Abstract
Background:
The European Association for Palliative Care (EAPC) acknowledges palliative sedation as an important, broadly accepted intervention for patients with life-limiting disease experiencing refractory symptoms. The EAPC therefore developed 2009 a framework on palliative sedation. A revision was needed due to new evidence from literature, ongoing debate and criticism of methodology, terminology and applicability.
Aim:
To provide evidence- and consensus-based guidance on palliative sedation for healthcare professionals involved in end-of-life care, for medical associations and health policy decision-makers.
Design:
Revision between June 2020 and September 2022 of the 2009 framework using a literature update and a Delphi procedure.
Setting:
European.
Participants:
International experts on palliative sedation (identified through literature search and nomination by national palliative care associations) and a European patient organisation.
Results:
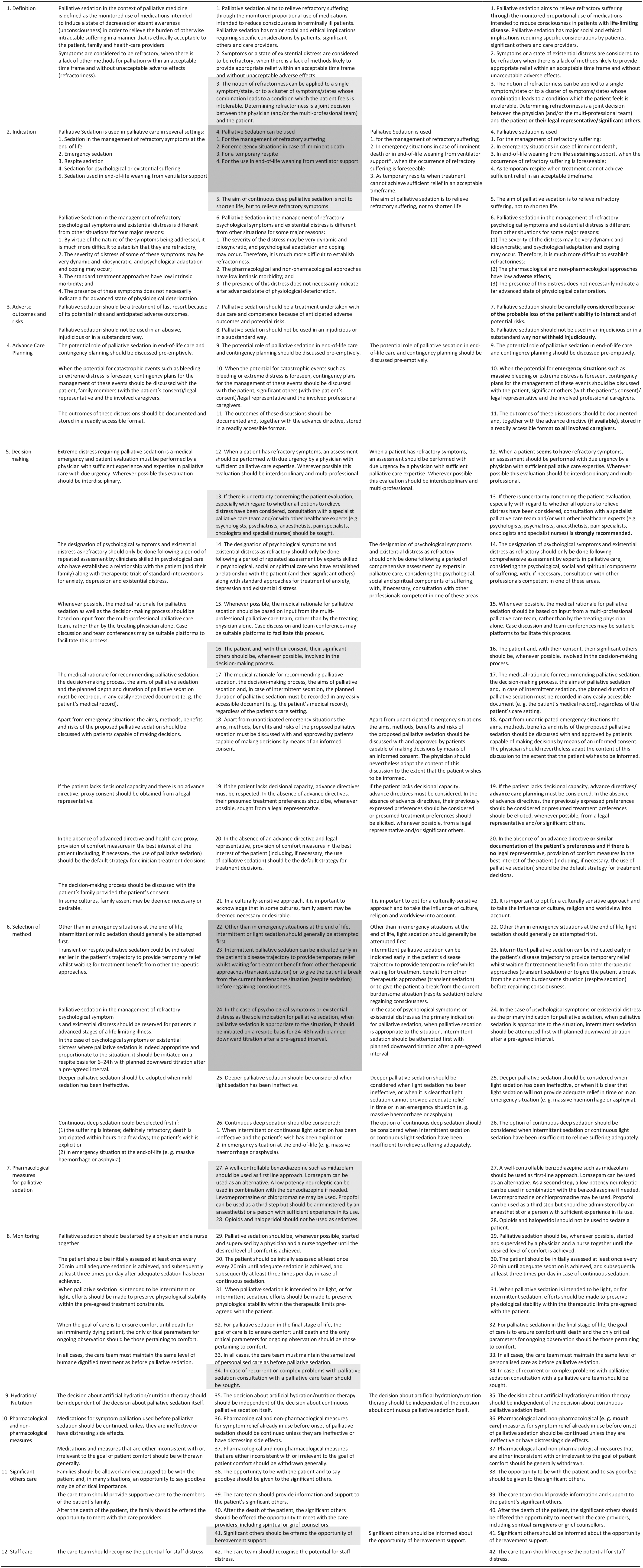
A framework with 42 statements for which high or very high level of consensus was reached. Terminology is defined more precisely with the terms suffering used to encompass distressing physical and psychological symptoms as well as existential suffering and refractory to describe the untreatable (healthcare professionals) and intolerable (patient) nature of the suffering. The principle of proportionality is introduced in the definition of palliative sedation. No specific period of remaining life expectancy is defined, based on the principles of refractoriness of suffering, proportionality and independent decision-making for hydration. Patient autonomy is emphasised. A stepwise pharmacological approach and a guidance on hydration decision-making are provided.
Conclusions:
This is the first framework on palliative sedation using a strict consensus methodology. It should serve as comprehensive and soundly developed information for healthcare professionals.
Keywords
The European Association for Palliative Care (EAPC) developed a framework on palliative sedation in 2009, acknowledging it as an important and ethically acceptable last resort intervention for terminally ill patients experiencing refractory symptoms.
Over the last two decades, a number of guidelines on palliative sedation have been developed in Europe and beyond.
The general weakness of their methodological development has been criticised and their lack of consensus on terminology and concepts make their applicability difficult.
Some issues, such as the complexity of assessing refractory symptoms or the differentiation between somatic, psychological and existential suffering, remain under debate.
This paper provides the first consensus-based guidance on palliative sedation structured in 42 statements and explanatory texts, for which a high or very high level of consensus has been reached among experts from 28 different countries with a broad range of professions, and a European patient organisation.
The importance of patient autonomy is emphasised in all phases of the process (timely discussion of patient preferences, shared decision-making process, informed consent by patient/legal representative).
The term suffering defined as to encompass distressing physical and psychological symptoms as well as existential suffering is used to reflect the shift towards a broader recognition of existential suffering as indication for palliative sedation.
No specific period of remaining life expectancy has been defined for the use of palliative sedation based on the three key principles of (1) refractoriness of suffering, (2) proportionality, which has been explicitly introduced in the definition of palliative sedation and (3) independent decision-making for hydration.
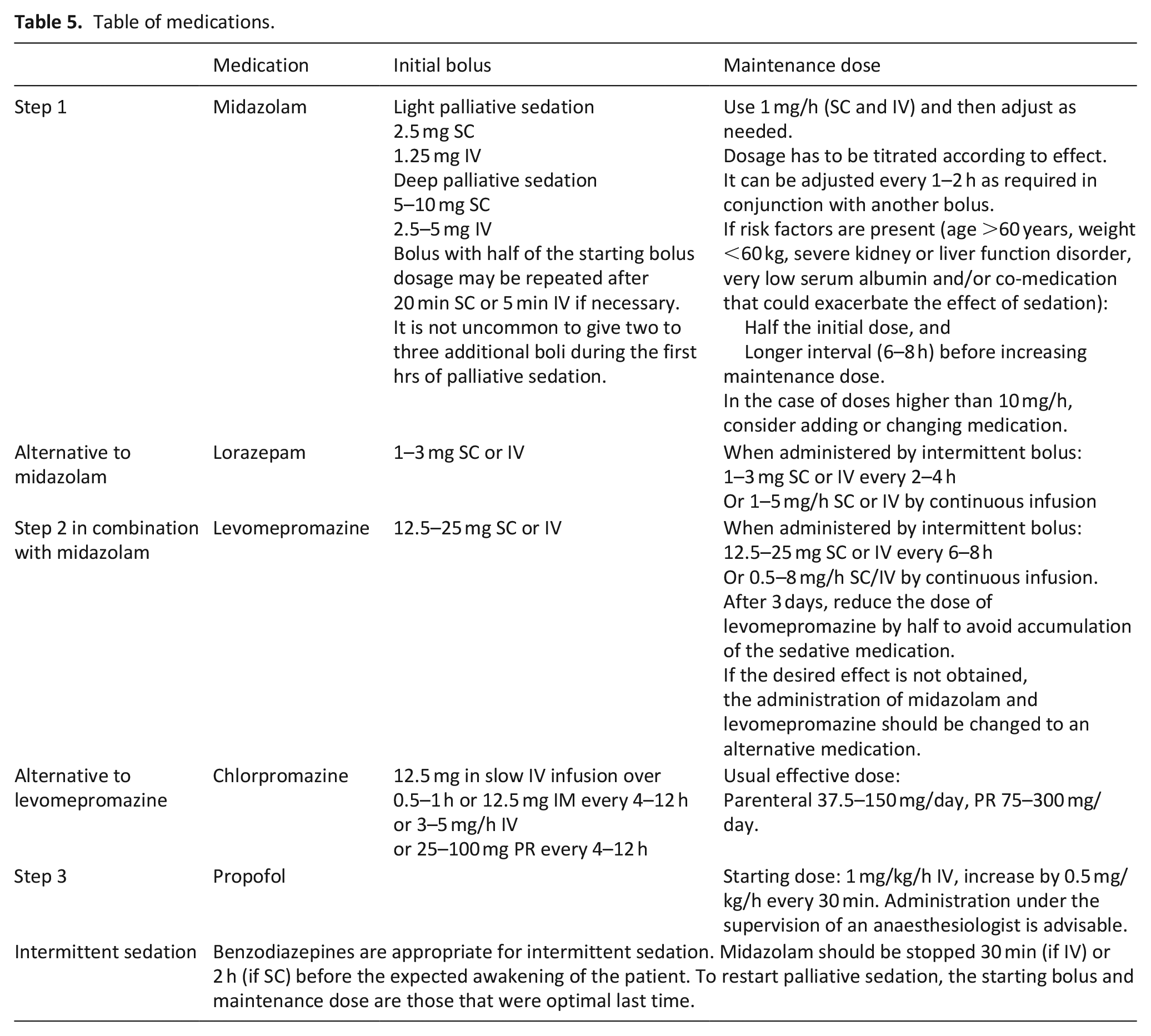
A step-by-step pharmacological approach with a detailed description of the recommended medications as well as a more detailed guidance on decision-making regarding hydration based on recent literature are provided.
This paper provides evidence- and consensus-based guidance for healthcare professionals involved in the care of adult patients with life-limiting disease in all settings, as well as for medical associations and health policy decision-makers.
Introduction
Even with comprehensive palliative care some patients with life-limiting diseases suffer from severe physical, psychological or existential distress at the end of life, for which conventional treatment options fail. In these cases of refractory suffering, the intentional reduction of consciousness (palliative sedation) may be indicated. It is estimated that palliative sedation precedes 10%–18% of deaths of palliative care patients in Europe, although its use is known to vary considerably across cultures and countries, and even between institutions.1–10 In 2009, the European Association for Palliative Care (EAPC) developed a framework on palliative sedation 11 to inform the development of local procedural guidelines. Over the last two decades, a number of national or regional guidelines on palliative sedation have been issued.12 –14
An update to the EAPC framework was deemed necessary as the general weakness of the methodological development and applicability of existing guidelines on palliative sedation 13 and the variations in the terminology used2,13 –16 have been criticised. More recently, a new terminology has been suggested, introducing the term ‘intentional sedation’, focussing on the causal role (intention to sedate) of the clinical action (sedation). 17 Even though new research on palliative sedation has been published,2,18,19 issues such as the complexity of assessing refractory symptoms14,20 or the differentiation between somatic, psychological and existential suffering 14 remain under discussion. In addition, a recent systematic review showed an underlying need to emphasise the principle of proportionality in palliative sedation practice with regard to the patient’s individual situation, considering the continuum from light to deep and from intermittent to continuous sedation. 2
Based on these issues, the Palliative Sedation (PalSed) project, 1 funded by the European Union (Horizon 2020 grant no. 825700), included the remit to update and revise the EAPC framework on palliative sedation using a rigorous Delphi consensus procedure with international experts and the European Cancer Patient Coalition (ECPC). The aim was to provide evidence and consensus-based guidance on palliative sedation for healthcare professionals involved in the care of adult patients with life-limiting disease in all patient care settings, as well as for medical associations and health policy decision-makers.
Methods
In light of the considerable differences in clinical practice of palliative sedation between cultures and countries a consensus methodology was performed in addition to the evaluation of the evidence in the literature. The four-step consensus procedure was based on the widely used Delphi methodology 21 and the reporting on the standards for Conducting and Reporting Delphi Studies (CREDES), which include clear criteria for consensus projects. 22 The development of the statements also considered all relevant methodological and content aspects of the Appraisal Guideline Research and Evaluation II (AGREE II), 23 an international tool to assess the quality and reporting of practice guidelines.
The study was approved by the Ethics Commission, University of Bonn, Germany, No. 268/20 (19 June 2020).
Literature update
The references of the 2009 framework were updated with scoping reviews in PubMed for key words of each framework section for the period 2009–2021 (articles published in English). In addition, experts were invited at each stage of the consensus procedure to propose additional references. As additional outcomes of the PalSed project two systematic reviews of prospective studies,2, 18 a review on monitoring 19 and a review of European guidelines on palliative sedation 12 were published. However, due to the workflow of the PalSed project the literature identified in these reviews was only available during step 2 and 3 of the Delphi process and were then implemented in the document.
Delphi procedure
Preparatory phase (step 1)
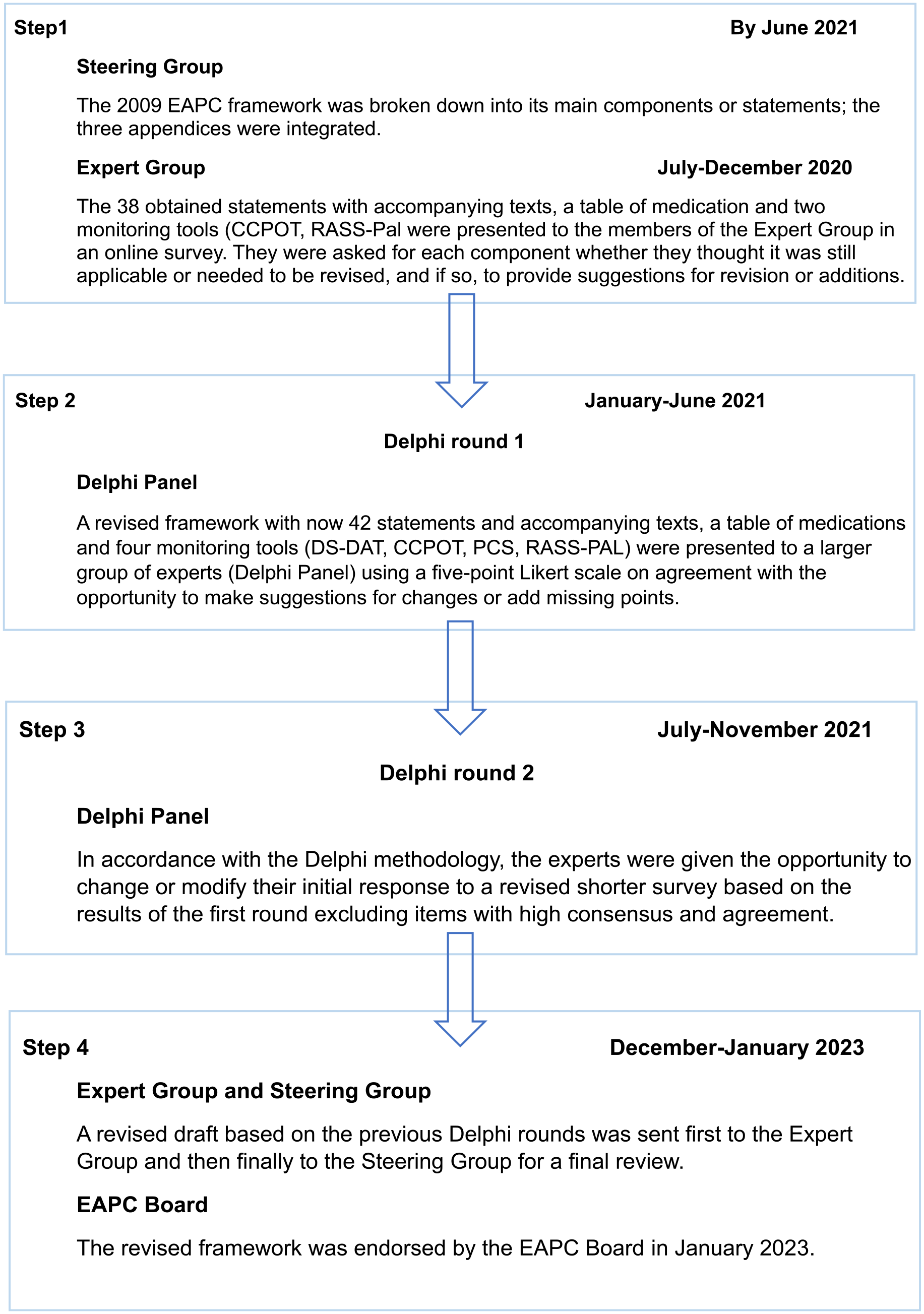
The consensus process was managed by two researchers (HB and SMS) and supervised by a Steering Group including palliative care experts and a patient representative from the European Cancer Patient Coalition (AC, EG, JH, BJ, SP and LR; see Figure 1).
Steps of the Delphi procedure.
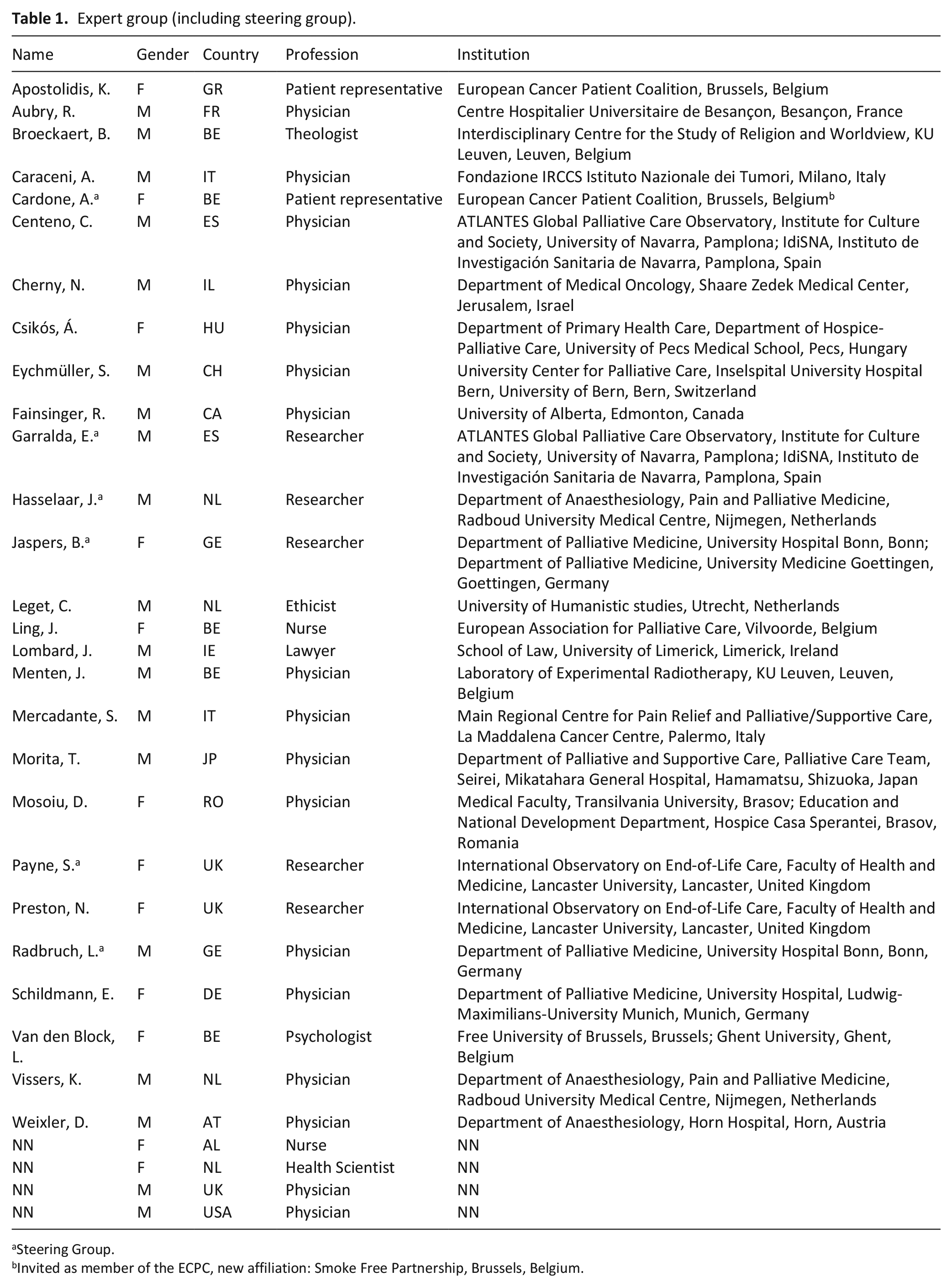
An Expert Group (Table 1) was recruited, including the Steering Group, work package leaders from the EU-funded PalSed project consortium, the advisory board members of the PalSed project and experts on this topic identified through a literature search (criteria: collaboration on palliative sedation guidelines and peer-reviewed publications on palliative sedation).
Expert group (including steering group).
Steering Group.
Invited as member of the ECPC, new affiliation: Smoke Free Partnership, Brussels, Belgium.
In June 2020, the original EAPC 2009 framework was broken down by the study team under the supervision of the Steering Group into 38 statements, some with explanatory text, two assessment tools for monitoring and the list of medications was formatted in the form of a table. They were presented in an online survey (July–November 2020) to the members of the Expert Group, who were asked for each component whether they thought it was still applicable or needed to be revised, and if so, to provide suggestions for revisions or additions including new and relevant literature references. They were also given the opportunity to suggest new statements.
Delphi rounds (steps 2 and 3)
A Delphi Panel of experts was recruited including the Expert Group and additional experts. The latter were identified from a broader literature search, from representatives of the national palliative care associations affiliated with the EAPC or nominated by other panelists. A balance was sought to cover the European regions as far as possible, including also experts from outside Europe, a large range of professional backgrounds (physicians, nurses, psychologists, social workers, ethicists, health researchers and legal experts) and an equal gender distribution. The invitation to take part in the different steps was made by e-mail, and the consensus consultation used an online survey tool (SoSci Survey).
The number of Delphi rounds was set at a maximum of two before the process began.
The level of consensus was rated on a five-point Likert scale either as
low: level of agreement <60%, median <4 and interquartile range (IQR) > 2;
moderate: level of agreement 60%–79% and median <4, IQR > 1;
high: level of agreement ⩾80%–89% and median 5, IQR = 1 and
very high: level of agreement ⩾90% and median 5, IQR = 0.
Statements that did not reach a high or very high level of agreement in the first Delphi-round, as well as statements that, despite reaching a high level of agreement, were the subject of comments and/or constructive suggestions, were to be revised and submitted to the second Delphi-round. If a statement should fail in the second Delphi-round (step 3) to reach the necessary level of consensus, it was decided to mention the statement and the lack of consensus on it and to describe the underlying reasons for the lack of consensus in the discussion.
Step 2: Based on the Expert Group’s feedback, a revised online questionnaire (Supplemental File 1) with 42 statements and explanatory text, a table of medications and four assessment tools for monitoring was presented to the Delphi Panel. In addition to the Likert scale assessment, the opportunity was given to provide comments and literature for each statement and explanatory text (April–June 2021).
Step 3: Statements requiring revision were presented in the second round to the Delphi Panel (Supplemental File 2), again with additional free-text entry fields for comments and literature (November–December 2021).
Final phase (step 4)
The revised draft based on the Delphi rounds and a short glossary (Supplemental Material Data 3) were sent to the Expert Group for a final review.
The final version approved by the Steering Group was presented to the EAPC board of directors for approval in September 2022.
Data management
The data was collected in excel sheets and the quantitative and qualitative analysis were carried out using SPSS Statistics 27.0 and NVivo R 1.61, respectively (HB and SMS). Each proposed amendment was reviewed by HB and SMS, discussed in the working group with BJ and LR and approved by the Steering Group.
Results of the Delphi procedure
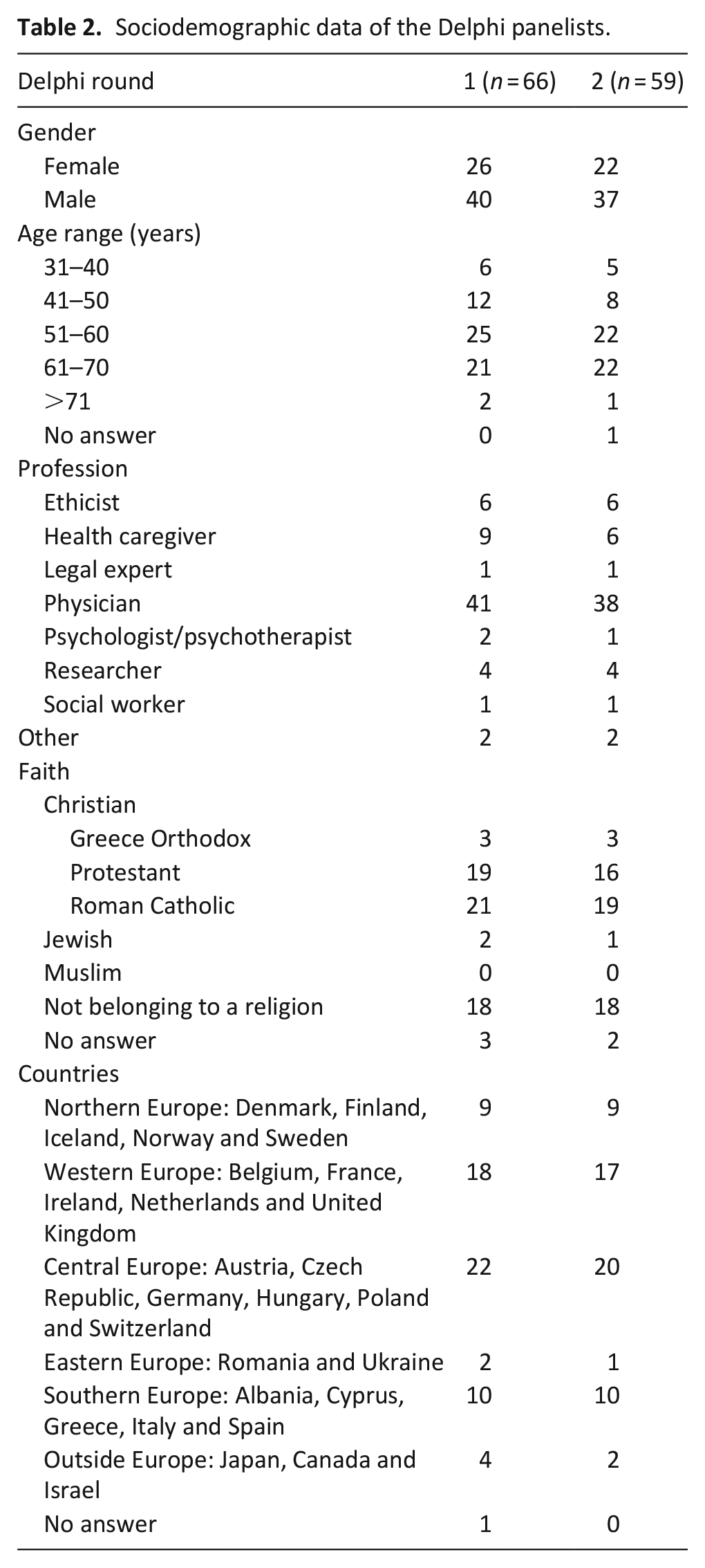
In addition to the Steering Group (n = 6), the work package leaders (n = 8) and advisory board members (n = 4) 15 experts were invited to join the Expert Group. Thirteen responded positively, resulting in an Expert Group of 31 members (Table 1). Twenty-five of them completed the online survey (step 1). All in all, a total of 193 experts (including Steering Group and Expert Group) were invited to participate to the Delphi Panel, and 91 agreed. The first and second Delphi rounds (steps 2 amd 3) were completed by 66 and 59 of these experts (response rate 73% and 65%), respectively (Table 2, Figure 2).
Sociodemographic data of the Delphi panelists.
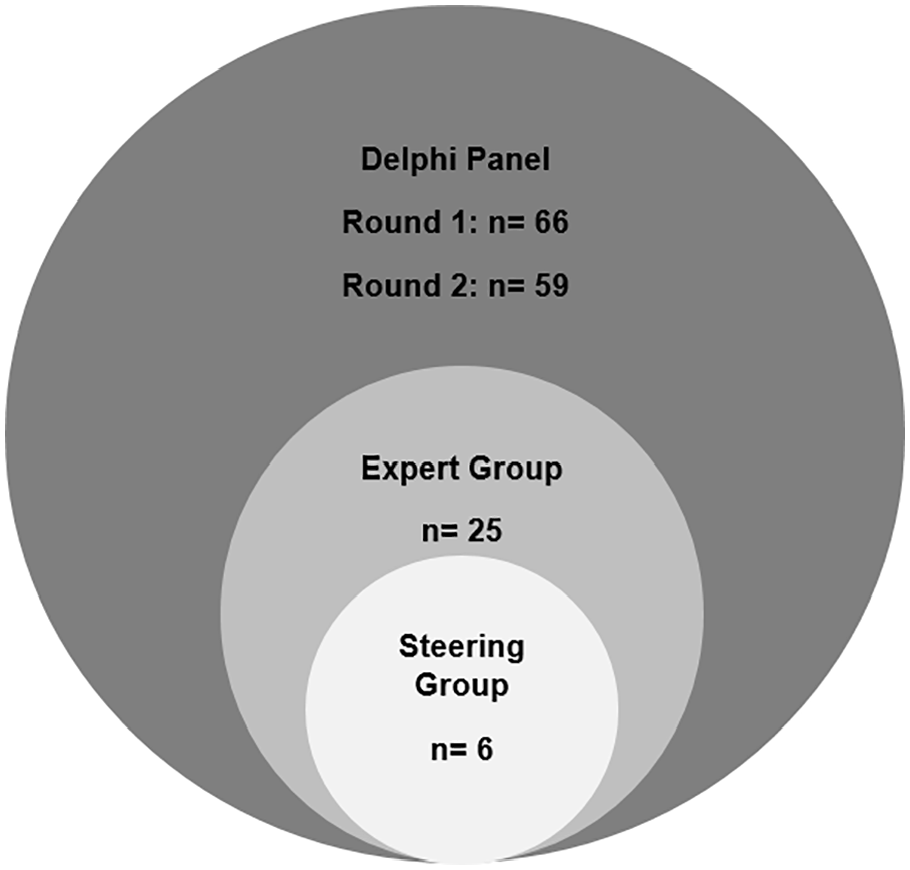
Delphi Panel.
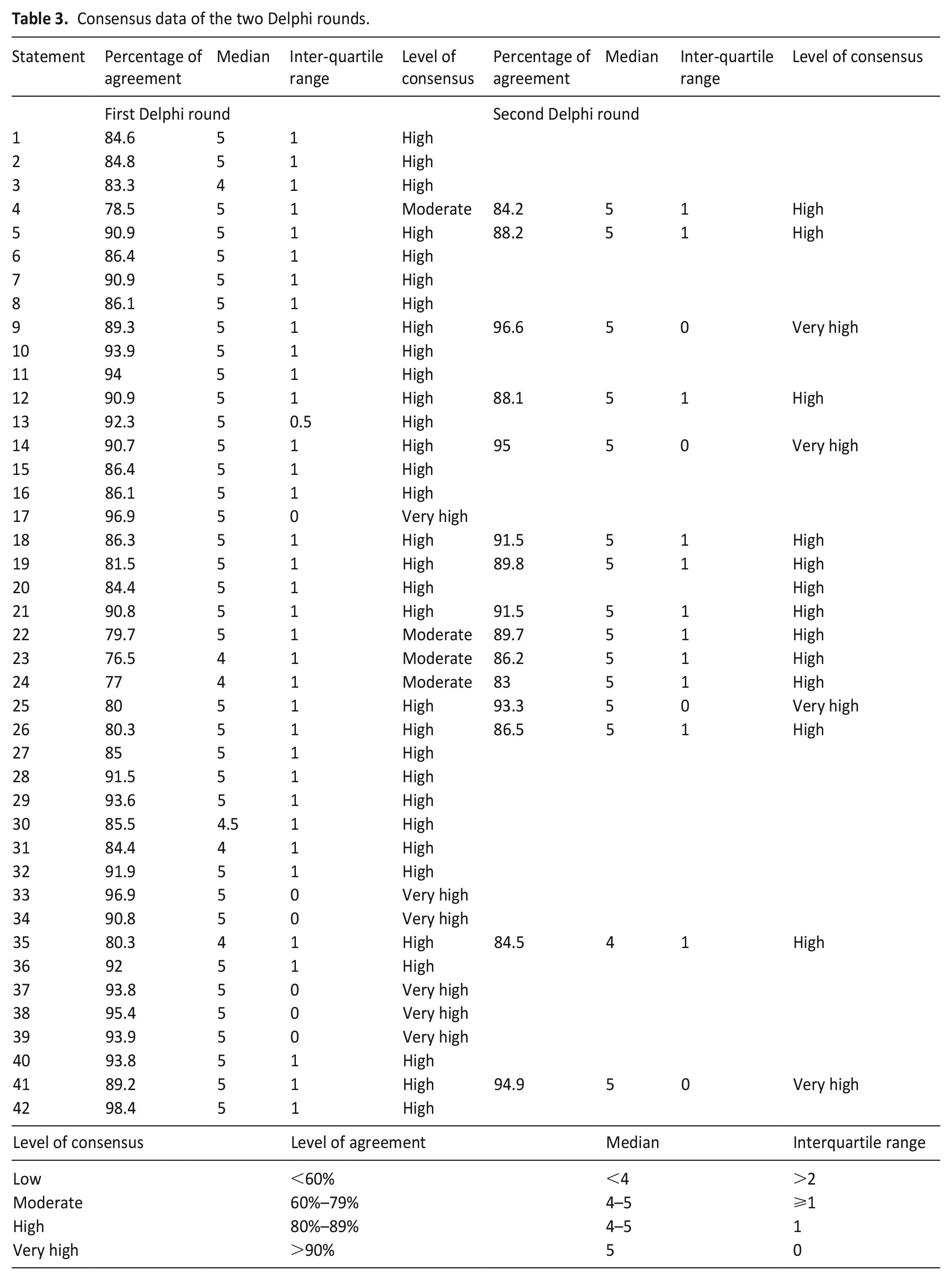
After the first Delphi round, 4 of the 42 statements reached a moderate, 32 a high and 6 a very high level of consensus. Feedback from participants led to significant revisions in 12 statements including explanatory text with high consensus. These statements as well as the four revised statements with moderate consensus concerning the domains indication (statement 4) and selection of method (statements 22–24) were presented to the Delphi Panel in the second round. Following the second Delphi round, 32 statements reached a high and 10 a very high level of consensus (Table 3). These 42 statements with their explanatory text are organised into 12 distinct domains (Table 4) and include a table of medications (Table 5) and four assessment tools for monitoring (the Discomfort Scale – Dementia of Alzheimer Type (DS-DAT),24,25 the Critical-Care Pain Observation Tool (CCPOT), 26 the Patient Comfort Score (PCS)27,28 and the Richmond Agitation Sedation Scale – Palliative Version (RASS-PAL).27,29 –32
Consensus data of the two Delphi rounds.
Domains and statements through the Delphi consensus procedure.

Terms in bold in the final version correspond to editorial changes made based on the expert comments during the Delphi rounds.
 New statements
New statements  Statements with moderate agreement
Statements with moderate agreement
Table of medications.
The amended version included an introduction, method and discussion section and was reviewed by 21 members of the Expert Group (response rate 70%) before being approved by the Steering Group and then by the EAPC Board of Directors, after minor modifications in January 2023.
Discussion
Main findings
More than a decade after the publication of the EAPC framework on palliative sedation this paper presents an update, using a rigorous consensus methodology. A range of experts were involved, with great commitment in the consensus process providing detailed comments and recommending literature, leading to a number of revisions with considerable impact on the framework and resulting in 42 statements in 12 distinct domains. After the two Delphi rounds, a high or very high consensus level was achieved for all statements.
What this study adds?
The main criticisms of the 2009 framework were due partly to the lack of a precise definition of the term refractoriness, the vaguely defined role of the patient (or their representative) in the determination of refractoriness, and the lack of clarity regarding the status of existential suffering as a possible indication for palliative sedation14,20. In the revised framework, the terminology has been more precisely defined. The term suffering (stat. 1) is used to encompass distressing physical and psychological symptoms as well as states of existential suffering to reflect a shift towards a broader recognition of refractory existential suffering as indication for palliative sedation. The term refractory (stats. 2 and 3) described the untreatable (healthcare professionals) and intolerable (patient) nature of the suffering. The consultation of palliative care experts and/or other healthcare experts to determine the untreatable nature of the suffering is strongly recommended before applying palliative sedation (stat. 13).
The literature search revealed that most of the existing publications deal with deep and continuous sedation, probably because this form of sedation is easier to compare between different settings. A broad consensus emerged among the experts on the importance of stressing the principle of proportionality in palliative sedation practice. This principle encompasses proportionality along two main dimensions. First in terms of depth of sedation, with the administration of sedative medications being titrated to the lowest dosage that will provide adequate relief of suffering and maintain interactive function as far as possible, instead of aiming straight at unconsciousness (stats. 1 and 5). Secondly, it also encompasses proportionality in terms of timing, as intermittent sedation will be more appropriate than continuous sedation earlier in the course of the illness to provide temporary relief whilst waiting for treatment benefit from other therapeutic approaches (transient sedation) or to give the patient a break from the current burdensome situation (respite sedation) before regaining consciousness (stat. 1). The physician will therefore first discuss with the patient the form of sedation which is appropriate to their individual situation. Apart from emergency situations at the end of life, a planned interruption of the sedation is recommended, whenever possible, to reassess the situation with the patient. This principle of proportionality has been explicitly introduced into the definition of palliative sedation. Consequently, a sedation that does not meet this principle – for example, deep sedation for moderate symptoms – should not be considered as palliative sedation.
Consideration of remaining life expectancy varied considerably from one guideline to another.12 –14 The revised framework clearly states (stat. 1) that no specific period of remaining life expectancy is defined for the use of palliative sedation based on the three key principles (1) refractoriness of suffering, (2) proportionality and (3) independent decision making for hydration, although deep and continuous sedation is generally indicated in the final stages of life. However, it was not possible to reach consensus in the Delphi process on a precise definition of the final stages of life.
The new statements emphasise the need of timely discussion of patient preferences (stats. 9 and 10) and shared decision-making between patient (and with their consent, their significant others/legal representative) and healthcare professionals (stats. 16, 18 and 19).
The applicability of the framework has been improved by the development of a step-by-step pharmacological approach (stats. 27 and 28) with a detailed description of the recommended medications (Table 5) and more detailed guidance on hydration decision-making based on recent literature (stat. 35).
Nevertheless, even with this high level of agreement, some aspects were debated until the final stage of the consensus process. The comments in question could not always be integrated into the respective statements as they sometimes presented opposing views. The different positions seem to be related to heterogeneous concepts of palliative sedation. Some participants seemed to understand palliative sedation as a merely medical intervention, requiring clear indications and concise definitions and checklists, others highlighted also the social and ethical challenges related to palliative sedation and advocated for a team-based approach with more ethical safety guards, and enough flexibility in the statements for adaptation for the specific patient or cultural context. As an example, several experts expressed concerns about discussing advance care planning (ACP) and in particular palliative sedation with patients at an early stage of the disease. This might reflect reluctance to discuss a medical intervention that is not yet indicated, even though these concerns are at odds with recent studies33,34 showing the benefit of ACP discussion for patient and significant others as a process that occurs in a continuum. 35
In addition, the discussion highlighted the differences in the legal, healthcare and social security systems between European countries. As an example, the role of the legal representative seems to be perceived differently. In some countries, their role is seen predominantly to communicate the patient’s preferences and thus participate in the decision-making process. Within this perspective, they seem to be somewhat protected from the weight of the decision. In other countries their role is to make the final decision after receiving all necessary medical information. Similarly, some expert feedback called for mandatory consultation with palliative care experts before initiation of palliative sedation, whereas others rejected this as it would disadvantage patients based on territorial disparity in access to specialist palliative care. 36
Strengths and limitations
In consequence of the above-mentioned differences, as with all European standards this framework might need adaptation by clinicians and health policy decision-makers to the specific national or local context with high quality procedural guidelines related to their specific legislation, regulations and culture.
Nevertheless, this is the first framework on palliative sedation using a strict consensus methodology involving experts from all over Europe and beyond (28 countries) with a broad range of professions, as well as a European patient organisation. All European regions (North, South, Central, East and West Europe; Table 2) were involved. Unfortunately, even though a gender balance in our invitations was sought, in the Delphi panel male participants predominated. The online Delphi survey was conducted in English after participants confirmed that the offer of translation into French, Spanish and Russian was not necessary. Literature search for the scoping reviews was limited to publications in English, resulting in a potential language bias. However, the analysis of existing guidelines on palliative sedation in the eight participating European countries11,12,37 –44 and the experts’ recommended literature informed the framework with a number of references in other languages than English. The updated literature shows that there is still a lack of clinical trials and prospective studies. 2 The EAPC task force on palliative sedation will therefore, five years after publication of the framework, evaluate the need for an update on the basis of new evidence and changing thinking patterns. They will also audit the use of the new framework in Europe.
The full framework with explanatory texts and references will be available on the EAPC website in English (Supplemental File 4), and also other languages. It will be also disseminated through an educational programme (MOOC; https://www.futurelearn.com/courses/dying-well-the-role-of-palliative-care) and an e-book.
Conclusion
The revised framework including proposed assessment tools for monitoring, a comprehensive table of medications, dosages and administration forms, serves as comprehensive and soundly developed information for healthcare professionals on the use of palliative sedation as well as for medical associations and health policy decision-makers. Since its development considered all relevant methodological and content aspects of AGREE II, 24 an international tool to assess the quality and reporting of practice guidelines, it may be used as a guideline or serve as a basis for cultural adaptation of guidelines on palliative sedation.
Supplemental Material
sj-docx-3-pmj-10.1177_02692163231220225 – Supplemental material for Revised European Association for Palliative Care (EAPC) recommended framework on palliative sedation: An international Delphi study
Supplemental material, sj-docx-3-pmj-10.1177_02692163231220225 for Revised European Association for Palliative Care (EAPC) recommended framework on palliative sedation: An international Delphi study by Séverine M Surges, Holger Brunsch, Birgit Jaspers, Kathi Apostolidis, Antonella Cardone, Carlos Centeno, Nathan Cherny, Àgnes Csikós, Robin Fainsinger, Eduardo Garralda, Julie Ling, Johan Menten, Sebastiano Mercadante, Daniela Mosoiu, Sheila Payne, Nancy Preston, Lieve Van den Block, Jeroen Hasselaar and Lukas Radbruch in Palliative Medicine
Supplemental Material
sj-docx-4-pmj-10.1177_02692163231220225 – Supplemental material for Revised European Association for Palliative Care (EAPC) recommended framework on palliative sedation: An international Delphi study
Supplemental material, sj-docx-4-pmj-10.1177_02692163231220225 for Revised European Association for Palliative Care (EAPC) recommended framework on palliative sedation: An international Delphi study by Séverine M Surges, Holger Brunsch, Birgit Jaspers, Kathi Apostolidis, Antonella Cardone, Carlos Centeno, Nathan Cherny, Àgnes Csikós, Robin Fainsinger, Eduardo Garralda, Julie Ling, Johan Menten, Sebastiano Mercadante, Daniela Mosoiu, Sheila Payne, Nancy Preston, Lieve Van den Block, Jeroen Hasselaar and Lukas Radbruch in Palliative Medicine
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163231220225 – Supplemental material for Revised European Association for Palliative Care (EAPC) recommended framework on palliative sedation: An international Delphi study
Supplemental material, sj-pdf-1-pmj-10.1177_02692163231220225 for Revised European Association for Palliative Care (EAPC) recommended framework on palliative sedation: An international Delphi study by Séverine M Surges, Holger Brunsch, Birgit Jaspers, Kathi Apostolidis, Antonella Cardone, Carlos Centeno, Nathan Cherny, Àgnes Csikós, Robin Fainsinger, Eduardo Garralda, Julie Ling, Johan Menten, Sebastiano Mercadante, Daniela Mosoiu, Sheila Payne, Nancy Preston, Lieve Van den Block, Jeroen Hasselaar and Lukas Radbruch in Palliative Medicine
Supplemental Material
sj-pdf-2-pmj-10.1177_02692163231220225 – Supplemental material for Revised European Association for Palliative Care (EAPC) recommended framework on palliative sedation: An international Delphi study
Supplemental material, sj-pdf-2-pmj-10.1177_02692163231220225 for Revised European Association for Palliative Care (EAPC) recommended framework on palliative sedation: An international Delphi study by Séverine M Surges, Holger Brunsch, Birgit Jaspers, Kathi Apostolidis, Antonella Cardone, Carlos Centeno, Nathan Cherny, Àgnes Csikós, Robin Fainsinger, Eduardo Garralda, Julie Ling, Johan Menten, Sebastiano Mercadante, Daniela Mosoiu, Sheila Payne, Nancy Preston, Lieve Van den Block, Jeroen Hasselaar and Lukas Radbruch in Palliative Medicine
Footnotes
Acknowledgements
We acknowledge for their input all H2020 PalliativeSedation (SEP-210506914) consortium members from KU Leuven (Belgium), University of Pécs (Hungary), La Maddalena Cancer Center (Italy), Radboud University Medical Center, Nijmegen (Netherlands), Hospice Casa Sperantei (Romania), University of Navarra (Spain), Lancaster University (UK), European Cancer Patient Coalition (ECPC) and European Association for Palliative Care (EAPC; ![]() ). We wish also to acknowledge the other experts who contributed to the development of this document including R. Aubry, B. Broeckaert, A. Caraceni, S. Eychmüller, C. Leget, J. Lombard, T. Morita, E. Schildmann, K. Vissers and D. Weixler. In parallel with the European Union funded project PalSed an EAPC Task Force on Palliative Sedation was established in 2019 with J. Hasselaar and L. Radbruch as chairs, with the remit to produce this update. This task force was approved by the EAPC board and has reported at recent EAPC conferences.
). We wish also to acknowledge the other experts who contributed to the development of this document including R. Aubry, B. Broeckaert, A. Caraceni, S. Eychmüller, C. Leget, J. Lombard, T. Morita, E. Schildmann, K. Vissers and D. Weixler. In parallel with the European Union funded project PalSed an EAPC Task Force on Palliative Sedation was established in 2019 with J. Hasselaar and L. Radbruch as chairs, with the remit to produce this update. This task force was approved by the EAPC board and has reported at recent EAPC conferences.
Author contributions
Conception and design of the study: HB, BJ, LR and SMS. Steering Group: AC, EG, JH, BJ, SP and LR. Data analysis: HB, BJ, LR and SMS. HB and SMS led the writing of the script. All authors (KA, HB, AC, ÀC, CC, NC, RF, EG, JH, BJ, JL, JM, SM, DM, SP, NP, LR, SMS and LVdB) provided feedback on one or more versions of the draft and contributed to the development of the paper. All authors revised and approved the final manuscript.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that there is no conflict of interest. The authors (apart from HB and SMS) participated in the Delphi process. However, the two researchers coordinating the consensus process and the evaluation (HB and SMS) did not participate, thus preventing any bias in the evaluation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 825700. The funding body had no role in the design and conduct of the study; collection, management, analysis and interpretation of the data; preparation, review and approval of the manuscript; or decision to submit the manuscript for publication.
Research ethics and patient consent
The study was approved by the Ethics Commission, University of Bonn, Germany, No. 268/20 (19 June 2020).
Data management and sharing
More detailed information on the consensus process results will be published separately.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.