Abstract
Ischemic stroke continues to be a leading cause of morbidity and mortality throughout the world. To protect and/or repair the ischemic brain, a multitiered approach may be centered on neural stem cell (NSC) transplantation. Transplanted NSCs exert beneficial effects not only via structural replacement, but also via immunomodulatory and/or neurotrophic actions. Unfortunately, the clinical translation of such promising therapies remains elusive, in part due to their limited persistence/survivability within the hostile ischemic microenvironment. Herein, we discuss current approaches for the development of NSCs more amenable to survival within the ischemic brain as a tool for future cellular therapies in stroke.
Keywords
Introduction
Stroke is one of the most common causes of death/long-term disability worldwide and current epidemiological data suggest that the burden of this disease will continue to increase in the coming decades, particularly in developing countries. 1 To date, therapeutic efforts have centered on the development of etiological therapies (e.g. thrombolysis) or the improvement of functional recovery for stroke patients (e.g. neurorehabilitation). However, despite the intensity of research performed, there are, as of yet, no therapies capable of regenerating the injured central nervous system (CNS) after a stroke has occurred. As such, there exists a need to re-evaluate stroke pathobiology in order to identify novel therapeutic targets and truly effective regenerative approaches. The vast majority of strokes in the United States are ischemic (87%), while 10% are intracerebral hemorrhagic strokes with the remaining 3% being subarachnoid hemorrhages. 1 Once cerebral ischemia occurs, it quickly triggers pathological pathways associated with an “ischemic cascade” that cause irreversible neuronal injury. 2 The primary driver of this pathobiology is the local crisis in energy availability caused by extreme changes within the parenchymal microenvironment, such as variations in oxygen/glucose concentrations and the depletion of cellular energy stores. 3 Concomitant release of neurotransmitters, inflammatory cytokines, chemokines, and reactive oxygen species furthers local ischemic damage.4,5 To be fully applicable and effective in ischemic stroke, regenerative therapies must ultimately modulate and counteract the complicated network dynamics operating in the context of post-ischemic tissue. 6
Neural stem cells and ischemic stroke
The proliferation and migration of endogenous NSCs in response to experimental stroke are key spontaneous regenerative processes that occur within the ischemic brain.7,8 Reports demonstrate that the endogenous neurogenic response influences the course of recovery in both short and long-term experimental settings.9,10 Stroke-induced compensatory neurogenesis occurs in the human brain as well, where it may contribute to post-ischemic recovery.
11
However, it is clear that this physiologic endogenous repair response is far from ideal, as patients continue to experience various levels of physical and cognitive morbidities post-ischemic injury.12–14 As such, the stroke-induced neurogenic response will clearly need to be supported and/or supplemented to become a clinically valuable tool. Current approaches seeking to accomplish the aforementioned encompass two predominant modalities: one acting on the endogenous compartment and the other focused on the delivery of exogenous stem cells. The NSCs and Ischemic Stroke. NSC proliferation within the SVZ is augmented after ischemic stroke, leading to the generation of neuroblasts that migrate along vessels toward gradients of chemokines produced locally by both glia and inflammatory cells. Furthermore, transplanted NSCs release several factors that can directly increase cell proliferation within the SVZ, thereby potentiating neuroblast migration. Transplanted NSCs can also positively affect the differentiation of endogenous neuroblasts and plasticity within the ischemic tissue via providing neurotrophic support and/or by direct differentiation into post-mitotic neurons, astrocytes, or oligodendrocytes. Most importantly, transplanted NSCs secrete myriad soluble molecules that modulate the activation of host microglia/macrophages thereby modifying the release of inflammatory mediators which amongst a litany of effects can serve to inhibit or increase endogenous neurogenesis. See Hermann et al.
17
for a comprehensive review. To prevent the death of transplanted exogenous NSCs and, by proxy, endogenous NSCs within ischemic microenvironments, one may employ strategies centered on preconditioning or cellular engineering prior to engraftment.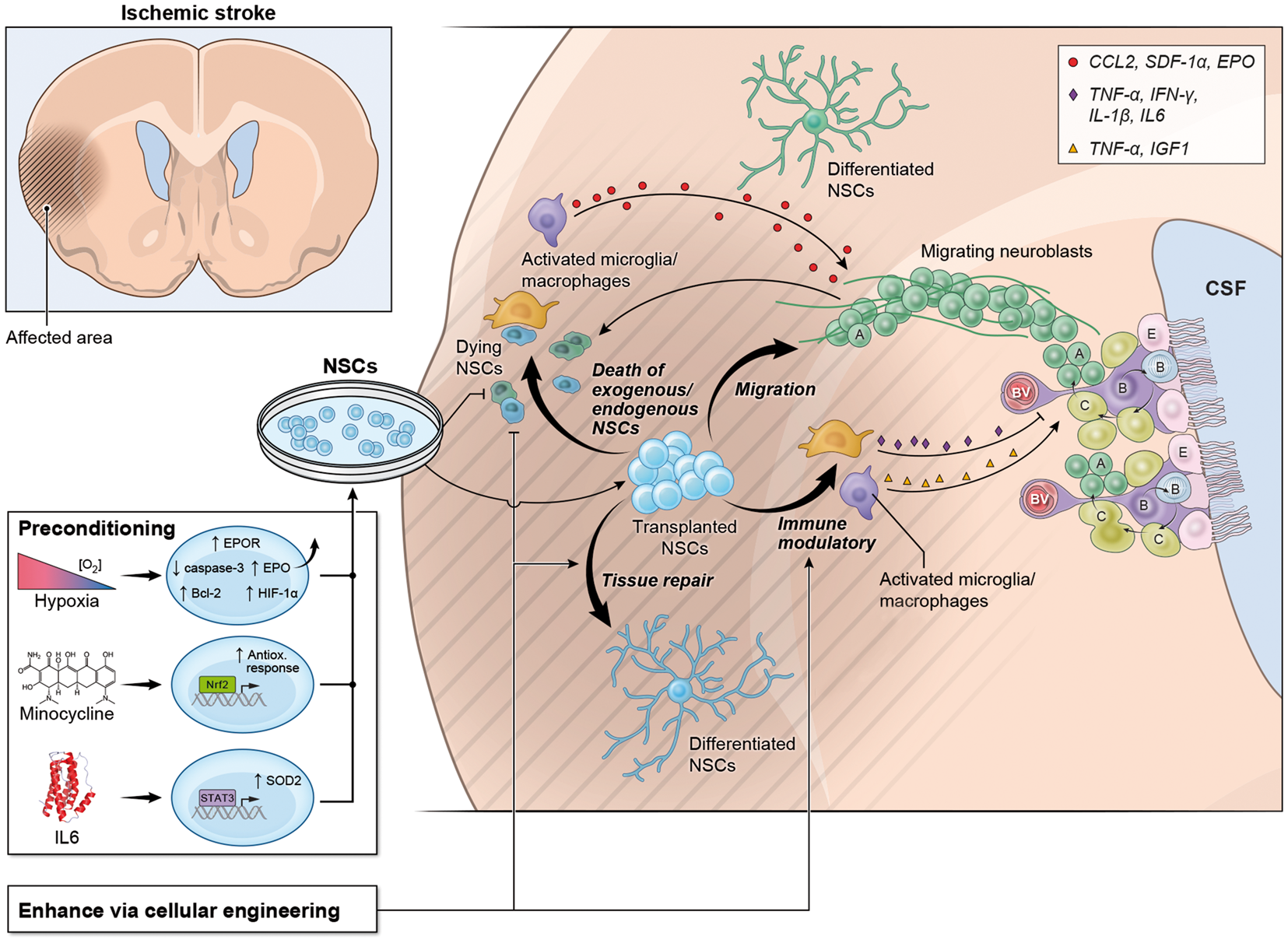
Engineering of NSCs equipped to survive/modulate the post-stroke microenvironment
NSCs genetically modified for ischemic stroke.

Bcl-x(L): B-cell lymphoma-extra-large; BDNF: brain derived neurotrophic factor; GDNF: glial cell-derived neurotrophic factor; HIF-1α: hypoxia-inducible factor-1α; Hsp70: heat shock protein 70; NGF: nerve growth factor; NT-3: neurotrophin-3; SOD1: copper/zinc-superoxide dismutase; TAT: trans-activator of transcription; tMCAO: temporary middle cerebral artery occlusion; VEGF: vascular endothelial growth factor.
Discussion/conclusions
The paucity of regenerative therapeutic options for those patients who suffer a cerebrovascular accident stands in stark contrast to the intensity of research efforts that have been performed to date. It is the authors’ contention that increasing the viability of NSCs after transplant will create more efficacious and stable cellular medicines. Several approaches have been investigated so far, and if proven successful they may find utility not only in the realm of stroke therapy, but may also be extended to a wide variety of other degenerative/inflammatory neurological disorders that share components of stroke pathobiology (e.g. Alzheimer’s, Multiple Sclerosis, etc.).
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Intramural Research Program of the NINDS/NIH and core support grant from the Wellcome Trust and MRC to the Wellcome Trust–Medical Research Council Cambridge Stem Cell Institute. Additionally, JDB was supported by a NIH-OxCam Fellowship, LPJ by a Wellcome Trust Research Training Fellowship (RRZA/057 RG79423) and FAG by a Gates-Cambridge Fellowship.
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
