Abstract
Background:
Chronic breathlessness adversely impacts people with chronic obstructive pulmonary disease and their caregivers (family and friends), who may, in turn, experience significant burden due to their caregiving role. Sustained-release morphine may reduce chronic breathlessness in some patients, which may have an impact on caregivers’ perceived burden.
Aim:
To explore the impact on caregiver burden of active treatment of people with chronic breathlessness (modified Medical Research Council (mMRC) ⩾ 3) and chronic obstructive pulmonary disease (COPD) with regular, low-dose, sustained-release morphine within a multi-site, double-blind, randomised, placebo-controlled trial.
Design:
Exploratory analysis of self-reported caregiver burden at baseline and end of week 3 in a randomised, double-blind, placebo-controlled study. Caregiver measures included: demographics and perceived burden (Zarit Burden Interview 12-item short-form questionnaire). Patient measures included: worst breathlessness and FitBitR-measures.
Setting/participants:
All consenting caregivers of trial patient participants in a multi-site study recruiting from palliative care and respiratory services.
Results:
Caregivers (n = 49; 59% women; median age 68 years [IQR 50–75]) reported median baseline caregiver burden 12 [IQR 5–17], with 53% reporting high burden (⩾13). Eighty-four percent of caregivers reported no change in burden. In people whose worst breathlessness improved, caregiver burden moved in the same direction, though the correlation was not significant (rs = 0.25, p = 0.17). Conversely, caregiver burden worsened as patients’ minutes lightly active increased, with the correlation being significant (rs = 0.56, p = 0.04).
Conclusions:
Caregivers reported high levels of caregiver burden, but patients’ response to treatment in terms of their symptom and function may influence change in caregiver burden over a three-week period.
Keywords
Key statements
Chronic breathlessness is a debilitating presentation for the individuals living with it and their caregivers.
Regular, low-dose, sustained-release morphine may be effective in reducing the symptom of chronic breathlessness (modified Medical Research Council (mMRC) ⩾ 3) in people with chronic obstructive pulmonary disease (COPD).
This study explored the impact of active treatment on caregiver burden in a multi-site, double-blind, randomised, placebo-controlled trial of regular, low-dose, sustained-release morphine for people with chronic breathlessness (mMRC ⩾ 3) and COPD.
The study found that caregivers of people with chronic breathlessness report high levels of caregiver burden that remains unchanged over time. In addition, patients’ changes in breathlessness and function may influence caregivers’ perceived burden.
Clinicians should routinely ask caregivers about any burden they may be experiencing while caring for a person with chronic breathlessness. This would help to ascertain the breadth of impacts of the symptom, including any unmet caregiver needs that might arise from increased burden, and help provide more optimal caregiver support.
The inter-relationship between patients’ breathlessness and function and caregivers’ burden highlights the importance of conducting caregiver assessments in palliative care clinical trials to help inform a more effective, evidence-based practice.
Delineating the precise nature of caregiver burden in the context of chronic breathlessness is also needed as the burden is likely to be multifaceted.
Introduction
Chronic breathlessness is a debilitating syndrome,1,2 with chronic obstructive pulmonary disease (COPD) being the most prevalent underlying condition. Severe breathlessness detrimentally affects people’s day-to-day functioning and is associated with increased anxiety, depression and reliance on others. 3 These challenges negatively impact caregivers (unpaid family or friends providing care and support) 4 who experience significant burden and associated poorer mental and physical health.5,6 Reducing the burden imposed by the symptom is important for optimising caregivers’ physical and emotional health, which may benefit patients, too.
Regular, low-dose, sustained-release (SR) morphine may reduce chronic breathlessness and severe functional impairment due to breathlessness (modified Medical Research Council (mMRC)
7
The aim was to explore the impact on caregiver burden of active treatment of people with chronic breathlessness (mMRC ⩾ 3) and COPD with regular, low-dose, SR morphine within a multi-site, double-blind, randomised, placebo-controlled trial.9,13
Methods
This was an exploratory analysis of prospectively collected data on self-reported caregiver burden at baseline and end of week three (BEAMS trial).9,13 Patient participants were randomised at baseline to different treatment trajectories.9,13 Baseline allocations followed a 1:1:1 ratio (placebo or SR morphine 8 or 16 mg); weeks 2 and 3 followed a 1:1 ratio (8 mg added to the previous week’s dose or placebo). Patient participants, caregivers and clinicians were blinded to the intervention. The primary outcome was change in worst breathlessness intensity in the previous 24 h. Details have been presented elsewhere.9,13 Caregivers were recruited through trial patient participants who identified the person who knew them best and could help understand any changes that may be seen as a result of the study.14,15 All consenting caregivers were eligible to participate.
Ethics approval for the trial was obtained from the Hunter New England Human Research Ethics Committee (Reference No. 15/12/16/3.06). The reporting follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Self-reported caregiver data encompassed demographic information and perceived burden. Caregiver burden was assessed using the Zarit Burden Interview 12-item (ZBI-12) short-form questionnaire, with answers provided on five-point Likert scales: 0 (never) to 4 (nearly always). 16,17 Scores range from 0 to 48; a total score of ⩾13 reflects higher burden. 18 Caregiver burden was assessed at baseline (W0), week 1 (W1) and week 3 (W3). Demographic and ZBI-12 questionnaires were provided to caregivers during trial patient participants’ scheduled visits.
Patient measures included: intensity of worst breathlessness in the previous 24 h measured on a 0–10 Numerical Rating Scale (NRS) anchored between 0 (no breathlessness) and 10 (worst possible breathlessness); and FitBitR-measures of physical activity. Active minutes were based on a person’s resting metabolic rate (MET) for ⩾10 continuous minutes, with lightly active 1.5⩽3METs, fairly active 3–6 METs and very active ⩾6 METs. 19
Given that improvements in patients’ physical activity by SR morphine may take some time to manifest, 20 the analyses focused on caregiver burden and patient measures at W0 and W3. Due to small numbers in the placebo arm and the fact the 8 mg SR morphine dose has not been tested in any trial in chronic breathlessness, placebo+8 mg were combined as the reference category to explore caregivers’ perceived burden in relation to higher doses (16 mg/24 mg/32 mg) which were achieved for at least 1 week before analysis.
Demographics and ZBI-12 scores were tabulated. Descriptive analysis of burden as dichotomous variable used a cut-off point of 13. 21 Changes in ZBI-12 scores from W0 to W3 were classified in four burden categories (low (no change); high (no change); low to high; high to low), and compared for two different groups (placebo + 8 mg SR morphine vs 16 mg/24 mg/32 mg SR morphine). Spearman’s rank correlation coefficient was used to analyse correlations between changes in ZBI-12 scores, breathlessness and physical activity measures (as continuous variables).
Results
Eighty-two caregivers were invited; of the 51 caregivers who consented, 49 were included in the analysis, having provided demographic information and self-reported burden. Median age was 69 years [IQR 50–75], and 59% were women (Table 1). More were spouses/partners (68%), having lived with the patient for a median of 33 years [IQR 4–49]. Hands-on care was provided by 60% of caregivers, over a median period of 3 years [IQR 0–8.5]. More caregivers (71%) reported spending >40 h with the patient in the previous week and being their sole care provider (78%). Almost half were retired (47%), with the balance mostly employed part-time or unemployed. Of those reporting changes in employment due to caregiving (14%), six reported working full-time previously. Caregiver burden reported at baseline was a median total score of 12 [IQR 5–17], of whom 53% reported high burden (⩾13).
Baseline characteristics of caregivers (n = 49).

ZBI-12: Zarit Burden Interview 12-item short-form questionnaire; IQR: interquartile range.
Eighty-four percent of caregivers reported no change in caregiver burden from W0 to W3 (median scores 11 and 10, respectively) (Figure 1). Of people reporting high caregiver burden scores in the treatment group, one third had their ZBI-12 scores drop below 13. Of those in the reference group, one fifth had their scores reduce below this same threshold.
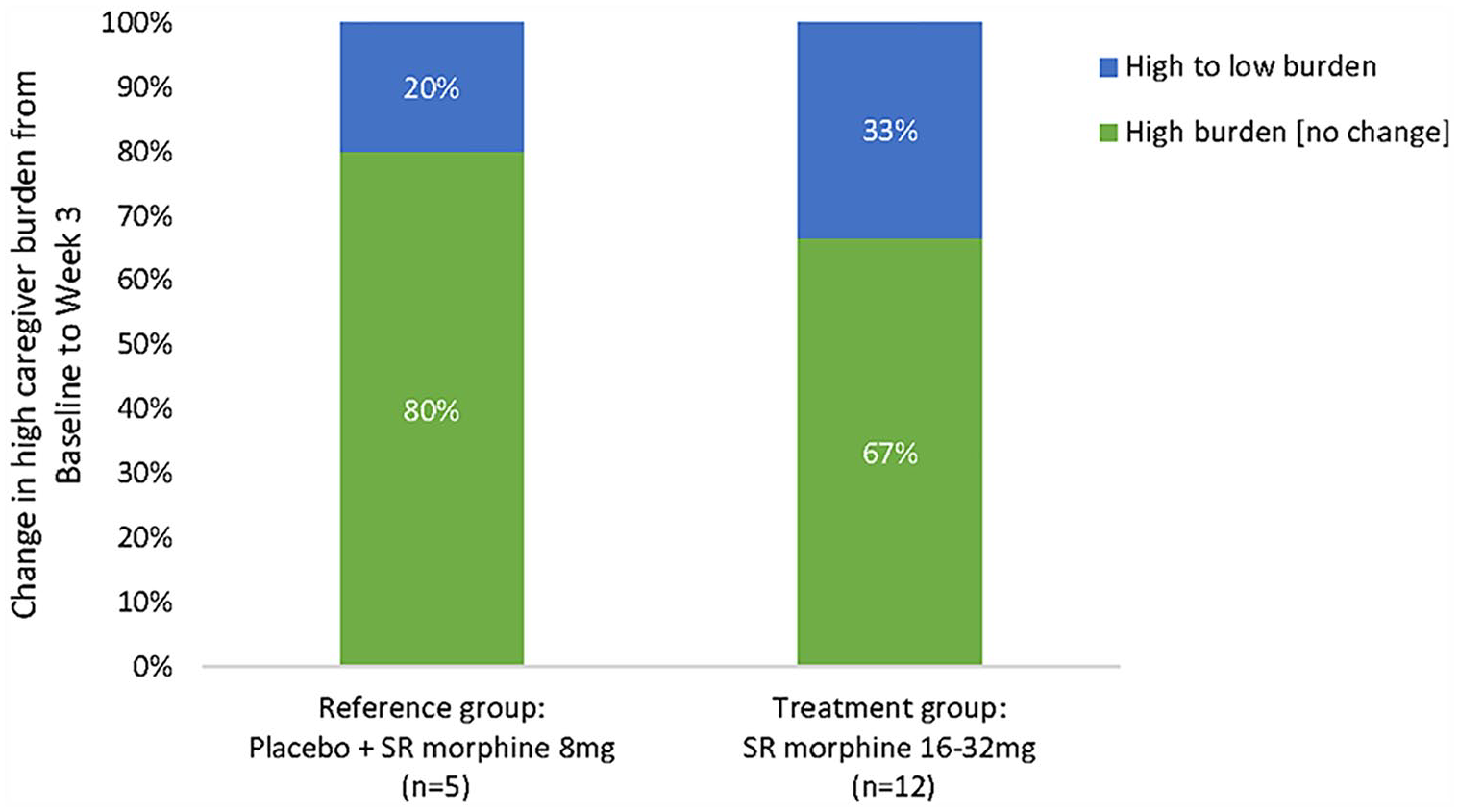
Proportion of caregivers with high caregiver burden by category of change in caregiver burden (ZBI-12) from baseline to week 3 (n = 17).
Caregiver burden decreased as patients’ worst breathlessness improved from W0 to W3 (Figure 2), though the correlation was not significant (rs = 0.25, p = 0.18). In contrast, caregiver burden worsened as patients’ minutes lightly active increased from W0 to W3 (Figure 3), with the correlation being significant (rs = 0.55, p = 0.04).
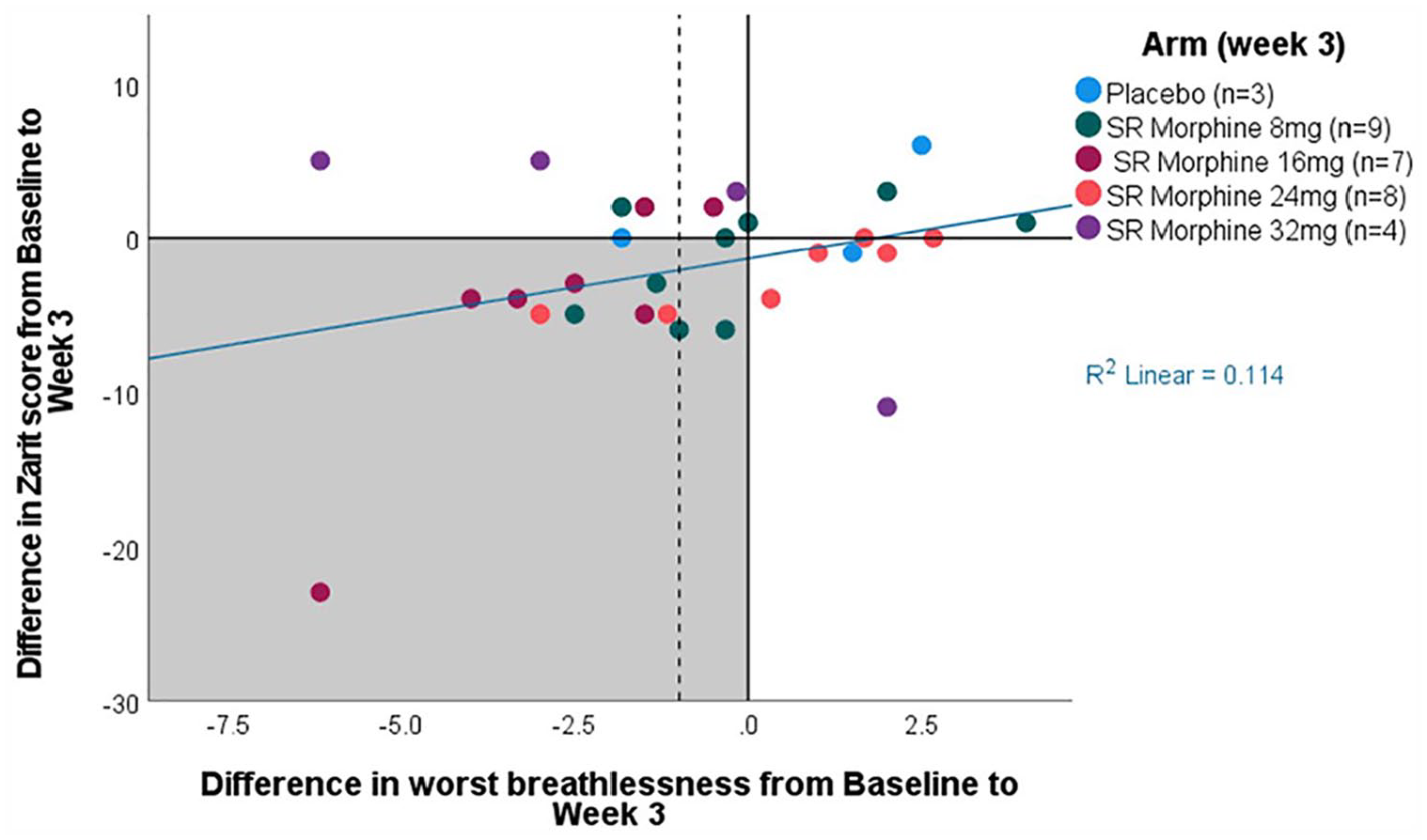
Change in caregiver burden (Zarit Burden Interview 12-item (ZBI-12) score) by change in patients’ worst breathlessness score from baseline to week 3 (n = 31).
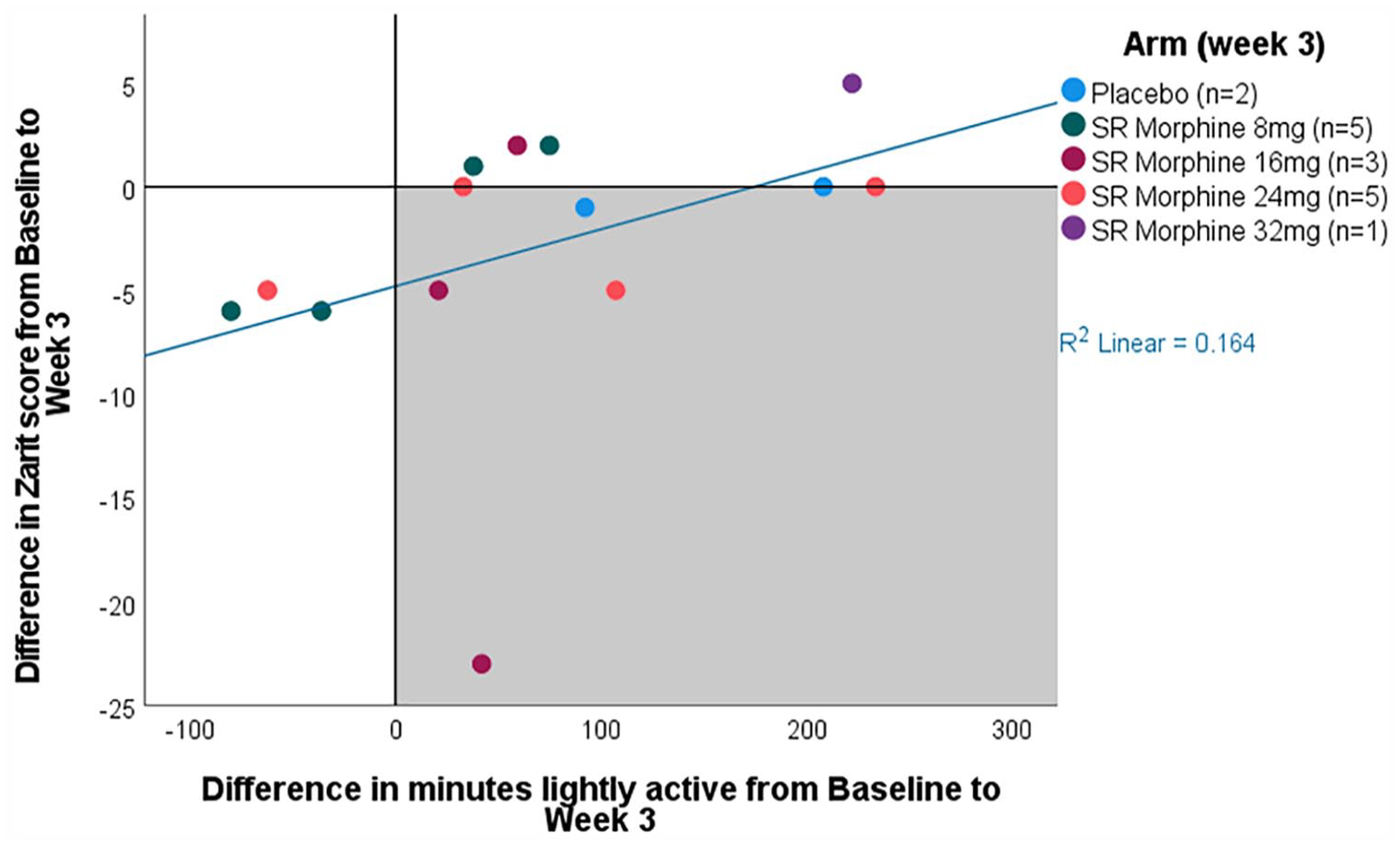
Change in caregiver burden (Zarit burden interview 12-item (ZBI-12) score) by change in patients' minutes lightly active from baseline to week 3 (n = 16).
Discussion
In this exploratory sub-study, a large proportion of caregivers reported high burden across the treatment and reference groups, which remained largely unchanged over time. Our study suggests that caregiver burden may increase as patients become active, a concept that needs to be investigated in future qualitative studies.
The direction of the relationship between symptom change and caregiver burden whereby reduction in breathlessness corresponds to reduction in caregiver burden is tantalising. Caring for a person with chronic breathlessness is demanding on caregivers’ physical, mental and social health. 11 Their often modified lifestyle sees declining social interaction and daily activities to accommodate the growing demands of proving care. 6 Navigating these demands is often with little or no support from health professionals,22,23 which only increases their psychological distress and sense of helplessness. 22 Improvements in breathlessness may reduce caregivers’ stress and anxiety, especially for those who live in close proximity to the patient, thus lessening the caregiver burden.
In contrast, the direction of the relationship between patients’ function and caregiver burden whereby caregiver burden increases as patients become active may seem counterintuitive. Considering that people with chronic breathlessness are constantly working to their physical limits, reductions in breathlessness may translate into higher activity levels, giving patients greater independence and thus, lessening the caregivers’ burden. However, it is also likely that as patients’ function improves, caregivers may be required to provide additional hands-on support with the new activities. A systematic review and meta-synthesis reported that people with chronic breathlessness found the use of oxygen beneficial because it improved their mobility; however, that also meant more work for their caregivers as they needed to carry the equipment. 24 It is possible that with improved function, patients may feel more confident to move beyond their immediate environment yet do not become completely autonomous, thus adding to the caregivers’ demands even when these changes are minimal. Given the complexity of these interactions, recognising the impact of chronic breathlessness and its treatment on both patients and caregivers is important, as is greater awareness by health professionals to assess and address any unmet needs 4 that might result from increased (yet potentially unidentified) caregiver burdens with appropriate support mechanisms.
Our findings highlight the importance of conducting caregiver assessments in clinical trials in palliative care. Evaluating caregiver burden in relation to the patients’ active treatment for breathlessness and function would align with the multi-dimensional nature of this disabling symptom to help inform a more effective, evidence-based practice. 25
The changes observed in this exploratory sub-study necessitate further investigation within a larger and more diverse caregiver cohort, especially in the context of higher doses of SR morphine where a greater proportion of caregivers reported improvement in burden. A more precise delineation of caregiver burden due to chronic breathlessness is also needed as the burden is likely to be multifaceted.
Conclusions
A large proportion of caregivers of people receiving placebo or SR morphine for chronic breathlessness reported high caregiver burden that remained unchanged over time. The complex relationships between caregiver burden and patients’ breathlessness and function should be evaluated in future powered treatment trials to evaluate both patient and caregiver outcomes.
Footnotes
Acknowledgements
We would like to thank the study participants for giving generously of their time and for their participation. We would like to thank Ms Debbie Marriott for her expertise and generous assistance in preparing this manuscript for publication.
Author contributions
S.K. contributed to formal analysis and wrote the original draft. S.C. and D.F. performed the formal analyses and contributed to writing (reviewing and editing). T.L., J.R. and M.E. contributed to writing (reviewing and editing). D.C.C. led the study conception, design and analyses; and contributed to writing (reviewing and editing).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.C.C. has received an unrestricted research grant from Mundipharma; is an unpaid member of an advisory board for Helsinn Pharmaceuticals; and has consulted Mayne Pharma and received intellectual property payments from them. J.R. reports personal fees from Amgen, unrelated to the submitted work, and consultancy with University of Birmingham Enterprise, unrelated to the submitted work. The other authors declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The BEAMS trial was funded by the National Health and Medical Research Council, Australia (Grant Number APP1065571) and sponsored by Flinders University, Adelaide, Australia. J.R. is supported by a Select Foundation fellowship. The funders and study sponsors had no role in the study design, nor in the data collection, analysis or dissemination of study results.
