Abstract
Importance:
Individuals with autism spectrum disorder (ASD) and intellectual disability often experience persistent challenges related to aggressive behaviour and agitation, highlighting the critical need for evidence-based pharmacological interventions among other strategies. Despite previous network meta-analyses (NMAs), the rapidly evolving landscape of treatment options necessitates ongoing and updated assessments.
Objective:
To evaluate the efficacy and tolerability of various pharmacotherapies in managing agitation in children and adults with ASD or intellectual disabilities (ID).
Methods:
Employing a systematic review and network meta-analysis methodology, we conducted an exhaustive search across multiple databases for double-blind, randomized controlled trials focusing on pharmacotherapies targeting agitation in these neurodevelopmental disorders. Adhering to Preferred Reporting Items for Systematic Reviews and Meta-analysis guidelines, our assessment of study quality utilized the Cochrane Risk of Bias Tool to ensure methodological rigour and accuracy in data synthesis. Primary outcomes encompassed measures of reduced agitation, as indicated by treatment response on standardized agitation scales, alongside dropout rates, providing a comprehensive overview of treatment efficacy and tolerability.
Results:
Our analysis included data from 38 eligible trials, involving 2503 participants across both pediatric and adult populations. Key pharmacological interventions, such as arbaclofen, risperidone plus buspirone, omega-3 fatty acids, risperidone plus palmitoylethanolamide, aripiprazole and risperidone, demonstrated significant efficacy in reducing agitation compared to placebo. Importantly, these treatments were generally well-tolerated, with no significant increase in all-cause dropouts compared to placebo, highlighting their suitability for clinical use in managing agitation in individuals with ASD or ID.
Conclusions:
This study underscores the efficacy and tolerability of several pharmacotherapies in managing agitation among children and adults with ASD or ID. Our findings provide robust evidence that specific treatments, such as arbaclofen, risperidone plus buspirone and omega-3 fatty acids, are both effective and well-tolerated, offering valuable therapeutic options for clinicians. The study emphasizes the need for ongoing research to ensure that treatment strategies remain aligned with the evolving clinical landscape, ultimately improving patient outcomes in this challenging population.
Keywords
Introduction
Neurodevelopmental disorders (ND) encompass a spectrum of conditions rooted in the neurological system and brain functioning, often emerging early in life and persisting into adulthood, and significantly influencing daily functioning, social interactions and cognitive abilities (European Psychiatric Association, 2015). Autism spectrum disorder (ASD) and intellectual disabilities (ID) are among the most prevalent NDs, characterized by distinct yet interrelated challenges. ASD entails persistent deficits in social communication and interaction, coupled with restricted interests and repetitive behaviours, impacting social relationships and adaptability (American Psychiatric Association, 2013). Conversely, ID denotes significant limitations in intellectual functioning and adaptive behaviour, hindering learning and practical application of knowledge (American Association on Intellectual and Developmental Disabilities, 2020).
One of the hallmark features shared by ASD and ID is the propensity for irritability and emotional dysregulation, often manifesting as tantrums, self-injurious behaviour and aggression (Ageranioti-Bélanger et al., 2012; Fitzpatrick et al., 2016). These behavioural challenges not only impact the affected individuals but also impose substantial stress and strain on their families, caregivers and broader support networks. It is important to distinguish between agitation and irritability, as these are related but not synonymous concepts. Agitation typically refers to a state of increased restlessness and heightened emotional arousal that may lead to aggressive behaviours, while irritability is characterized by a lower threshold for frustration and a predisposition to anger, which may or may not result in aggressive actions. Understanding these distinctions is crucial for accurately assessing and treating these symptoms.
In addressing the complex needs of individuals with ASD and ID, a multimodal approach combining behavioural interventions and pharmacotherapy is often employed (LeClerc and Easley, 2015). While behavioural therapies often aim to teach adaptive skills, coping strategies and social communication techniques, medications are utilized to manage specific symptoms such as aggression, agitation and emotional dysregulation.
Antipsychotic medications, particularly second-generation antipsychotics (SGAs), have garnered significant attention in the treatment of irritability and aggression associated with ASD and ID (Fallah et al., 2019; Park et al., 2016). Compared to first-generation antipsychotics (FGAs), SGAs are generally preferred because they tend to have a better safety profile compared to FGAs. This improved safety includes a reduced risk of extrapyramidal side effects, such as tremors and rigidity, and a lower potential for cognitive impairment, making them more suitable for long-term management of symptoms in individuals with ND (Solmi et al., 2017). For example, the incidence of treatment-emergent extrapyramidal side effects is lower, and there is minor impairment of cognitive function and treatment-related negative symptoms (Solmi et al., 2017). However, SGAs are not without drawbacks, as they are associated with adverse effects such as weight gain, metabolic abnormalities and cardiovascular risks, necessitating careful consideration of risks and benefits in treatment decisions (Solmi et al., 2017).
Despite the growing body of research on pharmacological interventions for ND-related symptoms, the field faces challenges in establishing definitive treatment guidelines and identifying optimal therapeutic strategies (Fallah et al., 2019; Fung et al., 2016; Hirsch and Pringsheim, 2016; Park et al., 2016). The lack of large-scale, head-to-head clinical trials comparing different pharmacotherapies hinders clinicians’ ability to make evidence-based treatment decisions tailored to individual patient needs (Yildiz et al., 2014). In this context, innovative methodologies such as network meta-analysis (NMA) offer promising avenues for synthesizing existing evidence (Cipriani et al., 2018; Miura et al., 2014; Yildiz et al., 2014), comparing multiple treatment options and informing clinical practice guidelines in the management of ND-related symptoms (Salanti, 2012).
Against this backdrop, this study seeks to contribute to the understanding of pharmacotherapeutic interventions for managing agitation in individuals with ASD and ID, with a focus on evaluating their comparative effectiveness and tolerability.
Methods
Protocol and registration
This review was registered with Prospective Register of Systematic Reviews (PROSPERO) (CRD42021258641) and followed the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) guidelines along with the extension for NMAs (Hutton et al., 2015; Liberati et al., 2009). As a secondary analysis of published data, no ethics committee review was necessary.
Eligibility criteria
We employed the populations-interventions-comparators-outcomes-study design framework to define eligibility criteria. Patients with a primary diagnosis of ASD or intellectual disability (ID) were included, based on diagnostic criteria from any edition of the Diagnostic and Statistical Manual of Mental Disorders or the International Classification of Diseases, as long as a validated diagnostic instrument was utilized. Any experimental study measuring the efficacy or safety of antidepressants, antipsychotics, mood-stabilizing agents, psychostimulants, N-methyl-D-aspartate receptor antagonists and other agents administered orally or parenterally for treating aggression or agitation in any age group was considered. Trials comparing medications to non-pharmacologic therapies were excluded.
The term ‘agitation’ encompassed a range of behaviour problems, including irritability, measured using validated instruments such as the Aberrant Behaviour Checklist, Irritability Subscale (ABC-I) (Aman et al., 1985; Schmidt et al., 2013), the Overt Aggression Scale, the Clinical Global Impressions – Improvement (CGI-I) and Severity (CGI-S) scales, the Developmental Behaviour Checklist – Teacher Behaviour Problems Subscale and other equivalent tools. Eligible studies reported at least one measure of efficacy or safety. In the absence of information or supplemental data, we calculated responses using a previously validated imputation method (Furukawa et al., 2005).
Primary outcomes were response (⩾50% reduction in agitation severity) and all-cause treatment discontinuation. Secondary outcomes included discontinuation due to adverse events, adverse events and reduction in irritability severity.
Search strategy
A comprehensive search strategy was developed and executed in multiple databases, including Cochrane Central Register of Controlled Trials, CINAHL, Embase, LILACS, MEDLINE, PsycINFO and PubMed, from inception through 2022 (Appendix 1). Bibliographies of included records and previous reviews were also reviewed.
Study selection
Study selection was performed independently by pairs of investigators using Covidence (Roy Rosenzweig Center for History and New Media, 2018; Veritas Health Innovation, 2019), a web-based systematic review manager. Discrepancies were resolved through consensus.
Data collection
Data abstraction was conducted independently by three reviewers (AB, EF and AN) using a Microsoft Excel Spreadsheet, including variables such as sample size, demographics, intervention details, outcome measures and study characteristics (Table 1).
Description of characteristics of included studies.

The pharmacotherapy acronyms are as follows: AMN: amantadine; ARI: aripiprazole; ASD: autism spectrum disorder; ATM: atomoxetine; BAC: baclofen; BUS: buspirone; CEL: celecoxib; Crossover: a crossover randomized controlled trial; D3: vitamin D3; DMQ: dextromethorphan/quinidine; ELT: eltoprazine; GAL: galantamine; HAL: haloperidol; ID: intellectual disability; LUR: lurasidone; MEM: memantine; NAC: N-acetylcysteine; NIC: nicotine; O3: omega 3 fatty acids; OLZ: olanzapine; Parallel: parallel randomized controlled trial; PEN: pentoxifylline; PLM: palmitoylethanolamide; RIL: riluzole; RIS: risperidone; TOP: topiramate; VPA: valproic acid; ZUC: zuclopenthixol. The agitation/aggression measure acronyms are as follows: ABC-C: Aberrant behaviour checklist; community subscale; ABC-I: Aberrant behaviour checklist; irritability subscale; ABC-SW: Aberrant behaviour checklist; social withdrawal/lethargy subscore; BDA: behavioural disorder assessment; BPI: behaviour problems inventory; CARS: childhood autism rating scale; CCC-2: children’s communication checklist – second edition; CE: clinical evaluation; CGAS: children’s global assessment scale; CGI-I: clinical global impression; improvement scale; CGI-S: clinical global impression – severity; CGI-SC: clinical global impression – symptom change; DAS: disability assessment schedule; GAS: global aggression score; HSQ: home situations questionnaire; MOAS: modified overt aggression scale; NCBRF: Nisonger child behaviour rating form; OAS: overt aggression scale; RBS-R: repetitive behaviour scale – revised; RF-RLRS: Ritvo-Freeman real-life rating scale; SIB-Q: self-injurious behaviour questionnaire; SNAP: Swanson Nolan and Pelham; SRS: social responsiveness scale; VAS: visual analogue scale; YBOCS-C: Yale-Brown obsessive compulsive scale – compulsion subscore; VABS: Vineland adaptive behaviour scale; and DBC-TBPS: developmental behaviour checklist – total behaviour problem score.
Risk of bias assessment
Study quality was assessed independently by three reviewers (AB, EF and AN) using the Cochrane Risk of Bias Tool (Sterne, 2019), which evaluates six domains: random sequence generation (randomization), allocation concealment, blinding of participants and personnel, incomplete outcome data (attrition), selective outcome reporting and other sources of bias. Overall, study-level bias was rated as high if any individual domain scored high or if there were two or more unclear fields. These domains are reported in Table 2. Additionally, we considered allegiance, adherence and attention biases (Jørgensen et al., 2021). Allegiance bias occurs when a treatment’s developer is also a randomized controlled trial
Risk of bias estimates in individual trials (n = 38).

Summary measures
Effect sizes were summarized using Cohen’s d standardized mean differences (SMDs) for continuous data (e.g. symptom severity) and rate ratios (RRs) for dichotomous data (e.g. all-cause treatment discontinuation). SMDs of 0.2, 0.5 and 0.8 were considered small, medium and large effect sizes, respectively (Cohen, 1988). Negative Cohen’s d (SMDs) or RRs < 1 indicated that the treatment reduced the parameter of interest relative to the control group, signifying a beneficial effect (Cohen, 1988; Lipsey and Wilson, 2001).
Planned methods of analysis
NMA is a statistical technique that allows for the comparison of multiple interventions in a single analysis, even when some of the treatments have not been directly compared in head-to-head trials. By integrating both direct and indirect evidence across a network of studies, NMA enables the estimation of the relative efficacy and safety of multiple treatments simultaneously. This method was chosen for our study to comprehensively assess the comparative effectiveness of various pharmacotherapies for managing agitation in individuals with ND. Given the heterogeneity of available studies and the diverse range of pharmacological agents used, NMA is particularly well-suited for synthesizing the evidence and providing a more complete understanding of the relative benefits and risks of each treatment option.
We followed the same analytic approaches of previous NMAs of psychiatric disorders (Bahji et al., 2020a, 2020b, 2020c, 2021a, 2021b; Wong et al., 2020) The netmeta package from R Studio (version 3.5.1, Posit PBC) was used for analysis, employing frequentist random-effects models to preserve randomization (Salanti, 2012) and accommodate different outcome measures across studies (RStudio Team, 2022; Rücker et al., 2019). A jointly randomizable network assumption was made, meaning participants were equally likely to be randomized to any treatments within the network (Cipriani et al., 2018; Huhn et al., 2019; Salanti, 2012; Salanti et al., 2008).
Forest plots were graphed for each outcome measure, with each treatment’s effect size plotted against placebo. League plots were used to visualize head-to-head comparisons by synthesizing direct and indirect evidence. The NMA goodness of fit was assessed through transitivity (measured by τ2 and I2; Borenstein et al., 2010; Higgins et al., 2003) and consistency (evaluated using Cochrane’s Q statistic; Rouse et al., 2017).
The quality of evidence was assessed using GRADE (Guyatt et al., 2008) guidelines, considering the risk of bias, imprecision and heterogeneity. Additional post hoc analyses included P-scores (Rücker and Schwarzer, 2015) for ranking treatments – where higher P-scores indicate a greater likelihood of a treatment being among the most effective. To assess publication bias and inconsistencies in the data, we utilized several graphical and statistical tools, including funnel plots (Sedgwick and Marston, 2015), heat plots (Krahn et al., 2013) and Egger’s test (Egger et al., 1997). Funnel plots were employed to visualize the relationship between study size and effect size; a symmetrical funnel shape suggests no publication bias, while asymmetry may indicate bias. Heat plots were used to detect inconsistencies between direct and indirect evidence in the NMA, with colour intensity indicating the level of inconsistency – lighter colours suggesting consistency and darker colours pointing to potential issues. Additionally, Egger’s test was applied to quantify the asymmetry in the funnel plots, with a significant result indicating potential publication bias. These tools collectively provide a comprehensive assessment of the robustness and reliability of the findings in our meta-analysis.
Use of artificial intelligence
This manuscript was proofread with the assistance of ChatGPT, an AI language model developed by OpenAI, used solely for linguistic and grammatical improvements. All intellectual content, data interpretation and conclusions presented in this manuscript are the sole responsibility of the authors.
Results
Study selection
The study selection process followed the PRISMA guidelines, as depicted in Figure 1. Initially, 323 unique records were identified, with 20 duplicates removed. Following screening, 303 citations were reviewed, resulting in the exclusion of 245 records due to reasons such as ineligible study design, population or intervention. Subsequently, 58 full-text articles underwent thorough evaluation, leading to the exclusion of 27 records primarily due to duplication, inappropriate study design, comparator, outcome measure or issues related to data integrity or availability. Specifically, three studies were excluded due to ineligible data, which refers to instances where the reported data were incomplete, inconsistently reported or not applicable to the predefined outcomes of interest. Additionally, seven records from previous reviews were incorporated, enhancing the breadth of evidence for analysis. Ultimately, 38 studies were deemed suitable for inclusion, providing a comprehensive dataset encompassing 2503 participants (Figure 2).
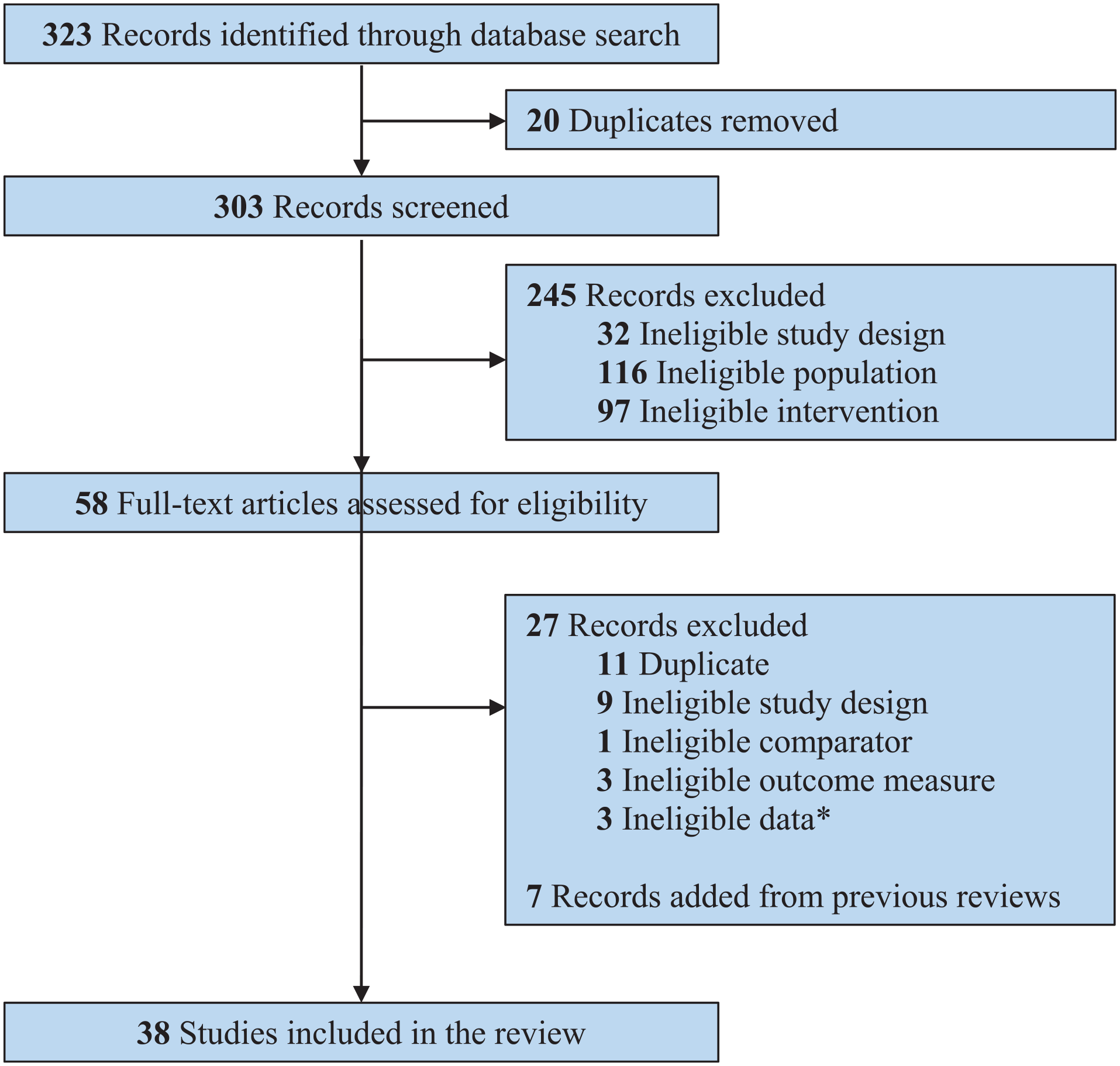
Study selection process using the PRISMA guidelines.
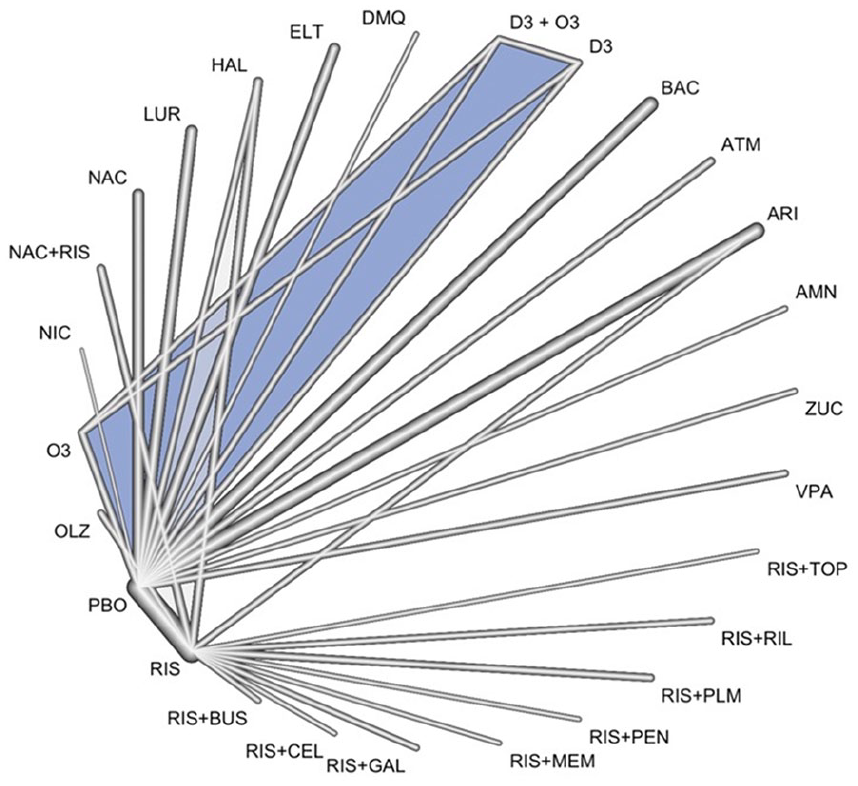
Network plot of eligible pharmacotherapy comparisons across all studies. Line width corresponds to the number of trials comparing pharmacotherapy pairs. The blue-shaded region highlights a specific comparison within the network of interventions.
Study characteristics
The majority of trials were conducted in two primary regions, namely the United States and Iran, comprising 16 and 11 studies, respectively. While these regions represent different healthcare systems and populations, the focus on these areas limits the broader generalizability of the findings to a truly global context. The average sample size across the studies was 66 participants, with notable variability indicated by a standard deviation of 45. This variability underscores the heterogeneity in study designs and participant demographics, reflecting the diverse nature of individuals with NDs and their responses to pharmacotherapy. Among the participants, 1629 received pharmacotherapy as part of an intervention to address agitation, highlighting the common practice of combining multiple interventions to meet the multifaceted needs of individuals with NDs.
The mean age of participants across the studies was 13.6 years, with a standard deviation of 10.8 years, indicating the inclusion of a wide age range from childhood to adolescence and young adulthood. This broad age representation is crucial for understanding the developmental aspects of pharmacotherapy response and long-term treatment outcomes. The median duration of treatment was 10 weeks, ranging from 3 to 112 weeks, indicating the varying lengths of intervention periods evaluated in the studies. This diversity in treatment duration allows for insights into both short-term and potentially longer-term effects of pharmacotherapies on agitation in individuals with NDs.
Risperidone emerged as the most frequently studied pharmacotherapy, with 21 trials evaluating its efficacy and tolerability. This emphasis on risperidone underscores its widespread use and clinical relevance in managing agitation in individuals with NDs. Aripiprazole and baclofen followed as the next most studied drugs, reflecting the interest in exploring alternative pharmacological approaches beyond traditional (dopamine type-2 receptor antagonist) antipsychotics.
The clinical focus of the studies predominantly revolved around individuals with ASD, comprising 31 studies, or ID, encompassing seven trials. This targeted approach ensures a specific focus on populations with distinct neurodevelopmental challenges, enabling tailored insights into the effectiveness of pharmacotherapies for agitation within these particular diagnostic categories.
Synthesis of results
Our analyses highlighted the efficacy of several treatments compared to placebo in the management of agitation in individuals with NDs (Figure 3, Appendix 2).

Forest plots of network meta-analyses for all trials for treatment efficacy (a: response rate; b: aggression severity) and safety (c: dropout rate; d: dropouts because of adverse events (DAE) rates; e: adverse event rate). We compared all pharmacotherapies to placebo (PBO).
Firstly, arbaclofen, risperidone plus buspirone, omega three fatty acids, risperidone plus palmitoylethanolamide, aripiprazole and risperidone demonstrated greater efficacy in reducing agitation compared to placebo, as evidenced by summary RRs for treatment response ranging from 2.76 (95% CI: 1.63–4.66) to 12.10 (1.36–107.89). These results underscore the potential of these pharmacotherapies in effectively managing agitation in individuals with NDs.
Moreover, the P-scores for treatment rankings provided in Appendix 2 corroborate our findings. For example, aripiprazole and risperidone consistently ranked higher in terms of efficacy compared to other treatments, with P-scores of 0.4411 and 0.4503, respectively. This further supports the favourable efficacy profile of these medications in managing agitation.
Additionally, our analysis indicates that none of the investigated therapies exhibited significantly lower tolerability for all-cause dropouts than placebo. Although risperidone plus buspirone showed a higher dropout-due-to-adverse-events rate compared to placebo, this difference was not statistically significant, as indicated by the RR crossing 1 in Figure 3.
In addition to the pediatric-focused studies, our analysis also included several trials conducted with adult populations. The studies by Chez et al. (2020), Hellings et al. (2006), Lewis et al. (2018), McDougle et al. (1998), Singh et al. (1992), de Koning et al. (1994), Tyrer et al. (2009) and Amore et al. (2010) specifically targeted adult participants. A sub-analysis of these adult-focused studies did not indicate any significant differences in treatment efficacy compared to the overall findings, reinforcing the generalizability of our results across age groups.
Upon conducting a sensitivity analysis by restricting the analysis to subjects with ASD only, we found no significant changes in our overall findings. However, due to the limited number of studies focused solely on individuals with ID (n = 7), we were unable to perform a reliable sub-analysis for the ID population, in accordance with Cochrane guidelines.
Overall, our findings suggest that while these treatments may vary in terms of efficacy, they generally maintain comparable tolerability profiles to placebo.
Risk of bias
The risk of bias within the included studies varied across different domains, as outlined in Table 2. Randomization was generally conducted appropriately in most studies, with low risk observed in 32 out of 38 trials (84.2%). However, allocation concealment was deemed to be at high risk in a notable number of studies, with 26 out of 38 trials (68.4%) exhibiting a high risk of bias in this domain. Double blinding was adequately implemented in the majority of trials, with low risk observed in 35 out of 38 studies (92.1%). Attrition bias, reflecting the completeness of outcome data, was generally low, with 36 out of 38 trials (94.7%) showing low risk in this domain. Other potential sources of bias, such as baseline imbalances or selective outcome reporting, were identified in some studies, contributing to an overall assessment of high risk in 15 out of 38 trials (39.5%). These findings indicate a mixed risk of bias across different aspects of study design and conduct within the included trials.
Exploration for intransitivity and inconsistency
Heterogeneity estimates were notably significant only for the reduction in agitation severity, as shown in Table 3, indicating variations in treatment effects among studies for this particular outcome. Conversely, other outcomes such as treatment completion, all-cause discontinuation, discontinuation due to adverse events and adverse events demonstrated non-significant heterogeneity, suggesting a consistent effect across these measures. The Q-between statistic for inconsistency between designs also revealed no significant inconsistencies for most outcomes, except for the reduction in agitation severity, highlighting the importance of cautious result interpretation due to differing treatment effects observed across studies.
Network meta-analysis indices.

k: number of studies; n: number of treatments; m: number of pairwise comparisons;
Consistency estimates, reflecting the agreement between direct and indirect evidence, were acceptable for all primary and secondary outcomes. This indicates that the NMA model effectively captured the underlying relationships between treatments, reinforcing the reliability of the synthesized evidence for most outcome measures. Nevertheless, further scrutiny and validation through additional well-designed studies are necessary, particularly concerning agitation severity reduction, given the observed heterogeneity and inconsistency in treatment effects across studies.
Risk of bias across studies
While no evidence of network publication bias was observed across the outcomes, it was not possible to investigate publication bias for individual agents, as indicated in Table 3.
Discussion
Summary of evidence
This review presents a comprehensive evaluation of the comparative effectiveness of pharmacotherapies in managing agitation within the context of NDs. We identified and analysed 38 trials meeting our eligibility criteria, with a collective sample size of 2503 participants and an average age of 13.6 years. The trials were primarily conducted in the United States and Iran, focusing predominantly on ASD. Risperidone emerged as the most frequently studied drug, followed by aripiprazole and baclofen. Notably, several pharmacotherapies, including arbaclofen, risperidone plus buspirone, omega 3s, risperidone plus palmitoylethanolamide, aripiprazole and risperidone, exhibited superior efficacy compared to placebo, with summary relative risks for treatment response ranging from 2.76 to 12.10.
Additionally, while none of the investigated therapies showed a significant increase in all-cause dropouts compared to placebo, it is important to note that all-cause dropout rates can be influenced by a variety of factors unrelated to the tolerability of the medication itself. Therefore, while the lack of a significant increase in all-cause dropouts suggests a favourable tolerability profile, this measure alone may not fully capture the complexity of treatment adherence and patient experience. In particular, certain medications did show higher dropout rates due to adverse events, which we have acknowledged and discussed in the context of their overall risk–benefit profile for managing agitation in individuals with ID and ASD.
Implications of findings
Behavioural disorders pose substantial challenges for individuals with ID, irrespective of their underlying aetiology. Our review, building upon previous meta-analyses, offers a more comprehensive and updated perspective by including both ASD and ID populations, thus expanding the scope of available evidence. The efficacy of risperidone and aripiprazole in reducing agitation symptoms is consistent with prior research, including reviews by Fallah et al. (2019) and Fung et al. (2016). Yet, our analysis extends this understanding by incorporating a broader range of pharmacotherapies. This expanded repertoire provides clinicians with a more nuanced understanding of available options, enhancing their ability to tailor treatment strategies based on individual patient needs.
Our review sheds light on essential insights regarding pharmacotherapy options for managing agitation in individuals with ID and ASD. Our analysis highlights several pharmacological interventions, such as arbaclofen, risperidone plus buspirone, omega 3s, risperidone plus palmitoylethanolamide, aripiprazole and risperidone, demonstrating greater effectiveness than placebo. These interventions showed summary relative risks for response ranging from 2.76 to 12.10, underscoring their potential clinical utility in effectively addressing agitation while maintaining tolerability. Importantly, none of the investigated therapies resulted in a significant increase in all-cause dropouts compared to placebo, further supporting their favourable profile for managing agitation in individuals with ID and ASD.
The effectiveness of specific pharmacotherapies, such as risperidone plus buspirone and risperidone plus palmitoylethanolamide, may be particularly attributed to their synergistic effects. Risperidone, a well-established antipsychotic, combined with buspirone, an anxiolytic that modulates serotonin receptors, likely provides a dual mechanism that targets both the dopaminergic and serotonergic systems, crucial in managing agitation and anxiety often observed in NDs. Similarly, the combination of risperidone with palmitoylethanolamide, a fatty acid amide involved in neuroprotection and anti-inflammatory processes, may enhance the therapeutic effects of risperidone by addressing neuroinflammation, which has been implicated in ASD and ID.
Moreover, arbaclofen’s effectiveness can be linked to its role as a GABA-B receptor agonist, which helps to reduce excitatory neurotransmission, a common issue in individuals with ASD. This mechanism may explain its utility in reducing agitation. Omega-3 fatty acids, known for their anti-inflammatory and neuroprotective properties, may also contribute to improved behavioural regulation in this population.
Our findings have implications for the duration of treatment in clinical practice, which often extends to months or even years due to the lifelong nature of these disorders. However, prolonged use of SGAs poses risks of long-term side effects such as metabolic syndrome and weight gain, necessitating additional strategies to manage these side effects, such as diet, exercise and additional pharmacotherapies. For instance, in a longitudinal study of aripiprazole over 52 weeks, participants experienced a mean change in body weight of 6.3 kg, challenging the perception that aripiprazole causes less weight gain than risperidone. Similarly, recent research indicates comparable long-term treatment effects of risperidone and aripiprazole on body weight gain.
The use of antipsychotics in the pediatric population requires careful consideration due to potential risks (Samaras et al., 2014), including the elevation of diabetes risk in children with or without ASD. Monitoring side effects, especially in this age group, is paramount. Additionally, the practice of polypharmacy, where patients receive multiple antipsychotic medications concurrently, is not uncommon and can significantly increase the risk of metabolic side effects, including weight gain (Wink et al., 2014), diabetes (Bobo et al., 2013) and life-threatening arrhythmias (Wink et al., 2017). Hence, caution is advised in combining multiple antipsychotics to minimize adverse outcomes.
A recent systematic review and meta-analysis by Groves et al. (2023), published in Lancet Psychiatry, examined the efficacy of both pharmacological and non-pharmacological interventions for managing behaviours that challenge individuals with ID. The study analysed data from 82 RCTs, encompassing 4637 participants, and found that both types of interventions produced small but significant effects in reducing overall challenging behaviours, including self-injury, aggression and irritability. Interestingly, the study reported no significant differences in efficacy between pharmacological and non-pharmacological interventions for any of the behavioural topographies examined. These findings underscore the importance of integrating psychological therapies with pharmacological approaches, as both can play crucial roles in a comprehensive treatment strategy for managing challenging behaviours in individuals with ID and ASD.
Limitations
This study represents the most exhaustive NMA to date for assessing pharmacotherapy targeting agitation symptoms in individuals with NDs. Given the proliferation of single-treatment RCTs and the scarcity of head-to-head RCTs, NMAs offer a distinct approach to synthesizing knowledge with the available data (Eccleston et al., 2014; Haugh et al., 2019). While NMAs are potent tools for comparative effectiveness research, it is crucial to consider several limitations associated with these findings.
In this study, we acknowledge the nuanced differentiation between ‘aggression’ and ‘agitation’ as distinct behavioural constructs. Although our search strategy encompassed both terms, it is important to note that aggression can manifest independently of agitation in individuals with NDs. We have factored in this distinction during our data analysis and interpretation, recognizing potential variations in the presentation of these behaviours across the included studies.
A significant portion of the included trials did not provide detailed information about co-morbidities, and in some cases, co-morbid conditions were explicitly excluded from the trial criteria. Consequently, we were unable to systematically analyse the impact of co-morbidities on medication effects or responses in this study. Future research focusing on explicitly examining the influence of co-morbid mental illnesses on treatment outcomes in individuals with NDs would offer valuable insights into this aspect.
The bulk of evidence in our NMA relied on indirect treatment comparisons, which are more susceptible to bias compared to head-to-head comparisons. Moreover, the limited number of RCTs and small sample sizes resulted in imprecise and potentially underpowered estimates for certain medications. Although we opted to pool studies by medication classification and specific symptom clusters to enhance statistical power, our review’s generalizability to other symptom clusters (e.g., depression, anxiety, psychosis) is limited (Riley et al., 2017; Willis and Riley, 2017). Consequently, there was notable heterogeneity in some outcomes, partly due to the lack of standardized protocols in some medication-based studies, which was an inherent limitation of this review (Thorlund and Mills, 2012).
While the majority of the included studies were conducted in pediatric populations, the inclusion of a few studies focusing on adults allowed for a broader assessment of pharmacotherapy efficacy across age groups. However, due to the limited number of adult-focused studies, further research is warranted to confirm these findings in larger adult cohorts.
Furthermore, the risk of bias in individual RCTs was elevated due to various factors such as inherent challenges with blinding medication trials (Shean, 2014), response bias and social desirability, which could have biased self-reported, subjective measures, especially for agitation and aggression symptoms (Rosenman et al., 2011). Despite an extensive search, we might have missed relevant studies, as evidenced by the presence of publication bias in at least one of our outcomes. Although network-level publication bias was not detected for most other outcomes, individual medications might have been influenced by publication bias, particularly for pharmacotherapies with only one study available. Additionally, therapy-comparator differences might have been mitigated by some comparator conditions’ active, therapeutic nature, further warranting cautious interpretation of our findings. Another limitation of our study is the inability to perform a separate sensitivity analysis for individuals with ID alone, as the number of studies available (n = 7) did not meet the Cochrane-recommended minimum of 10 studies, which precluded a reliable sub-analysis.
Lastly, the limited number of studies measuring longer-term outcomes adds another layer of complexity to our analysis.
Future research
To build upon the insights gained from this study, larger, multicentred Phase III trials are imperative to replicate findings obtained from the small, single-site and underpowered studies. Longitudinal studies are also essential to elucidate the long-term efficacy, treatment response with varying doses, and the side effects associated with these medications. Moreover, conducting head-to-head trials would provide more robust evidence compared to placebo controls. The efficacy of newer antipsychotics such as brexpiprazole and paliperidone remains minimally explored, despite their availability in injectable formulations that allow for convenient dosing, particularly for individuals with swallowing difficulties in NDs.
Seizures are a prevalent issue in NDs, and antiepileptic drugs like valproic acid play a role in managing seizures and reducing irritability. However, our review identified only two studies focusing on the efficacy of valproic acid. Therefore, exploring trials involving other antiepileptics could be beneficial, especially in cases where the side effect profile of antipsychotics is unsuitable or contraindicated.
Efforts to standardize outcome measures and trial durations in future RCTs would facilitate comparisons of findings across studies. Additionally, there is a pressing need for studies that delve deeper into refining our understanding of the pathophysiology of agitation and aggression in NDs and their treatment modalities. Emphasizing functional outcomes over symptom-based assessments and incorporating biological measures may provide more meaningful insights and complement clinical data. Lastly, more research focusing on special populations, such as older adults and individuals with concurrent disorders like anxiety, ADHD, psychosis and substance abuse comorbid with NDs, is warranted.
Conclusions
In conclusion, our review underscores the efficacy of various pharmacotherapies such as arbaclofen, risperidone with buspirone, omega 3s, risperidone with palmitoylethanolamide, aripiprazole and risperidone in managing agitation in NDs. These interventions showed favourable response rates without a significant increase in dropouts compared to placebo. However, the limited strength of evidence for most agents calls for additional high-quality RCTs to establish a definitive treatment hierarchy. Future research should focus on larger multicenter trials, longitudinal studies, and head-to-head comparisons to validate and build upon our findings. Standardizing outcome measures, exploring newer antipsychotics and investigating the role of antiepileptics like valproic acid are also critical areas for further investigation. Addressing these gaps will enhance treatment options and improve outcomes for individuals with NDs.
Supplemental Material
sj-docx-1-jop-10.1177_02698811241303654 – Supplemental material for Navigating agitation in neurodevelopmental disorders: A comparative study of pharmacotherapies via network meta-analysis in children and adults with autism spectrum disorder or intellectual disabilities
Supplemental material, sj-docx-1-jop-10.1177_02698811241303654 for Navigating agitation in neurodevelopmental disorders: A comparative study of pharmacotherapies via network meta-analysis in children and adults with autism spectrum disorder or intellectual disabilities by Anees Bahji, Evan Forth, Amina Nasar, Ahmed Waqas, Emily R Hawken and Muhammad Ayub in Journal of Psychopharmacology
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: I am a co-founder of a research organization in Pakistan. There was no input from the organization about any decisions about this paper. It was planned and written before the organization was founded. Bahji volunteers on professional committees and contributes as an unpaid editor for CJA and a mental health educator for TED-Ed. They do not receive compensation for other activities like royalties, consulting fees or board memberships.
Funding
The author disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Bahji received funding for their doctoral studies from CIHR and the University of Calgary, with additional support from the Calgary Health Trust. They also earn a small honorarium for teaching at the University of Calgary.
Registration
PROSPERO (CRD42021258641).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
