Abstract
The typical US diet has >30% calories from fat; yet, typical laboratory diets contain 17% calories from fat. This disparity could confound the clinical relevance of findings in cerebral ischemia models. We compared outcomes after neonatal brain injury in offspring of rat dams fed standard low-fat chow (17% fat calories) or a higher fat diet (34% fat calories) from day 7 of pregnancy. On postnatal day 7, hypoxic-ischemic injury was induced by right carotid ligation, followed by 60, 75 or 90 min 8% oxygen exposure. Sensorimotor function, brain damage, and serum and brain fatty acid content were compared 1 to 4 weeks later. All lesioned animals developed left forepaw placing deficits; scores were worse in the high-fat groups (p < 0.0001, ANOVA). Similarly, reductions in left forepaw grip strength were more pronounced in the high-fat groups. Severity of right hemisphere damage increased with hypoxia-ischemia duration but did not differ between diet groups. Serum and brain docosahexaenoic acid fatty acid fractions were lower in high-fat progeny (p < 0.05, ANOVA). We speculate that the high-fat diet disrupted docosahexaenoic acid-dependent recovery mechanisms. These findings have significant implications both for refinement of neonatal brain injury models and for understanding the impact of maternal diet on neonatal neuroplasticity.
Introduction
Brain injury is a major cause of neonatal mortality and chronic neurodevelopmental disabilities. A puzzling and incompletely understood clinical observation is that neonates who experience similar hypoxic-ischemic insults have widely varying neurodevelopmental outcomes. The metabolic milieu in which hypoxia-ischemia occurs may be a potent factor that influences the impact of injury in the brain, and thereby contributes to this variation in outcomes. In both fetal and early postnatal periods, inflammatory challenges may amplify the deleterious effects of hypoxia-ischemia1–3; however, much less is understood about the influence of maternal metabolic factors on susceptibility of the developing brain to hypoxic-ischemic injury.
Epidemiological evidence implicates maternal obesity during pregnancy as a risk factor for perinatal mortality and adverse neurodevelopmental outcomes.4,5 This could be due, in part, to the well-recognized link between obesity and chronic low-grade inflammation. 6 The pathophysiological effects of dietary fat composition and obesity are challenging to distinguish, both in human populations and experimentally. In this study, we sought to evaluate the impact of maternal dietary fat intake during pregnancy and lactation on outcomes after neonatal brain injury.
We utilized a well-characterized experimental model of neonatal hypoxic-ischemic brain injury, elicited by unilateral carotid artery ligation and subsequent timed exposure to 8% oxygen in seven-day old (P7) rats: this procedure results in quantifiable contralateral sensorimotor deficits and ipsilateral forebrain tissue damage.7–9 Although it is difficult to replicate the multifactorial complexity of clinical pathophysiology, strengths of this model include the high long-term survival rates of offspring, ability to titrate injury severity by varying the duration of hypoxia, and feasibility to incorporate functional and pathological outcome measures. A significant limitation of the neonatal rodent model that has not been previously considered is that experiments are typically performed in offspring of dams that are fed a grain-based diet that derives about 17% calories from fat. In contrast, US women of child-bearing age typically ingest a diet that contains roughly twice as much fat. 10
We sought to determine the impact of maternal dietary fat intake on the outcomes of neonatal brain injury. Our initial approach was to devise a high-fat (HF) rat diet that replicates the amount and distribution of dietary fatty acids consumed by US women of child-bearing age (34% calories from fat). 10 We evaluated the impact of this diet, administered to pregnant dams and continued postnatally, in comparison to a conventional, grain-based, relatively low-fat (LF) rodent diet, on outcomes after neonatal hypoxic-ischemic brain injury. This standard chow typifies the diet used in the majority of studies of cerebral hypoxia-ischemia in rodents. We found that a maternal HF diet during pregnancy and lactation resulted in worse functional outcomes after hypoxic-ischemic brain injury, and was associated with significant alterations in serum and brain fatty composition in their offspring.
Materials and methods
Diets
A modified AIN93G-based rodent HF diet was developed to match dietary fat intakes of US women aged 20–39, as reflected in the 2007–2008 National Health and Nutrition Examination Survey (NHANES).10,11 This diet was prepared by blending oils to achieve 40%, 22%, and 38% of fat intake as monounsaturated fatty acid (MUFA), polyunsaturated fatty acid (PUFA) including docosahexaenoic acid (DHA), and saturated fatty acids (SFA), respectively, an n-3:n-6 fatty acid ratio of 0.11, and a total fat content of 34% calories from fat (Dyets Inc., Bethlehem, PA). 10 Cornstarch was reduced to accommodate the higher fat content (see online Supplementary Table 1). The LF diet group was fed a standard grain-based chow with 17% of calories from fat (5008 Chow, LabDiet, St. Louis, MO, USA). Fatty acid content of samples of each diet was assayed in triplicate (see online Supplementary Table 1).
Animals
Animals were treated in accordance with protocols approved by the University of Michigan Committee on the Use and Care of Animals, which complies with all U.S. Federal regulations. Timed pregnant Wistar rats (purchased from Charles River Laboratories, Portage, MI) were allocated to LF grain-based chow or the HF diet on embryonic day 7 (E7) and were housed under SPF conditions with a 12 h light/dark cycle and ad libitum access to water and diet. Weight of maternal diet intake was recorded daily. Animal lesioning was performed on postnatal day 7 (P7). Experiments always included male and female pups in both diet groups. Experiments and this report comply with ARRIVE guidelines. 12
Physiological measures
Dams were weighed on E7, the day of birthing, P7 and P14. Offspring were weighed on P7, and then weekly up until the time of sacrifice. Body temperature was routinely measured pre-surgery, and at 15 and 120 min post-hypoxia-ischemia. Blood glucose was measured immediately before initiation of euthanasia from a drop of tail vein blood (Contour Blood Glucose Meter and Blood Glucose Test Strips, Bayer Health Care, Mishawaka, IN, USA). Total serum protein was determined in blood collected at the time of euthanasia (P14) in one HF and one LF litter by the biuret method using a commercial bench-top dry chemistry analyzer (VetTest 8008, IDEXX Laboratories, Inc. Westbrook, ME).
Hypoxic-ischemic lesioning
Isoflurane-anesthetized pups underwent right carotid artery ligation, as previously described.9,13 After a 90 min recovery period in a circulating air incubator (Hovabator, set at 36.5℃), they were exposed to 8% oxygen (for 60, 75, or 90 min) to titrate the severity of resulting right forebrain injury (temperature maintained at 36.5℃). They recovered in a temperature-controlled incubator (at 36.5℃, 15 min) and were returned to their dams after resumption of normal activity.
Vibrissae-stimulated forepaw placement testing
Bilateral forepaw placement testing, a quantifiable measure of sensorimotor function (10 trials/forepaw), was done at selected postnatal times. The normal complete response to unilateral vibrissae stimulation is full forepaw extension to contact the stimulus surface (score = 1); the impaired forepaw either does not move (score = 0) or extends partially (score = 0.5). All testing was video-recorded. In this injury model, forepaw scores ipsilateral to carotid ligation remain normal, contralateral forepaw deficits are common, and these deficits generally persist to P35. 9
Grip traction test
Measurement of forepaw grip strength using a grip strength meter (three measurements/forepaw; Digital Force Gauge DFIS-2, Chatillon Force Measurement Products, Largo, FL) was used as a complementary quantifiable indicator of forepaw sensorimotor function in older animals: P14 pups cannot be reliably tested with this method. This test measures both ability to sustain forepaw traction on a thin wire and the force exerted. In protocols that included the one-month outcome assessment, bilateral forepaw grip was measured on P21, P28, and P35. Normal animals have equal strength bilaterally, and grip strength increases from P21-P35. 9 Grip strength and left/right forepaw grip strength ratios were calculated. In this injury model, contralateral forepaw grip strength is reduced 2–4 weeks later. 9
Histopathology
Animals were euthanized, and brains were removed and frozen on P14 or on P35. Coronal 20-mm sections were stained with cresyl-violet. Using ImageJ software (US National Institutes of Health, Bethesda, MD, http://rsb.info.nih.gov/ij/), bilateral volumes were calculated from hemisphere and regional area measurements made in regularly spaced sections from the level of the anterior genu through to the posterior genu of the corpus callosum, by an observer blinded to treatment identity. In this model, hypoxic-ischemic lesioning at P7 results in right cerebral hemisphere tissue loss by P14; the duration of hypoxia exposure strongly influences the magnitude of tissue damage. Examples of histopathology representative of the different hypoxia-ischemia durations used in Protocol 1 are presented in Figure 1, panels B through G. As a measure of right cerebral hemisphere damage, ratios of right (R)/left (L) hemisphere volumes were calculated.
Study overview and representative histopathology: (a) Experimental protocols including diet allocations, hypoxia-ischemia conditions, and outcome measures are summarized in this flow chart. (b–g) Representative cresyl-violet-stained coronal brain sections at the levels of striatum (b, c, d), and hippocampus (e, f, g) illustrate the typical extent of damage one week after hypoxia-ischemia (HI) of 60 (b, e), 75 (c, f), or 90 (d, g) min duration on postnatal day (P) 7. Scale bar = 1 mm.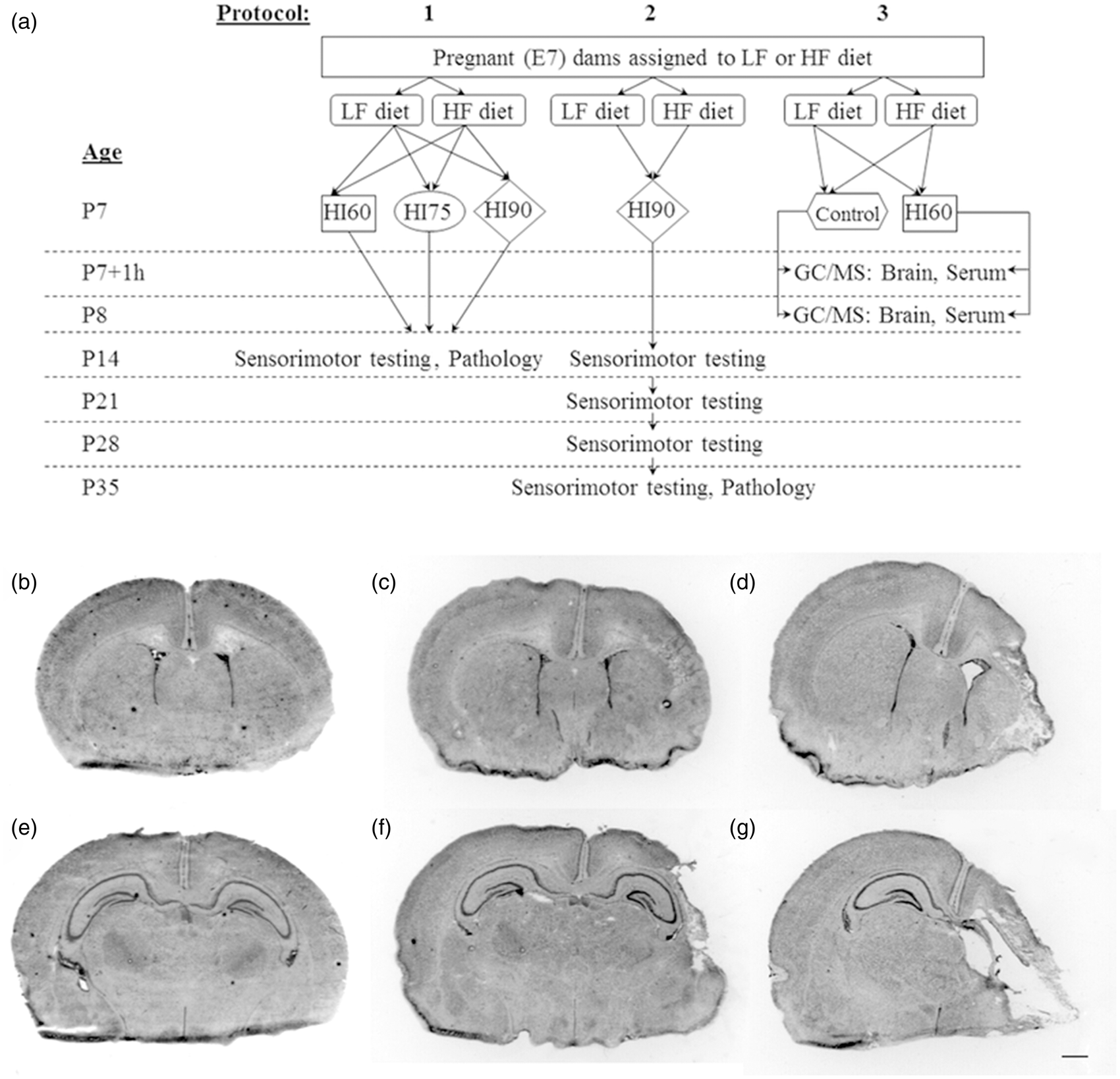
Fatty acid analysis
Brain tissue was removed from animals, frozen on dry ice and stored at −80℃ until analysis. Brain tissue was pulverized in liquid nitrogen, and 180 mg of pulverized tissue was mixed with 800 µl of ice-cold phosphate-buffered saline containing 1 mM EDTA and 0.1 mM indomethacin. The tissue was homogenized by vortexing followed by sonication in an ice-water bath (3 min). The homogenate was aliquoted to retain 150 µl for fatty acid analysis and 20 µl for protein analysis by the Bradford assay (Bio-Rad, Hercules, CA).
Folch reagent (chloroform: methanol 2:1, v/v) was used for extraction of brain homogenates, experimental diets, or sera. Fatty acid methyl esters were prepared using METH-PREP II derivatization reagent (Alltech, Deerfield, IL) at room temperature prior to analysis by gas chromatography with mass spectral detection using an Agilent 7890 GC and a 5975C mass-selective detector. Major fatty acids were quantified against standard curves using triheptadecanoin as the internal standard (Nucheck Prep, Elysian, MN) as previously described and expressed as % of total fatty acids.14–16 All fatty acid assays were performed blinded to treatment identity.
Study design
Typical experiments included four pregnant (E7) dams. Each experiment included dams allocated to both diet groups, and progeny from both groups were lesioned concurrently. The overall study design, with the intervention groups and testing procedures from three distinct protocols, is outlined in Figure 1, Panel A. Initial studies (Protocols 1 and 2) primarily evaluated post-hypoxic-ischemic functional outcomes and brain damage to P14 or P35, comparing diet groups. Subsequent studies (Protocol 3) compared serum and brain fatty acid composition on P7 and P8 in tissue samples from both hypoxic-ischemic lesioned and normal control progeny from both groups.
Protocol 1
Timed pregnant (E7) Wistar rats (n = 19) were allocated to standard LF grain-based chow (17% calories from fat, n = 7) or the HF diet (34% calories from fat, n = 12) for gestation and lactation (more dams were allocated to HF diet based on an initial experience of reduced fecundity). Pups underwent hypoxic-ischemic lesioning concurrently on P7. After a 90 min recovery period, pups were exposed to 8% oxygen (for 60, 75, or 90 min) (≥20 pups/time/group). On P14, sensorimotor function was scored, 9 and brain damage was evaluated by morphometry. 9 Blood was collected from dams on postpartum day 14 (tail vein puncture) and progeny on P14 (from neck veins), and serum was frozen at −80℃ for later fatty acid analysis on two representative pups (one male, one female) per litter. Serum glucose was measured from P14 blood samples on all pups from the 60 min hypoxia-ischemia experiments.
Protocol 2
To determine if there were more sustained differences in outcomes between diet groups, Protocol 1 was replicated with 90 min 8% oxygen exposure (n = 4 litters; 2 HF, 2 LF), and sensorimotor function was evaluated weekly up to P35 (n = 20–24/group). On P35, animals were sacrificed, serum was collected for fatty acid analysis and serum glucose measurement, and brains were frozen for histopathology evaluation. Animals remained on their designated diets after they were weaned on P22.
Protocol 3
To determine if the HF diet altered brain fatty acid composition in unlesioned and/or in hypoxic-ischemic animals, Protocol 1 was replicated with 60 min HI (n = 10 litters; 5 LF, 5 HF); non-lesioned littermates were included in each experiment. Right (lesioned) hemisphere tissue was collected 1 h and 24 h after hypoxia-ischemia, left hemisphere samples were collected from normal control P7 and P8 littermates, and left hemisphere samples were also collected from lesioned animals 24 h after hypoxia-ischemia. Serum samples were collected from all P7 and P8 animals in protocol 3. Brain tissue and sera were frozen at −80℃ and assayed in multiple independent batches from each time point for fatty acids.
Statistics
Minimum sample sizes for assessment of functional and pathology outcomes (≥16/group) were selected based on power calculations with data from prior similar experiments. Performance measures on sensorimotor testing were compared using ANOVA or repeated measures ANOVA. Post hoc comparisons of diet group means were evaluated with Tukey Kramer t-tests, and post hoc comparisons of left- vs. right-sided grip strength were evaluated with Sidak multiple comparison tests. A linear mixed models analysis of variance (ANOVA) was applied to evaluate differences in brain damage. Serum fatty acid percentages for dam and P14 progeny serum were analyzed by ANOVA factoring diet and assay batch. P8 or P35 serum samples were assayed concurrently for each age group and compared between diet groups by t-tests. Brain fatty acid values were analyzed by two-way ANCOVA with diet and lesioning status as the main factors and controlling for assay batch. The relationship between serum and brain DHA fractions was evaluated in samples from P8 hypoxic-ischemic and control animals using general linear models, with brain DHA as the outcome and serum and diet as independent variables. Differences in serial body temperatures and weights were assessed by repeated measures ANOVA, factoring diet and sex. A p value of <0.05 was considered significant.
Results
Diet fatty acid content
The measured fatty acid constituents of each diet are presented in online Supplementary Table 1. The measured DHA content of the HF diet and the LF chow was 103 and 145 µg/g of diet, respectively. The measured linoleic acid (18:2n6) content of the HF diet was about nine-fold higher than in the LF diet and the α-linolenic acid (18:3n3) content of the HF diet was about 11-fold higher than in the LF diet.
Survival, growth and physiologic measures
Maternal characteristics.
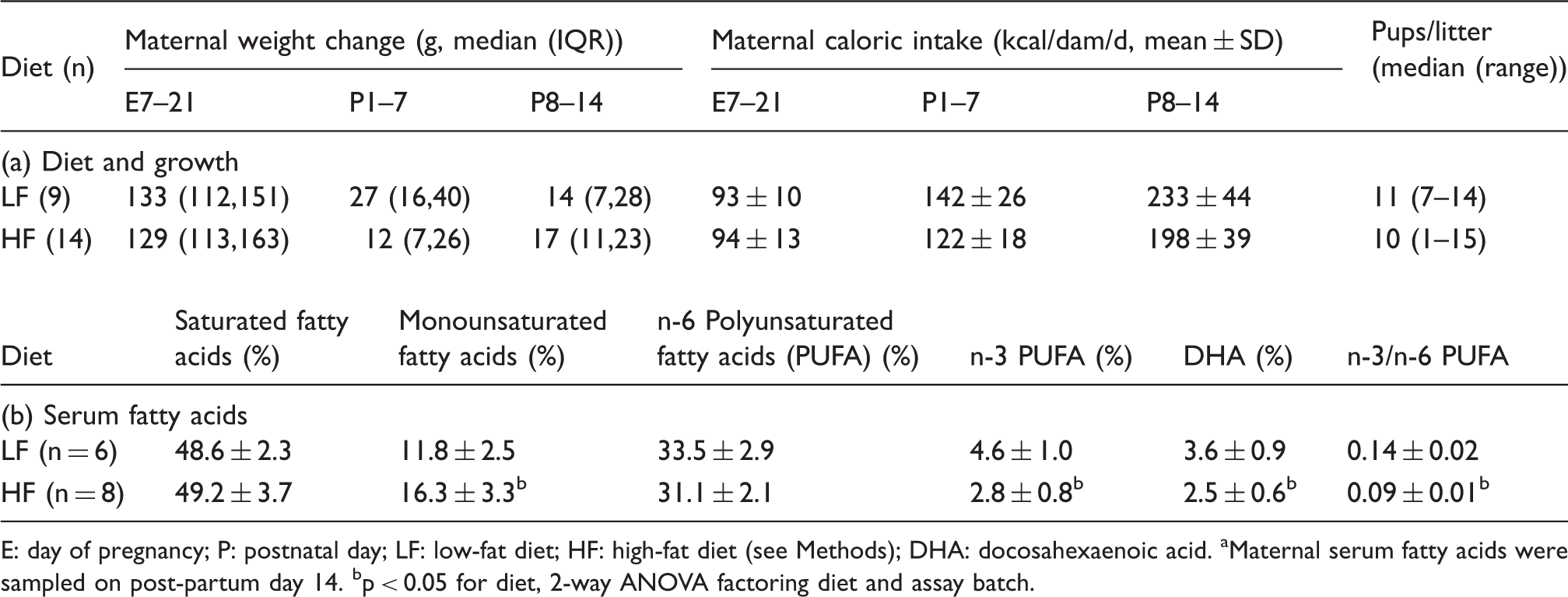
E: day of pregnancy; P: postnatal day; LF: low-fat diet; HF: high-fat diet (see Methods); DHA: docosahexaenoic acid.
Maternal serum fatty acids were sampled on post-partum day 14.
p < 0.05 for diet, 2-way ANOVA factoring diet and assay batch.
Physiologic measures in progeny.
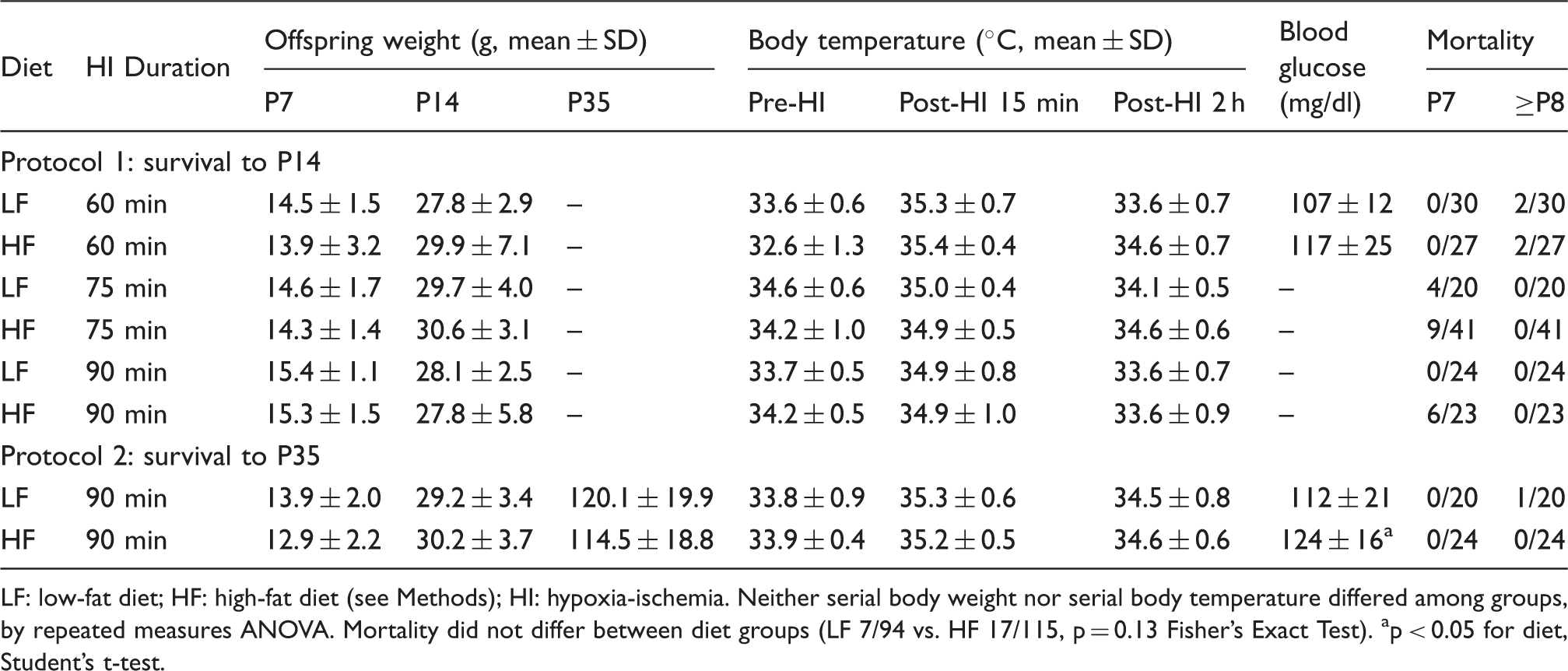
LF: low-fat diet; HF: high-fat diet (see Methods); HI: hypoxia-ischemia.
Neither serial body weight nor serial body temperature differed among groups, by repeated measures ANOVA.
Mortality did not differ between diet groups (LF 7/94 vs. HF 17/115, p = 0.13 Fisher’s Exact Test).
p < 0.05 for diet, Student’s t-test.
Protocol 1: Functional and pathology outcomes on P14
Sensorimotor function
Right forepaw placing scores were normal (10/10) in all animals (not shown). Among LF diet group pups, left forepaw median scores were close to normal after 60 min hypoxia-ischemia and were lower with longer duration hypoxia-ischemia (p < 0.0001 ANOVA, Figure 2(a)). With all three duration periods of hypoxia-ischemia, left forepaw function was substantially worse in the HF than the LF diet animals (p < 0.0001, ANOVA with Sidak’s post hoc test, Figure 2(a)).
Comparison of sensorimotor deficits and damage severity on postnatal day 14 (P14). Pups from LF (open bars) and high-fat (HF) diet (striped bars) groups underwent right carotid ligation, followed by 8% O2 exposure, for 60, 75, or 90 min on P7. Boxes extend from the 25th to 75th percentile, horizontal lines within each box are medians, and whiskers represent the minimum and maximum values. (a). Forepaw placement was scored on P14 (normal 10/10). Right forepaw scores were normal in all (not shown). Left forepaw placing deficits were more severe with increasing HI duration (*p < 0.0001 ANOVA), and deficits were more pronounced in all high-fat groups (†p < 0.0001, ANOVA with Sidak’s post hoc test). (b). Brain damage was quantified morphometrically and expressed as ratio of right/left (R/L) hemisphere volumes. In both diet groups, there was increasing brain damage (decreasing R/L ratios) with increasing duration of HI (*p < 0.0001, ANOVA): for each HI duration, brain damage did not differ between diet groups.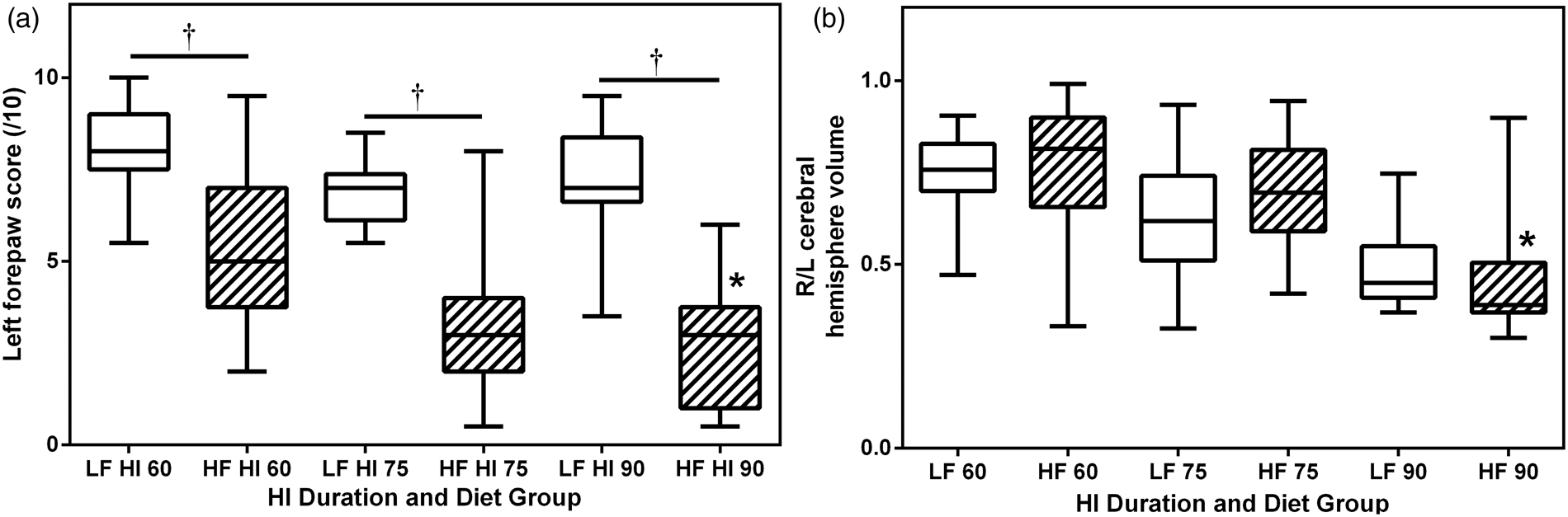
Neuropathology
Right (R)/left (L) cerebral hemisphere volume ratios were calculated, as measures of forebrain damage, and compared (Figure 2(b)). Damage was more severe (lower R/L ratio) with longer hypoxia-ischemia duration (p < 0.0001, ANOVA) but did not differ between diet groups at any of the three hypoxia-ischemia durations. Comparison of regional volume measurements confirmed these trends (online Supplementary data Table 2).
Protocol 2: Functional and pathology outcomes up to P35
Forepaw placing was tested weekly from P14 through P35. Right (R) forepaw scores were all normal (not shown). In the HF diet group, left (L) forepaw placing was persistently impaired (p < 0.0001 repeated-measures ANOVA, Figure 3(a)). Similarly, measurement of bilateral grip strength from P21 to P35 revealed that left forepaw grip weakness was more pronounced in the HF group (e.g. P35 L/R grip ratio, mean ± SD: LF 0.88 ± 0.17, vs. HF 0.47 ± 0.16; p < 0.0001 repeated-measures ANOVA, Figure 3(c)). An additional analysis of grip strength, independently in the two diet groups indicated that there was also a significant left–right strength difference (p < 0.05, ANOVA) at P35 in the LF group (14% difference) vs. 54% left–right difference in the HF group. Right hemisphere damage severity did not differ on P35 (R/L hemisphere volume ratio, mean ± SD for LF 0.51 ± 0.24 vs. HF 0.66 ± 0.28).
Comparison of sensorimotor deficits persisting to postnatal day 35 (P35). Pups from low-fat (LF) (open bars) and high-fat (HF) diet (striped bars) groups underwent right carotid ligation, followed by 90 min 8% O2 exposure, on P7. (a) Forepaw placement was scored weekly from P14–P35 (normal 10/10). Right (R) forepaw scores were all normal (not shown). Left (L) forepaw deficits remained more pronounced in the high-fat group (*: p < 0.0001 repeated measures ANOVA; boxes and whiskers are defined as in Figure 2). (b) Bilateral grip strength (3 trials/forepaw) and L/R grip strength ratios, (c), from P21–P35. Strength increased over time in all animals (b). Left forepaw weakness was more pronounced in the high-fat group at all ages (b and c, *: p < 0.0001 from repeated measures ANOVA; error bars represent standard deviations). Post hoc Sidak multiple comparison testing revealed a significant left–right forepaw grip difference at P35 in the LF group (14%) vs. 54% left–right difference in the HF group.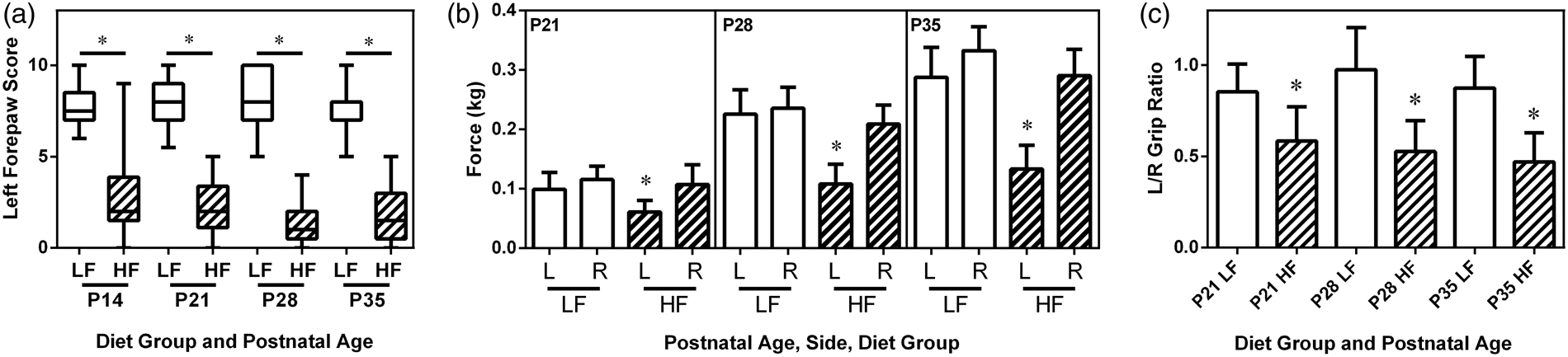
Fatty acid composition
Maternal diet influenced blood and brain fatty acid composition in both dams and their progeny.
Serum total fatty acids
Influence of maternal and post-weaning diet on serum fatty acid analysis in progeny on P14 and P35.
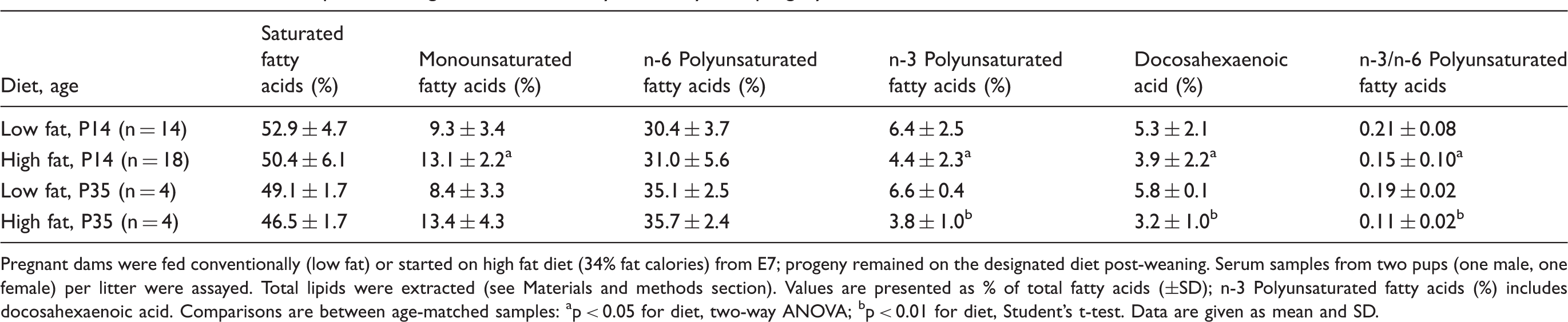
Pregnant dams were fed conventionally (low fat) or started on high fat diet (34% fat calories) from E7; progeny remained on the designated diet post-weaning.
Serum samples from two pups (one male, one female) per litter were assayed. Total lipids were extracted (see Materials and methods section). Values are presented as % of total fatty acids (±SD); n-3 Polyunsaturated fatty acids (%) includes docosahexaenoic acid.
Comparisons are between age-matched samples: ap < 0.05 for diet, two-way ANOVA; bp < 0.01 for diet, Student’s t-test.
Data are given as mean and SD.
Brain total fatty acids
Influence of maternal dietary fat intake on brain fatty acid composition in progeny.
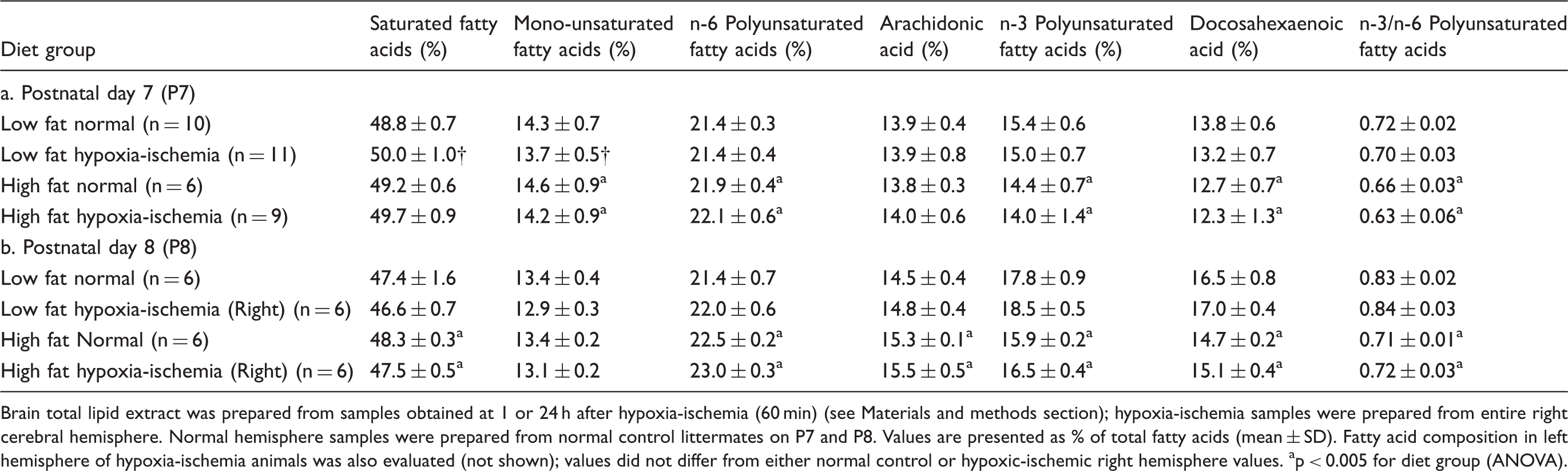
Brain total lipid extract was prepared from samples obtained at 1 or 24 h after hypoxia-ischemia (60 min) (see Materials and methods section); hypoxia-ischemia samples were prepared from entire right cerebral hemisphere. Normal hemisphere samples were prepared from normal control littermates on P7 and P8. Values are presented as % of total fatty acids (mean ± SD). Fatty acid composition in left hemisphere of hypoxia-ischemia animals was also evaluated (not shown); values did not differ from either normal control or hypoxic-ischemic right hemisphere values.
p < 0.005 for diet group (ANOVA).
In both diet groups, DHA made up a much higher percentage of the total fatty acid content in brain than in serum (e.g. P8, mean% ± SD: brain 15.8 ± 1.0 vs. serum 5.4 ± 1.9, p < 0.0001 paired t-test). Combining data from all P8 samples in both diet groups to evaluate the brain–serum DHA relationship, the slope was significant (standardized beta coefficient = 0.25 (95% CI = 0.02–0.48), p = 0.036), and the relationship between brain and serum DHA % values did not differ between diet groups (p = 0.56).
Discussion
Our results demonstrate that a modified AIN93G HF diet during pregnancy and lactation impairs recovery after neonatal cerebral hypoxic-ischemic injury, when compared to a conventional LF diet, in a rodent model. This finding is congruent with recent reports that a HF diet impairs functional outcome after cerebral ischemia in adult rodents17,18 and after traumatic brain injury in juvenile rodents. 19 With the maternal HF diet, progeny had more severe and persistent sensorimotor deficits than conventionally fed controls, although there was no difference in the severity of overt brain damage.
The HF diet resulted in persistent and congruent changes in maternal and neonatal serum fatty acid composition with a reduced n-3/n-6 fatty acid ratio and depletion of DHA, an n-3 polyunsaturated fatty acid (n-3 PUFA) with neurotrophic and anti-inflammatory actions. 20 Moreover, we found congruent alterations in brain phospholipid fatty acids of these progeny including reductions in the n-3/n-6 ratio and depletion of DHA. These findings are consistent with a recent report that a maternal diet with similar total fat content but a higher n-6/n-3 ratio, when continued into adulthood in the progeny, decreased progeny brain DHA content. 21 Our findings are also consistent with published data that feeding adult rats an HF diet depletes brain DHA content, reduces brain n-3/n-6 fatty acid ratio and impairs multiple plasticity mechanisms, including brain-derived neurotrophic factor expression. 22
In our study, diet affected the functional endpoint to a greater extent than the neuroanatomic endpoint. In neonatal brain injury models, there are several precedents for disparate effects of therapeutic interventions on functional and pathologic outcomes. These include cannabinoid, 23 vascular endothelial growth factor receptor antagonist, 24 erythropoietin, 25 and inhaled xenon with delayed hypothermia. 26 The relatively greater benefits of the LF diet on sensorimotor recovery rather than reduction in tissue damage suggest that a pivotal mechanism underlying the deleterious effects of the HF diet could be attenuation of neurotrophic signaling.20,22,27–29 An intriguing question for future study in this model is whether neurotrophin signaling pathways in the immature brain are modifiable by dietary fat manipulations and/or DHA supplementation. A diet high in fat with a high n-6/n-3 ratio decreased evidence of hippocampal neurogenesis in developing rats that did not undergo brain injury. 21 Thus, another question for future study is whether dietary fat intake could modulate injury-induced neurogenesis or oligodendrogliogenesis, for example in the subventricular zone and nearby striatum or sensorimotor cortex.30–32
Therapeutic supplementation with DHA, using acute systemic therapy or dietary manipulations, is neuroprotective in neonatal and adult rodent cerebral ischemia,13,33–35 and adult traumatic brain injury models. 36 These studies were all conducted in conventionally fed (i.e. LF diet) animals. Of interest, in the same neonatal model as was used in this study, post-hypoxia-ischemia treatment with a single injection of DHA (2.5 mg/kg) improved sensorimotor function without attenuation of brain damage. 13 The effects of this acute DHA treatment on blood and brain DHA content were not measured.
The present results support the hypothesis that the deleterious effects of the HF diet are mediated in part by DHA depletion in brain and/or blood. However, reduced preformed DHA in the diet may not be the sole responsible factor. The HF diet, designed to match American dietary intake, incorporated only a modest (29%) reduction in preformed DHA relative to the LF chow, and the DHA precursor α-linolenic acid was present at an 11-fold higher level in the HF diet. Other aspects of the fatty acid composition might also contribute to the HF diet’s deleterious effects. For example, the HF diet contains more than 10 times as much of the n-6 PUFA linoleic acid as of the n-3 PUFA α-linolenic acid, a metabolic precursor of DHA. Since both linoleic and α-linolenic acid compete for the same metabolic pathways, 37 excess dietary linoleic acid could limit production of DHA in liver, the main site of endogenous DHA synthesis. 38 Linoleic acid is also a substrate for the 15-lipoxygenase that is the first step in enzymatic conversion of DHA to protectin-D1 and related protectins and resolvins. 39 Thus, the high-linoleic acid content might competitively restrict production of beneficial neurotrophic or anti-inflammatory DHA metabolites.
We also evaluated the effects of hypoxia-ischemia on brain phospholipid fatty acid composition. Fatty acids with increasing numbers of double bonds are chemically less stable, and thus might be selectively degraded in this injury model. 40 Hypoxia-ischemia alone did not affect brain fatty acid composition in total brain lipids at either 1 h or 24 h after hypoxia-ischemia. These results confirm and amplify a previous report of brain fatty acid composition from 1 to 7 days after hypoxia-ischemia. 41
In randomized trials done at our institution and others, n-3 fatty acid supplementation resulted in slightly longer gestation and increased birth weight of infants.42,43 There has been substantial enthusiasm for DHA supplementation to pregnant women and their infants; however, inconsistent clinical benefits have been reported.44–48 The present results suggest that variation in dietary fat intake is one factor that could influence the impact of exogenous DHA and account for these disparate trends.
Other nutritional factors could also influence susceptibility to neonatal hypoxic-ischemic brain injury. In this model, other studies have indicated that heavier P7 pups develop more severe hypoxic-ischemic brain damage,49,50 but in our study weight did not differ between diet groups, either at the time of hypoxia-ischemia or later. We also considered protein adequacy, since protein malnutrition during pregnancy and lactation results in impaired sensorimotor function after neonatal cerebral hypoxia ischemia. 51 In both groups of pups, weight gain was the same (Table 2), as was serum protein. Although blood glucose was higher in HF progeny at P35, this was not the case at P14; moreover, hyperglycemia reportedly does not increase the severity of hypoxic-ischemic damage in the neonatal rodent brain.52,53 Finally, natural cereal grains used in conventional chows are a source of flavonoids that could reduce reperfusion inflammation.54–56
Limitations to the study design other than the content of the two diets should be considered. The present design does not address the impact of high dietary fat intake (or obesity) either before conception or in early pregnancy. Our maternal and progeny weight data (Tables 1 and 2) indicate that this design did not model maternal obesity or its common neonatal correlate of large for gestational age progeny. Inflammatory mechanisms contribute to many consequences of obesity 6 and potentiate HI injury. 2 Whether the HF diet is pro-inflammatory and could thus amplify neonatal cerebral hypoxic-ischemic damage is uncertain. However, if the HF diet amplified inflammation, more severe tissue damage, in addition to worse functional outcomes, would have been expected. 1 The present design also does not address whether dietary fat manipulations modulate injury-induced effects on specific neuronal subpopulations, e.g. striatal interneurons 57 or on oligodendrocytes. 58 In unlesioned rodents, a maternal HF diet and obesity have been shown to adversely affect cognitive development.59,60 In this study, the focus was on evaluation of sensorimotor function that is readily quantifiable and can be examined repeatedly to evaluate the trajectory of recovery.9,61 Evaluation of hypoxia-ischemia-induced cognitive impairments is an important question for future studies.
Conclusion
Maternal diet pre- and postnatally influenced functional recovery after neonatal hypoxic-ischemic brain injury. The deleterious effects of a higher maternal dietary fat intake using a purified diet were associated with depletion of serum and brain DHA in the offspring. These findings highlight the need to examine the impact of the diet during pregnancy and lactation on recovery after neonatal brain injury both in experimental models and clinically.
Availability of data and materials
The underlying research materials (data) related to the paper and samples of the diet can be accessed by written request to the corresponding author.
Footnotes
Funding
This work was funded by: U.S. National Institutes of Health R21 AT006636; U.S. National Institutes of Health R21 HD073692; Charles Woodson Acceleration Award from the Department of Pediatrics, University of Michigan.
Acknowledgments
The authors thank Nahid Keshavarzi M.S. and Nicola Kaciroti Ph.D. for providing statistics support through the Charles Woodson University of Michigan Department of Pediatrics Biostatistics Consultation Program. DHASCO Oil used in preparation of the high-fat diet was a gift from Martek Biosciences which played no role in the scientific conduct of experiments or in analysis or reporting of results.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
John Barks: Designed experiments, performed statistical analyses, interpreted data, drafted manuscript, final approval of manuscript. Yiqing Liu: Performed all animal experiments and histopathology preparation, review of manuscript, final approval of manuscript. Yu Shangguan: Performed all image analysis of histopathology, assisted with animal behavior scoring, review of manuscript, final approval of manuscript. Zora Djuric: Designed high-fat diet, supervised all fatty acid assays, assisted in drafting of the manuscript, final approval of manuscript. Jianwei Ren: Performed all fatty acid assays, review of manuscript, final approval of manuscript. Faye Silverstein: Designed experiments, assisted in drafting manuscript, final approval of manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
