Abstract
Vascular changes during spontaneous headache attacks have been studied over the last 30 years. The interest in cerebral vessels in headache research was initially due to the hypothesis of cerebral vessels as the pain source. Here, we review the knowledge gained by measuring the cerebral vasculature during spontaneous primary headache attacks with the use of single photon emission tomography (SPECT), positron emission tomography (PET), magnetic resonance imaging (MRA) and transcranial Doppler (TCD). Furthermore, the use of near-infrared spectroscopy in headache research is reviewed. Existing TCD studies of migraine and other headache disorders do not provide solid evidence for cerebral blood flow velocity changes during spontaneous attacks of migraine headache. SPECT studies have clearly shown cortical vascular changes following migraine aura and the differences between migraine with aura compared to migraine without aura. PET studies have shown focal activation in brain structures related to headache, but whether the changes are specific to different primary headaches have yet to be demonstrated. MR angiography has shown precise changes in large cerebral vessels during spontaneous migraine without aura attacks. Future development in more precise imaging methods may further elucidate the pathophysiological mechanisms in primary headaches.
Introduction
Comparison of practical issues for TCD, SPECT, PET and MRI.
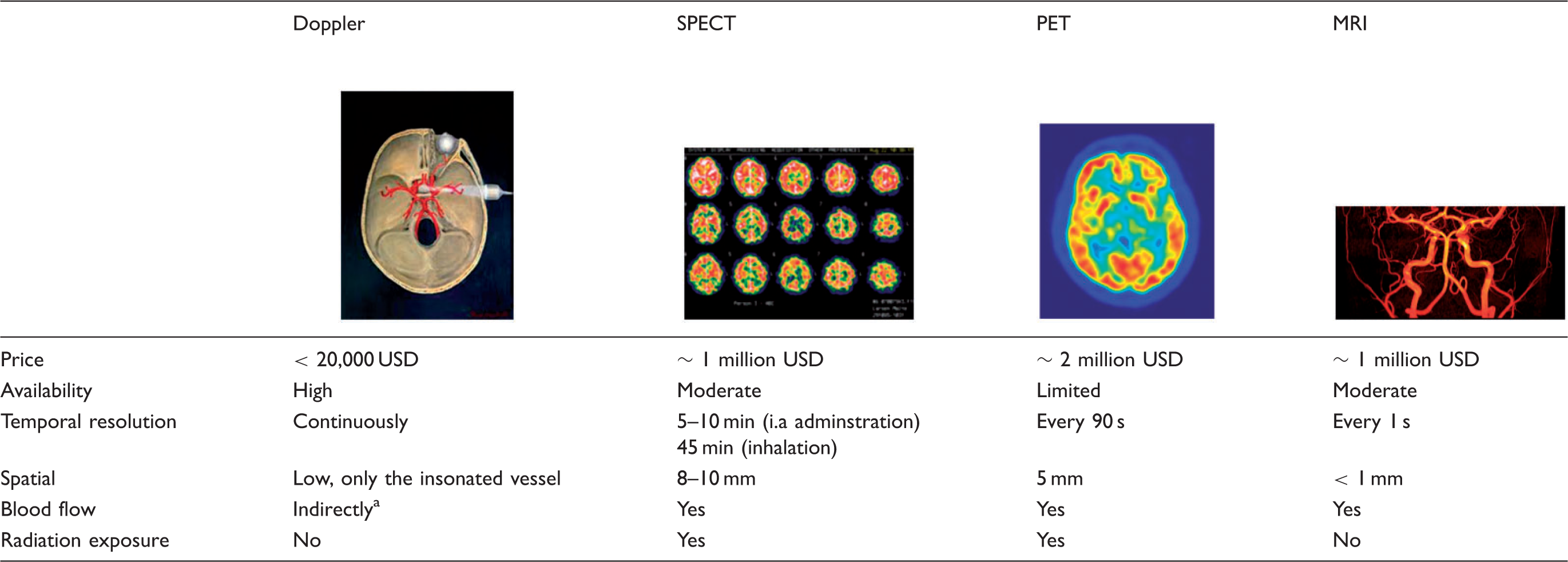
blood flow assessment assumes constant diameter in the insonated vessel.
i.a: intra-arterial; PET: positron emission tomography; SPECT: single photon emission computer tomography; TCD: transcranial Doppler; MRI: magnetic resonance imaging.
Methods
TCD
TCD was introduced in 1982 by Aaslid et al.
14
and has over the last 30 years been used to measure blood flow velocity (BFV) in the middle cerebral artery (MCA), the anterior cerebral artery (ACA) and the posterior cerebral artery (PCA) in headache and other neurological disorders such as stroke.
15
This was achieved by changing the Doppler frequency from the previous conventional 5–10 MHz to 1–2 MHz for which the attenuation in bone is much less. Furthermore, to achieve in-depth recordings, an acoustic lens with a focal length of 5 cm was applied.
14
It is necessary to assume that the angle between the ultrasound beam and the intracranial artery is sharp, between 0 and 30°, which results in a maximum error of 15%.
14
The blood velocity V can then be calculated by measuring the Doppler shift via the equation: V = 0.039 × f, where f is the Doppler frequency shift. When measuring changes in velocity and assuming that the cerebral blood flow is constant, it is also possible according to Dahl et al.
16
to retrieve the diameter change Δd of the MCA by the equation:

The Dahl et al. study proposed this equation following infusion of the vasoactive drug nitroglycerin, but assuming or controlling for no changes in CBF during a spontaneous migraine, this equation can also be applied for analysis of spontaneous attacks. One important issue is that changes in respiration frequency may often lead to either hypocapnia or hypercapnia and thereby to a compensational change in CBF, whereas the diameter of the large arteries such as the MCA remains constant, which results in a proportional change in MCA blood velocity. This effect can be corrected by measuring end-tidal carbon dioxide partial pressure (PCO2), and computing a corrected velocity normalized to a standard PCO2 value of 40 mmHg as detailed by Markwalder et al.
17
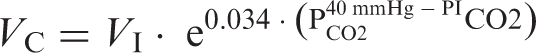
This correction factor may, however, underestimate the effect of CO2 on the velocity change in some conditions. In a human migraine model testing pituitary adenylate cyclase activating peptide 38 (PACAP38), which causes hyperventilation, 18 the MCA diameter dilation measured with TCD and calculated with the above equation was 9.5% at the end of a 20-min infusion of 10 ρ m/kg/min PACAP38. 18 However, the exact same dosage in a MR angiography study only induced an insignificant 0.8% change in MCA circumference at 20 min. 19 Thus, it is possible that under some circumstances, such as infusion of vasoactive substances, there is an underestimation of the CO2 effect on the velocity change in intracranial arteries as assessed by TCD.
The fact that some primary headaches, such as migraine and cluster headache (CH), are often or always unilateral in presentation makes TCD an ideal tool. Vessels from both hemispheres can be assessed simultaneously, with the MCA velocities being nearly equal on the two sides in the healthy adult, 14 and with day-to-day variations of only 16% being reported. 20 Today, the TCD technique can be used at the bedside with a swift application of the ultrasound probes, which makes the system ideal for experimental monitoring during headache attacks.
SPECT
SPECT is a nuclear imaging technique, which measures and quantifies cerebral blood flow. In SPECT, a gamma ray is generated by the decay of a radioisotope, and directly or indirectly recorded by a gamma camera rotating around the head, which can then, via reconstruction algorithms, create two- or three-dimensional images. SPECT has a lower reconstructed spatial resolution from 8 to 10 mm, 21 is less expensive, and is more widely available than PET (Table 1), and it utilizes radioisotopes with longer half-lives from hours to days, which makes it possible to have the radioisotopes delivered from an off-site supplier. Corrected CBF values, as with TCD, have to be corrected for significant changes in PetCO2 by 2% for each mmHg change in PetCO222 in order to measure changes related to changes occurring as a result of the headache attack and not due to a change in respiratory rate. Regional and global CBF measurements with 133Xe inhalation SPECT have been shown to be a reproducible method for assessing absolute changes 17 with a day-to-day coefficient of variation of 8%, 23 which makes it slightly better than MRI and approximately equal to PET. 24 SPECT ictal recordings have been performed in headache research since the late 1970s to measure CBF alterations during migraine aura and migraine without aura attacks. The first studies used up to 16 collimated sodium iodide (NaI) crystal scintillation detectors placed over both hemispheres and thereby having a very limited spatial resolution. 25 Later, a 254-channel instrument was developed leading to an improved spatial resolution. 7 The temporal resolution can be brought down to 5–10 min using intra-arterial administration, but this is a relatively invasive procedure and therefore the use of SPECT in spontaneous headache attacks in larger studies has ceased over the last two decades.
PET
PET can be used to study cerebral blood flow changes and spontaneous activation of brain areas. PET uses radiotracers that emit positrons when they decay. The emitted positron travels a few millimetres and then interacts with an electron, leading to annihilation of both particles, and thereby creation of two gamma rays going in opposite directions, which can then be detected simultaneously. Through this detection, a line of coincidence can be algorithmically constructed and the location of the original emitted proton can be topographically mapped from the detection of thousands of gamma ray pairs. The radiotracers used for PET can be physiologically relevant tracers such as [ 15 O]H2O for CBF measurements 26 or 2-[ 18 F]flouro-2-deoxy-D-Glucose (FDG) for metabolism measurements. 27 Specifically designed PET radiotracers can also be used to study pharmacological effects in the brain. As an example, Hostetler et al. 28 showed in vivo quantification of calcitonin gene-related peptide (CGRP) receptor occupancy by telcagepant in rhesus monkey and human brain using the PET radiotracer [11C]MK-4232, and showed a low receptor occupancy in the brain at an efficacious dose. Thus, it is unlikely that antagonism of central CGRP receptors are required for migraine efficacy, which point to extracerebral mechanisms being responsible for the generation of pain in migraine Similarly, Schankin et al. 29 recently demonstrated in a PET study with the radiotracer (11)C-dihydroergotamine (DHE), which is chemically identical to pharmacologically active DHE, that DHE does not cross the blood–brain barrier (BBB) during an induced migraine attack by a nitric oxide donor. Future studies may show the exact location of a CGRP or other signalling molecule antibodies used for migraine treatment.
Magnetic resonance angiography
Three-dimensional magnetic resonance angiography (MRA) using the time-of-flight (3D-TOF-MRA) method has been one of the most important non-invasive approaches for in vivo examination of the human cephalic and cerebral arteries in headache studies during the last ten years.19,30–34 The 3D-TOF-MRA method provides high resolution (< 1 mm voxel size) images of the arteries in and the scanning requires no contrast agent, 35 making it the method of choice when repeated measurements are required. The technique is based on flow-related enhancement of magnetically unsaturated blood entering stationary regions which have been previously magnetically saturated by multiple radio frequency (RF) pulses. Thus, the ‘fresh’ blood appears brighter compared to the background, enabling measurement of the luminal diameter and circumference of a given artery. This method was first introduced in the field of migraine research in 2008 34 and has since been used in several headache and migraine studies.11,19,32,33,36,37 The intra- and inter-observer variations using this method are less than 5%, while day-to-day and side-to-side variations in the luminal circumference of the MCA and the middle meningeal artery (MMA) are below 10%. 37 Apart from non-contrast and radiation-free scans allowing repeated recordings, the advantages of this method are that it can measure several arteries simultaneously. Disadvantages of this method include the long image acquisition time compared to conventional computerized axial tomography (CAT-scan) angiography (i.e. acquisition time < 1 min), increasing the risk of movement artefacts and the audible noise produced by the scanner machine, which can aggravate pain in patients suffering from phonophobia during the migraine attack.
NIRS
NIRS is a non-invasive imaging modality that uses near-infrared light to measure changes in cerebral oxy- (HbO) and deoxy-hemoglobin (HbR) concentrations. It has become an effective tool to study normal and pathological brain physiology.38,39 Following the first proof-of-principle experiment in a human subject in 1977, 40 the 1980s saw the development and clinical implementation of cerebral oximeters that use the NIRS technology to monitor trends in cerebral oxygenation, mainly for neuromonitoring during cardiovascular surgeries.41,42 In 1993, came the first demonstrations of the capability of NIRS to measure the functional hemodynamic response to cortical activation. 43–45
Functional NIRS (fNIRS) uses near-infrared light emitters – laser diodes or light-emitting diodes (LEDs) – at typically two wavelengths to illuminate the scalp. The light propagates diffusively through the scalp, skull and brain, and is partly absorbed by different chromophores, mainly the hemoglobin of the microvasculature blood. A photodetector – generally a photodiode or avalanche photodiode (APD) – is placed a few centimeters away from the source (3–5 cm) and detects part of the diffused light (Figure 1). As cerebral blood volume and oxygenation vary, the local absorption of light and therefore the detected light intensity change accordingly. Through models of light propagation in the head,46–48 and taking advantage of the differential absorption spectra of HbO and HbR, one can retrieve the concentration changes in these two hemoglobin species, and consequently total hemoglobin (Hbt). The normal hemodynamic response to the increased metabolic demand arising from neuronal activity is a focal increase in blood flow largely exceeding the increased oxygen consumption,
49
resulting in the typical increase in HbO and decrease in HbR measured by fNIRS (Figure 1).
(a) Array of NIRS sources and detectors (dots), and corresponding channels (lines). (b) Sensitivity profile of an fNIRS channel (source-detector pair) from Monte Carlo simulations. (c) Typical hemodynamic response to cortical activation measured by fNIRS.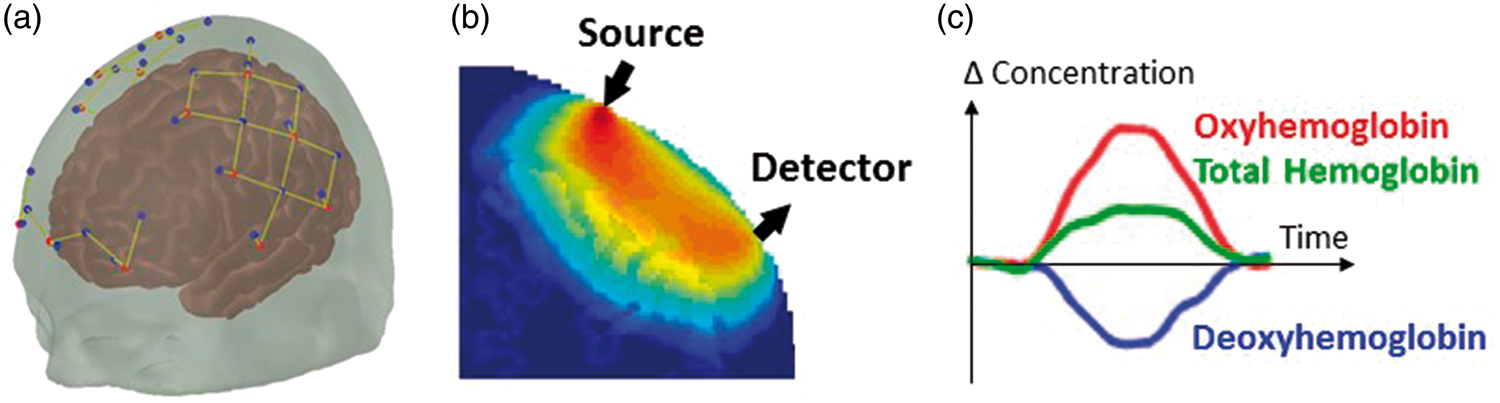
Generally, an array of light sources and detectors is placed on the head, with each source-detector pair creating a measurement channel, and the combination of all channels allowing the retrieval of a two-dimensional map of the hemodynamic response to brain activation. Most traditional fNIRS systems employ optical fibers to deliver light to and from the head, but recent wearable devices instead place sources and detectors directly on the scalp.50–54 A variety of fNIRS devices are available commercially and in research settings, 55 varying in flexibility and complexity, from simple forehead-only monitors to high-density whole-head systems. 56
fNIRS offers many attractive features for a wide range of functional studies: relatively low-cost, non-invasiveness, portability, tolerance to motion, high temporal resolution, and functional contrast. Despite these assets, the modality also has limitations that need to be considered. First, its spatial resolution is roughly a couple of centimeters (roughly the source detector separation), although advances in instrumentation (e.g. high-density arrays) and image reconstruction algorithms (e.g. anatomical guidance57–59) can improve resolution potentially by a factor of 3. 60 Second, as the light must travel back and forth through the scalp to reach the brain, measurements are only sensitive to the superficial cortex and are contaminated by the extracerebral vasculature that reflects systemic physiology. Multiple approaches have been proposed and are still being actively developed to minimize this contamination.61–63 Finally, because fNIRS is particularly suited to populations where subjects are moving or talking, it is prone to motion artifacts. Hardware and software methods can reduce this motion contamination,50,64 or partially correct for them during post-processing.65–67
The range of fNIRS applications and users has grown exponentially since the first demonstration of the technology,38,39 encompassing notably the fields of developmental neuroscience,68,69 psychiatric conditions, 70 and neurological disorders and injuries such as migraine, epilepsy, movement disorders and stroke. 71 It is particularly adapted to challenging populations that may not be amenable to other imaging modalities, including infants and children, neurocritical care patients, and freely-moving, behaving participants (testing of cognitive or executive functions, gait studies, speech studies). In addition to traditional evoked-response functional studies, some research has focused on the spontaneous oscillations in the fNIRS signals, driven either from neuronal or vascular fluctuations. 72 In this context, fNIRS has been applied to study resting-state functional connectivity,73–76 and cerebral autoregulation and vascular reactivity.15,66,77
Studies of spontaneous attacks in migraine
TCD in migraine
Studies investigating vascular changes during spontaneous migraine attacks using SPECT.
SPECT: single photon emission computer tomography; MA: migraine with aura; FHM: familial hemiplegic migraine; MO: migraine without aura; MA: migraine with aura; TTH: tension-type headache.
SPECT in migraine
In the late 1970s, SPECT studies began to emerge, which recorded regional CBF (rCBF) changes during spontaneous migraine attacks. Sakai and Meyer 25 investigated migraine and CH patients during attacks compared to healthy controls as well as the interictal headache using the Xenon inhalation method with 16 detectors placed over both hemispheres and thereby having a very limited spatial resolution. The study compared three migraine without aura (MO) and two migraine with aura (MA) patients with serial images during the headache phase and when headache free. 25 The authors found a mean increase of 31% in total CBF during attacks in these five patients with no apparent differences between migraine subtypes. Interestingly, one MA patient was described in detail, in which there was a local CBF reduction in the post-central gyrus corresponding to the contralateral loss of sensation. 25 Three years later, the groundbreaking study by Olesen et al. 7 was published, which used the intra-arterial Xenon 133 method and 254 channel equipment with repeated measurement by 5–10 min, which was able to detect that in migraine with aura, attacks were initiated by focal hyperemia and then an oligemia which gradually spread anteriorly in the course of 15 to 45 min. 7 Thus, the study was the first to clearly show what may be the vascular footprint of cortical spreading depression in migraine aura. Then followed a series of studies, which replicated the findings.80–83 This was achieved using Xenon intraarterial injection and Xenon inhalation (Table 2). The studies from the Copenhagen group also showed that during familial hemiplegic migraine (FHM) attacks, a pattern similar to that of MA is observed. 83 Furthermore, they observed no increase in CBF during MO attacks.82,84 This was also confirmed using the technetium Tc99m hexemethyl-propyleneamineoxime method. 85 In contrast, Kobari et al. 86 investigated MO and MA patients during attacks and observed that CBF increased by 20–40% in the fronto-temporal cortex, thalamus and basal ganglia, which may be due to cortical activation as a result of pain. 87 In addition, they observed no differences between MO and MA patients, which is in contrast to the majority of studies, but similar to the Sakai study. 25 The SPECT studies in migraine laid the foundation to describing the pathophysiological background behind migraine with aura and showed, in most cases, a clear difference in vascular alterations between spontaneous MO and MA attacks.
PET in migraine
Studies investigating vascular changes during spontaneous migraine attacks using PET.
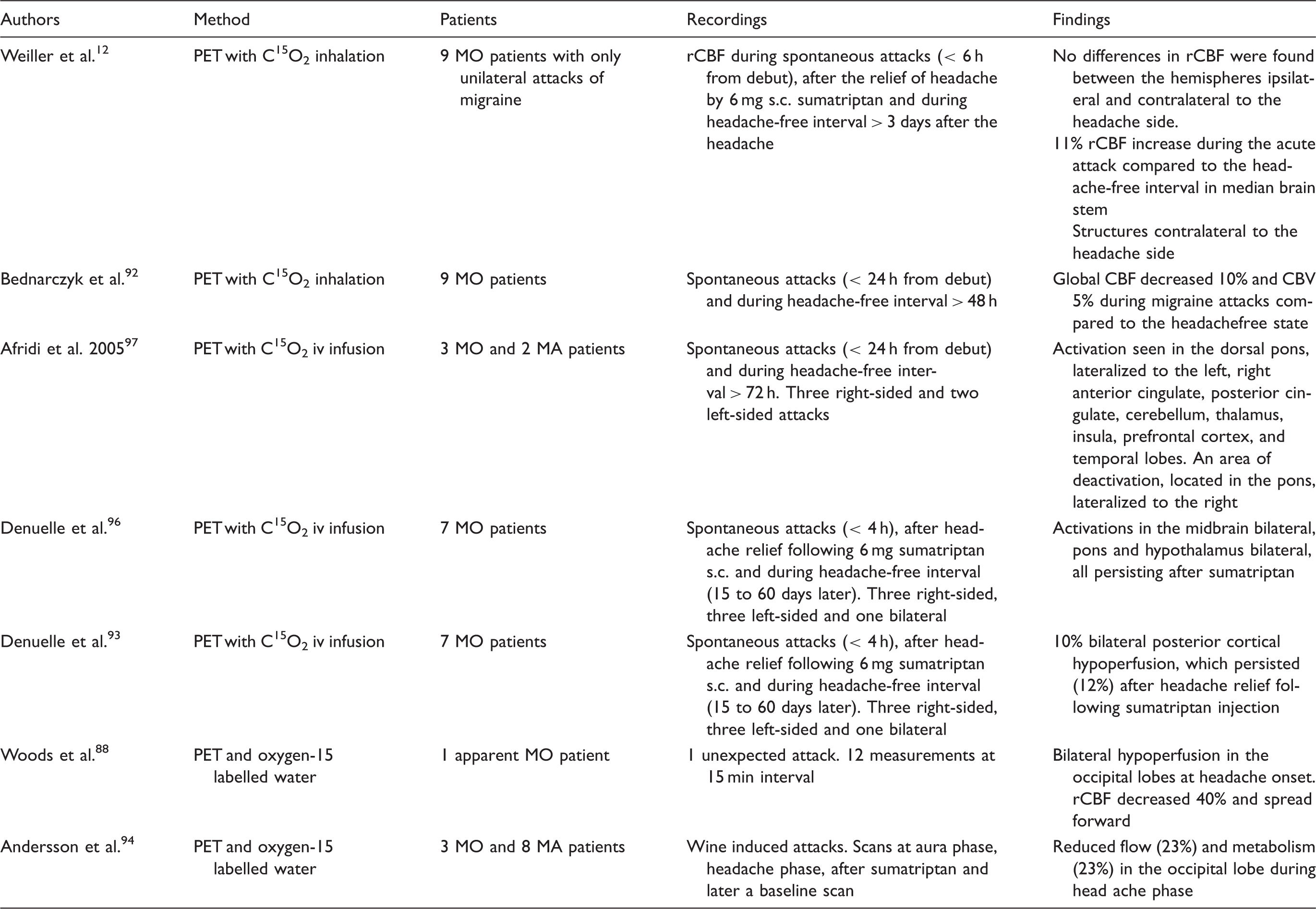
PET: positron emission tomography; MA: migraine with aura; MO: migraine without aura; MA: migraine with aura; rCBF: regional cerebral blood flow.
MRA in migraine
Although several headache and migraine studies have been conducted using this method, there is only a single study reporting data on spontaneous migraine attacks. 11 In this study, the long-standing theory of extracranial vasodilation as the cause of the pulsating migraine pain was tested using a 3D-TOF-MRA scan during spontaneous attacks of unilateral migraineous pain in 19 patients suffering from migraine without aura. 11 These patients were subsequently scanned 30 min after subcutaneous injection of 6 mg sumatriptan on the migraine attack day, and again on another migraine- and headache-free day. The circumference of several intra- and extracranial arterial segments was measured and compared between the three conditions. 11 Data revealed a 13.0% dilation of the MCA, 11.5% dilation of the cerebral ICA and 11.4% dilation of the cavernous ICA on the pain-side during attack compared to outside of attack. A modest dilation was also reported on the non-pain side MCA (6.1%) and cavernous ICA (4.9%). 11 However, the side-to-side comparison of these arteries showed 9.1–14.4% more dilation on the pain-side versus non-pain side during the attack day. No day-to-day or side-to-side differences of the extracranial arteries were demonstrated in the study. 11 After effective treatment with sumatriptan, all extracranial arteries and the cavernous ICA were constricted by 10–20%, whereas the MCA and cerebral ICA remained dilated. 11 These segments are thought to be protected by the blood–brain barrier, which may explain the lack of constrictor effect of sumatriptan. The cavernous ICA was the only arterial segment that was dilated during attack and constricted after sumatriptan. This part of ICA is in the bone and is covered by nerve plexus, which may be compressed by the surrounding bone when the artery dilates. May et al. 98 reported dilation of the cavernous ICA after activation of the trigeminovascular system by ipsilateral injection of capsaicin in the forehead. Thus, the conclusion of this study was that migraine pain may be associated with intra- but not extracranial vasodilation. 98 However, dilation of the MCA and ICA was not regarded as the cause of the pain, as effective treatment with sumatriptan did not reverse the dilation of these arteries. The strengths of this study are the investigation of spontaneous migraine attacks and the fact that the attacks were unilateral. Thus, the non-pain side can be used as control for the pain side. The limitation of the study was that only 19 patients were included, so that subgroup analyses were not possible. Examination of untreated spontaneous migraine attacks in a MRI scanner is a difficult task that requires an effective and demanding collaboration between many different departments. Although, no statistical significant extracranial dilation was found, data revealed that circumferences of all extracranial arteries were numerically greater on the pain side versus non-pain side. Thus, subtle circumference changes (less than 10%) cannot be rejected based on these findings. A small (5%) dilation of MMA on the pain side has previously been reported in a case-report of one patient. 99 Subtle dilation of the MMA per se may not be painful as drug-induced 30% dilation of this artery has been reported in migraine without aura patients who had no pain. 36 The ipsilateral lateralisation of small circumference changes could indicate perivascular inflammation causing slight relaxation of the vessel walls.
NIRS in migraine
NIRS has been used in migraine research to investigate cortical vasomotor reactivity during metabolic,100–102 physiological,103,104 pharmacological105,106 and functional tests.107,108 NIRS has also been used in one study investigating neurostimulation in CH. 109
Using a breath hold (BH) test to investigate vasoreactivity as a result of changes in arterial CO2 concentration, 110 Akin et al. 100 showed that interictal HbR increase following BH was smaller in MO than in healthy controls, while HbO increase was similar in the two groups. In a different study, Liboni et al. 101 showed smaller HbO increases and lower HbR decreases in 30 MO patients than in healthy controls during BH, which may have been biased by a shorter breath holding duration in MO patients than in healthy subjects. Another method of assessing vasomotor reactivity is via head-down maneuver inducing an increase in frontal lobe pressure, which usually leads to elevated CBF. This maneuver produced a significantly smaller increase in right-sided HbT in migraineurs when compared to volunteers. 103 Migraine patients showed a smaller increase in right-sided HbT than in left-sided HbT following the head down maneuver. In addition, migraine patients had a greater increase in right-sided HbT than healthy volunteers in left-sided HbT. 103 It is difficult to draw clear conclusions from this study as the patients were not coded as having MO or MA. The lateralized vasoreactivity is difficult to understand, but has been attributed to a sympathetic lateralization in the right hemisphere,111,112 even though it is not known if this may lead to asymmetric vasomotor responses. Vernieri et al. 102 investigated side differences in MA patients in the interictal period, showing an increased cerebral vasoreactivity to CO2 inhalation measured by NIRS on the side of most frequent attacks. A recent study investigated 10 MA and 10 MO patients during the interictal period of migraine in comparison to healthy controls. 113 The time-delay between the R-wave of an electrocardiogram and the arterial pulse wave of cerebral microcirculation, detected by NIRS on the frontal cortex of both sides, was used to evaluate the presence of cerebral arteriolar vasoconstriction. Both migraine groups had a significantly longer time delay than the control subjects, but there were no differences between sides. The same group measured the amplitude of the arterial pulse wave in patients with prolonged aura (1 h to 7 days) and found during aura a significant decrease of the arterial pulse wave and an increase of cerebral tissue oxygen saturation ipsilateral to the pain side and contralateral to aura symptoms compared with the headache-free periods. 104 Overall, the studies on vasomotor reactivity are few and do not translate into one concept of altered vasomotor reactivity following metabolic or physiological changes in migraine patients.
NIRS has been used to investigate sumatriptan treatment investigated MO patients during attacks followed by subcutaneous sumatriptan or sham injections. 105 After sumatriptan injection HbO decreased, 105 which was correlated to changes in skin blood flow measured by laser Doppler flowmetry. Human studies have not shown that sumatriptan constricts extracerebral arteries more than cerebral arteries,11,30,114 and the decrease found by may therefore very likely be a systemic extracerebral effect. Thus, NIRS monitoring using scalp probes are probably not entirely brain specific, and the clinical value of this type of investigation, when systemic changes are expected, is questionable. Studies attempting to separate superficial and cerebral signals would be useful to further explore these types of pharmacological effects. In patients with familial hemiplegic migraine (FHM), Schytz et al. 106 investigated the changes in low-frequency oscillations following glyceryl trinitrate (GTN), a nitric oxide donor. Interestingly, only FHM patients with common migraine subtypes had a lower increase in LFO amplitude compared to healthy controls and pure FHM patients, which suggests that the vascular differences between FHM patients and healthy controls were due to the common migraine subtypes. 106
fNIRS studies have been performed in a few studies. Schytz et al. 107 investigated frontal activation using a Stroop test interictally in 12 MO patients compared to 12 healthy controls and observed a clear vascular response in both groups, but no differences in amplitude or time response between groups, pointing to a normal neurovascular coupling in the frontal cortex. In contrast, Coutts et al. 108 investigated hemodynamic response to a range of visual stimuli in 15 MA and 5 MO patients. There were no differences in the amplitude of the response between migraine and control groups, but migraine patients had a faster hemodynamic response to visual stimuli, which, however, normalized after applying active lenses that reduced distortions of text and improved comfort. The underlying mechanism for the time differences in hemodynamic response and their normalization is unknown and needs further investigation. Thus, there may be differences in neuronal, vascular or neurovascular coupling in migraine patients following stimuli, but so far this has only been shown in the occipital cortical vasculature interictally. NIRS may also be used to investigate responses to neurostimulation, which was shown in a small study with five CH patients with sphenopalatine ganglion (SPG) stimulators, in which high frequency SPG stimulation induced a relative increase in HbO in the frontal region primarily on the stimulated ipsilateral side. 109 In conclusion, NIRS has been used as a non-invasive tool to measure cortical changes following different types of experimental stimulation paradigms. So far, there are no studies investigating vascular changes during spontaneous attacks. The development of new portable NIRS devices, 115 which can even be controlled by smartphones 116 may result in the opportunity to investigate cortical changes during spontaneous attacks. It is possible that patients could apply the equipment during spontaneous attacks at home and measure continuously for hours. The challenges of NIRS recording during spontaneous attacks would be the lower spatial resolution and motion artifacts.
Studies of spontaneous attacks in trigeminal automatic cephalalgias
TCD in TACs
TCD studies in trigeminal autonomic cephalalgias (TACs) are very rare, most likely due to the nature of the disorders. Most patients are agitated during the attacks making it difficult to lie still for TCD measurements. Afra et al. 117 reported decreased BFV in the MCA on the pain side during CH compared to an attack-free period. In an earlier study, 118 bilateral decreased BFV in the MCA was found during CH attacks in patients. However, the attacks were induced by nitroglycerin and the decrease was most probably an effect of the drug 119 rather than attack-related changes.
SPECT in TACS
Studies investigating vascular changes during spontaneous TAC attacks using SPECT and PET.
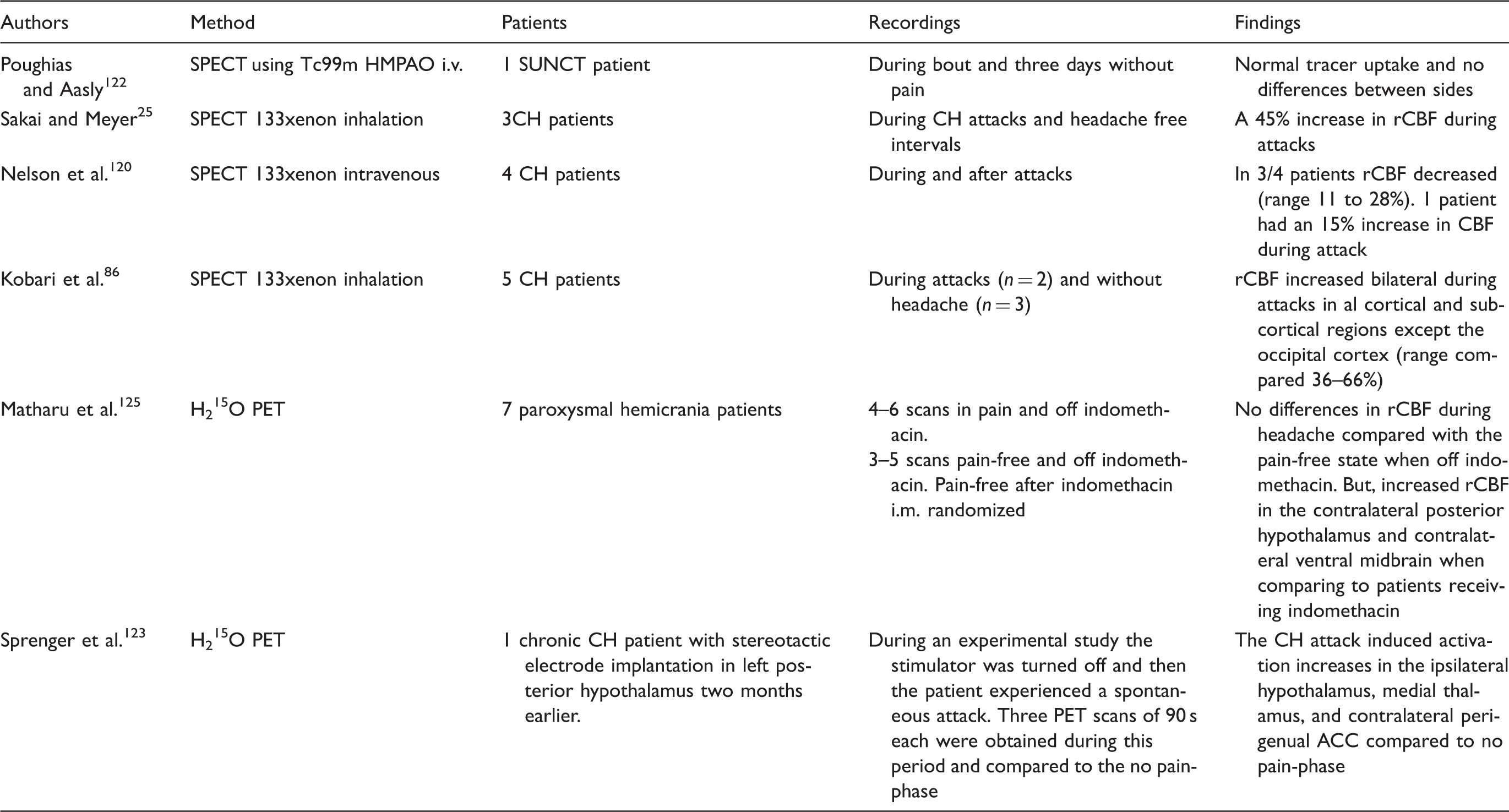
SPECT: single photon emission computer tomography; PET: positron emission tomography; SUNCT: short-lasting unilateral neuralgiform headache attacks; CH: cluster headache; rCBF: regional cerebral blood flow. ACC: anterior cingulate cortex; TAC: trigeminal automatic cephalalgia.
PET in TACs
Two PET studies exist in TACs patients during spontaneous attacks (Table 4). Sprenger et al. 123 published a case report of a spontaneous CH attack in a CH patient who two months earlier had underwent stereotactic electrode implantation for deep brain stimulation of the left posterior hypothalamus. 123 During a planned experimental study of brain activation, the stimulator was turned off, and the patient had a spontaneous attack in which three PET scans of 90 s each were obtained over 30 min and compared to the no-pain phase. The study found activation increases in the ipsilateral hypothalamus, medial thalamus and contralateral perigenual ACC, which are similar to activation found during nitroglycerin-induced CH attacks. 124 In paroxysmal hemicranias, Matharu et al. 125 showed no differences in rCBF during headache compared with the pain-free state when patients were off indomethacin, whereas an increased rCBF in the contralateral posterior hypothalamus and contralateral ventral midbrain was demonstrated when comparing to patients receiving indomethacin treatment. Thus, no pronounced cortical alterations are found in TAC patients interictally using PET.
Studies of spontaneous attacks in tension-type headache
Andersen et al. 126 investigated rCBF in 41 chronic tension-type headache (TTH) patients during habitual headache using Xenon inhalation or Tc99m HMPAO i.v. SPECT and found normal regional and global CBF when comparing to 33 healthy controls. 126 In 21 headache-free episodic TTH patients, increased blood flow velocities were measured in the anterior, middle and posterior cerebral, with no significant asymmetries of blood flow velocities in corresponding arteries. 127 The same group did not observe increased BFV in 20 chronic TTH patients. 128 Thus, there is no clear evidence of hemodynamic changes in TTH patients.
Conclusion and future directions
This review has gathered the available studies of vascular changes during spontaneous attacks in primary headaches. In migraine, aura symptoms clearly have a pattern of gradually spreading cortical hyperperfusion followed by gradual spreading hypoperfusion as shown by SPECT studies. There is not a pattern in migraine without aura of uniform vascular changes during attacks, but local changes may occur due to neuronal activation. MRA and TCD studies point to a small increase in cerebral vessel diameter during the headache phase, which may be the result of vasoactive peptides being released from perivascular nociceptors or parasympathetic nerve fibers. There is no proof that the vasodilation per se leads to pain. In TACs, there is no evidence of a uniform vascular change during attacks, but focal changes likely reflect neuronal activity in pain pathways. NIRS may be a new tool to further investigate cortical changes, especially during migraine with aura, which may elucidate underlying pathophysiological mechanisms. Moreover, the MR-based arterial spin labeling (ASL) sequence may also provide important information of the cortical blood flow changes in future studies. MRI images may be improved and patients may be scanned during attacks with motion correction. 129 The MMA has been suspected as the source of pain in migraine using primarily animal models and from surgical experience. To date, no studies have investigated the intracranial segment of the MMA during spontaneous attacks, which would be extremely valuable in future studies.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.