Abstract
Background:
The 9-item Concise Health Risk Tracking – Self-Report (CHRT-SR9) is a widely used patient-reported outcome measure of suicidal risk. The goal of this article is to provide an evidence-based interpretation of the CHRT-SR9 total score in terms of four clinically actionable categories of suicidal risk (none, mild, moderate, and severe).
Methods:
Data from two large programs involving adolescents and adults were combined in this paper. In these studies, the CHRT-SR9 was anchored against an independent measure of suicidal risk, the suicide item (Item #9) in the Patient Health Questionnaire (PHQ-9), with categories 0 (none), 1 (mild), 2 (moderate), and 3 (severe). In the combined data (n = 1945), we calculated the cumulative percentage of data across these four categories and the percentile score of the CHRT-SR9 total score that corresponded to these percentages; from this, we developed ranges of the CHRT-SR9 total score that corresponded to the four categories of Item #9 of PHQ-9. We also calculated similar ranges for two broad subscales of the CHRT-SR9 total score; Propensity and Suicidal Thoughts. To assess the robustness of our findings, we repeated the analysis at another timepoint across studies.
Results:
Findings indicated that the CHRT-SR9 total score (range: 0–36) can be categorized as none (0–14), mild (15–21), moderate (22–26), and severe (27–36). Similar categories were calculated for the Propensity and Suicidal Thoughts subscales. The findings were the same when repeated at another timepoint.
Conclusion:
This categorization of the CHRT-SR9 total score can place patients into clinically meaningful and actionable categories of suicidal risk.
Keywords
Introduction
Over 45,900 persons died by suicide in the United States in 2020, according to the Centers for Disease Control and Prevention (CDC), making it the 12th leading cause of death overall. Between the ages of 10–14 and 25–34, suicide was the second most common cause of death. Between the ages of 15 and 24, it was the third most common cause, and between the ages of 35 and 44, it was the fourth most common (Centers for Disease Control and Prevention, 2020).
There is an immense complexity to suicidal phenomena. Shneidman (1993), a psychologist who founded the nation’s first comprehensive suicide prevention center and revolutionized the study of suicide, characterized suicide as an escape from intolerable mental suffering. He stated that the objective of a therapist is to collaborate with suicidal individuals to identify alternative methods of alleviating their mental distress.
The risk of suicide is a critical clinical parameter in managing patients. The reduction of pain and suffering in patients who have suicidal thoughts or tendencies is a critical aspect of patient management (Pompili, 2024). Clinicians are sometimes required to conduct suicidal risk assessments that are both precise and beneficial, often in emergencies. The single most pressing issue may be the assessment of suicide risk. The Concise Health Risk Tracking – Self-Report (CHRT-SR) has been widely used as a patient-reported outcome measure in research to identify suicidal risk. The psychometric properties of the CHRT-SR of various lengths (16, 14, 12, and 7 items) have been studied in various age groups and diagnostic settings (Mayes et al., 2018; Ostacher et al., 2015; Reilly-Harrington et al., 2016; Sanchez et al., 2018; Trivedi et al., 2011; Trombello et al., 2023). We have settled on the 9-item version (henceforth, CHRT-SR9), as it is user-friendly, can easily fit on a phone application, and has excellent psychometric properties across the age spectrum (Nandy et al., 2023a, 2023b).
The CHRT-SR9 comprises nine self-referent statements of how the respondent felt over the past week. The total score on the CHRT-SR9 is a measure of overall suicidal risk, with a higher score indicating a greater risk of suicide. To make the CHRT-SR9 widely applicable in the real world, it is important to provide clinically meaningful interpretations of the CHRT-SR9 total score. In other words, in terms of the suicidal severity shown in the scale total, what might be considered “none,” “mild,” “moderate,” and “severe” suicidal risk?
This report synthesized data from two substantial patient populations, in which the 9-item Patient Health Questionnaire (PHQ-9) and the CHRT-SR9 were administered concurrently. The objective was to determine the risk associated with different thresholds on the CHRT-SR9 total score, as well as the two associated subscales of the CHRT-SR9 (“Propensity” and “Suicidal thoughts”), juxtaposed against the suicide item (Item #9) from the PHQ-9. We sought to establish clinically actionable categories of risk that were evaluated based on the frequency of suicidal thoughts.
Methods
Data from two large programs, comprising three cohorts with data from two timepoints, were combined in this paper. These programs and the corresponding cohorts are described below briefly.
VitalSign6
The University of Texas Southwestern Medical Center and primary care and specialized care clinics in Texas collaborated on the VitalSign6 quality improvement initiative to expand access to evidence-based depression screening and treatment (Trivedi et al., 2019). The VitalSign6 program gathers demographic information and data from self-report forms and clinician evaluations and then uses treatment algorithms to provide measurement-based care recommendations. The initiative began in 2014 and is still underway. The VitalSign6 app has been used in 37 clinics around Texas to screen over 70,000 patients for depression.
Patients evaluated through VitalSign6 include adolescents and adults. For these analyses, we divided the sample into two cohorts based on age. Psychometric analyses of the CHRT-SR9 have been previously published with these samples. The first cohort (Nandy et al., 2023a) included 657 adolescents (<18 years of age), with most from mental/behavioral health settings (n = 592) and some from primary care clinics (n = 65), who screened positive for depression at study entry. From this cohort, our final sample consisted of 648 adolescents with PHQ-9 and CHRT-SR9 data at an initial clinic visit and 647 with complete data at a consecutive visit. The second cohort (Nandy et al., 2023b) involved 369 adults (>18 years of age), mostly from primary care clinics, who screened positive for depression at study entry. From this cohort, 367 adolescents provided data on both the PHQ-9 and the CHRT-SR9 at 2 consecutive clinic visits and were thus included in the analysis. We used data from both cohorts and timepoints in the analyses for this paper. VitalSign6 is a quality improvement initiative and thus the University of Texas Southwestern Medical Center Institutional Review Board (IRB) approved data collection from the above-referenced studies with a waiver of the need to obtain informed consent.
Texas Youth Depression and Suicide Research Network
The Texas Youth Depression and Suicide Research Network (TX-YDSRN) was established in August 2020 and spans the state’s 12 academic medical institutes (Trivedi et al., 2023). The network supports a registry of youth (ages 8–20 years) who screen positive for depression or suicidal ideation/behavior or who are currently receiving care for depression at participating clinics. Psychometrics of the CHRT-SR9 were previously published using the adolescent sample, ages 12–20 (n = 933), from TX-YDSRN, who completed their baseline visit by June 2021 (Nandy et al., 2023c). Of this sample, 930 youth had complete PHQ-9 and CHRT-SR9 data from an initial clinic visit, and 787 had complete data at a consecutive visit; these participants were included as the third cohort in this analysis. The University of Texas Southwestern Medical Center IRB served as a single IRB of record for all study Nodes; Node IRBs also reviewed and approved the study.
Measures
CHRT-SR9: Suicidal risk was measured by the CHRT-SR9 (Nandy et al., 2023a, 2023b). The scale consists of four subscales: pessimism, helplessness, despair, and suicidal thoughts. Each item was measured on a 5-point Likert scale, from strongly disagree (0) to strongly agree (4), with higher ratings indicating greater risk. The items were constructed as self-referent statements. The total score ranges from 0 to 36, with higher scores indicative of a greater degree of behavior and cognitions associated with elevated suicide risk.
PHQ-9: The PHQ-9 (Kroenke et al., 2001) and the Patient Health Questionnaire for Adolescents (PHQ-A) (Johnson et al., 2002) are both nine-item, self-report inventories including all nine criterion symptoms that define a major depressive episode. The frequency of each symptom is rated 0 (“Not at all”), 1 (“Several days”), 2 (“More than half the days”), and 3 (“Nearly every day”) for the prior 2 weeks. Higher scores indicate greater depression severity. Item #9 relates to suicidal ideation and is identical in the PHQ-9 and PHQ-A. Adults in VitalSign6 completed the PHQ-9; adolescents in both VitalSign6 and TX-YDSRN completed the PHQ-A. Henceforth, we refer to Item #9 of both versions as Item #9 of the PHQ-9.
Statistical data analysis
Data from the three cohorts were used to calculate the cumulative percentage of the combined data across the four categories of Item #9 of the PHQ-9. Next, we calculated the percentile scores on the CHRT-SR9 total score that corresponded to these cumulative percentages. These gave us the ranges for the CHRT-SR9 total score that corresponded to the four categories representing 0 (“Not at all”), 1 (“Several days”), 2 (“More than half the days”), and 3 (“Nearly every day”).
We repeated this analysis for the two subscales, propensity (items 1–6 of CHRT-SR9) and suicidal thoughts (items 7–9 of CHRT-SR9), to obtain ranges for each that correspond to 0–3 of the PHQ-9.
To assess the robustness of the ranges, we repeated the analysis at a second timepoint across all three cohorts. For the VitalSign6 program, the second timepoint was within 3 months of the first visit, while for the TX-YDSRN study, it was 1 month after the first clinic visit.
Results
After combining the data from the first clinic visit from the three cohorts, we calculated the percentile score on the CHRT-SR9 total score that corresponded to the cumulative frequencies of the four response categories on the PHQ-9 Item #9 (Table 1). For example, 47.15% of the combined sample had a score of 0 on PHQ-9 Item #9. The 47.15th percentile on the distribution of the CHRT-SR9 total score was 14. So, a score of 0 on PHQ-9 Item #9 corresponded to a CHRT-SR9 total score in the range of 0–14 in the combined sample. Similarly, a CHRT-SR9 total score in the range of 15–21 corresponded to a score of 1 on PHQ-9 Item #9 in the combined sample; 22–26 corresponds to a score of 2 on PHQ-9 Item 9 and 27–36 corresponds to a score of 3 on PHQ-9 Item 9. For the Propensity subscale (range: 0–24), the ranges were as follows: none: 0–11; mild: 12–16; moderate: 17–20; and severe: 21–24. For the Suicidal Thoughts Subscale score (range: 0–12), the ranges were none: 0–3; mild: 4–6; moderate: 7–8; and severe: 9–12. Details are shown in Table 1.
Benchmarking the CHRT-SR9 total score for suicidal risk by anchoring it against the PHQ-9 Item #9 by synthesis across three large studies at Time 1 (n = 1945).
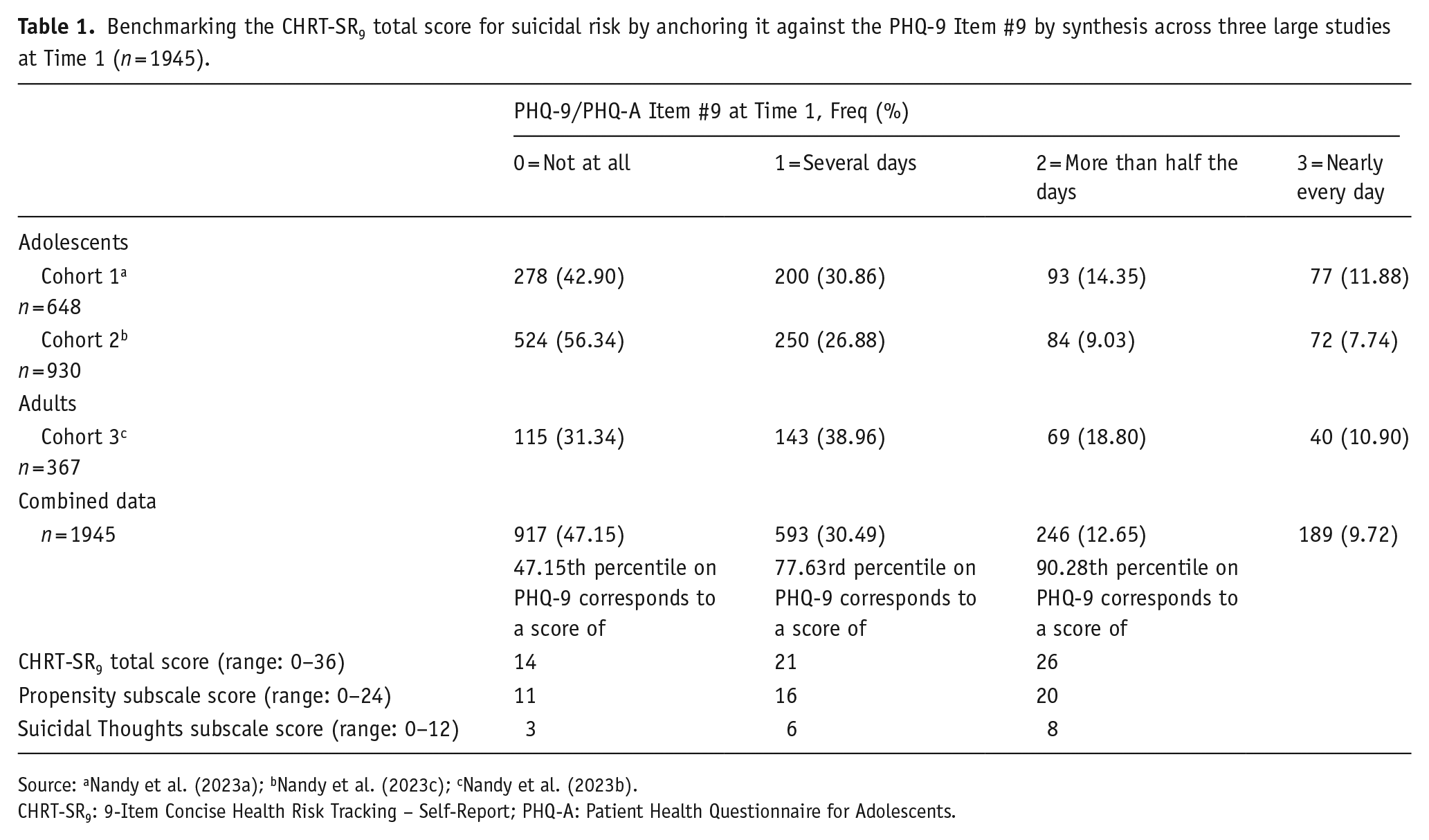
Source: aNandy et al. (2023a); bNandy et al. (2023c); cNandy et al. (2023b).
CHRT-SR9: 9-Item Concise Health Risk Tracking – Self-Report; PHQ-A: Patient Health Questionnaire for Adolescents.
We replicated the analysis at the second timepoint to assess the stability of these ranges for the CHRT-SR9 scores (Table 2). Results from the second timepoint were similar: none: 0–13; mild: 14–21; moderate: 22–26; and severe: 27–36. Because they are essentially the same, we concluded that none (0–14), mild (15–21), moderate (22–26), and severe (27–36) were the four clinically actionable categories of suicidal risk. Results for Propensity and Suicidal Thoughts were also consistent at the second timepoint.
Benchmarking the CHRT-SR9 total score for suicidal risk by anchoring it against the PHQ-9 item #9 by synthesis across three large studies at Time 2 (n = 1801).
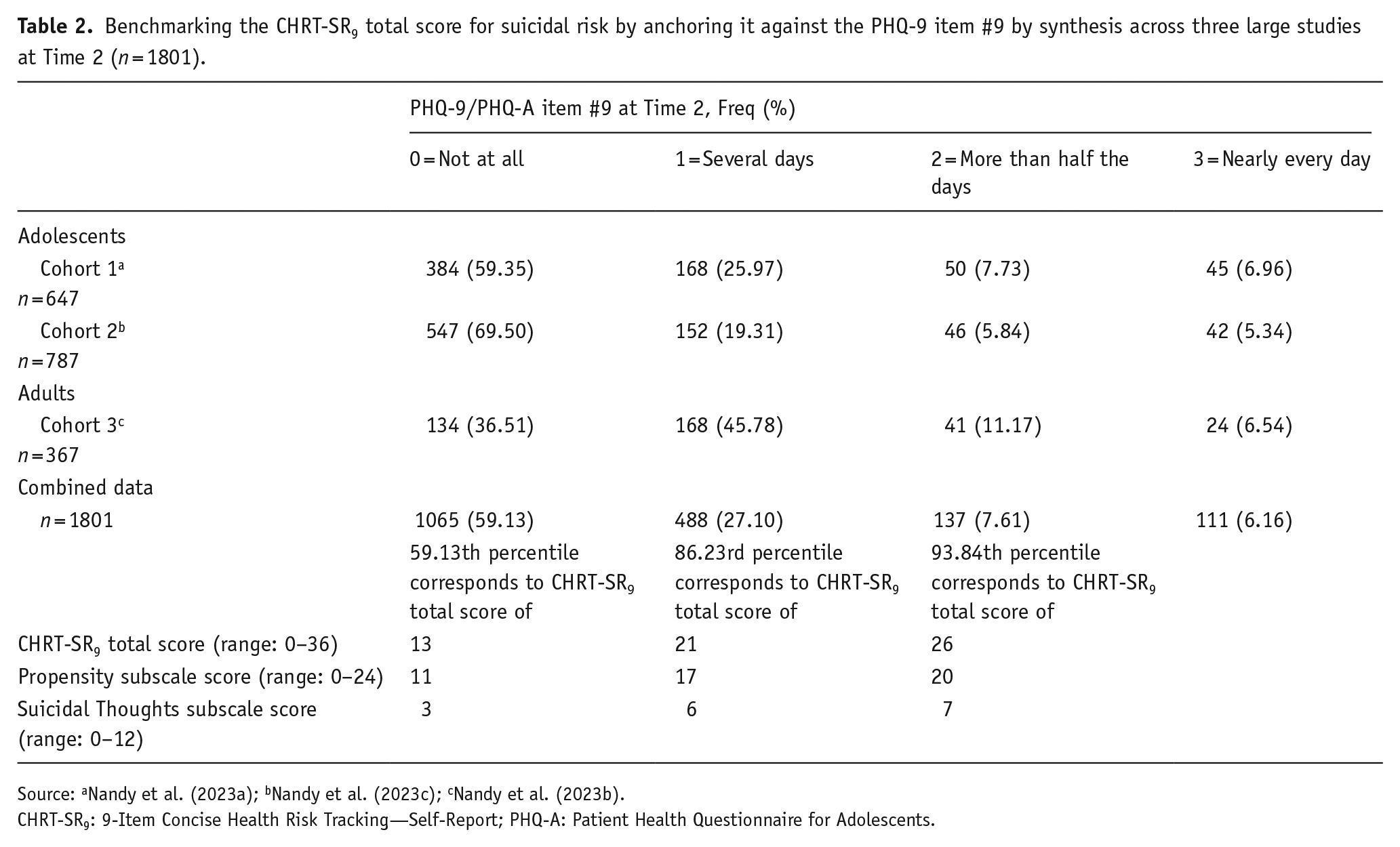
Source: aNandy et al. (2023a); bNandy et al. (2023c); cNandy et al. (2023b).
CHRT-SR9: 9-Item Concise Health Risk Tracking—Self-Report; PHQ-A: Patient Health Questionnaire for Adolescents.
Discussion
The care of potentially suicidal patients can be facilitated by estimating risk and implementing services based on those estimates. The goal of this study was to propose cutoff values for patients’ suicidal risk assessment, as measured by the CHRT-SR9, by leveraging data from multiple large-scale studies. Cutoff scores thus obtained allow for the categorization of risk in terms of clinical relevance such as mild (i.e. monitor but likely does not need clinical intervention) versus severe (i.e. needs immediate clinical attention and/or intervention).
The CHRT-SR9 has already been evaluated as a tool for the prediction of suicidal attempts (Mayes et al., 2023). It can identify severely suicidal adolescents who will experience an event or attempt, as well as capture critical parameters that are associated with subsequent suicidal events or attempts in adolescents. Given the growing popularity of the CHRT-SR9 as a patient-reported, continuous measure of suicidal risk, it will be beneficial to understand the likelihood of a significant or subthreshold risk at various cut points.
Item 9 of the PHQ-9 is widely considered to be an indicator of suicidal ideation. In fact, a large 2013 study (Simon et al., 2013) found that the response to Item 9 of the PHQ-9 identified outpatients who were at an elevated risk of suicide attempt or death. A 2016 study (Louzon et al., 2016) found that patients in the Veterans Health Administration system were at an increased risk of suicide when they exhibited higher levels of suicidal ideation, as indicated by Item 9 of the PHQ-9. This substantiated our decision to benchmark the CHRT-SR9 total score to Item 9 of the PHQ-9 to categorize suicidal risk into four broad categories.
We reported clinically actionable categories in terms of CHRT-SR9 total score. The lower limits of mild, moderate, and severe suicidal risk are determined by the cumulative scores of 15, 22, and 27 on the CHRT-SR9. Particularly, scores below 15 are uncommon in individuals who are at risk of suicide, while scores exceeding 27 typically indicate the presence of significant suicidal risk. Cutoffs for CHRT-SR9 total score thresholds at the second timepoint were the same as those from the first timepoint, save a 1-point shift in the “none” category. The stable findings provided confidence in the categories of suicidal risk hereby proposed.
Limitations
The work was based on three cohorts of depressed outpatients. Its generalizability beyond major depressive disorder remains to be determined. Only outpatients contributed to the thresholds, so the thresholds for the more severely ill are undetermined.
Conclusion
This work is the first to benchmark the CHRT-SR9 total score into clinically meaningful actionable categories of suicidal risk. This gives clinicians a means to estimate a patient’s suicidal risk by placing them into categories that can inform clinical decision-making.
Footnotes
Acknowledgements
The authors would like to thank the patients, clinics, staff, and colleagues who made this project possible. We acknowledge the TX-YDSRN teams from the following sites: University of Texas Southwestern Medical Center: Graham J. Emslie, Betsy D. Kennard, and Laura Stone; Baylor Medical Center: Gabrielle Armstrong, Emily Bivins, Kendall Drummond, and Eric Storch; Texas A&M University System Health Science Center: Tri Le and Olga Raevskaya; Texas Tech University Health Science Center Lubbock: Chanaka Kahathaduwa, Robyn Richmond, Anuththara Lokubandara, and Victoria Johnson; Texas Tech University Health Science Center El Paso: Sarah L. Martin, Zuber Mulla, Alejandro Fornelli, and Caitlin Chanoi; University of Texas at Austin Dell Medical School: Ryan Brown, Tyler Wilson, Caroline Lee, and Stephanie Noble Hernandez; University of Texas Health San Antonio: Norma Balli-Borrero, Sofia Ballesteros, Abigail Cuellar, and Presley Pargin; University of Texas Rio Grande Valley: Cynthia Garza, Diana Chapa, Catherine Hernandez, and Alessandra Rizzo-Esposito; University of Texas Health Science Center Houston: Cesar Soutullo, Jair C. Soares, Giovana B. Zunta-Soares, and Sofia Galarza Estrella; University of Texas Health Science Center Tyler: Jamon Blood, Kiley Schneider, Pamela Thurman, and Kale Boesel; University of Texas Medical Branch: Michaella Petrosky, Layla Kratovic, and Akila Gopalkrishnan; University of North Texas Health Science Center: Cynthia Claassen, David Farmer, Summer Ladd, and Amanda Rosenberg.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.J.R. has received consulting fees from Compass Inc., Curbstone Consultant LLC, Emmes Corp., Evecxia Therapeutics, Inc., Holmusk Technologies, Inc., ICON, PLC, Johnson and Johnson (Janssen), Liva-Nova, MindStreet, Inc., Neurocrine Biosciences Inc., Otsuka-US; speaking fees from Liva-Nova, Johnson and Johnson (Janssen); and royalties from Wolters Kluwer Health, Guilford Press, and the University of Texas Southwestern Medical Center, Dallas, TX (for the Inventory of Depressive Symptoms and its derivatives). He is also named co-inventor on two patents: U.S. Patent No. 7,795,033: Methods to Predict the Outcome of Treatment with Antidepressant Medication, Inventors: F. J. McMahon, G. Laje, H. Manji, A. J. Rush, S. Paddock, A. S. Wilson; and U.S. Patent No. 7,906,283: Methods to Identify Patients at Risk of Developing Adverse Events During Treatment with Antidepressant Medication, Inventors: F. J. McMahon, G. Laje, H. Manji, A. J. Rush, S. Paddock. M.H.T. has provided consulting services to Acadia Pharmaceuticals, Alkermes Inc., Alto Neuroscience Inc., Axsome Therapeutics, Biogen MA Inc., Cerebral Inc., Circular Genomics Inc., Compass Pathfinder Limited, GH Research, GreenLight VitalSign6 Inc., Heading Health, Janssen Pharmaceutical, Legion Health, Merck Sharp & Dohme Corp., Mind Medicine Inc., Myriad Neuroscience, Naki Health Ltd, Navitor, Neurocrine Biosciences Inc., Noema Pharma AG, Orexo US Inc., Otsuka America Pharmaceutical Inc., Perception Neuroscience Holdings, Pharmerit International, Policy Analysis Inc., Praxis Precision Medicines Inc., PureTech LYT Inc., Relmada Therapeutics Inc., Rexahn Pharmaceuticals, Inc., SAGE Therapeutics, Signant Health, Sparian Biosciences, Titan Pharmaceuticals, Takeda Pharmaceuticals Inc., WebMD. He has received grant/research funding from NIMH, NIDA, NCATS, American Foundation for Suicide Prevention, Patient-Centered Outcomes Research Institute (PCORI), and Blue Cross Blue Shield of Texas. Additionally, he has received editorial compensation from Engage Health Media, and Oxford University Press. K.N., R.R.N., and T.L.M. declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was funded by the Texas Youth Depression and Suicide Research Network (TX-YDSRN), a research initiative of the Texas Child Mental Health Care Consortium (TCMHCC). The TCMHCC was created by the 86th Texas Legislature and, in part, funds multi-institutional research to improve mental health care for children and adolescents in Texas. The TX-YDSRN is implemented under the leadership of the central UT Southwestern Hub (Madhukar Trivedi, M.D., Principal Investigator; Sarah Wakefield, M.D., Medical Director (Texas Tech University Health Science Center Lubbock), Abu Minhajuddin, PhD, Data/Statistics Lead, Lynnel Goodman, PhD, Scientific Lead, and Holli Slater, PhD, Operations Lead). The development of the VitalSign6 program was funded in part by the Center for Depression Research and Clinical Care (CDRC) at UT Southwestern, The Rees-Jones Foundation, and the Meadows Foundation. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding organizations. The University of Texas Southwestern Medical Center holds a copyright on the CHRT. At the time of publishing, the CHRT is available without charge to non-commercial users. Fees may apply to commercial users, IT companies, funded academic users, or healthcare organizations. Requests for information and licensing of the CHRT should be sent to M.H.T. (
Disclaimer
The Intellectual Property of VitalSign6 belongs to the University of Texas Southwestern Medical Center (Principal Investigator, Dr. M.H.T.) and is now licensed to GLVS6 for future distribution.
