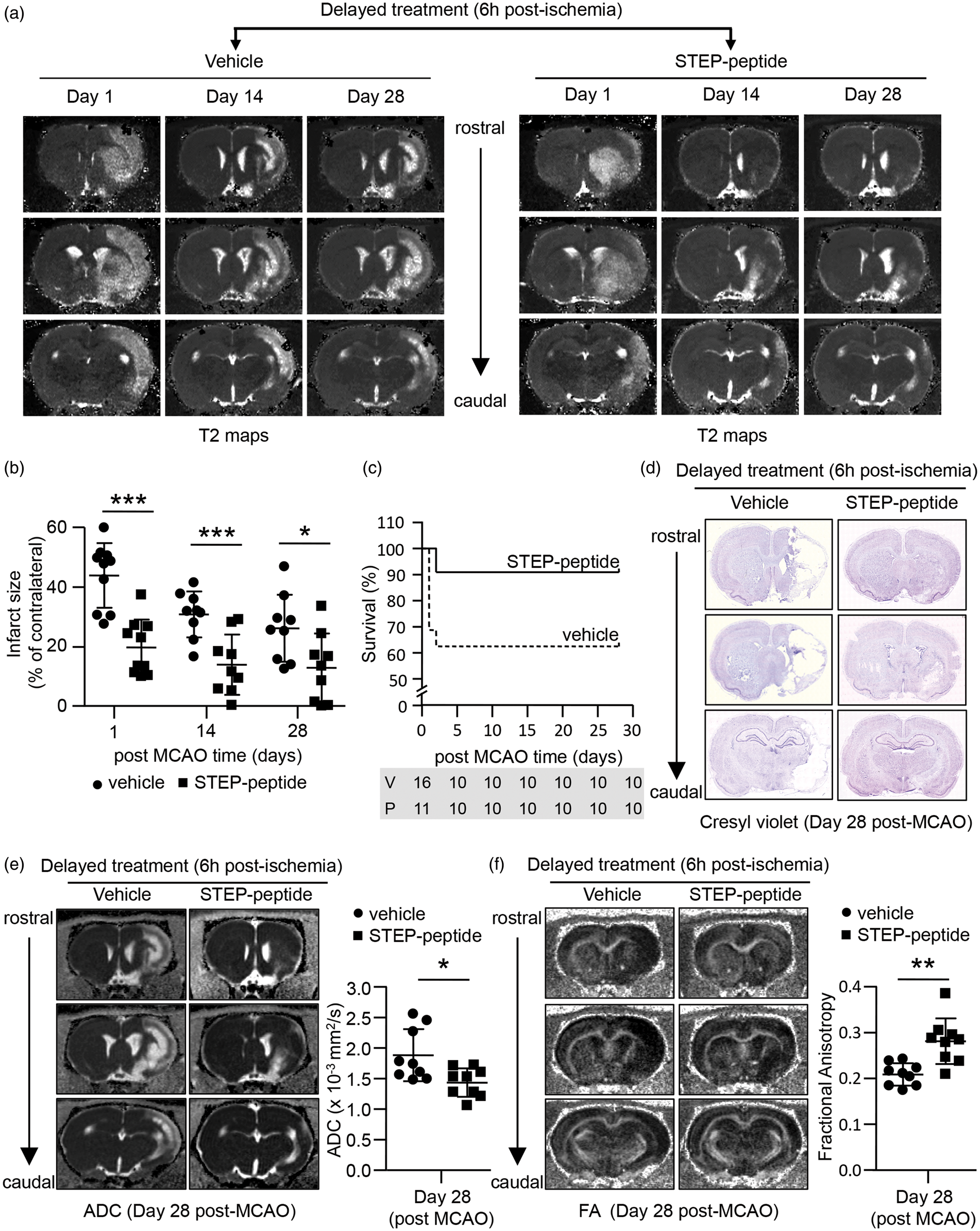
Both early and delayed treatment with the STEP-derived peptide facilitates functional recovery from ischemic stroke
Normal gait analysis
Post-stroke gait dysfunction is a measure of stroke severity in humans, and a primary focus of rehabilitation program for stroke patients is to facilitate as much as possible normal movement and postural stability.26,27 Varying degree of gait impairment has also been observed in different rodent models of stroke.28–30 To assess the changes in gait following stroke and the efficacy of the peptide in restoring normal gait, we used a computer-assisted, gait imaging and analysis system for rodents (CatWalk). Changes in several static (maximum contact area and paw print area) and dynamic (swing speed and stride length) gait parameters of the affected forepaw (contralateral) were assessed two weeks after stroke in both vehicle- and peptide-treated groups. Figure 5(a) and (e) shows that the maximum area of the affected forepaw that comes in contact with the floor during the stance phase of gait (maximum contact area), is significantly larger in the peptide-treated groups as compared to the untreated control, regardless of early (vehicle 0.89 ± 0.28 mm2 vs. peptide 1.4 ± 0.42 mm2, p = 0.01, r = 0.6) or delayed (vehicle 0.85 ± 0.30 mm2 vs. peptide 1.3 ± 0.29 mm2, p = 0.007, r = 0.6) peptide treatment. Similarly, the maximum floor area that comes in contact with the affected forepaw during the stance phase (print area) is significantly more for the group treated with peptide at either early (vehicle 1.1 ± 0.34 mm2 vs. peptide 1.7 ± 0.50 mm2, p = 0.02, r = 0.6) or delayed (vehicle 0.9 ± 0.35 mm2 vs. peptide 1.5 ± 0.33 mm2, p = 0.004, r = 0.6) time point (Figure 5(b) and (f)). Comparison of the speed at which the affected forepaw moves through the air between two consecutive paw placements (swing speed) also show significantly faster movement in the peptide-treated group (Figure 5(c) and (g)) for both early (vehicle 43.1 ± 10.6 m/s vs. peptide 55.2 ± 7.6 m/s, p = 0.02, r = 0.6) and delayed (vehicle 39.3 ± 7.2 m/s vs. peptide 51.8 ± 10.5 m/s, p = 0.009, r = 0.6) treatment paradigm. In addition, the distance between successive placements of the affected forepaw during maximal contact (stride length) is significantly larger with both early (vehicle 6.6 ± 0.9 mm vs. peptide 8.7 ± 2.3 mm, p = 0.03, r = 0.5) and delayed (vehicle 6.4 ± 0.9 mm vs. peptide 7.6 ± 1.4 mm, p = 0.04, r = 0.5) peptide treatment (Figure 5(d) and (h)).
Effect of early and delayed peptide treatment on ischemia-induced normal gait impairment. Quantitative analysis of (a, e) Maximum contact area (mm2); (b, f) print area (mm2); (c, g) swing speed (m/s) and (d, h) stride length (mm) in the affected forepaw (contralateral) 14 days after MCAO in rats treated with vehicle or peptide (a–d) at the onset of reperfusion or (e–h) 6 h after the onset of the insult. Data are expressed as mean ± SD with individual values in the scatter plot (for early treatment – vehicle: n = 9, peptide n = 7; for delayed treatment – vehicle n = 9, peptide n = 9). *p < 0.05 and **p < 0.01 for vehicle- versus peptide-treated group.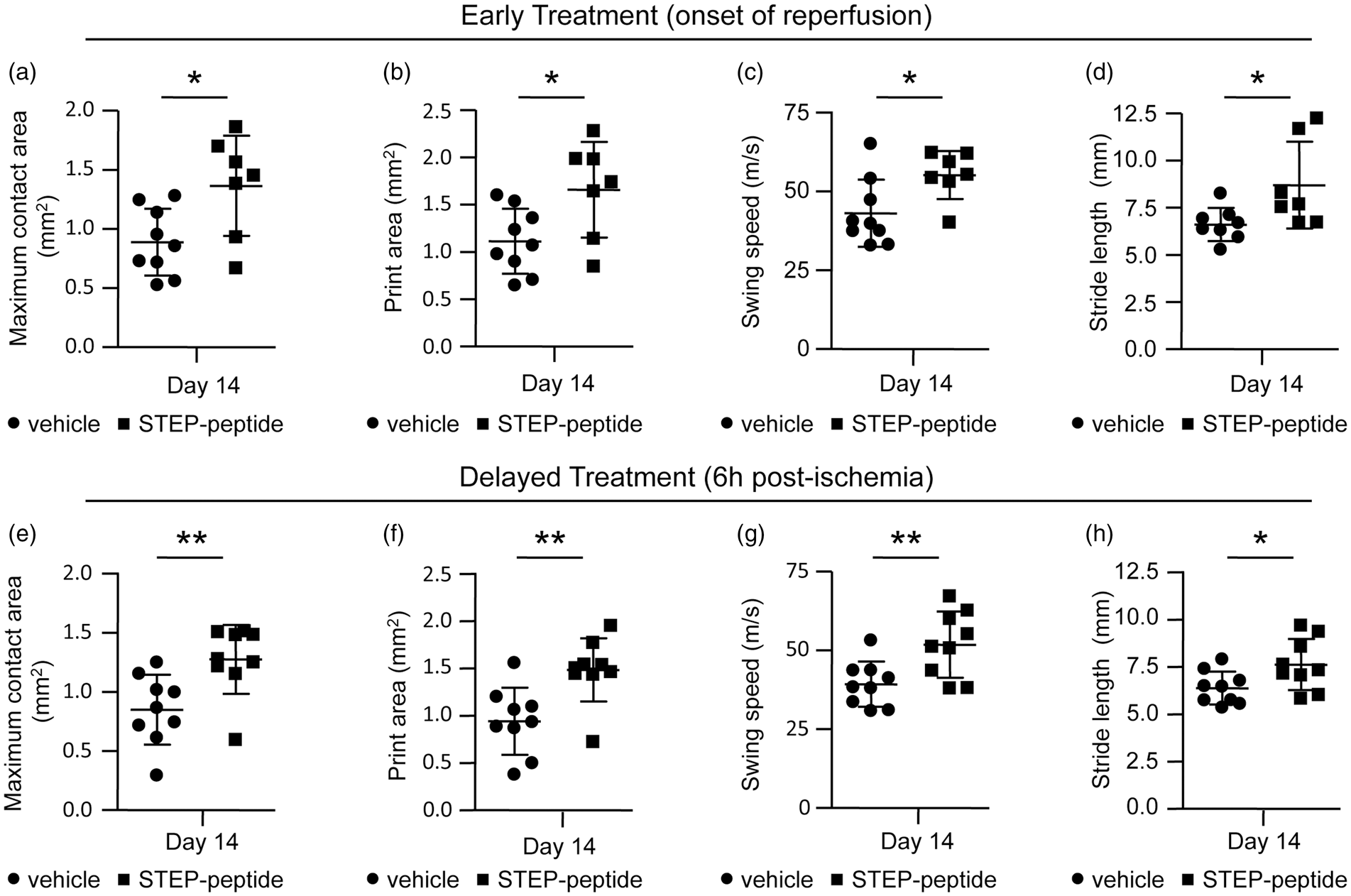
Motor coordination and balance
Next, we evaluated the deficit in spontaneous usage of the affected forelimb of the vehicle- and peptide-treated rats using the cylinder test on 7th, 14th and 21st day after MCAO (Figure 6(a) and (b)). Significant group difference was observed between vehicle and early treatment groups [F(1, 14) = 30.424, p < 0.001], as well as vehicle and delayed treatment groups [F (1, 18) = 71.374, p < 0.001]. A significant day effect was also evident between vehicle- and peptide-treated groups for both early [F (2,28) = 58.518, p < 0.001] and delayed [F (2,36) = 38.137, p < 0.001] treatment. Evaluation of spontaneous forelimb use on individual testing days shows significantly higher usage of the affected forelimb in the peptide-treated group on day 7 (51.7 ± 12.1% vs. 83.7 ± 16.4%, p < 0.001, r = 0.8), day 14 (71.3 ± 12.4% vs. 93.86 ± 6.9%, p < 0.001, r = 0.8) and day 21 (87.6 ± 5.9% vs. 105.9 ± 6.5%, p < 0.001, r = 0.8) when compared to the vehicle-treated group. Similarly, delayed intervention with the peptide also leads to significantly higher forelimb use on day 7 (52.8 ± 9.6% vs. 86.7 ± 10.3%, p < 0.001, r = 0.9), day 14 (65.7 ± 11.1% vs. 97.9 ± 10.6%, p < 0.001, r = 0.8) and day 21 (81.8 ± 3.8% vs. 97.9 ± 9.2%, p < 0.001, r = 0.8).
Effect of early and delayed peptide treatment on ischemia-induced motor coordination and sensory motor deficits. (a, b) Spontaneous contralateral forelimb use was assessed using cylinder test on day 7, 14 and 21 after MCAO, following (a) early peptide treatment (on days 7, 14 and 21 - vehicle: n = 8, peptide: n = 8) or (b) delayed peptide treatment (on days 7, 14 and 21 – vehicle: n = 10, peptide: n = 10). Results are expressed as a percentage of pre-stroke values. (c, d) Motor impairment and balance was assessed using the rotarod test on day 7, 14 and 28 after MCAO, following (c) early peptide treatment (on day 7 – vehicle: n = 9, peptide: n = 9; at day 14 – vehicle: n = 10, peptide: n = 9; at day 28 – vehicle: n = 9, peptide: n = 9) or (d) delayed peptide treatment (on day 7 – vehicle: n = 10, peptide: n = 9; at days 14 and 28 – vehicle: n = 10, peptide: n = 10). Time spent on rotarod is expressed as a percentage of pre-stroke values. (e, f) Mean latency to remove adhesive label from contralateral forepaw (time in seconds), as a measure of sensorimotor function, was assessed on day 7, 14 and 21 after MCAO, following (a) early peptide treatment (on day 7 – vehicle: n = 8, peptide: n = 6; on days 14 and 21 – vehicle: n = 10, peptide: n = 6) or (b) delayed peptide treatment (on days 7 and 14 – vehicle: n = 10, peptide: n = 9; on day 21 – vehicle: n = 10, peptide: n = 10). All data are expressed as mean ± SD with individual values in a scatter plot. *p < 0.05, **p < 0.01 and ***p < 0.001 for vehicle- versus peptide-treated group.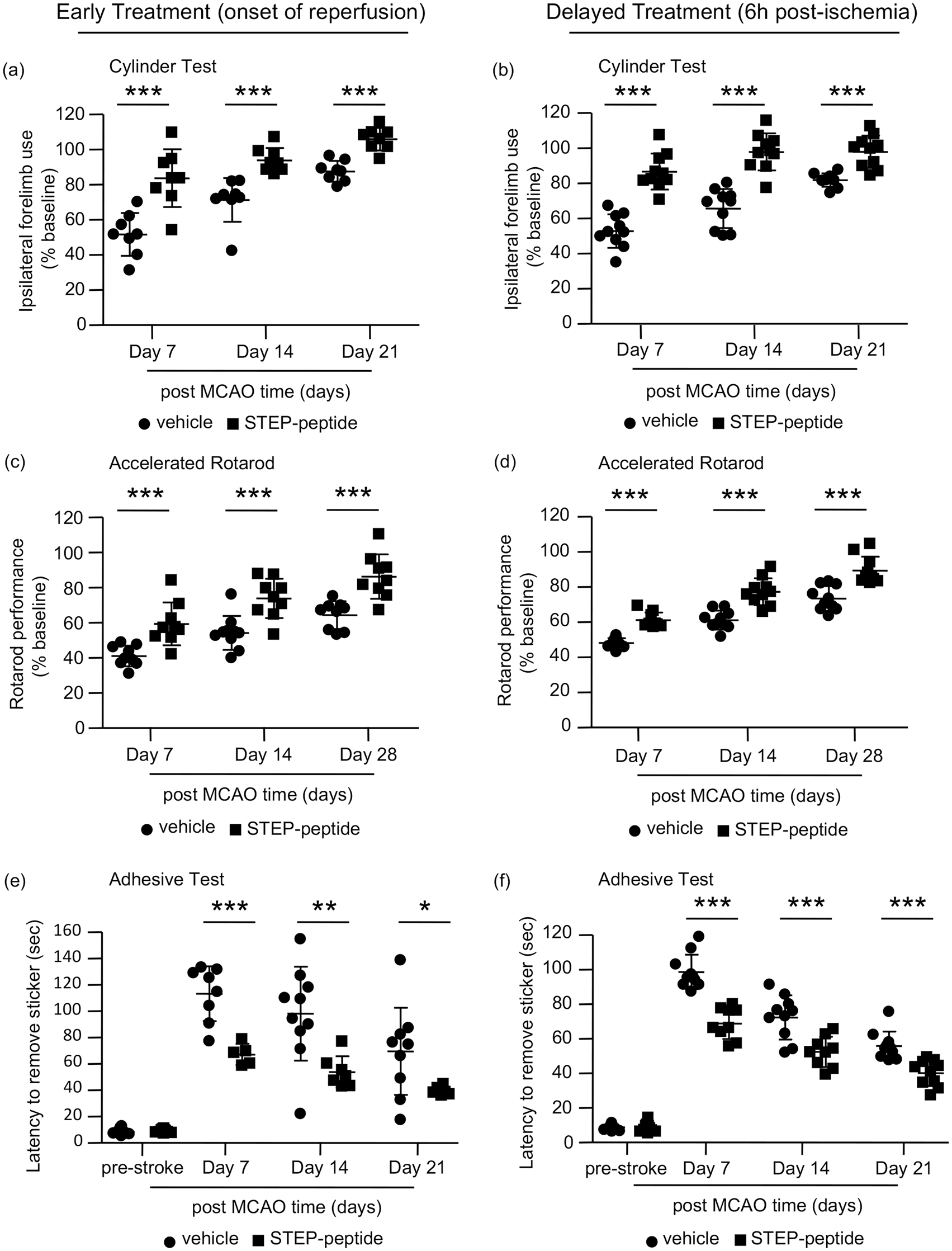
To assess the effect of peptide treatment on stroke-induced deficits in balance and motor coordination (coordinated alteration of fore and hind paw), we measured the latency to fall from an accelerated rotarod on 7th, 14th and 28th day after MCAO. Data analysis reveals significant group difference between the two conditions (vehicle vs. peptide-treated groups) for both early treatment [F (1, 15) = 25.072, p < 0.001] and delayed treatment [F (1, 17) = 33.99, p < 0.001] with the peptide (Figure 6(c) and (d)). In addition, significant day difference is also observed for both early [F (1.431,30) = 74.039, p < 0.001] and delayed [F (2,34) = 354.098, p < 0.001] treatment (for early treatment Greenhouse-Geisser correction was applied as Maulchy’s sphericity test failed). Comparison of rotarod performance on each of the three testing days, between vehicle- and peptide-treated groups, shows significantly better performance on day 7 (41.1 ± 5.9% vs. 59.3 ± 12.2%, p < 0.001, r = 0.7), day 14 (54.2 ± 9.7% vs. 73.9 ± 11.2%, p < 0.001, r = 0.7) as well as day 28 (64.3 ± 7.9% vs. 86.3 ± 12.6%, p < 0.001, r = 0.7) after early peptide treatment. Delayed treatment also shows significantly better performance on the rotarod on day 7 (48.1 ± 2.7% vs. 61.1 ± 4.3%, p < 0.001, r = 0.9), day 14 (61.2 ± 5.4% vs. 77.3 ± 7.6%, p < 0.001, r = 0.8) and day 28 (73.4 ± 7.0% vs. 89.4 ± 7.9%, p < 0.001, r = 0.7) when compared with vehicle-treated control.
Sensory motor assessment
Sensory motor deficit in the contralateral forelimb of vehicle- and peptide-treated groups was evaluated by measuring the latency to contact and removal of a stimuli (adhesive tape) from the contralateral forepaw on 7th, 14th and 21st day after MCAO. Comparison between vehicle- and peptide-treated groups show significant group effect for both early treatment [F(1, 9) = 8.278, p = 0.018] and delayed treatment [F (1, 17) = 33.94, p < 0.001] with the peptide (Figure 6(e) and (f)). A significant day effect is also observed for both early [F (2,18) = 4.28, p = 0.03] and delayed [F (2,34) = 151.26, p < 0.001] treatment. Evaluation of sensory motor deficit on each testing day shows significant difference in the latency to remove the tape between vehicle- and peptide-treated groups on day 7 (113.3 ± 20.8 s vs. 68.6 ± 8.0 s, p < 0.001, r = 0.8), day 14 (98.1 ± 35.7 s vs. 53.6 ± 13.1 s, p = 0.007, r = 0.6) as well as day 21 (69.5 ± 33.1 s vs. 39.4 ± 3.2 s, p = 0.04, r = 0.5). Similarly, following delayed peptide treatment, a significantly enhanced sensory motor function is observed on day 7 (98.7 ± 9.9 s vs. 68.9 ± 9.1 s, p < 0.001, r = 0.9), day 14 (72.4 ± 12.8 s vs. 52.4 ± 8.7 s, p < 0.001, r = 0.7) and day 21 (55.9 ± 8.3 s vs. 40.1 ± 7.5 s, p < 0.001, r = 0.7).
Spatial memory task
To assess the effect of the peptide on post-ischemic cognitive deficits, we next evaluated spatial learning (escape latency) and memory (probe trial) performance using Morris Water Maze. Comparison of mean escape latencies to reach the hidden-platform during training (22nd to 25th days post-MCAO) shows that group differences between vehicle- and peptide-treated groups are not significant for both early [F (1, 8) = 5.143, p = 0.05] and delayed [F (1, 15) = 2.877, p = 0.11] treatment paradigm (data not shown). In the probe test (26th day after MCAO), in which the escape platform was removed from the tank, vehicle-treated rats spent significantly less time in the target quadrant compared to the rats treated with the peptide for both early (vehicle 23.5 ± 6.6% vs. peptide 37.3 ± 13.2%, p = 0.01, r = 0.6) and for the delayed (vehicle 17.2 ± 6.9% vs. peptide 33.1 ± 7.6%, p < 0.001, r = 0.8) treatment paradigm (Figure 7(b) and (e)). In the visible platform test (cue test on 27th day after MCAO), the escape latencies to reach the platform did not significantly differ between the vehicle- and peptide-treated groups, for both early and delayed treatment (Figure 7(c) and (f)).
Effect of early and delayed peptide treatment on ischemia-induced spatial memory impairment. (a, d) Representative traces illustrating the swim pattern of vehicle- and peptide-treated rats in the target quadrant during the probe test following (a) early or (d) delayed peptide treatment. (b, e) Quantitative analysis of the time spent in the target quadrant (%) during probe test for both vehicle- and peptide-treated groups following (b) early peptide treatment (vehicle: n = 10, peptide: n = 9) or (e) delayed treatment (vehicle: n = 10, peptide: n = 9). (c, f) Quantitative analysis of mean escape latency (time in sec) to a visible platform for both vehicle- and peptide-treated groups in cue test following (c) early peptide treatment (vehicle: n = 10, peptide: n = 9) or (f) delayed treatment (vehicle: n = 10, peptide: n = 9). All data are expressed as mean ± SD with individual values in a scatter plot. *p < 0.01 and **p < 0.001 for vehicle- versus peptide-treated group.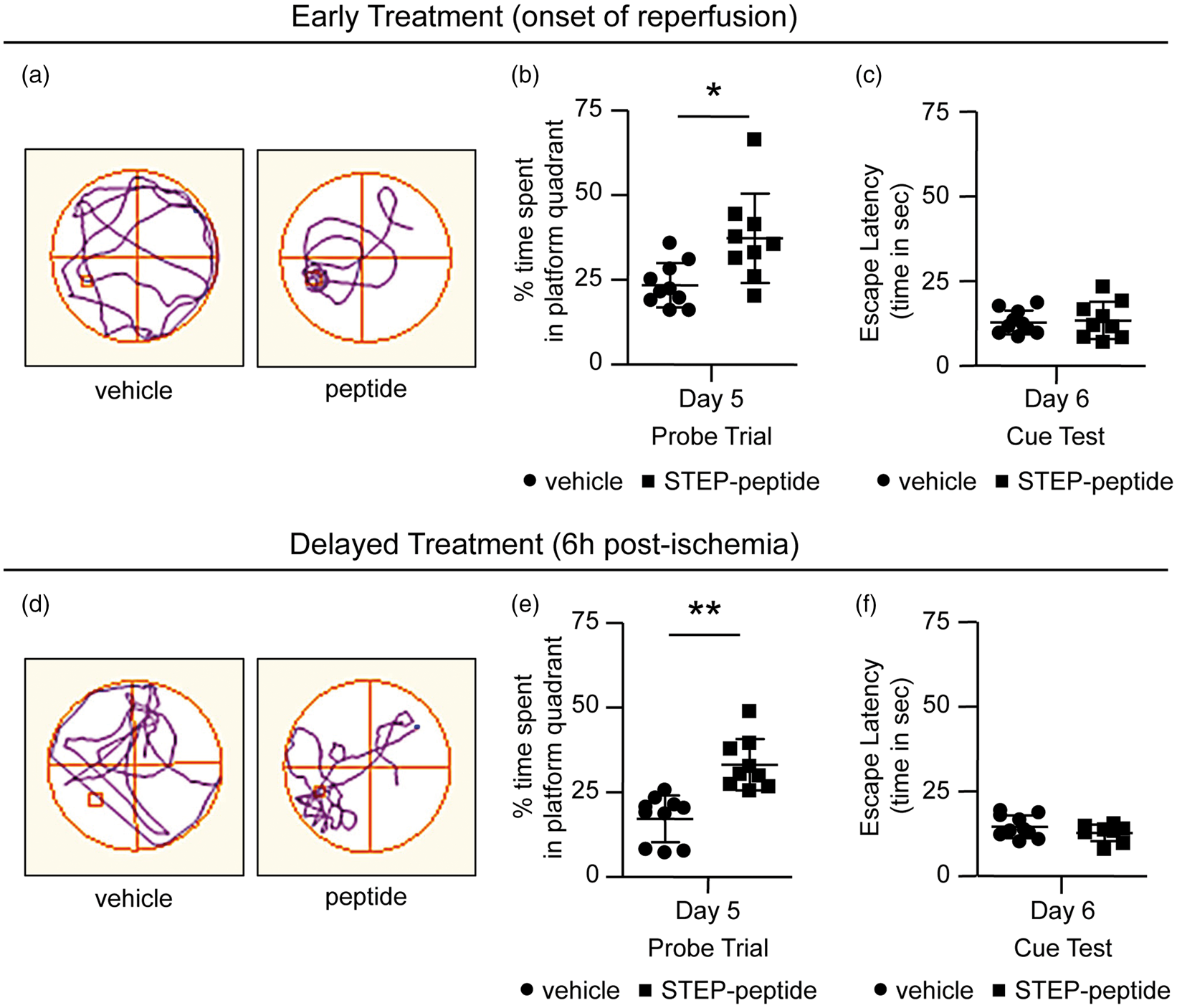
Finally, analysis of the longitudinal MRI and behavioral data for post hoc actual power calculation (Supplementary Table S1) shows that the sample size used for each of the outcome measure was large enough to convincingly establish that both early and delayed peptide treatment leads to significantly better outcome in terms of lesion volume and functional deficits.
Discussion
The current study demonstrates for the first time the efficacy of a peptide mimetic derived from the tyrosine phosphatase STEP in facilitating long-term recovery from stroke, using MRI and a series of clinically relevant indicators of functional outcome. The significant reduction in infarct size and improvement in functional deficits observed in our study, even when the peptide is administered 6 h after the onset of the insult emphasizes the therapeutic potential of the peptide.
Our findings show that acute administration of a single dose of the peptide at the onset of reperfusion substantially reduces stroke mortality. The size of the lesion visualized by anatomical T2 maps in the surviving animals initially reduces from day 1 to day 14 in both the vehicle- and peptide-treated groups but remains unchanged from day 14 to day 28 after MCAO. Such initial reduction in lesion size after transient MCAO suggests dynamic maturation of the lesion in the first two weeks and has also been observed in earlier studies. 31 However, the lesion size observed in the peptide-treated group at day 1 is significantly lower compared to vehicle-treated group and remain significantly lower at both 14 and 28 days after MCAO. In addition, evaluation of ADC and FA values in the lesion area 28 days after MCAO shows significant difference between vehicle- and peptide-treated groups. Earlier studies have reported that the temporal evolution of ADC in stroke lesion involves an acute (6 h), sub-acute (1–5 day) and chronic phase (>5 days). ADC value decreases in the ischemic infarct in the acute phase due to restricted diffusion triggered most likely by cytotoxic edema. The ADC value then pseudonormalizes in the sub-acute phase reflecting a co-existence of cytotoxic edema (associated with decreased diffusion) and a vasogenic edema (associated with increased diffusion). Thereafter, the ADC of the infarct area increases again reflecting the accumulation of extracellular water, tissue cavitation and gliosis.32–34 On the other hand, the FA value continues to decrease as the infarct evolves and remains significantly reduced as a consequence of progressive loss of structural integrity and orientation of white matter tissue resulting in increased radial diffusivity.34–36 Based on these earlier observations, the lower ADC values in our study with peptide treatment most likely reflect reduced tissue breakdown and less accumulation of extracellular water in the residual stroke cavity. Whereas the larger FA value reflects increase in axial diffusivity and decrease in radial diffusivity, which is consistent with better tissue structural integrity and orientation at this latter stage. These findings suggest that limiting the severity of the initial brain injury with TAT-STEP peptide treatment could facilitate long-term recovery.
Consistent with the above interpretation, the peptide-treated group demonstrates higher recovery in static and dynamic gait parameters (CatWalk), spontaneous forelimb use (cylinder test), motor coordination (rotarod) and sensory motor (adhesive test) function at all the time points tested. This overall recovery of normal gait and sensorimotor function strongly suggests that peptide treatment attenuates the loss of structural integrity of the cortical sensory-motor network. In addition, peptide treatment improves post-ischemic deficit in learning and memory evaluated by Morris water maze. Specifically, the time spent in the target quadrant during the probe trial is significantly enhanced with peptide treatment without a significant difference in the latency to reach the platform during training or cue test, suggesting a recovery in spatial memory. This would imply that the potential benefit of the peptide is not restricted to the sensorimotor cortex but also extends to hippocampus, since hippocampus is known to play a key role in spatial memory. 37
A key objective of the present study was to evaluate the outcome when peptide treatment is delayed for 6 h after MCAO, to mimic what might happen in a clinical setting. Our findings show that acute peptide treatment when delayed to 6 h post-occlusion is also able to reduce stroke mortality and significantly reduce the temporal evolution of the lesion size over 28 days when compared to the vehicle-treated control group. The lower ADC value as well as the higher FA value in the peptide-treated group at day 28, compared with the untreated group is also indicative of improved structural integrity of the infarcted area following delayed treatment. Functional evaluation of normal gait, motor coordination, sensory motor function and cognitive impairment shows significant improvement following delayed intervention with the peptide. An important outcome of these longitudinal studies is that with both early and delayed peptide intervention, there is smaller lesion size and limited tissue loss in the peri-infarct area in the surviving animals. Since post-stroke lesion in the brain compromises neural networks connecting different regions of brain, limiting the lesion size and loss of neurons in the peri-infarct areas would mean less number of compromised neural circuits. 38 The enhanced post-stroke functional recovery observed in the peptide-treated group in our study would therefore suggest that the recovering peri-infarct region has less compromised neural circuits, and could connect more effectively with the healthy adjacent tissues to restore crucial function of the affected area more efficiently. Alternatively, it is also possible that the local environment in the peri-infarct area of the peptide-treated group is more optimal for development of new brain circuits during long-term recovery resulting in enhancement of sensory and motor function. Thus, it is conceivable that the TAT-STEP peptide facilitates post-stroke functional recovery by limiting ischemic and reperfusion injury. The ability of the peptide to limit reperfusion injury would also suggest that the peptide could be used in combinational treatment with thrombolytic agents such as tissue plasminogen activator, which in addition to restoration of normal blood flow also leads to reperfusion injury through induction of oxidative stress.7,10
The pathophysiology of cerebral stroke induced brain damage involves interaction between multiple deleterious pathways, 39 and in view of this complexity, effective stroke therapy will very likely require approaches that target multiple mechanisms of damage. Previous studies have identified several substrates of STEP that include NR2B-NMDAR subunit, tyrosine kinases Pyk2 and Fyn, extracellular-regulated kinase 1/2 and the stress-activated p38 mitogen-activated protein kinase (p38 MAPK).17,18,40–43 Activation of these target proteins of STEP following an ischemic insult has been shown to play a role in Ca2+-overload and excitotoxicity, nitric oxide signaling, oxidative stress, microglial activation and neuroinflammation that leads to neurotoxicity and subsequent brain damage.44–52 Since STEP inhibits the activity of this diverse group of signaling proteins through de-phosphorylation of specific tyrosine residues, it is conceivable that the STEP-derived peptide confers neuroprotection against ischemic brain damage by targeting these proteins. This interpretation is further supported by our earlier findings demonstrating that transduction of neurons with this peptide inhibits p38 MAPK phosphorylation and prevents neuronal injury in an in vitro model of glutamate excitotoxicity. 18 In addition, administration of the peptide also blocks p38 MAPK phosphorylation and prevents neuronal injury in both in vitro and in vivo models of ischemia. 19 To further our understanding of the multimodal effect of the peptide in ischemic stroke treatment, future studies will focus on the contribution of the peptide in regulating the other targets of STEP.
In conclusion, these findings demonstrate that acute treatment with a single dose of the STEP-derived peptide within 6 h of the onset of an ischemic insult reduces the rate of mortality, which is a key measure of success for stroke treatment in a clinical setting. The peptide also promotes recovery in the surviving animals by limiting the loss of brain tissue in the early stage. The overall effect size quantifying the difference in ischemic lesion size and functional outcome between vehicle- and peptide-treated groups, points towards a large impact of the peptide. Taken together, this study provides a valuable basis for further development of this promising new neuroprotective agent for stroke therapy.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Health grant numbers NS059962 (Paul, S) and NS083914 (Poddar, R).
Acknowledgements
We would like to thank Dr. Gary A Rosenberg, University of New Mexico, for his helpful comments and Dr. Yirong Yang at the preclinical MRI core facility for performing the MRI scans and assistance with data analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
RP has contributed towards concept and design; acquisition, analysis and interpretation of data; critical revision of the manuscript for intellectual content. SR has contributed to acquisition and analysis of data, and critically reviewed the manuscript for content. LW has contributed to acquisition and analysis of data, and reviewed the manuscript for content. AA has contributed towards analysis and interpretation of data and critically revised the manuscript. SP has contributed towards concept and design; analysis and interpretation of data; drafting and revision of manuscript. All the authors have approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
