Abstract
Neurovascular coupling reflects the close temporal and regional linkage between neural activity and cerebral blood flow. Although providing mechanistic insight, our understanding of neurovascular coupling is largely limited to non-physiological ex vivo preparations and non-human models using sedatives/anesthetics with confounding cerebrovascular implications. Herein, with particular focus on humans, we review the present mechanistic understanding of neurovascular coupling and highlight current approaches to assess these responses and the application in health and disease. Moreover, we present new guidelines for standardizing the assessment of neurovascular coupling in humans. To improve the reliability of measurement and related interpretation, the utility of new automated software for neurovascular coupling is demonstrated, which provides the capacity for coalescing repetitive trials and time intervals into single contours and extracting numerous metrics (e.g., conductance and pulsatility, critical closing pressure, etc.) according to patterns of interest (e.g., peak/minimum response, time of response, etc.). This versatile software also permits the normalization of neurovascular coupling metrics to dynamic changes in arterial blood gases, potentially influencing the hyperemic response. It is hoped that these guidelines, combined with the newly developed and openly available software, will help to propel the understanding of neurovascular coupling in humans and also lead to improved clinical management of this critical physiological function.
Introduction
The human brain comprises only 2% of body weight, yet consumes more than 20% of oxygen and glucose at rest, with almost all adenosine triphosphate in the brain being produced by oxidative metabolism of glucose. 1 In addition to a great need for substrate provision and by-product clearance, the metabolic circumstances in the brain are compromised by a limited intra-cellular capacity for energy storage. These two characteristics, combined with the paramount importance of brain function in comparison with other end-organs, create a situation where coupling of blood flow requires careful matching to changes in neuronal activation. The brain is well equipped to provide suitable blood flow for a given metabolic demand due to two components: extremely high vascularization, as well as redundant and sophisticated regulation of blood flow. 2 One primary factor responsible for ensuring appropriate blood supply within the brain is neurovascular coupling, which describes a close temporal and regional linkage between neural activity and cerebral blood flow (CBF) responses. This manuscript will review the major issues related to neurovascular coupling including: the anatomic structure of cerebral vasculature, organ-level mechanisms regulating CBF; provide insight into the mechanisms underlying the neurovascular response; outline the various methods used to measure neurovascular coupling in humans; present new guidelines for standardizing the assessment of neurovascular coupling in humans; and highlight the related alterations in neurovascular coupling in clinical conditions. Furthermore, this manuscript will describe a new analytical process to enable reliable and consistent assessments of human neurovascular coupling and provide directions for future research into this exciting and fascinating line of inquiry.
Anatomic structure
The internal carotid and vertebral arteries (ICA, VA), which branch off of the subclavian arteries, are the two conduit vessels providing blood delivery to the brain. The internal carotid arteries transmit approximately 70% of total CBF, while the remainder is provided by the VAs. In healthy individuals under typical scenarios, the VAs primarily provide blood flow to the brainstem, cerebellum, and occipital cortex. The VAs converge to form the basilar artery which then serves as a tributary to the circle of Willis and emerge as the posterior cerebral arteries. The circle of Willis is a crucially redundant anastomotic safety net evolutionarily commissioned to maintain perfusion if blood flow if one or more extracranial cerebral arteries is disrupted. It also represents the terminus of the internal carotid, which then contributes to the middle cerebral artery (MCA). The anterior cerebral arteries branch off the anterior aspect of the circle of Willis. The anterior, middle, and posterior cerebral arteries branch extensively before wrapping the brain surface in an array of vascularized tissue within the pia mater. Pial arteries then penetrate into the cortex. Upon penetration, arterioles are located within the Virchow-Robin space (i.e., pia mater protrudes into the brain with the arteriole). Once the arteries completely penetrate the parenchyma, they are considered “parenchymal” and are completely enveloped by astrocytic end-feet. 3
Extracranial arteries (e.g. internal and vertebral arteries), large arteries of the brain (e.g. MCA), as well as pial arteries on the brain surface, are richly innervated by “extrinsic” perivascular postganglionic neurons from both sympathetic and parasympathetic origins.4,5 On the other hand, parenchymal arterioles are regulated by “intrinsic” factors within the parenchyma, which are mediated by neuronal activation and astrocytic modulation. 6 Parenchymal arteries have greater basal tone and do not respond effectively to neurotransmitters, including norepinephrine and serotonin, which do elicit changes in the tone of upstream pial arteries. 6 All arteries in the brain contain a specialized endothelial layer, which functions as a barrier in an attempt to tightly regulate exchange between the blood supply and the brain (i.e., blood-brain barrier). In addition to this, tight junctions prevent paracellular passage and connect the innermost layer of epithelial cells.
Regulation of cerebral blood flow
Regulation of CBF is achieved through several factors including metabolic, myogenic, neurogenic, and systemic control. 7 Specifically, the primary controllers of CBF are partial pressure of arterial blood gases (CO2 and O2), cerebral metabolism, and the autonomic nervous system.8,9 Under conditions of sufficient perfusion pressure, the remaining factors interact to regulate CBF by modulations in cerebrovascular resistance. 10 Cerebral blood flow is particularly sensitive to partial pressure of arterial CO2, as reflected in a 1-mm Hg increase or decrease from eupnoeic CO2 levels leading to a 3−6% elevation or a 1−3% reduction in CBF, respectively.
The large conduit arteries of the neck provide a large degree of influence in the regulation of CBF. Early canine in situ work showed that the arteries of the neck (i.e., ICA and VA) may provide as much as 50% of cerebrovascular resistance at rest.11–13 These large arteries actively change diameters in response to various stimuli including alterations in arterial blood gas partial pressures, 14 and orthostatic challenges. 15 Both extracranial and pial arteries are innervated by sympathetic neurons originating from the superior cervical ganglion, which may serve to regulate CBF through the release of norepinephrine and neuropeptide Y.4,16–19 Parasympathetic neurons from cranial nerves of the otic and pterygopalatine ganglia also modulate cerebrovascular tone by releasing acetylcholine, vasoactive intestinal peptide (VIP), and nitric oxide (NO), while the trigeminal ganglion fibres release a variety of other vasorelaxant mediators.4,20 This is in contrast to the vast majority of systemic vasculature where the parasympathetic nervous system does not actively regulate tone. Despite extensive autonomic innervation in the brain, findings in humans support only a modest and somewhat frequency-dependent role of sympathetic21–23 and parasympathetic activity 24 in influencing dynamic cerebral autoregulation. The details of the complex interactions involved in human CBF regulation have recently been reviewed. 9 The focus of this article is on the relatively unexplored topic of neurovascular coupling in humans. Herein, particular focus will be made on the physiology and pathophysiology of neurovascular coupling, methodological limitations, and advances in this field.
Mechanisms of neurovascular coupling
The neurovascular unit is made up of three major components: (1) the vascular smooth muscle, (2) the neuron, and (3) the astrocyte glial cell (Figure 1). At a rudimentary level of detail, modulations in neuronal activity cause changes in local blood flow mediated by transmission through the astrocyte. This interaction is termed “neurovascular coupling,” which describes coupling between neuronal activity and the vasculature. Tight temporal and amplitudinal linkage between neuronal activity and CBF delivery has been observed for >120 years.25–27 There is much nuance and redundancy in this simplistic explanation of the neurovascular unit’s interactions. For example, the neuron also alters vascular tone independently of astrocytic mediation, and these two cells play a synergistic or antagonistic role depending on the physiological environment. Readers are referred to recent reviews on this topic for further details.2,28,29
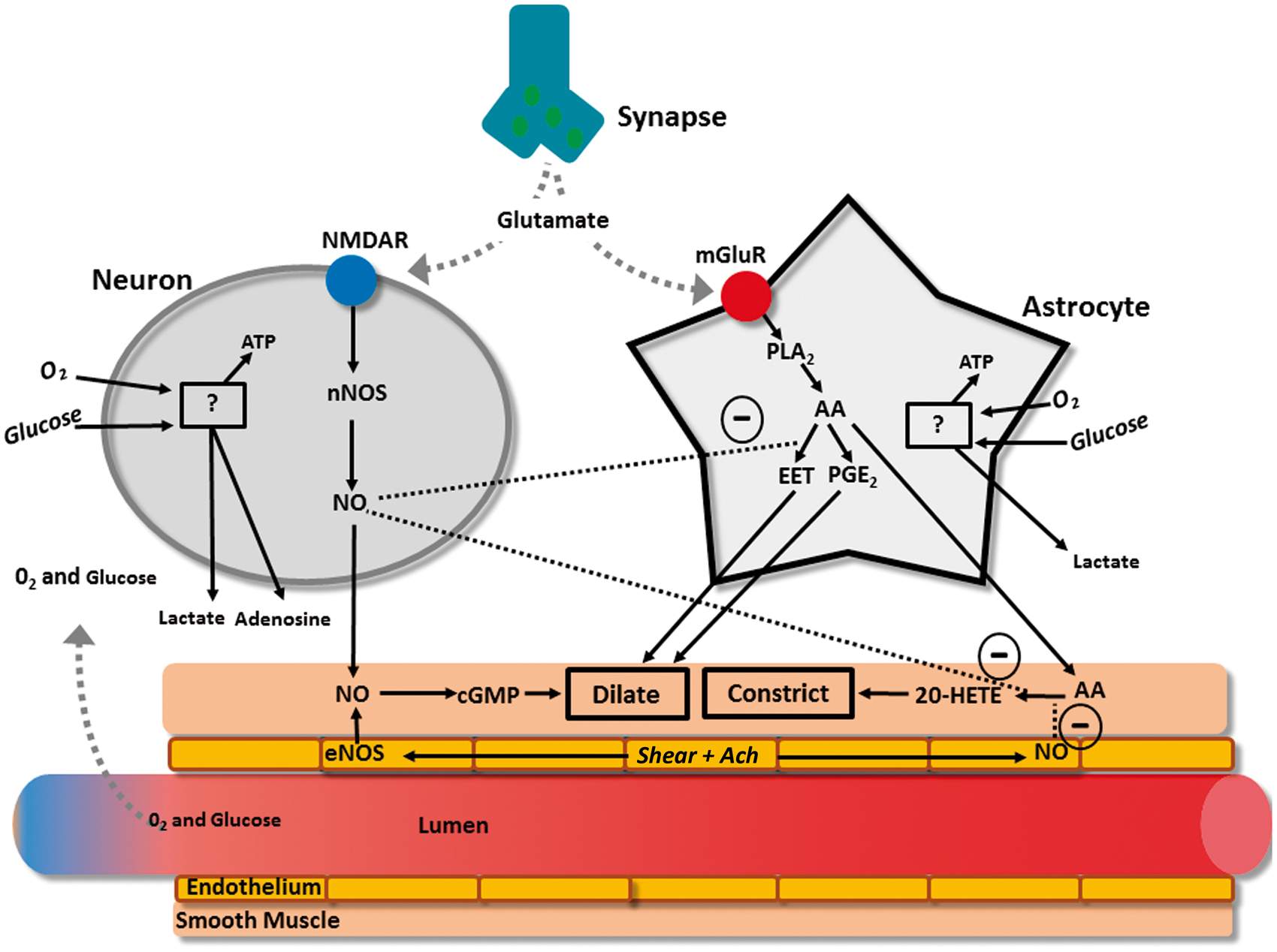
Both neurons and astrocytes respond to increased extracellular glutamate to transmit direct and indirect vasoactive signals for the appropriate delivery and distribution of CBF. 2 For example, glutamate released from presynaptic terminals activates N-methyl-D-aspartate receptors on neurons, which in the presence of sufficient intracellular oxygen and glucose, will stimulate the activation of neuronal NO synthase that provides NO to act directly on the parenchymal arterioles as a dilator. 30 Adenosine will also be directly released to act as a vasodilator when neuronal adenosine triphosphate is low. 31 Similarly to neurons, astrocytes respond to glutamate through metabotropic glutamate receptors, which again in the presence of sufficient intracellular oxygen and glucose, activate a cascading pathway involving the production of arachidonic acid which then produces epoxyeicosatrienoic acid and prostaglandins. 32 Both prostaglandin and EET serve to dilate parenchymal arterioles, while arachidonic acid, after diffusing out of the astrocytes and into the smooth muscle, produces vasoconstriction. 33 There also appears to be a complex relationship between astrocytes, incoming neuronal signals, and GABA interneurons that are thought to play a local integrating role precisely regulating local CBF delivery.29,34,35 These interneurons are ideally placed between glutaminergic pyramidal cells and project onto microvessels.35,36 Indeed, evoked firing of GABA interneurons resulted in increased concentration of a variety of vasoactive substances (e.g., NO, acetylcholine, neuropeptide-Y, VIP) that led to both constriction and dilation of cerebral microvasculature.34,35
Under normal conditions, the double stimuli (i.e., neuronal and astrocytic) leads to a 4-fold greater increase in CBF relative to the increase in ATP consumption. 37 This overcompensation of functional hyperemia appears to support the feed-forward mechanism. 37 It should be noted however that a feed-backward system capable of functional hyperemia has also been proposed as a compensatory mechanism in a model incorporating capillary transit time heterogeneity, maximum oxygen extraction fraction, metabolic rate of oxygen and CBF. 38 Together, these factors demonstrated that more homogenous capillary flow and exaggerated CBF response compensated for reduced oxygen extraction during functional hyperemia. 38 This overabundance of blood flow relative to metabolism is the theoretical foundation of blood-oxygen level dependent (BOLD) magnetic resonance imaging; which relies on the assumption that increases in deoxygenated blood are associated with increased neuronal activity. 39
Although astrocytes are sufficient to evoke a neurovascular coupling response, it is unclear whether astrocytes are necessary for functional hyperemia to occur.40,41 Recently, it has been proffered that the role of astrocytes during neurovascular coupling is isolated to maintaining blood flow suitable for basal neuronal activity and to producing a “slow-onset” hyperemia (i.e., 3−4 s post neuronal activation) but not to be active in the immediate hyperemic effect that occurs in <1 s. 41 Recent and sophisticated assessments of the neurovascular coupling response to visual stimulation showed that astrocytic endfeet/Ca2+ signalling was not responsible for the rapid functional hyperemia; 40 readers are referred to more in-depth reviews on this topic.28,42 This is a somewhat emerging but compelling contention, such that it deserves mention in the context of the present review.
It is not just the arteriole smooth muscle that regulates neurovascular coupling, although this has been the primary understanding until very recently. Pericytes (i.e., small cells expressing contractile tissue located at 50 µm intervals along the length of capillaries) 43 likely also play a large role in neurovascular regulation, as suggested by their anatomic arrangement. 44 For example, neurons are more often closer to pericytes than arterioles, creating the plausible scenario whereby neuronal activation first alters resistance through modulations in pericyte tone on capillaries, and signals are then transmitted ‘up-stream’ to arterioles. 45 Pericytes are theorized to be regulated in much the same way as arterioles. 44 Brain slice preparations show that glutamate and norepinephrine lead to dilation and constriction of pericytes, respectively, and blocking glutaminergic receptors leads to vasoconstriction. 46 The intervals between pericytes necessitate the transmission of vasoactive signals through gap junctions; however, whether these travel between pericytes/endothelial gaps remains to be elucidated.44,46 In any case, there is a huge capacity for the pericytes to influence neurovascular coupling. For example, it has been modelled that anywhere from 16−70% of resistance within the brain parenchyma is produced by capillaries.47,48 In reality this may be even larger, as capillaries are so narrow that white and red blood cells have to deform to pass through, creating even more resistance than Poiseuille's law predicts. 49
The combination of incongruent findings,40,50 the elucidation of highly redundant mechanistic pathways, 41 and limited translational capacity has led to the acknowledgment that the mechanisms of neurovascular coupling need to be better explored in vivo/human models rather than just in animals and isolated well-controlled preparations (e.g., two-photon imaging, brain slices, etc.).41,51 Establishing a standardized protocol for evaluating human neurovascular coupling in vivo would allow for insight into not only the mechanisms underlying this fundamental response but also how neurovascular coupling is altered in diverse populations (e.g., clinical populations, aging, etc.).
Human neurovascular coupling
Our present understanding of neurovascular coupling in humans is limited, and the available knowledge is generated from a small number of laboratories, using a variety of unstandardized assessments.52–63 Although there is mechanistic insight provided by the isolated preparations of neurovascular coupling, 41 little is known about which mechanisms are sufficient versus necessary for a functional and effective neurovascular coupling response in humans. The available literature related to this topic is reviewed below.
Data indicate that the neurovascular coupling response is comprised of highly redundant mechanistic regulation and is well maintained under a variety of physiological and pharmacological stressors. Such mechanisms are largely unexplored in humans to date. Recently, we have attempted to specifically antagonize cerebrovascular L-type calcium channels using nimodipine, which theoretically blunts the capacity for the cerebrovasculature to respond to an array of vasoactive mediators thought to play a role in neurovascular coupling from isolated preparations (i.e., bradykinin, vasopressin, dopamine, epinephrine, norepinephrine, histamine, glutamate, acetylcholine, serotonin, and prostaglandin). 64 Even with a clinically relevant dose of 60 mg, which is used to treat vasospasm, the neurovascular coupling response was normal as compared to a placebo control trial. 64 Although impaired in various pathologies (as described in detail below), neurovascular coupling is also maintained during exercise when a number of physiological parameters a significantly altered. 55 Together, these data indicate that human neurovascular coupling is remarkably robust and demonstrate the inability of isolated models to predict in vivo responses.
Outside of using physiological and pharmacological stressors, another approach to provide insight into the mechanisms underlying neurovascular coupling has been to use mathematical dynamic multivariate/autoregressive modelling. 54 Through subcomponent modelling, alterations in critical closing pressure (i.e., the pressure inside a blood vessel below which the vessel will collapse) and resistance area product (i.e., resistance with acknowledgement of unknown vessel diameter; inverse slope of instantaneous beat-by-beat relationship between CBF velocity and blood pressure) during neurovascular coupling are thought to provide selective indices of metabolic and myogenic cerebrovascular regulation, respectively, while the influence of blood pressure and changes in end-tidal partial pressure CO2 (ETCO2) on neurovascular coupling are also modelled.65–67 Using this approach, a number of interesting studies have been completed with the following conclusions: (1) hypercapnia impairs the metabolic regulation of neurovascular coupling, (2) breath-by-breath changes in ETCO2 during cognition can impact the neurovascular coupling response, and (3) the blood pressure response to cognitive activation plays a large role in the neurovascular coupling response, particularly the initial spike.53,66,68 These interesting studies have yet to confirm a direct mechanistic link between the physiological regulatory pathways of interest (i.e., metabolic versus myogenic influences) and the subcomponent models/metrics being utilized (i.e. critical closing pressure versus resistance area product).
Some insight into neurovascular coupling in humans has been provided by unique models measuring concurrent cerebral tissue oxygenation and CBF. Although technically and environmentally challenging, these studies can provide detailed insight into flow-metabolism coupling. For example, using functional MRI, it is now known that a consistent BOLD signal over childhood development is underpinned by decreasing neuronal activation/metabolism counterbalanced by increasing neurovascular coupling. 39 Also when using this approach in the healthy human brain, oxygenated venous blood rises (i.e., increased BOLD signal) to a greater extent than CBF during visual stimulation, indicating the effect of exaggerated hyperemia beyond cerebral metabolic demand. 69 Furthermore, although reduced neural activity to a given stimulus has been reported after ischemic stroke, 70 a recent study combining BOLD, fluid attenuation inversion recovery, and arterial spin labelling has shown a variety of neurovascular coupling phenotypes in this population. 69 For example, individuals with ischemic stroke can exhibit a blunted, exaggerated or reduced functional MRI BOLD signal during visual stimulation, with exaggerated or blunted hyperemia. 69 Although these data indicate a mismatch between CBF delivery and oxygenation extraction/metabolism, 69 the mechanisms explaining the variability in the functional MRI BOLD signal responses during visual stimulation are unknown.
Clinical implications
Neurovascular coupling in various clinical conditions.

Mechanistic insight provided only by animal models/isolated preparations.
Mechanisms not clearly established.
An interesting relationship between cognitive activation and neurovascular coupling in humans has been demonstrated, 79 which provides a mechanistic link in conditions where cognition is impaired (Table 1). For example, chronic hypotension depresses cognitive activity and resting CBF, which is directly mediated by neurovascular coupling.72,80 Conversely, cognitive activity can be improved with pharmacological treatment of hypotension, through improvements in neurovascular coupling;52,81 demonstrating that cognitive function is positively related to neuronal tissue oxygen delivery. This link between impaired neurovascular coupling and cognitive decline needs to be further established in other clinical conditions, but if confirmed, may be particularly alarming in the smoking population, where neurovascular coupling does not normalize after cessation. 82 In order to increase the clinical utility of neurovascular coupling as an end-point treatment target, there is a need for an automated and standardized protocol for analysis, which produces interpretable metrics.
Finally, although not a feasible technique in most scenarios, treatment of traumatic brain injury (where the brain is exposed) can allow for direct measurement of CBF, local metabolism (via microdialysis) and/or oxygenation across brain tissue. 83 Under these conditions, it has been demonstrated that during cortical spreading depolarization, the neurovascular coupling response often becomes inverse with neuronal activation, leading to a hypo-emic response. 77 The mechanisms underlying this inversion are unknown; however, this response likely further compromises survival of cerebral tissue which is already ischemic. Clearly, a plethora of future studies are required to delineate the major factors and redundancies in the human in vivo neurovascular coupling response.
Assessing neurovascular coupling in humans
In the following sections, we will illustrate how transcranial Doppler (TCD) and/or near-infrared spectroscopy (NIRS) approaches are used for neurovascular coupling evaluation in humans and describe a standardized approach for eliciting the neurovascular coupling response. Furthermore, we will outline a suggested standardized protocol for neurovascular coupling evaluation, description and quantification in humans, which employs newly developed automated software.
Transcranial Doppler
In humans, the most commonly used approach to study neurovascular coupling involves the use of transcranial Doppler (TCD). The high temporal resolution and non-invasive nature of TCD make it a useful tool in the assessment of integrative cerebrovascular function in terms of neurovascular coupling. New technologies further increase the utility of TCD. A TCD machine is relatively inexpensive ($25,000–$50,000 USD); moreover, TCD is easy to use and is safe in healthy and disease states alike. Therefore, it is practical to use TCD in the clinical setting to assess a variety of different cerebrovascular pathologies. Although the diameter of major arteries may change with fluctuations in arterial blood gases,14,84 the range of arterial CO2 at which TCD accurately estimates changes in CBF is not exceeded during standard neurovascular coupling protocols.52,67 The functional anatomy of the brain allows neurovascular coupling to be easily and reliably examined by measurement of the sensorimotor or cognitive stimulatory effects on cerebral blood velocity; a method termed functional TCD. This technique was first utilized by Aaslid in 1987, who showed that blood velocity in the posterior cerebral artery (PCA) changed with visual stimulation (Figure 2),
85
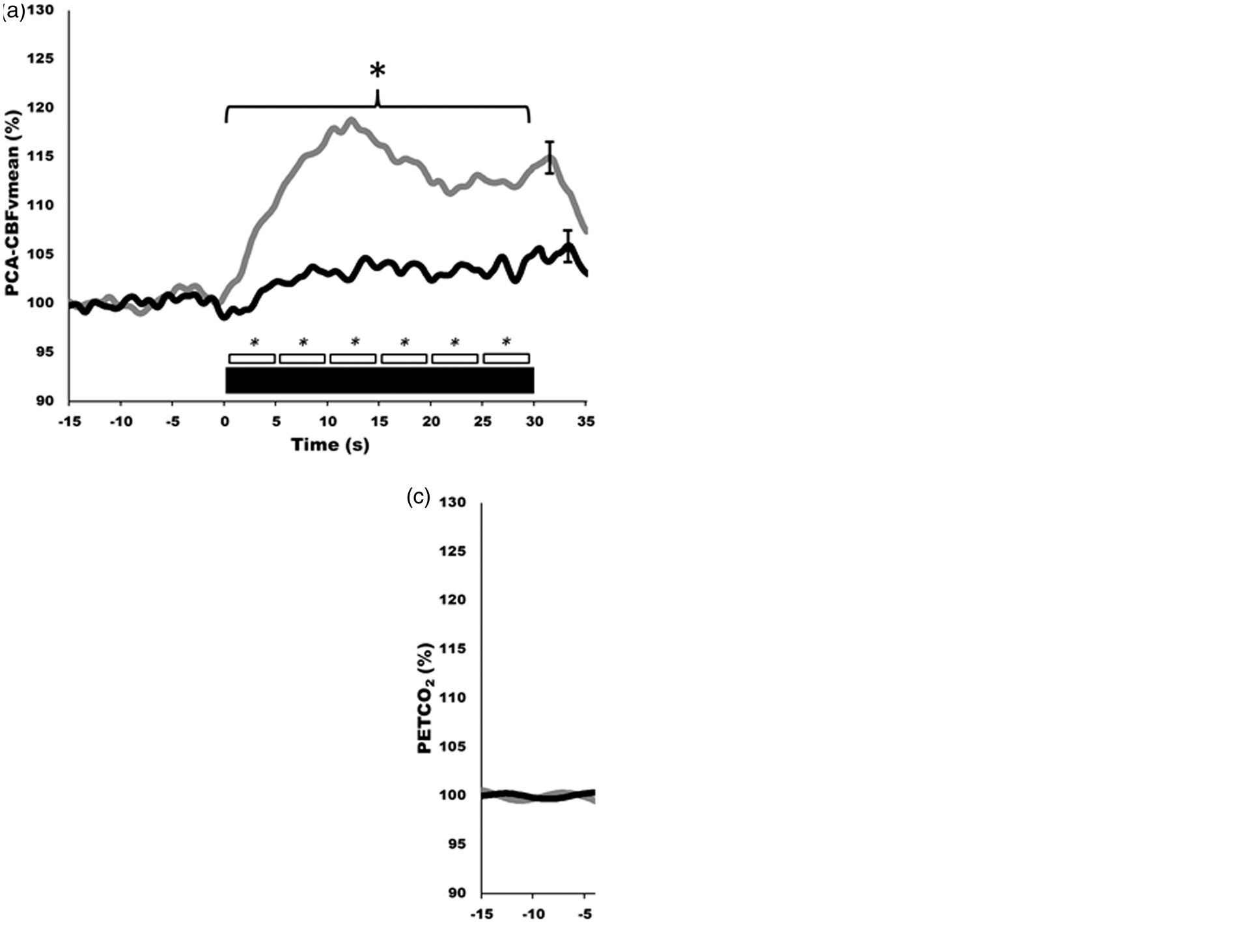
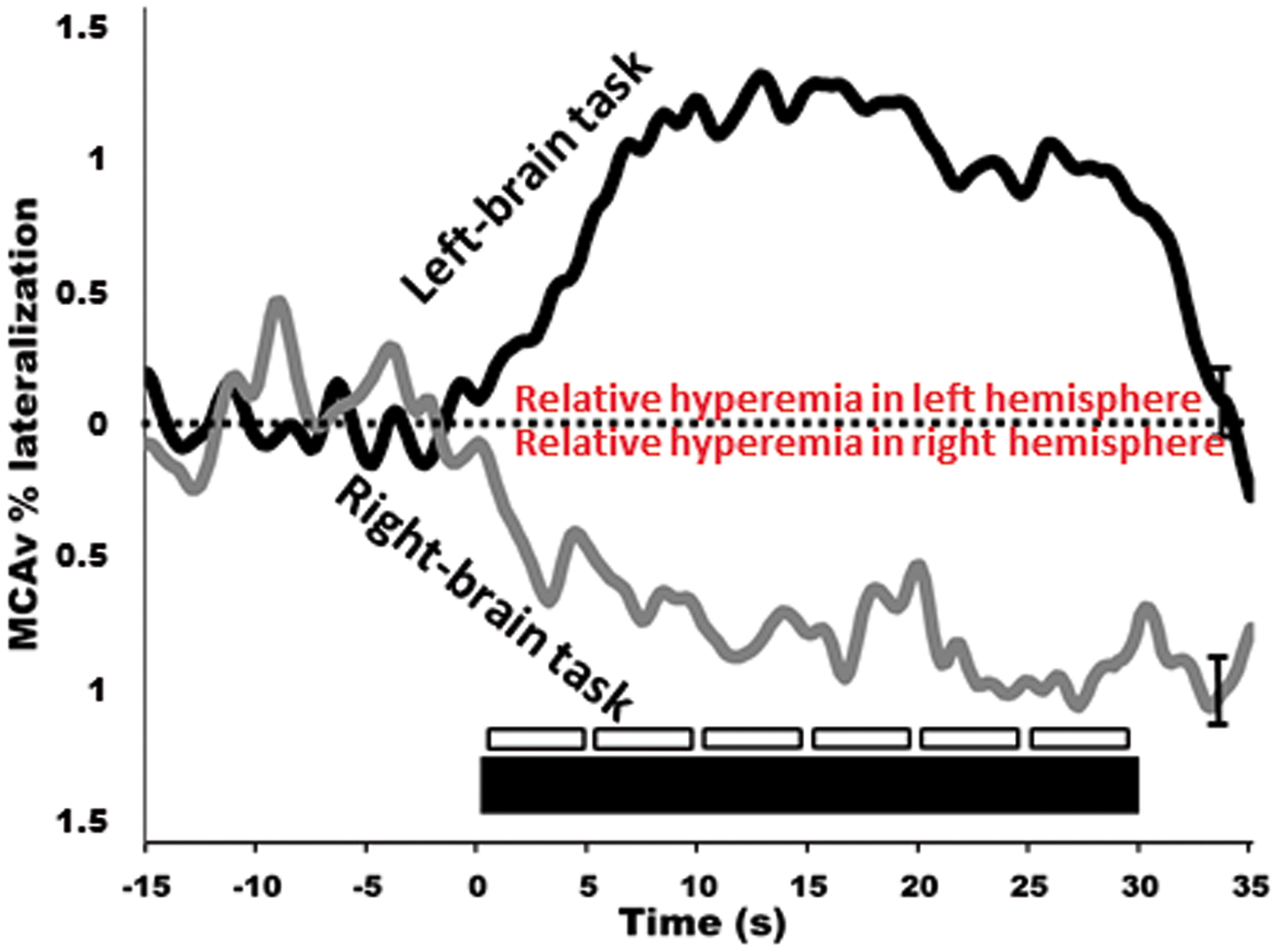
but there are numerous studies in the neuro-cognitive literature that demonstrate consistent CBF changes in response to cognitive, verbal and motor tasks.85–88 In healthy individuals, this response will lead to an average of 10−20% increase in CBF in the PCA and a 5−8% increase in the MCA (Figures 2 and 3).59,86,89 Further, our group and others have explored hemispheric lateralization of CBF delivery using either right-handed/verbal tasks or left-handed motor paradigms to preferentially activate the left and right MCA.66,90 This latter technique has shown that indeed verbal-tasks combined with right-handed motor function lead to hyperemia preferentially in the left hemisphere, while spatial-tasks combined with left-handed motor function lead to hyperemia preferentially in the right hemisphere (Figure 4).66,90
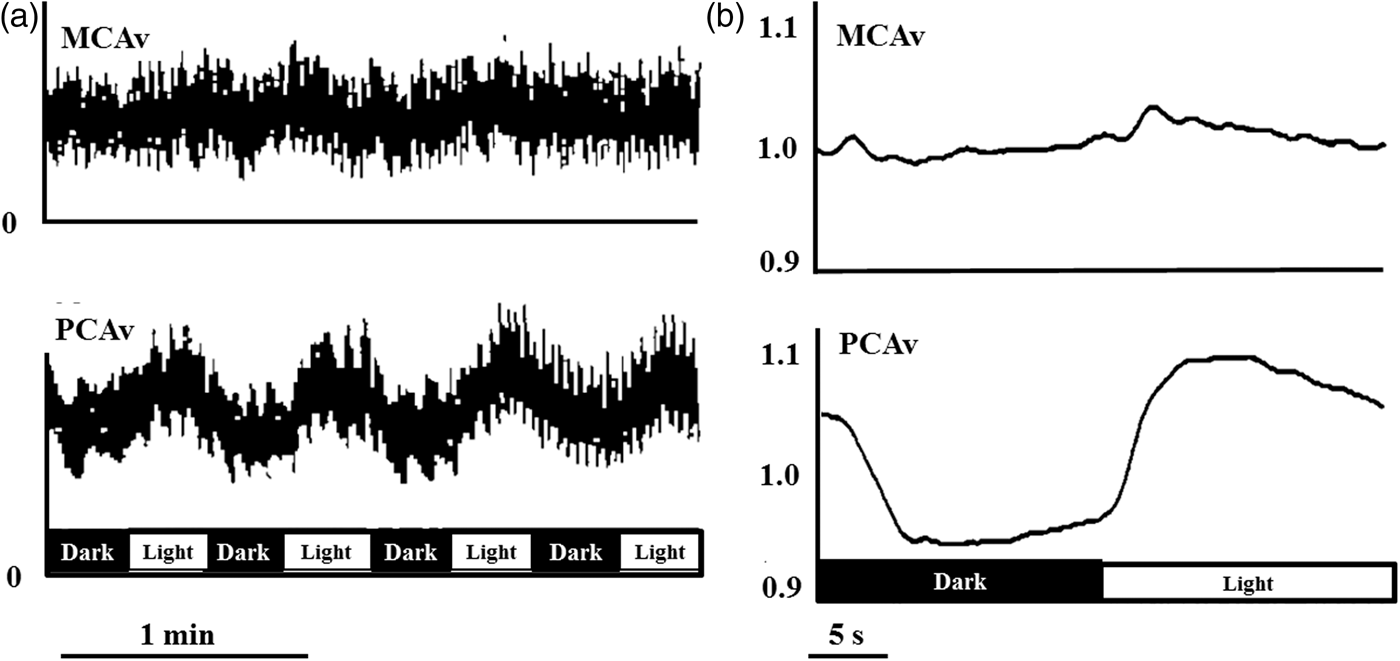
Detailed instructions on insonating the MCA and PCA have been provided elsewhere. 91 Briefly, MCA blood velocity is generally viewed at 35−60 mm depth, after placing the Doppler probe on the middle transtemporal window oriented in line with the coronal axis. The PCA can then be viewed most commonly at 60−70 mm using the anterior transtemporal window by tilting the Doppler probe slightly posteriorly from the MCA. The P1 segment of the PCA will result in blood flow toward the Doppler probe, while the P2 segment curves away from the probe. Both the P1 and P2 segments have been utilized with similar results, however a consistent segment should be maintained throughout a given study.52,55,73,92,93 Lastly, often only the posterior window is suitable for insonating intracranial vessels in the elderly, due to skull thickening with advancing age. 94 Some studies have reported that 10−15% of individuals will have inadequate windows for insonating intracranial cerebral vessels; 95 as such, it may be advantageous to utilize other techniques such as NIRS (see next section) in addition to TCD for studies in the elderly or those with related cerebrovascular pathology.
It is now clear that the diameter of the MCA (and hence possibly the PCA) is influenced by marked changes in arterial PCO2 (>8 mmHg above or below resting) and arterial PO2 (<50 mmHg)84,96 (reviewed in literature9,97). It is less clear that changes in blood pressure will affect the diameter of these intra-cranial arteries. However, it would seem highly unlikely that hyperemic responses noted in the MCA/PCA are being affected by changes in arterial diameter over the neurovascular coupling response. The major reason is that changes in vasoactive influencers (i.e., arterial blood gases) do not occur and the NVC response is very rapid (∼1 s). For example, no discernable change in PETCO2 has been noted during 30−60 s of stimulation. 52,98 Furthermore, if the diameter of the MCA (or PCA) was dilating during the NVC response and being directly impacted by metabolic demand/hypoxia, this would be reflected in a blunted or absent hyperemic response (i.e., the dilation of the MCA/PCA would lead to a reduction in blood velocity). Thus, the major site driving the NVC response seems to be the neurovascular unit rather than the larger intra-cranial arteries.
Near infrared spectroscopy
In addition to TCD, NIRS can also be used for assessing neurovascular coupling in humans.99–102 Incorporating NIRS can indirectly quantify changes in oxygenated and deoxygenated hemoglobin, based on the relative transparency of tissue to near-infrared light, and oxygen-dependent light absorption caused by hemoglobin chromophores. As such, both quantitative and regional indexes of CBF can be provided utilizing the blood-oxygen level dependent signal to detect changes in oxygenated (O2Hb), deoxygenated, (HHb) and total hemoglobin (tHb) within for the most part, the venous vessels of the brain (i.e. <1 mm). 103 Like the TCD, NIRS has very high temporal resolution and can most commonly provide analog outputs at >20 Hz. Moreover, NIRS is also similar to TCD in terms of cost and ease of use. As NIRS also provides some insight into changes in CBF, the underlying arterial CO2 must be considered, as alterations will affect CBF independently from changes in neuronal activity. 104 Also similar to TCD, NIRS can evaluate CBF responses to neuronal activation using a variety of strategies of activation including various emotional, sensory, motor, and pure cognitive tasks.105,106 The history of the development of NIRS is well documented (reviewed in the literature 103 ). The NIRS response during NVC is similar to that generated using TCD and typically results in increased O2Hb and tHb with a concurrent small decrease in HHb; these responses are similarly regionally specific (i.e., visual stimulation leads to posterior cortex hyperemia). 99 During repeated squat-stand maneuvers where blood pressure rapidly increases and decreases, changes in NIRS and TCD were of similar amplitude however changes in NIRS signals trailed those from TCD by 1.8−2.6 s, which may be the result of arterial transit time and fundamental differences in anatomic location between TCD and NIRS. 107 Further, when using other vasoactive stimuli, such as hypercapnia, the NIRS-derived percent hyperemic response is markedly less (1−15% increases vs. 20−100% increases) of that demonstrated by NIRS. 108 This discrepancy, likely reflecting different physiological mechanisms, potentially impacts the sensitivity of NIRS to detect significant differences between clinical and control populations. 109 Clearly, more studies are needed to address the congruency of TCD and NIRS signals during NVC. Care must be taken to ensure that the optodes are located in appropriate locations of the participant’s head to allow for the evaluation of hyperemic responses in relevant portions of the brain, and that the sources and detectors are appropriately spaced. These guidelines are somewhat less stringent as compared to TCD and involve placing the optodes over the general area of interest (i.e., posterior cortex, frontal, partietal lobe, etc.). 99 Apart from the frontal cortex, however, hair will impede signal quality in other brain regions unless the hair is removed. Nevertheless, the simultaneous assessment of TCD (as an index of blood flow delivery) with NIRS (as an index of cerebral tissue oxygenation, the key end-point of interest for neuronal viability) during NVC will provide complimentary insight into integrated cerebrovascular physiology and will permit the inclusion of more subjects, especially when TCD is not possible.
Although TCD and NIRS are by far the most widely used tools for neurovascular coupling measurement in humans, there still exists significant variability in the software and strategies used to analyze these metrics. For example, many studies simply report the maximal change in CBF recorded during the “activation” period with55,92,110 or without111,112 addressing the temporal characteristics, while others attribute specific components and characteristics of the hyperemic contour to specific influences (i.e., perfusion pressure, neurogenic, autoregulation, blood gas concentration).54,74 Our approach is to incorporate both amplitudinal and temporal characteristics by plotting the entire hyperemic contour, while extracting data from temporal subdivisions (i.e., early versus late/5 s duration bins, etc.) for further analysis. 52 The amount of data analysis required for the latter two approaches is significant, with a variety of processing steps required to coalesce contours into interpretable metrics.
Eliciting the neurovascular coupling response
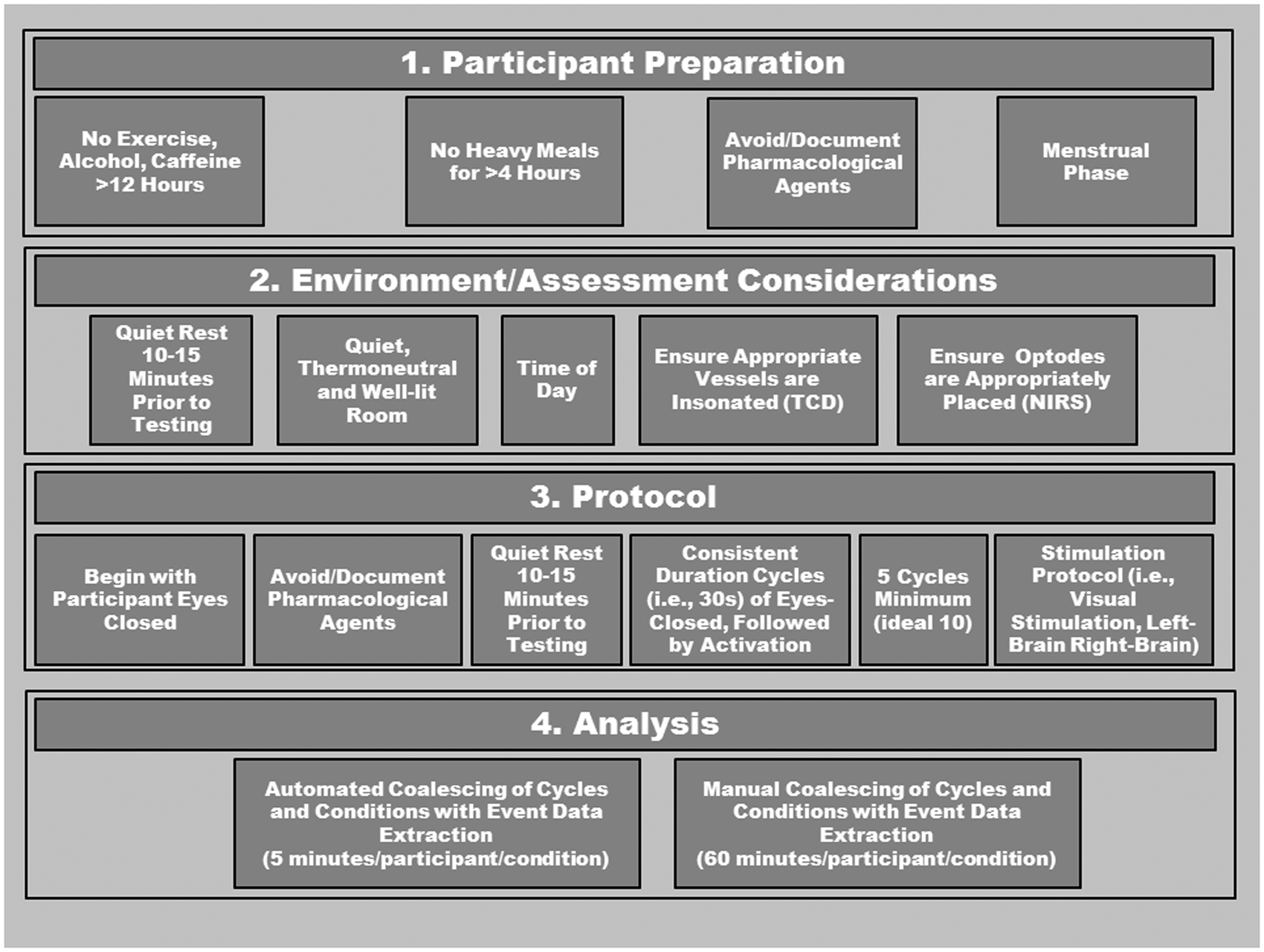
A robust neurovascular coupling response can be assessed by simple visual stimulation (i.e., opening eyes/reading) during beat-by-beat measurement of blood flow/velocity in the PCA relative to the MCA, the prior of which is associated with perfusion of the visual cortex. 52 The selective activation of the occipital lobe is achieved using repeatable and reliable stimuli involving an eyes-open task against a bright visual stimuli (reading, flashing screen, eyes tracking tester’s moving hand, etc.). The visual stimulation provides a strong mechanistic model of neurovascular coupling that is somewhat independent of variable neuronal activation schemes (i.e., a variety of different visual spatial tasks elicit similar PCA hyperemic responses); 73 however, less is understood about the hyperemia response in MCA or anterior cerebral arteries to tasks of varying difficulties or under different states of motivation. Conventionally, after a 10−15 min period of quiet rest (to allow stabilization of blood volume and hemodynamic signals), 5−10 cycles are repeated where each cycle consists of a 20−30 s period of eyes-closed time followed by 30−40 s of eyes-open time.52,73 The greater the number of cycles, the higher the signal-to-noise ratio; however, this obviously must be balanced with the time constraints of the study and the impact on the participants whom may be from a clinical population. Patient positioning can vary depending on study design. Often for clinical relevance, the seated position is used; however, participants may be positioned supine if studying different loading conditions. Similarly, the left or right PCA can be chosen while assessing the contralateral MCA, although this must be consistent throughout the study as perfusion properties can vary between the left and right sides and often CBF is elevated to the left hemisphere. 113
All testing should take place in a quiet, temperature-controlled and well-lit room. Depending on the study design, participants should avoid alcohol, caffeine, and strenuous exercise at least 12 h prior to testing, and be fasted for at least 4 h as these factors can influence vascular tone and function.114–125 Furthermore, assessments should take place at the same time of day, and in the same menstrual phase for premenopausal women.
115
Additionally, medications should be avoided for at least four half-lives if possible, whereas non-steroidal anti-inflammatories should be discontinued for 2−3 days.126,127 This latter recommendation may be problematic in many clinical conditions; however, in any case, all medications should be carefully documented and considered during data interpretation. Due to the ease of administering the test, if artifact occurs within a cycle, another cycle should be added and used to replace the aberrant cycle. If a trial with an artifact needs to be included, the problematic data points should be linearly interpolated using one data point on either side of aberrant data. If more than two aberrant data points occur, this cycle should be removed. Please see Figure 5 for a detailed description of proposed standardization protocol.
Analysis considerations
List of metrics and binning options available with current software.
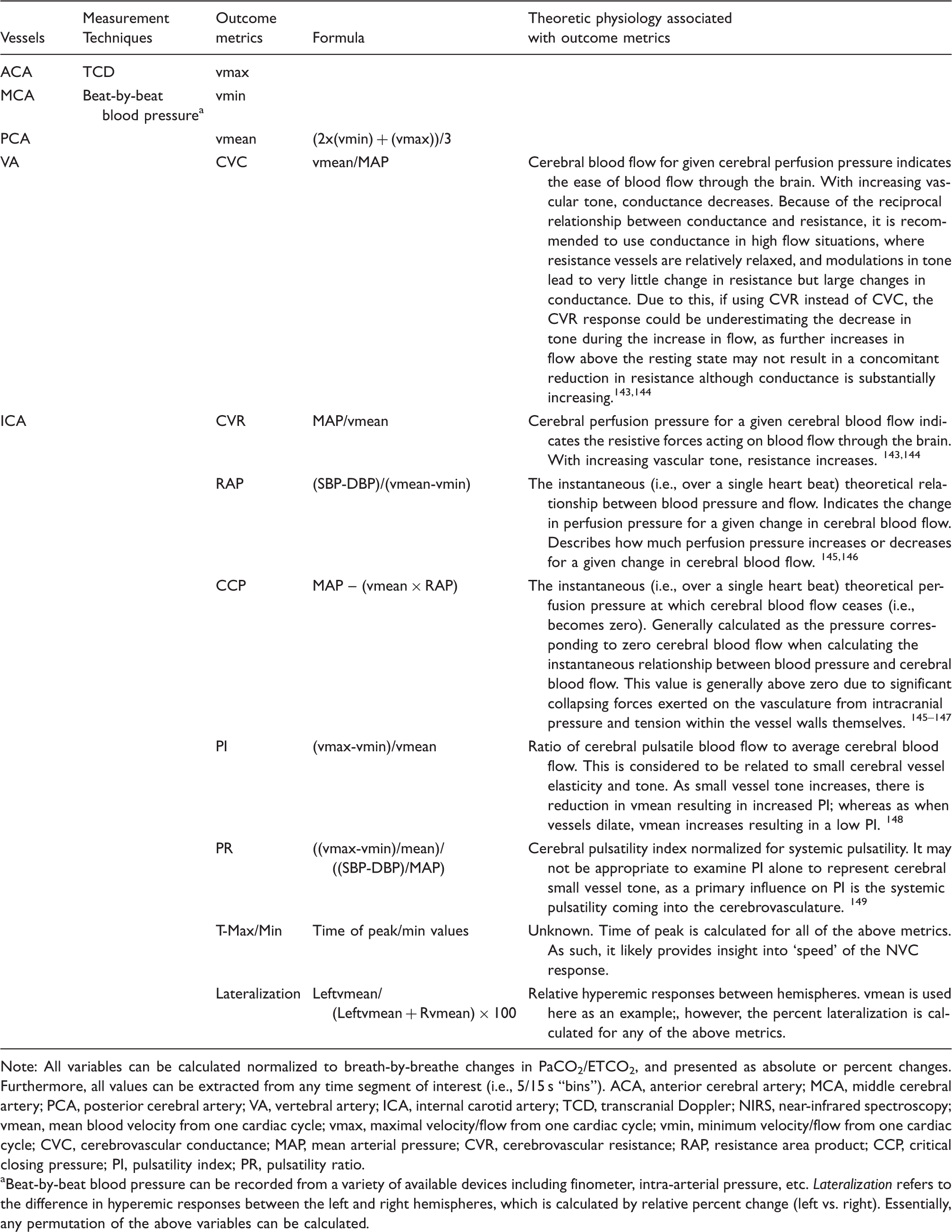
Note: All variables can be calculated normalized to breath-by-breathe changes in PaCO2/ETCO2, and presented as absolute or percent changes. Furthermore, all values can be extracted from any time segment of interest (i.e., 5/15 s “bins”). ACA, anterior cerebral artery; MCA, middle cerebral artery; PCA, posterior cerebral artery; VA, vertebral artery; ICA, internal carotid artery; TCD, transcranial Doppler; NIRS, near-infrared spectroscopy; vmean, mean blood velocity from one cardiac cycle; vmax, maximal velocity/flow from one cardiac cycle; vmin, minimum velocity/flow from one cardiac cycle; CVC, cerebrovascular conductance; MAP, mean arterial pressure; CVR, cerebrovascular resistance; RAP, resistance area product; CCP, critical closing pressure; PI, pulsatility index; PR, pulsatility ratio.
Beat-by-beat blood pressure can be recorded from a variety of available devices including finometer, intra-arterial pressure, etc. Lateralization refers to the difference in hyperemic responses between the left and right hemispheres, which is calculated by relative percent change (left vs. right). Essentially, any permutation of the above variables can be calculated.
In order to normalize the time-scale to when heart/breathing rates are different between subjects/trials or within trials (which is critical when input values are sampled at different frequencies (i.e., heart rate and breathing frequency)), y-axis data are cubic spline interpolated and re-sampled in the x-axis at 5 Hz. As opposed to linear interpolation, which resamples data from a constructed straight line between data points, the cubic spline function has contributions from second derivative and degree-3 polynomial pieces that together allow for prediction of data points transitioning between extracted data points. Due to increased accuracy, a cubic spline interpolation likely provides more physiologically relevant transitions between data points.
Future directions
In an attempt to propagate our understanding of neurovascular coupling in humans, we have taken the step of developing standardized and automated software for neurovascular coupling analysis. Using the high temporal resolution of TCD or NIRS, this program automatically detects and coalesces multiple trials within participants and generates a hyperemia contour for each condition (see Figure 3). Furthermore, this software extracts a vast array of metrics for statistical analysis between groups and/or conditions (Table 2). Moreover, this software generates group mean hyperemia contours for presentation. The use of an approach such as this would serve to standardize the field interested in human neurovascular coupling, increase sensitivity of studies in laboratories currently limited to rudimentary peak responses, and provide a platform for between-laboratory comparisons. The software can be used with data collected on any charting software where data extraction can occur on a beat-by-beat basis and should ideally be gated by the QRS component of the ECG. Furthermore, this software merges with breath-by-breath PETCO2 values. Beat-by-beat, and breath-by-breath data is cubic spline interpolated at 5 Hz to generate a single contour for cycles of stimulus on/off tasks (i.e., eyes-open/closed). Once interpolated and re-sampled, this software then calculates a number of metrics such as conductance, normalized to changes in PETCO2 on a beat-by-beat basis. A list of the key metrics that can be obtained, and the various time-bins are outlined in Table 2. Furthermore, the mathematical formulas and theoretical physiological underpinnings are presented in Table 2.
Conclusions
Neurovascular coupling is a critical component of CBF regulation, having implications for cerebrovascular, autonomic, and cognitive dysfunction. Detailed knowledge has been accumulated related to neurovascular coupling in vitro, or in non-human models that unfortunately suffer from non-physiological approaches and/or unintended secondary consequences of vasoactive sedatives/anaesthetics. We are at an embryonic stage regarding neurovascular coupling function using an in vivo human model. One primary reason for this lack of development is inappropriate and inconsistent analysis strategies and stimulation paradigms.
A major concern related to neurovascular coupling is the lack of association with primary risk factors and relatively small sample sizes in clinical studies. This is due partially to the extremely time-consuming manual data analysis required for each individual in a study. In order to progress beyond this issue, a streamlined analysis using standardized software is needed to allow for large data set incorporation with associated risk-factor prediction for neurovascular coupling impairment within healthy and clinical populations, as well as across the life-span. Further, more studies will be able to develop an adjunct and clinically relevant cerebrovascular outcome measure when evaluating therapeutic interventions; with the capacity to impact management strategies for a number of risk factors and clinical conditions. It is our vision that a widespread implementation and utilization of standardized software would rapidly progress the field and benefit our understanding of neurovascular coupling.
Footnotes
Funding
A.A.P. is a Research Fellow and Postdoctoral Fellow funded by the Craig H. Neilsen Foundation, the Heart and Stroke Foundation of Canada (Focus on Stroke), and Michael Smith Foundation for Health Research. M.M.Z. is funded by Canadian Institute for Health Research (Scholarship). A.V.K. is funded by a Chair in Rehabilitation Medicine, the Canadian Institute for Health Research (Team Grant), Rick Hansen Foundation Clinical Outcome Measures and Optimizing Neurorecovery Program, the Craig Neilson Foundation, Christopher and Dana Reeves Foundation, and the Heart and Stroke Foundation of Canada. P.N.A. is supported by a Canada Research Chair in Cerebrovascular Physiology and Natural Sciences and Engineering Research Council (Canada) Discovery Grant.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Aaron A. Phillips – Conception and design, drafting and editing manuscript; Franco Chan − Conception and design, drafting and editing manuscript; Mei Mu Zi Zheng − Conception and design, drafting and editing manuscript; Andrei V. Krassioukov − Conception and design, editing manuscript; Philip N. Ainslie − Conception and design, drafting and editing manuscript. All authors provided final approval of the present version for publication. Our proposed software is openly accessible for assessments, testing and further development by contacting Dr. Aaron Phillips:
