Abstract
We used functional magnetic resonance imaging to investigate whether hemispheral hemodynamic impairment can play an independent role in the functional reorganization of motor-related activity in the brain. Fourteen patients with large vessel occlusion but no infarct performed a simple motor task with the hand contralateral to the occluded vessel. Statistical parametric maps of regional activity were generated to compare the distribution of motor-related activity among patients with that of control subjects. Patients were classified into normal or abnormal cerebral hemodynamics on the basis of intracerebral vasomotor reactivity using transcranial Doppler and carbon dioxide inhalation. Controls and patients with normal vasomotor reactivity showed typical motor activity in contralateral motor areas. When the 9 patients with abnormal vasomotor reactivity were compared with the 14 control subjects in a single analysis, unique motor activation was identified in ipsilateral motor regions in the nonhypoperfused hemisphere. In a confirmatory analysis, blood oxygen level-dependent (BOLD) signal intensity was averaged in prespecified motor regions of interest. A significant group by hemisphere interaction was identified, driven by higher ipsilateral and lower contralateral hemisphere BOLD signal in patients with abnormal vasomotor reactivity compared with controls (F = 12.40, P=0.002). The average ipsilateral motor region signal intensity was also significantly higher in the subgroup of patients with abnormal vasoreactivity and no TIA compared with controls (P =0.04). Our results suggest that hemodynamic impairment in one hemisphere, even in the absence of any focal lesion or any symptoms can be associated with a functional reorganization to the opposite hemisphere.
Introduction
Functional reorganization in the human brain is thought to occur as a result of focal brain injury, as demonstrated by the appearance of new task-related regional cerebral blood flow responses in positron emission tomography and functional magnetic resonance imaging (fMRI) (Butefisch et al, 2005; Thiel et al, 2005). One commonly reported finding is the presence of ‘contralesional’ neuronal activity. Movement of the affected limb after stroke-induced hemiparesis, for example, may be associated with neuronal activity in the hemisphere ipsilateral to the hand being moved (Cramer et al, 1997; Weiller, 1998). What remains uncertain is the range of cerebral insults that can induce this type of brain reorganization. Focal brain injury appears to be sufficient to induce activity in the contralesional hemisphere, but the question of whether it is necessary to have a focal injury to see this type of activity appear remains unresolved. We recently demonstrated that unilateral cerebral hypoperfusion in the absence of stroke correlated with increased fMRI responses in the opposite hemisphere, ipsilateral to the hand being moved, generating the hypothesis that impaired cerebral hemodynamics was responsible for the ipsilateral activity (Krakauer et al, 2004). In that study, however, five of six patients had transient ischemic attack (TIA), and one had only borderline impairment in cerebral hemodynamics, leaving open the possibility that it was the presence of symptoms that induced the shift to ipsilateral activation, rather than the hypoperfusion per se. We now explore these alternatives by examining a group of patients with large vessel stenosis or occlusion and no focal brain injury on magnetic resonance imaging (MRI), with and without abnormal vasoreactivity, and with and without TIA. We use vasomotor reactivity as a measure of hemodynamic impairment because it can be quantified on an absolute scale (Kastrup et al, 1997), it closely reflects neurovascular physiology (Matsumoto et al, 2000; Vernieri et al, 2001; Cipolla and Curry, 2002) and it correlates well with stroke. In addition, impaired vasomotor reactivity (VMR) as a consequence of large vessel stenosis is particularly relevant to motor dysfunction because much of the cortex involved in motor function lies in the distal field of the internal carotid artery (ICA) and middle cerebral artery (MCA). We wished to test the hypothesis that in patients with large vessel disease, abnormal cerebral hemodynamics is an independent variable in determining the appearance of ipsilateral motor activity, even in patients who have never had stroke or TIA.
Materials and methods
Subjects
We recruited 14 right-handed patients ages 27–83 years with ICA or MCA high-grade stenosis or occlusion but no stroke. Internal carotid artery stenosis was defined by duplex Doppler or angiogram (80%–99% stenosis or complete occlusion). Middle cerebral artery stenosis was defined by segmental flow dropout with distal reconstitution on MRA. There were 14 age-matched controls. Patients had no neurologic symptoms and normal neurologic examination at the time of testing. Standard elements of the National Institutes of Health stroke scale were performed by a stroke neurologist (RSM) to determine strength, coordination, and dexterity of the upper extremities. Five of the patients had TIA 1 week to 6 months before testing. No patient had evidence of infarct in the territory of the stenosed or occluded vessel by fluid attenuated inversion recovery (FLAIR) or diffusion weighted imaging (DWI) MR. The age-matched controls had normal neurologic examination, no evidence for focal lesions on FLAIR or DWI MR images, and normal carotid and transcranial Doppler studies. Handedness was determined using the Edinburgh inventory. Six of the patients and six of the control subjects were part of a previous study (Krakauer et al, 2004). All participants gave written informed consent for the study using an IRB-approved and HIPAA-compliant protocol.
Vasoreactivity Testing
To determine cerebral hemodynamic status, all patients underwent cerebral VMR testing. Bilateral transcranial Doppler monitoring (Pioneer TC 4040; Nicolet Biomedical, Madison, WI, USA) of the MCAs was performed at an insonation depth of 50 to 56 mm as described previously (Marshall et al, 2003). Briefly, partial pressure of carbon dioxide (pCO2) was measured continuously with an inline capnometer (Datex-Ohmeda, Helsinki, Finland) connected via snorkel mouthpiece, with the nasal airway occluded by nose clip. After 2 mins of baseline measurements, subjects breathed a 5% CO2 and air mixture (carbigene) for 2 mins. Vasomotor reactivity was calculated as percent rise in the ipsilateral MCA mean flow velocity (MFV) per mm Hg pCO2 once the MFV curve reached its highest level during the 2-min inhalation period. The contralateral VMR was measured as a control. ‘Normal’ VMR was defined as a rise in MCA MFV of at least 2.0%/mm Hg pCO2, corresponding to 2 standard deviations below the mean of control data from a previous study (Marshall et al, 2003)
Functional Imaging and Voxel-Wise Analysis
We used a ‘block design’ fMRI protocol to generate statistical parametric maps of motor task-related activity in control subjects, patients with normal VMR, and patients with abnormal VMR. For the fMRI session, all patients and controls performed a unilateral repetitive hand closure task in synchrony with a 1 Hz metronome tone during 7 alternating 20-sec rest and activation periods (1.5 T, 128 × 128 image matrix, FOV = 19 cm, TR = 4,000 ms, TE = 60 ms, FA = 60°, 25 slices, 4.5 mm slice thickness, 0 mm gap). For left occlusion, images were flipped to make the ‘right’ hemisphere the ‘affected’ hemisphere in all cases. Thus, the ‘affected hand’ is the ‘left’ hand, contralateral to the hypoperfused right hemisphere; ‘ipsilateral’ brain activity refers to fMRI activity appearing ipsilateral to the hand being moved, in the nonhypoperfused left hemisphere. For controls, we used left hand data because in 10 of 14 patients the ‘affected’ hand was the left. Images were normalized to the Montreal Neurological Institute (MNI) template, smoothed to 6 mm gaussian kernel, then entered into a ‘random effects’ model for group analysis, or ‘fixed effects’ model for subgroup analyses containing fewer than 10 patients (SPM2).
Regions of Interest Analysis
To test our hypothesis that VMR was a significant factor in producing ipsilateral motor region activation with movement of the ‘affected’ hand, we performed a mixed analysis of variance (ANOVA) among all subjects using the blood oxygen level dependent (BOLD) signal intensity in cortical regions of interest (ROIs) that have been consistently shown to be activated in motor tasks in healthy subjects. As described previously (Krakauer et al, 2004), we drew five ROIs using an MNI template and criteria described by Fink et al (1997). The regions were primary sensorimotor cortex (M1S1), lateral premotor cortex (PM), superior parietal lobule, inferior parietal lobule, and supplementary motor area (SMA). We calculated percent BOLD signal change in each ROI in each patient, then averaged to give a motor region hemispheral value for each group or subgroup. The ANOVA was performed to compare control subjects with patients, with hemisphere (ipsilateral to the hand being moved versus contralateral to the hand being moved) examined as a within-group variable. A second ANOVA was performed comparing controls with patients who had abnormal VMR, and patients with abnormal VMR versus those with normal VMR, again using hemisphere as the within-group variable. Student's t-test was used to compare specifically the average BOLD signal in the ipsilateral hemisphere ROIs for controls versus patients with abnormal VMR and no TIA. Threshold for significance was P > 0.05.
Results
All patients and controls had normal neurologic function at the time of testing. All 14 controls and 5 patients had normal VMR; 9 patients had abnormal VMR (one was markedly impaired on one side, moderately impaired on the other; all others were abnormal on one side only). Four patients with abnormal and one patient with normal VMR had TIA (see Table 1).
Clinical and hemodynamic characteristics of the 14 patients
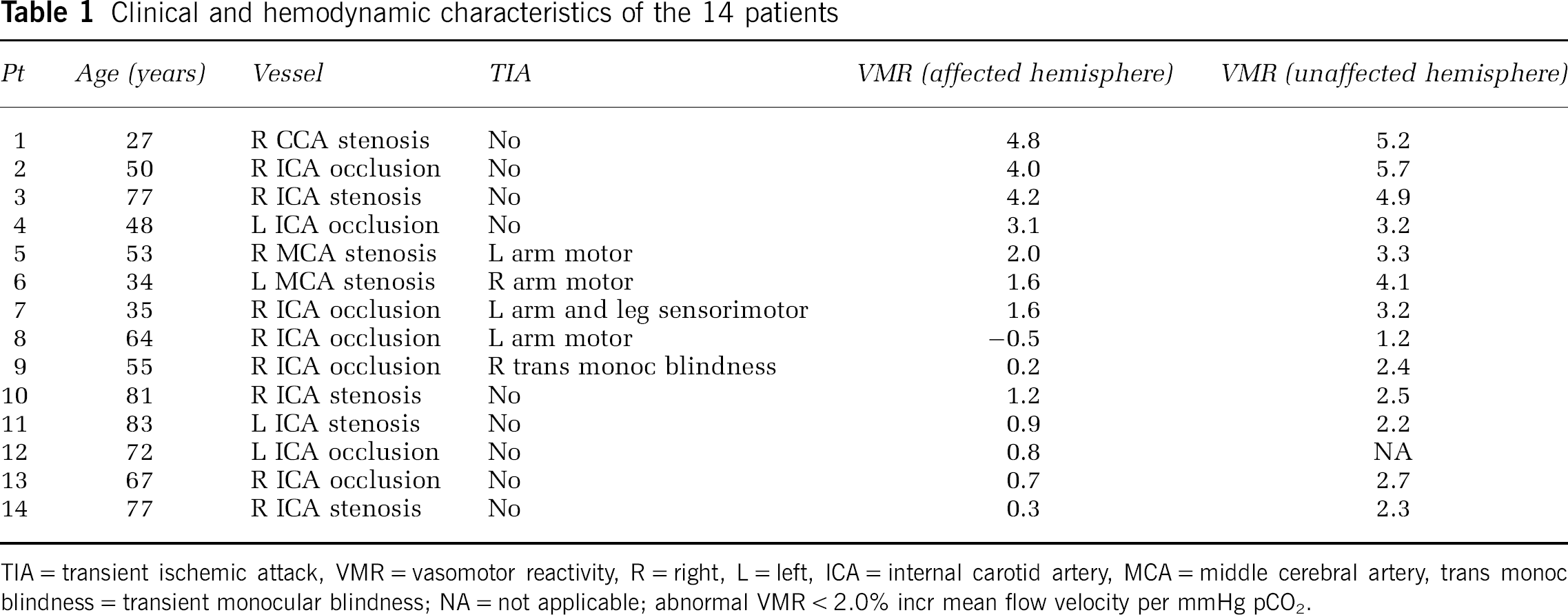
TIA = transient ischemic attack, VMR = vasomotor reactivity, R = right, L = left, ICA = internal carotid artery, MCA = middle cerebral artery, trans monoc blindness = transient monocular blindness; NA = not applicable; abnormal VMR < 2.0% incr mean flow velocity per mmHg pCO2.
Voxel-Wise Comparison
Control subjects performing the motor task (repetitive hand closure) showed typical activation in the contralateral sensorimotor cortex (M1S1C), bilateral PM and SMA (Figure 1A). Patients with large vessel occlusion but normal VMR showed a nearly identical pattern when moving the ‘affected hand,’ contralateral to the occlusion (Figure 1B). Patients with abnormal VMR, in contrast, showed reduced activation in the contralateral hemisphere and increased activation in the ipsilateral sensorimotor cortex (M1S1i), which was not seen in the normal VMR or control groups (Figure 1C). There was no difference in degree of stenosis between patient subgroups.
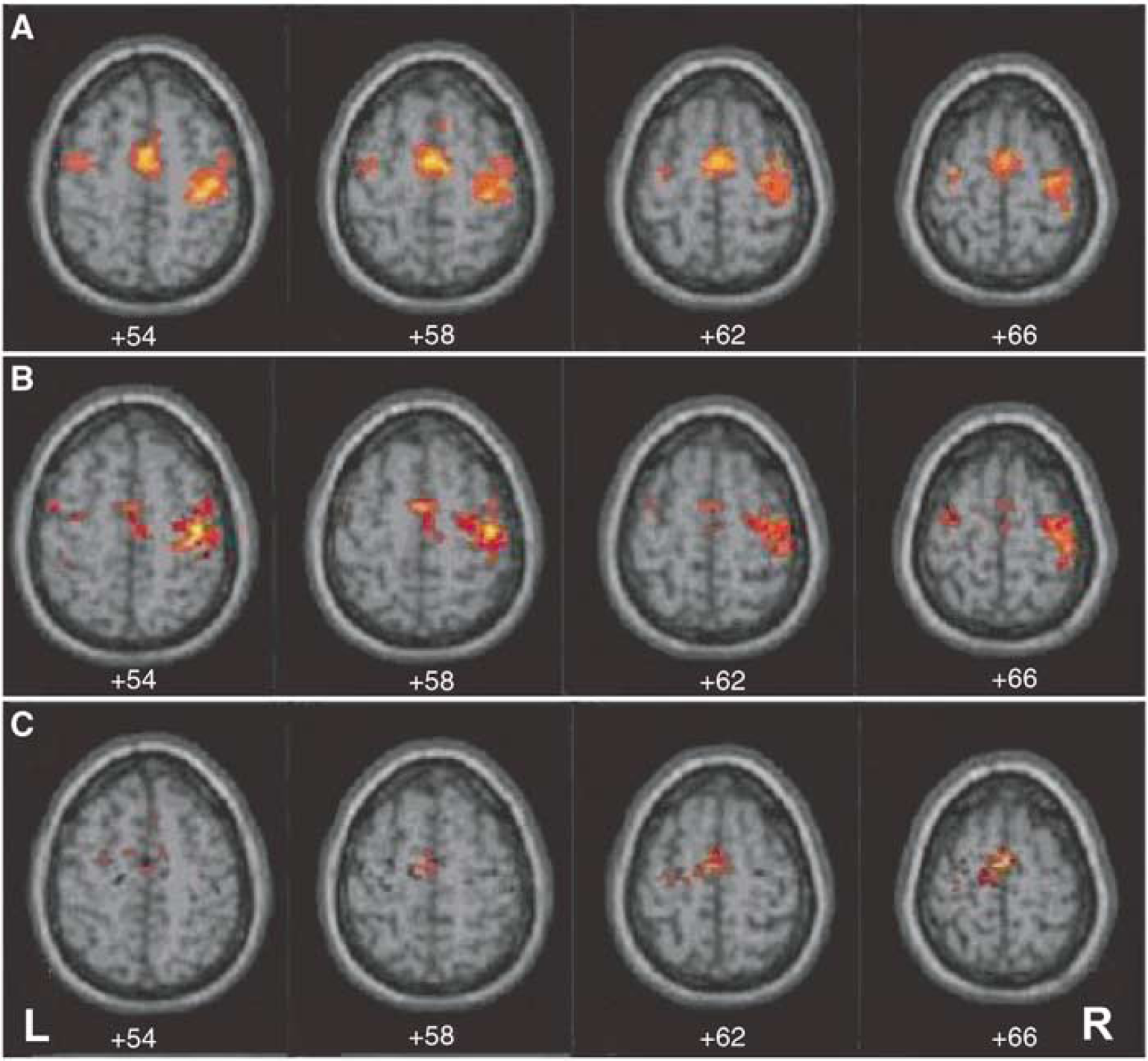
Axial fMRI with BOLD response pattern associated with left (‘affected’) hand repetitive grasp movement. (
We previously showed that patients with large vessel stenosis or occlusion, activated ipsilateral motor regions during ‘affected’ hand movement; however, the numbers were not large enough to permit direct comparisons in a single model. In this study when the 14 control subjects were compared with the 9 patients with abnormal VMR in a single analysis (‘affected’ hand grasp, abnormal VMR patients > controls, fixed effects model, P < 0.001 uncorrected), unique motor activation was identified in ipsilateral M1S1, ipsilateral dorsal PM and SMA (Figure 2A). A comparison of controls with patients with normal VMR yielded no significant differences. Comparing patients with abnormal VMR with those with normal VMR showed a similar pattern of ipsilateral motor activity to the comparison between abnormal VMR patients and controls, with ipsilateral activity apparent also more laterally in the PM cortex (Figure 2B). Finally, to address the hypothesis that the same pattern could occur even in the absence of TIA, we performed the same analysis (patients > controls, fixed effects model, P < 0.001 uncorrected) using the 5 patients with abnormal VMR but no TIA. As shown in Figure 2C, the ipsilateral activity was still present, well seen in the ipsilateral SMA, and M1S1 cortex.
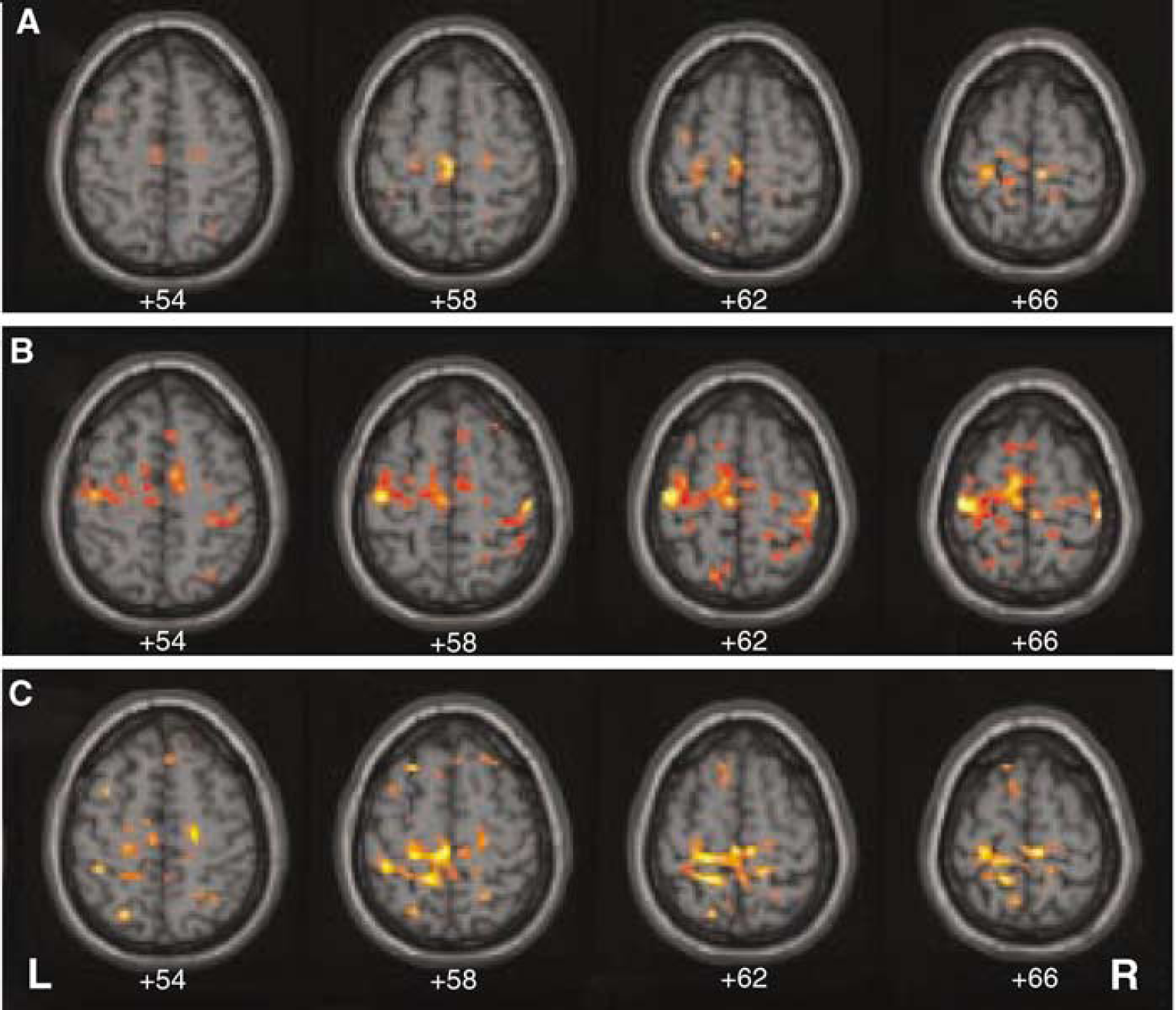
Group comparisons for axial fMRI BOLD response associated with left (‘affected’) hand repetitive grasp movement in patients with abnormal vasomotor reactivity. (
Regions of Interest Analysis
For the average BOLD signal in the 5 motor region ROIs, there was no significant main effect for group (patients (n = 14) versus controls (n = 14)) or for hemisphere (hypoperfused/contralateral versus nonhypoperfused/ipsilateral), but there was a significant group by hemisphere interaction driven by a higher ipsilateral and lower contralateral hemisphere BOLD signal in patients compared with controls (F = 9.49, P = 0.045). When we examined the abnormal VMR subgroup (n = 9) versus controls, the group and hemisphere main effects still remained nonsignificant, but the group by hemisphere interaction became more robust (F = 12.40, P = 0.002, see Figure 3). The same relationship of higher ipsilateral and lower contralateral BOLD activity held true in each individual motor ROI as well. In M1S,1 ipsilateral signal = 2.81E-03; contralateral = 4.31E-03 for the abnormal VMR group; ipsilateral = 6.35E-04; contralateral = 7.39E-03 for the normal VMR group. In PM, ipsilateral 1.58E-03; contralateral = 6.86E-04 for the abnormal VMR group; ipsilateral 1.25E-03; contralateral = 1.51E-03 for the normal VMR group. There was no significant group, hemisphere, or group by hemisphere interaction effect for the comparison between controls and patients with normal VMR.
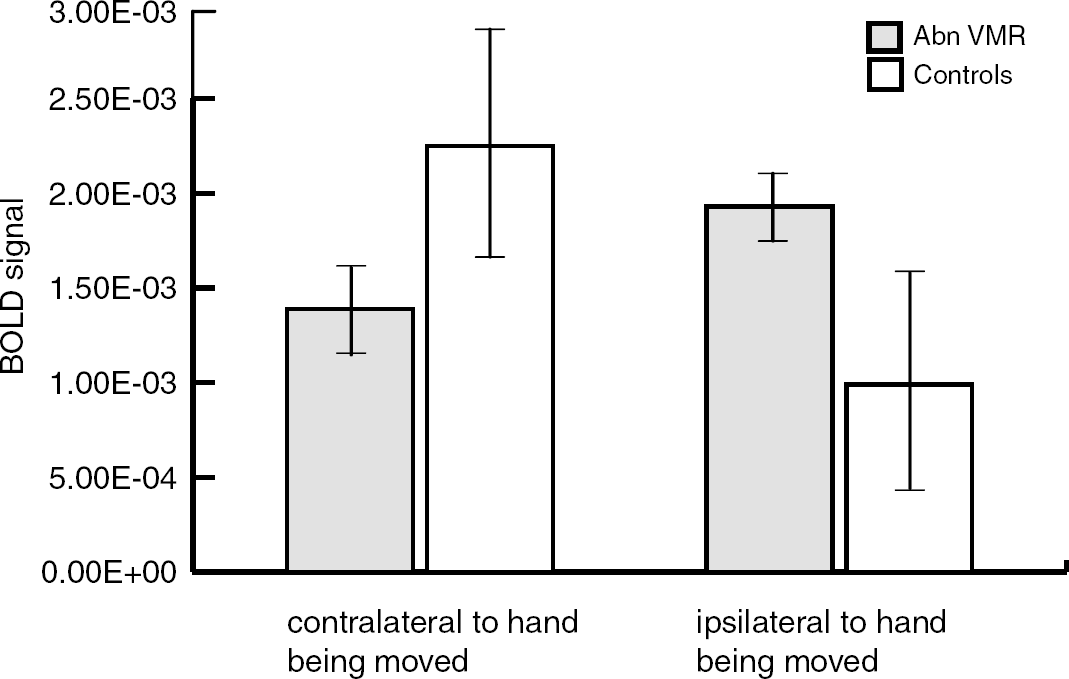
Graph showing average BOLD signal intensity in five motor regions in the hemisphere contralateral or ipsilateral to the hand being moved for controls versus patients with abnormal vasomotor reactivity (Abn VMR). For patients, the hemisphere contralateral to the hand being moved was the hypoperfused hemisphere; the hemisphere ipsilateral to the hand being moved was nonhypoperfused.
When we specifically compared the average ipsilateral BOLD signal in the motor region ROIs in the subgroup with abnormal VMR but no TIA with that of controls, we found it to be significantly higher (3.21E-03 versus 1.00E-03, two-tailed t-test, P = 0.04). There was no significant difference in degree of impairment of VMR between the TIA subgroup and the abnormal VMR/no TIA subgroup.
Discussion
Impaired cerebral hemodynamics in patients with unilateral ICA/MCA stenosis or occlusion was associated with the appearance of increased task-related activity in the opposite hemisphere, ipsilateral to the hand being moved. In this larger study, we were able to address the question of the specificity of hemodynamics that was raised in our previous work (Krakauer et al, 2004). In this study, we compared directly the BOLD activity in patients versus controls, with and without abnormal VMR using both a voxel-wise statistical parametric map and an ROI-based quantitative analysis. We were thus able to isolate abnormal hemodynamics as a variable associated with the ipsilateral activity pattern. The hypothesis that the impaired cerebral hemodynamics was the salient variable in altering the motor activity pattern was further supported by the finding of significantly higher ipsilateral BOLD activity in the subgroup with abnormal VMR who had never had TIA, confirming that the presence of overt symptoms was not required to induce reorganization. Although the lower average BOLD signal in the contralateral (hypoperfused) hemisphere in patients as compared with controls could be attributable to neuro-vascular uncoupling that has been hypothesized to occur in the setting of large vessel occlusion and low blood flow (Powers et al, 1988; Bilecen et al, 2002; Carusone et al, 2002; Hamzei et al, 2003; Rossini et al, 2004) any BOLD activity in the normally perfused hemisphere would not have been subject to uncoupling, and therefore would more reliably reflect underlying neuronal activity(Logothetis, 2002).
The timing and circumstances of our findings differ from the ipsilateral motor activity reported in the setting of focal brain injury even though the topography of the ipsilateral activity is similar. After stroke, the ipsilateral activity occurs early after stroke onset (Marshall et al, 2000; Dijkhuizen et al, 2001; Foltys et al, 2003; Nhan et al, 2004), and appears to correlate inversely with degree of motor recovery over time (Calautti et al, 2001; Ward et al, 2003). In our study, the ipsilateral activity was present in the absence of any deficit and in the absence of any injury on structural imaging. To reconcile these apparently conflicting findings, we propose that the reorganization is induced by brain injury in both cases. In the setting of stroke, the brain responds acutely to a focal injury to critical structures such as the cortical spinal tract (Konishi et al, 2005), compensating with ipsilateral activity until more canonical contralateral pathways are re-established. In our patients, the injury is diffuse and mild, but nonetheless creates an environment that prevents normal neuronal networks from operating. In both settings, the computational work required to accomplish the target motor task is only achieved by recruiting alternative brain regions to share the neuronal burden of the task. In the case of hemispheral hypoperfusion, because the nonconducive environment is broadly distributed, relatively mild, and in most cases develops gradually, the reorganization establishes itself without overt clinical manifestation and persists as long as the adverse physiologic environment is present. Analagous brain states in which this type of reorganization is reported include slow-growing brain tumors and cerebral arteriovenous malformations in which, in the absence of symptoms, alternative patterns of brain activity are shown (Lazar et al, 1997, 2000; Fandino et al, 1999; Petrovich et al, 2004).
Neuronal cellular dysfunction is presumed to occur when the brain's protective responses of arteriolar dilatation and increased oxygen extraction fraction fail to preserve normal tissue metabolism (Powers et al, 1985). In a hyperacute setting, specific levels of perfusion failure have been correlated with specific neurologic dysfunction: hemiparesis has been shown to appear at an average 23mL/100gmin (Jones et al, 1981; Marshall et al, 1999) and disruption in sustained attention has been demonstrated at an average 37 mL/100 g min (Marshall et al, 1999). The minimum reduction in cerebral perfusion sufficient to alter neuronal function at the cellular level has not been described under long-term conditions in humans, although both cellular and metabolic changes have been described in animal models of chronic hypoperfusion (Plaschke et al, 1997; Schmidt-Kastner et al, 2005). Magnetic resonance spectroscopy findings that may indicate neuronal injury have been variable in showing changes after carotid occlusion in humans (Lythgoe et al, 2001; Rutgers et al, 2003). Chronically impaired hemodynamics can be demonstrated physiologically, however (Widder et al, 1994; Derdeyn et al, 2002), and has been shown to increase the risk of future stroke, even in asymptomatic patients (Silvestrini et al, 2000; Markus and Cullinane, 2001; Marshall et al, 2003). The BOLD response itself may also reflect a state of chronic hypoperfusion. An exaggerated ‘initial negative dip,’ (Yacoub and Hu, 2001) reduced amplitude in the positive response (Shiino et al, 2003), and the prolonged time-to-peak of the hemodynamic response function (Ances et al, 2000) have been reported in patients with large vessel occlusions, suggesting that the local microcirculation is barely meeting the second-to-second changes in neuronal metabolic demand during task-related focal brain activity. One might postulate that further reductions in perfusion in that region would prevent blood flow from meeting the metabolic demands of the tissue, and at this point the alternative brain regions would be recruited to preserve normal neurologic functioning.
The brain has extremely limited energy stores that demand adequate blood flow on a continuous basis to maintain normal neurologic function. Our results suggest that if hemodynamic compensatory mechanisms are exhausted, dysfunctional or injured tissue may be present in the hemodynamically compromised region, even if it is not demonstrable on structural imaging. Under such circumstances, the brain will alter its pattern of neuronal activity to accomplish the necessary work of a given neurologic function within the constraints of available hemodynamic physiology.
