Abstract
Perinatal hypoxic-ischemic (HI) brain injury remains highly associated with neurodevelopmental disability after preterm birth. There is increasing evidence that disability is linked with impaired white matter maturation, but there is no specific treatment. In this study, we evaluated whether, in preterm fetal sheep, delayed intranasal infusion of human amnion epithelial cells (hAECs) given 1, 3 and 10 days after severe HI, induced by umbilical cord occlusion for 25 min, can restore white matter maturation or reduce delayed cell loss. After 21 days recovery, asphyxia was associated with reduced electroencephalographic (EEG) maturation, brain weight and cortical area, impaired maturation of oligodendrocytes (OLs), no significant loss of total OLs but a marked reduction in immature/mature OLs and reduced myelination. Intranasal infusion of hAECs was associated with improved brain weight and restoration of immature/mature OLs and fractional area of myelin basic protein, with reduced microglia and astrogliosis. Cortical EEG frequency distribution was partially improved, with reduced loss of cortical area, and attenuated cleaved-caspase-3 expression and microgliosis. Neuronal survival in deep grey matter nuclei was improved, with reduced microglia, astrogliosis and cleaved-caspase-3-positive apoptosis. These findings suggest that delayed intranasal hAEC administration has potential to alleviate chronic dysmaturation after perinatal HI.
Introduction
Preterm birth is highly associated with substantial long-term disability. 1 Although survival rates have significantly improved in developed nations, there have only been modest reductions in long-term cognitive, sensory and motor disabilities. 2 It is conservatively estimated that as many as 50% of extremely preterm infants have cognitive and behavioral difficulties. 2 Neurodevelopmental disability has been closely associated with impaired cerebral maturation, with reduced grey and white matter volumes, cortical folding and gyral complexity, delayed cellular maturation, reduced dendritic arborization and impaired synaptogenesis.3–5 Currently, there are no specific neuroprotection or neurorepair therapies for the preterm brain.
The etiology of preterm brain injury is multifactorial, but key factors include hypoxia-ischemia and inflammation.6,7 These adverse events may start well before birth and both the preterm fetus and newborn are at risk of exposure to combined and multiple-insults.5,6,8,9 Key mechanisms mediating impaired brain development include acute and chronic inflammation and loss of survival and cell development cues.5–7 This combination of uncertain timing and variable mechanisms is a key challenge for the development of neuroprotective interventions.5,7,8
Exogenous stem cells can promote angiogenesis, neurogenesis, synaptogenesis and neurite outgrowth, and so may have potential to improve outcomes from preterm brain injury.10–12 Their longevity in situ may help ameliorate chronic or repeated insults, and repeated doses may build upon evolving endogenous repair processes and the regenerative niche created by earlier administration.13,14 In particular, human amnion epithelial cells (hAECs) harvested from the inner lining of the amniotic membrane have pluripotent potential because they are formed from embryonic ectoderm before gastrulation. 11 They have low tumorigenicity and are immune privileged, due to their low expression of human leucocyte antigen and co-stimulatory molecules. 11 This allows allogenic transplantation without the need for immune suppression. 11 hAECs have been reported to be beneficial for healing in multiple tissues, including eyes, skin and lungs, 11 likely by modulating local inflammation and releasing trophic factors. 15
In adult rats, intra-cerebral administration of hAECs, 24 h after middle cerebral artery occlusion (MCAO), reduced infarct size, peri-infarct apoptosis, and behavioral dysfunction two weeks post-MCAO. 16 hAECs, given intra-cerebrally, also reduced cerebral edema and improved motor deficits after hemorrhage. 17 Similar beneficial effects are observed with human amnion mesenchymal cells, and amniotic fluid-derived cells, given intra-cerebrally after MCAO.18,19 In term and preterm fetal sheep, three systemic doses of hAECs, started either during or after exposure to lipopolysaccharide (LPS), reduced inflammation and white matter apoptosis and loss of oligodendrocytes (OLs).20,21 Similarly, in preterm lambs, hAECs reduced ventilation-induced cerebral injury. 22 In newborn mice, hAECs reduced apoptosis and astrocytosis due to LPS exposure in utero and post-natal hyperoxia. 23 To date, there have been no studies in large animals evaluating the long-term effects of hAECs on brain injury after perinatal hypoxia-ischemia.
In the present study, we examined the effect of three intranasal infusions of hAECs given to preterm fetal sheep commencing 24 h after asphyxia induced by reversible umbilical cord occlusion at an age when brain maturation is broadly equivalent to 28–30 weeks gestation humans. 24 We hypothesized that hAECs would significantly reduce secondary grey and white matter injury and improve white matter maturation, and that protection would be associated with reduced cerebral inflammation.
Materials and methods
Experimental preparation
All procedures were approved by the Animal Ethics Committee of the University of Auckland and in accordance with the Animal Welfare Act (1999) of New Zealand and reported according to the ARRIVE guidelines (Animal Research: Reporting In Vivo Experiments). Eighteen Romney-Suffolk cross fetal sheep were surgically instrumented at 98–100 days of gestational age (ga; term is ∼147 days) as previously described. 25 Food, but not water was withdrawn 18 h before surgery. Ewes were given long acting oxytetracycline (20 mg/kg, Phoenix Pharm Distributors, Auckland, New Zealand) intramuscularly 30 min before surgery for prophylaxis. Anesthesia was induced by i.v. injection of propofol (5 mg/kg, AstraZeneca, London, U.K.) and general anesthesia was maintained using 2–3% isoflurane (Medsource, Ashburton, New Zealand) in oxygen. The depth of anesthesia, maternal heart rate and respiration was constantly monitored by trained anesthetic staff. Ewes received a constant infusion of isotonic saline (approximately 250 ml/h) to maintain fluid balance.
Surgical instrumentation
A midline abdominal incision was made to expose the uterus, and the fetus was partially exteriorized for instrumentation. Polyvinyl catheters (SteriHealth, Dandenong South, VIC, Australia) were placed in a fetal brachial artery to measure blood pressure and for pre-ductal blood sampling. An additional catheter was placed into the amniotic sac for measurement of amniotic fluid pressure. Electrodes (AS633-3SSF, Cooner Wire, Chatsworth, CA, USA) were placed subcutaneously over the right shoulder and at the level of the left fifth intercostal space to measure the fetal electrocardiogram from which fetal heart rate (FHR) was derived. Two pairs of electroencephalograph (EEG) electrodes (AS633-7SSF; Cooner Wire) were placed through burr holes onto the dura over the parasagittal parietal cortex (5 and 10 mm anterior to bregma and 5 mm lateral) and secured with cyanoacrylate glue. A polyvinyl catheter was inserted into the right fetal nostril and placed against the cribriform plate for delivery of hAECs and vehicle. The catheter was sutured in place and the nostrils sealed with cyanoacrylate glue. An inflatable silicone occluder (In Vivo Metric, Healdsburg, CA, USA) was placed around the umbilical cord to allow umbilical cord occlusion. All fetal leads were exteriorized through the maternal flank, and a maternal long saphenous vein was catheterized for maternal post-operative care. The maternal laparotomy incision was repaired and the skin infiltrated with 10 ml of 0.25% bupivacaine plus adrenaline (AstraZeneca).
Post-surgical recovery
After surgery, ewes were housed in separate metabolic cages in a temperature-controlled room (16 ± 1℃, humidity 50 ± 10%) with a 12-h light/dark cycle (6.00 am lights on) and ad libitum access to water and food. Ewes received i.v. antibiotics daily for four days (600 mg benzylpenicillin sodium; Novartis, Auckland, New Zealand, and 80 mg gentamicin sulfate; Pfizer, Auckland, New Zealand). Fetal catheters were maintained patent by continuous infusion of heparinized saline (20 U/ml at 0.2 ml/h). Physiological parameters were recorded continuously from 24 h before umbilical cord occlusion, 26 until the end of experiment, 21 days after occlusion. At the end of the experiment, the ewes and their fetuses were killed by an intravenous overdose to the ewe of sodium pentobarbitone (9 g; Pentobarb 300, Chemstock International, Christchurch, New Zealand).
hAEC collection and handling
Tissue collection to isolate hAECs was performed with approval from the Monash Health Human Research Ethics Committee, Monash, Melbourne, Australia. Women gave written, informed consent for the collection of their placentae, and all procedures conformed to the standards set by the World Medical Association Declaration of Helsinki. hAECs were isolated from term placentae donated by healthy volunteers who underwent elective caesarean section delivery as previously described. 27 Briefly, the amnion was manually separated from the chorion, and blood removed. The hAECs were enzymatically removed by two 1 h digestion periods using TrypZean (Sigma-Aldrich, Sydney, NSW, Australia) and TrypZean was then inactivated with soybean trypsin inhibitor (Sigma-Aldrich). The hAECs were collected by centrifugation for 10 min at 1000g and re-suspended for cell counts and viability testing (Supplementary Table 1). As previously characterized, hAECs are epithelial cell adhesion molecule positive but negative for CD31, CD45, CD90 and CD105. 27 Cells were cryopreserved using standard methods at 5 × 106 cells/ml. Frozen cells were thawed, washed, counted and placed in culture 24 h before administration. 27 For culture, 10 million hAECs were seeded in a T175 flask and maintained at 5% CO2 in Dulbecco's Modified Eagle Medium: Nutrient Mixture F-12 (Invitrogen New Zealand Limited, Auckland, New Zealand) with 10% heat-inactivated fetal bovine serum. The hAECs were then taken out of culture, washed, counted and their viability assessed before the cells were suspended in 2 ml sterile 1 × Dulbecco's phosphate-buffered saline (PBS) without calcium and magnesium balanced salt solution (Invitrogen New Zealand Limited) for intranasal administration (Supplementary Table 1).
Experimental recordings
Fetal mean arterial blood pressure (MAP), corrected for maternal movement by subtraction of amniotic pressure, electrocardiogram (ECG), and EEG were recorded continuously for offline analysis using custom data acquisition software (LabVIEW for Windows, National Instruments, Texas, USA). The blood pressure signal was collected at 64 Hz and low pass filtered at 30 Hz. The fetal ECG was analog filtered between 0.05 and 100 Hz and digitized at 512 Hz, and used to derive FHR. The analog fetal EEG signal was low pass filtered with a cut off frequency set with the −3 dB point at 30 Hz, and digitized at a sampling rate of 512 Hz. EEG power was derived from the power spectrum signal between 0.5 and 20 Hz, while spectral edge frequency (SEF) was calculated as the frequency below which 90% of the power was present. 28 For data presentation, total EEG power was normalized by log transformation (dB, 20 × log power). Maturational development was assessed by changes in the frequency distribution of SEF as described by Szeto 29 over 24 h periods at days 104 (i.e. the 24 h before occlusion), 110, 115, 120 and 125 ga. For this analysis, the proportion of time that the SEF fell within one Hz bands from 0.5 to 19.5 Hz was calculated from 1 min average data for each 24 hour period.
Experimental protocol
Ewes and fetuses were allowed to recover for four days before experiments commenced. On day 104 ga, fetuses were randomly assigned to receive boluses of either 2 ml of PBS (asphyxia-vehicle group, n = 6) or 40.7 ± 1.7 × 106 hAECs intranasally in the same volume (asphyxia-hAEC group, n = 6), at 1, 3, and 10 days after occlusion. Sham-control fetuses did not receive occlusion or hAEC treatment (n = 7). Randomization was stratified by cohort to control for time of year. Sample size was determined based on an estimated population standard deviation of 3.5 for 2′,3′-cyclic-nucleotide 3′-phosphodiesterase (CNPase) fractional area. A sample size of 6 was required to provide 80% power to detect an improvement in fractional area of CNPase of 6.5 or greater. Fetuses were killed 21 days after the end of occlusion for histology in which the assessor was blinded to the treatment group. In addition, one additional asphyxia-hAEC fetus was killed four days after occlusion for histological assessment of migration of hAECs.
Systemic asphyxia was induced by 25 min of reversible complete umbilical cord occlusion. 25 Successful occlusion was determined by rapid onset of bradycardia and blood pressure elevation, and confirmed with pH and blood gas analysis. All occlusions were started in the morning between 9 and 9.30 a.m. Fetal arterial blood samples were collected at 1 h before, 5 and 17 min during, and 1, 2, 4, and 6 h after occlusion. Samples were taken daily between 8 and 9 a.m. Fetal pre-ductal pH, PaCO2, PaO2 ((ABL 800, Radiometer, Copenhagen, Denmark), and glucose and lactate (YSI model 2300, Yellow Springs, Ohio, USA) measurements were made.
Histology
Fetal brains were perfusion fixed in situ by 1 m height gravity perfusion, starting with 500 ml of 0.9% saline solution followed by 1000 ml of 10% phosphate-buffered formalin. Brains were immersion fixed for a further three to five days before being embedded in paraffin. Coronal slices (10 µm thick) were cut using a microtome (RM235, Leica Microsystems, Wetzler, Germany) at the level of the mid-striatum and the dorsal hippocampus. Slides were dewaxed in xylene, rehydrated in decreasing concentrations of ethanol and washed in 0.1 mol/L PBS. For antigen retrieval, the pressure cooker technique, with citrate buffer (450 ml of distilled water, 8 ml citric acid, 42 ml sodium citrate, pH 6.5), was used (EMS Antigen 200 Retriever, Aptum Biologics, Southampton, UK). Endogenous peroxidase was quenched by incubation in 1% H2O2 in methanol (or in PBS for Olig-2). Blocking was performed for 1 h at room temperature in 3% normal goat serum for all antibodies, except ionized calcium binding adaptor molecule 1 (Iba-1) for which normal horse serum was used. Sections were labeled with monoclonal antibodies; 1:200 mouse anti- neuronal nuclear antigen (NeuN, Chemicon International, Temecula, CA, USA), 1:200 mouse anti-synaptophysin (Merck-Millipore, Billerica, MA, USA), 1:200 rabbit anti-cleaved caspase-3 (Chemicon International), 1:400 rabbit anti-oligodendrocyte transcription factor 2 (Olig-2, Merck-Millipore), 1:200 mouse anti-CNPase (Merck-Millipore), 1:200 rabbit anti-myelin basic protein (MBP, Merck-Millipore), 1:200 rabbit anti-glial fibrillary acidic protein (GFAP, Abcam, Cambridge, United Kingdom), 1:200 goat anti-Iba-1 (Abcam), 1:200 mouse anti-tumor necrosis factor alpha (TNFα, AbD Serotec, Pucheim, Germany) or 1:200 mouse anti-human cytoplasmic marker STEM121 (Stem Cells Inc. Newark, CA, USA) overnight at 4℃. Sections were washed in PBS and incubated for 3 h with the species appropriate biotin-conjugated monoclonal 1:200 anti-mouse, 1:200 anti-rabbit, or 1:200 anti-goat in 3% normal goat serum for all except Iba-1 which was incubated in 3% normal horse serum. Slides were incubated in ExtrAvidin (1:200, Sigma-Aldrich) in PBS for 2 h and then reacted with diaminobenzidine tetrachloride (Sigma-Aldrich). The reaction was halted by immersion in distilled H2O and the slides washed and dehydrated before being mounted.
Data analysis
Brain regions used for analysis of the striatum (including the caudate nucleus and putamen) were assessed on sections 23 mm anterior to stereotaxic zero as defined in the stereotaxic atlas for fetal sheep. 30 The parasagittal intragyral white matter (IGWM) and periventricular white matter (PVWM) tracts as well as the parasagittal and lateral cortex were assessed on sections taken 17 mm anterior to stereotaxic zero. Images were obtained from labeled sections by light microscopy at 20 × or 40 × magnification on a Nikon 80i microscope equipped with a DS-Fi1-U3 camera and NIS Elements Br 4.0 imaging software (Nikon Instruments, Melville, NY, USA), using five fields in the striatum (Supplementary Figure 1(a)), three fields in the white matter (two parasagittal intragyral, one periventricular) and three fields in the cortex (Supplementary Figure 1(b)). Average scores from both hemispheres from two sections were calculated for each region. Cortical and white matter areas were assessed on acid fuchsin stained slides taken 17 mm anterior to stereotaxic zero (Supplementary Figure 1(b)).
All analyses were performed by an assessor (LGH) who was blinded to the treatment groups by independent coding of slides and data files. Healthy neurons were counted based on morphological assessment; neurons showing a pyknotic morphology were excluded. 31 Microglia showing either an amoeboid or ramified morphology were included. Cleaved caspase-3 positive cells were counted based on morphological assessment; only cells with both staining and apoptotic bodies were counted. Total and CNPase positive and MBP positive oligodendrocyte, microglia, astrocyte numbers and the area fraction of GFAP and MBP staining were quantified using the isodata threshold filter (ImageJ software, National Institutes of Health, USA).
Data were compared between groups using two-way analysis of variance (ANOVA, IBM SPSS 22.0 for Windows, (Armonk, NY: IBM Corp, USA) followed by Tukey’s post hoc test when a significant effect of group was found. The statistical analysis was performed masked to group. Region and time were treated as repeated measures as appropriate. Statistical significance was accepted at P < 0.05. Data are mean ± standard error of the mean (SEM).
Results
Blood chemistry and hemodynamic measurements during umbilical cord occlusion
Fetal arterial pH, blood gases, glucose and lactate concentrations 60 minutes before, at 5 and 17 min during umbilical cord occlusion, and 10 min, 1 h and 2 h after the end of occlusion in sham-control, asphyxia-vehicle and asphyxia-hAEC fetuses.
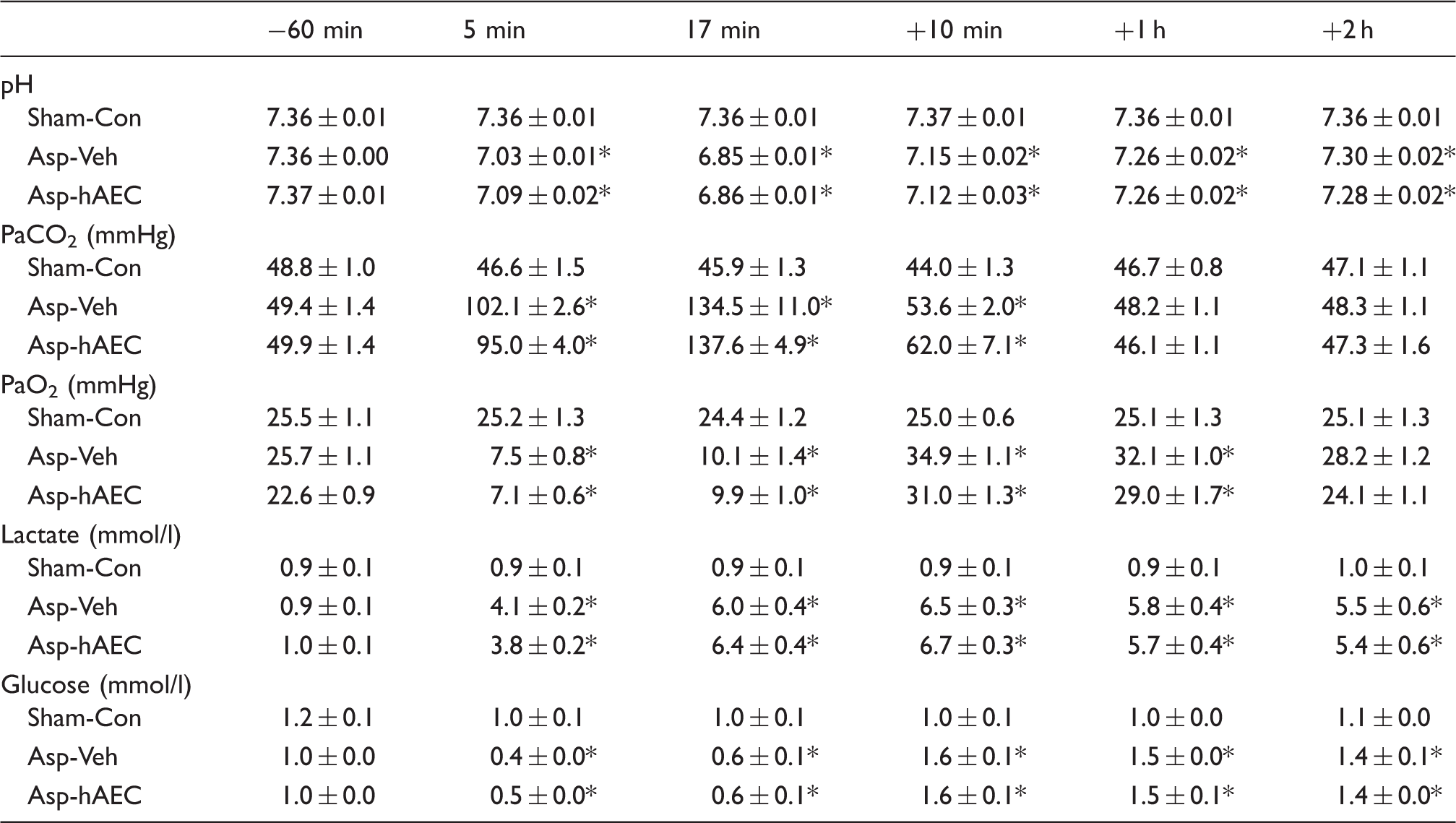
P < 0.05 vs. sham-control.
Sham-Con: sham control; Asp-Veh: asphyxia-vehicle; Asp-hAEC: asphyxia-hAECs.
Post-mortem findings
Asphyxia-vehicle was associated with reduced brain and lung weights compared to sham-control values (P < 0.05, Supplementary Table 2), with no significant effect on total body, heart, spleen or liver weight. In contrast, the asphyxia-hAEC group showed significantly improved brain weight (P < 0.05 vs. asphyxia-vehicle), but no other significant effect.
Cortical EEG and SEF
Cortical EEG power was rapidly suppressed during asphyxia and remained suppressed for the duration of the insult. There was no difference in EEG power between asphyxia groups and both remained modestly but significantly lower than the sham-control group for the remainder of the experiment (P < 0.05, Figure 1(a)).
Changes in electroencephalographic (EEG) power (dB, Panel A) and EEG spectral edge frequency (SEF, Hz, Panel B) from 24 h before until 21 days after umbilical cord occlusion (data not shown for occlusion). Sham-control, open circles (n = 7), asphyxia-vehicle, black circles (n = 6), asphyxia-hAECs, grey triangles (n = 6). Data are mean ± SEM, for hourly intervals from −24 to 0 h, three hourly intervals from 0 to 72 h, and 6 hourly intervals from 6 to 504 h. *P < 0.05 vs. sham-control, ANOVA + Tukey’s test.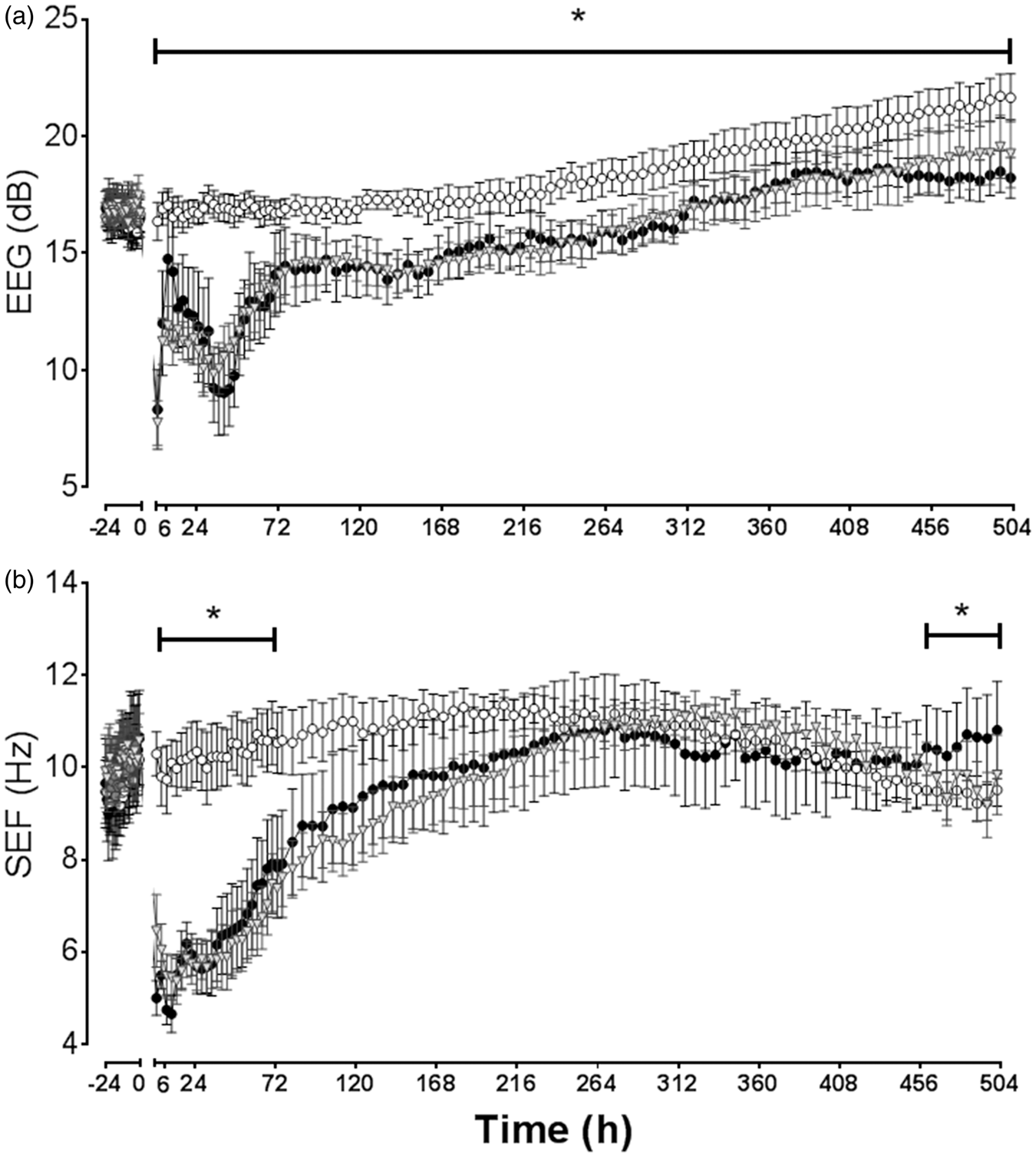
Cortical SEF was suppressed during asphyxia (data not shown) and remained significantly lower than the sham-control group in both asphyxia groups for the first 64 h (P < 0.05, Figure 1(b)). SEF was significantly higher in the asphyxia-vehicle group for the last 30 h (P < 0.05). SEF in the asphyxia-hAEC group was not different from either group at the end of experiment.
The baseline distribution of SEF values at 104 days ga was not different between groups (Figure 2). Sham-controls showed maturation of the distribution of SEF over time, with a shift from predominance of higher frequencies at 104 days ga, to a much broader mixture of low and high frequencies at 125 days ga, consistent with emerging sleep state cycling.
29
Asphyxia-vehicle was associated with relative loss of higher frequencies after asphyxia at 110 days ga (P < 0.05 vs. sham-controls). This was followed by a persistence of the higher frequency peak at ∼ 12 Hz that persisted to 125 days ga, with a corresponding reduction in the proportion of lower frequency SEFs (P < 0.05 vs. sham-controls). The asphyxia-hAEC group showed a similar loss of high frequency SEF at 110 days to the asphyxia-vehicle group (P < 0.05 vs. sham-controls). However, at 115, 120 and 125 days gestation, the asphyxia-hAEC group showed attenuation of the large peak in the distribution of SEF values at 12 Hz (P < 0.05). By 125 days, asphyxia-hAEC group showed a similar pattern of distribution of SEF to sham-controls, with significantly reduced proportion of SEF at 11 and 12 Hz than the asphyxia-vehicle group and a greater proportion at 8 Hz (P < 0.05).
Relative distribution of spectral edge frequency (SEF, %) at 104 (i.e. the 24 h before occlusion), 110, 115, 120 and 125 days ga. Sham controls show progressive maturation from predominantly higher frequency activity, to a much broader distribution by 125 days ga with an increasing proportion of lower frequency activity, consistent with emerging sleep state cycling. Asphyxia was associated with initial loss of higher frequency SEF at 110 days ga, followed by persistence of the higher frequency peak to the end of recordings at 125 days ga, and a reduced proportion of time spent in low frequency compared to sham-controls. The asphyxia-hAEC group showed partial restoration of the high frequency peak to sham-control values. Sham-control, open circles (n = 7), asphyxia-vehicle, black circles (n = 6), asphyxia-hAECs, gray squares (n = 6). Data are mean ± SEM. (a) sham-control vs. both asphyxia groups (P < 0.05); (b) asphyxia-vehicle vs. both sham-control and asphyxia-hAEC (P < 0.05); (c) sham-control vs. asphyxia-vehicle (P < 0.05), ANOVA + Tukey’s test.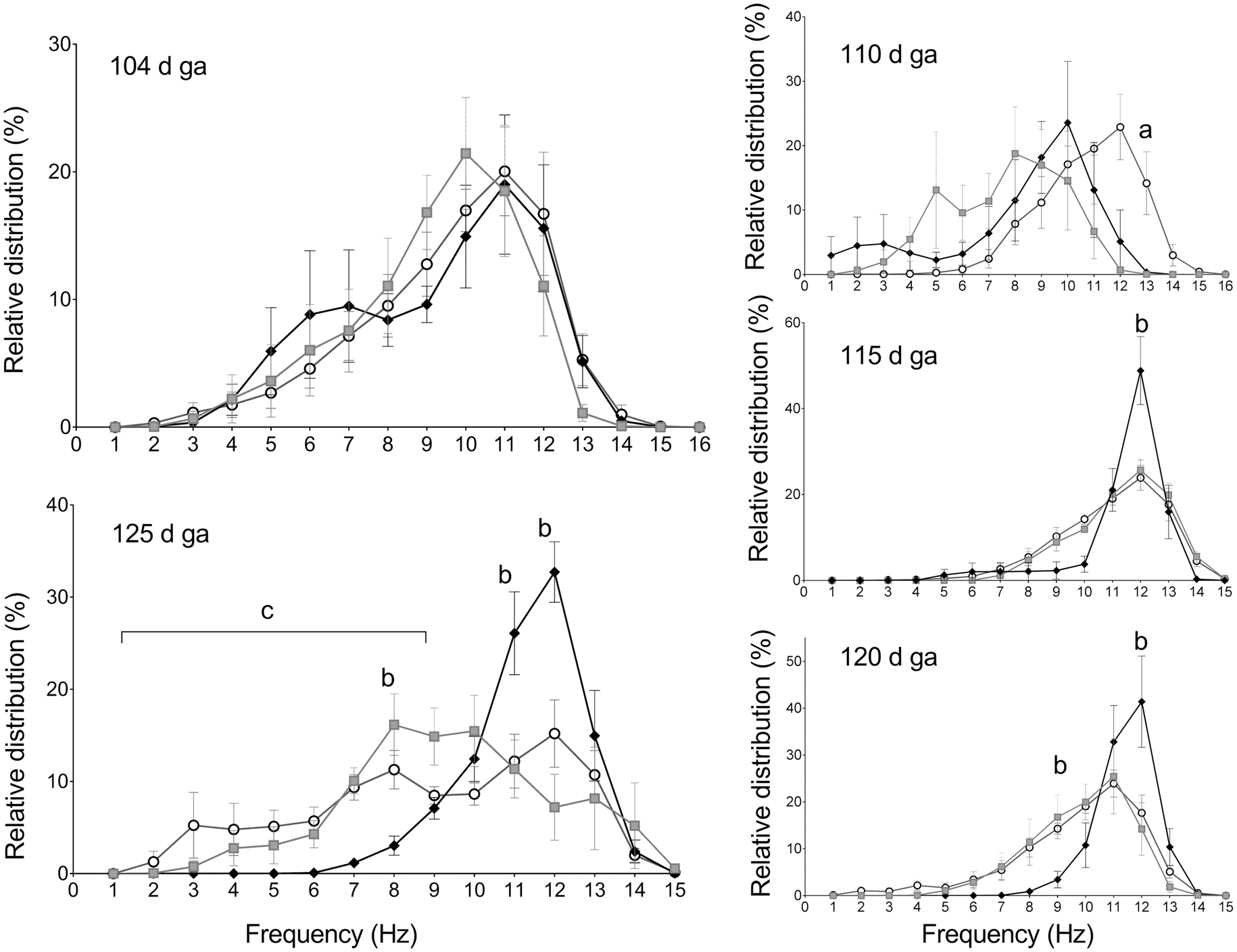
White matter histology
In the fetus killed four days after asphyxia (i.e. 3 days after the intranasal infusion), hAECs were observed in the medial septal nucleus. In the main study, small numbers of hAECs were observed in all asphyxia-hAEC fetuses in the PVWM 21 days after occlusion (Supplementary Figure 2).
Asphyxia was associated with a marked reduction in total white matter area after 21 days recovery: 76.7 ± 33.5 mm2 in sham-controls vs. 52.5 ± 24.2 mm2 in the asphyxia-vehicle group (P = 0.010). The asphyxia-hAEC group showed an intermediate area of 64.0 ± 25.9 mm2 (N.S. vs. sham-control or asphyxia-vehicle). There were no significant effects of asphyxia or hAEC infusion on total numbers of Olig-2 positive cells compared to sham-control (P = 0.25, Figure 3(a)). In contrast, asphyxia was associated with a significant overall reduction in CNPase positive area in the three white matter regions assessed, compared to sham-controls (P < 0.001, Figure 3(b)). hAEC treatment was associated with an overall increase in CNPase positive area vs. asphyxia-vehicle (P = 0.003), and a statistically borderline reduction compared to sham-controls (P = 0.05). Further, asphyxia was associated with an overall reduction in the ratio of CNPase to Olig-2 positive cells in the white matter to 0.64 ± 0.05 vs. 0.89 ± 0.05 after sham-occlusion (P = 0.003). hAEC infusion was associated with restoration of the ratio to sham-control values (0.88 ± 0.05 vs. asphyxia-vehicle, P = 0.004). Consistent with these findings, asphyxia-vehicle was associated with a significant decrease in the area of MBP compared to sham-controls (P < 0.001, Figure 3(c)). Intranasal hAEC infusion was associated with a significant increase in area fraction of MBP (P < 0.001 vs. asphyxia-vehicle), to sham-control values (P = 0.13).
Changes in numbers of oligodendrocyte transcription factor 2 (Olig-2)-positive oligodendrocytes (a), 2′,3′-cyclic-nucleotide 3′-phosphodiesterase (CNPase)-positive-area fraction and CNPase-positive oligodendrocytes (b) and area fraction of myelin basic protein (MBP) (c) in the intragyral white matter of the first and second parasagittal gyri (IGWM1 and IGWM2, respectively) and the periventricular white matter (PVWM) 21 days after asphyxia. Sham-control group, white bars (n = 7), asphyxia-vehicle group, black bars (n = 6) and asphyxia-hAEC group, striped bars (n = 6). Photomicrographs of Olig-2-positive cells (d), CNPase-positive oligodendrocytes (e) and MBP staining (f) in the PVWM. The inserts show enlargement of the boxed area, with intact cells indicated by arrows. Asphyxia was not associated with any change in numbers of Olig-2-positive cells, but was associated with a significant reduction in CNPase and MBP, which was attenuated by hAEC treatment. Data are mean ± SEM. *P < 0.05 vs. sham-control, #P < 0.05 vs. asphyxia-vehicle, ANOVA + Tukey’s test. Scale bar = 50 µm.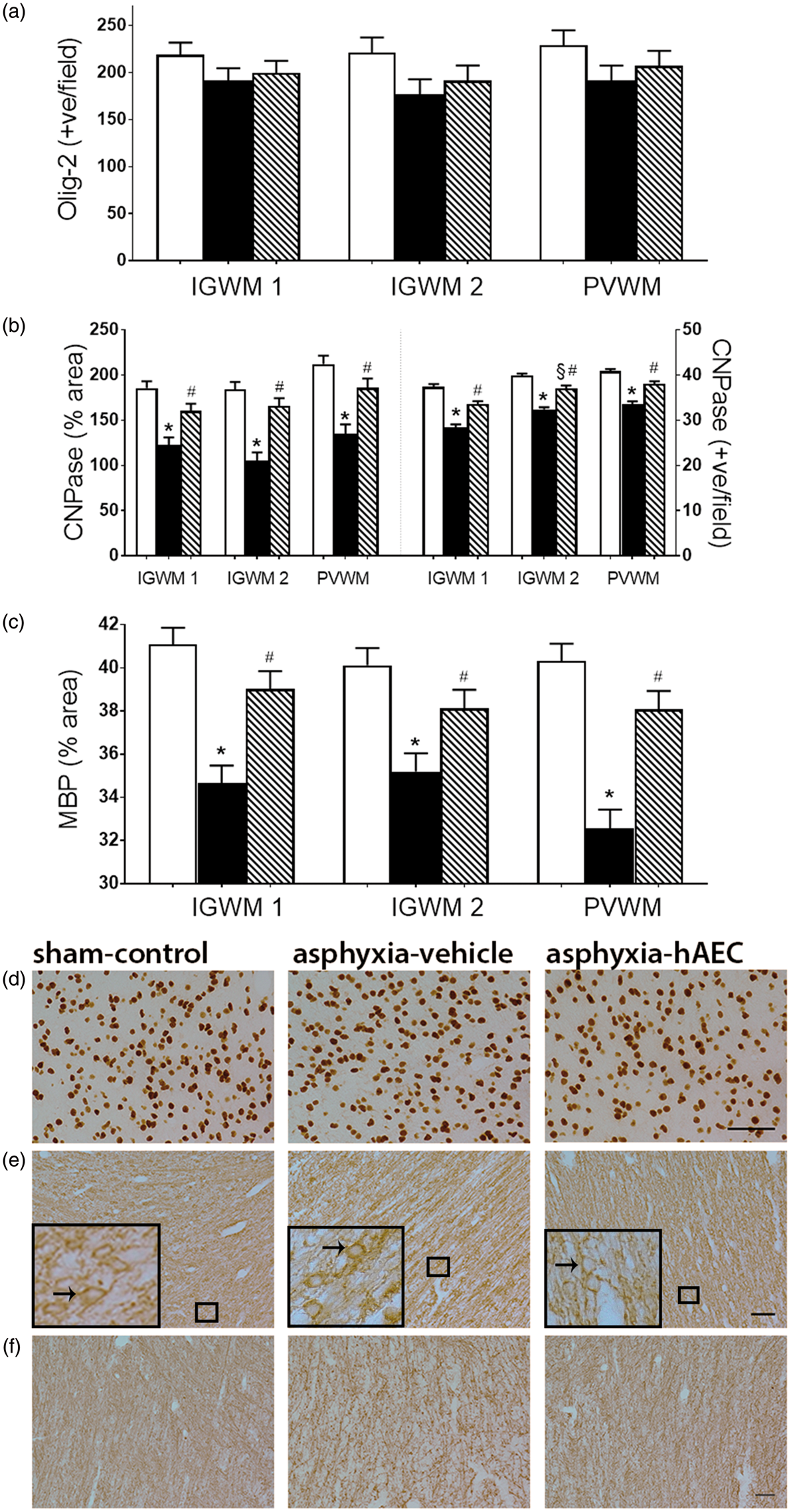
There was no significant effect of asphyxia or hAEC treatment on numbers of GFAP-positive astrocytes in the white matter (P = 0.11, Figure 4(a)), with no statistical interaction between group and region. In contrast, there was an increase in the area fraction of GFAP labeling after asphyxia (P < 0.001, Figure 4(b)). The asphyxia-hAEC group showed an overall reduction in GFAP area fraction (P = 0.001 vs. asphyxia-vehicle, Figure 4(b), to sham-control values (P = 0.29). Further, asphyxia was associated with markedly increased numbers of Iba-1 positive microglia in the white matter (P < 0.001 vs. sham-controls, Figure 4(c)). Asphyxia-hAEC was associated with reduced numbers of microglia (P = 0.002 vs. asphyxia-vehicle, Figure 4(c)), to sham control values (P = 0.08).
Changes in anti-glial fibrillary acidic protein (GFAP)-positive astrocytes (a), GFAP-positive area fraction (b), and ionized calcium binding adaptor molecule 1 (Iba-1)-positive microglia (c) in the intragyral white matter of the first and second parasagittal gyri (IGWM 1 and IGWM 2, respectively) and the periventricular white matter (PVWM). Sham-control group, white bars (n = 7), asphyxia-vehicle group, black bars (n = 6) and asphyxia-hAEC group, striped bars (n = 6). Photomicrographs from the IGWM of GFAP (d) and Iba-1-positive microglia (e). Asphyxia was associated with a significant increase in GFAP-positive astrocyte area and numbers of Iba-1-positive microglia. hAEC treatment significantly attenuated this increase in GFAP-positive astrocytes and Iba-1-positive microglia. Arrows show GFAP-positive or Iba-1-positive cells, respectively. Data are mean ± SEM. *P < 0.05 vs. sham-control, #P < 0.05 vs. asphyxia-vehicle, ANOVA + Tukey’s test. Scale bar = 50 µm.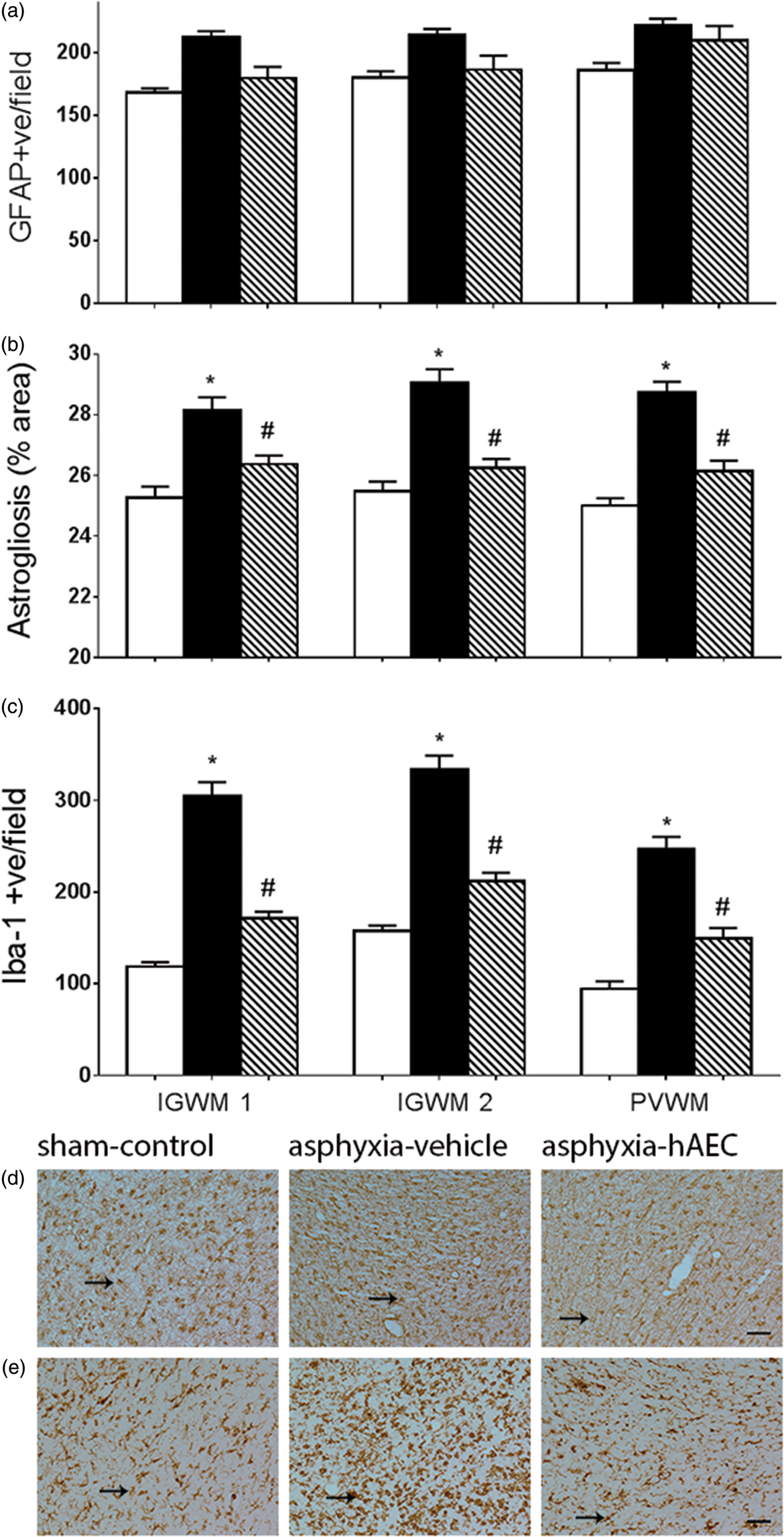
Cortical histology
Asphyxia was associated with a marked reduction in total cortical area after 21 days recovery, from 92.1 ± 30.6 mm2 in sham-controls to 73.9 ± 35.7 mm2 in the asphyxia-vehicle group (P = 0.015). The asphyxia-hAEC group showed an intermediate area of 85.2 ± 25.8 mm2 (N.S. vs. either sham-control or asphyxia-vehicle). There was no significant overall difference in numbers of cortical neurons between groups (Figure 5(a)). Asphyxia was associated with reduced synaptophysin staining compared to sham-controls (P = 0.001, Figure 5(b)), with no difference between asphyxia-hAECs and asphyxia-vehicle (P = 0.37).
Changes in neuronal nuclear antigen (NeuN)-positive neuronal number (a), synaptophysin (Synap) intensity (b), cleaved-caspase-3-positive (CC-3) cells (c), ionized calcium binding adaptor molecule 1 (Iba-1)-positive microglia (d) and tumor necrosis factor (TNF)-α intensity (e) in the cortex. Sham-control group, white bars (n = 7), asphyxia-vehicle group, black bars (n = 6) and asphyxia-hAEC group, striped bars (n = 6). Photomicrographs of NeuN-positive neurons (f), synaptophysin intensity (g), Iba-1-positive microglia (h) and TNF intensity (i) in the first parasagittal gyrus (1 PSG). Asphyxia was not associated with significant loss of cortical neurons, but was associated with significant reduction in synaptophysin intensity and increased numbers of CC-3-positive cells. hAEC administration was associated with significant attenuation of numbers of CC-3-positive apoptotic cells in the 1 PSG and LC (lateral cortex), but no significant effect in the 2 PSG (second parasagittal gyrus) or on synaptophysin staining in all areas assessed. Asphyxia was associated with an increase in Iba-1-positive microglia, which was partially attenuated by hAEC treatment. Data are mean ± SEM. §P < 0.05 vs sham-control, *P < 0.01 vs sham-control, #P < 0.05 vs asphyxia-vehicle, ANOVA + Tukey’s test. Scale bar = 50 µm.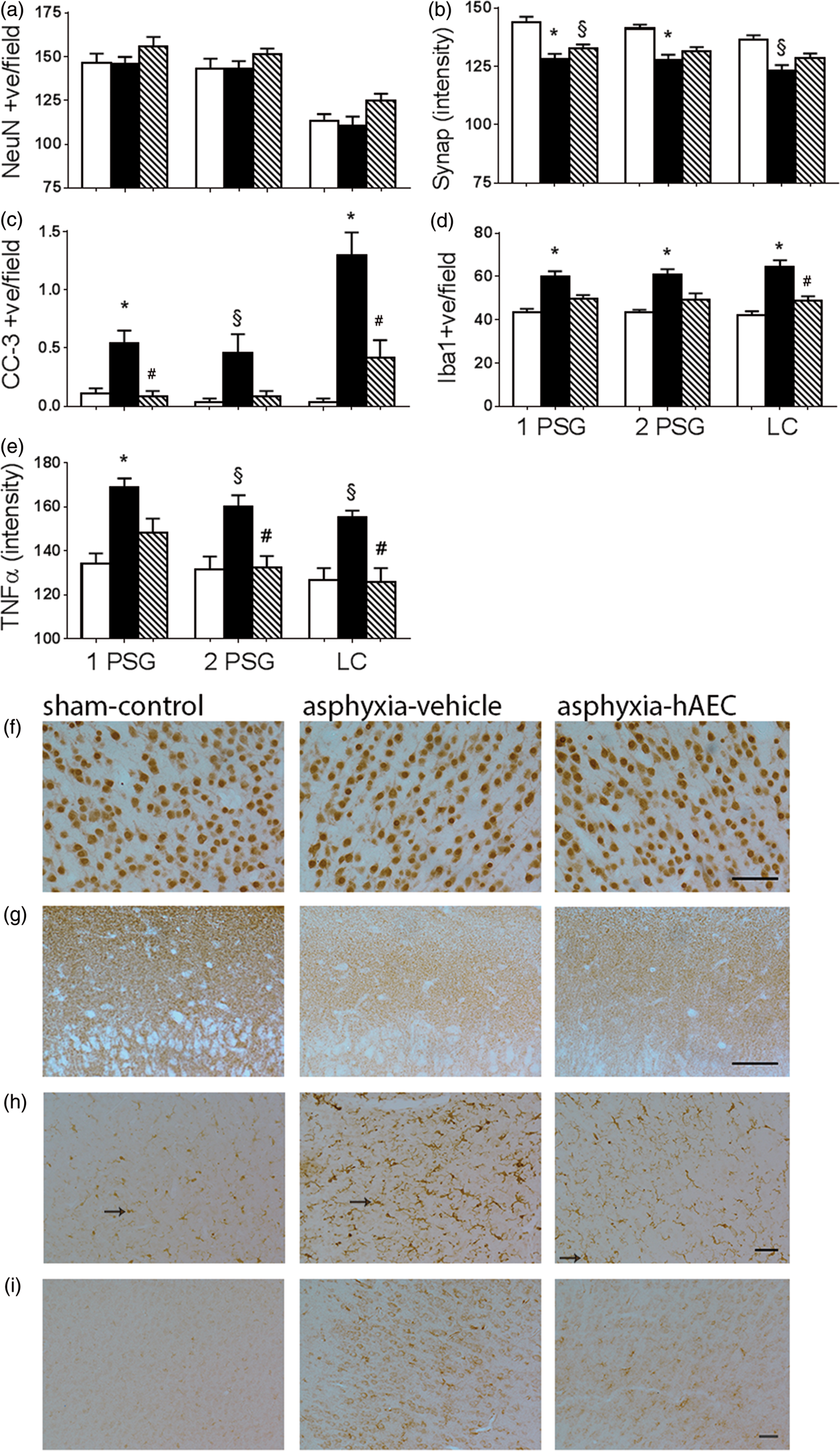
Further, numbers of caspase-3 positive apoptotic cells were increased after asphyxia compared to sham-controls (P < 0.0001, 5(c)), and reduced by hAEC treatment (P < 0.0001 vs. asphyxia-vehicle). There was a statistical interaction between region and group (P = 0.049), such that post hoc tests suggested greater residual caspase-3 induction in the asphyxia-vehicle group in the lateral cortex compared with the other cortical regions.
Asphyxia was also associated with a small increase in numbers of Iba-1 positive microglia in the cortex compared to sham-controls (P = 0.003, Figure 5(d)). hAEC treatment was associated with a statistically borderline reduction in cortical microglia (P = 0.05 vs. asphyxia-vehicle), to sham-control levels (P = 0.40 vs. sham controls). Post hoc testing suggested that hAEC treatment was associated with intermediate numbers of microglia in the parasagittal cortex that were not significantly different from sham or asphyxia-vehicle, but a significant reduction in the lateral cortex (P = 0.036 vs. asphyxia-vehicle, Figure 5(d)). Similarly, the intensity of TNFα staining was increased in the cortex after asphyxia (P < 0.008, Figure 5(e)), which was significantly reduced in the asphyxia-hAEC group (P < 0.022 vs asphyxia-vehicle), to sham-control values (P = 0.86).
Basal ganglia histology
Asphyxia was associated with loss of neurons in the caudate nucleus and the putamen of the striatum compared to sham-controls (P = 0.02, Figure 6(a)). hAEC treatment was associated with improved neuronal survival (P = 0.035 vs. asphyxia-vehicle), which on post hoc testing was significant in the putamen (P = 0.047), with no statistically significant change in the caudate nucleus. Asphyxia was associated with an overall increase in striatal Iba-1 positive microglia (P < 0.0001, Figure 6(b)). This was reduced by hAEC treatment (P < 0.0001 vs. asphyxia-vehicle), to sham-control values (P = 0.24). Further, asphyxia was associated with increased numbers of cleaved caspase-3-positive apoptotic cells compared to sham-controls in both the caudate nucleus and putamen (P < 0.0001, Figure 6(c)), which was reduced in the asphyxia-hAEC group (P = 0.02 vs. asphyxia-vehicle), and was not significantly different from sham-controls (P = 0.08).
Change in numbers of neuronal nuclear antigen (NeuN)-positive neurons (a), ionized calcium binding adaptor molecule 1 (Iba-1)-positive microglia (b) and cleaved caspase-3 (CC-3)-positive apoptotic cells (c) in the caudate nucleus and putamen of the striatum. Sham-control group, white bars (n = 7), asphyxia-vehicle group, black bars (n = 6) and asphyxia-hAEC group, striped bars (n = 6). Photomicrographs of NeuN-positive neurons (d), Iba-1-positive microglia (e) and CC-3-positive cells (f), in the caudate nucleus of the striatum. Asphyxia was associated with a significant reduction in NeuN-positive neurons, increased Iba-1-positive microglia and increased CC-3-positive apoptotic cells in the caudate nucleus and putamen. hAEC administration increased NeuN-positive neurons overall, which was significant on post hoc testing in the putamen, with a corresponding reduction of CC-3-positive apoptosis and Iba-1-positive microglia in both nuclei. Data are mean ± SEM. §P < 0.05 vs sham-control, *P < 0.01 vs. sham-control, #P < 0.05 vs. asphyxia-vehicle, ANOVA + Tukey’s test. Scale bar = 50 µm.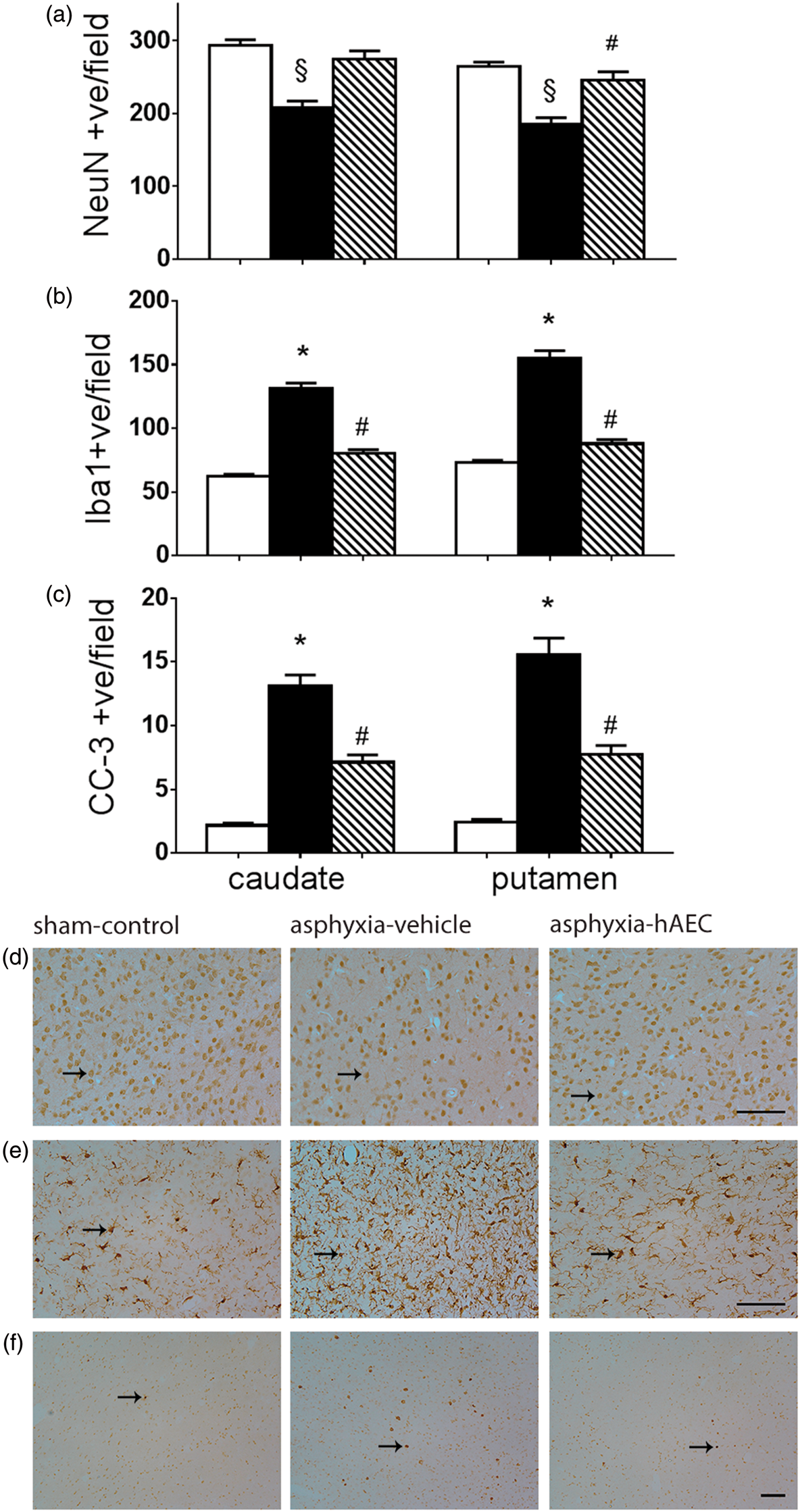
Discussion
The most common pathological substrate of long-term disability in preterm infants is diffuse white matter loss and long-term myelination failure, 5 with reduced cortical and subcortical grey matter volumes.32,33 Neurodevelopmental outcomes are independently correlated with the magnitude of both white matter and grey matter deficits. 34 At present, there are no specific treatments to prevent injury or subsequent impaired neurodevelopment. In the current study, we demonstrate for the first time that delayed, repeated intranasal infusion of hAECs, after asphyxia in utero, increased the proportion of mature OLs and improved myelination in the white matter tracts, reduced neuronal loss in striatal nuclei, and partially alleviated loss of brain weight and cortical and white matter area after 21 days recovery. Improved maturation of white matter was associated with reduced astrogliosis and microgliosis, reduced expression of the inflammatory mediator TNFα, and suppression of caspase-3 positive apoptosis. These findings suggest that delayed treatment with hAECs can attenuate white matter dysmaturation, inflammation, and ongoing low-level apoptosis in both white and grey matter.
The model of asphyxia induced by acute umbilical cord occlusion used in this study led to a clinically relevant pattern of preterm brain injury characterized by diffuse white matter loss without focal necrosis, subcortical grey matter loss and evolving impairment of cortical development, consistent with the clinical pattern of injury after acute perinatal asphyxia on magnetic resonance imaging. 35 We now report that this paradigm leads to a chronic reduction in the area of white matter, with hypomyelination and reduced area fraction and proportion of immature/mature (CNPase-positive) OLs despite no changes in total (Olig-2-positive) OLs. These changes are consistent with impaired maturation of OLs. 5 Importantly, this occurs despite complete resolution of the insult so that fetuses matured in a stable, normoxic environment.
We have previously reported that this paradigm triggers acute loss of pre-OLs three days post-asphyxia, but that new cells proliferate to restore total numbers of OLs by day seven.36–38 Similarly, after reversible cerebral ischemia in preterm fetal sheep, Riddle et al. 39 reported that numbers of pre-oligodendrocytes recover in the first week and can double in two weeks, 39 but pre-myelinating OLs were significantly reduced over the same period. These findings suggest that pre-OLs undergo active restorative proliferation after a hypoxic-ischemic (HI) insult in utero, but may fail to differentiate into myelin producing cells.39–42 We now show that repeated intranasal hAEC administration improved both the proportion of mature OLs, and restored myelination to near sham control levels.
The precise mechanisms of maturational arrest are not known, 5 but likely include excessive accumulation of hyaluronic acid produced by reactive astrocytes,42,43 chronic microglial activation, and changes in the cell cycle of oligodendrocyte progenitors. 44 The present study is consistent with this, as the improved maturation of white matter tracts seen after hAECs treatment was closely associated with attenuation of the chronic microgliosis and astrogliosis. Astrocytes and microglia are important regulators of normal development of oligodendrocyte precursor cells and myelination 45 and contribute to brain recovery in part though the neuro-restorative aspects of the innate immune system. 46 However, they also contribute to impairment of oligodendrocyte maturation.6,40,42,45
Both microglia and astrocytes release pro-inflammatory mediators, including TNFα as observed in this study, in addition to interleukins and interferon signaling proteins.45,47 Preterm infants who develop white matter injury have high levels of IL-1β, IL-6 and TNFα in the amniotic fluid, 48 plasma and cerebrospinal fluid at birth. 49 Elevated levels of TNFα and other inflammatory markers in preterm infants 21–28 days after birth are correlated with impaired brain development and adverse neurodevelopmental outcomes at 2 and 10 years of age.50–52 The mechanisms mediating persistent inflammation are multifactorial. Some infants are exposed to repeated inflammatory stimuli, 53 and thus there is a failure to remove the stimulus. For others, the biological cues that signal inflammation resolution both centrally and peripherally may be inadequate or interrupted, leading to chronic non-resolving inflammation.54,55
hAEC infusion into the lungs of newborn immature lambs before ventilation down-regulated both microglia and astrocytes, as well as levels of TNFα during inflammation induced by LPS,20,21 and microglial activation in intragyral, but not PVWM. 22 Similarly, in a mouse model of autoimmune encephalomyelitis, hAECs suppressed both specific and non-specific T-cell proliferation, decreased pro-inflammatory cytokine production, and inhibited the activation of stimulated T-cells. 15 hAECs also modulate macrophage recruitment and promote transition of macrophages from the potentially deleterious M1 to the beneficial M2 phenotype in liver 56 and lung injury. 57 Collectively, these data support the hypothesis that hAECs provide protection by moderating both central and peripheral immune responses.
In this study, asphyxia did not affect cortical neuronal density, but was associated with reduced brain weight and cortical area and neuronal loss in the caudate nucleus and putamen of the striatum, with microgliosis and ongoing upregulation of caspase-3 positive apoptosis in both the cortex and subcortical nuclei. The first of the three doses of hAECs was administered 24 h after asphyxia, demonstrating that at least some cell loss is still potentially reversible well outside the therapeutic window for treatment with hypothermia in the same paradigm as used here, 58 through additional active mechanisms supported by hAECs. Alternatively, there is evidence that exogenous stem cells can promote endogenous precursors and so might play a role in neurorestoration. 59 Prematurity is associated with reduced volume and complexity of cerebral cortical grey matter, and reduced size of the thalamus, hippocampus and brainstem nuclei, at term-equivalent and later, as previously reviewed. 60 Since the preterm cortex is consistently spared from acute injury, both clinically,61,62 and experimentally,38,63 the long-term maldevelopment is proposed to involve secondary mechanisms such as loss/disruption of neural transmission, leading to secondary cell loss and simplification of neural architecture. 7
Although therapeutic hypothermia is not currently clinically established for treating preterm HI encephalopathy, it is likely that the combination of early hypothermia to suppress evolving cell death, 58 with delayed hAEC treatment to promote normalization of neural maturation, as suggested by the present study, might be more beneficial or allow a wider window of opportunity for treatment. This is a significant consideration for preterm brain injury as the timing of injurious events is often unclear, and may begin before birth. 7 Moreover, there is some evidence that hypothermia alone may be less effective after infection-sensitized hypoxia-ischemia in neonatal rats. 64 Given the high rate of exposure of preterm infants to infection/inflammation, 6 the combination of hypothermia with interventions such as hAECs that have anti-inflammatory properties may be particularly beneficial.
In absolute terms, the total number of cells undergoing apoptosis after three weeks was relatively low; however, there was still increased apoptosis defined by a combination of caspase-3 expression and the presence of apoptotic bodies compared to sham-controls. This finding supports the hypothesis that upregulation of physiological apoptosis contributes to the long-term impairment of brain growth after preterm brain injury. In the developing brain, damaging, low-grade inflammation may be triggered by this cell death, while conversely, activated glia down-regulate trophic factors required for survival, proliferation and maturation.6,65,66 Thus, at least in part, hAECs likely improved neuronal survival by promoting local trophic factor release.15,67 For example, in previous studies, lipoxin A4 released by hAECs triggered expansion of regulatory T cells and limited inflammatory infiltration. 68 Supporting this, other studies have found that lipoxin A4 inhibits microglial activation, 69 and can regulate neural stem cell proliferation and differentiation. 70
There is increasing experimental evidence that intranasal administration, as tested here, is an effective and relatively non-invasive route of administration. 13 The barrier between the nasal cribriform plate and the brain parenchyma is minimal, and so allows cells to move up the olfactory nerves towards the areas of injury, possibly though tracking of the chemokine stromal derived factor. 71 In rats, it has been shown that intranasal administration allows rapid dispersion throughout the brain and into the brain stem within 4 h. 71 Clinically, non-cell treatments are already routinely administered intranasally, and offer better accumulation in the brain or CSF. 72 Naturally, the absolute distance traveled is smaller in rodents than it would be in humans and the human olfactory track is not as well developed as in sheep. However, the present study in a large animal paradigm offers encouraging proof of principle that it may be worth exploring in humans.
Treatment was delayed until 24 hours after asphyxia in the present study to test a realistic delay for diagnosis and evaluation. Studies using other stem cell types support the potential for very delayed treatment. In term equivalent mice, MSCs given intranasally as a single dose of 0.5 × 106 at 10 days after hypoxia-ischemia could improve outcomes assessed at 9 months, 73 but treatment was less beneficial if delayed to 17 days. 74 Similarly, bone-derived mesenchymal stem cells given to neonatal rats at one week after hypoxia-ischemia at postnatal day 7 improved numbers of striatal medium-spiny projection neurons at 90 days, and much higher doses (750,000–1,000,000 MSCs vs. 85,000–120,000 MSCs per animal) were associated with behavioral improvements. 75 In preterm fetal sheep, a single bolus dose of multipotent adult progenitor cells, mesenchymal stem cells or cord blood cells given immediately or 12 h after asphyxia reduced white matter loss at 7 to 10 days,47,76,77 but when treatment with cord blood cells was delayed to 5 days, it was not beneficial. 47 Repeated administration of stem cells, as tested in the present study, is likely to be important for optimal benefit. In postnatal day 9 rats, a single intracerebral injection of mesenchymal stem cells three days after hypoxia-ischemia improved sensorimotor function and reduced infarct size, 14 but a second injection of cells 10 days after hypoxia-ischemia further enhanced sensorimotor improvement and promoted recovery of staining for myelin basic protein and microtubule-associated protein 2, which is expressed by neurons, and may have enhanced axonal remodeling. 14
In conclusion, the present study is the first to demonstrate that delayed, repeated intranasal infusions of hAECs is associated with improved white matter maturation and reduced neuronal injury in a large animal, translational model of asphyxia. hAECs have advantages over other stem cell types in that they are readily available with low ethical concern regarding harvesting, and are non-tumorigenic and non-immunogenic. Further studies are required to directly assess the impact of cell therapy on long-term neurological recovery, to determine the window of opportunity for delayed treatment after different insults, and to optimize hAEC treatment for perinatal HI encephalopathy at different ages and for other types of insults. Nevertheless, this study demonstrates that hAECs have potential clinical utility to help promote normalization of brain development after preterm birth.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Research Council of New Zealand [grant numbers 11/576, 12/613]; and LGH was an Auckland Medical Research Foundation Doctoral Scholar [grant number 1211003]. SM, GJ, EW & RL acknowledge the support of the Victorian Government’s Operational Infrastructure Support Program.
Acknowledgements
The authors thank Maree Schollum for technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
These experiments were conducted in the Fetal Physiology and Neuroscience Group laboratory, at the University of Auckland. LB and AJG conceived the hypothesis, experimental design and analysis protocols. LGH and MF were responsible for cell preparation and data collection. SM, EW, RL and GJ collected and provided the hAECs and contributed to the study design; MF assisted with cell culture and preparation of cells. LGH, CAL, JD, GW, LB and AJG were responsible for data analysis. LGH drafted the manuscript. All authors were involved in data interpretation, and in the editing and revision of the manuscript. All authors qualify for authorship on the paper, are listed on the paper and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
