Abstract
Cerebral cortical microinfarcts (CMI) are small ischemic lesions that are associated with cognitive impairment and probably have multiple etiologies. Cerebral hypoperfusion has been proposed as a causal factor. We studied CMI in patients with internal carotid artery (ICA) occlusion, as a model for cerebral hemodynamic compromise. We included 95 patients with a complete ICA occlusion (age 66.2 ± 8.3, 22% female) and 125 reference participants (age 65.5 ± 7.4, 47% female). Participants underwent clinical, neuropsychological, and 3 T brain MRI assessment. CMI were more common in patients with an ICA occlusion (54%, median 2, range 1–33) than in the reference group (6%, median 0; range 1–7; OR 14.3; 95% CI 6.2–33.1; p<.001). CMI were more common ipsilateral to the occlusion than in the contralateral hemisphere (median 2 and 0 respectively; p<.001). In patients with CMI compared to patients without CMI, the number of additional occluded or stenosed cervical arteries was higher (p=.038), and cerebral blood flow was lower (B −6.2 ml/min/100 ml; 95% CI −12.0:–0.41; p=.036). In conclusion, CMI are common in patients with an ICA occlusion, particularly in the hemisphere of the occluded ICA. CMI burden was related to the severity of cervical arterial compromise, supporting a role of hemodynamics in CMI etiology.
Keywords
Introduction
Cerebral cortical microinfarcts (CMI) are small ischemic lesions that are a common finding in patients with stroke and dementia.1,2 CMI can be visualized on brain autopsy and recently also with in vivo high-resolution MRI. 3 Studies have shown associations between CMI and cognitive impairment, emphasizing their clinical relevance. 2
CMI can have multiple underlying causes that are linked to large vessel disease, small vessel disease or cardiac disease. 2 Hypoperfusion is proposed as a possible underlying mechanism. We recently showed a relation between reduced global brain perfusion and CMI in a memory clinic population. 4 However, because of the heterogeneous nature of the etiology of cerebral pathologies inherent to this population, the question remains if this observed association could be causal, where low perfusion induces CMI, or just reflects shared risk factors.
In the current study we further investigated hypoperfusion as a possible cause for CMI. We studied CMI in patients with an internal carotid artery (ICA) occlusion, as a model for cerebral hemodynamic compromise. An ICA occlusion causes an altered, more vulnerable, hemodynamic brain state. Although only some patients with permanent (i.e. non-acute) ICA occlusion have resting state hypoperfusion, they are clearly at risk of hypoperfusion (temporary cerebral ischemia) in case of temporary drops in perfusion pressure. 5 We studied if CMI occur more often in patients with an ICA occlusion than in a reference group without ICA occlusion. Among patients with a unilateral ICA occlusion, we examined if CMI occurred more often in the ipsilateral hemisphere than in the contralateral hemisphere. Lastly, within the patient group we examined if occurrence of CMI is associated with the severity of collateral steno-occlusive disease, and with lower cerebral blood flow.
Materials and methods
Study population
Participants took part in the Heart-Brain Connection study, a Dutch multicenter study focusing on cardiovascular and hemodynamic contributions to cognitive impairment. 6 Data collection took place in four university medical centers (UMCs) in The Netherlands (Leiden UMC, Maastricht UMC, Amsterdam UMC and UMC Utrecht). Reference participants were recruited among spouses of patients and through advertising leaflets in the four UMCs.
Generic inclusion criteria for both patients and reference participants in the Heart-Brain Connection study were age of 50 years or older, being independent in daily life and able to undergo cognitive testing and MRI. Exclusion criteria for all participants were a life expectancy of less than three years, current atrial fibrillation, and neurologic or psychiatric disease affecting cognitive performance other than vascular injury or possible co-occurring Alzheimer’s disease. Additional criteria applied for patients with an ICA occlusion. Before inclusion, an ICA occlusion had to be confirmed either with ultrasound, magnetic resonance angiography or computed tomography angiography. Also, patients with an ICA occlusion should not have had a brain infarct or transient ischemic attack in the three months prior to inclusion, and should have no planned carotid surgery or participation in an intervention trial. There were no additional criteria regarding the occlusion being symptomatic or asymptomatic.
For the current study five patients with an ICA occlusion and one reference participant were excluded because brain MRI was not available. In addition, two patients and two reference participants were excluded because of missing MRI sequences and three patients due to insufficient MRI scan quality for the sequences necessary to rate CMI. This resulted in a study population of 95 patients and 125 reference participants.
The Heart-Brain Connection study was approved by the Medical Ethics Review Committee of the Leiden UMC (number P.14.002) and local boards of the other UMCs. The study was conducted in accordance with the declaration of Helsinki and the Medical Research Involving Human Subjects Act (WMO). Written informed consent was obtained from all participants prior to enrolment in the study. The data that support the findings of this study are available from the corresponding author upon reasonable request.
Clinical characteristics
For the patients with an ICA occlusion we recorded the side of the occlusion and whether the ICA occlusion had been symptomatic, which we defined as an ipsilateral transient ischemic attack (TIA) or ischemic stroke. This was based on medical records. Possible occlusions and stenosis in other cervical arteries (i.e. left and right internal carotid arteries and left and right vertebral arteries) were recorded from medical records as well (i.e. duplex ultrasound, computed tomography angiography and/or magnetic resonance angiography).
Demographics and vascular risk factors were recorded for both patients and reference participants. Hypertension was based on presence in medical history. Current office hypertension was defined as a mean systolic tension >140mm/Hg or diastolic tension >90mm/Hg measured during the research day. Hypercholesterolemia was defined as presence in medical history or medication use. Diabetes mellitus was based on medical history. Smoking was defined as current or previous smoking and obesity was defined as a body mass index over 30.
All participants underwent a standardized neuropsychological test battery. Raw test-scores were corrected for age, sex and education relative to the reference group and a mean composite cognitive z-score was calculated for every patient with an ICA occlusion. 6
MRI protocol and analysis
Brain MRI was acquired at 3 T Philips Ingenia, Philips Achieva and Philips Gemini MRI scanners (Philips, Best, the Netherlands). The brain protocol included a 3 D T1-weighted image (TR/TE/TI = 8.2/4.5/990 ms; shot interval 3000 ms; flip angle 8°; voxel size 1.0×1.0×1.0 mm3), fluid-attenuated inversion recovery (FLAIR) image (TR/TE/TI = 4800/313/1650 ms; TSE factor 182; voxel size 1.11×1.11×1.11 mm3), susceptibility-weighted image (SWI) (TR/TE = 45/31 ms; flip angle 13°; EPI factor 3; voxel size 0.8×0.8×1.6 mm3) and phase-contrast flow measurement (TR/TE = 12/8.2 ms; flip angle 10°; Venc 200 cm/s; untriggered; 10 averages; voxel size 1.17×1.17×5 mm3).
CMI were rated in the cortex according to previously established criteria for 3 T MRI. 2 In short, CMI had to be hypointense lesions on T1-weighted MRI with a corresponding hyper- or isointense signal on FLAIR and SWI, <4 mm in diameter, and located strictly intracortical. Of note, these criteria can currently not be used to identify subcortical CMI. 2 CMI had to be distinct from perivascular spaces and visible in at least two planes (i.e. sagittal, transversal, coronal). Lesions neighboring a larger stroke (i.e. <1 cm in the same gyrus) were excluded. 2 CMI were rated by three experienced raters (HvdB, DF, RvdB) with the use of an in-house developed tool in MeVisLab (MeVis Medical Solutions AG, Bremen, Germany). Raters were blind to the clinical condition of the participants. Interrater agreement was good (interclass correlation = 0.97 in a random subset of 31 scans).
Infarcts, microbleeds, enlarged perivascular spaces and white matter hyperintensities (WMHs) were rated by a neuroradiologist (JdB) according to the STRIVE-criteria. 7 Small vessel disease (SVD) presence was then summed in a previously developed SVD burden score for all patients. 8 Total brain volume, grey matter volume and WMH volume were segmented with an automated pipeline (Quantib brain, Rotterdam, the Netherlands), taking into account manually segmented infarcts and other pathologies. Blood flow in the basilar artery and ICAs was quantified (ml/min) based on phase-contrast flow scans. The arteries were manually contoured using the flow analysis tool of Mass software (Division of Image Processing, Department of Radiology, Leiden University Medical Center, Leiden, the Netherlands). We then calculated total cerebral blood flow by summing blood flow in all arteries, dividing this by the individual’s brain volume and multiplying the result by 100 (thus obtaining total blood flow in ml/min/100 ml).
Statistics
Differences in baseline characteristics between patients and the reference group were assessed with an independent t-test for age and cerebral blood flow, χ2 for sex and vascular risk factors, and Mann-Whitney U test for education. Logistic regression was used to determine whether an ICA occlusion (versus reference group) was associated with CMI presence, also when controlling for differences in baseline characteristics between the groups. Total numbers of CMI for patients and the reference group where compared with a Mann-Whitney U test.
Further analyses were only carried out in the group of patients with an ICA occlusion. Differences between patients with and without CMI were assessed with an independent t-test for age and cognition, χ2 for sex, and Mann-Whitney U test for education. CMI count in the ipsilateral and contralateral hemispheres was compared with a Wilcoxon signed rank test. The relation between number of occluded or stenosed cervical arteries and CMI was tested with a Mann Whitney median test and the relation of cerebral blood flow with CMI presence was tested with regression analysis. The relations of risk factors and MRI markers with CMI presence were analyzed with logistic regression for categorical data and Mann-Whitney U test or regression for continuous data. Brain volumes were corrected for age, sex and intracranial volume and white matter hyperintensity volume was log transformed additionally.
Analyses were carried out in SPSS Statistics version 25 with the significance threshold set at p<.05.
Results
Comparison between patients with an ICA occlusion and the reference group
ICA occlusion was confirmed for all patients and ruled out for all reference participants on the research MRI scan. The characteristics of patients with an ICA occlusion and participants from the reference group are shown in Table 1. Patients with an ICA occlusion were of similar age as the reference group, but more often male, and lower educated. As expected, the burden of vascular risk factors was higher among patients. Cerebral blood flow was 14% lower in patients with an ICA occlusion (average 47.6 ml/min/100 ml; SD 12.8) than in the reference participants (average 55.5 ml/min/100 ml; SD 13.6; p<.001).
Demographics and vascular risk factors of patients and reference group.
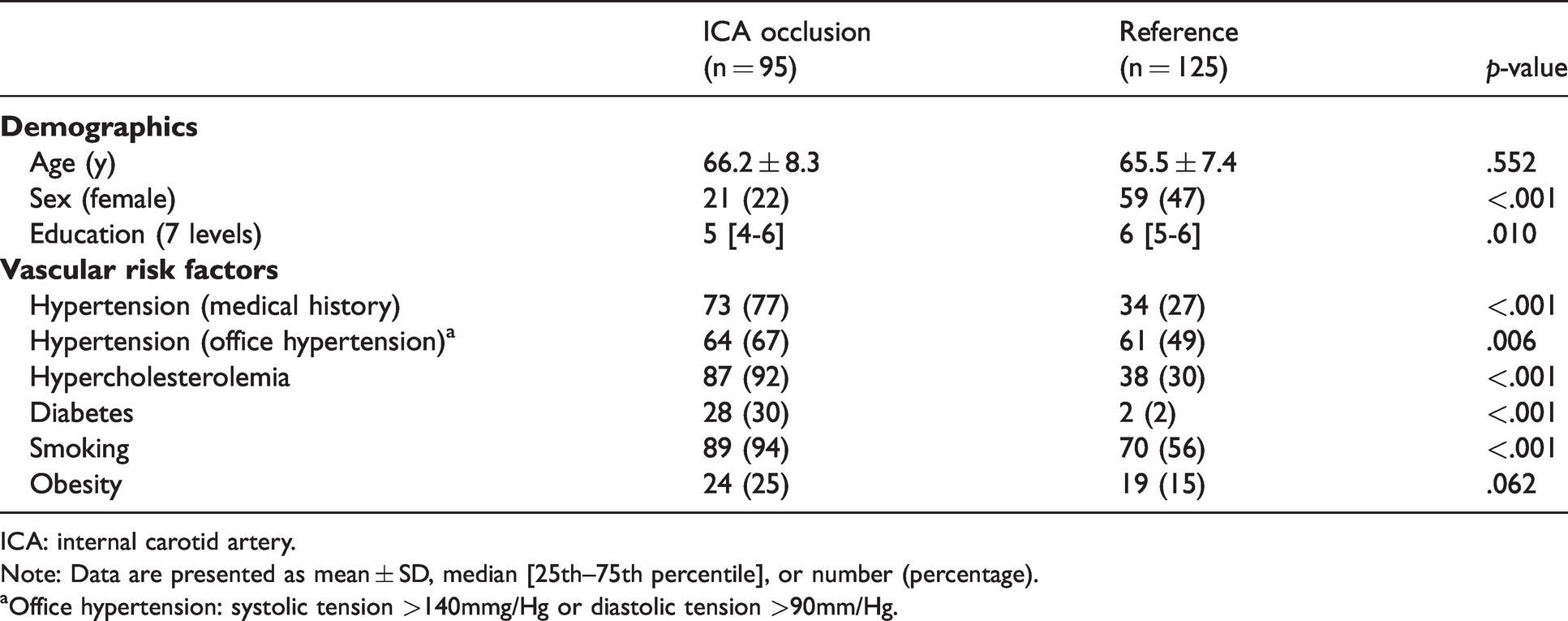
ICA: internal carotid artery.
Note: Data are presented as mean ± SD, median [25th–75th percentile], or number (percentage).
aOffice hypertension: systolic tension >140mmg/Hg or diastolic tension >90mm/Hg.
CMI were present in 54% of the patients with an ICA occlusion and in 6% of the reference group (crude OR 17.0; 95% CI 7.5–38.6; p<.001; OR corrected for sex and education 14.3; 95% CI 6.2–33.1; p<.001; additional correction for all defined vascular risk factors OR 6.9; 95% CI 2.4–20.0; p<.001). Among the participants with CMI, 69% of the patients and 25% of the reference participants had multiple CMI (range 2–33 and 2–7 respectively). The total number of CMI per participant was significantly higher in patients than in the reference group (p<.001).
Analyses within the patient group
The characteristics of patients with an ICA occlusion with CMI and without CMI are shown in Table 2. Only hypertension and hypercholesterolemia were associated with CMI presence in patients.
Demographics and vascular risk factors for patients with and without CMIs.
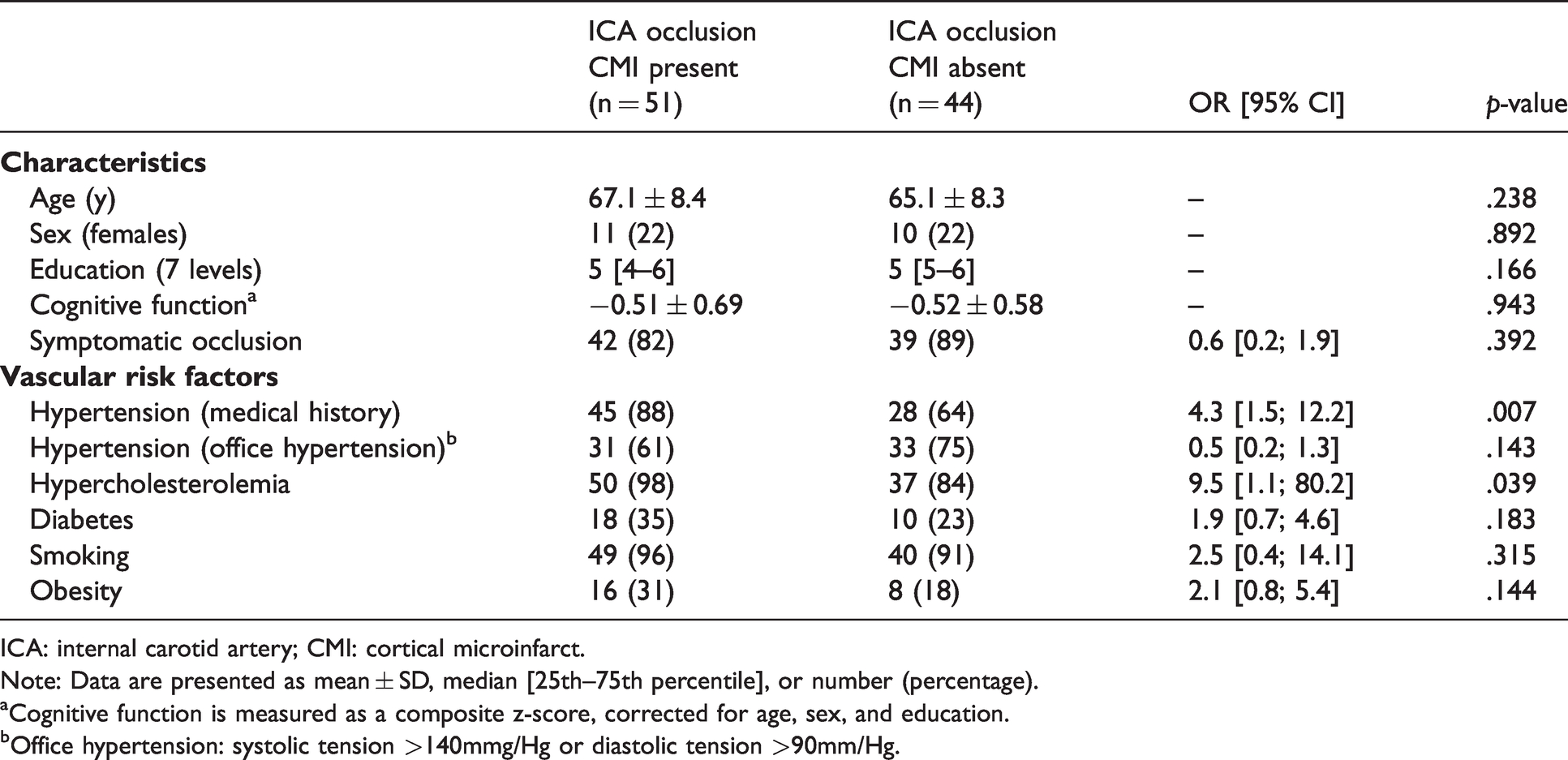
ICA: internal carotid artery; CMI: cortical microinfarct.
Note: Data are presented as mean ± SD, median [25th–75th percentile], or number (percentage).
aCognitive function is measured as a composite z-score, corrected for age, sex, and education.
bOffice hypertension: systolic tension >140mmg/Hg or diastolic tension >90mm/Hg.
In patients with an ICA occlusion, 80% had one or more CMI in the hemisphere ipsilateral to the occlusion and 48% had one or more CMI in the contralateral hemisphere. The number of CMI was significantly higher in the hemisphere ipsilateral to the occlusion (median 2, interquartile range 3) than in the contralateral hemisphere (median 0, interquartile range 1) (p=.001). Figure 1 shows the 3 D distribution of CMI relative to an ICA occlusion. CMI were preferentially located in the cranial part of the frontal and parietal lobes. The vast majority of CMI were located in the perfusion territory of the ICA, with an apparent predilection for watershed areas. The distribution of CMI in the ipsilateral hemisphere was not different from that in the contralateral hemisphere.
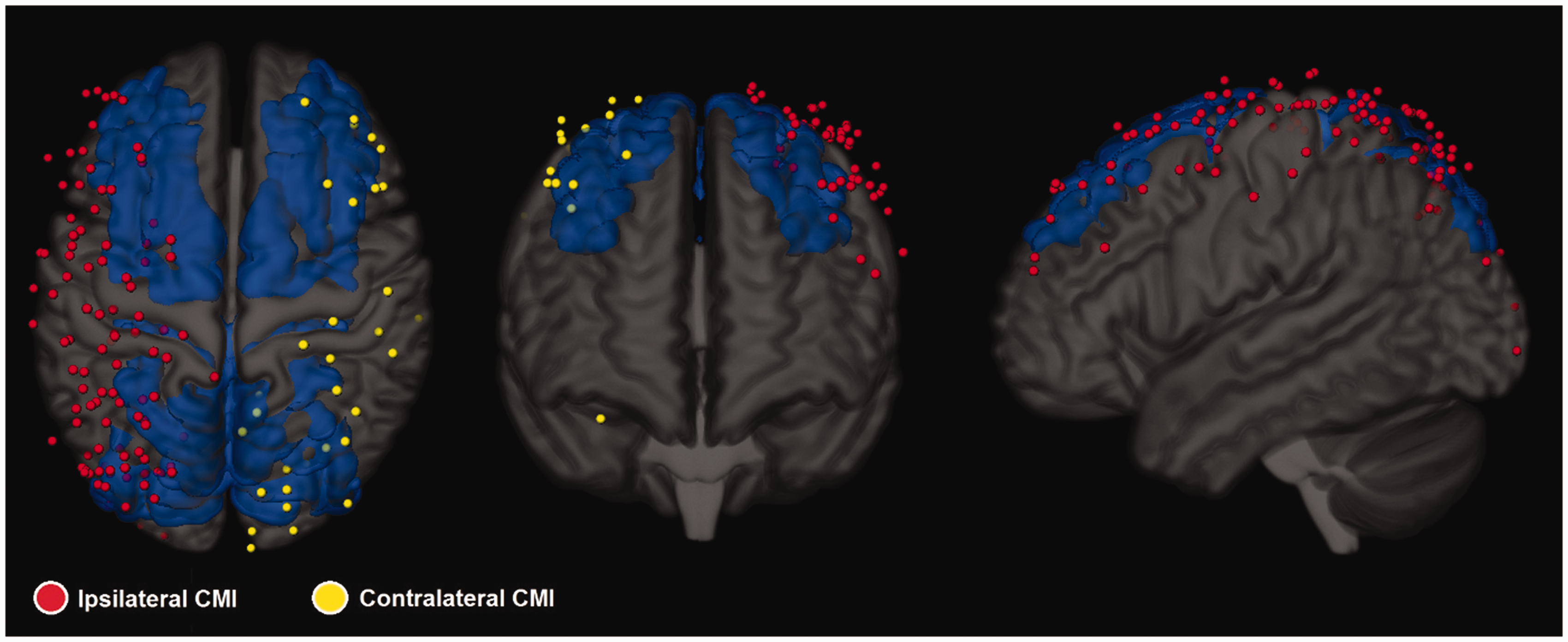
3D distribution across the cortex on an MNI-standard brain of all CMI in 40 patients with a unilateral ICA occlusion and CMI. For patients with a right ICA occlusion the CMI distribution was flipped to create this image where CMI are presented ipsilateral (red) and contralateral (yellow) to the occlusion. An atlas based representation of the watershed areas is included in blue. 9 CMI burden is higher in the hemisphere ipsilateral to the occlusion (p=.001). From visual inspection CMI appear to have a predilection for watershed areas. This distribution was not evaluated by formal statistical testing, because sensitivity of CMI detection may vary by brain region and because the actual watershed region varies between individuals.
By design, all patients had at least one occluded carotid artery. An occlusion of the other carotid was present in 15 (15%) and >50% stenosis in 1 (1%) of the patients. Moreover, an occlusion or >50% stenosis of one or both vertebral arteries was present in 10 (11%) and 19 (20%) respectively. Figure 2 shows that a higher number of occluded or >50% stenosed cervical arteries (i.e. left and right carotid arteries, and left and right vertebral arteries) was associated with CMI presence (p=.038). In patients with a unilateral ICA occlusion, decreased cerebral blood flow also related with CMI presence. Total cerebral blood flow was 12% lower in patients with CMI than in patients without CMI (B -6.2 ml/min/100 ml; 95% CI -12.0:-0.41; p=.036). This was mainly due to lower flow in the contralateral ICA (B -62.3 ml/min; 95% CI -117.5:-7.1; p=.028), rather than the basilar artery (B 13.5 ml/min; 95% CI -30.0:56.8; p=.54), see Figure 3.
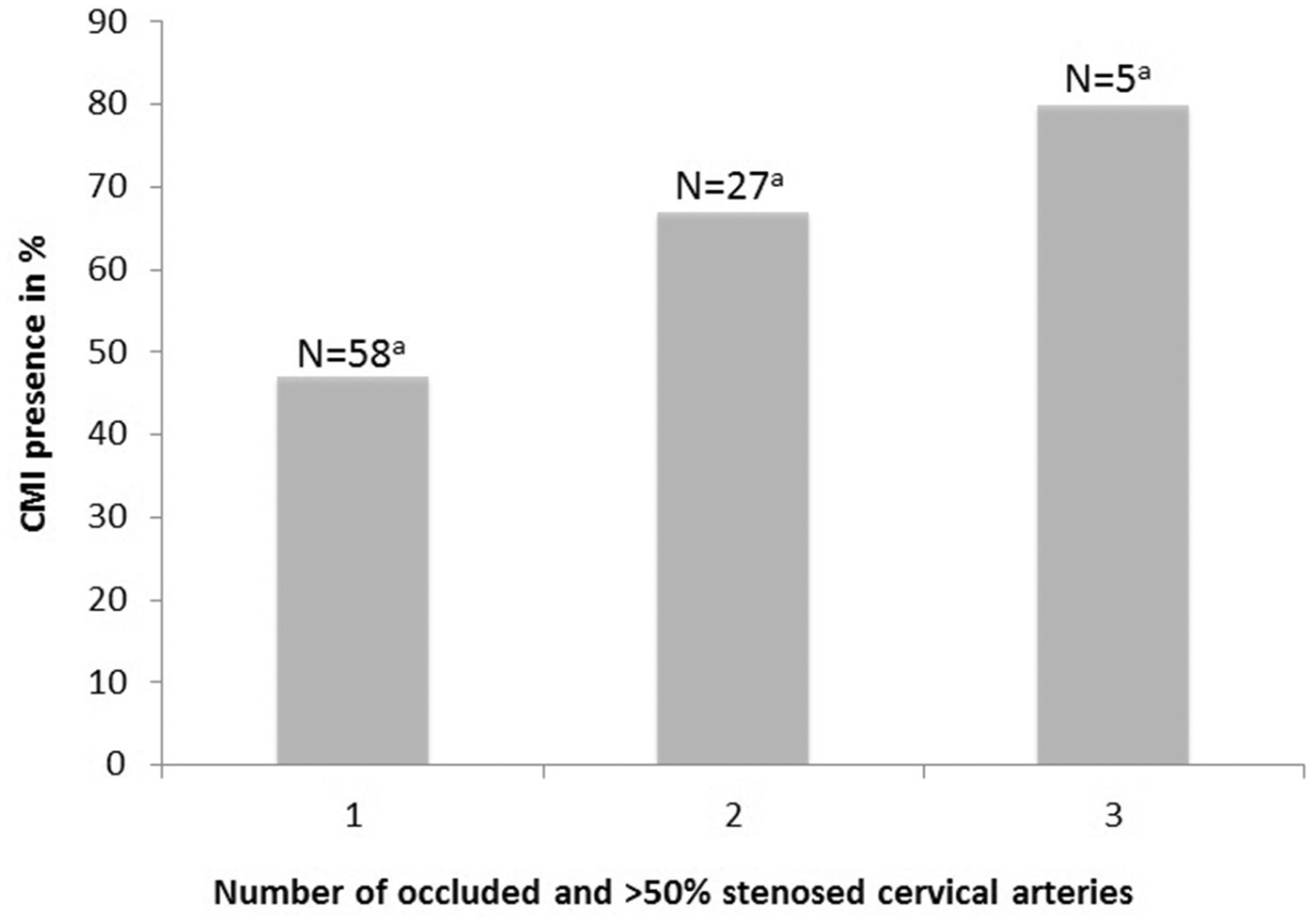
CMI presence in patients with 1-3 occluded or >50% stenosed cervical arteries (i.e. left and right internal carotid arteries and left and right vertebral arteries). None of the patients had occlusions or >50% stenosis in all 4 cervical arteries. aNumber of patients with 1, 2 or 3 occluded or >50% stenosed cervical arteries. N = 90 because of 5 missings.
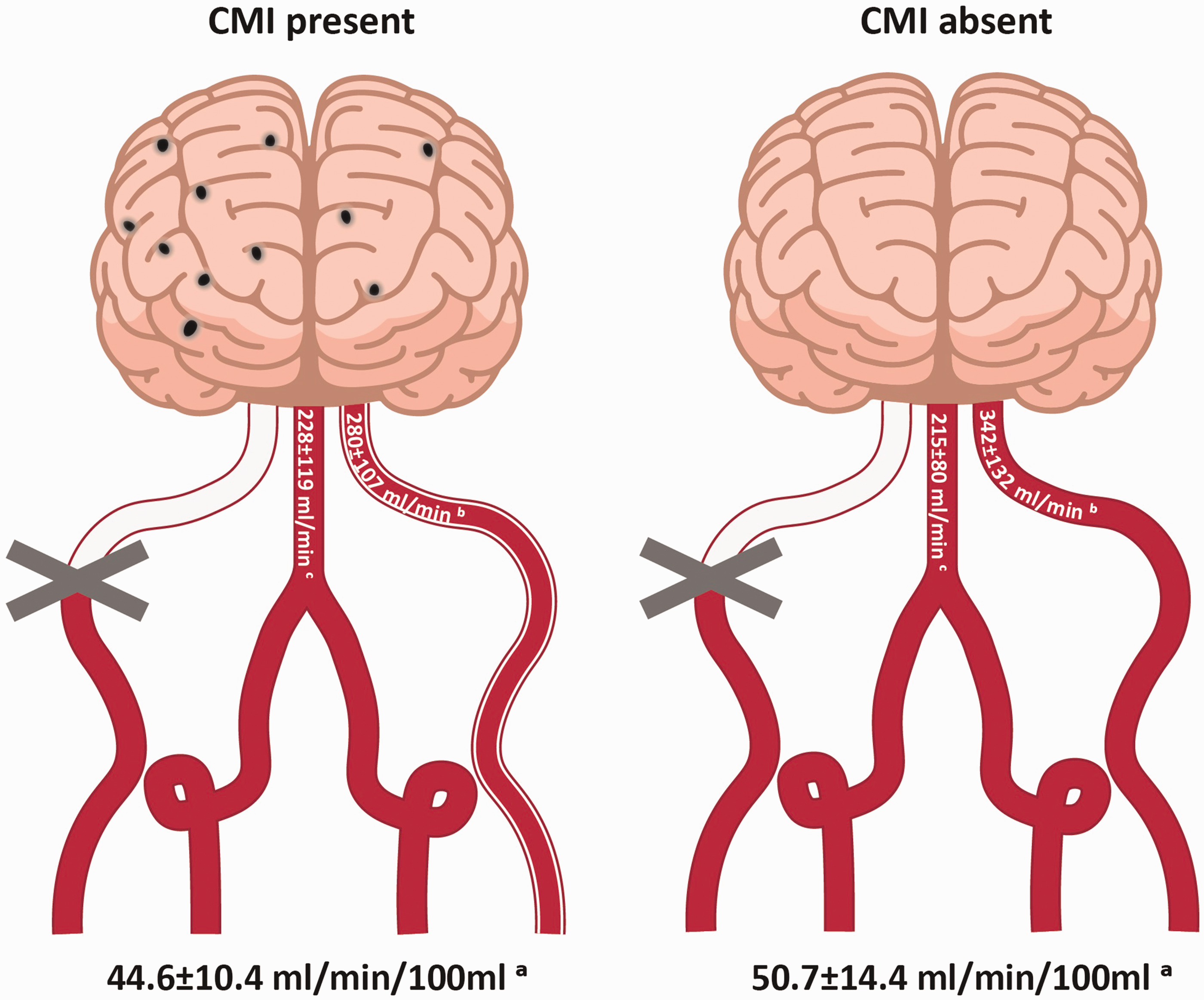
Total cerebral blood flow (mean ± SD, ml/min/100 ml) to the brain and blood flow through the non-occluded ICA and basilar artery (mean ± SD, ml/min). aTotal cerebral blood flow was significantly lower in patients with CMI than in patients without CMI. bThis difference was attributable to a lower flow in the contralateral ICA cnot in the basilar artery. N=75 (CMI present N=38, CMI absent N=37) because of 5 missings and because 15 patients were excluded from this analysis due to a double-sided ICA occlusion.
Table 3 shows the relations between CMI presence in patients with an ICA occlusion and other MRI-markers of cerebral small vessel and large vessel disease. Lacunar infarcts and microbleeds were more common among patients with CMI and SVD burden score was positively associated with CMI presence. There were no significant associations between CMI presence and any of the other MRI markers. Also, there was no correlation between large artery infarcts or lacunar infarcts ipsilateral to the ICA occlusion and ipsilateral CMI (τb = .13; p=.30).
Cerebral MRI-markers in patients with and without CMIs.
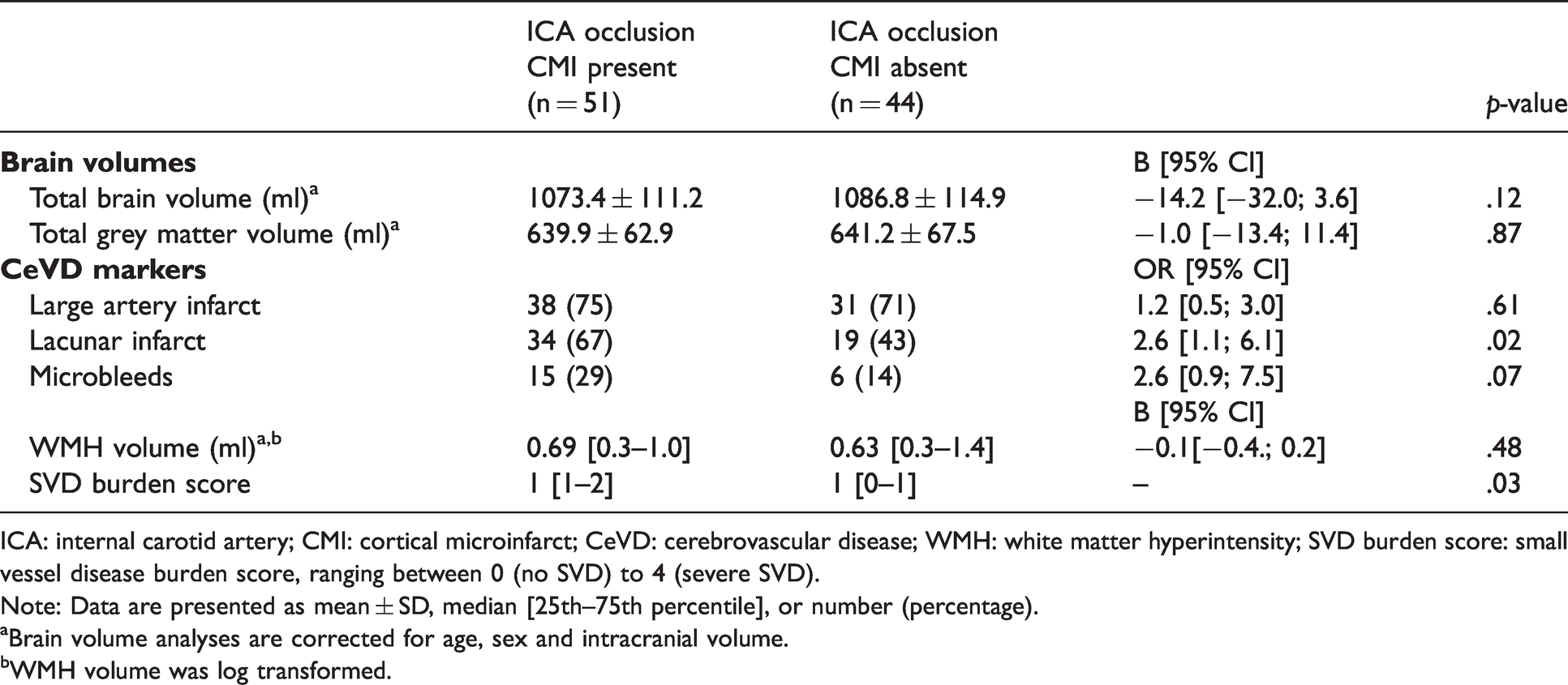
ICA: internal carotid artery; CMI: cortical microinfarct; CeVD: cerebrovascular disease; WMH: white matter hyperintensity; SVD burden score: small vessel disease burden score, ranging between 0 (no SVD) to 4 (severe SVD).
Note: Data are presented as mean ± SD, median [25th–75th percentile], or number (percentage).
aBrain volume analyses are corrected for age, sex and intracranial volume.
bWMH volume was log transformed.
Discussion
CMI are common in patients with an ICA occlusion and occur more frequently in the hemisphere ipsilateral to the occlusion. Within the patient group, the severity of arterial compromise, in terms of number of vessels affected by severe atherosclerosis and blood flow, is related to CMI occurrence, suggesting that hemodynamics indeed play a role in CMI etiology.
To the best of our knowledge this is the first study to examine CMI in patients with a complete ICA occlusion. CMI burden in terms of occurrence and total number is much higher in the patients (54%) than in our reference group and control participants from previous studies (6-12%10–12) Earlier studies in patients with ICA stenosis also reported high CMI prevalence. Specifically, CMI prevalence was 26% in a 3 T MRI study in patients with a > 30% ICA stenosis, 13 and 67% in a 7 T MRI study in patients with a > 50% ICA stenosis. 14 Of note, the prevalence of this last study is not readily comparable due to higher sensitivity of CMI detection on 7 T MRI. 3
We considered patients with an ICA occlusion as a model for hemodynamic compromise. Accordingly, we hypothesized that CMI would be more common in patients with an ICA occlusion than in the reference group, that CMI would be more common in the hemisphere ipsilateral than contralateral to the occlusion, and that the severity of cervical arterial compromise among patients with an ICA occlusion (i.e. worse collateral steno-occlusive disease and worse collateral blood supply) would relate to CMI presence. All these hypotheses were supported by the study data, and there appeared to be a higher occurrence of CMI in watershed areas, which seems to support the notion that worse hemodynamics indeed contribute to CMI etiology. The causal pathway that links hypoperfusion to CMI could be direct, with temporary hypoperfusion directly causing small ischemia in the most distal and smallest vessels. 15 Alternatively, hypoperfusion could be associated with washout disturbances in insufficiently perfused arterioles and/or venules which could lead to embolisms that produce CMI.15,16 Additionally there is evidence for an interaction of hypoperfusion with small vessel disease, specifically cerebral amyloid angiopathy (CAA). A study with a CAA mouse model showed markedly higher CMI burden in CAA mice that were subjected to chronic hypoperfusion than in CAA mice with normal brain perfusion. 17 Yet, additional explanations for the high CMI occurrence in our patient population with high vascular burden need to be considered. Vascular risk factors could contribute to CMI, since we found a relation of CMI presence with hypertension and hypercholesterolemia. However, vascular risk factors cannot explain all our findings, particularly not the within patient between hemisphere differences in CMI occurrence. Moreover, patients with an ICA occlusion are strongly affected by large vessel disease which could give rise to microemboli through different mechanisms. Microemboli could for example have originated from large thrombi that caused ischemic stroke. Of note, we excluded CMI that were located in tissue directly adjacent to large artery infarcts. We did not find that CMI occurred more often in patients that had been symptomatic (versus asymptomatic) and we did not find a relation of CMI with large artery infarcts on MRI, but microemboli could also have originated from pre-occlusive stenosis in earlier disease stages. Indeed it has previously been reported that CMI are related with vulnerable ICA plaques. 13 Yet, this possible mechanism cannot explain that current cerebral blood flow was lower in patients with CMI compared to patients without CMI. We encourage future research to study acute CMI on diffusion weighted imaging in clinically stable patients with an ICA occlusion. If acute CMI would be observed in the hemisphere of the occluded ICA, this would provide further support for hemodynamics as cause of CMI.
The findings of this study may be of clinical relevance to patients with an ICA occlusion, or patients that are in other ways vulnerable to hemodynamic compromise. In other settings, acute CMI have been associated with increased risk of poor clinical outcome after two years, 18 and with (vascular) cognitive impairment. 2 Since patients with an ICA occlusion are known to be at risk of cognitive impairment, 19 the prognostic relevance of CMI in this setting should be topic of future research. Moreover, CMI might be a marker of future stroke risk, even in patients with apparently stable ICA occlusion, but this should be explored in longitudinal studies.
An important strength of this study is the relatively large sample size of patients with an ICA occlusion that were examined in a stable disease phase. Also, due to the high CMI occurrence in the cohort, we had enough power to investigate the relation of CMI with vascular risk factors and measures of collateral compromise. A limitation of our study is that we cannot exclude microemboli from large vessel disease as (partly) cause of CMI in our cohort. We therefore encourage future research to study acute CMI in patients with a clinically stable ICA occlusion. Another limitation of our study is that we studied CMI with 3 T MRI which can only image the largest CMI. Therefore we may have missed smaller CMI that might have a different distribution and pathophysiology. Lastly, we did not measure perfusion at tissue level, but used a measure derived from the main feeding arteries of the brain. We preferred this approach over arterial spin labeling (ASL), because transit time effects in patients with arterial occlusive disease is a well-known source of quantification errors in ASL. 20 Yet, a limitation of our method is that we cannot provide local measures of cortical perfusion and for the analysis presented in figure 3 we cannot take possible collateral flow through the external carotid artery into account.
In conclusion, in this study we explored hypoperfusion as possible cause for CMI, in patients with an ICA occlusion as a model condition of hemodynamic compromise. We found that CMI are common in patients with an ICA occlusion, even more so in the hemisphere of the occluded ICA. Moreover, CMI presence related with the severity of cervical arterial compromise. These findings support a role for hemodynamics in the pathophysiology of CMI.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Netherlands CardioVascular Research Initiative: the Dutch Heart Foundation (CVON 2018-28 & 2012-06 Heart Brain Connection). GJB and HvdB acknowledge support by the Netherlands Organisation for Scientific Research (NWO) (Vici Grant 918.16.616). EEB acknowledges additional support from the Dutch Heart Foundation (PPP Allowance, 2018B011).
Acknowledgements
We want to acknowledge Roos van den Berg for assistance in microinfarct ratings. We acknowledge the contribution of Heart-Brain Study researchers and employees.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Study concept and design: HvdB, DF, LJK, GJB. Acquisition, analysis, or interpretation of data: HvdB, DF, JdB, EEB, LPO. Statistical analysis: HvdB. Drafting of the manuscript: HvdB, DF. Critical revision of the manuscript for important intellectual content: HvdB, DF, JdB, EEB, LPO, LJK, GJB.
