Abstract
Stroke-associated pneumonia is a frequent complication after stroke associated with poor outcome. Dysphagia is a known risk factor for stroke-associated pneumonia but accumulating evidence suggests that stroke induces an immunodepressive state increasing susceptibility for stroke-associated pneumonia. We aimed to confirm that stroke-induced immunodepression syndrome is associated with stroke-associated pneumonia independently from dysphagia by investigating the predictive properties of monocytic HLA-DR expression as a marker of immunodepression as well as biomarkers for inflammation (interleukin-6) and infection (lipopolysaccharide-binding protein). This was a prospective, multicenter study with 11 study sites in Germany and Spain, including 486 patients with acute ischemic stroke. Daily screening for stroke-associated pneumonia, dysphagia and biomarkers was performed. Frequency of stroke-associated pneumonia was 5.2%. Dysphagia and decreased monocytic HLA-DR were independent predictors for stroke-associated pneumonia in multivariable regression analysis. Proportion of pneumonia ranged between 0.9% in the higher monocytic HLA-DR quartile (≥21,876 mAb/cell) and 8.5% in the lower quartile (≤12,369 mAb/cell). In the presence of dysphagia, proportion of pneumonia increased to 5.9% and 18.8%, respectively. Patients without dysphagia and normal monocytic HLA-DR expression had no stroke-associated pneumonia risk. We demonstrate that dysphagia and stroke-induced immunodepression syndrome are independent risk factors for stroke-associated pneumonia. Screening for immunodepression and dysphagia might be useful for identifying patients at high risk for stroke-associated pneumonia.
Introduction
Stroke associated pneumonia (SAP) is the most frequent and a potentially preventable complication after stroke showing consistent association with poor outcome.1,2 Initial concepts on the aetiology of SAP focussed on stroke-facilitated aspiration. In fact, dysphagic patients are particularly threatened with an up to 11-fold increased risk for SAP. 3 However, dysphagia alone is not sufficient for the development of SAP. 4 Moreover, therapeutic measures to prevent aspiration do not effectively protect against SAP. 5 Accumulating experimental and clinical evidence suggests that stroke induces a rapid, temporary immunodepression mainly mediated by the autonomic nervous system.6–8 Stroke-induced immunodepression syndrome (SIDS) is characterized by a down-regulation of systemic cellular immune responses, i.e. rapid numerical decrease in peripheral blood lymphocyte subpopulations and functional deactivation of monocytes resulting in an increased susceptibility for SAP.8–11 A strong association between SAP and decreased monocytic HLA-DR expression (mHLA-DR) as a marker for immunodepression has been shown in previous studies.12,13 Infection markers such as lipopolysaccharide-binding protein (LBP), essential for the response to bacterial lipopolysaccharides and the pro-inflammatory cytokine interleukin-6 (IL-6) are further interesting candidate markers for the prediction of SAP.14,15 Therefore, we designed the PREDICT-study (a) to confirm that markers of immunodepression predict SAP independently from dysphagia and (b) to determine the diagnostic value of selected markers of immune function (mHLA-DR), inflammation (IL-6) and infection (LBP) on day 1 in improving prediction of SAP between 2 and 7 days after stroke onset.
Materials and methods
The PREDICT study (www.clinicaltrials.gov, NCT01079728) is a prospective, international, multicenter study with 11 study sites in Germany and Spain experienced in biomarker studies. In 9 from 11 study centres, patients have been recruited. German study sites were selected by asking all members of the Berlin Stroke Alliance (BSA) for participating in the study. Participating German study sites comprised the three hospitals of the University Hospital Charité Berlin (Campus Mitte, Campus Virchow-Klinikum and Campus Benjamin Franklin), five non-university hospitals of the Berlin-Brandenburg region and one university hospital in Spain (Vall d’Hebron in Barcelona). All study sites were instructed according to GCP standards (e.g. initiation visit of the centres, monitoring of all centres and CRFs). The study protocol required daily screening of new admissions to the study site’s stroke units for eligible patients. Daily screening for eligible patients was performed during the week and on weekends. All eligible patients were asked for participation in the study. Patients participating in stroke trials were not included. Reasons for non-participation were not documented. All participating patients or their legal representatives gave written informed consent in accordance with the Declaration of Helsinki. The study was approved by the ethics committees of the Charité – Universitätsmedizin Berlin (EA1/216/09) and Hospital Vall d’Hebron Barcelona (PR_IR_170-2012). Patients were included between January 2010 and December 2012. Inclusion criteria were acute ischemic stroke of any severity (NIHSS ≥ 1 at study inclusion, TIA excluded) and any localization, symptom onset ≤36 h and age ≥18 years. Exclusion criteria were intracerebral haemorrhage, preexisting dysphagia, mechanical ventilation, antibiotic treatment or immunosuppressive therapy within the last 4 weeks and clinical/paraclinical signs of infection at study inclusion. Blood samples were obtained as soon as possible but no longer than 36 h after symptom onset (day 1) and repeated on days 2–4.
Clinical data
Sociodemographics, neurological deficits, comorbidities, stroke severity on admission, complications processes and outcomes during hospitalization were documented. The following data definitions were used in the current analyses: age (categorized a priori as <65 years, 65–74 years, 75–84 years, ≥85 years); sex; comorbidities including hypertension and diabetes mellitus (any treatment and/or patient’s self report and/or new diagnosis during hospital admission); previous stroke (neurological deficit > 24 h prior to current event); atrial fibrillation (patient’s self report and/or documented by standard ECG or Holter monitor); stroke severity on admission assessed by the National Institute of Health Stroke Scale (NIHSS) and categorized based on previously defined cut points into: mild (NIHSS < 5), moderately severe (NIHSS 5–15), severe/very severe (NIHSS ≥ 16) impairment; clinical symptoms on admission including evidence for any type of dysphagia documented by a dysphagia bedside screening test. Etiological subtype of ischemic stroke was described according to the TOAST criteria. 16 Patients were assessed for signs of pneumonia on days 1–4 and day 7 (or day of discharge in case of length of stay < 7 days). Diagnosis of SAP during hospitalization was defined by the study/treating physician based on clinical symptoms and/or suggestive clinical examination and/or radiological findings and/or pathogen detection of pulmonary infection. Criteria for ‘clinically defined pneumonia’ by the US Centers for Disease Control and Prevention (CDC) 17 were assessed at each visit. On each visit, provision and reason (pneumonia/urinary tract infections/other) for antibiotic treatment was inquired. Antibiotic choice was left to the discretion of the treating physician, type of antibiotics used was not documented. Outcome assessment was performed at three months after stroke via a structured telephone interview assessing the modified Rankin Scale (mRS). Poor outcome was defined a priori as mRS score of 3 or higher including death. 18
Immunological data
Blood samples parameters were obtained on days 1–4 after stroke onset. For plasma collection samples were centrifuged (200 g, 4℃, 10 min) and stored at −80℃. Plasma concentrations of IL-6 and LBP were determined with the IMMULITE™ semi-automatic chemiluminescent immunoassay (Siemens Medical Solutions, Bad Nauheim, Germany). The detection limit for IL-6 is 2 pg/ml and 0.8 µg/ml for LBP. Expression of human leukocyte antigen-DR (HLA-DR) on monocytes was determined by flow cytometry using a highly standardized quantitative assay as described earlier. 19 In short, whole blood in Vacutainer tubes (BD Biosciences, San Jose, CA, USA) containing EDTA was stained with 20 µl of monoclonal phycoerythrin-conjugated anti–HLA-DR and PerCP-Cy5.5–conjugated anti-CD14 antibodies (Quantibrite™ HLA-DR/monocyte; BD Biosciences) in the dark at room temperature for 30 min. Erythrocyte lysis was achieved with 0.5 ml of lysing solution (BD Biosciences) for another 30 min at room temperature. Finally, the cells were washed with 1 ml PBS buffer containing 2% FCS and analyzed on a Navios flow cytometer (Beckman Coulter, Krefeld, Germany). HLA-DR surface expression on monocytes is given as monoclonal antibodies bound per cell (mAb/cell).
Statistics
The t-test was used to test differences in continuous variables, and the χ2 test or Fisher’s exact test for those in proportions. To assess the association between biomarkers and SAP risk, levels of biomarkers were categorized into quartiles according to their underlying distribution in the study population. To estimate Odds Ratio (OR) and the resulting 95% confidence intervals (CI) of the association between quartiles of biomarkers and SAP risk, logistic regression analyses were performed. Analyses were stratified for dysphagia status. As in one of the biomarkers (IL-6), no SAP was observed in the lowest quartile, the problem of quasi-separation appears resulting in non-existence of OR and CI estimates. In these instances, Firth logistic regression was used and the SAS-macro ‘%fl’ was applied, estimating ORs and CIs with penalized likelihood estimation methods and corresponding penalized likelihood ratio tests. To assess the influence of log-transformed levels of biomarkers on pneumonia risk independent of dysphagia status, clinical evidence for dysphagia was included in the regression model. Nagelkerke’s R2 and C-statistics were calculated for assessing the fit and discrimination of the logistic regression models. Analyses were restricted to patients without missing values. The number of patients with missing values ranged from 0% for age to 14% for dysphagia. Time course of biomarker levels from day 1 to 4 is presented as box-and-whiskers plots with whiskers representing the 2.5 and 97.5 percentiles. Between-group differences were analysed using Kruskal–Wallis one-way ANOVA with Dunn’s multiple comparison test. All tests were two-tailed and statistical significance was determined at an alpha level of 0.05. Statistical analyses were performed with SPSS 20.0, SAS 9.3 and GraphPad Prism 6.0 software packet. The underlying PREDICT data set is available as supplementary file.
Results
Patient’s demographic and clinical characteristics
Baseline characteristics stratified by pneumonia. a
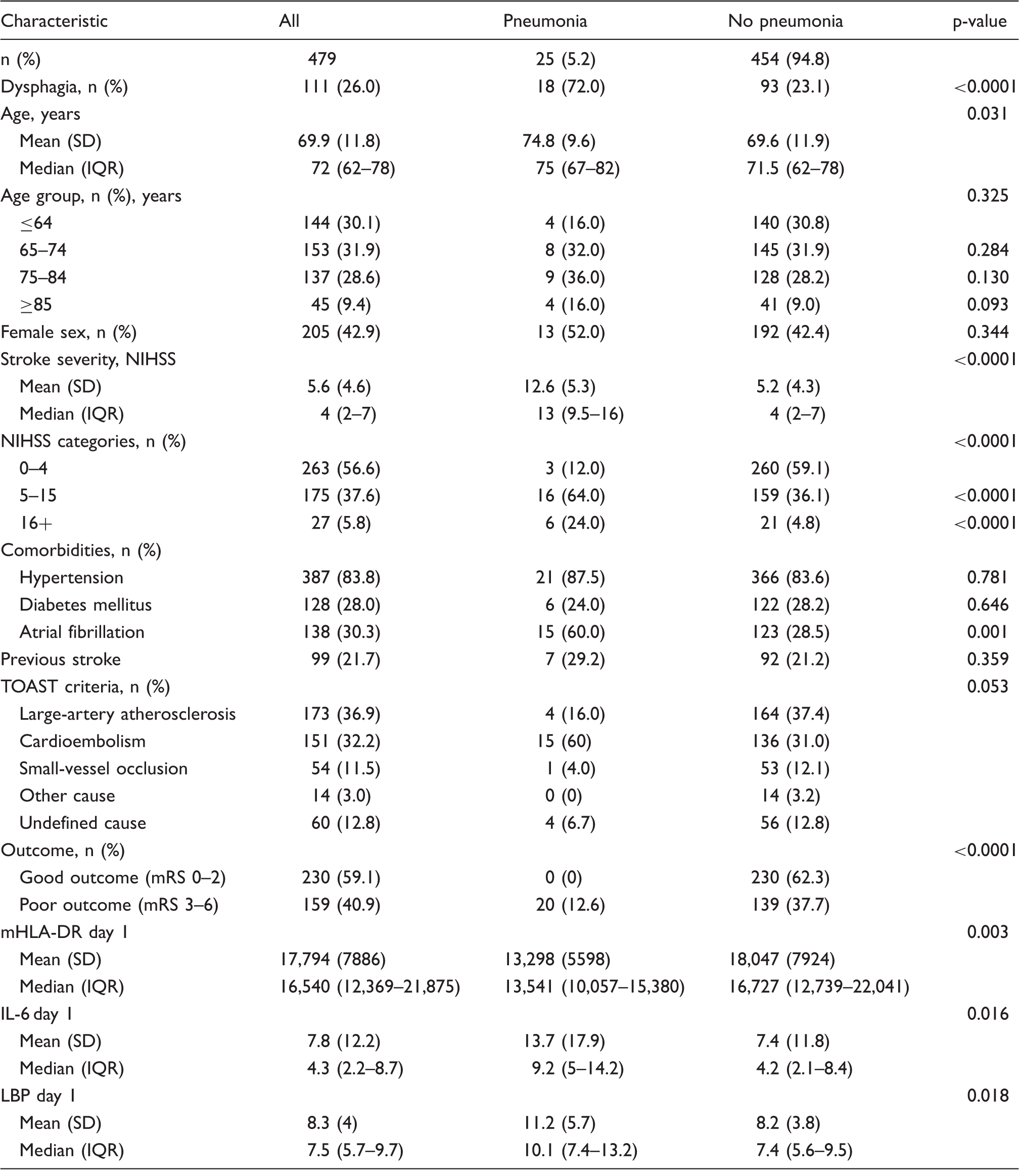
Analysis were restricted to patients without missing values for pneumonia and the respective variable. Percentages within the groups pneumonia/no pneumonia refer to patients with dysphagia.
Association between biomarkers, dysphagia and pneumonia
Level of biomarkers (quartiles) and frequency of pneumonia by dysphagia status. a
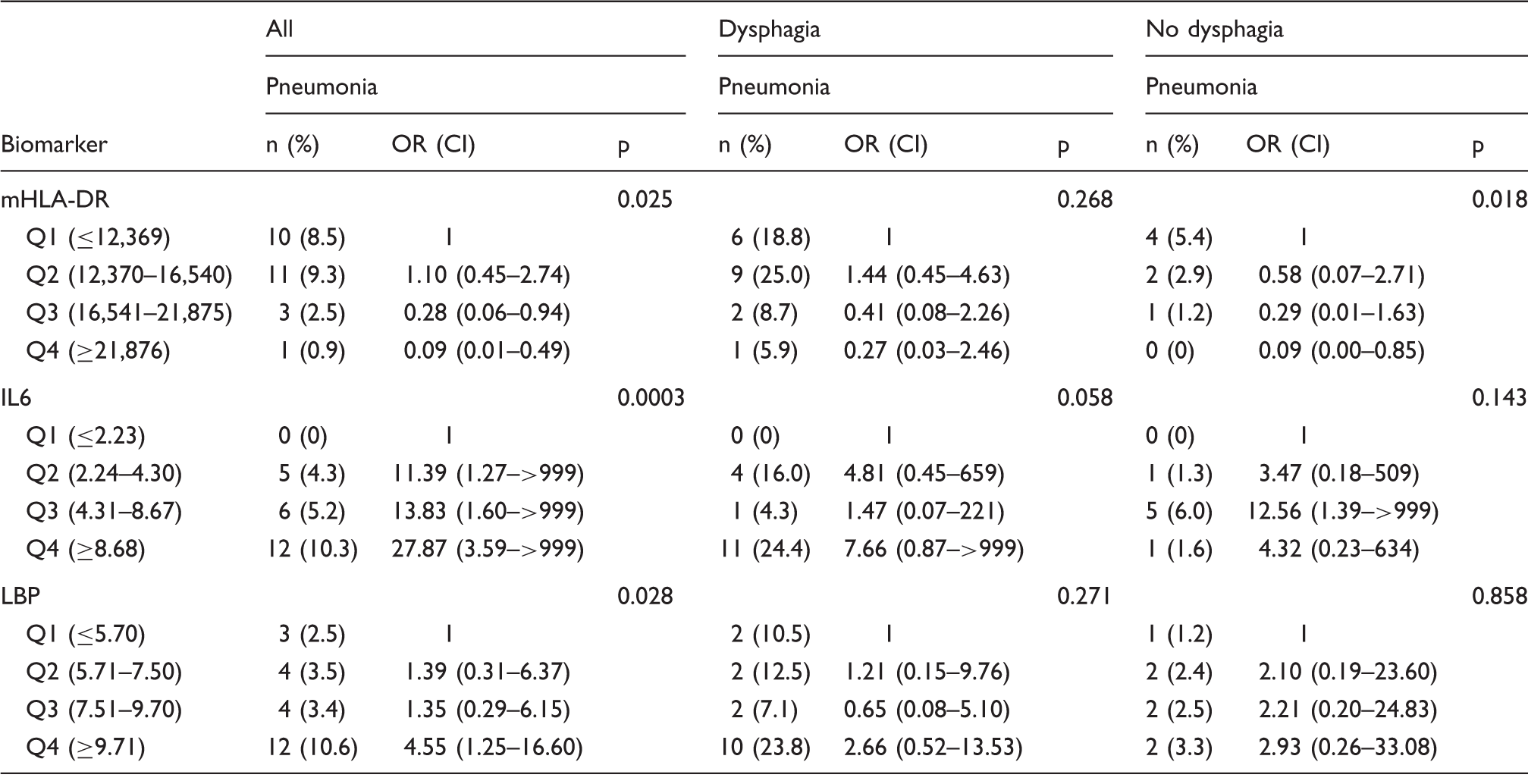
Analysis restricted to patients without missing values; p-value refers to proportion of pneumonia across quartiles of respective biomarker in patients with known status of pneumonia.
mHLA-DR as marker of immunodepression
Proportion of pneumonia ranged between 0.9% in the higher mHLA-DR quartile (>21,876 mAb/cell) and 8.5% in the lower quartile (<12,369 mAb/cell). In the presence of dysphagia, proportion of pneumonia increased to 5.9% and 18.8% in the higher and lower quartile, respectively. Patients without dysphagia and low mHLA-DR expression had a moderate risk of pneumonia (5.4%). Within the stratum of patients without dysphagia and normal mHLA-DR expression no SAP (0%) was observed.
IL-6 as inflammation marker and LBP as infection marker
SAP is associated with increased IL-6 levels. Patients in the lower quartile (≤2.23 pg/ml) had no risk for SAP. Even in the presence of dysphagia, risk for pneumonia remained 0%. Proportion of pneumonia in the higher quartile (>8.68 pg/ml) was 10.3% and increased to 24.4% in the presence of dysphagia. A similar relationship was found for dysphagia and increased LBP levels. Proportion of pneumonia ranged between 2.5% in the lower (≤5.70 µg/ml) and 10.6% in the higher quartile (≥9.71 µg/ml, p-value = 0.028). In the presence of dysphagia, pneumonia increased to 10.5% and 23.8% in the higher and lower quartile, respectively. Patients without dysphagia and high LBP levels had a moderate risk (3.3%) of SAP.
Biomarkers and dysphagia as predictors for SAP: Univariate and multivariable regression analysis
Level of biomarkers (quartiles and log-transformed) and its association with pneumonia. a
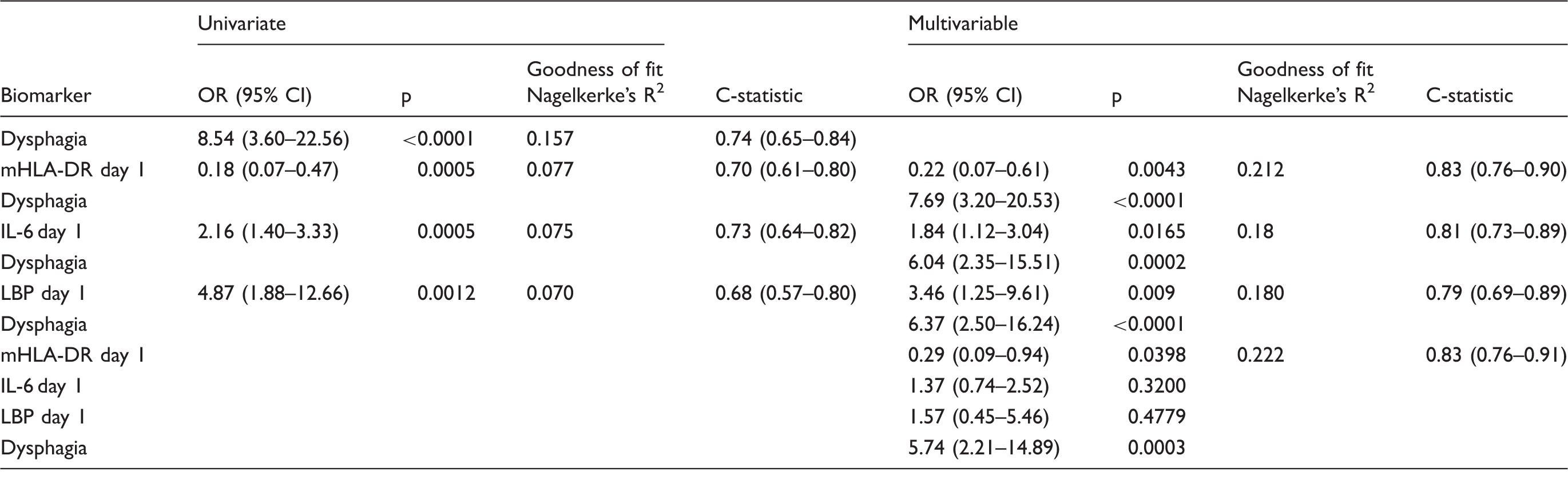
Analysis restricted to patients without missing values; multivariable analyses adjusted for dysphagia status.
Effect modification of predictive value of IL6, LBP and dysphagia on SAP depending on the mHLA-DR level.
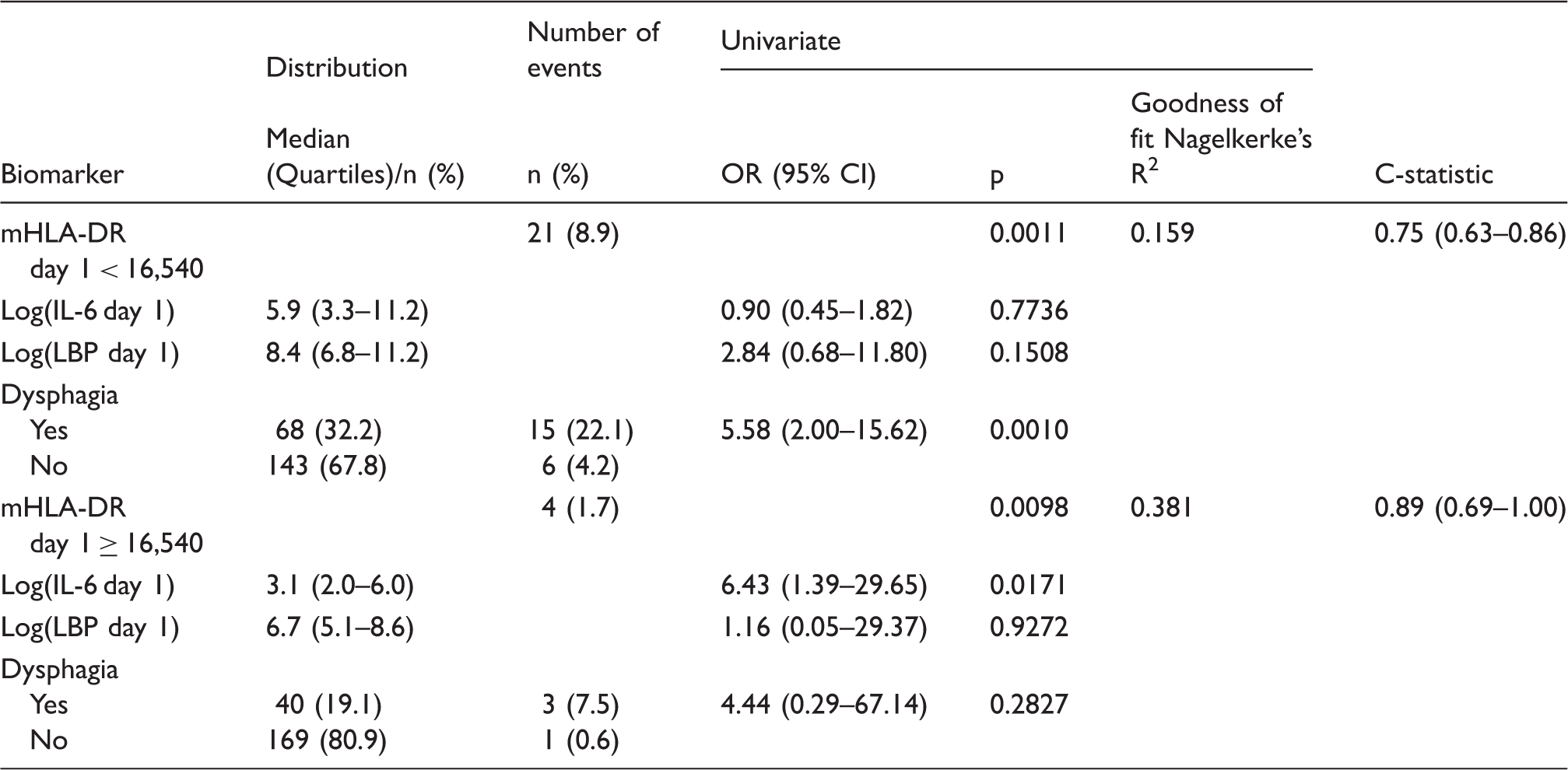
Time course of immune, infection and inflammation markers in patients with and without SAP
Figure 1 shows the time course of biomarker levels (mHLA-DR, IL-6 and LBP) in patients with and without SAP as well as for patients receiving antibiotic treatment compared with patients not receiving antibiotic treatment. Patients with SAP and receiving antibiotic treatment had significantly lower mHLA-DR levels on days 1–4 after stroke onset. mHLA-DR expression was lowest on day 3 in patients with SAP (mean 9635 mAb/cell). Patients with SAP and receiving antibiotic treatment had furthermore significantly increased IL-6 and LBP levels on days 1–4 compared with patients not suffering SAP. IL-6 levels were highest on day 2 (mean 55.3 pg/ml) in patients with SAP, LBP levels were highest on day 4 (mean 22.9 µg/ml).
Time course of biomarkers after stroke onset (days 1 to 4). Monocytic HLA-DR (mHLA-DR) levels were significantly decreased while IL-6 and LBP-levels were significantly increased in patients with stroke-associated pneumonia (SAP, panels a–c) and those receiving antibiotics (at any time point during the observation period) compared with patients not receiving antibiotics (panels d–f), respectively. Grey boxplots indicate patients with SAP or antibiotic treatment and open boxplots indicate patients without SAP or antibiotic treatment. In patients with SAP and those receiving antibiotics, alterations in biomarkers were significant already on day 1 but became even more pronounced on days 2 to 4. Dashed lines indicate the lower (mHLA-DR) and upper limit of normal (IL-6, LBP), respectively. *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001 (Kruskal–Wallis one-way ANOVA with Dunn’s multiple comparison test).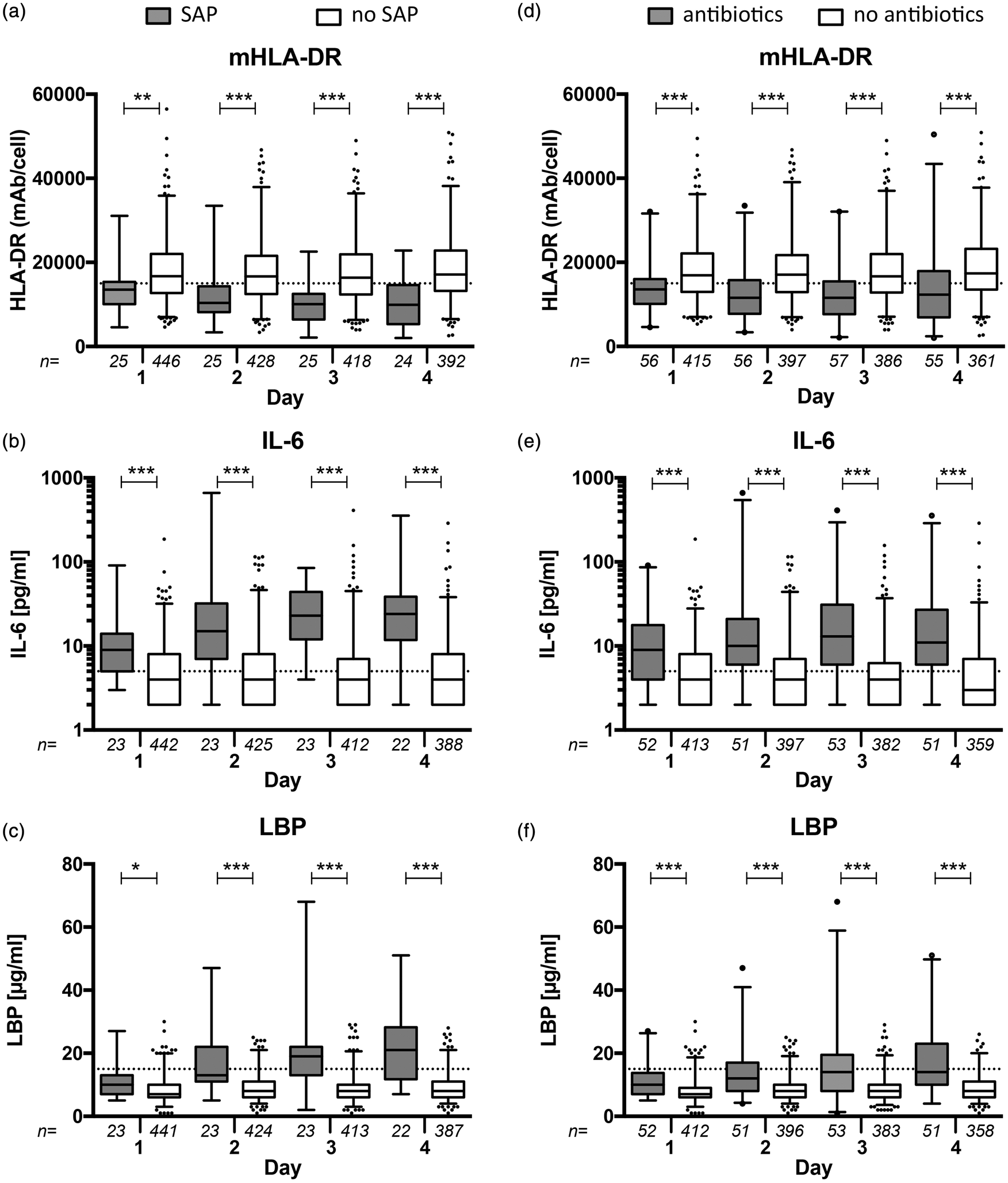
Discussion
Despite improvements in acute stroke care, SAP remains the most frequent complication after stroke and is associated with poor outcome. 2 Early identification of patients at high risk for SAP might help to justify increased monitoring and tailored prophylactic measures in clinical routine. Currently, aspiration and SIDS are the two pathophysiologically postulated concepts on the development of SAP. 11 However, there is an ongoing debate as to whether SIDS actually is a standalone risk factor for SAP or if immunological changes are an epiphenomenon of brain injury and/or imminent infections. The PREDICT-study is a proof of concept study designed to confirm that selected biomarkers are predictors for SAP independently of presence of dysphagia. For this purpose, we performed a prospective observational study and assessed dysphagia as well as early markers for immunodepression, inflammation and infection within 36 h after symptom onset for their predictive properties.
The main finding of PREDICT is that, both, dysphagia and immunodepression were independent predictors for SAP. Interestingly, dysphagia was only significantly associated with SAP in patients with mHLA-DR levels below the median. This finding supports our hypothesis that SAP is the result of two independent mechanisms, aspiration and SIDS, leading to an increased susceptibility for pulmonary infections. Patients with dysphagia and low mHLA-DR expression are at a particularly high risk for SAP.
Reduced mHLA-DR expression was a robust predictor for SAP in all of the calculated models. Monocytes from stroke patients have decreased expression of major histocompatibility complex (MHC) class II. 7 Impaired monocyte functions result in insufficient antigen-presentation and decreased expression of secreted or membrane-bound costimulatory molecules and may, therefore, contribute to reduced lymphocyte responses. 20 In previous studies, we and others demonstrated that reduced mHLA-DR expression on day 1 after stroke onset is a strong independent predictor for SAP.12,14,19 However, none of these previous studies assessed dysphagia and mHLA-DR expression for their independent predictive properties on SAP.
Higher IL-6 levels on day 1 were associated with a higher risk for SAP even after adjustment for dysphagia but not after additional adjustment for mHLA-DR and LBP. Further analysis showed that a significant association between IL-6 levels and SAP was only seen in patients with mHLA-DR levels above the median. We hypothesize that this finding might be explained by the immunodepressive state in patients with a low mHLA-DR expression resulting in a reduced production of the proinflammatory cytokine IL-6. Yet, this hypothesis needs further investigations reproducing our findings. IL-6 has recently been shown to be associated with post-stroke infections. 21 However, the study by Kwan et al. did not assess dysphagia status and blood samples were obtained within 72 h after stroke onset hampering early SAP prediction since about half of pneumonia cases occur within the first 48 h after stroke onset. 1
Higher LBP levels on day 1 were associated with SAP only after adjustment for dysphagia but not after additional adjustment for mHLA-DR and IL-6 indicating that markers of immunodepression and inflammation are more suitable in the early prediction of SAP. LBP is an acute phase protein of the host in response to bacterial lipopolysaccharides of Gram-negative bacteria. 22 Therefore, LBP might be considered as a marker for infections caused by the most frequently identified pathogens in SAP.23,24 An association between overall post-stroke infections and increased LBP levels has been shown in two previous studies without providing data on microbiological findings.14,15
An important measure to assess the accuracy of a prediction model is discrimination describing the ability to distinguish subjects with different outcomes. 25 Interestingly, mHLA-DR expression and presence of dysphagia had comparable, good discrimination abilities, which could be increased to excellent discrimination abilities when combining information on both parameters.
SAP is an early complication after stroke, usually occurring between days 2 and 7 after stroke. Biomarkers and clinical parameters were obtained within 36 h after stroke facilitating early prediction of SAP. Although we observed altered levels of mHLA-DR, IL-6 and LBP in patients with SAP already within 36 h after stroke onset, differences became even more pronounced on the following 3 days (Figure 1). In fact, multivariable regression analysis of biomarkers obtained on days 2–4 reveals that the association of biomarkers with SAP becomes so strong that presence of dysphagia is eliminated as independent predictor for SAP though this might be due to beginning infection (data not shown). The number of patients receiving antibiotics is considerably higher than the number of patients with SAP (Figure 1). This is mainly explained by indications for antimicrobial therapy other than SAP. Those included urinary tract infections (17 patients), sepsis (1 patient) and ‘other indications’ that were not specifically listed (16 patients). Moreover, clinicians seem to treat with antibiotics when in doubt about a bacterial infection, which has also been shown in PASS and STROKE-INF.26,27
Our study has several limitations that need to be acknowledged. The early rise of inflammation and infection markers on day 1 in patients with subsequent clinically diagnosed SAP might indicate that SAP already starts on day 1 after stroke onset and that beginning of SAP is not caused by but results in immunodepression as indicated by decreased mHLA-DR levels. However, the majority of patients were diagnosed with SAP on day 3 making it more likely that SAP is the consequence of and not the reason for SIDS. To further increase the causality between immunodepression and SAP, future studies on SAP should include very early assessment of immune markers within the first hours after stroke onset.
Patients with SAP and those receiving antibiotics (including patients with SAP, urinary tract infections, sepsis and ‘other infections’) show comparable alterations in biomarker levels. Stroke disturbs the normally well-balanced interplay between the nervous system and the immune system and thereby induces SIDS, mainly mediated by the autonomic nervous system. SIDS is characterized by a down-regulation of systemic cellular immune responses, i.e. rapid numerical decrease in peripheral blood lymphocyte subpopulations and functional shift or deactivation of lymphocyte subpopulations 11 as well as monocytes, lung macrophages and epithelial cells resulting in an increased susceptibility for SAP.8,10,28 However, the crosstalk between the CNS and the immune system in acute stroke does not only result in an increased susceptibility for SAP but also bacterial infections in other organs such as the urinary tract (UTI) or intestines. 6 Interestingly, patients receiving antibiotics due to UTI do not show significant alterations in mHLA-DR-, IL-6- and LBP-levels (data not shown). Similarities between biomarker levels in patients with SAP and those receiving antibiotics are mainly due to SAP and ‘other infections’. Recent experimental data suggests that autonomic changes after stroke also alter intestinal immune barriers29,30 and future clinical studies on SIDS should include colitis as post-stroke infection to assess if changes in gut microbiota also occur in humans.
Diagnosis of SAP was based on clinical judgement of the treating physicians. CDC criteria (developed for the diagnosis of hospital-acquired pneumonia) were assessed at each visit but did not need to be fulfilled for diagnosis of SAP. However, this might reflect clinical routine. Diagnostic approaches to SAP vary considerably. 31 A consensus development of operational diagnostic criteria for SAP was started only very recently. 32 In current practice, clinicians use in part CDC criteria, biomarkers such as CRP, stroke severity and dysphagia. Chest X-ray – the central diagnostic item in the CDC criteria of ‘clinically defined pneumonia’ – is assigned only minor importance. 33 Thus, the Pneumonia in Stroke Consensus Group recently recommended excluding typical chest X-ray changes for probable diagnosis of SAP. 32 Our approach in diagnosing SAP and dysphagia is in line with the recently published PASS and STROKE-INF trials.26,27 Moreover, rates of SAP and dysphagia are comparable with those of other studies in the field.1,34 Both, our rate of 40% of SAP patients undergoing microbiological testing as well as the high number of negative test results is reflecting the current clinical practice and in line with other studies.19,24,35 Cultures in SAP often remain negative without bacterial growth, 23 especially if specimens are obtained in the early phase after of post-stroke infections. 24 This might have several reasons. Among others, standard methods of bacterial testing might be insufficiently sensitive, thus pathogens may escape these investigations. Consequently, diagnostic standards such as the commonly used CDC criteria do not require the identification of a relevant pathogen. 17 With the available data of the PREDICT study, we aim to assess the clinical validity of the CDC criteria as well as the recently proposed criteria for SAP in a future publication by analyzing the clinical decision making in the diagnosis and treatment of SAP compared with documented CDC criteria.
Another limitation of this study is the lack of standardized criteria for the diagnosis of dysphagia. All our patients were treated on stroke units according to the European Stroke Organization (ESO) recommendations requiring routine screening for dysphagia for all admitted patients within 24 h after admission and to withhold oral feeding until the patient has demonstrated intact swallowing with small amounts of water and intact coughing on command. 36 FEES is considered as gold standard for dysphagia assessment with a higher sensitivity and specificity compared with clinical examination of swallowing. 37 However, current international stroke guidelines lack specific recommendations on the method of dysphagia screening and clinical dysphagia screening remains the standard in the majority of stroke units. This pragmatic approach does not only reflect clinical routine but is also used in many clinical stroke studies.38,39 Even the recent STROKE-INF trial that specifically assessed the effectiveness of antibiotic prophylaxis for reducing pneumonia in dysphagic stroke patients did not use FEES for dysphagia assessment. 26 Since FEES had and even has not been established as standard procedure for diagnosing dysphagia in most stroke units in general as well as in the participating study sites, we decided against using the gold standard and applied a pragmatic approach for the work up in dysphagia diagnostics.
To find implementation in clinical practice predictive markers should ideally entail therapeutic consequences. Preventive antibiotic treatment raised hope for a more effective prevention of SAP. Two recent large randomized-controlled trials investigated whether or not prophylactic antibiotic therapy prevents post-stroke infections and improves long-term outcome in acute stroke. In PASS, 27 preventive antibiotic therapy did not improve long-term outcome after stroke but reduced the overall infection rate. However, incidence of SAP was not significantly reduced. In STROKE-INF, 26 antibiotic prophylaxis also failed in reducing the frequency of SAP and did not improve 3-month outcome in dysphagic stroke patients. Despite these negative findings, PASS and STROKE-INF stimulate research to improve the pathophysiological understanding and diagnostic criteria of SAP as well as approaches to prevent and treat SAP. 40 For example, more sophisticated approaches to prevent SAP might be developed based on a refined SAP prediction using biomarkers and immune modulating strategies starting already in the hyperacute phase of stroke.
In conclusion, our study suggests that dysphagia together with stroke-induced immunodepression facilitates SAP. Early screening for both, dysphagia and immune markers allows for an identification of patients at high risk for SAP. Future clinical trials on SAP prevention might tailor patient selection on clinical and immunological parameters to assess the effects on SAP and outcome.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by German Research Foundation (Exc257), German Federal Ministry of Education and Research (01EO0801), European Community FP7 (201024) and Siemens Healthcare Diagnostics.
Acknowledgements
The authors gratefully acknowledge the support of the recruiting study sites and all PREDICT investigators: Department of Neurology, Charité – Universitätsmedizin Berlin, Germany (Campus Mitte, Campus Virchow Klinikum, Campus Steglitz): Andreas Meisel, MD; Sarah Hoffmann, MD; Gerhard Jan Jungehuelsing, MD; Lena Ulm, MD; Jos Göhler, MD; Thomas Krause, MD; Robert Steinicke, MD. Department of Neurology, St.-Josefs Krankenhaus Potsdam, Germany: Hendrik Harms,MD; Olaf Hoffmann, MD; Jens-Eric Röhl, MD. Department of Neurology, Vivantes Klinikum Neukölln, Berlin, Germany: Darius Günther Nabavi, MD; Marcella Hermans, MD; Olaf Crome, MD, Jens Offermann, MD. Department of Neurology, Vivantes Auguste Viktoria Klinikum, Berlin, Germany: Bruno-Marcel Mackert, MD; Frank Hamilton, MD; Nguyen, MD. Department of Neurology, Hospital Vall d’Hebron Barcelona, Spain: Joan Montaner, MD; Alejandro Bustamante, MD. Department of Neurology, Unfallkrankenhaus Berlin, Germany: Ingo Schmehl, MD, Lehmann, MD. Department of Neurology, Vivantes Klinikum Spandau, Berlin, Germany: Jörg Müller, MD; Thiem, MD. Department of Neurology, Klinikum Frankfurt Oder, Germany: Andreas Hartmann, MD. Department of Neurology, Vivantes Klinikum im Friedrichshain, Berlin, Germany: Hans-Christian Koennecke, MD. Department of Immunology, Labor Berlin - Charité Vivantes GmbH, Berlin, Germany: Christian Meisel, MD, Nadine Unterwalder, Uwe Kölsch, MD.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christian and Andreas Meisel have issued of a patent on anti-infective and immunomodulatory agents used for preventive antibacterial therapy after stroke (EP1480646). Peter U Heuschmann reports research grants from the German Ministry of Research and Education, European Union, Berlin Chamber of Physicians, German Parkinson Society, University Hospital Würzburg, Robert-Koch-Institute, Charité–Universitätsmedizin Berlin (within MonDAFIS; MonDAFIS is supported by an unrestricted research grant to the Charité from Bayer), University Göttingen (within FIND-AFrandomized; FIND-AFrandomized is supported by an unrestricted research grant to the University Göttingen from Boehringer-Ingelheim), and University Hospital Heidelberg (within RASUNOA-prime; RASUNOA-prime is supported by an unrestricted research grant to the University Hospital Heidelberg from Bayer, BMS, Boehringer-Ingelheim), outside submitted work. The other authors report no conflict of interest.
Authors’ contributions
S Hoffmann: Design and conceptualization of the study, acquisition of data, analysis and interpretation of data, drafting of manuscript, final approval of the manuscript version to be published. H Harms: Design and conceptualization of the study, acquisition of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. L Ulm: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. D Nabavi: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. BM Mackert: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. I Schmehl: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. GJ Jungehuelsing: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. J Montaner: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. A Bustamante: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. M Hermans: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. F Hamilton: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. J Göhler: Acquisition of data, interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. U Malzahn: Analysis and interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. C Malsch: Analysis and interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. PU Heuschmann: Design and conceptualization of the study, analysis and interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. C Meisel: Design and conceptualization of the study, analysis and interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published. A Meisel: Design and conceptualization of the study, analysis and interpretation of data, revision of manuscript for intellectual content, final approval of the manuscript version to be published.
