Abstract
Background:
Palliative sedation is the monitored use of medications intended to relieve refractory suffering. The assessment of palliative sedation has been focused on the assess of the level of consciousness but a more comprehensive approach to assessment is needed.
Aim:
To understand how the potential effects and possible adverse events of palliative sedation in Palliative Care patients are measured.
Design:
Integrative review of most recent empirical research.
Data sources:
Cochrane Library, Embase, Medline, PubMed, and CINAHL were searched (2010–2020) using the terms sedation, palliative care, terminal care, assessment. Limits included studies in English and adults. Inclusion criteria were: scientific assessment papers, effects and complications of palliative sedation; patients with incurable illness.
Results:
Out of 588 titles, 26 fulfilled inclusion criteria. The Discomfort Scale-Dementia of Alzheimer Type and Patient Comfort Score were used to assess comfort. The Richmond Agitation-Sedation Scale and The Ramsay Sedation Scale are the most used to measure its effect. Refractory symptoms were assessed through multi-symptom or specific scales; except for psychological or existential distress. Delirium was assessed using the Memorial Delirium Assessment Scale and pain through the Critical Care Pain Observation Tool. The use of technical approaches to monitor effects is upcoming. There is lack of measurement of possible adverse events and variability in timing measurement.
Conclusions:
There are palliative care validated instruments to assess the sedation effect but this review shows the need for a more standardized approach when assessing it. Instruments should be used within an experienced and trained expert, providing a holistic assessment.
Keywords
In the context of patients with incurable disease palliative sedation is used for refractory symptom control.
A minority of articles measure the effect of palliative sedation and current assessment of parameters of such effect is limited.
The literature about palliative sedation measurement has mainly focused on medication use and level of sedation.
Discomfort Scale-dementia of Alzheimer Type (DS-DAT) and Patient Comfort Score (PCS) are assessment instruments being used to measure the effect of palliative sedation on patient comfort, the latter being validated for palliative care context.
There is limited evidence on the timing of assessment, reported use ranges from daily assessment to six times per day, with often hourly measurements until adequate sedation is achieved.
There is limited data available on the training and preparation of the health professional who has the responsibility for assessing refractory symptoms and palliative sedation.
There is a lack of evidence, regarding measurement approaches or instruments for assessment of existential and psychological distress leading to palliative sedation; and also, for measuring adverse events.
Measurement instruments adapted to palliative care context should be used to assess palliative sedation, as these will facilitate practice improvement and comparability of the study’s results.
Adding measurement instruments for comfort can contribute to assessing palliative sedation effects.
A more standardized approach to assessing the effect of palliative sedation and possible adverse events, paying special attention to adequate training of health care professionals and timing of measurements, is needed to improve the quality of palliative sedation.
Background
Patients nearing death can experience physical, psychological or existential discomfort that causes major distress. In some cases, symptoms can become refractory, which means that treatment options are exhausted either because they fail, the results are not available in sufficient time, or the risk-benefit ratio is no longer acceptable to the patient. In such cases, palliative sedation may be considered.1 –3
The European Association of Palliative Care (EAPC) defined palliative sedation as “the monitored use of medications intended to induce a state of decreased or absent awareness (unconsciousness) in order to relieve the burden of otherwise intractable suffering in a manner that is ethically acceptable to the patient, family and healthcare providers” (p. 581). 3 The EAPC highlights that different terms exist for palliative sedation. Most articles agree that palliative sedation needs to be administered exclusively to patients close to death who are suffering from refractory symptoms,4 –11 with the aim of relieving suffering by administering medication.1,3 The administration of medication must be proportional to the relief of suffering,10,12 –18 which means that the degree of sedation must not be deeper than necessary to relieve suffering.
Depending on the frequency of the administration of medication, palliative sedation can be intermittent or continuous. Palliative sedation can be light, intermediate or deep depending upon the levels needed to ensure comfort for the patient. 16
In Europe, the proportion of deaths associated with palliative sedation is between 7% and 18%.19,20 However, the proportion of palliative sedation used is not easily assessed due to the existence of several definitions and the alternative terms used referring to it,19,21 the existing different types of sedation and the lack of standardized assessment instruments to measure it. 22
Often in health contexts the gold standard used to measure distress and other symptoms is patient reported outcome measures (PROMs). 23 In the case of light or intermittent palliative sedation, this is possible. However, it can be a challenge in cases of deep continuous sedation where there is impaired capacity to communicate.2,24 Therefore, there is a need for a comprehensive approach to explore options for assessment of palliative sedation; its effects and the possible subsequent adverse events.
In these cases, subjective assessments by professionals or via observer rating instruments in sedated patients are more commonly used. 2 It is important that a selection of suitable methods is utilized to assess the effects of palliative sedation on patients and also the appearance of subsequent adverse events.
The last review by Brinkkemper et al. focused on observational scales to monitor symptom control and depth of sedation in patients requiring palliative sedation. 18 They reported the scarce use of scales to measure the effect of palliative sedation, and they also suggested considering the frequency and timing of assessment. No other review has focused on the assessment of palliative sedation by considering a more inclusive approach where assessment goes beyond the use of observational instruments, for example including aspects such as adequacy, timing, and expertise.
The main aim of this article is to understand how the potential effects of palliative sedation in palliative care patients are assessed in the literature including a more inclusive methodology. The secondary aim is to explore the measurement of possible adverse events during palliative sedation.
Design
An integrative review method was selected as the “broadest type of research review method allowing for the simultaneous inclusion of experimental and non-experimental research in order to more fully understand a phenomenon of concern” (p. 547). 25 Whittemore and Knafl’s 25 five stages were used to conduct the integrative review and the PRISMA framework 26 was used to report findings.
Three main concepts were combined: sedation, palliative care, and assessment.
Search strategies were revised with an expert librarian in biomedical databases and adjusted for each database. The key terms were sedation, palliative care, terminal care and assessment, as these were a robust and valid strategy to identify the relevant literature (Table 1). Established limits are shown in Table 1. A balance was made between the sensitivity and specificity of the search strategy (Supplemental Appendix 1). Experts who have been involved in research in the field suggested key articles on the topic and these were used to test the sensitivity of the search strategy.
Search strategies on the different search engines.
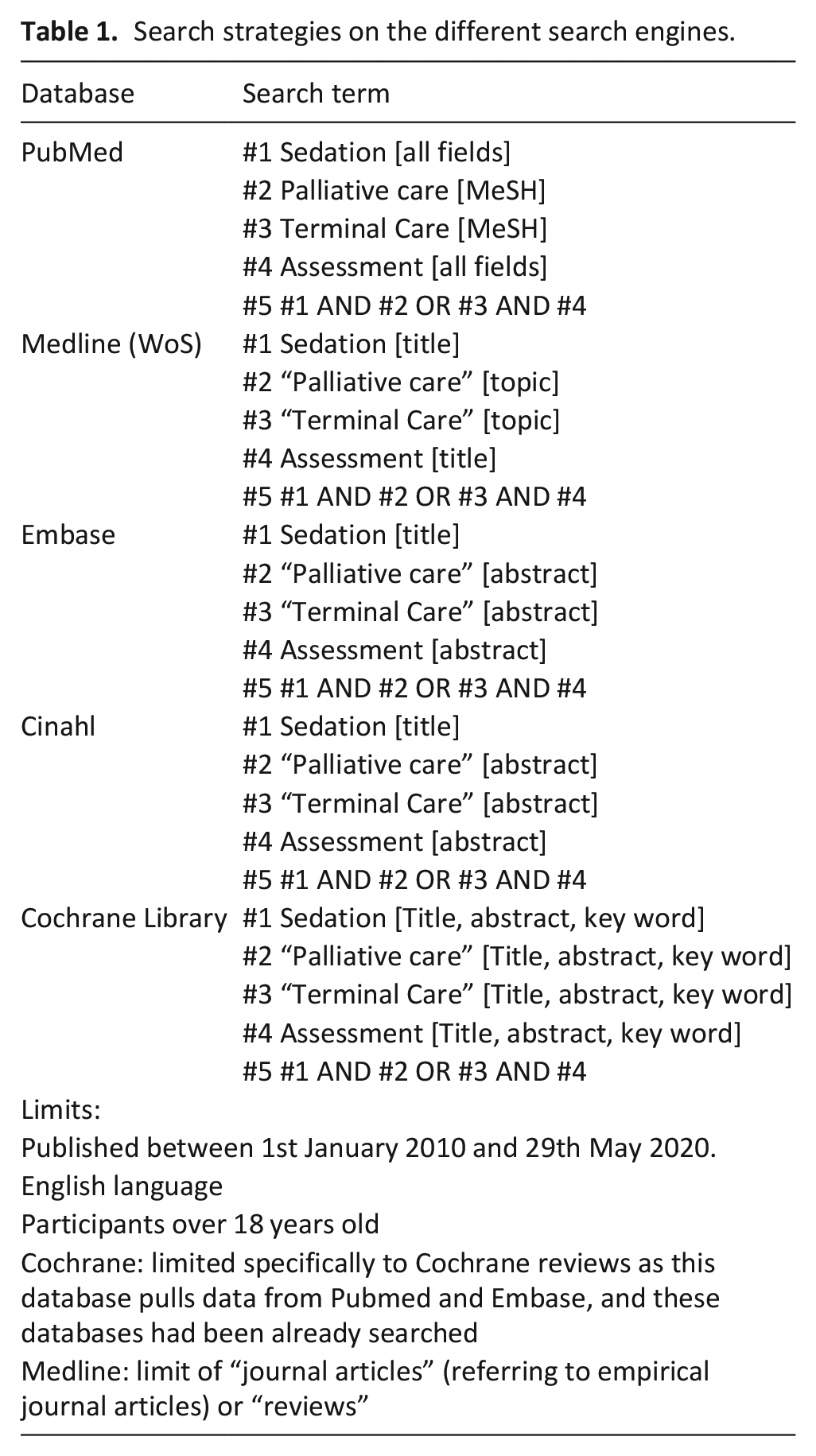
Reference lists of included articles were explored to detect additional cited articles. Citations of the included articles were tracked to identify other eligible articles. 27 In the case of review articles, as their search time was out of our time frame and included older articles, the reviews were used to identify articles.
The article eligibility stage was carried out according to inclusion and exclusion criteria (Table 2) using Covidence software, which allows blind reviewing of titles and abstracts by two independent reviewers. Then full text assessment was conducted. Discrepancies were managed through discussion with a third researcher. Articles were fully read to identify included articles. Reasons for exclusion of articles not meeting the inclusion criteria were systematically recorded.
Inclusion and exclusion criteria.
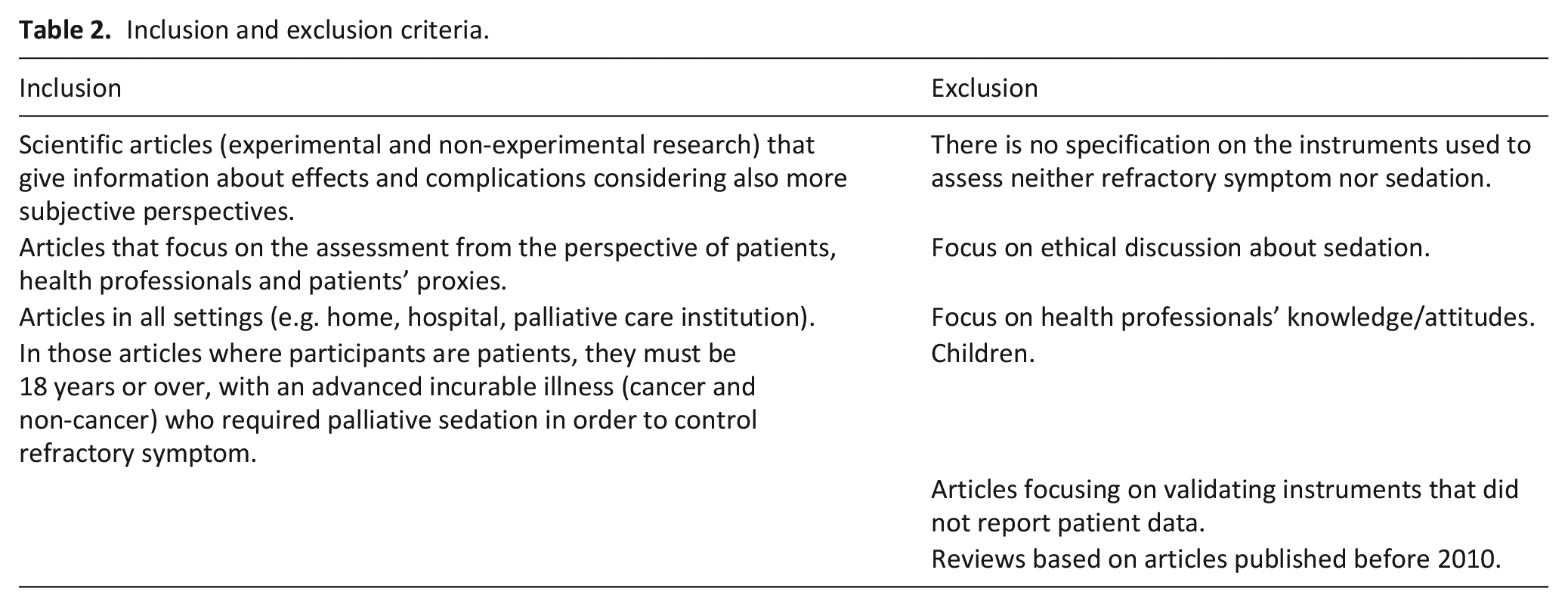
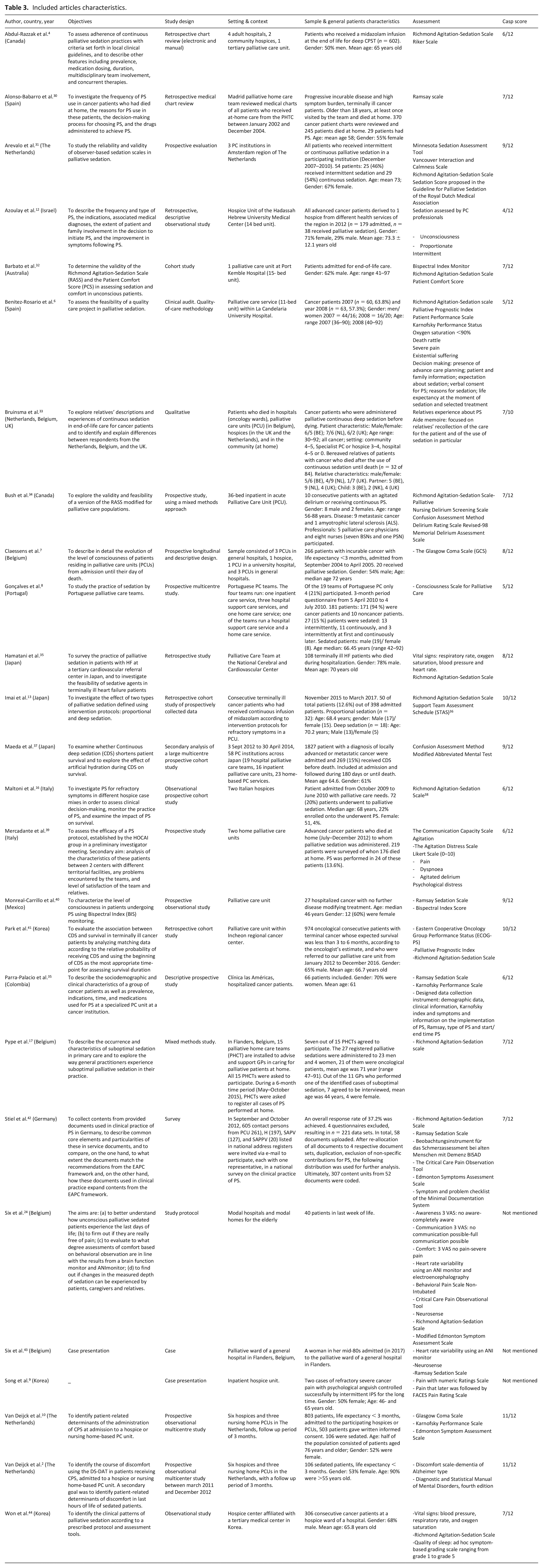
Both researchers used a predefined data extraction checklist that was pilot tested with five articles and adjusted (Supplemental Appendix 2). When multiple articles from the same study were identified these were presented following each other and separated by discontinuous dots (Table 3).
Included articles characteristics.
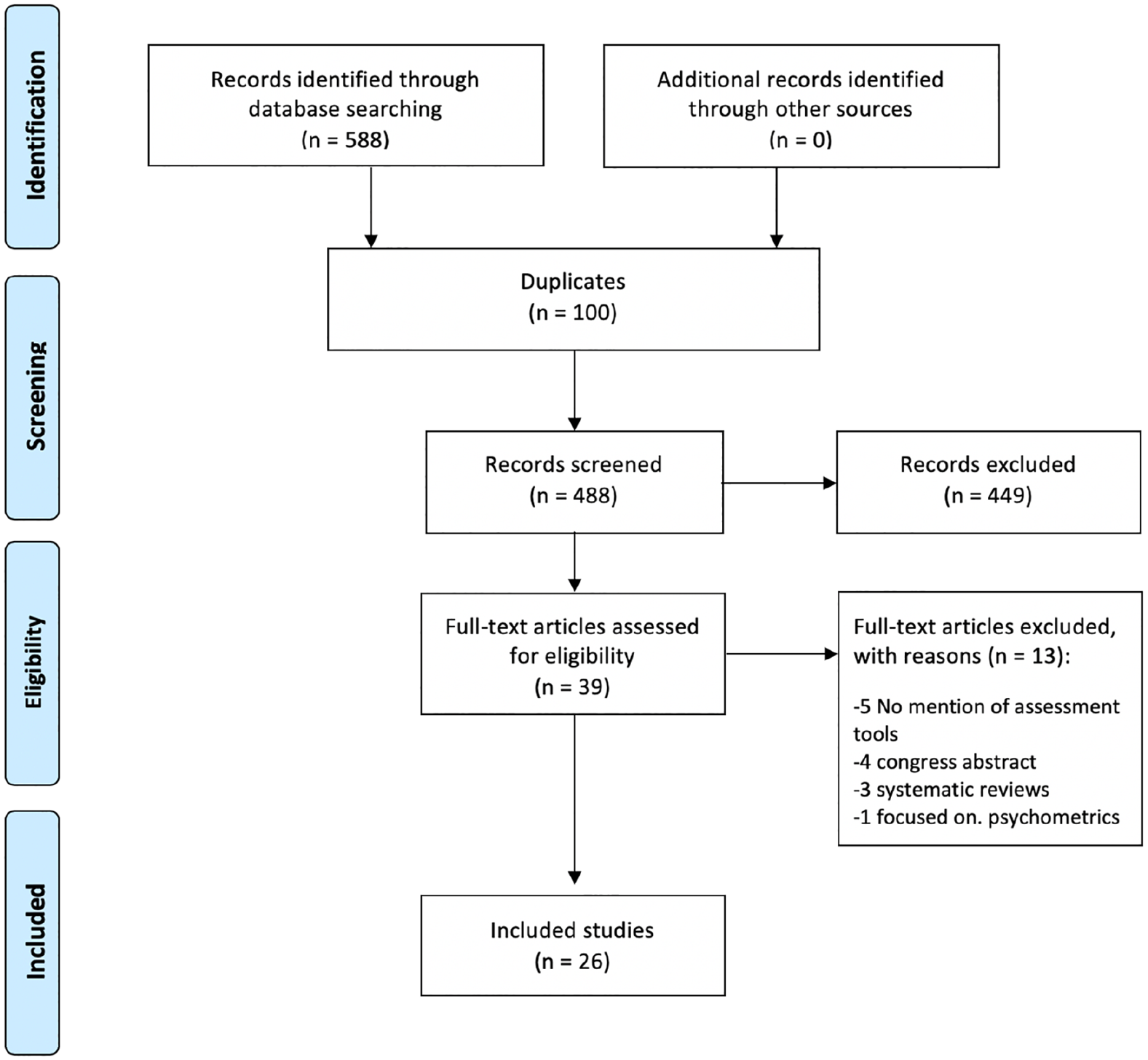
PRISMA flow chart from the search strategy.
Results
A total of 588 articles were screened, 26 met the inclusion criteria. Two articles were part of the same study although they were referenced independently.2,10 Citation tracking of the 26 included articles was conducted in PubMed with no additional inclusions.
Included study characteristics, quality assessment and type of sedation
The 26 articles originated from 14 countries including Belgium and Netherlands (n = 4); Korea and Japan (n = 3); Canada, Italy and Spain (n = 2); Australia, Columbia, Germany, Israel, Mexico, Portugal, and the United Kingdom (n = 1). The main characteristics of the articles are presented in Table 3.
Fourteen articles reflected work conducted in a single site.6,9,11,13,24,30,32,34,35,40,43 Ten studies included multiple sites within the same country or region2,7,8,10,16,17,31,37,39,42 and one included an international collaboration between three countries (Belgium, United Kingdom, and the Netherlands). 15 The majority of studies were conducted in palliative care services. Eight articles included sedated patients with non-malignant disease,2,8,10,17,31,32,34,45 three did not specify a diagnosis4,24,42 and the rest included only cancer patients.
Fourteen articles reported on cohort studies.2,6,7,10,13,16,31,32,35,37,40,41,44,45 Seven studies used a cross-sectional design.4,8,12,30,39,42,44 There were two mixed method studies17,34 and two case studies9,43 and one qualitative methods study. 33 Fifteen articles reported collecting data prospectively with samples ranging from 10 to 269 participants.2,7,8,10,13,15 –17,31,32,34,35,37,39,40 Six studies were conducted retrospectively through chart or clinical documents review.4,12,13,30,41,45
The quality of articles was relatively good7,13,16,17,30 –35,37,39,40 –42,44,45 except for three studies with lower scores (scoring lower than 6/12).6,8,12 The two articles of van Deijck et al. stand out for their excellence (CASP 11/12).2,10
Context and palliative sedation types need to be considered as background information when considering its assessment. All the articles clearly set the use of sedation in the context of end of life, but there are nuances regarding timing. This varied between the last 6 months of life of an incurable disease12,41 to the last phase of terminal illness,2,7,13,37,43 which could be specified as a life expectancy of 2 weeks or less10,15 or last days or hours of life. 44
Five articles mentioned cases of intermittent sedation,8,9,12,35,44 the others referred to continuous sedation, although using a variety of terms: palliative sedation,6,7,16,17,24,30 –32,34,39,40,42,43,45 palliative sedation therapy,6,13,37 continuous palliative sedation therapy, 4 continuous sedation8,33,35 and proportional palliative sedation. 12 The concept of proportionality is mentioned in several articles10,12,13,16,17,33 relating the level of sedation to the degree of patient symptom control needed. Some articles mention specific outcome measures such as: describing suboptimal continuous deep sedation considering their own definition 17 ; measuring the quality of sleep using an ad hoc symptom-based grading scale or palliative sedation success considering their set standards 44 or the quality of the administered sedation regarding the patient comfort and physician estimation of success. 2
Assessment process of palliative sedation and its indications
This process refers to the instruments used, the people involved in the assessment, the timing of assessments and related-adverse events of palliative sedation (Table 3). Summary information about the instruments mentioned below is presented in Table 4.
Description and validation information about identified instruments.

The reference numbers included in this table belong to the original scale reference.
Indications for palliative sedation
Several refractory symptoms such as pain, delirium, dyspnoea or vomiting as well as psychological and existential distress were registered as main reasons for starting palliative sedation. Some articles reported general assessment of symptoms by instruments validated in palliative care, containing multiple symptoms such as anxiety, depression, nausea or shortness of breath. General symptom assessment instruments such as the Support Team Assessment Schedule (STAS), 13 the ten-item Edmonton Symptom Assessment System (ESAS), 42 the Modified Edmonton Symptom Assessment Scale (M-ESAS) 24 and the Minimal Documentation System (MiDOS) were used. 42 Some other articles used team developed numerical instruments to measure pain,9,24,39,42 nausea, 42 vomiting, 42 dyspnoea,39,42 anxiety, 42 psychological aspects,39,42 disorientation, 42 and agitated delirium 39 in sedated patients.
During sedation, delirium and pain were the most frequently assessed symptoms using validated instruments.9,24,34,36,37 Concerning the assessment of delirium, the articles reported the use of various instruments. The Memorial Delirium Assessment Scale (MDAS) is the only scale validated in palliative care patients. 34 This instrument quantifies the severity of delirium over an extended time frame by rating ten individual items at a 4-point scale. Other articles used the Confusion Assessment Method (CAM)34,37 and the Delirium Rating Scale-Revised-98. 34 These are validated instruments, in non-palliative care patients, which explore delirium by scoring aspects such as consciousness, attention/concentration, orientation, behavior and psychomotor activity (Table 4).
According to the Diagnostic and Statistical Manual of Mental Disorders delirium was determined in the article of van Deijck et al. 2 Finally, the Modified Abbreviated Mental Test 37 validated to assess dementia in older patients, was also used for assessing patients mental status, by considering the orientation and consciousness.
Pain was measured as an item within general symptom assessment instruments, such as ESAS and its modified version (M-ESAS), but also with specific pain assessment instruments. Specific instruments validated to be used for pain measurement were: (a) The Faces Pain Rating Scale with a series of faces representing no pain until the worst pain imaginable 9 ; (b) the Critical Care Pain Observational Tool which considers the facial expression, body movements, muscle tension and the compliance with the ventilator or vocalisation24,42 and (c) in the Behavioral Pain scale for non-intubated patients facial expression, movements of upper limbs and compliance with ventilation or the vocalization were scored. 24 From all reported instruments, the Critical Care Pain Observational Tool,24,42 the ESAS10,42 and the M-ESAS 24 are validated for palliative care.
Assessment of life expectancy was performed using various instruments including: the Palliative Prognostic Index 10 (PPI)6,41; Palliative Performance Score (PPS) 6 ; functional status with Karnofsky Performance Status Scale (KPS)6,10,35; Eastern Cooperative Oncology Group performance status scale; and an observer-rated scale of physical ability. 41
Some studies reported assessment of vital signs when presenting their results on palliative sedation.6,44,45
Palliative sedation assessment
Most articles explained how they monitored sedation and in which domain: level of sedation, comfort level or symptom control.2,6 –8,10,13,16,17,24,30 –34,37,39,40,42,43 The majority also reported the use of the Richmond Agitation-Sedation Scale6,13,16,17,24,31,32,41,42,44,45 or the Ramsay Sedation Scale to objectify the effects of palliative sedation.30,35,40,42,43
The Richmond Agitation-Sedation Scale (RASS) is an observational scale that assesses the level of sedation and agitation without requiring patient input. It was validated in intensive care unit patients 38 and adapted for a population with palliative care needs, calling it Richmond Agitation Sedation Scale-Palliative (RASS-PAL). 34 Both instruments measure the patients’ level of sedation and agitation, scoring from +4 (combative) to −5 (not arousable).
Although not validated in a palliative care population, the Ramsay Sedation Scale is often used to measure effects of palliative sedation 64 by scoring the patients’ sedation level within six categories ranging from severely agitated to not responsive.
Less frequently mentioned instruments for assessment of sedation levels, but validated in palliative care, are the assessment presented in the Guideline for Palliative Sedation by the Royal Dutch Medical Association, 31 which considers sedation and the response to stimuli; the Consciousness Scale for Palliative Care, 8 which assesses the consciousness level through stimulation, and the Agitation Distress Scale assessing agitation through observation of the patient. 39
Specific instruments validated in other populations also utilized in sedation monitoring are the Minnesota Sedation Assessment Tool (MSAT) 31 which scores the motor activity and the arousal of patients, and the Riker Sedation Agitation scale that considers the agitation and sedation of the patient. 39
Effects of sedation have also been measured by considering the consciousness of the patient with the Glasgow Coma Scale recording eye opening and the motor and verbal response7,10; the Vancouver Interaction and Calmness Scale (VICS) considering the interaction with the environment 31 ; and the Communication Capacity Scale Score assessing interaction level of the patient. 39
Few clinical articles use comfort as an outcome measurement in sedated patients. Comfort of the sedated patients was measured through the Discomfort Scale-Dementia of Alzheimer Type (DS-DAT), 2 based on observing different behavioral indicators; and the Patient Comfort Score (PCS), 70 that considers pain and level of consciousness.
In some articles,24,32,40,43 sedation was measured using physiological factors monitored by technical approaches. The Neurosense24,43 assesses hypnotic depth of anesthesia by displaying two EEG signals and calculating several parameters, including the Wavelet Anesthetic Value for Central Nervous System (WAVcns), ranging from 100 (awake) to 0 (flat EEG). 24
The Bispectral Index Score (BIS)24,32,40 is a non-invasive and validated instrument to measure the hypnotic effect of sedative and anesthetic medications, ranging from 100 (fully awake and aware) to 0 (brain death). Each patient is connected to a Quatro sensor applied to the forehead and analyses frontal EEG input using an algorithm.
Another reported non-invasive technique is the continuous monitoring of Heart Rate Variability (HRV) transformed into an Analgesia Nociception Index (ANI, 0–100),24,43 which assesses parasympathetic activity as a possible measure of nociception as HRV reflects the effect of the vagus nerve on the heart which is inhibited during pain. ANI has been shown to be effective in detecting pain in deeply sedated critically ill patients. 24 It is based on the analysis of the respiratory fluctuations of heart rate that reflect the variability in the parasympathetic tone.24,43
Assessment responsibility and timing
Sedation was monitored by different professionals. Most assessments were performed by nurses4,7,10,17,24,31,32,40,44 with fewer assessments undertaken by physicians,24,35,40 researchers and palliative care professionals. 6
Almost half of the articles included mentioned the timing of assessments. Daily assessment was common as a minimum requirement.7,24,32,39,44 Other articles reported hourly measurements,13,42 hourly during the first 4 h, 34 6 hourly until reaching adequate sedation, 16 maximum of five times 31 or six times per day.32,40 In the case of technical approaches for monitoring, continuous registration of values is described.32,43
Adverse events
During the administration, unintended effects of palliative sedation might occur. Only one study grades the severity of adverse events based on the Common Terminology Criteria for Adverse Events version 4.0. 13 They considered the causality of adverse events when grading 3 or 4. They reported that apnea occurred in 1/32 patients receiving proportional sedation group and in 4/18 of the deep sedation group. No fatal events were considered as probably or definitely related to the intervention, and protocol-based sedation was continued in all cases. Other adverse events such as decreased respiratory rates (entire cohort of 32 patients 35 ; (10/89 patients), 42 decreased oxygen saturation (3/89 patients), 44 and paradoxical agitation (3/89 patients) 44 were reported. The majority of the studies did not explain how these adverse events were measured, only one referred to the use of oximeter for oxygen saturation. 44 In the rest it can be deduced that adverse events were measured through observation as consisted on assessing and registering vital signs (i.e. respiratory rate).
One of the most controversial adverse events, raising ethical concerns, is whether palliative sedation hastens death. In this review, several articles demonstrated that palliative sedation does not shorten survival.6,37,44 Survival was calculated considering the period from hospitalization to death6,44 or from enrolment in the study to death. 37 Azoulay et al., 12 analyzed the survival of palliative sedated patients considering who initiated the decision to use it (i.e. patient, medical staff or family) and the type of sedation, with no differences on survival. Finally, other study compared survival between the patients who were administered continuous deep sedation and those who were not. 41 Survival was statistically significantly longer in the continuous deep sedation group than the non-continuous deep sedated group. 41
Discussion
The results of this review of 26 articles from 14 different countries, clearly demonstrates an increasing international interest in the use of palliative sedation. The majority of the articles report on studies conducted in a single site or several sites within the same country or region. Studies mainly included patients with cancer but some included patients with non-malignant conditions.
This review demonstrates improvements in comparison with the review of Brinkkemper 18 as there is an increase in available and validated monitoring instruments of refractory symptoms and the effects of palliative sedation over the last ten years. Articles unanimously agree that refractory symptoms are a prerequisite when considering sedation. Articles tend to name the refractory symptoms, but often their assessment is not clearly reported with limited or no information on the evaluation instruments used or scores obtained. Among the instruments validated for palliative care and most frequently used are the Edmonton Symptom Assessment System (ESAS),51 the Memorial Delirium Assessment Scale (MDAS)55 and the Critical Care Pain Observational Tool (CPOT).48 The ESAS assesses a variety of symptoms, but it has been mainly used in the studies to assess pain while others used the CPOT; whereas delirium has been assessed with MDAS. These instruments are commonly used assessment instruments, suggesting that in clinical practice instruments are used to identify refractory symptoms. This practice is important as it allows comparison between studies and settings. In addition, when patients are unable to provide details about their symptoms (i.e. due to delirium, the depth of sedation), proxies can be an important source of information. Thus, it is also important that the symptom assessment instruments have been validated for proxy-as well as for self-assessment.
Assessment of profound psychological or existential distress
It is noteworthy that occasionally profound psychological or existential distress are mentioned as reason for starting palliative sedation.6,12,17 No instruments to assess them have been identified in the review. This may be due to lack of awareness and underreporting related to professionals being more geared towards documenting physical sign and symptoms more than existential distress; rather than low incidence of this indication. There may be several explanations for this. There is a lack of consensus on a definition of existential suffering. 71 Existential distress has been linked to aspects such as loss of personal meaning and purpose to life, fear of death, despair, hopelessness, loss of dignity, sense of isolation.72,73 No instruments were reported in the included articles. However, a review on available instruments to assess suffering for use in palliative care has identified instruments for assessing psycho-existential suffering. 71 These instruments may be used to improve awareness in this area. Moreover, assessment of existential distress requires a complete multidimensional approach including psychologists, psychiatrists and/or spiritual caregivers, in order to identify it as a refractory situation. 71
Instruments to monitor level of sedation in palliative care context
Seven different instruments to monitor level of sedation have been identified in this review. Four of these are validated in palliative care, compared with the two, the Ramsay sedation scale, the RASS and the Communication Capacity Scale, reported in the 2013 published review. 18 This provides more choice of instruments that can be utilized in palliative care. Two instruments assess the agitation level of the patient, which also assess level of sedation, 34 and the Agitation Distress Scale. 39 The RASS-PAL has received the highest rating on psychometric properties together with the Consciousness Scale for Palliative Care, according to a recent systematic review evaluating instruments to monitor level of consciousness on palliative patients. 73 The other two instruments identified in the current review and validated for palliative care are the KNMG sedation score of the Royal Dutch Medical Association 62 and the Consciousness Scale for Palliative care. 8 The former being mainly used in Dutch contexts and the latter reported as easy to use. 66 Articles identified did not discuss the use of neurological levels of somnolence, stupor and coma as a means for monitoring depth of sedation.
Technical approaches to assess physiological responses coming from anesthesiology are being used to assess level of sedation6,32 or parasympathetic activity.24,43 In a case report the use of Neurosense monitor was described by families as quite acceptable and non-intrusive. 43 However, reliability of these methods has not been proven outside the controlled setting of an operating theatre. 32 The technical equipment, but also the wide range of BIS values in deeply sedated and comfortable patients make its use in routine clinical practice unlikely. 74
Measuring palliative sedation effect
As the aim of palliative sedation is relief from refractory symptoms and not achievement of a specific level of consciousness,3,75 instruments that assess symptom relief or patient comfort are recommended. Two instruments for comfort assessment, the Discomfort Scale Dementia of Alzheimer Type (DS-DAT) 69 and Patient Comfort Score 70 were identified; the latter being validated in palliative care context. These instruments use observational criteria indicating that the patient seems relaxed, does not grimace and is not agitated.
Adverse events
Potential adverse events need to be considered when assessing the effect of palliative sedation, but this review found little information relating to this and how these are assessed. The risk of hastening death is a serious adverse event, but a Cochrane review 5 found no evidence that palliative sedation hastens death. Documentation of respiratory rates, blood pressure and cardiac arrest were reported as safety measures in one publication. 76 Documenting vital signs has little consequences on these patients as there is a decline in them as part of the dying process and might lead even to wrong consequences, for example reduction or withdrawal of sedation in the dying patient when he needs it. This review has identified very few cases of adverse events being reported and even less information about how these are assessed. Future research should include systematic assessment and documentation of adverse events.
Assessment of palliative sedation: expertise, timing, and proportionality
In the reviewed articles, assessment was conducted by healthcare professionals, mainly nurses. There is very limited information about professionals’ training to assess sedation and refractory symptoms. Only one article reports that regular in-service training and information sessions were provided to ensure competency and proficiency to undertake assessments. 32 As many of the included studies were conducted within palliative care services, it might be assumed that the professionals were adequately trained for monitoring palliative sedation. However, there is an ongoing discussion in the literature about who should monitor sedation and about the need for consultation of a palliative care specialist for expert assessment of refractory symptoms.3,77,78 The use of validated instruments for palliative care patients is recommended, but a holistic clinical assessment needs to contain more than the use of them. Although, it is recommended that family members can provide input with the assessment of patients distress. 3 Further studies are needed due to the limited evidence on this. 15
There is broad consensus that patients should be assessed and monitored at the initiation as well as continuously throughout the sedation process. However, there was a wide range of assessment times reported in the literature. The EAPC framework recommended assessment initially at least once every 20 min until adequate sedation is achieved and subsequently at least three times per day. 3 This is in line with a systematic review on published clinical guidelines that assessed recommendations on monitoring. 79 Guidelines recommended frequent monitoring during initiation of palliative sedation with intervals of 15–30 min. For ongoing sedation, they reported monitoring intervals ranging from hourly to once a day. 79 The clinical guidelines agreed that it is essential to monitor that the patient is comfortable, does not receive too much or too little sedation and that possible adverse events can be identified and acted on.79,80 It would be interesting to know if monitoring reported on papers is part of the daily clinical practice or as part of the study monitoring, as it may explain the variations.
The literature on sedation implicitly refers to continuous sedation: only a few articles referred to intermittent sedation.8,9,31 Even though, definitions of sedation deliberately made no distinction between continuous and intermittent, and light and deep sedation, 75 there seems to be an underlying trend to assume that palliative sedation is always or mainly continuous deep sedation. The concept of proportionality is quite often mentioned in the literature, making a concept already implied in the definition more explicit,4,8,12,75,81 emphasizing this way that palliative sedation needs to be adjusted progressively to control the refractory symptom although it seems that often requires reaching deep sedation to manage the symptom.
The studies in this review investigated palliative sedation in an end-of-life context, though there was considerable variability on the timeframe. Sometimes palliative sedation was offered during a period where patients had an estimated prognosis of 6 months 12 and in other studies 2 weeks (REF Abdul-Razzak era k 2019) or for the last hours of life. 30 The tendency reported in the clinical cases is within days except in individual cases when intermittent sedation was administered for months (< 6 month). 9 This has implications for the method and timing of assessment, as intermittent sedation may not need such a close assessment as continuous cases and can count with patient perspective. In consequence, the European Society for Medical Oncology (ESMO) guideline suggests that patient monitoring should be determined by the clinical situation, 82 taking into account also the level of unconsciousness.
What this study adds
This study updates the information about instruments available to assess the effect of palliative sedation, beyond observational scales to measure the level of sedation. It provides information on assessment tools used to measure other potential effects of palliative sedation such as symptom control, comfort or related-adverse events, specifying if they are validated for palliative care context. It adds a comprehensive view of the assessment, considering the expertise and involvement of people on it and the timing. The EAPC framework on palliative sedation highlighted the need to define the quality of sedation. 3 For this aim, a well oriented and comprehensive assessment is needed. Assessing refractory symptom relief, patient comfort with the minimum decrease on consciousness lowering of conscious level and adverse events, can be a way to describe the quality of the intervention in clinical practice. This study provides information about it and suggestions to improve palliative sedation assessment. However, this study do not want to be prescriptive considering that instruments need to be clinically applied being available in local language and being culturally adequate. In order to specify further recommendations, it would be interesting to consider if the tools are available in the language needed and this was out of the scope of the review. Further studies should be done measuring the effects of palliative sedation with more adequate assessment strategies to show more clearly the contribution of it to patient comfort and symptom relief.
Strengths and limitations
Rigorous methodological steps were used to decrease the risk of bias, for example using independent and blinded assessment of articles. However, a few limitations apply to this review. Only articles in English were included and there may be relevant articles published in other languages (e.g. French) that have not been included. However, this review provides information from 14 countries. The different research methods used in the articles, and variability in the assessments reported, complicated compilation of findings and did not allow for meta-analysis. The under representation of vulnerable groups and cultural minorities in the studies is also a limitation. One article mentioned that they did not include patients of indigenous descent as it required additional approval and based on the very small number of indigenous people usually admitted to the unit. 32
Conclusion
The review identified validated instruments in palliative care context that allow assessing the effect of palliative sedation including its outcomes, relief from refractory symptoms and patient’s comfort. These instruments should be used within an expert interdisciplinary team who can provide a complete clinical assessment. A standardized approach for assessment, including timing and documentation, and adequate training for healthcare professionals is needed to improve both clinical practice and support comparison between research studies. Adverse events are not commonly reported on palliative sedation, possible risk of hastening death is the most studied. Future studies need to specify the systematic assessments conducted including possible adverse events.
Supplemental Material
sj-docx-1-pmj-10.1177_0269216320974264 – Supplemental material for How to measure the effects and potential adverse events of palliative sedation? An integrative review
Supplemental material, sj-docx-1-pmj-10.1177_0269216320974264 for How to measure the effects and potential adverse events of palliative sedation? An integrative review by Alazne Belar, María Arantzamendi, Sheila Payne, Nancy Preston, Maaike Rijpstra, Jeroen Hasselaar, Lukas Radbruch, Michael Vanderelst, Julie Ling and Carlos Centeno in Palliative Medicine
Footnotes
Acknowledgements
We acknowledge all consortium H2020 Palliative Sedation (SEP-210506914) members for their inputs: Stichting Katholieke Universiteit, Pecsi Tudomanyegyetem-Universtiy of Pecs, Katholieke Universiteit Leuven, Hospice Casa Sperantei, Universitaetsklininikum Bonn, Lancaster University, European Association of Palliative Care and La Maddalena S.p.a.
Author Contributions
Conception or design of the review: MA, AB, SP, MR, NP, ME, LR, JH, JL and CC. MA, AB conducted the search strategy and data analysis. SP, MR, NP, MV, LR, JH, JL and CC provided feedback on each stage of the review and contributed to the development of the paper. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work of ‘PalliativeSedation’ is funded by the Horizon 2020 Framework Programme of the European Union under Grant Agreement nº No. 825700.
Research ethics and patient consent
Not applicable.
Data management and sharing
Complementary data is available online. Further data are not publicly deposited by and may be available upon request to the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
