Abstract
In acute brain disorders, elimination of the excitatory output from an injured brain region reduces activity in connecting brain regions remote from the lesion site (i.e., diaschisis). The authors examined the effect of functional ablation of the left cerebral cortex by cortical spreading depression (CSD) or topical application of tetrodotoxin on single cell spiking activity, baseline CBF, and neurovascular coupling in the right rat sensory cortex. CSD or tetrodotoxin in left cortex reduced the right cortical spontaneous spike rate by 36% and 45%, respectively. Baseline CBF in the right cortex was unaffected by a left-sided CSD, but decreased by 12% for left cortical application of tetrodotoxin. This suggested dissociation between spontaneous spiking activity and basal CBF. Left infraorbital nerve stimulation evoked local field potentials in right cerebral cortex that were reduced in amplitude by 19% for left CSD and by 23% for left tetrodotoxin application. The corresponding declines in the evoked CBF responses were 42% for CSD and 23% for tetrodotoxin. Vascular reactivity to adenosine remained unchanged in right cortex. Thus, transhemispheric diaschisis produced a pronounced decrease in the spontaneous spike rate accompanied by no reduction or a small reduction in basal CBF, and an attenuation in amplitudes of evoked synaptic responses and corresponding rises in CBF. The findings suggest that disturbed neurovascular coupling may contribute to the disturbance in brain function in acute transhemispheric diaschisis.
Keywords
Acute brain disorders cause interruption of the excitatory projections from the lesioned brain area to anatomically intact brain regions (Kempinsky, 1958). This phenomenon, known as diaschisis, causes a transient decrease in spontaneous activity in the intact regions due to attenuated function in the projecting region. This may present itself in neuroimaging studies as decreases in glucose and oxygen metabolism, and CBF in connecting brain regions (Andrews 1991; Feeney and Baron, 1986; Ginsberg et al., 1977; Ito et al., 2002; Lenzi et al., 1982; Martin and Raichle, 1983; Reivich et al., 1978). We recently showed that focal cerebral ischemia in rats caused diaschisis in the contralateral cerebellar cortex that was characterized by pronounced decreases in Purkinje-cell spiking activity and small decreases in baseline cerebellar blood flow. The findings were explained by a decrease in the excitatory input to the cerebellar cortex (i.e., deactivation) because cerebellar neuronal excitability and vascular reactivity were preserved. Functional ablation of the cerebral cortex by either spreading depression or tetrodotoxin reproduced the changes in cerebellar function with complete recovery of Purkinje-cell activity and cerebellar blood flow concomitant with recovery of cortical function (Gold and Lauritzen, 2002). The fact that the decrease in spiking activity was three to seven times larger than the decrease in basal flow suggested that assessment of cerebellar function based on baseline blood flow represented an underestimation (Gold and Lauritzen, 2002).
The express purpose of the present study was to examine whether our findings for crossed cerebellar diaschisis could be generalized to other neuronal circuits. Our first hypothesis was that functional ablation of the cerebral cortex on one side would lead to a reduction in the spiking activity in the contralateral cortex accompanied by disproportionately small reductions in baseline CBF. If confirmed this would support the hypothesis that important changes in neuronal activity may prevail even though a constant baseline level of CBF is observed, and that single cell spike activity has only little influence on baseline CBF. Our second hypothesis was that the coupling relationship between synaptic activity and CBF was preserved in cortical regions affected by acute diaschisis. If confirmed this would imply that activity in projecting pathways do not contribute to neurovascular coupling. Although the first hypothesis was confirmed the second was disproved, suggesting that the consequences of acute diaschisis may vary among brain regions.
MATERIALS AND METHODS
Animal anesthesia and preparation
Experiments were performed in 24 male Wistar rats (356 ± 39 g) bred at the Panum Institute. All experiments were in compliance with the guidelines of the European Community of the Care and Use of Laboratory Animals. The rats were anesthetized with isoflurane (Vapor, Dräger, Germany; 5% at induction and 2.5% during surgery) in O2 30% and N2O 70%. Lidocaine (5 mg/mL) was used at the operation sites and at the contact spots for ear pins. The trachea was cannulated for mechanical ventilation with a respirator to maintain arterial pH at 7.41 ± 0.05, Pa
After surgery the anesthesia was changed to α-chloralose (1,2–0-[2,2,2-trichloro-ethylidene]-α-d-gluco-furanose), which was dissolved in saline (10 mg/mL) and heated to 69°C. The filtered α-chloralose was introduced as an intravenous bolus of 45 mg/kg. Isoflurane and N2O were discontinued and another 15-mg/kg bolus was given after 3 to 5 minutes. During the rest of the experiment, 15 to 20 mg/kg was given intravenously every 20 minutes. This anesthetic regime has been shown to be ideal for studies of cerebrovascular physiology (Bonvento et al., 1994). The level of anesthesia was checked by observing arterial blood pressure during stimulation and by tail pinch. The experiments were performed after a postoperation recovery period of at least half an hour to obtain a stable level of anesthesia, a stable laser-Doppler baseline, and an arterial blood pressure of between 90 to 120 mm Hg, not varying more than 10% in the same animal. Arterial blood pressure remained constant during electrical stimulation. Thus, the CBF increases noted were not due to fluctuations in blood pressure. The signals were A/D converted, amplified, and sampled via the 1401 plus interface (Cambridge Electronic Design [CED], Cambridge, U.K.) connected to a PC running the Spike2 software (CED). At the end of the experiment, the heart was stopped by an intravenous bolus injection of air and the zero level of the recording parameters was recorded.
Infraorbital nerve stimulation
In all experiments, the left ramus infraorbitalis of the trigeminal nerve was stimulated by a set of custom-made bipolar electrodes inserted percutaneously (Nielsen and Lauritzen, 2001). The cathode was positioned at the hiatus infraorbitalis and the anode was inserted in the masticatory muscles. The infraorbital nerve was stimulated using constant current (ISO-flex, A.M.P.I., Jerusalem, Israel) with 1-millisecond-long pulses with an intensity of 1.5 mA, 1 Hz in trains lasting for 10 seconds using 20-second interstimulus intervals. Synaptic and vascular responses were stable for hours using these stimulus variables. We used recordings from three stimulus trains immediately before cortical spreading depression (CSD) elicitation or tetrodotoxin application as controls.
Diaschisis
Diaschisis was produced by functional ablation of the left cerebral cortex by CSD (n = 6 rats) elicited with a fine-needle stab in the frontal cortex, avoiding induction of cortical bleeding. CSD induces a spontaneously reversible suppression of cortical function as outlined in details elsewhere (Bures et al., 1984). In alternating experiments, we used topical application of the sodium channel blocker tetrodotoxin dissolved in artificial CSF at 20 μmol/L (n = 6 rats) (Gold and Lauritzen, 2002). Suppression of spontaneous activity by either CSD or tetrodotoxin was monitored by the electrocorticogram (ECoG) in the left cerebral cortex.
For comparison, tetrodotoxin was applied to the contralateral cerebellar cortex in another six rats. Because of the weak nature of the cerebellar connections to the sensory cortex, this was expected to give rise to no change or only very small changes in cortical function.
Laser-Doppler flowmetry
Optic probes (PF 410; Perimed, Stockholm, Sweden; 780-nm wavelength, 250-μm fiber separation) were used for laser-Doppler measurements of CBF (Periflux 4001 Master, Perimed). The probes were placed on the cortical surface of a region devoid of large vessels (>100 μm) as close as possible to the microelectrode at coordinates -3 mm posterior to bregma and -6 mm lateral to the midline corresponding to the barrel cortex (Chapin and Lin, 1984). After stable baseline recordings had been obtained, the probe was left for the duration of the experiment. The signal was A/D converted and recorded using the CED 1401 plus interface and the CED Spike 2 software (200-Hz digital sampling rate). All changes of CBF were calculated as percent of the baseline value immediately preceding the test as described previously (Fabricius and Lauritzen, 1994). The laser-Doppler flowmetry monitor displays blood flow readings in arbitrary units that do not allow for measurement of CBF in terms of absolute values, but the method is valid in determining relative changes of CBF during moderate flow increases (Fabricius and Lauritzen, 1996).
Adenosine protocol
The vascular reactivity in the region affected by diaschisis was examined by topical application of 1-mmol/L adenosine dissolved in artificial CSF using a micropipette. Adenosine was left on the brain surface for 1 minute. The test was performed two times before and two times 45 minutes after tetrodotoxin was applied and diaschisis, as evidenced by a large reduction in the spontaneous spike rate of the right sensory cortex, had been produced (n = 6 rats).
Electrophysiology
We used single-barreled glass microelectrodes, filled with 2-mol/L saline (impedance, 2 to 3 MΩ; tip, 2 μm). Single unit activity (spike rate) and extracellular local field potentials (LFPs) were recorded with a single electrode at a depth of 300 to 600 μm in the right sensory cortex. Neuronal signals were continuously displayed on a digital storage oscilloscope (DS-203; Pintek, Cambridge, U.K.). The ECoG was recorded at a depth of 500 μm in the left cerebral cortex with a bipolar montage using the same type of microelectrodes (2-mm tip distance). An Ag/AgCl ground electrode was placed in the neck muscle. The preamplified (x10) signal was A/D-converted, amplified, and filtered (spikes: x5,000/300- to 2,400-Hz bandwidth; LFP and ECoG: × 1,000/1- to 1,000-Hz bandwidth), and digitally sampled using the 1401 plus hardware (CED) connected to a PC running the CED Spike 2.3 software. Digital sampling rates were at 20 kHz for spikes, and 5 kHz for field potentials and ECoG.
Data analysis and statistics
Values are expressed as mean ± SD. Spike 2.3 software with a 1401 plus interface (CED) was used for on- and off-line analysis of CBF, spikes, LFPs, and the ECoG. Spikes were identified by shape and amplitude before data analysis. Automatic spike sorting was used to remove noise contributions to the calculated event rate. The spontaneous neuronal activity is expressed as the spike rate (Hz). The LFPs were averaged and amplitudes were calculated as the difference between the baseline and the first negative peak for each stimulation period (mV). Evoked CBF responses were expressed as percent increase of baseline. Student's t-test or Wilcoxon test was used for statistical analysis. Values were considered statistically significant at P < 0.05.
RESULTS
The effect of CSD or tetrodotoxin in the left cortex on electrical activity and CBF in the contralateral right cerebral cortex was detected by extracellular recordings from either individual cortical cells (spikes) or groups of neurons (LFP), combined with laser-Doppler flowmetry on a real-time basis. The elicitation of CSD induced a dramatic decrease in the ECoG amplitude in the left cortex. Tetrodotoxin usually took 20 to 30 minutes to decrease the ECoG amplitude, which presumably reflects the time it takes for tetrodotoxin to diffuse to deeper cortical layers. The effect of tetrodotoxin was more variable than for CSD, which immediately caused total abolition of the ECoG due to the well-known large-scale depolarization of cortical neurons.
In the right cortex the simple spikes in layers III through IV occurred randomly with a firing rate of about 2 to 5 Hz. Functional ablation of the left cerebral cortex by CSD or tetrodotoxin decreased the spontaneous spike rate in the right sensory cortex. Figure 1A shows original spike measurements from one animal exposed to tetrodotoxin. The spike rate was approximately 6 Hz under control conditions, and declined as a function of time after tetrodotoxin application. Figure 1B shows the normalized data for all animals (n = 6). The spike rate had decreased by 45% ± 22% at 15 to 30 minutes after tetrodotoxin application. CSD induced a spontaneously reversible functional decortication and a mean decrease in the spontaneous spike rate of 36% ± 19%. The spike rate in the right sensory cortex returned to normal as the left ECoG returned to normal (Fig. 1C and 1D, n = 6). For technical reasons we are unable to report the effect of tetrodotoxin application on the left cerebellar cortex on the spike rate in the right sensory cortex. Baseline CBF decreased by 12.4% ± 7.8% when tetrodotoxin was applied to the left sensory cortex, but remained constant in response to a left CSD, and to tetrodotoxin applied to the left cerebellar cortex.
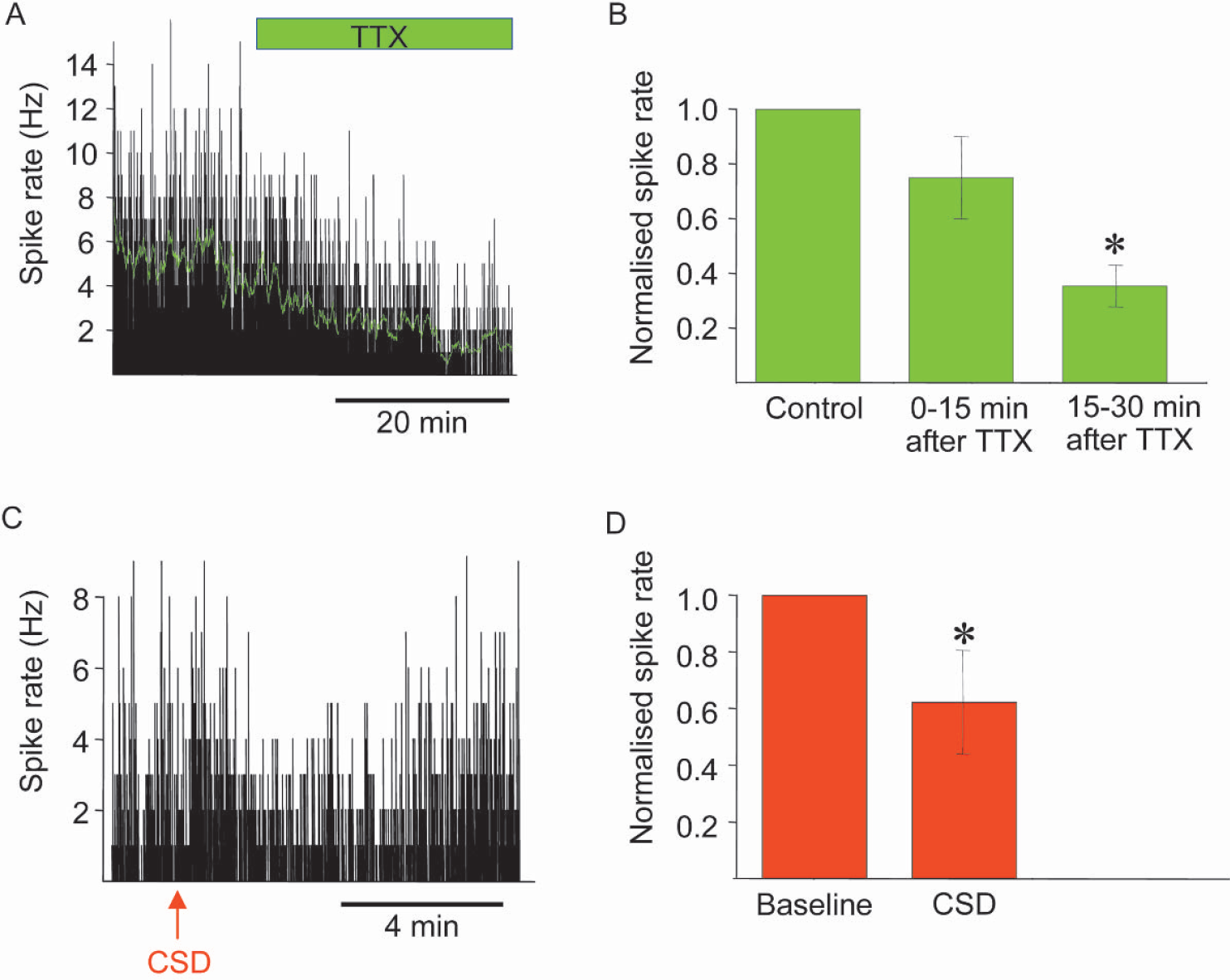
Spike rate in right sensory cortex in response to tetrodotoxin (TTX) application and cortical spreading depression (CSD) in left sensory cortex. (
To gain information about the coupling between synaptic activity and CBF we stimulated the left infraorbital nerve, which evoked robust increases in LFP and CBF amplitudes in the right somatosensory cortex (see Figs. 2 and Fig. 3) (Nielsen and Lauritzen, 2001). Functional ablation of the left sensory cortex by tetrodotoxin or CSD decreased the LFP amplitudes in the right sensory cortex by 21.9% ± 12.4% and 18.5% ± 13.7%, respectively (Figs. 2A andFig. 2C Figs. 2B and Fig. 2D show original data from one animal). Tetrodotoxin and CSD also significantly attenuated the CBF increases evoked by infraorbital nerve stimulation at 1 Hz for 10 seconds by 22.7% ± 15.9% and 41.7% ± 6.2%, respectively (Fig. 3A, C; Fig. 3B and Fig. 3D show original data from one animal). This suggests that preserved function of transhemispheric projections are necessary to maintain normal neuronal and vascular responses evoked by somatosensory stimulation. Tetrodotoxin application to the left cerebellar cortex attenuated the LFP amplitudes by 27.4% ± 13.4% in the right sensory cortex, while the evoked CBF responses remained the same (Fig. 4). This suggests that the cerebellar cortex has a facilitating effect on activity in the sensory cortex evoked by somatosensory stimulation. Figure 5 summarizes the findings from all animals in the study.
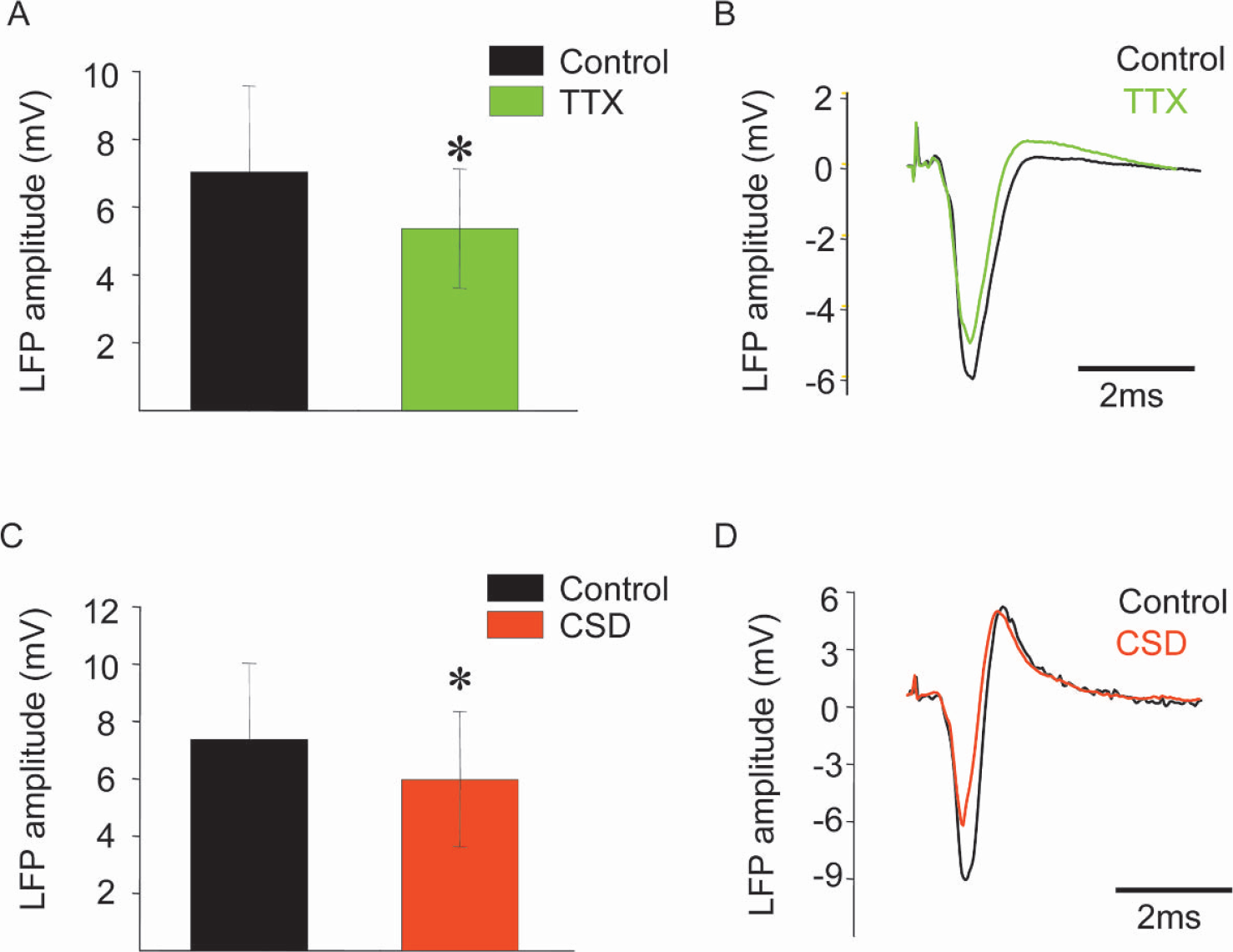
Attenuation of evoked local field potential amplitude in right sensory cortex by tetrodotoxin application or cortical spreading depression (CSD) in left sensory cortex. Stimulation of left infraorbital nerve evoked local field potentials (LFP) in right sensory cortex that were recorded at depth of 300 to 600 μm. Left panel (
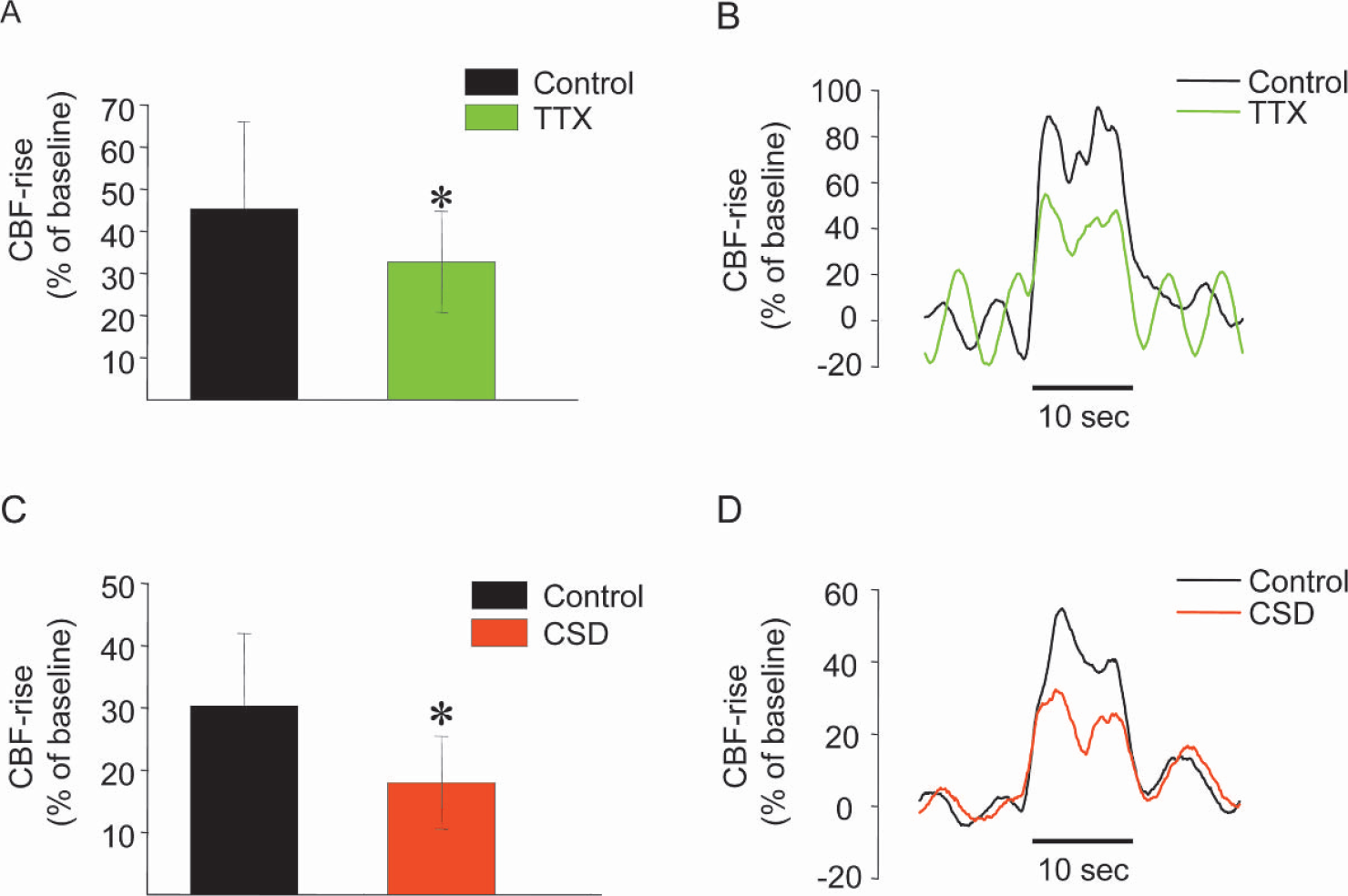
Effects of tetrodotoxin application or cortical spreading depression (CSD) in left cortex on evoked CBF responses in right sensory cortex. Left panel (
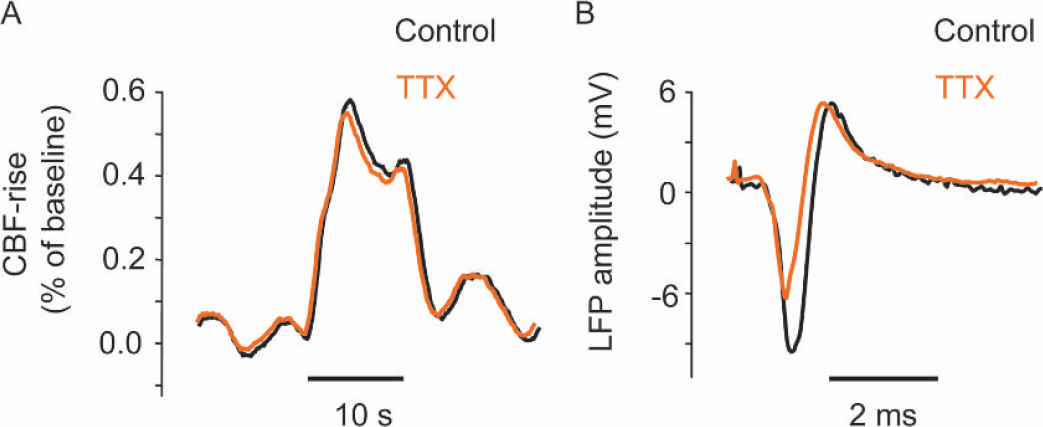
Effect of tetrodotoxin (TTX) application in left cerebellar cortex on evoked CBF response and local field potentials in right sensory cortex. Figure shows original data recorded in the right sensory cortex from one rat before and after functional ablation of the left cerebellar cortex with tetrodotoxin. Infraorbital nerve was stimulated at frequency of at 1 Hz. The left part of figure shows that the evoked CBF response in right sensory cortex remained unchanged under conditions of functional ablation of left cerebellar cortex. In contrast, the amplitude of the local field potentials decreased by 27.4% ± 13.4% (P < 0.05, Student's t-test). Black indicates control conditions, whereas orange indicates responses after tetrodotoxin application.
The increases in CBF evoked by adenosine in the right cortex were the same before and after tetrodotoxin was applied in the left cortex (138% ± 25% versus 138% ± 25%). This suggests that large-scale vascular function is preserved in the region affected by diaschisis, but does not exclude subtle changes of cerebrovascular reactivity.
DISCUSSION
Transhemispheric diaschisis
We here show that unilateral impairment of cortical neuronal activity leads to large-scale reductions in the spontaneous spike activity of the contralateral cortex that are accompanied by small or insignificant reductions in baseline CBF. In addition, we show that diaschisis affects neurovascular coupling.
The effect of tetrodotoxin and CSD on function in the contralateral somatosensory cortex may be explained by the important role that callosal projections play in the functional integration of the two hemispheres. Callosal projections from the rat sensory cortex arise from neurons in layers III and V that terminate in the superficial layers in the opposite cortex (Conti and Manzoni, 1994). Callosal projections are expected to have a facilitating influence on synaptic activity on the contralateral site, but inhibitory interneurons are excited as well, and a small percentage of the callosal axons are directly inhibitory (Bures et al., 1974; Conti and Manzoni, 1994; Reggia et al., 2001). Nevertheless, callosal projections are mainly excitatory and glutamatergic, and are dependent on D,L-alpha-amino-3-hydroxy-5-methylisoxazole-4propionic acid (AMPA) and N-methyl-
Application of tetrodotoxin to the left cortex decreased both the spontaneous spike rate and baseline CBF in the right cortex, but the effect was approximately three to four times larger for the spontaneous spike rate than for CBF. A left-sided CSD reduced the spontaneous spike rate by approximately 36% while basal CBF remained constant on the right side. This is consistent with our previous study of crossed cerebellar diaschisis produced by focal ischemia, CSD, or tetrodotoxin, which showed a disproportionate decrease in the cerebellar spike rate compared with baseline CBF (Gold and Lauritzen, 2002). A study of intrahemispheric diaschisis reported deafferentated zones without spikes, but with normal CBF (Graf et al., 1986). Taken together, these findings may suggest that the basal spike frequency has only a limited influence on the baseline level of CBF. Previous studies have shown that large ischemic lesions produce CBF reductions in the contralateral hemisphere whereas small insults do not (Andrews, 1991). This may suggest spreading of the pathologic process from the ischemic to the nonischemic region. For example, brain edema distributes not only on the side of the ischemic lesion, but also on the contralateral side (Izumi et al., 2002). Therefore, if a decrease in baseline CBF is observed contralateral to a defined lesion, this may be explained by factors other than diaschisis (Andrews, 1991; Feeney and Baron, 1986).
Tetrodotoxin on the left cortex attenuated the amplitude of the LFP and CBF responses in the right cortex to the same degree whereas a CSD in the left cortex attenuated the right-sided CBF response twice as much as the LFP amplitude. This difference is not explained by differences in the effect on the spontaneous spiking activity because tetrodotoxin reduced the spike rate in the right sensory cortex more than CSD. The difference may relate to the different ways in which tetrodotoxin and CSD affect cortical function. Tetrodotoxin decreases neuronal function by blocking action potential propagation and hence all ongoing afferent and efferent neuronal activity. The effect of tetrodotoxin may be more pronounced in superficial cortical layers because it is applied topically. In contrast, during CSD the whole cerebral cortex undergoes a profound depolarization that eradicates all spontaneous and evoked electrocortical activity for 5 to 10 minutes (Bures et al., 1974; Leao 1944).
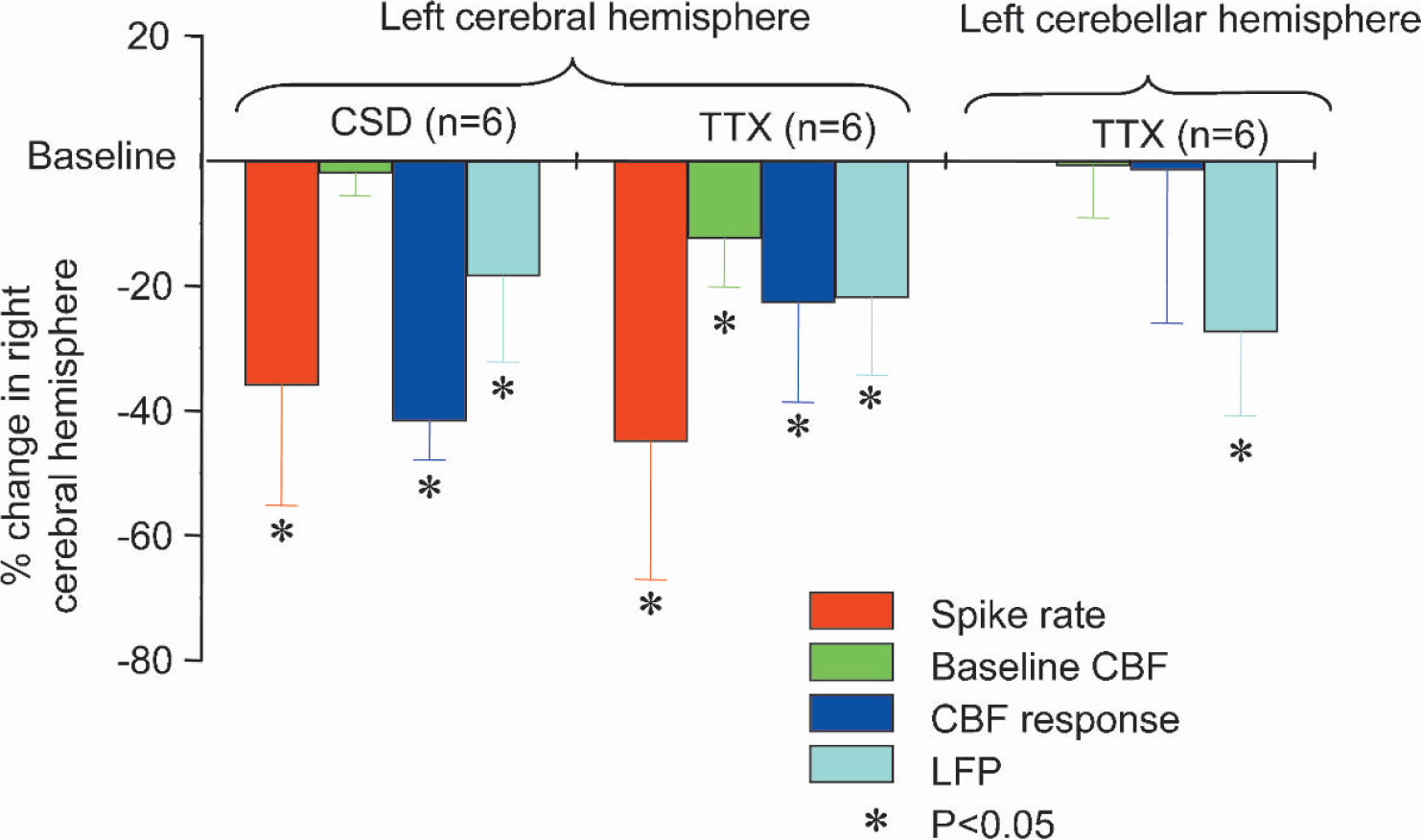
Summary of influence of diaschisis on spontaneous spike activity, baseline CBF, and local field potentials (LFP) and CBF amplitudes evoked by sensory stimulation. Ordinate indicates decline of variables in right cerebral cortex caused by functional ablation of left cerebral cortex in percent of the control condition. Transhemispheric diaschisis was induced by topical application of tetrodotoxin (TTX; n = 6 rats) or cortical spreading depression (CSD; n = 6 rats) in left cerebral cortex. Cerebellocortical diaschisis was induced by topical application of tetrodotoxin to the left cerebellar cortex (n = 6). Asterisk indicates a significant decrease (P < 0.05).
Our data are consistent with previous studies that reported decreases and increases in the cortical somatosensory evoked potentials in the healthy cortex produced by cerebral cortical lesions on the opposite side (Andrews and Muto, 1992; Bo et al., 1987; Graf et al., 1986; Hossmann et al., 1985; Lopes da Silva et al., 1985; Matsumiya et al., 1990; Sakatani et al., 1990). A reduction in the amplitudes of the evoked responses may reflect not only transhemispheric diaschisis, but also spread of the pathologic process to the opposite side. However, increases in the amplitude of the somatosensory evoked potentials reported in some studies might reflect a reduction in cortical excitation of the inhibitory interneurons in the nuclei gracilis and cuneatus. This is expected to increase the amplitude of somatosensory evoked potentials in the healthy cortex due to an increase in the amplitude of the afferent input to the thalamus (Sakatani et al., 1990). Still other studies have suggested increases in neuronal excitability by disinhibition of cortical neurons (Witte et al., 2000).
Our results conform to the classical definition of diaschisis as a transient decrease in spontaneous activity due to attenuated function in the projecting region. This decrease in overall excitability may also explain the matched decrease of synaptic activity and CBF that accompanied cortical application of tetrodotoxin on the left side. However, this cannot explain that CSD induced a comparatively larger attenuation of the CBF response compared with the decrease in synaptic activity. CSD may induce changes on the opposite side of which we are ignorant at the moment, but it is important to note that a CSD does not propagate from one hemisphere to the other, and that a CSD on one side does not interfere with the circulation or cause edema in the contralateral cortex (Bures et al., 1974).
Cerebellocortical diaschisis
Cerebellocortical diaschisis has been investigated in both clinical (Gasparini et al., 1999; Komaba et al., 2000; Tecco et al., 1998) and experimental studies (Botez-Marquard et al., 2001; Middleton and Strick, 1997; Sasaki et al., 1972a,b). Cerebellocortical projections first synapse in the thalamus and then reach the frontal, sensorimotor, and associative areas (Botez-Marquard et al., 2001; Middleton and Strick, 1997). Cerebellothalamocortical projections mainly target the motor and parietal cortex. (Sasaki et al., 1972a,Sasaki et al., 1972b; Wannier et al., 1992), but activity in the sensory cortex is also modulated by cerebellothalamic projections (Blakemore et al., 1999). Suppressed function of the left cerebellar cortex had no effect on baseline or evoked CBF in the right sensory cortex despite a significant decrease in the evoked LFP amplitude, suggesting that the cerebellum facilitates cortical synaptic activity in response to stimulation of somatosensory afferents. This is in agreement with observations from two clinical studies in humans, which reported that a unilateral cerebellar lesion decreased the contralateral cortical excitability (Restuccia et al., 2001), and that baseline hemispheric CBF remained constant contralateral to a cerebellar lesion (Gasparini et al., 1999).
CONCLUSION
Acute transhemispheric diaschisis was characterized by a marked decrease in the spontaneous spike rate accompanied by small or insignificant changes in baseline CBF. Somatosensory evoked potentials were reduced in the cortex affected by transhemispheric diaschisis, and neurovascular coupling was affected as well. This is in contrast to crossed cerebellar diaschisis in which cerebellar excitability and neurovascular coupling is preserved (Gold and Lauritzen, 2002). The data suggest differences in manifestations of diaschisis between brain regions.
Footnotes
Acknowledgment
The authors thank Lillian Grøndahl for expert technical assistance.
