Abstract
Magnetic resonance imaging employing administration of iron oxide-based contrast agents is widely used to visualize cellular and molecular processes in vivo. In this study, we investigated the ability of
Keywords
Introduction
Over the past several years, there has been an increasing interest and effort in developing magnetic resonance imaging (MRI) assays to visualize cellular and molecular processes in vivo. To this end, iron oxide particle-based contrast agents like superparamagnetic iron oxide (SPIO) or ultrasmall superparamagnetic iron oxide (USPIO) nanoparticles have been used.
1
Cellular MRI is performed either by intravenous injection of iron oxide nanoparticles, which are subsequently taken up by phagocytic cells, or by adoptively transferring cells, which have been exogenously labeled with the nanoparticles. Iron oxide particle-based contrast agents increase the transverse relaxation rate by an amount ΔR2,CA and alter the local magnetic susceptibility (ΔχCA), which enhances the reversible fraction to the transverse relaxation rate by ΔR2’,CA and thereby signal dephasing. Thus, the local effective transverse relaxation rate
Several MRI methods have been developed for quantification of iron oxide-based contrast agents. One common approach is relaxometry, i.e., the analysis of the magnitude signal decay to extract transverse relaxation rates R2,CA or effective transverse relaxation rates
Quantitative susceptibility mapping (QSM), a recently introduced post-processing technique for GRE phase information, promises to overcome this limitation. QSM converts phase images into maps of bulk magnetic susceptibility distribution, which can be analyzed directly.11–13 The benefit of QSM in quantifying the amount of iron oxide nanoparticles in biological tissue has recently been demonstrated for nanoparticles injected into skeletal muscle ex vivo, 14 as well as in vivo with nanoparticles that had been functionalized against the intercellular adhesion molecule-1 in a lipopolysaccharide-induced model of acute inflammation. 15
In the present work, we demonstrate the feasibility of QSM to visualize and quantify USPIO accumulation in the brain of arcAβ mice at 9.4 T. The arcAβ mouse strain is a genetic model of cerebral amyloidosis and exhibits progressive accumulation of amyloid-β (Aβ) protein in brain parenchyma and in the walls of cerebral vessels (cerebral amyloid angiopathy, CAA) with pronounced vascular remodeling.16–18 Hence, the strain is well suited to serve as a model of hereditary cerebral hemorrhage with amyloidosis,
19
and Alzheimer’s disease.
20
We evaluated the visibility of USPIO accumulation on quantitative susceptibility and
Materials and methods
Animals
All experimental procedures conformed to the national guidelines of the Swiss Federal Act on Animal Protection and were approved by an official committee (license 194/2011, Cantonal Veterinary Office, Zurich, Switzerland). We confirm compliance with the ARRIVE guidelines on reporting animal experiments. Transgenic (arcAβ) mice and non-transgenic littermates (NTL) of either sex were used. Animals were kept at standard housing conditions with a 12-h dark/light cycle and were provided with water and food ad libitum.
USPIO
P904 (Guerbet, France) is an USPIO and consists of an iron oxide core and a hydrophilic coating of a monomeric organic molecule with 20 hydroxylic groups, resulting in a mean hydrodynamic diameter of 21 nm. 21 P904 is cleared from the blood with a rate of 0.73 mL/min/kg. 22 As P904 is less prone to liver uptake compared to SPIO its blood half-life and thus its uptake capacity by macrophages is increased. 23 The P904 that is taken up by macrophages has been found to be degraded to non-toxic ferritin. 24
Magnetic resonance imaging
The study was performed using four batches of animals: The first batch consisted of six arcAβ mice and six NTL of 5.8–6.5 months of age and was assessed with MRI without USPIO injection. The second batch consisted of five arcAβ mice and six NTLs of 6–6.7 months of age and comprised animals from the first batch that were, after one week of recovery, assessed with MRI a second time 18 h after intravenous injection of P904 (1000 µmol/kg body weight). The third batch consisted of 11 arcAβ mice and 12 NTL of 9.6–12.2 months of age that were measured with MRI without USPIO injection. The fourth batch of mice consisted of 11 arcAβ mice and 7 NTL of 11.5–12.5 months of age which were assessed with MRI 18 h after intravenous injection of P904.
MRI data were acquired on a Bruker BioSpec 94/30 (Bruker BioSpin GmbH, Germany) small animal MR system operating at 9.4 T. The system was equipped with a cryogenic receive only 2 × 2 phased-array mouse head surface coil (Bruker BioSpin AG, Fällanden, Switzerland). A linearly polarized volume resonator was used for homogeneous transmission. Anesthesia was induced with 3 % isoflurane (Abbott, Cham, Switzerland) in a 4:1 air/oxygen mixture. The mice were endotracheally intubated and mechanically ventilated during the measurements with 80 breaths/minute and a respiration cycle of 25 % inhalation and 75 % exhalation (MRI-1 Volume Ventilator; CWI Inc., Ardmore, USA) using 1.2 % isoflurane. Body temperature was monitored with a rectal temperature probe (MLT415, ADInstruments) and kept at 36.0 ± 0.5℃ using a warm-water circuit integrated into the animal support (Bruker BioSpin). Anatomical reference data acquired in coronal and sagittal orientations served for accurate positioning of the animal’s head. Global first-order shimming followed by fieldmap-based local shimming was performed on the mouse brain using the automated MAPshim routine to reduce field inhomogeneities. A 3D multi-echo GRE sequence was applied using a field-of-view of 25.6 mm × 25.6 mm × 8 mm and an acquisition matrix of 256 × 256 × 80, resulting in an effectively isotropic spatial resolution of 100 µm × 100 µm × 100 µm. Four echoes were recorded (echo times (TE) = 3.2 ms/7.8 ms/12.4 ms/17 ms) with repetition time = 80 ms, flip angle = 15°, bandwidth = 78 kHz, monopolar echo readout, and no averaging within an acquisition time of ∼20 min.
The relaxivity (
Histology and immunohistology
Upon completion of MRI measurements, mice were transcardially perfused with phosphate-buffered saline (PBS, pH 7.4), followed by 4% buffered paraformaldehyde (PFA; pH 7.4) for fixation. Brains were removed from the skull and fixed for another 24 h, after which horizontal slices (approx. 2 mm) of the entire brain were prepared and embedded in paraffin wax. Sections (4 µm) were prepared and stained with hematoxylin-eosin (HE) for histopathological assessment and with Perls’ Prussian blue for the demonstration of iron (Fe3+). Briefly, slides were incubated in a 1% potassium ferrocyanide and 0.5% hydrochloric acid solution, followed by incubation with a 0.1% solution of nuclear fast red with aluminum sulfate for nuclear counterstaining. For diaminobencidine (DAB) enhancement of the Perls’ Prussian blue stain, slides were washed in TBS-Tween and incubated for 15 min with Peroxidase Blocking Solution (Dako), then washed with tap water and counterstained with hemalaun for 1 s. Demonstration of iron within macrophages/microglial cells was achieved by staining with Perls’ Prussian blue (blue), nuclear fast red, and immunohistology for Iba1. Consecutive sections were used for triple immunohistology, using rat anti-mouse CD31 (PECAM-1; clone SZ31, Dianova, Germany) for the demonstration of endothelial cells, rabbit anti-Iba1 (anti-peptide antibody, Wako Pure Chemicals) for the staining of microglial cells/macrophages, and mouse anti-beta amyloid (Aβ; clone 6E10, BioLegend) for the demonstration of Aβ deposition. Antigen retrieval was achieved by incubation in Retrieval-Buffer 2 (Leica AR9640) for 45 min at 100℃ (CD31) and for 10 min at 95℃ (Iba1). Sections were incubated for 30 min at room temperature with the primary antibodies diluted 1:200 (anti-CD31), 1:2,500 (anti-Iba1), and 1:5,000 (anti-Aβ) in Antibody Diluent (Leica AR9352) followed by incubation with rabbit anti-rat Ig (1:1,000, Abcam) and the Refine AP-Kit with new fast red as chromogen (Leica) for CD31, the Refine HRO kit and DAB as chromogen for Iba1, and the Refine HRP-kit (Leica) and permablue as chromogen (Diagnostic Biosystems) for Aβ.
MRI data processing
Single-channel GRE magnitude images were combined using the sum-of-squares method,
25
whereas single-channel GRE phase images were combined by taking the argument of the complex summed single-channel images after subtracting the channel-dependent phase offset estimated in the center of the three-dimensional volume of the first echo.
26
Quantitative susceptibility maps were computed based on these combined phase images. To this end, the combined phase images for each echo were unwrapped using a 3D best-path algorithm,
27
divided by
Maps of the effective transverse relaxation rate,
Data analysis
VOI analysis of
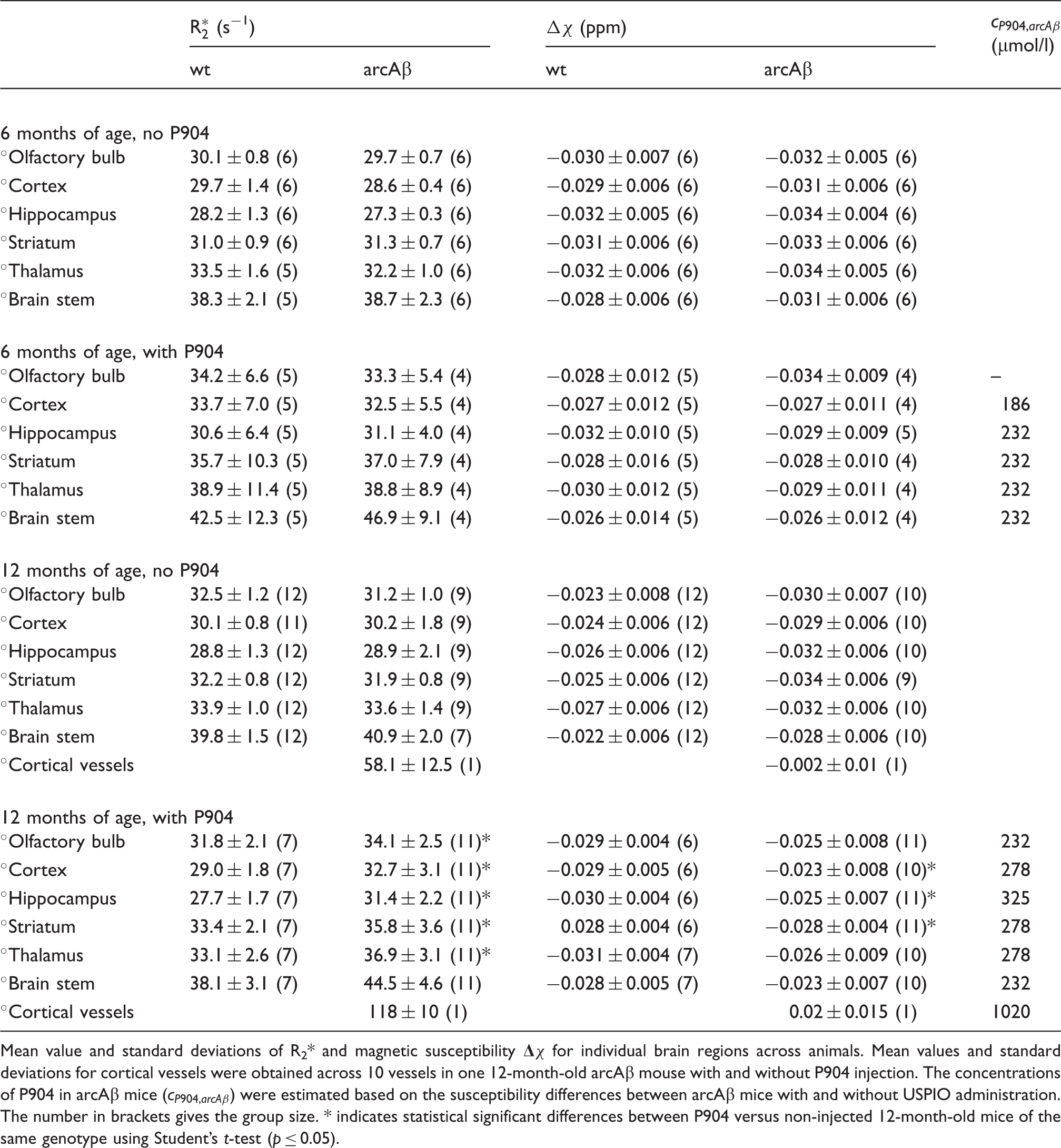
Mean value and standard deviations of R2* and magnetic susceptibility
Linear regression between P904 concentration (cP904) and the magnetic susceptibility difference with respect to the surrounding water and between cP904 and the
Results
GRE data of 6 - and 12-month-old NTL and arcAβ mice were acquired without and 18 h after intravenous injection of P904. Representative horizontal sections of (a) Representative horizontal sections of (a) Axial brain sections of susceptibility maps of a 12-month-old non-transgenic littermate (NTL) and an arcAβ mouse taken at different anatomical locations. The white dashed boxes indicate the anatomical region highlighted in the enlarged sections (b). (b) Enlarged images reveal areas of high susceptibility in the NTL animal, presumably veins, that contain a high amount of paramagnetic deoxyhemoglobin (black arrow). Focal areas of high susceptibility in cortical regions, the striatum, and hippocampus of the arcAβ mouse are indicative of USPIO particle accumulation in these regions (white arrows).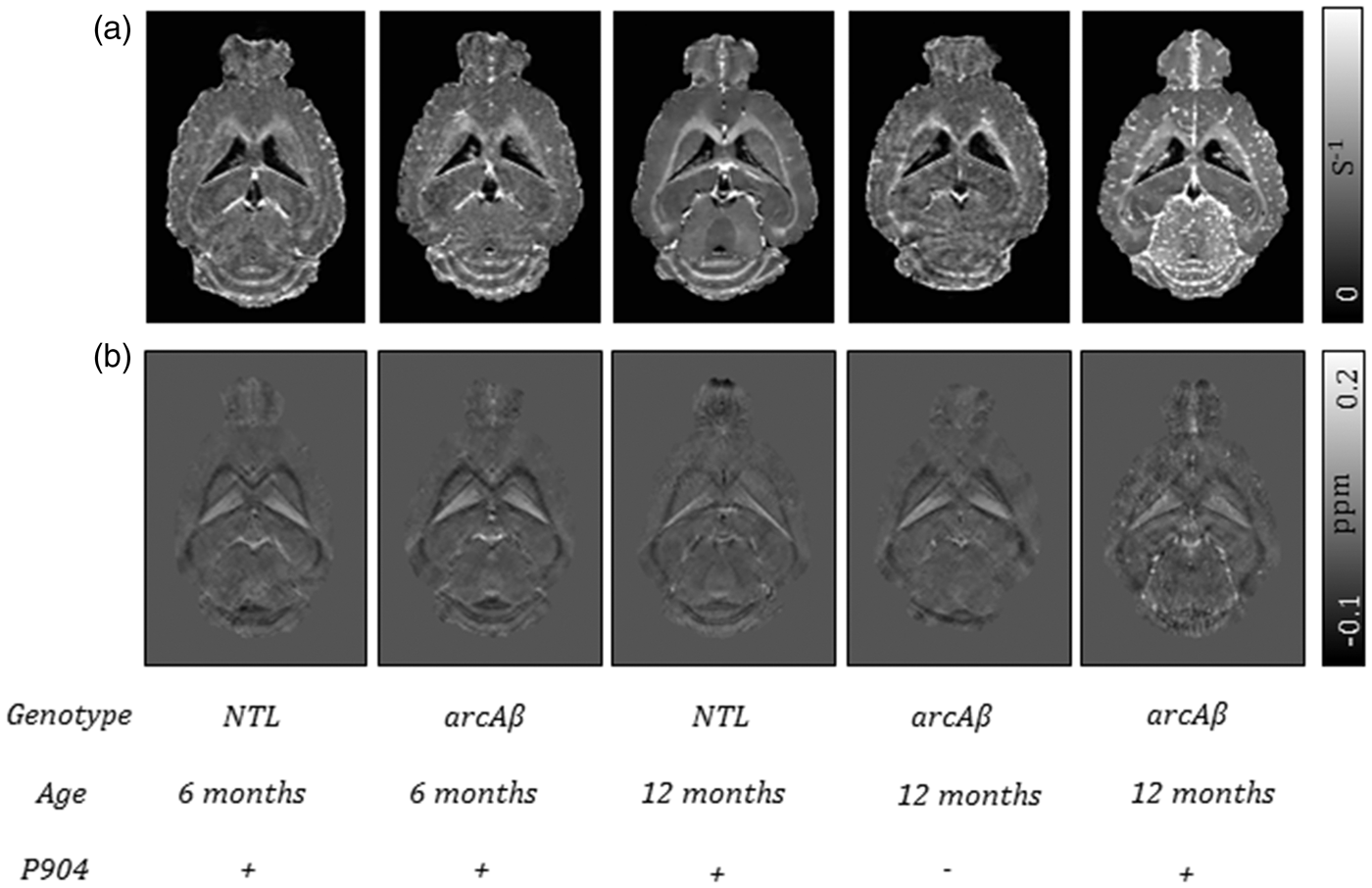

To obtain a quantitative estimate of USPIO accumulation, we performed VOI analysis of susceptibility and
Histological analysis was used to investigate vasculopathy and cellular uptake of USPIOs. This analysis did neither reveal pathological changes in the 6-month-old arcAβ mice nor in the NTL. However, in all 6-month-old arcAβ mice, small caliber vessels occasionally exhibited a variable degree of vascular Aβ deposition, ranging from very small granular over larger patchy to circular deposits, but showed no parenchymal Aβ deposition (Figure 3(a)). Cerebral vessels were not associated with the presence of activated perivascular macrophages and there was no evidence of iron uptake.
Histological findings in the cortex of arcAβ mice and non-transgenic littermates (NTL). (a) Twelve-month-old NTL. Small caliber vessels (CD31-positive endothelial cells: red) with Iba-1-positive (brown) perivascular macrophages (arrowhead) and Iba-1-positive (adjacent) inactive microglial cells (arrows). (b) Six-month-old arcAβ mouse. Small caliber vessels (CD31-positive endothelial cells: red) exhibit patchy Aβ deposition (blue, arrowheads). Staining for Iba-1 (brown) highlights the inactive microglial cells with their slender processes (short arrows) and the flatter perivascular macrophages (long arrow). (c) to (h) Twelve-month-old arcAβ mouse. (c) Small caliber vessels with CAA changes on a HE stain. One vessel exhibits homogenous eosinophilic thickening of the wall (*) due to Aβ deposition. On a more severely affected vessel, the eosinophilic material spreads out into the adjacent parenchyma (arrowheads), where activated microglial cells (arrows) are seen. (d) Triple staining of CD31, Aβ, and Iba1 confirms the deposition of Aβ (blue) in the vessel walls (CD31-positive endothelial cells: red), often circular over a stretch of a vessel (arrowheads), and with an occasional apposed activated microglial cell (arrows). (e) Closer view of affected vessels on the CD31/Aβ/Iba1 triple stain, with Aβ stretching from the vessel wall into the adjacent parenchyma (arrowheads) and closely apposed activated microglial cells (arrows). (f) Parenchymal Aβ plaque. Left: The Aβ plaque is represented on the HE stain by dense amorphous eosinophlic material (*) which is surrounded by cells with the morphology of activated microglia (arrows). Right: The CD31/Aβ/Iba1 triple stain reveals that the eosinophilic material represents Aβ (blue) and highlights the morphology of the associated activated microglial cells with Iba-1 (brown, arrows). (g) Focus with a few small caliber vessels with Perls’ Prussian blue positive, iron laden perivascular macrophages (arrows) and scattered iron laden macrophages in the parenchyma (arrowhead). Inset: Double stain for iron (Perls’ Prussian blue) and Iba-1 (brown) confirms the iron laden cells (arrows) as macrophages. Nuclear fast red counterstain. (h) In the DAB-enhanced Perls’ stain, the shape of the iron-laden perivascular macrophages (arrows; inset: arrowheads) is better defined, and some positive cell processes stretching into the parenchyma can be seen (arrowheads). a, b, d–f (right): Triple immunohistology for Aβ (blue), Iba-1 (brown), and CD31 (red). Bars = 10 µm.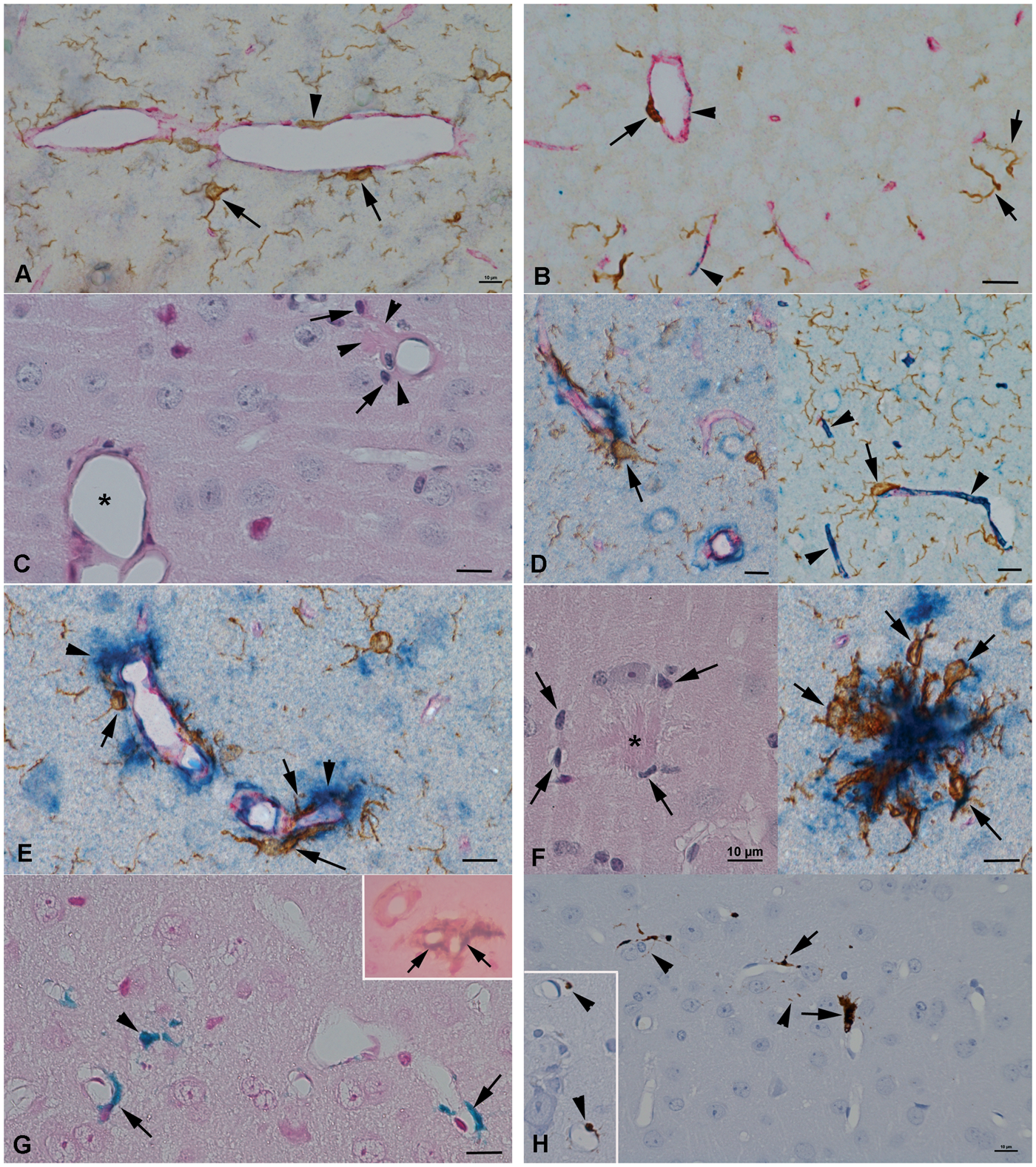
The brains of both USPIO injected and non-injected 12-month-old arcAβ mice revealed vascular changes consistent with CAA (Figure 3(c)), 17 together with multifocal parenchymal Aβ plaques with associated microgliosis (Figure 3(f)), which was most prominent in the cortex. Immunohistology identified a variable degree of vascular Aβ deposition in the walls of small caliber vessels, which was patchy or circular and often extended over a considerable length of the vessels (Figure 3(d) and (e)). Around more severely affected vessels, Aβ deposits stretched into the adjacent parenchyma, where one or occasionally several Iba1-positive cells with the morphology of activated microglia were found (Figure 3(d) and (e)). Immunohistology also confirmed the multifocal parenchymal deposits as dense Aβ aggregates which, as previously described, 34 were surrounded by or intermingled with Iba1-positive, activated microglial cells (see Figure 3(f)). In USPIO injected mice, a few small foci were identified, in which individual small caliber vessels (radius: 2–8°µm) exhibited one or a few perivascular elongated cells that contained iron-positive material, consistent with cellular uptake of USPIOs (Figure 3(g) and (h)). Combined Perls’ Prussian blue and immunohistological staining for Iba1 confirmed these cells as macrophages. However, their morphology was consistent with perivascular macrophages and not with activated microglia. 35 These vessels did not exhibit any histopathological changes, in particular there was no morphological evidence of CAA. In contrast, in CAA-affected vessels we did not detect iron-positive material. Rarely, individual iron-containing cells with the morphology of macrophages/microglial cells were also found in the parenchyma within these areas (Figure 3(g), arrows).
Discussion
GRE MRI techniques are highly sensitive to the underlying magnetic susceptibility distribution. Iron oxide-based contrast agents are used as label for cellular MR studies because they generate localized variations in magnetic susceptibility (Δχ) and R2 (or
Quantifying USPIO concentrations on the basis of
The effect of an injected iron oxide contrast agent on
We also found differences in the sensitivity between
Histological analysis was performed to demonstrate the cellular uptake of USPIOs. We found that USPIOs target to CAA affected vessels with radii between 2 and 8 µm meaning that P904 accumulates at the microvasculature but not at larger cerebral vessels which is in a good agreement with the literature.37,38 Although the actually measured radii of CAA affected vessels are lower than the radii of 10 to 40 µm reported previously which might be due to differences in the technical approaches, 18 the current study supports the finding that microvasculature is affected by the amyloid-induced vasculopathy.
In addition, we found that perivascular macrophages contained iron-positive material following injection of P904 in 12-month-old arcAβ mice whereas iron-containing cells in brain parenchyma were rarely seen (Figure 3(g) and (h), arrow heads). This confirms findings by Beckmann et al. 39 who detected iron oxide particles ingested by perivascular macrophages after intravenous injection in APP23 mice. Since, we have not seen morphological evidence of perivascular and parenchymal macrophage infiltration it is most likely that the residing perivascular macrophages have taken up P904 in situ.
Interestingly, Beckmann et al. 39 observed a mean number of 10 foci in the brain when assessing the 28-month-old APP23 mice with MRI, while we observed a much higher number of foci in arcAβ already at 12 months of age. This difference might be attributed to the different mouse strains but also to differences in the contrast agent. While Endorem, which was used in the study by Beckmann et al., 39 is a SPIO particle with a hydrodynamic diameter of 150 nm, the hydrodynamic diameter of USPIO P904 is only 21 nm. 21 Although the uptake of Endorem by phagocytes is higher compared to P904 under in vitro conditions, the labeling efficiency may be reduced in vivo due to the shorter blood half-life of Endorem. 23
Beckmann et al. 39 showed that iron oxide particle accumulation depends on the accumulation of vascular Aβ by comparing contrast agent accumulations in different mouse strains. While they observed only minor particle accumulation in the brains of PP23xPS45, APP24, and APP51 mice which show very little CAA, Endorem accumulated substantially in the brain of APP23 mice for which the degree of CAA was markedly higher. We have further analyzed the relationship between USPIO accumulation and CAA in the current study. In 6-month-old arcAβ mice, minimal vascular Aβ deposition was observed (Figure 3(b)), while in 12-month-old animals CAA was more pronounced (Figure 3(d) and (e)). We have previously shown that CAA in arcAβ mice results in vascular remodeling with deposition of fibrinogen, loss of smooth muscle cells, blood–brain barrier impairment, and occurrence of cerebral microbleeds.16–18 Surprisingly, we did not observe iron-positive perivascular macrophages around CAA-affected vessels but only around morphologically unaltered vessel without morphological evidence of CAA (Figure 3(g) and (h)). An explanation for this could be that in CAA-affected vessels, despite vascular remodeling, Aβ and fibrinogen deposition might physically prevent the USPIO from extravasating and thereby the uptake by tissue-resident macrophages. However, the fact that non-CAA-affected vessels show uptake of P904 indicates their increased permeability and thereby a functional vasculopathy. Using immunohistology, we observed activated microglia associated with vascular and parenchymal amyloid deposition. Activated microglia can change their microenvironment by secreting for example proangiogenic factors, 40 which in the vicinity of vessels might lead to blood–brain barrier impairment and thus extravasation of injected USPIO. We have found no evidence of Gd-DTPA leakage using dynamic contrast-enhanced MRI in arcAβ mice, 36 while blood–brain barrier damage has been described with histology in this mouse strain. 17 Hence, the USPIO-enhanced GRE MRI might provide a more sensitive read-out for vascular leakage in vivo than the dynamic contrast-enhanced MRI because of the cellular ingestion of particles by perivascular macrophages. The mechanisms of the functional impairment and their temporal dynamics are not known. The effect was clearly age-dependent as it was seen in 12-month-old but not in 6-month-old arcAβ mice. USPIO-enhanced GRE MRI provides a powerful non-invasive tool to study these vascular lesion loads during the disease course quantitatively, enabling a correlation with image-based read-outs of vascular dysfunction and thus shedding more light on the role of functional vasculopathy in the disease pathogenesis.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by the Swiss National Science Foundation (Grant PZ00P3_136822 to JK, Grant 310030-126029 to MR), the German Research Foundation (Grant RE-1123/9-2 to JR), a seed grant awarded to AD by the Interdisciplinary Center for Clinical Research (IZKF) in Jena, Germany, and seed grants awarded to FS by the International Society for Magnetic Resonance in Medicine (ISMRM) and the Friedrich Schiller University Jena.
Acknowledgements
The authors thank Guerbet (France) for providing the P904.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
JK and AD conceived and designed the experiments. GDI, DK, MV, and AK performed the experiments. JK, AD, and AK analyzed the data. JK, AD, AK, JRR, and MR interpreted results. AS, FS, and JRR, contributed reagents/materials/analysis tools. JK, AD, and AK wrote the paper. All coauthors made critical revisions to the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

