Abstract
The potential effects of changes in blood flow on the delivery and washout of radiotracers has been an ongoing question in PET bolus injection studies. This study provides practical insight into this topic by experimentally measuring cerebral blood flow (CBF) and neuroreceptor binding using simultaneous PET/MRI. Hypercapnic challenges (7% CO2) were administered to non-human primates in order to induce controlled increases in CBF, measured with pseudo-continuous arterial spin labeling. Simultaneously, dopamine D2/D3 receptor binding of [11C]raclopride or [18F]fallypride was monitored with dynamic PET. Experiments showed that neither time activity curves nor quantification of binding through binding potentials (
Keywords
Introduction
The effect of changes in radiotracer delivery on outcome measures of receptor-specific positron emission tomography (PET) studies has been a recurrent discussion in the literature.1–3 Pharmacological challenges or activation paradigms can cause large and potentially rapid changes in cerebral blood flow (CBF), as shown with PET flow tracers, blood oxygen level dependent (BOLD), CBF or cerebral blood volume (CBV) measurements in experiments using functional magnetic resonance imaging (fMRI) techniques.4–9 Typically, the purpose of pharmacological challenges in receptor imaging studies with PET is to measure reductions in PET signal that can be attributed to reductions in specific receptor availability. However, if the delivery and washout of the tracer are also altered during the timeframe of a PET study or in conjunction with a pharmacological challenge, a central question is how PET measures of receptor binding could be affected.
In PET blocking or competition studies, it is usually assumed that the kinetic parameters of the radiotracer are constants throughout a study, except for the binding term
Previously, investigations have simulated whether specific binding measures can be affected by changes in blood flow. Various assumptions have been made to simulate the precise mechanism of how blood flow could affect kinetic parameters in a compartmental model. Several studies postulated that blood flow changes affect both the plasma-to-tissue transfer rate
One experimental investigation in humans was reported 14 in which hyperventilation was used to decrease blood flow physiologically while measuring dynamic PET time-activity data from [11C]raclopride. While changes in the overall shape of TACs were observed, decreases in blood flow for the duration of the experiment did not affect binding potentials of [11C]raclopride. Other radiotracers, especially those that may have a large extraction fraction in vivo, irreversible binding, or very high affinity, are suspected to be vulnerable to bias arising from changes in flow. Cumming et al. 15 postulated that differences in [18F]fallypride binding potentials, an example of a high affinity radiotracer, are due to differences in CBF values and thus tracer delivery in healthy volunteers and that these could even be used as a surrogate marker for CBF. This finding and the confound of CBF changes would potentially question the validity of studies that have used [18F]fallypride to investigate dopamine displacement and characterize extra-striatal D2/D3 receptor binding.16,17
Simultaneous measurements of blood flow and receptor binding would be able to experimentally measure the effects of blood flow on radiotracer binding. By repeatedly injecting a short-lived blood flow tracer in the presence of a long-lived neuroreceptor ligand, measurements of both CBF and receptor occupancy are possible using PET alone, 18 but such experiments are highly technically demanding and have not been performed to our knowledge. With the availability of combined PET/MRI scanners, however, it is now possible to continuously monitor CBF using arterial spin labeling techniques19,20 during a neuroreceptor PET imaging experiment. This method has the advantage that changes in flow can be tracked continuously at high temporal resolution (seconds to minutes) throughout a PET study. By perturbing CBF using modest respiratory challenges between or during the scan in a way that does not directly influence neuroreceptor availability, the effect of changes in blood flow on radiotracer binding can be directly evaluated using simultaneous PET/MRI technology.
The goal of this study was to experimentally measure controlled changes in CBF and their influence on PET time activity curves (TACs) with simultaneous PET/MRI in a variety of settings simulating a range of experimental paradigms of general interest. CBF was increased by inducing hypercapnia in non-human primates (NHP) for specific periods of time during the dynamic PET acquisition, and changes in CBF were simultaneously measured with arterial spin labeling. Two tracers specific to the dopamine D2/D3 receptors were investigated: [11C]raclopride, a well-validated tracer for displacement studies, and [18F]fallypride, a tracer previously suggested to be susceptible to flow changes. We carried out simulations of flow changes and compared these to experimental results, thus providing insight into the assumptions used for kinetic simulations.
Materials and methods
Animal model
Imaging studies were carried out in two healthy baboons (
Study design with hypercapnia
To induce hypercapnia (and thus increase CBF), animals were ventilated with 7% CO2 mixed with air for a period of 10–15 min at a time. Exposure to 7% CO2 was repeated in a block design fashion, interleaved with a period of 10–15 min of ventilation with air.
Two experimental conditions were carried out with [11C]raclopride that mimicked the timing normally used for within-scan pharmacological or behavioral challenges: In one condition (1×CO2), one hypercapnia period of 12 min was applied 34 min after injection of the radiotracer. In another condition (3×CO2), hypercapnia was applied three times for a duration of 10 min, starting at 20, 40 and 60 min after tracer injection, which would allow to observe the effects of increasing blood flow on the TAC in a dynamic fashion. Experiments with [11C]raclopride were carried out in the male baboon. Using [18F]fallypride, a baseline and two varying hypercapnia conditions were tested in the female baboon: One experimental design employed five hypercapnia challenges (5×CO2) in an ON-OFF block design, with the timings representing within-scan challenge conditions. Each hypercapnia challenge lasted 12 min, with onsets at 30, 60, 90, 120 and 150 min. after injection of the radiotracer. In a different experimental design (2×CO2), the onset of the first hypercapnia challenge was timed to start 4 min before injection of the radiotracer and to last for 16 min. This design allowed for measurements of flow effects during the uptake of the tracer. An additional hypercapnia period was added at the end of the scan at 110 min (for a duration of 15 min) after radiotracer injection.
PET/MR image acquisition
Simultaneous PET and MR data were acquired on a whole-body human PET/MR scanner at 3 T (mMR, Siemens AG, Healthcare Sector, Erlangen Germany). A custom-built PET compatible 8-channel NHP receive array together with the body coil for transmit was used for MR imaging. 21 For quantitative flow measurements, pseudo-continuous arterial spin labeling (pcASL) optimized for NHPs 22 was employed for the entire duration of the experiment (FOVread = 128 mm, resolution = 2 × 2×4 mm, TE = 15 ms, TR = 3.5 s, post-labeling delay = 800 ms, BW = 1474 Hz). The phased-array receiver enabled a two-fold acceleration with GRAPPA 23 in the anterior-posterior direction.
PET emission data were acquired in list-mode format for 90 min ([11C]raclopride) or 150–180 min ([18F]fallypride) starting with radiotracer injection, which was given as a bolus by hand injected over 30 s. Injected activities were kept similar for each radiotracer injection, with an average of 5.01 ± 0.02 mCi for [11C]raclopride and 3.12 ± 0.08 mCi for [18F]fallypride experiments. Average specific activities of the produced radiotracers were 1.1 mCi/nmol for [11C]raclopride and 22.7 mCi/nmol for [18F]fallypride. Images were reconstructed with the ordinary Poisson expectation maximization algorithm with 3 iterations and 21 subsets. Corrections for scatter and attenuation of the head and the radiofrequency coil were applied during reconstruction. The final reconstructed volume consisted of 4 × 4×2 mm voxels in a 172 × 172×127 matrix, with framing intervals of 10 × 30 s, followed by 1 min frames.
ASL data analysis
Both MR and PET data were registered to the Saleem-Logothetis stereotaxic space, 24 in which regions of interest were defined standardly. All data analysis and processing were performed with open-access software. 25
ASL data were first motion-corrected and spatially smoothed with a 4 mm Gaussian kernel, after which they were skull-stripped and registered. To compute perfusion-weighted or relative CBF maps, a pairwise subtraction of tag and control data was performed. Images were averaged over experimentally defined blocks of baseline and hypercapnia conditions and normalized to the respective tag condition. Quantitative CBF voxelwise maps for each condition were computed for a single post-labeling delay as described in previous methods. 22
PET data analysis: Kinetic modeling
PET [11C]raclopride and [18F]fallypride data were analyzed with a GLM formulation of the three-parameter simplified reference tissue model (SRTM)26–28 using the cerebellum as a reference region. The SRTM is based on the following basic equations
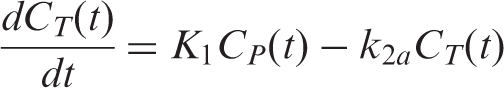
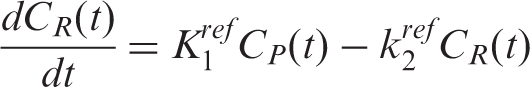
Substituting the expression for 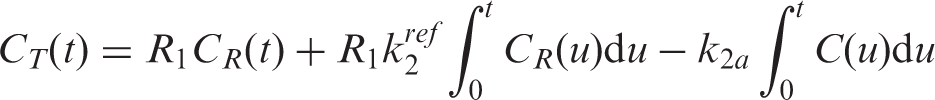
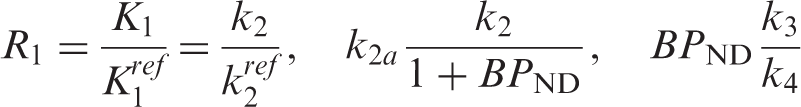
For [11C]raclopride, SRTM was used to analyze the full TAC (90 min of data acquisition). In a second analysis, only the first 20 min of the TAC was used for the SRTM analysis. Using the first 20 min of the scan provides an estimate to a no-flow-change condition in the same scan (within-scan baseline) because hypercapnia was only introduced after 20 min. In a third analysis, the first 60 min of the TAC were taken into account for an SRTM analysis for comparison purposes to the previous cases.
In addition to steady state kinetic modeling, analyses with a time-dependent 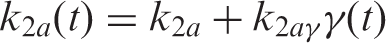
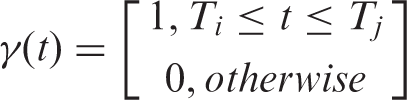
The parameters
The operational equation including the time-dependent binding term is thus expressed as
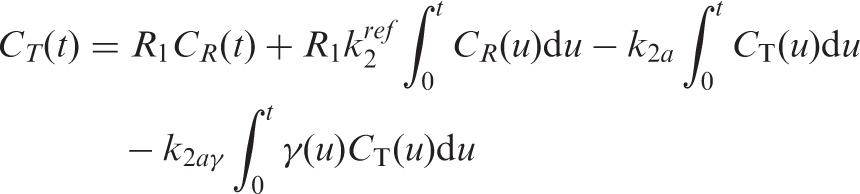
In order to quantify binding changes over time, the time-dependent binding quantity, dynamic binding potential
29
(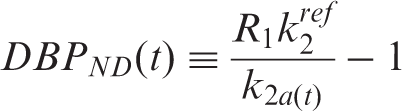
For each analysis, the error for estimating the binding potential (
Simulations: Reference tissue model
The effect of flow changes on radiotracer kinetics was simulated using a forward model implementation of the full reference tissue model (FRTM),
30
i.e. a two-tissue compartmental model, for the radiotracers [11C]raclopride and [18F]fallypride. The inputs to the forward model were a simulated plasma input function and kinetic rate constants
The plasma input function was calculated using a model that consists of two compartments representing the plasma concentration 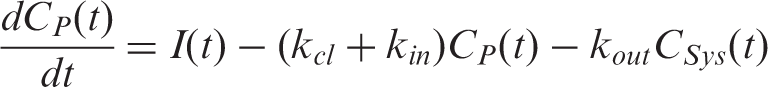
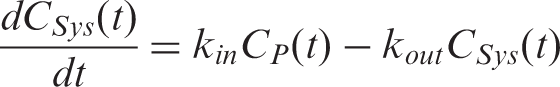
These equations were solved numerically for the plasma concentration
Simulations of the FRTM were based on the operational equations of a two-tissue compartmental model
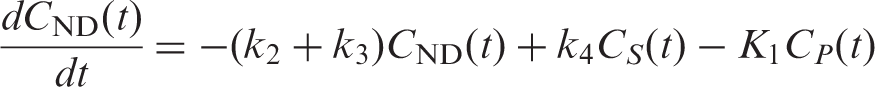
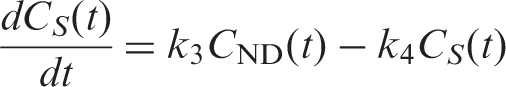
Noise was added to the simulated TACs using a noise model proposed by Logan et al.
31
The model uses random noise that is dependent on counting statistics and thus increases in later time frames according to the following formula
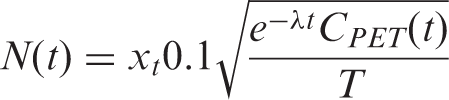
Parameter values for simulations of a specific binding region with [11C]raclopride were assumed to be
Simulations: Changes in blood flow
The hypercapnia interventions were modeled by including a time-dependent change in the kinetic rate constants
Changes in blood flow were simulated with the following assumptions: (i) Blood flow
For the purpose of validating assumptions, simulation results with the above assumptions were compared to the following alternative assumptions: (iv′) Surface area S stays constant, i.e. is flow-independent. (v′) Only
A larger parameter space, with kinetic rates (
Results
Experimental results: [11C]raclopride
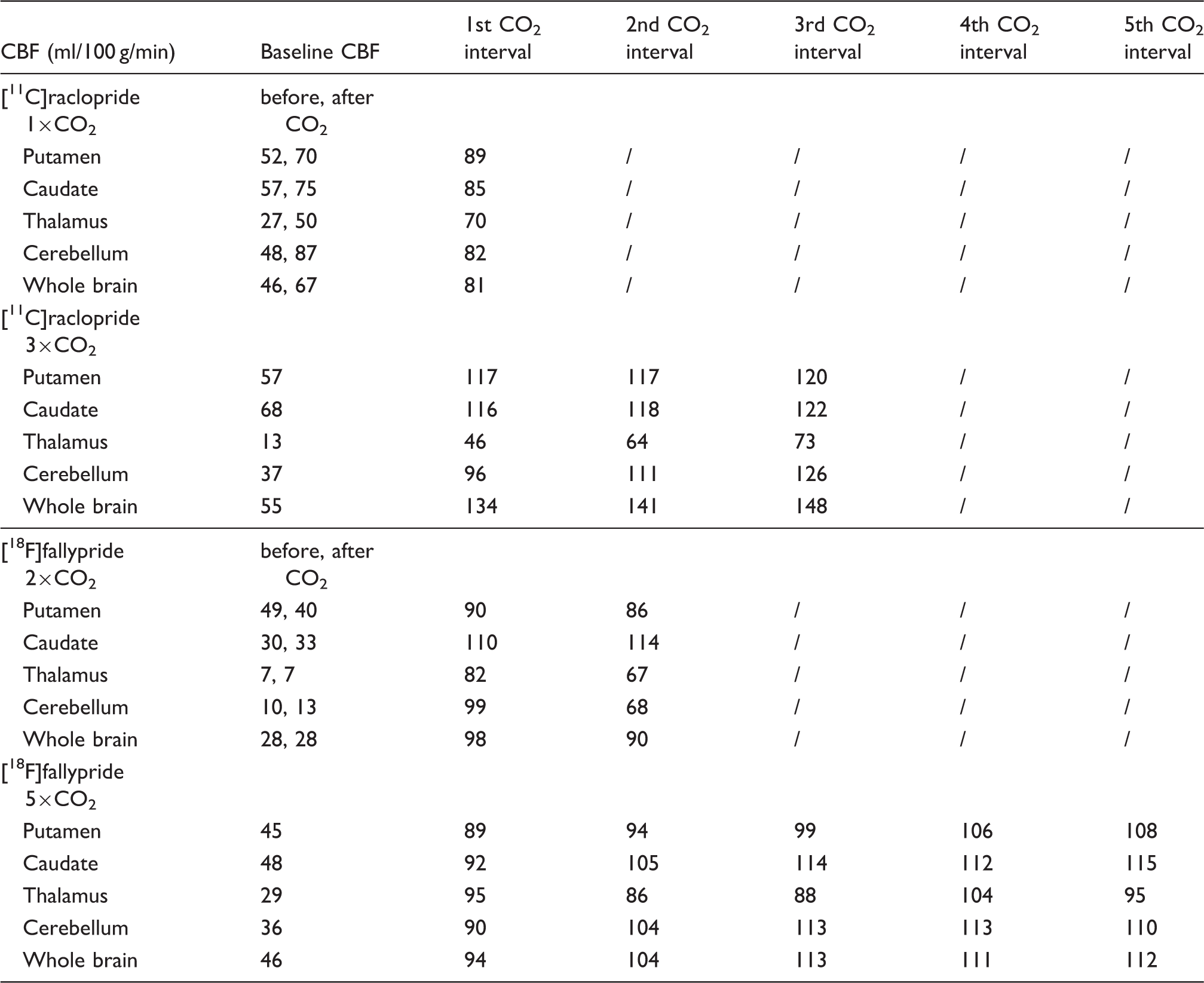
Experimental results from the 3×CO2 study are shown in Figure 1 as a representative scanning session for [11C]raclopride. Increases in CBF, as measured by pcASL, in response to the hypercapnic challenges were measured in all regions, albeit with different magnitudes, as shown for the putamen and cerebellum ROIs in Figure 1(c). Flow increased up to 110% in the putamen and up to 245% in the cerebellum, relative to baseline values. Quantitative analysis of the pcASL data in the putamen showed that flow increased from a baseline value of 59 ml/100g/min to 114, 116 and 119 ml/100g/min for the first, second and third hypercapnic challenge, respectively. In the cerebellum, baseline values of flow were lower (37 ml/100g/min) but increased to 96, 111 and 126 ml/100g/min for the three hypercapnic challenges. See Table 1 for a full list of quantitative CBF values during hypercapnia conditions.
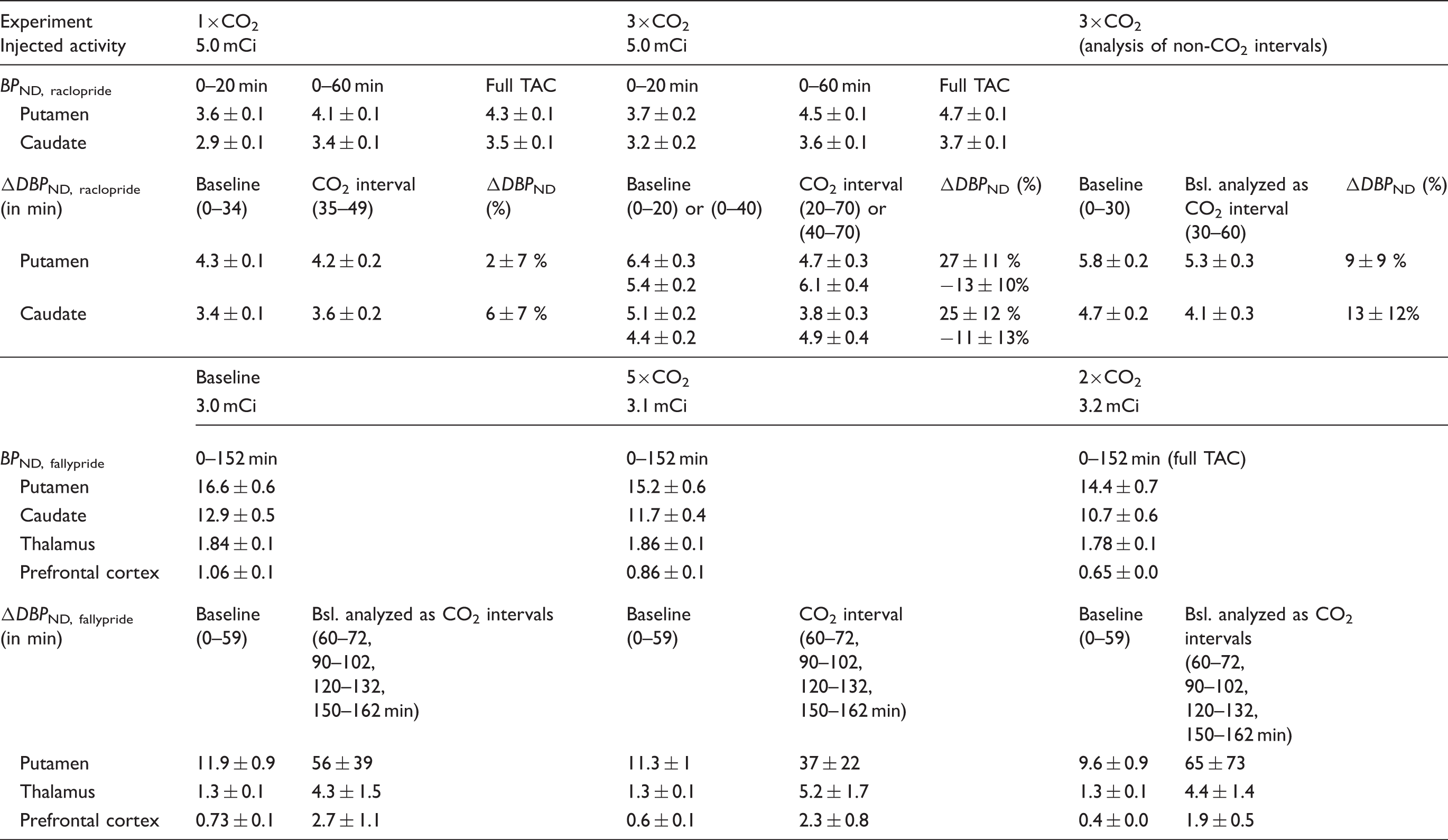
Experimental results from a [11C]raclopride bolus injection and hypercapnia challenges of 7% CO2 applied three times during the session (3×CO2). (a) Time activity curves for putamen and cerebellum show no effects during hypercapnia (blue-shaded areas). (b) Residuals between the kinetic modeling fit and data do not show any pattern associated with the timing of the challenge. (c) Measured flow changes with pcASL show increases in flow in the putamen and cerebellum ROIs. (d) End-tidal CO2 values correspond to measured flow changes. Absolute cerebral blood flow (CBF) values in ml/100g/min during baseline and for each hypercapnia interval. Note: CBF values were measured with pcASL and are listed in ml/100 g/min. The numbered CO2 columns denote chronological hypercapnia periods within each experiment. CBF: cerebral blood flow; pcASL: pseudo-continuous arterial spin labeling. Binding potential estimates for different intervals of the baseline and hypercapnia conditions. Note: SRTM was used for estimating SRTM: simplified reference tissue model; 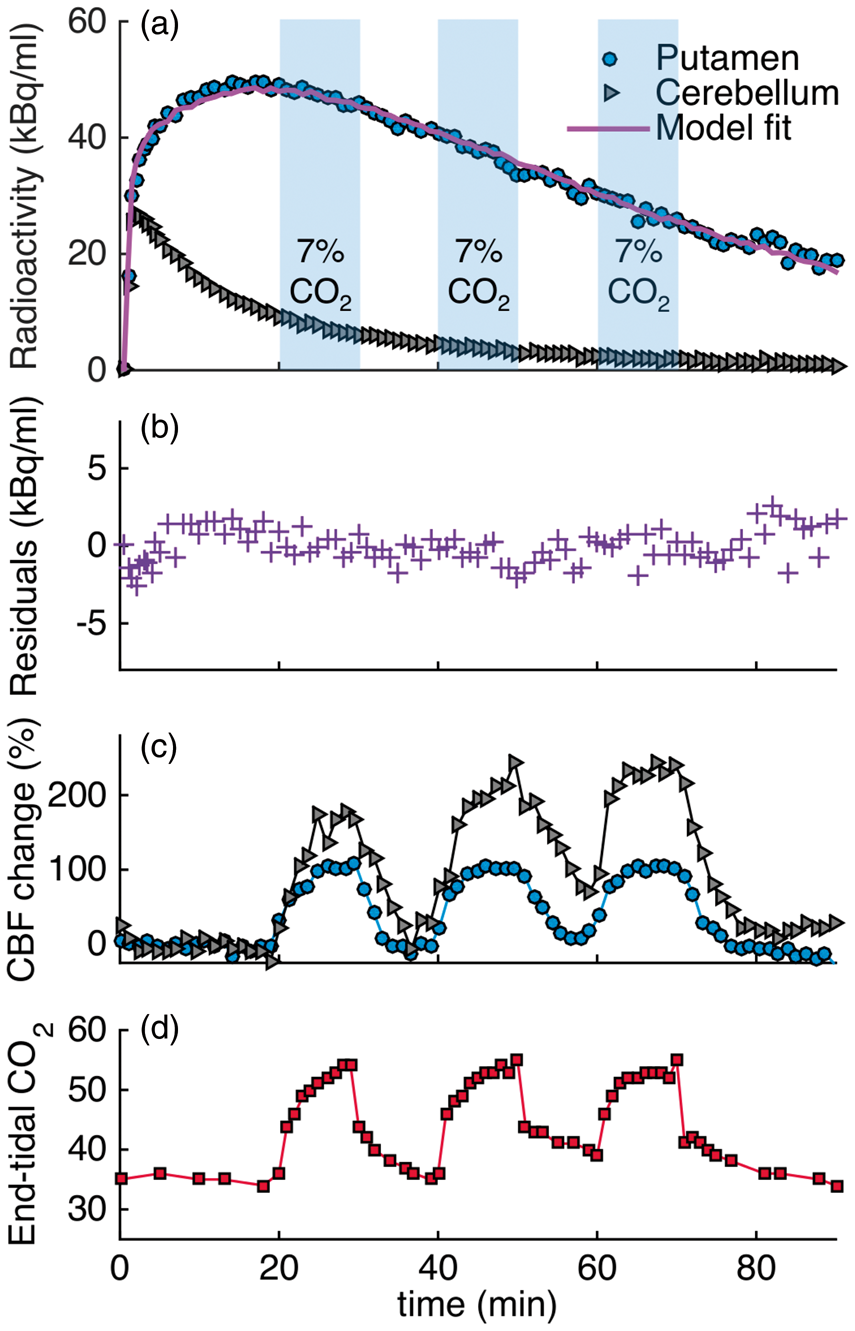
End tidal CO2 (etCO2, Figure 1(d)) correlated well in time with the measured changes in CBF by pcASL. Baseline values started at 35 mmHg and increased by 20 mmHg up to a maximum value of 55 mmHg during the administration of hypercapnia.
Figure 1(a) shows [11C]raclopride TACs for the putamen and cerebellum that were acquired simultaneously with the flow changes. Qualitatively, the TACs do not show prominent changes due to the hypercapnia condition. In a separate experimental session (1×CO2), in which hypercapnia was applied once for a duration of 12 min at 34 min after injection of the radiotracer, no qualitative change in the TAC was observed either.
Quantification of the [11C]raclopride PET data with the SRTM kinetic model and constant kinetic rates resulted in good fits to the entire TAC (solid purple line in Figure 1(a)). Figure 1(b) shows the residuals between the fit and the data. No periodic changes that match the flow pattern are detectable. Based on the Durbin–Watson test of autocorrelation, the residuals are not significantly correlated at the
In a second analysis, a time-dependent
Experimental results: [18F]fallypride
Figure 2 shows the experimental results for applying five hypercapnia challenges (5×CO2) in an ON-OFF block design over the timecourse of the experiment of 180 min. The timing of the cyclic hypercapnia challenge is indicated by the shaded areas of 7% CO2 exposure. TACs did not show any noticeable changes that matched the pattern of flow modulations (Figure 2(a)). Relative blood flow values measured with pcASL are shown in Figure 2(b). In the putamen, flow increased by up to 178% and in the cerebellum flow increased by up to 246% during hypercapnia. Quantitative CBF values in putamen were 45 ml/100g/min at baseline and increased up to 108 ml/100 g/min during the fifth hypercapnic condition with the highest average value. In cerebellum, baseline values started at 36 ml/100 g/min and increased up to 113 ml/100 g/min. Initial baseline etCO2 values were 32 mmHg and increased to a maximum of 52 mmHg during hypercapnia, as shown in Figure 2(c). The temporal changes in etCO2 matched the pattern of hypercapnia conditions and flow changes very well.
Experimental results from an [18F]fallypride bolus injection and hypercapnia challenges of 7% CO2 applied five times for 12 min during the experiment (5×CO2). (a) Time activity curves for putamen, thalamus and cerebellum are not noticeably affected by hypercapnia (timing of challenge indicated by shaded areas). (b) Measured cerebral blood flow changes with pcASL show increases in flow in the putamen and cerebellum ROIs during hypercapnia. (c) End-tidal CO2 values are well-matched with flow changes for the duration of the experiment.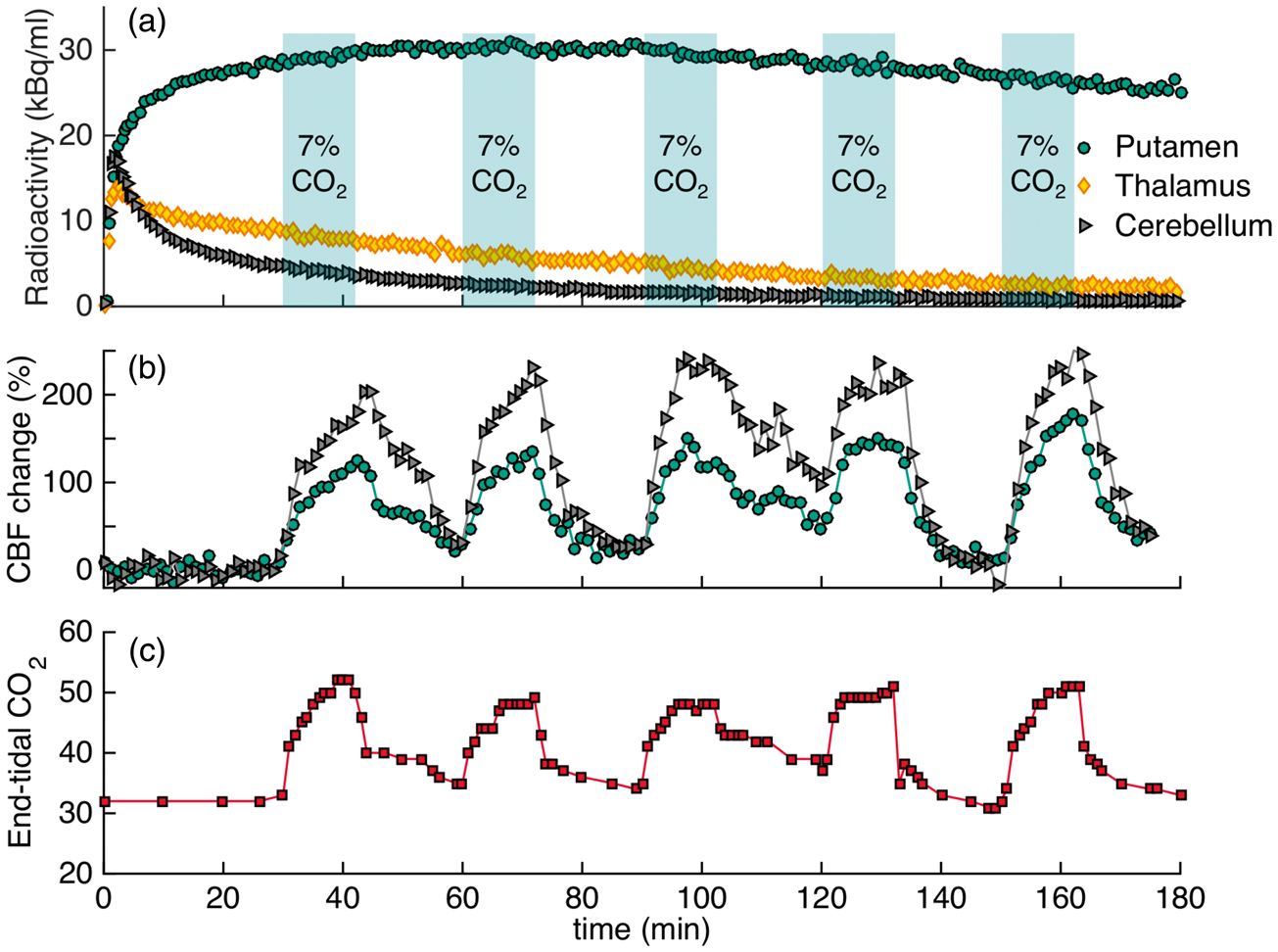
To assess flow effects during radiotracer uptake, the timing of the hypercapnia challenge was altered to start before and persist during injection of the radiotracer in a separate [18F]fallypride experiment (2×CO2, Figure 3). Despite the change in the timing of the challenge, no changes in the shape of the TACs were observed. Changes in flow, measured by pcASL, and etCO2 values measured throughout the timecourse of the experiment are shown in Figure 3(b) and (c). Flow changes corresponded well to the changes observed in etCO2, as shown in the graphs. The flow measurements show that CBF was increased by up to 145% in the putamen during hypercapnia. Average quantitative values in the putamen ranged from 40 ml/100g/min at baseline up to 90 ml/100g/min during hypercapnia. Additional regions of interest and their CBF values are listed in Table 1.
Experimental results from an [18F]fallypride bolus injection and 7% CO2 hypercapnia challenges applied before the start of the experiment to observe flow-induced uptake differences (2×CO2). (a) The shapes of the time activity curves for putamen, thalamus and cerebellum are not affected by hypercapnia (timing of challenge indicated by shaded areas). (b) Measured flow changes with pcASL show large increases in flow during hypercapnia. (c) End-tidal CO2 values for the duration of the experiment.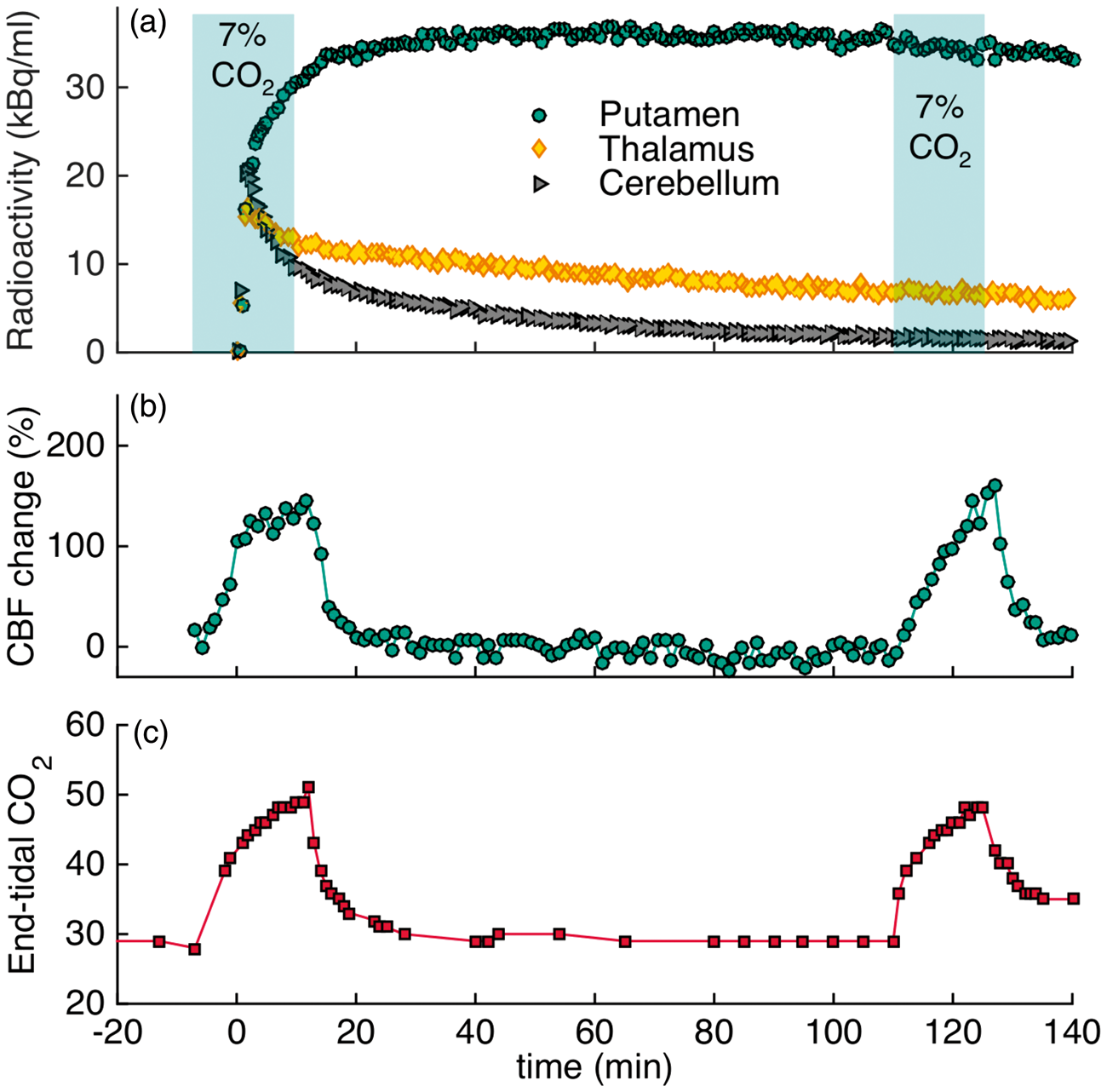
The kinetic modeling results for [18F]fallypride with the SRTM model did not show measurable changes in
The use of a dynamic binding term (time-dependent
Figure S1 in Supplementary Materials shows a plot of the TACs from the 5×CO2 and 2×CO2 hypercapnia conditions compared to the baseline [18F]fallypride curve for the putamen and cerebellum. The curves are normalized to their peak values. Compared to the baseline curve, there are only minor differences in uptake and washout. CBF was increased in the 2×CO2 condition at the beginning during the uptake phase but the TAC does not exhibit a different pattern compared to the other two TACs, in which CBF was at baseline during the first 30 min.
Simulation results
We simulated the effect of flow changes on radiotracer kinetics using a full reference tissue compartmental forward model (FRTM) 30 for the radiotracers [11C]raclopride and [18F]fallypride. Simulations that mimicked the ON-OFF design from the experiments were carried out, and analyzed with SRTM. A larger parameter space with different kinetic rates, extraction fractions and CBF changes was also investigated in order to understand the effect of CBF changes on a wider range of radiotracers with varying properties. The full set of assumptions and parameters used for the simulations are described in the Methods section.
Figure 4(a) shows simulated TACs for the radiotracer [11C]raclopride in a specific binding region (e.g. putamen) and for a reference region (e.g. cerebellum). For comparison, a baseline TAC without flow changes is plotted. Changes in the rate constants Simulations of a reference tissue model with kinetic parameters for [11C]raclopride. (a) Time activity curves for the PET signal from a specific binding region show very small deviations compared to a baseline PET curve. (b) Timecourse of simulated rate constants 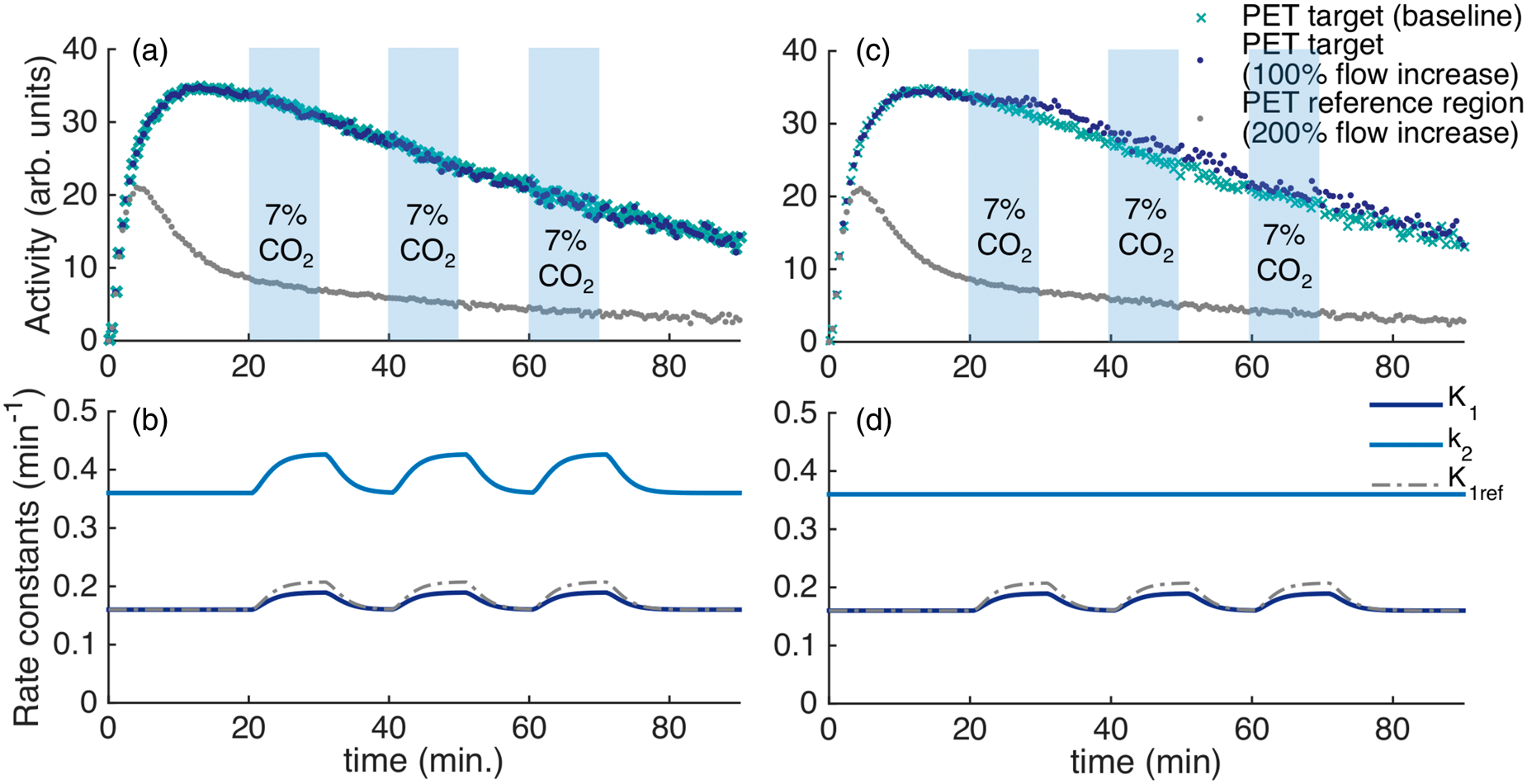
Figure S2 shows simulated TACs for the radiotracer [18F]fallypride in a high specific binding region (e.g. putamen), a low specific binding region (e.g. thalamus) and for a reference region (e.g. cerebellum). The TACs are simulated with an increase in flow with the timing of the (A–B) 5×CO2 and (C–D) 2×CO2 design. Timecourses of the simulated rate constants
To investigate the sensitivity of other radiotracers with varying kinetic properties to changes in blood flow, we simulated the effect of doubling blood flow (100% increase) for a combination of relevant radiotracer parameters. The results are shown in Figure 5(a) and (b), which show the decrease in (a, b) Change in 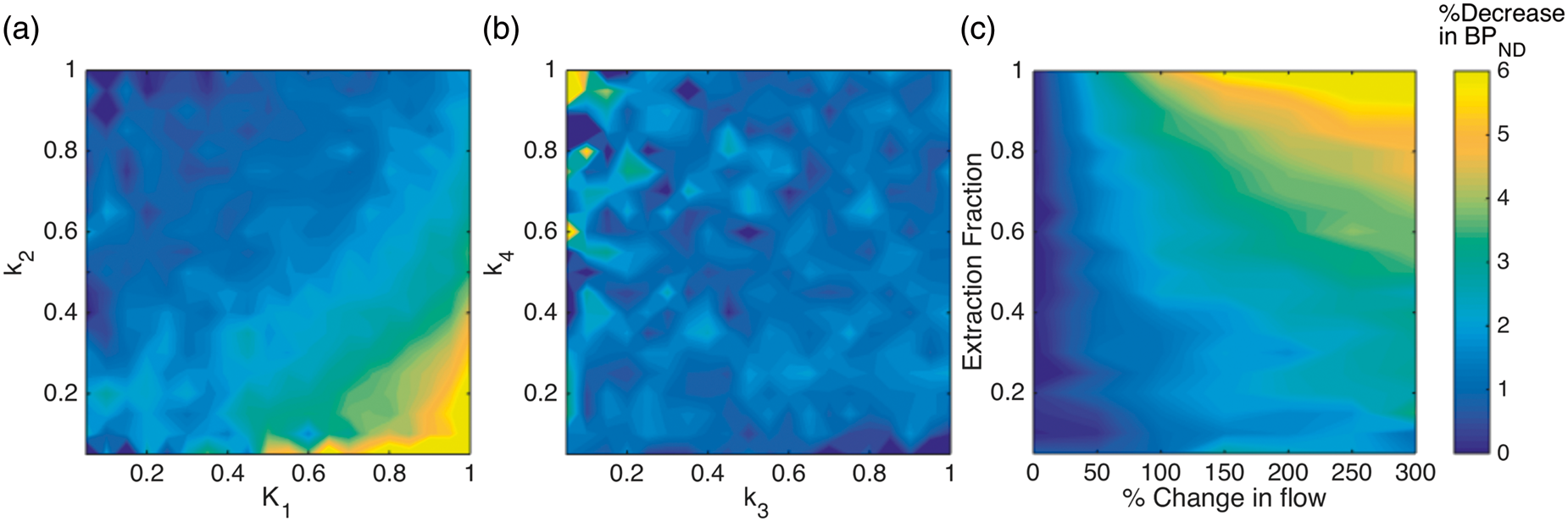
Figure 5(c) shows a plot of the percent decrease in
We investigated several assumptions that determine how flow changes affect
Discussion
In this study, we investigated the effect of increasing blood flow on radiotracer kinetics. By applying hypercapnia challenges in non-human primates, CBF increased up to 2.7-fold in whole-brain. Simultaneous acquisition of dynamic PET data and pcASL with MRI enabled the measurement of radiotracer kinetics together with induced flow changes. For the investigated radiotracers [11C]raclopride and [18F]fallypride, flow changes did not change the shape of the time activity curves or the quantification of binding potentials, independent of the timing of the hypercapnia challenge. Overall, our results suggest that changes in blood flow do not significantly affect specific binding parameters or kinetics of these radiotracers.
[11C]raclopride and [18F]fallypride were chosen as radiotracers in this study due to their differences in extraction fractions, kinetics and affinity, while being specific to the same target (D2/D3 receptors). Raclopride has a fairly low extraction fraction (0.16), 33 whereas fallypride is considered to have a higher extraction fraction (0.4–0.5), 34 higher affinity and slower washout kinetics, which has led to suggestions that it should be more susceptible to blood flow changes. Our result that [11C]raclopride is not sensitive to CBF changes is in agreement with previous simulations1–3 and experimental results, 14 and provides experimental evidence that even extremely large increases in CBF do not affect quantification of binding parameters. This result may help interpret other study results, e.g. in light of our study, CBF is likely not the reason why differences in binding potentials are observed under different anesthesia regimens, 35 even if anesthesia methods can differentially affect CBF. Perhaps more surprisingly, [18F]fallypride did not show measurable sensitivity to CBF increases either, which supports previous theoretical investigations 16 but is in contrast to conclusions from experimental data by Cumming et al. 15 In the latter study, CBF changes were not measured directly, rather, changes in CBF were attributed to the area under the curve from a region of interest in the venous confluence. Our study did not find measurable changes in [18F]fallypride binding with experimentally controlled and determined CBF changes that are much larger than are expected among healthy subjects across a large age range. Hence, our findings suggest that the correlation between the flow metric and [18F]fallypride binding potential by Cumming et al. is not caused by CBF changes.
Hypercapnia challenges were employed because hypercapnia produces large CBF changes that exceed blood flow changes occurring in the brain under many conditions, while also minimally impacting neuronal metabolism
36
and presumably not changing neuroreceptor availability. Our CBF results agree with other studies that reported similar CBF increases using 7% CO2 using either PET or MR measurements, with cerebellum generally showing larger CBF increases compared to cerebral gray matter structures.37–40 PET studies with
Kinetic modeling of PET data was carried out using different models and datapoints in order to quantify potential effects of CBF increases on binding measures. In order to quantify both a baseline
In this study, we investigated two representative radiotracers but the question of flow sensitivity in other radiotracers remains to be experimentally tested. We thus attempted to generalize results using simulations. The majority of other radiotracers have extraction fractions that lie within the range of [11C]raclopride and [18F]fallypride, and we would expect that similar radiotracers would exhibit negligible sensitivity to CBF changes as well. Experimentally, we have previously shown that infusions of [18F]-FDG are not affected by changes in blood flow
47
– an example of a radiotracer that binds irreversibly to its target and is not blood flow dependent. Our simulations and analysis of flow sensitivity for wide ranges of kinetic parameters
The primary goal of this study was to investigate how CBF changes, which are known to occur during pharmacological or other functional challenges, influence inferences about specific binding. From fMRI studies 29 based upon CBF, BOLD signal or CBV, 12 we know that flow changes induced by injected drugs can rapidly increase (within minutes) and the timecourse may or may not be correlated with specific binding, depending upon the nature of the drug or stimulus. 46 In PET competition studies, injection of pharmacological challenges can be given before, during or after the radiotracer administration. Consideration of the timing of CBF changes is important because varying parts of the radiotracer curve may be affected differently with kinetic rate constants dominating during update and washout phase. In our experiments, we varied the timing of the challenge in order to address these potential differences. Yet, we did not detect any effects of flow in either scenario on TACs or on radiotracer quantification.
The conclusions from this study are expected to hold for other PET infusion paradigms and measurements. In a separate study, we demonstrated that continuous infusion of [18F]-FDG is not affected by changes in blood flow. 47 With continuous infusion-based methods, steady-state can be reached after an initial equilibration period. 48 In this scenario, flow changes should minimally affect tracer kinetics when blood and tissue concentration are at steady state. In this study, we used the bolus method to maximize flow-sensitivity as a worst-case scenario. Since we did not find that flow affected our measurements, these results should be applicable to continuous infusion, or mixed bolus plus infusion methods as well.
Our simulations assumed that flow alters both
Pharmacological challenges usually produce flow patterns that differ in regions of the brain. Especially when using reference region models 44 for a kinetic analysis in PET, differences in flow between the two regions can potentially affect the estimation of parameters. In the data presented here, we observed differences in flow between the striatal regions (specific binding region) and the cerebellum (reference region), which suggests that the CBF changes we induced are an appropriate model for challenges at neuroreceptors that might act locally. In addition, since we did not observe apparent alterations in the TACs during cycles of modulated flow, our results can be applied even for competition studies, in which only specific regions of the brain are targeted.
It is possible that anesthesia affects the absolute quantification of our study parameters. Yet, a strength of our experimental paradigm is that we specifically used a periodic modulation of hypercapnia and thus CBF. Any change in anesthetic levels that could affect the data would not be expected to be periodic, rather it would be slow-changing. We closely monitored the animals under anesthesia and hypercapnia, which allowed us to observe animal physiology continuously. Since we did not detect changes specifically due to blood flow changes, and the changes induced by hypercapnia were very large, this study conclusion is likely not affected by the anesthetic drugs.
Our study provides experimental and theoretical support for continuing efforts to understand nuances of receptor binding during behavioral 51 or pharmacological challenges17,52 and their relationships to changes in hemodynamic parameters29,53 and functional networks. 54 This study confirms hemodynamic parameters as independent measures from receptor binding in PET studies. Despite previous suggestions in the literature, 15 but in agreement with previous findings from several studies,1,2,16 flow effects on most radiotracers sensitive to neuroreceptors are likely to be a negligible confound for quantification of PET binding parameters.
Conclusions
In summary, we have experimentally investigated the effect of flow changes on neuroreceptor PET radiotracer kinetics using simultaneous measurements of PET and MRI. Even with very large flow changes induced with a hypercapnic challenge, [11C]raclopride and [18F]fallypride TACs did not show measurable changes, indicating that flow does not influence these radiotracers. The flow-insensitivity of the two radiotracers investigated in this study validates a number of previous literature findings. Our findings are important for kinetic modeling approaches and their assumptions on flow. Furthermore, our results show experimental validation of the effect of flow for within-scan challenges and provide a framework for testing the flow effects of other existing or novel radiotracers.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National Institute of Health grants P41EB015896, S10RR026666, S10RR022976, S10RR019933 and S10RR017208.
Acknowledgements
We thank Helen Deng, Judit Sore, Kari Phan, Garima Gantam, Shirley Hsu, Grae Arabasz and Regan Butterfield for their support on radiotracer synthesis and animal setup.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors' contributions
CYS: Designed study, planned and conducted experiments, analyzed data, performed simulations, interpreted results, wrote manuscript. JBM: Assisted with experiments, data analysis and interpretation, edited manuscript. HYW: Assisted with experiments and data analysis, edited manuscript. CC: Assisted with data reconstruction and analysis, edited manuscript. JMH: Provided radiotracers, assisted with interpretation of results, edited manuscript. BRR: Assisted with study design, interpretation of results, edited manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
