Abstract
Subjects with asymptomatic carotid stenosis (ACS) may be at risk of cognitive impairment due to cerebral hypoperfusion. In this study, we aimed to detect a threshold of cerebral hemodynamics which is able to identify subjects at risk of cognitive deterioration. In subjects with ACS, cerebral vasomotor reactivity (CVR) was assessed with the breath-holding index (BHI) transcranial Doppler-based method. Cognitive deterioration was defined as a decrease in the MMSE score by ≥2 points after one year. In order to define the threshold of impaired BHI, a ROC curve analysis was performed adopting the binary difference of MMSE score as the outcome and continuous BHI as the testing variable. A total of 548 subjects completed the follow-up. Cognitive deterioration was observed in 119 patients (21.7%). The BHI value ipsilateral to the stenosis was the strongest predictor of cognitive deterioration among the variables tested. The best cut-point to discriminate between normal and abnormal BHI resulted ≤0.89. The post-test probability of cognitive deterioration for an abnormal BHI was 44%, while a normal BHI showed a post-test probability of 9% for the same outcome. The present investigation provides a threshold of reduced CVR that can be useful to identify subjects with ACS at risk of cognitive deterioration.
Introduction
Recent evidence suggests that cognition may be a sensitive index of brain integrity in subjects with carotid disease.1–3 This aspect seems particularly relevant for potential clinical implications in the case of an incidental finding of a severe carotid stenosis in subjects with no other symptoms of cerebrovascular disease. In this respect, asymptomatic carotid stenosis (ACS) has been associated to poor neuropsychological performance4–6 and, more recently, with neuroimaging indices of brain ischemia and aging and with cognitive impairment after adjusting for standard vascular risk factors.7,8
Following these findings, the emerging question is whether to consider symptomatic only those subjects with previous ipsilateral transient ischemic attacks (TIA) or strokes, or to extend this condition to subjects with cognitive impairment. Conversely, previous investigations have suggested that the mere presence of carotid disease is not able to produce cognitive deficits unless it is associated with impaired cerebral hemodynamics. 9
Accordingly, the beneficial effects of treatments, including carotid revascularization, are supposed to be more indicated for the subgroup of subjects with an unfavorable cerebrovascular status.
The availability of specific markers of increased disease risk could help to identify subjects with ACS who are particularly prone to developing cognitive deterioration and could therefore benefit from more aggressive treatment strategies.10,11 Previous investigations have demonstrated a significant link between reduced cerebrovascular reactivity (CVR) to hypercapnia and the risk of cognitive deterioration in subjects with carotid stenosis.3,10,12
In the present study, we aimed to detect a specific threshold of impaired CVR that predicts in ACS the risk of cognitive deterioration. Based on the widely shared concept that any therapeutic intervention to contrast cognitive impairment requires an early diagnosis, we established to take into consideration even slight changes in cognitive perormance during a one-year period.
Materials and methods
This prospective study was carried out at the Vascular Ultrasound Laboratory of the Neurological Clinic at the Marche Polytechnic University Hospital (Ancona, Italy). During a four-year period, we considered for enrollment all consecutive subjects referred by their primary care physicians or by other specialists to receive an extra- and intra-cranial ultrasound examination screening for suspected large-artery atherosclerotic disease due to their vascular risk profile in the setting of a local primary prevention initiative.
Standard protocol approvals, registrations, and patient consents
The study was conducted according to the ethical guidelines of the Helsinki Declaration of 1975 (and as revised in 1983) and was approved by the Ethics Committee of the Marche Polytechnic University. All participants provided their written informed consent.
Study protocol
Extra- and intra-cranial arteries were assessed by continuous wave Doppler and color flow B-mode Doppler ultrasound (Philips iU22, Bothell, WA, USA). Quantification of stenosis was made on the basis of the presence of plaque at the grayscale or color Doppler imaging and on velocity criteria: internal carotid artery (ICA) peak systolic velocity (PSV), end diastolic velocity, and ICA/common carotid artery PSV ratio. 13 Subjects with ultrasound evidence of ≥70% diameter reduction in one ICA without clinical history of stroke or TIA were considered for enrollment. As part of a standardized neuroimaging assessment, all subjects underwent brain MRI or CT examination, and we selected only subjects with no or minimal vascular impairment using Wahlund classification (grade 0: no vascular lesions) or (grade 1: small subcortical focal lesions), defined as areas with high signal intensity on T2 but isointense with normal brain parenchyma on T1). 14
Exclusion criteria were age over 85 years, evidence of carotid occlusion or vertebro-basilar and intracranial steno-occlusive disease, referred or documented cardiac failure (defined as a left ventricular ejection fraction below 50%), referred or documented or treated dementia arising from any cause or any severe psychiatric disease, baseline Mini-Mental State Examination (MMSE) score <25, previous disability defined by modified Rankin Scale score >0, absence of temporal acoustic window/noncompliance with the cerebrovascular reactivity assessment with transcranial Doppler ultrasonography, preexisting cerebrovascular disease (defined as a history of stroke, TIA or neuroradiological signs of significant ischemic lesions according to the Wahlund scale) 14 , or coexisting severe medical conditions interfering with the possibility to perform a follow-up.
In addition, the history of major vascular risk factors and complete blood analysis were obtained from each patient. Hypertension, diabetes, and dyslipidemia were defined according to international guidelines.15–17 With regard to smoking, we considered the current status, while alcohol abuse was defined as a >60 g/day intake. History of coronary artery disease (CAD) defined as documented symptomatic myocardial infarction (MI), angina, coronary artery bypass surgery, or coronary artery angioplasty, and atrial fibrillation (AF), and history of peripheral arteriopathy (PAD) were also obtained. For subjects with a newly diagnosed condition, a specific medical treatment was advised. In each case, best medical treatment for vascular risk factors was recommended to all subjects according to international guidelines. 18
Intracranial vessels were examined by means of a MultiDop X/TCD instrument (DWL Elektronische Systeme GmbH, Singen, Germany). Cerebral vasomotor reactivity (CVR) to hypercapnia was measured using the breath-holding index (BHI). 19 Two transducers placed on the temporal bone window with a stable angle of insonation were used to obtain a bilateral continuous measurement of flow velocity of middle cerebral arteries. The highest flow signal was sought at a depth of insonation ranging from 48 to 54 mm. Subjects were requested to hold their breath for a period of 30 s, which was monitored by a capnometer. This index was calculated by dividing the percent increase in mean flow velocity (MFV) occurring during breath-holding by the length of time (in seconds) that subjects held their breath after a normal inspiration ([MFV at the end of breath-holding − rest MFV/rest MFV] × 100/seconds of breath-holding).
Subjects were followed for 12 months by telephone interviews or clinical examinations at three-month intervals. Particular attention was paid to check and stimulate patients to a proper intake of pharmacologic treatment and adaptation to behavioral recommendations for the control of vascular risk factors.
Global cognitive function was evaluated by using the MMSE adjusted for age and education as assessed for the Italian population. 20
All subjects underwent MMSE testing at baseline and at the end of the follow-up period. All MMSE were administered by the same expert neuropsychologist. Cognitive deterioration was defined as a decrease in the MMSE score of 2 points or more during the 12-month follow-up period. The threshold of two points was decided prior to the study performance.
Neurosonology and neuropsychology operators were blinded to all other data.
Statistical analysis
Subjects’ characteristics, such as age, baseline MMSE score, MMSE at 12 months and years of education, were stored as continuous variables. Sex, smoking, diabetes, dyslipidemia, hypertension, alcohol abuse, and cardiovascular pathologies (CAD, AF, and PAD) were collected as binary variables. Drug therapy was also recorded: antiplatelet agents, oral anticoagulants, statins, antidiabetic, and antihypertensive drugs were synthesized into different dichotomic variables.
The difference between baseline and final MMSE score was stored as continuous and binary variables (<2 or ≥2). Both ipsilateral and contralateral BHI were stored as continuous variables. Binary variables were compared with the chi-squared test, and continuous variables with the t-test.
The relationship between ipsilateral BHI and the MMSE score change was explored with multiple regression models, choosing the best-fitting function.
A ROC curve analysis was performed adopting the binary difference of MMSE score and continuous BHI as the testing variable. We compared ROC curves of mean, ipsilateral and contralateral BHI with the DeLong method and then chose the best cut-off for each curve both with the Youden index and a critical evaluation of the curves. 21 For each cut-off, we calculated sensitivity (Se), specificity (Sp), positive likelihood ratio (LR+), negative likelihood ratio (LR-), positive predictive value (PPV), and negative predictive value (NPV).
The power of ROC curve study was calculated considering an alpha level of 0.05, a sample size of 548 subjects, with 119 positive subjects in the first group, excluding the H0 hypothesis that the obtained AUC is equal to 50%.22,23 After the identification of the optimal cut-off value for the prediction of cognitive deterioration, we prepared a new binary variable. In order to validate our observation internally, we prepared a Cox regression model which was set up using the dichotomous ipsilateral BHI variable with a cut-off value at 0.89 as the main predictor, cognitive deterioration as the main outcome and age, education, sex, smoking, dyslipidemia, hypertension, alcohol abuse, CAD, AF, and PAD as covariates. Finally, we obtained a dichotomous variable applying the classical cut-off value, 0.69, in the ipsilateral BHI variable. Then we cross-tabulated the binary outcome variable with the two dichotomous variables containing the different cut-off variables, and compared the number of correctly diagnosed cognitive deteriorations with the two methods. Statistics were performed with SPSS 13.0 and MedCalc 12.0 for Windows Systems.
Results
From a total of 623 subjects with severe ACS screened, 56 were excluded (seven for the presence of dementia at the baseline, 15 for preexisting cerebrovascular disease, six because of coexisting severe medical conditions, nine for poor temporal windows, and 19 that underwent carotid revascularization).
During the follow-up period, six subjects had a vascular event (two strokes and four MI), and 13 were lost at follow-up (three died and 10 declined to attend the second cognitive evaluation). The final analysis was then performed on 548 subjects who completed the follow-up. Among them, 257 subjects had already been enrolled in our previous study exploring the relationship between cerebrovascular impairment and the long-term risk of cognitive deterioration. 10 The mean age was 69.95 (±4.81) years, with an M:F ratio of 0.51 (males 33.8%) and a mean education of 10.35 (±3.63) years; 19.2% of the subjects were smokers and 4.6% abused alcoholic drinks. Diabetes was present in 17.5%, hypertension in 57.3%, and dyslipidemia in 24.5%; 2.7% of the sample was affected by AF, 6.4% by previous MI and 4.9% by PAD. Mean MMSE was 26.73 (±1.106) at baseline and 25.49 (±1.865) at the end of the follow-up period. Mean BHI ipsilateral to the stenosis was 0.87 (±0.32) and mean BHI contralateral to the stenosis was 1.09 (±0.16). Cognitive deterioration was observed in 119 subjects of the overall sample (21.7%) vs. 429 (78.3%) who remained stable.
In the overall population, 70.3% (n = 385) of the subjects regularly assumed antiplatelet drugs, 5% (n = 28) oral anticoagulants, 60.7% (n = 333) statins, 9.48% (n = 52) antidiabetics and 54.9% (n = 301) antihypertensives. No significant difference in the use of the single classes of drugs was found between patients with and without cognitive decline: 68.9% (n = 82) vs. 70,6% (n = 303) for antiplatelet drugs; 5.04% (n = 6) vs. 5.12% (n = 22) for oral anticoagulants; 59.6% (n = 71) vs. 61.1% (n = 262) for statins; 6.72% (n = 8) vs. 10,2% (n = 44) for antidiabetic drugs; 57.9% (n = 69) vs. 54.1% (n = 232) for antihypertensives (respectively: p = 0.734, p = 1.000, p = 0.832, p = 0.291, p = 0.468, chi-squared test).
The relationship between cognitive deterioration and ipsilateral BHI, both considered as continuous variables, was best explained by an inverse-logarithmic function (r2 = 0.318; p < 0.0001; Figure 1). The BHI ipsilateral to stenosis had an area under the ROC curve (AUC) estimated at 0.736 (95% CI: 0.697–0.773; p < 0.0001) in predicting the cognitive outcome, while the BHI contralateral to stenosis obtained an AUC estimated at 0.502 (95% CI: 0.459–0.544; p = 0.1079). The difference between the ROC curves obtained by ipsilateral and contralateral BHI was significantly different (difference between areas: 0.252; 95% CI: 0.172–0.332; p < 0.0001; Figure 2).
Relationship between ipsilateral BHI and difference between MMSE at the end of the follow-up period (T12) and baseline MMSE (T0). Comparison of ipsilateral and contralateral BHI in the prediction of cognitive deterioration.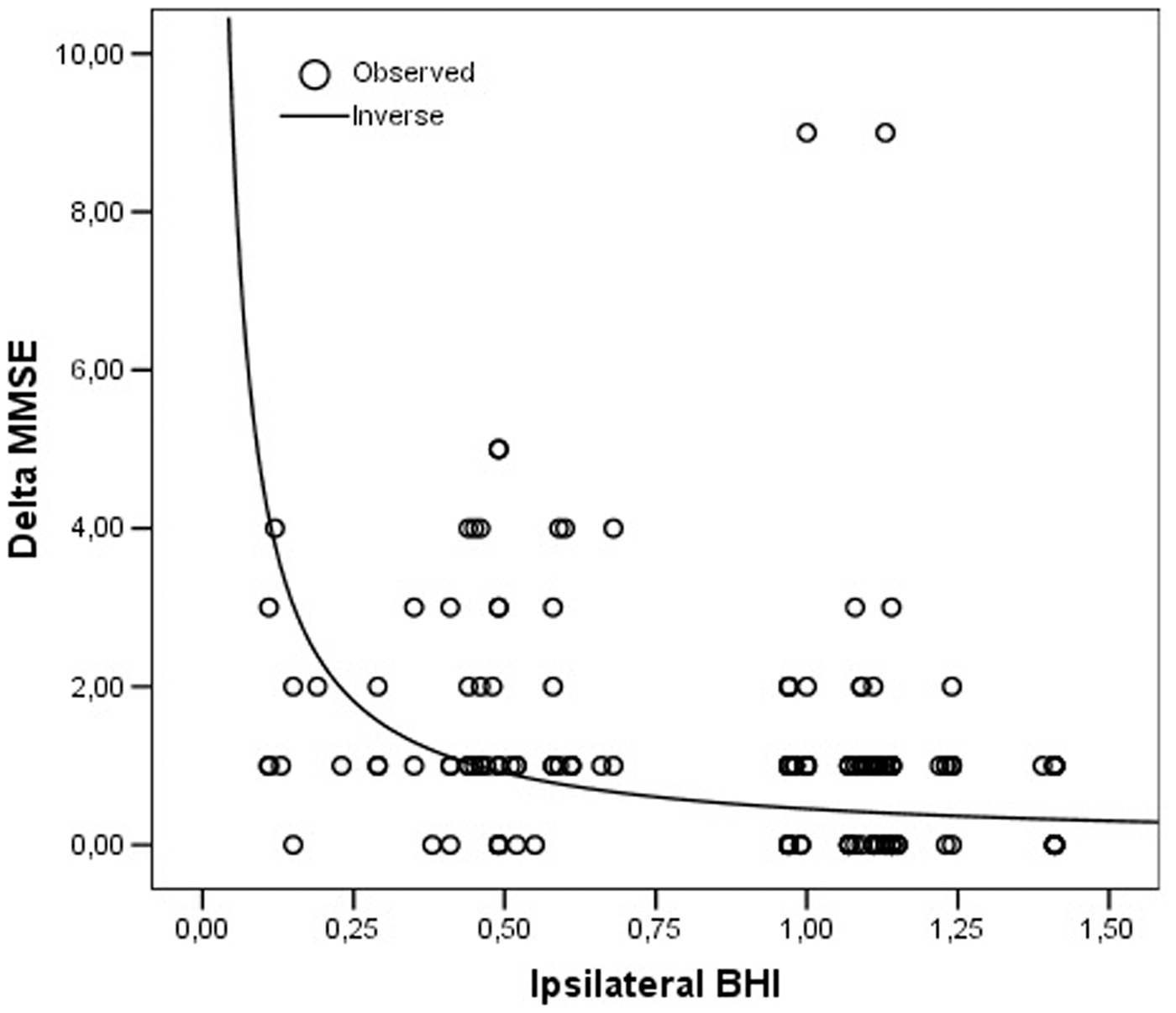
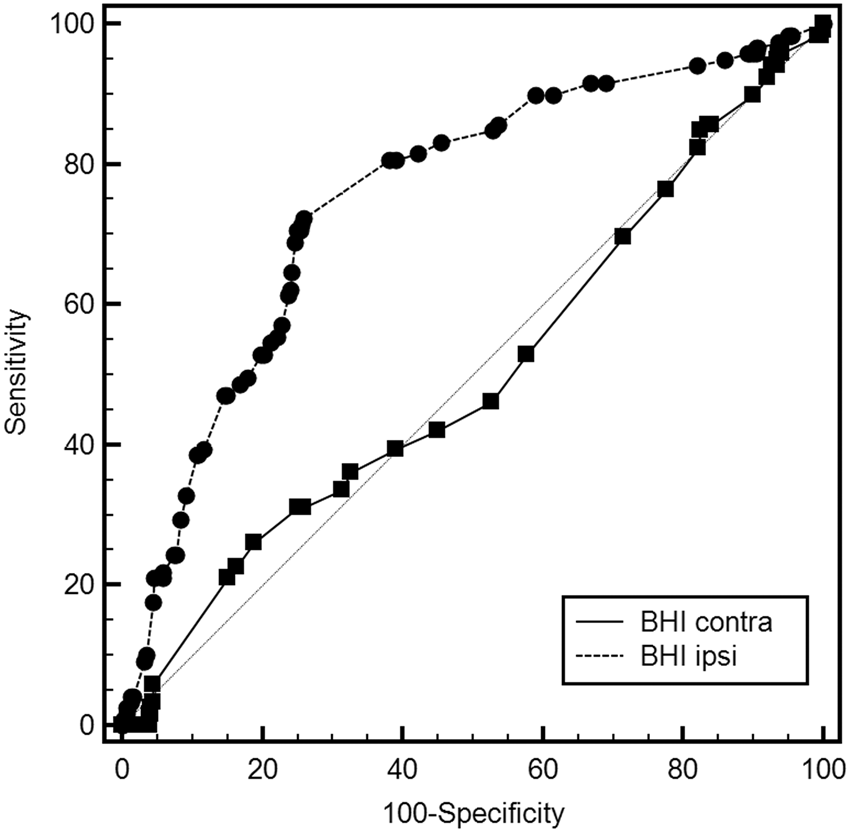
Considering the 119 subjects with cognitive deterioration and the 429 who remained stable, as derived from the ipsilateral BHI ROC curve analysis, we achieved a 100% power to detect a difference of 0.236 between AUC under the H0 hypothesis (0.500) and an AUC under the H1 hypothesis of 0.736 using a two-sided z-test, at a significance level of 0.05.
The optimal ipsilateral BHI cut-off value for predicting cognitive decline in the sample was ≤0.89. This cut-point was set with critical evaluation of both Se and Sp charts and Youden index J (0.4639). At this level, Se was 72.27% (63.3–80.1), Sp 74.13% (69.7–78.2), LR+ was 2.79 (2.3–3.4) and LR− 0.37 (0.3–0.5), with a PPV of 43.7% (36.6–50.9) and an NPV of 90.6% (87.1–93.4).
Baseline characteristics of the sample.
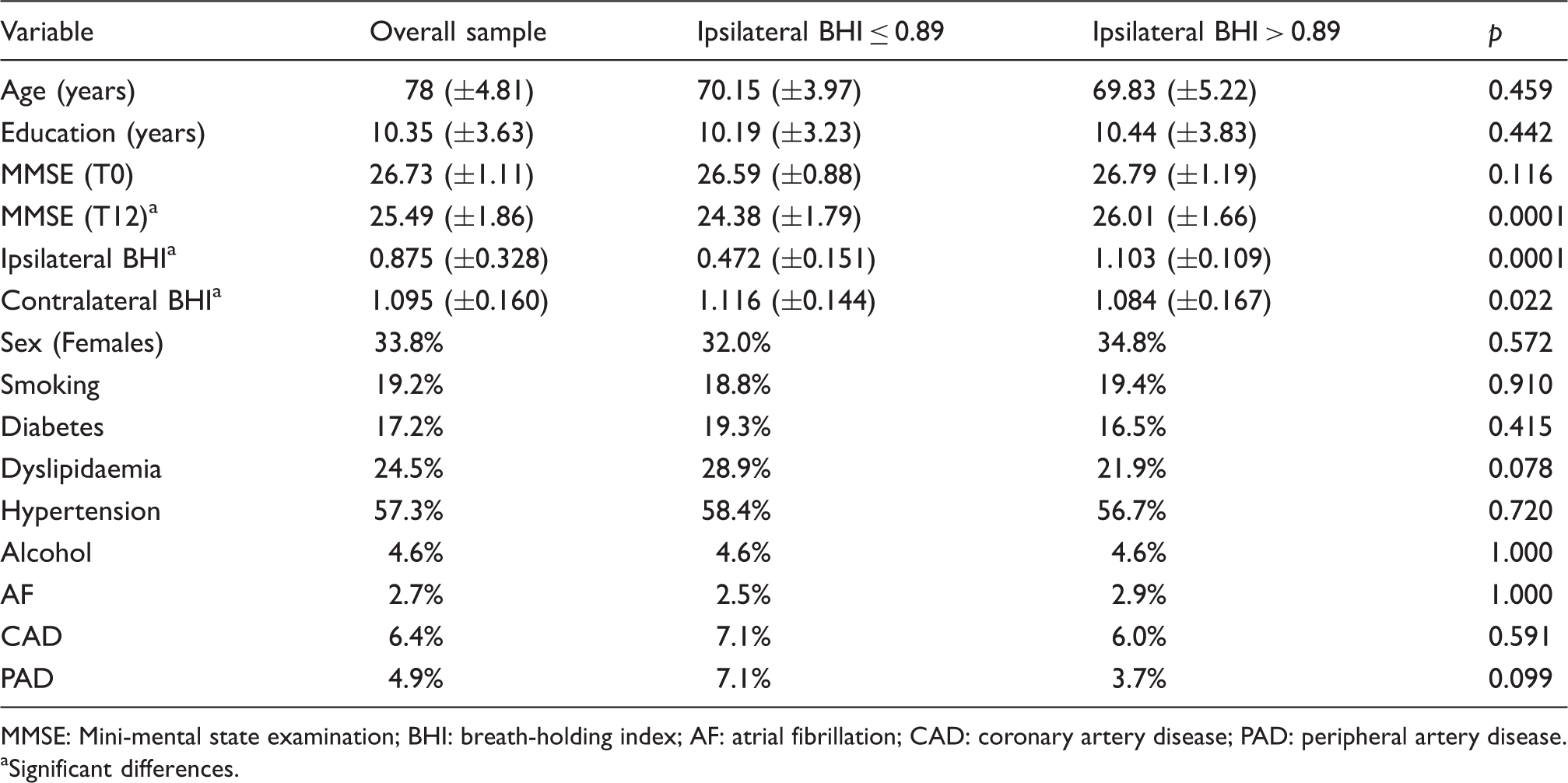
MMSE: Mini-mental state examination; BHI: breath-holding index; AF: atrial fibrillation; CAD: coronary artery disease; PAD: peripheral artery disease.
Significant differences.
Results of the multivariate Cox regression model.
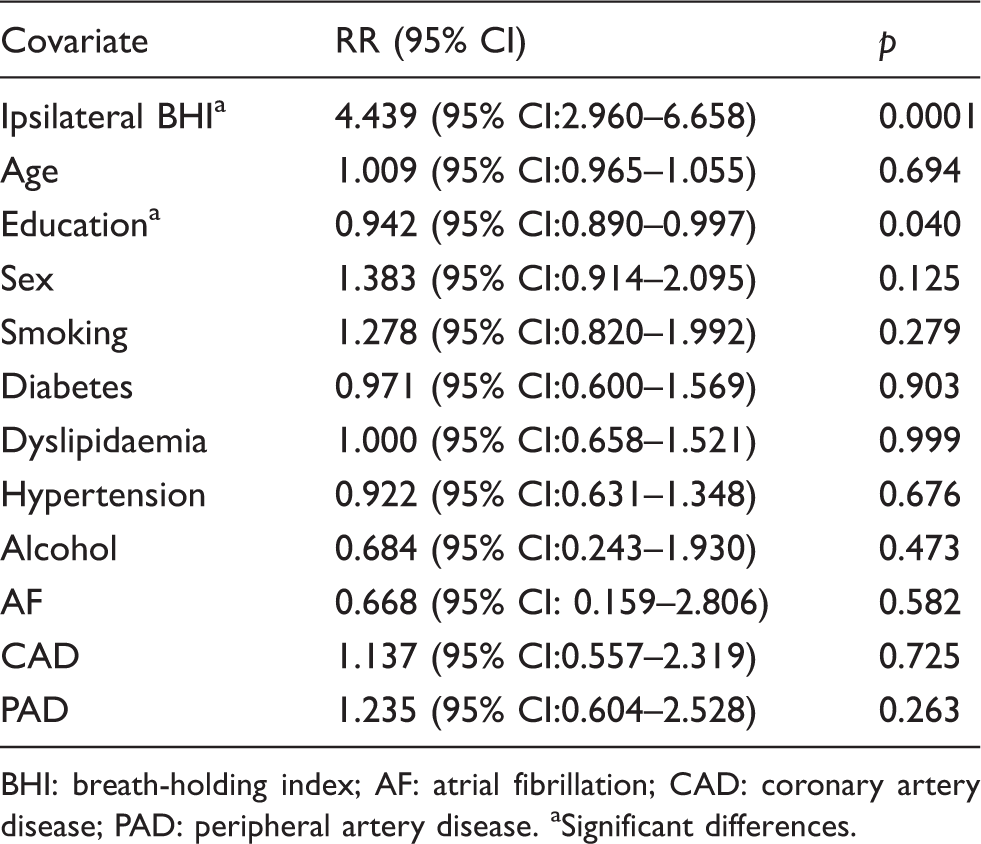
BHI: breath-holding index; AF: atrial fibrillation; CAD: coronary artery disease; PAD: peripheral artery disease.
Significant differences.
Discussion
In the present investigation, we aimed to detect a specific threshold of CVR to hypercapnia which is able to identify subjects with ACS at increased risk of developing cognitive deterioration over a short time period.
In line with our previous studies, we found a direct relationship between hypoperfusion-induced cerebral hemodynamic impairment and reduced cognitive performance.3,10 The specific and new information provided by the present investigation is the identification of a critical level of hemodynamic impairment that can predict an unfavorable cognitive outcome in subjects with otherwise ‘apparently asymptomatic’ carotid disease.
The ability to identify subjects with ACS who are more prone to develop cognitive deterioration is indeed important as they may benefit from more aggressive treatment strategies to potentially reverse or delay cognitive deterioration. Accordingly, the ‘hemodynamically based’ cognitive impairment occurring in subjects with carotid stenosis may be considered as a reversible form of dementia and therefore any attempt to resolve/reverse it should be made. 24 The fact that in the present study we did not find any effect of antiplatelets, antidiabetics, antihypertensives, and statins on the risk of cognitive deterioration, suggests the possibility that the correction of vascular risk factors is more effective in the prevention of thromboembolic events than on the risk of cognitive impairment. Then, the correction of carotid stenosis, with the consequent expected improvement of cerebral hemodynamics, could be taken into consideration as a possible approach to improve cognitive prognosis in apparently asymptomatic subjects.
Carotid atherosclerotic disease is responsible for a significant percentage of ischemic stroke. 25 Therefore, the treatment of carotid stenosis is a well-established therapeutic target for stroke prevention. Nowadays, carotid endarterectomy is considered to be an effective and relatively safe treatment for symptomatic subjects, 26 while carotid artery stenting represents an equally effective and safe alternative when performed by highly qualified centers on those subjects who are deemed poor candidates for surgery. 27 The role of both revascularization procedures in the case of ACS still remains controversial. 26 Many attempts to obtain reliable prognostic indicators for this condition have been made in the last few years. 28 Increasing evidence supports the concept that subjects with ACS can be stratified according to their individual risk of suffering from cerebral ischemic attacks based on the hemodynamic consequences of the stenosis. In particular, a reduction in CVR to hypercapnia has been demonstrated as a useful method to predict the risk of stroke or TIA ipsilateral to the stenosis. 29 In the present study, a very low number of strokes and, in general, of vascular events occurred during the one-year follow-up period. This could be imputed to the fact that during the observation period, each patient was continuously monitored with a careful assessment of compliance to the therapeutic and behavioral recommendations for the control of vascular risk factors. By using this approach, all subjects regularly took prescribed medications (including statins in above 60% of cases) able to reduce the risk of thromboembolic events. Such an effective compliance to treatment recommendations is probably rather difficult to obtain for longer periods, suggesting the possibility that the risk of vascular events can progressively increase over time. For this reason, the classification of patients with carotid atherosclerosis according to their degree of impairment of cerebrovascular reactivity maintains, in our opinion, its importance in order to quantify the medium- and long-term risk of cerebrovascular events. In this respect, the probability of thromboembolic events of causing an ischemic injury can be significantly increased in the presence of an alteration in cerebral hemodynamics. 30
Aiming to identify a new target of treatment strategies, the effect of ACS on cognitive function has received considerable interest over the past two decades, and different investigations have considered the possibility of a direct relationship between stenosis and cognitive impairment. 2 The apparent contradictory results obtained in the investigations exploring the possible impact of carotid stenosis on cognition might be explained by considering the fact that the risk of cognitive impairment may be differently expressed in subjects with an apparently similar anatomic status of the carotid arteries. Accordingly, different investigations have suggested that cognitive deficits may specifically affect a subgroup of subjects in which the carotid lumen narrowing is associated to an impaired cerebral hemodynamic status.1,2,9 This consideration may also explain the lack of strongly encouraging results obtained by the surgical correction of carotid stenosis or occlusion in terms of improvement of cognitive performance31,32 and further underlines the need to find a marker of a preclinical status preceding loss of cognition.33–35
By considering the complexity of the brain network subtending cognitive functions, its vascular-metabolic balance is probably more fragile and susceptible to reach a critical threshold for ischemic damage at an earlier stage with respect to that involved in the control of other functions. Accordingly, the cut-point of BHI value obtained in the present study was higher than that found for the prediction of acute cerebrovascular events identified at 0.69. 36
For this reason, detecting and treating potential causes of alteration in cerebral hemodynamics before they become severe enough to cause irreversible effects should be considered. 37 This could be of keen interest for clinical and practical implications as a potential target of treatment and due to the current lack of any effective medical treatment available for subjects after disease occurrence. 38
In the present study, we took into consideration the previously mentioned concepts and, for this reason, we evaluated the possibility to stratify subjects with ACS without cognitive impairment at baseline in the attempt to identify a preclinical condition preceding a presumably irreversible brain damage. Accordingly, in order to detect possible early signs of the unfavorable consequences of carotid stenosis, we performed a relatively short-time follow-up. Our results suggest that, with a relatively simple and non-invasive approach to evaluate the cerebral hemodynamic status, it is possible to identify a group of subjects with an increased risk of developing unfavorable cognitive changes defined as a loss of at least two points in the MMSE score within 12 months. This extent of MMSE score change is lower than that previously used for the definition of cognitive decline over a longer follow-up period. In a previous investigation, we found that a 0.69 BHI value was able to predict at least a three-point loss in MMSE score during a three-year follow-up period. 10 The results of the present study show that the individuation of subjects at risk of a subtle cognitive decline in a shorter time-period may require the consideration of less severe degrees of cerebral hemodynamic impairment. Accordingly, the use of a cut-off BHI value of 0.69 in the present investigation would not have allowed the identification of five (4.2%) subjects at risk of cognitive impairment. Even if this difference may not appear so significant, from a practical point of view it becomes relevant given the dramatic social consequences of dementia. It is important to underline that the established level of two-point difference in MMSE score was established in order to consider even minimal unfavorable changes in cognitive performances that may be neglected by patients and relatives but could anticipate irreversible negative cognitive changes with the risk to delay any timely and potentially effective therapeutic approach.
Our results show that while we were able to characterize with a relatively high level of precision ‘low risk’ subjects, the detection of a hemodynamic impairment was able to identify about 40% of ‘high risk’ subjects and therefore cannot be considered fully exhaustive. A possible explanation to this finding is that in a considerable percentage of subjects, cognitive deterioration is indeed the result of the interaction of different factors. Considering other possible mechanisms or parameters characterizing carotid stenosis ‘at risk’ of developing cognitive deterioration, i.e. detecting microemboli, 39 defining the ‘unstable’ plaque, 40 monitoring the progression of the stenosis, 41 together with assessing the hemodynamic status, could help to identify subjects with ACS that could benefit the most from revascularization strategies. The lack of this kind of information represents a main limitation of the present study.
Given all the implications for individuals and societies of ‘loss of cognition’, its prevention, along with stroke events, should be considered as a factor in determining the balance of the risks and benefits of therapy for carotid steno-occlusive diseases. Further investigations are needed to test whether implementing a more aggressive treatment approach in stroke-free ICA stenosis subjects with hemodynamic impairment may reverse the impairment and ultimately reduce their risk of cognitive deterioration. Therefore, it would be desirable to design future comparative studies of treatment strategies by considering cognitive outcome as an endpoint and the evaluation of CVR in the selection of subjects.
Furthermore, future research should focus on the identification of other markers of increased risk of cognitive deterioration in order to improve our understanding of the mechanisms behind the development of cognitive dysfunction in carotid stenosis.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
MS, LB and FV contributed to the study concept and design; LB, GV and CP contributed to the acquisition of data; CB, LF, GV and LP contributed to the analysis and interpretation of data; LF contributed to the statistical analysis; MS, FV, LB and CA contributed to the drafting of the manuscript; LP, CA and LB contributed to the critical revision of the manuscript for important intellectual content and MS and LP contributed to the study supervision.
