Abstract
Mercury pollution in aquatic systems poses a critical environmental challenge due to its high mobility, toxicity, and bioaccumulation potential. In this study, the synthesis of floating alginate–bentonite (FAB) and floating alginate–
Introduction
Heavy metal pollution in aquatic environments has become a growing global concern, mainly due to increasing anthropogenic activities (Aziz et al., 2023). Among these pollutants, mercury (Hg) species are particularly critical because of its high mobility and bioaccumulation potential, which lead to severe ecological and human health risks. In ecosystems, mercury can inhibit plant growth and photosynthetic activity while disrupting key metabolic processes (Singh, 2023). In humans and aquatic species, chronic exposure is associated with pulmonary dysfunction, renal impairment, acute neurotoxicity, and central nervous system damage (Wang et al., 2021).
Colombia is currently among the 21 countries with the highest per capita mercury emissions, releasing more than 100 tons annually, which are subsequently distributed into major rivers and tributaries nationwide (Diaz et al., 2022). This widespread contamination has caused adverse effects on biodiversity and poses severe risks to local communities that depend directly on these water resources (Harry et al., 2018). Alarmingly, mercury levels as high as 47.0 μg g⁻1 have been detected in human populations from the Colombian Amazon, primarily due to the consumption of contaminated fish and water (Olivero-Verbel et al., 2016).
To mitigate mercury contamination in aquatic systems, several treatment technologies have been developed, including chemical precipitation (Matebese et al., 2024), ion flotation (Dhokpande et al., 2024), ion exchange (Zhao et al., 2019), coagulation–flocculation (Mac Mahon, 2022), and electrochemical methods (Wu et al., 2019). Among these, adsorption stands out as the most efficient, cost-effective, and versatile approach for the removal of Hg(II) from aqueous media (Rajendran et al., 2022). Adsorption also provides advantages in terms of reusability and adaptability to a wide range of natural, mineral, and polymeric adsorbents (Upadhyay et al., 2021). Various sorbents have been reported, such as biochar (Yang et al., 2021), conjugated nanomaterials (Rani et al., 2022), and natural clays, which are particularly attractive due to their high specific surface area, ion-exchange capacity, structural stability, low toxicity, and wide availability (Prabhu and Prabhu, 2018).
Recently, composites based on natural biopolymers such as sodium alginate (SA) have gained increasing attention owing to their unique features, including high biocompatibility, biodegradability, and abundant hydroxyl and carboxyl groups that strongly interact with metal ions (Gao et al., 2019). When combined with clay minerals or zeolites, alginate-based composites display enhanced adsorption capacity and improved mechanical resistance, as reported for alginate–zeolite and alginate–clay beads (Das et al., 2021). Despite these advances, a major limitation remains their recovery after use, which often requires additional separation steps and increases the overall cost of treatment.
To overcome this challenge, the development of floating and reusable alginate-based composites represents a promising strategy. Floating adsorbents not only facilitate recovery after treatment but also expand their applicability in dynamic aquatic environments. In this context, the present study aims to design and evaluate floating alginate–bentonite (FAB) and floating alginate–
Material and methods
Preparation of aluminosilicate precursors
For the synthesis of the adsorbent composites, inorganic phases based on aluminosilicates were used, selected for their known cation exchange capacity, thermal stability, and affinity for heavy metals. In this study, purified natural bentonite (B) and hydrothermally synthesized
Purification Of bentonite
A natural bentonite from Valle del Cauca, Colombia, classified as sonoite type, was used. A suspension of the clay was prepared at a concentration of 83 g L−1 in deionized water, to which 1.0 wt% sodium hexametaphosphate was added. The mixture was stirred for 24 h. Subsequently, the supernatant was separated by centrifugation at 4000 r/min for 5 min. The recovered supernatant was dried at 60 °C, ground in an agate mortar, and sieved through a 1 mm mesh.
Synthesis of β -zeolite
The hydrothermal synthesis of
Synthesis of floating alginate/aluminosilicate composites
The synthesis of the alginate/aluminosilicate composites was carried out based on the procedure described by (Wang et al., 2018). Initially, two SA solutions were prepared: one at 2% w/v by dispersing 4 g of SA in 100 mL of deionized water, and the other at 4% w/v by dispersing 8 g in the same volume. Both solutions were stirred at 1000 r/min for 24 h. Subsequently, these solutions were used to prepare suspensions with either B or
After completing the previous steps, the composites were subjected to a crosslinking process in 100 mL of a 0.5 mol L−1 CaCl2·2H2O solution for 4 h at room temperature, aiming to achieve complete gelation. Afterwards, the composites were rinsed with deionized water to remove any residual calcium ions. The composites were subsequently frozen again at −41 °C for 24 h and lyophilized to obtain a porous sponge-like structure. The materials were cut into small cubes with dimensions of 6 mm × 6 mm (Figure 1) and stored in a desiccator until characterization and subsequent use in experimental procedures. Composites formulated with bentonite were designated as FAB, whereas those prepared with

Macroscopic appearance and dimensions of floating alginate-based composites: FAZ1.0 (alginate–
Statistical optimization of floating alginate–aluminosilicate composites
A statistical study was conducted to optimize the synthesis process of floating composite materials based on aluminosilicate precursors (B and
A central composite design (CCD) was employed using Design-Expert® software version 11 to evaluate the combined effect of three independent factors on the response variable, namely the buoyancy percentage (
Model fitting was validated through analysis of variance (ANOVA), and its adequacy was assessed using statistical indicators such as the coefficient of determination (
The resulting regression equation, corresponding to a quadratic model, is expressed as follows:
Functional evaluation: Buoyancy study
The buoyancy evaluation was conducted based on the procedure developed by Dalponte et al. (2019). A total of 250 mg of the FAB or FAZ composites were added to Erlenmeyer flasks containing 100 mL of deionized water and subjected to constant stirring at 250 r/min at room temperature. After 24 h, the floating and sedimented composites were separated and dried individually. Subsequently, the respective fractions were weighed to calculate buoyancy using the following equation: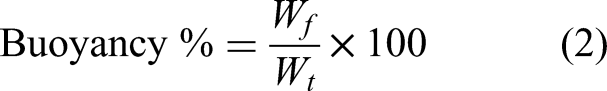
Material characterization
TG–DTA was performed on an SDT 650, TA Instruments, at a heating rate of 10 °C/min from room temperature to 800 °C under nitrogen atmosphere. FTIR–ATR spectra were recorded on a Nicolet IS50, Thermo Scientific spectrometer, in the range of 4000–400 cm⁻1. XRD patterns were obtained on a Rigaku Miniflex diffractometer, operating at 40 kV and 30 mA, equipped with a D/teX Ultra detector and a Cu
Kinetic adsorption study
The kinetic adsorption study of Hg(II) in aqueous solution on FAB or FAZ was conducted using 250 mg of the adsorbent material and 100 mL of HgCl2 aqueous solution (CAS No. 7487-94-7, 99.5% purity, Thermo Scientific Chemicals) at an initial concentration of 200 mg L⁻1, adjusted to pH ∼ 6.5 with 0.1 M NaOH or HCl. The experiments were carried out in Erlenmeyer flasks under constant agitation in an orbital shaker (Maxshake™ OB2) at 250 r/min and room temperature (22 °C) over a period of 240 min. Aliquots of 100 μL were periodically withdrawn for mercury quantification by differential pulse anodic stripping voltammetry, following the methodology previously reported (Ruiz-Bravo et al., 2025), every 5 min during the first 60 min and subsequently every 30 min until the completion of the 240-min run. All experiments were performed in triplicate.
The amount of Hg(II) adsorbed at time 
The kinetic data were fitted using nonlinear models (Wang et al., 2024), as described in Table 1.
Kinetic adsorption nonlinear models.

PFO: pseudo-first-order; PSO: pseudo-second-order.
Evaluation of reusability cycles
To experiment the reusability of the material, three adsorption cycles were carried out following the methodology described in the “Kinetic adsorption study” section. After each cycle, the composites were washed with 0.1 mol L−1 HCl for 2 h to remove the adsorbed Hg(II). Subsequently, they were rinsed with deionized water and dried in an oven at 55 °C for 3 h. After the wash step using HCl, the Hg(II)-containing acidic solutions were collected and stored in appropriate, labeled containers and handled in accordance with institutional safety protocols and local environmental regulations for hazardous mercury-containing waste. No direct discharge to the environment occurred at any stage of the experimental procedure.
Results and discussion
Experimental optimization and statistical analysis
The optimization of composite buoyancy was evaluated using a full factorial CCD with three independent factors: SA mass fraction (
Experimental factors, coded levels, and design matrix with flotation response in the central composite design.
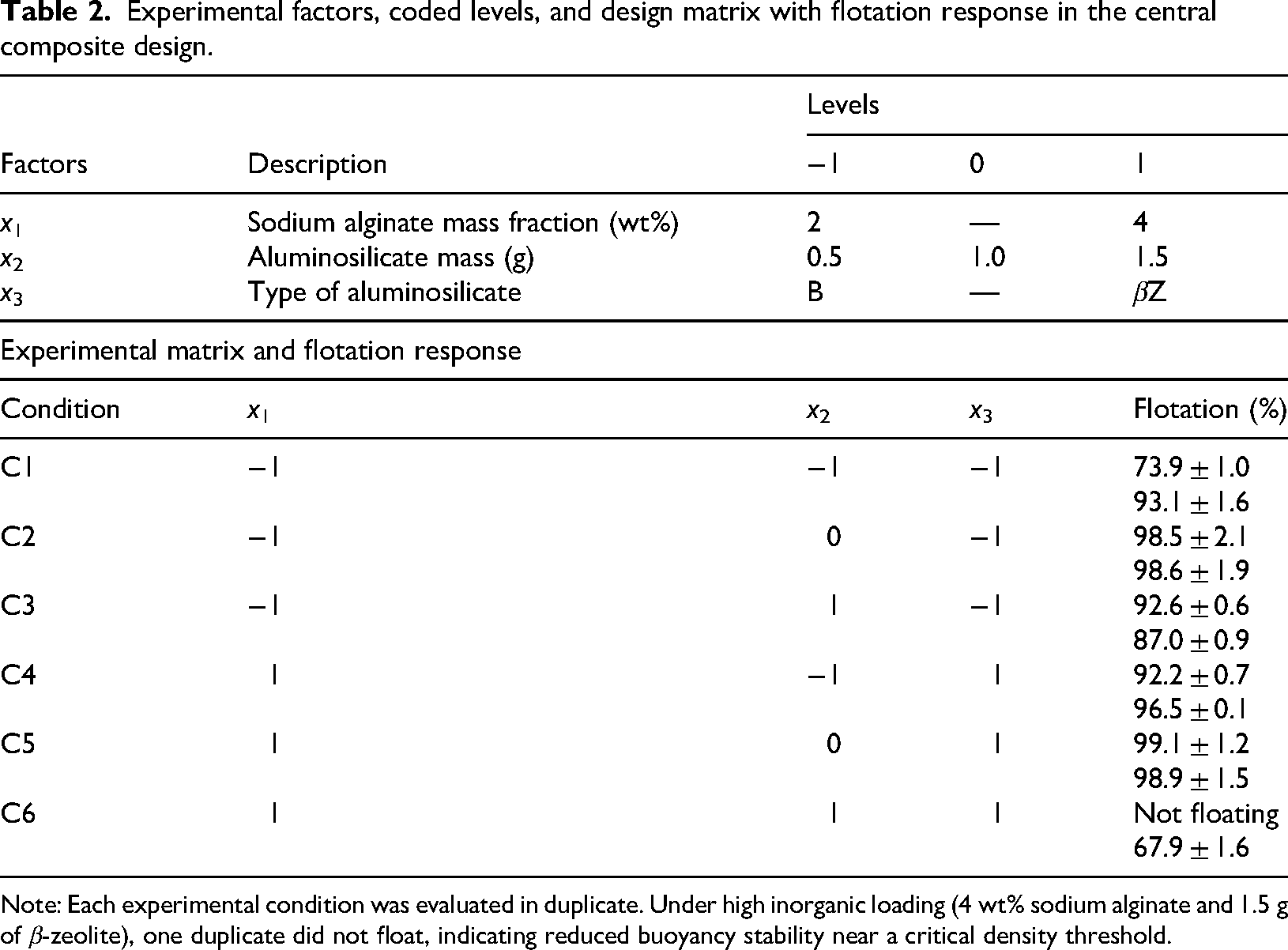
Note: Each experimental condition was evaluated in duplicate. Under high inorganic loading (4 wt% sodium alginate and 1.5 g of
The ANOVA for the significant terms of the quadratic model (Table 3) revealed a statistically significant relationship between the independent variables and the flotation of the composites (
ANOVA, significant terms of the quadratic model for composite flotation, and model fitting statistics.
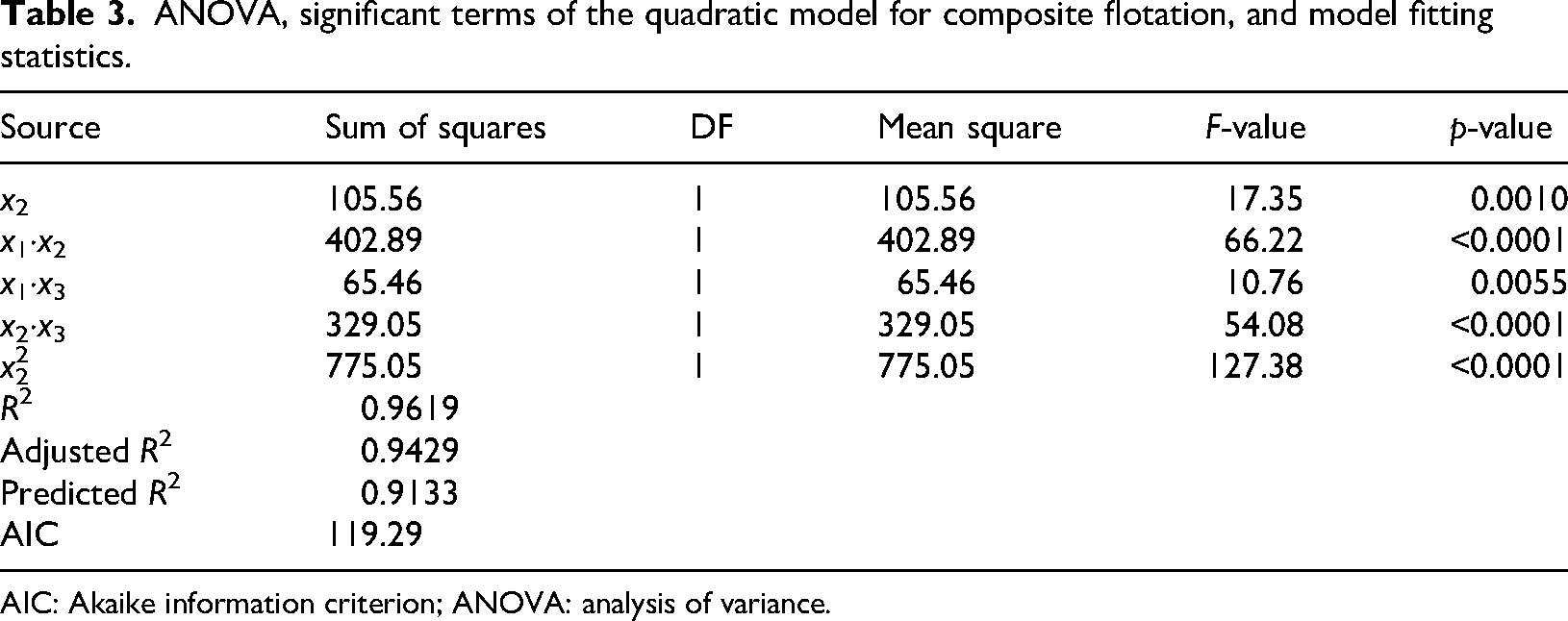
AIC: Akaike information criterion; ANOVA: analysis of variance.
The polynomial equation for flotation (Equation (4)) indicates that this property is primarily influenced by factor interactions rather than by individual effects. The interaction terms 
The response surfaces (Figure 2(a) and (b)) and the corresponding contour plots (Figure 2(c) and (d)) for the set of synthesized composites confirm the aforementioned findings. An optimal flotation region was observed under intermediate to high aluminosilicate mass condition (∼1.0 g) combined with a 4% alginate fraction. The surface corresponding to
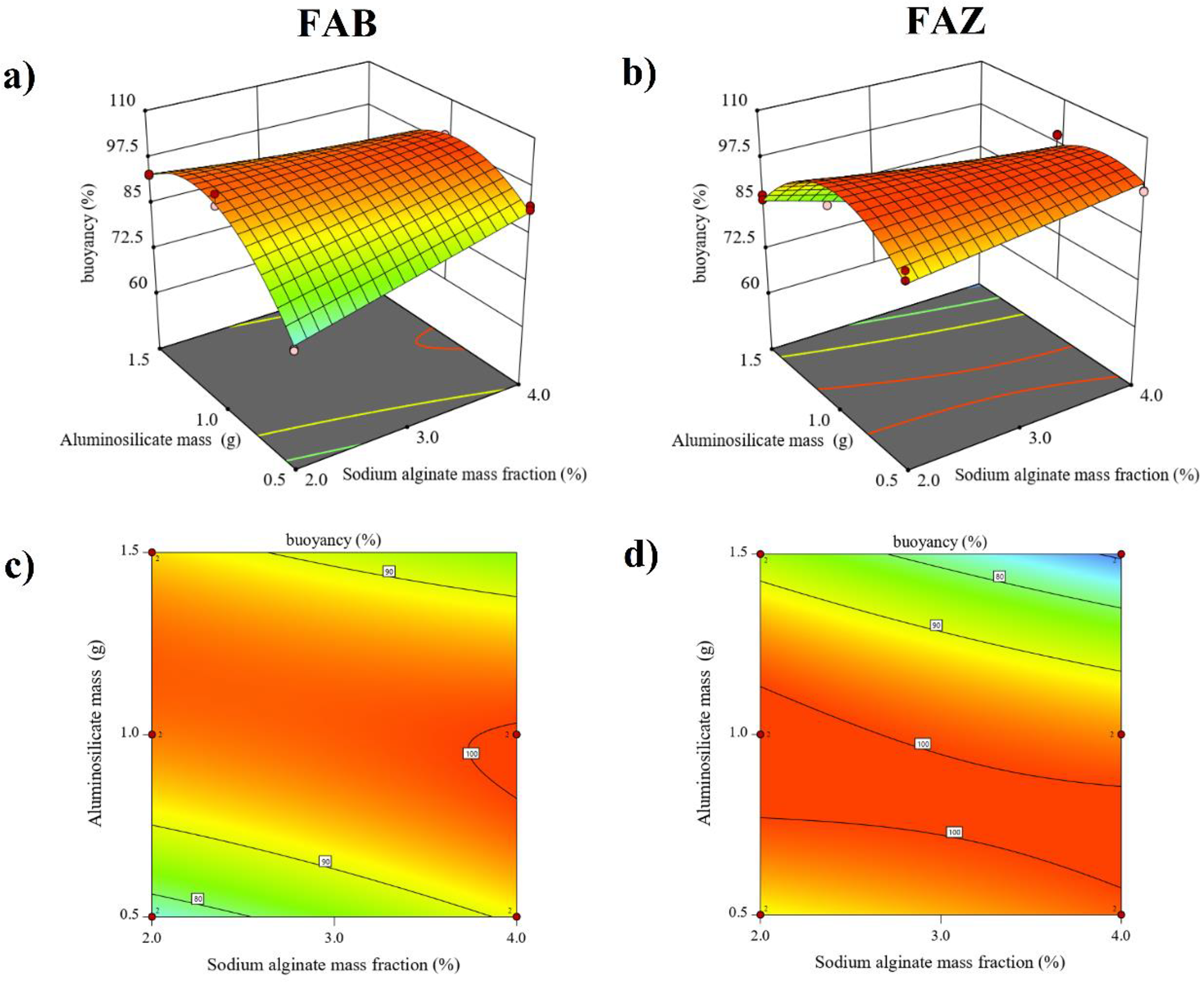
3D surface plots of (a) FAB and (b) FAZ composites; 2D contourn plots of (c) FAB and (d) FAZ composites. 3D: three-dimensional; 2D: two-dimensional; FAB: floating alginate–bentonite; FAZ: floating alginate–
Based on the statistical and graphical analysis, the CCD predicted an optimal formulation corresponding to 4% SA and 1.0 g of aluminosilicate for both bentonite and
Thermal, structural, morphological, and surface characterization of the optimized composites
Thermogravimetric and differential thermogravimetric analysis
Thermogravimetric analysis of the FAB1.0 and FAZ1.0 composites (Figure 3(a), blue and green lines, respectively) revealed total mass losses of approximately 54.4% and 55.8%. These losses reflect the combined decomposition of the organic fraction (alginate), the removal of adsorbed and structural water, and the partial carbonization of residual material. Although the initial preparation involved a 4 wt% SA solution, most of the water is removed during drying and crosslinking, and not all of the polymer becomes fully integrated into the composite matrix. Consequently, the final solid exhibits a relatively high organic content, a phenomenon widely reported for alginate-based hybrid materials (Baigorria et al., 2020; Bukit et al., 2024; Edathil et al., 2018; Giriyappa Thimmaiah et al., 2022; Liaqat et al., 2022; X. Wang et al., 2022b; Xu et al., 2024).
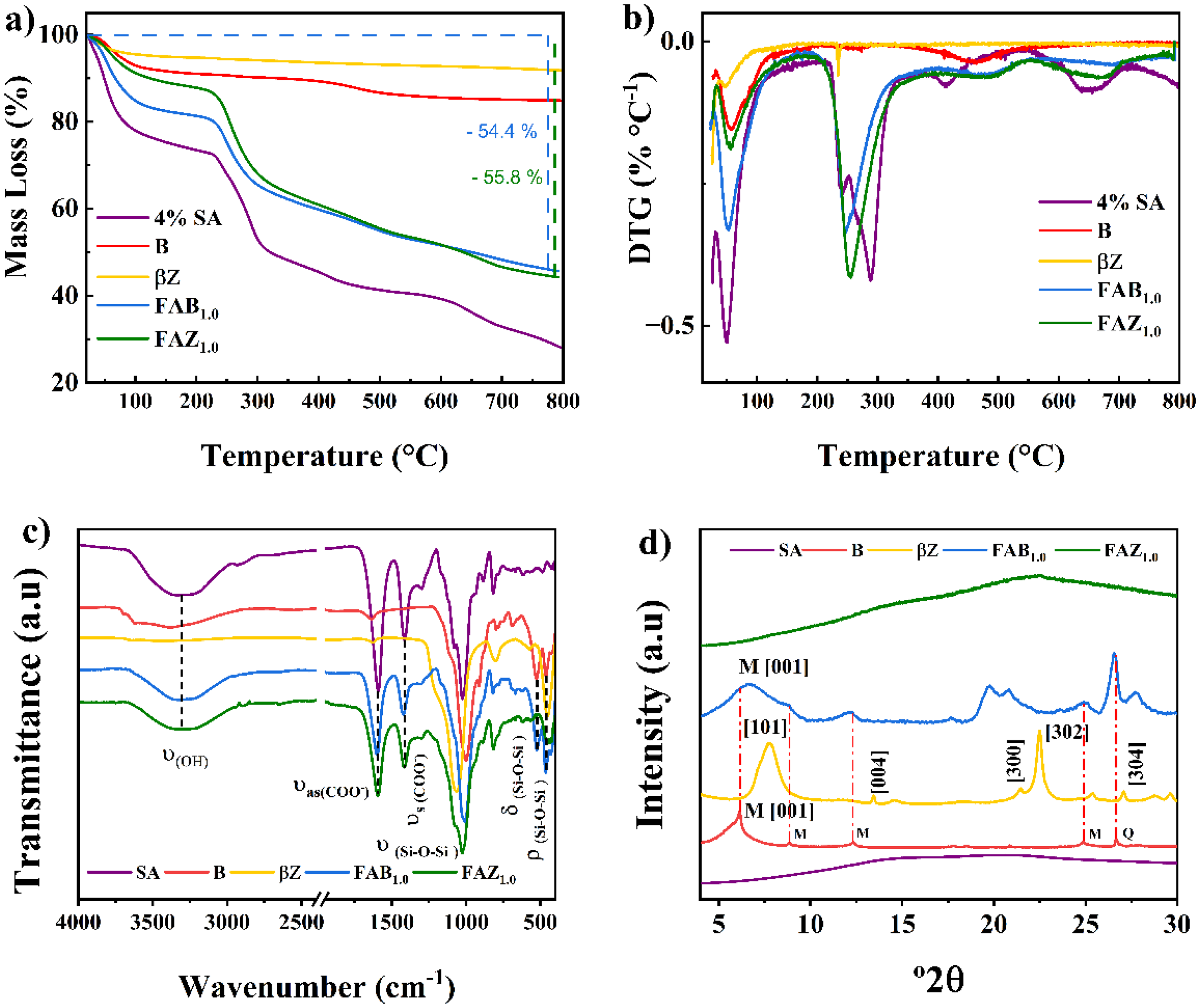
(a) TG of SA, B,
The main thermal event associated with alginate degradation occurred between 250 and 400 °C, while the ranges of 30–150 °C and 400–550 °C corresponded to water desorption and final carbonization, respectively. Accordingly, the functional organic content (alginate + structural water + carbonaceous residues) was estimated to account for 50%–56% of the total mass, consistent with the observed losses. The remaining 44%–50% of the mass was attributed to the inorganic phase (B or
The relatively high proportion of the organic phase is a key factor in conferring flotation capacity to the composite (>98%), as it reduces the apparent density of the material and generates a porous structure. In addition, the presence of alginate functional groups (–COO⁻, –OH) could potentially contribute to the adsorption of metal cations such as Hg(II) through coordinated or electrostatic interactions, as discussed later.
DTG analysis (Figure 3(b)) showed a pronounced decomposition peak around 120 °C, associated with the removal of weakly bound water within the polymeric matrix. In the 270–320 °C range, more intense peaks corresponding to the thermal degradation of SA were identified. In the FAB1.0 and FAZ1.0 composites, these peaks shifted slightly toward higher temperatures and displayed lower intensities compared to pure alginate. This behavior indicates a stabilizing effect induced by the inorganic phase, which is attributed to interfacial interactions such as hydrogen bonding between alginate carboxyl groups and aluminosilicate surface sites. These interactions restrict polymer chain mobility and delay thermal decomposition. Similar stabilization effects have been reported for clay–alginate hybrid systems (Coşkuner Filiz et al., 2025; Fernando et al., 2019; Hellal et al., 2023; Pawar et al., 2018; Radoor et al., 2020; Syah et al., 2025) and are consistent with the enhanced macroscopic cohesion observed experimentally in the composites.
FTIR–ATR
The infrared spectrum of SA (Figure 3(c), purple line) exhibited a broad band centered at
In the FAB1.0 and FAZ1.0 composites (Figure 3(c) blue and green lines), characteristic bands of alginate were preserved, although with slight shifts. For the FAB1.0 composite, bands were identified at
X-ray diffraction
The XRD analysis (Figure 3(d)) revealed a contrasting structural behavior between the FAB1.0 and FAZ1.0 composites. The FAB1.0 composite retained diffraction peaks characteristic of bentonite (Figure 3(d), red line) corresponding to montmorillonite and quartz phases (M and Q), indicating partial intercalation of the biopolymer within the montmorillonite layers rather than complete exfoliation. This effect is consistent with the decrease in the basal spacing of the [001] reflection from
In contrast, the FAZ1.0 composite exhibited an amorphized pattern, without defined diffraction signals, indicating a complete loss of the crystalline order of
Field emission scanning electron microscopy
FE-SEM micrographs at the macroscale (500 µm) revealed remarkable structural contrasts among the materials (Figure 4(a)–(c)). The SA sponge (SA, Figure 4(a)) exhibited a homogeneous porous network with large, well-defined cavities, which is characteristic of freeze-dried gels without inorganic loading. This open architecture is consistent with the features observed at the microscale (10 µm) (Figure 4(d)) and indicates good pore connectivity. However, such structures typically present limited mechanical strength, as commonly reported for freeze-dried alginate hydrogels (Wang et al., 2022c). In contrast, the FAB1.0 composite (Figure 4(b)) displayed a denser macrostructure with partially collapsed regions and scarce visible porosity. This morphology is consistent with the homogeneous incorporation of laminar bentonite particles and the rough surface observed at the microscale (Figure 4(e)). The presence of bentonite promotes the formation of a more compact framework, which arises from strong interactions between alginate carboxylate groups and clay surfaces. Similar structural features have been described for alginate–montmorillonite nanocomposites and are associated with improved cohesion of the polymeric matrix (H. Zhang et al., 2020a). The FAZ1.0 composite (Figure 4(c)) exhibited a markedly heterogeneous macrostructure, characterized by crystalline aggregates and irregular pore distribution. These features correlate with the limited dispersion of

Representative FE-SEM images of the floating composite sponges SA, FAB1.0, and FAZ1.0 at 500 µm (a–c) and 10 µm (d–f). FE-SEM: field emission scanning electron microscope; FAB: floating alginate–bentonite; FAZ: floating alginate–
Textural properties
The N2 adsorption–desorption isotherms of the pristine aluminosilicate precursors (B and
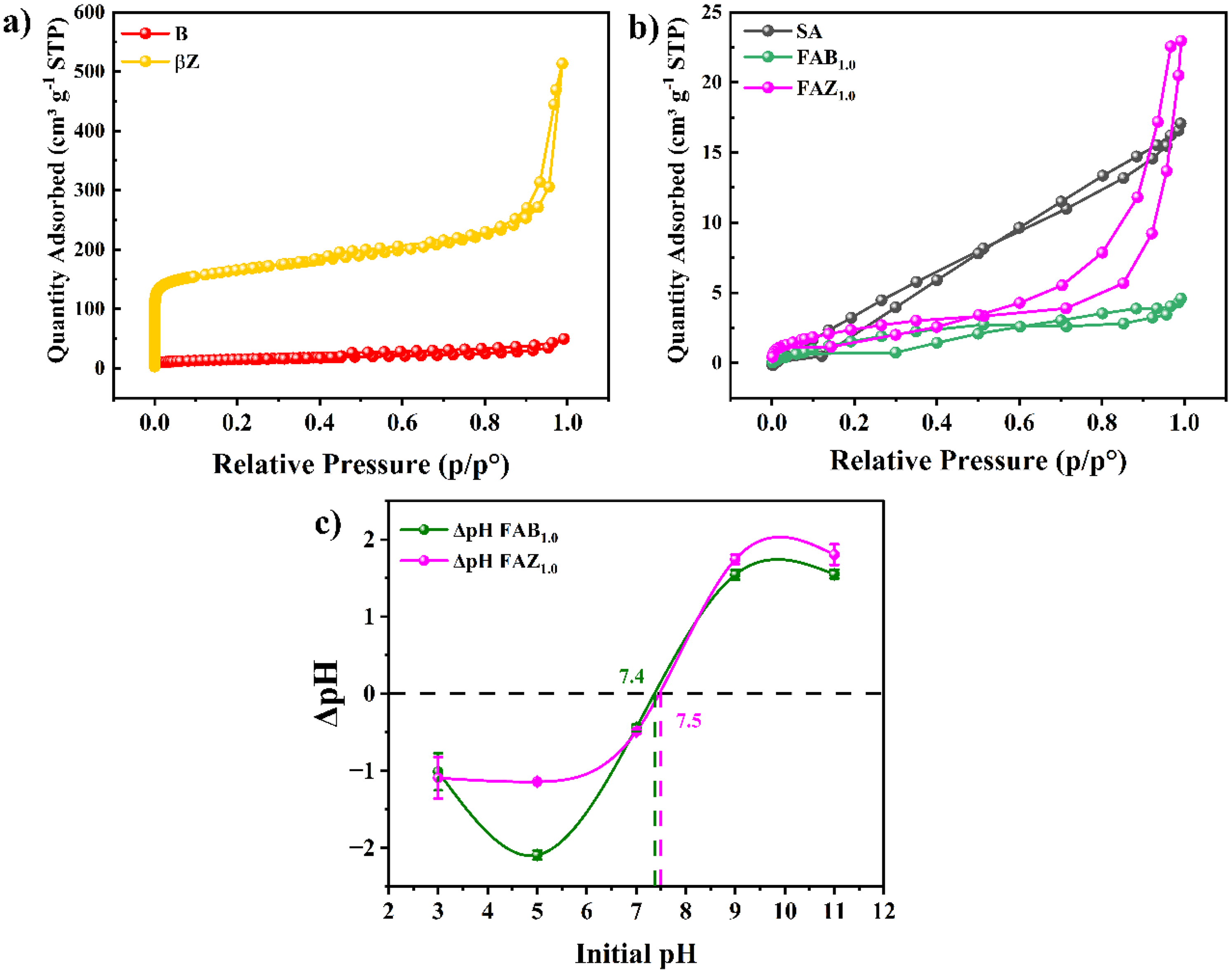
(a) Adsorption–desorption isotherms of the pristine inorganic precursors (B and
Textural properties of pristine inorganic precursors and floating composites.

FAB: floating alginate–bentonite; FAZ: floating alginate–
The SA material (Figure 5(b)) displayed a type III isotherm according to the IUPAC classification (Rahman et al., 2019), characterized by a convex adsorption curve over the entire relative pressure range and the absence of a well-defined plateau, indicative of weak adsorbate–adsorbent interactions. Narrow hysteresis loop were observed both in the low to mid relative pressure region (
Upon incorporation of bentonite, FAB1.0 (Figure 5(b)) exhibited an incipient type IV isotherm with an H3-type hysteresis loop, characteristic of materials containing slit-shaped mesopores or inter-aggregate porosity arising from lamellar structures (Zhang et al., 2020a). Despite the intrinsic mesoporosity of B, FAB1.0 showed a pronounced decrease in BET surface area and total pore volume relative to both pristine B and SA (Table 4). This behavior strongly suggests partial pore blocking and structural densification of the hybrid matrix, likely caused by alginate infiltration into clay interlayers and collapse of interparticle voids during freeze-drying, as previously reported for alginate–clay composites (H. Zhang et al., 2020a). The average pore diameter of FAB1.0 remained within the lower mesoporous range, consistent with a compacted hybrid structure.
FAZ1.0 displayed a type IV isotherm with a pronounced adsorption increase at high relative pressures (
With respect to surface affinity, the BET constants increased systematically with the incorporation of aluminosilicates, following the order SA < FAB1.0 < FAZ1.0 (Table 4). This trend reflects a substantial enhancement in adsorbate–adsorbent interaction energy, which can be attributed to the presence of inorganic active sites and stronger solid–gas interactions provided by the aluminosilicate phases (Wang et al., 2022c). Notably, although FAZ1.0 does not recover the high BET constant of pristine
The textural results confirm that the combination of alginate with aluminosilicates alters the porous architecture of the floating composite sponges. While hybridization leads to a substantial reduction in BET surface area relative to pristine inorganic precursors, it simultaneously promotes the development of mesoporous domains, enhanced surface affinity, and tailored adsorption capacities. Such a textural redesign is particularly advantageous for environmental remediation and controlled-release applications, where pore size distribution, accessibility, and interaction strength are often more critical than absolute surface area (Panayotova, 2022; Wang et al., 2022c).
Surface charge behavior and point of zero charge (pHPZC)
The point of zero charge (pHₚzc) of the FAB1.0 and FAZ1.0 composites, determined by the pH drift method, was found at 7.4 and 7.5, respectively (Figure 5(c)). These values indicate that at pH < pHpzc, the material surfaces carry a net positive charge due to the protonation of functional groups such as carboxyl (–COOH) moieties from alginate and silanol groups (≡Si–OH) from the inorganic phase. This surface environment modulates the interaction with dissolved mercury. However, according to the Pourbaix diagram of the Hg–H2O system, free Hg2+ ions are stable only at pH < 2 (Powell et al., 2005). At higher pH values, including the range 5–7, the predominant mercury species correspond to neutral or monovalent hydroxyl complexes such as Hg(OH)+ and Hg(OH)2, depending on the redox potential and metal concentration (Ciavatta and Grimaldi, 1968; Ruiz-Bravo et al., 2025). These uncharged or weakly charged species can efficiently interact with electron-donor functional groups (–COO⁻, ≡Si–O⁻) present in the composites through surface complexation mechanisms or coordinated bond formation, even in the absence of a favorable electrostatic gradient (Liu et al., 2021; Hosseinpour et al., 2023).
At pH > pHₚzc, surface functional groups (carboxyl and hydroxyl groups) become fully deprotonated, generating a surface with a high density of negative charge, which could promote the adsorption of cationic species (Salehi, 2025). Nonetheless, under such conditions, anionic mercury species such as Hg(OH)3− or Hg(OH)42− may also form, potentially hindering adsorption depending on the surface affinity of the material. Considering this context, subsequent adsorption experiments were performed at pH 6.5, which on the one hand simulates natural water conditions and, on the other, prevents the formation of non-adsorbable anionic species while enabling specific interactions with Hg(OH)+ or Hg(OH)2, the predominant species under these conditions.
Kinetics of Hg(II) adsorption on floating composites
The kinetic adsorption study of Hg(II) (Figure 6) revealed a behavior characterized by a rapid initial uptake within the first 30 min, likely associated with the abundant availability of surface-active sites and the predominance of electrostatic interactions between Hg(II) ions and functional groups on the adsorbent surface, as commonly reported for alginate and clay-based systems (Crini et al., 2019; Das et al., 2023). In addition, Hg(II) removal is strongly influenced by the porous structure of the adsorbents and the effective hydrated radius of mercury species, which governs mass transfer and accessibility to internal active sites. This initial stage was followed by a slower approach to equilibrium, during which the adsorption capacity gradually reached a plateau. Among the evaluated materials, the floating composite FAB1.0 exhibited the highest adsorption capacity (≈33 mg g−1), values kept but wording tightened, followed by FAZ1.0 (≈28 mg g−1) and pristine alginate (≈21 mg g−1). The superior performance of FAB1.0 is attributed to the high cation-exchange capacity and interlayer accessibility of bentonite, which synergistically interact with alginate carboxylate groups, thereby enhancing the effective retention of metal cations (Gao et al., 2020). In contrast, although the incorporation of
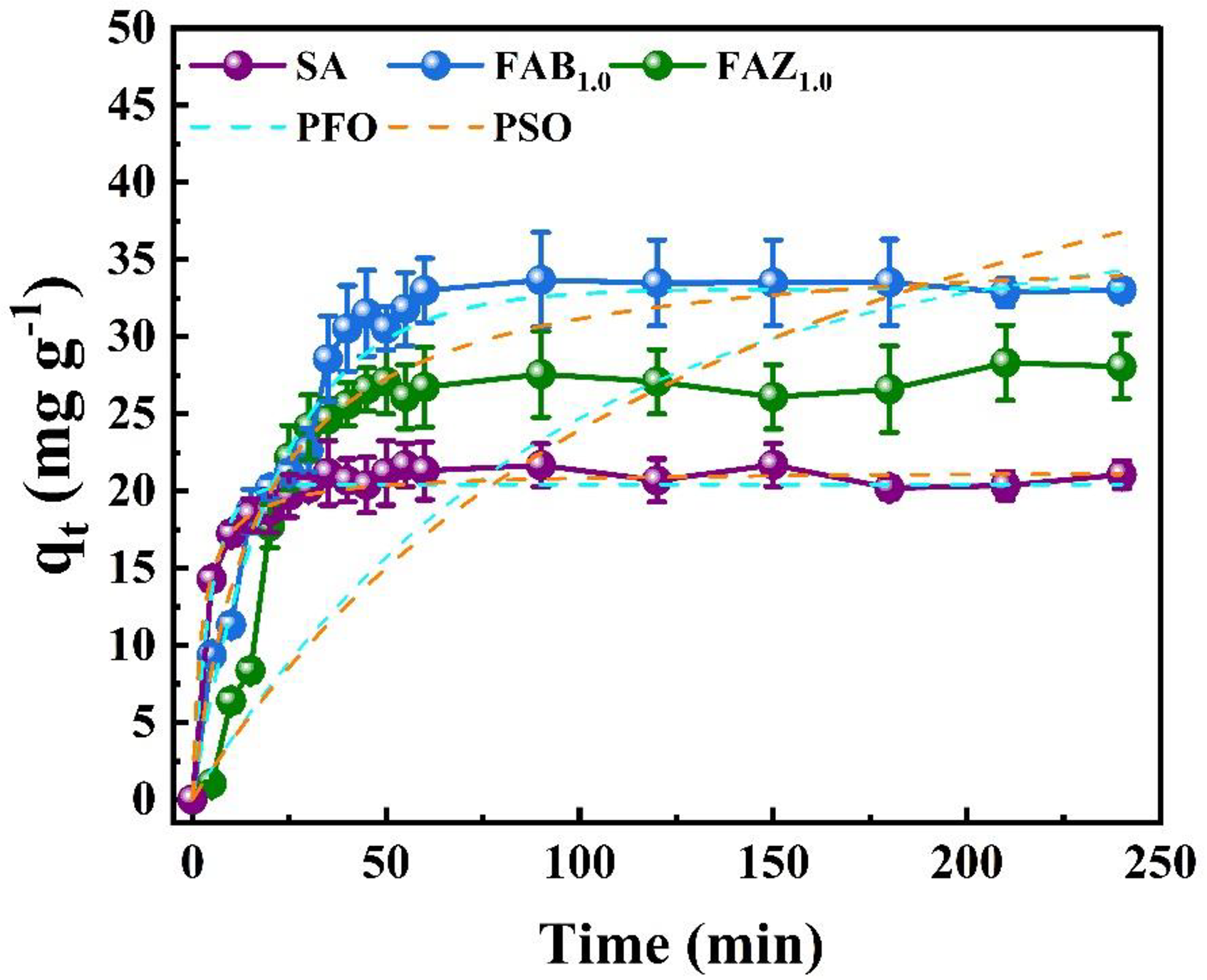
Adsorption kinetics of Hg(II) onto SA, FAB1.0, and FAZ1.0 composites fitted to nonlinear PFO and PSO models. Experimental conditions: [Hg(II)]0 = 200 mg L−1, pH 6.5, adsorbent dosage of 250 mg per 100 mL solution. PFO: pseudo-first-order; PSO: pseudo-second-order; FAB: floating alginate–bentonite; FAZ: floating alginate–
The experimental kinetic data were fitted to pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetic models through nonlinear regression. Model comparison was performed using
Kinetic parameters of Hg(II) adsorption onto SA, FAB1.0, and FAZ1.0 obtained from nonlinear fitting to PFO and PSO models.
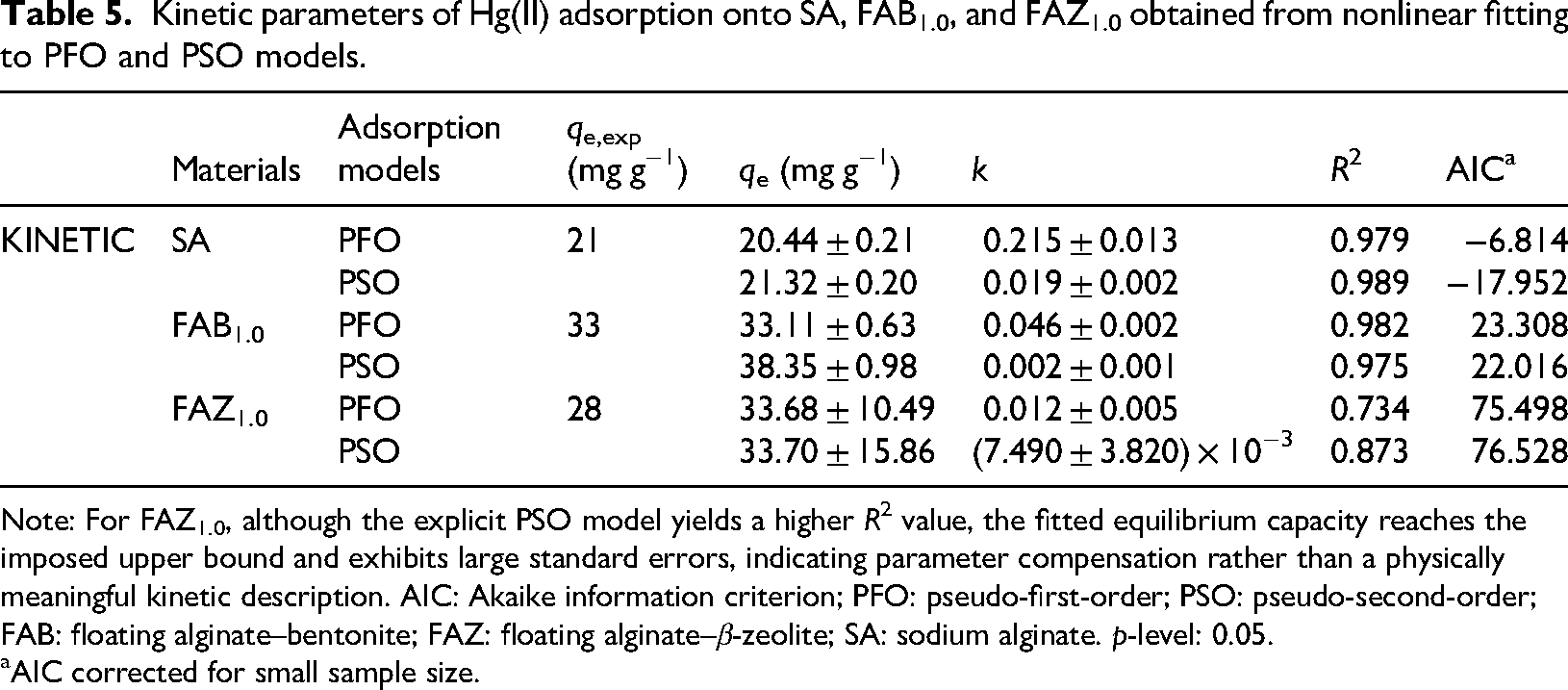
Note: For FAZ1.0, although the explicit PSO model yields a higher
AIC corrected for small sample size.
The removal efficiencies obtained for the floating and reusable adsorbent composites developed in this study (FAZ1.0 and FAB1.0) fall within a competitive range compared to materials reported in the literature (Table 6). In its first adsorption cycle, FAZ1.0 achieved 37% removal, outperforming encapsulated organobentonite/SA beads (21%) (Belhouchat et al., 2017). FAB1.0 reached a removal efficiency of 44%, higher than that of iron oxide/bentonite/activated carbon/SA composite beads (42%) (Gao et al., 2019) and comparable to barium alginate/graphene oxide nanocomposite hydrogels (43%) (Wang et al., 2022a). These results demonstrate that, even in the first adsorption cycle, the composites exhibit efficiencies equivalent to or higher than other reported alginate–clay systems, highlighting their potential not only as high-performance materials but also as promising candidates for reuse in consecutive adsorption cycles.
Adsorbent materials based on SA for contaminant removal after a single use cycle.

FAB: floating alginate–bentonite; FAZ: floating alginate–
Proposed mechanism for Hg(II) adsorption
Based on the experimental evidence obtained from pHₚzc analysis, FTIR characterization, textural properties, adsorption kinetics, and reuse experiments, a plausible mechanism for Hg(II) adsorption onto the floating alginate–aluminosilicate composites is proposed. At pH values around 6.5, the predominant mercury species correspond mainly to neutral or weakly charged hydroxyl complexes, such as Hg(OH)⁺ and Hg(OH)₂, which can effectively interact with electron-donor functional groups present in the composite matrix. In this context, alginate carboxylate (–COO⁻) and hydroxyl (–OH) groups, together with surface ≡Si–OH and ≡Al–OH sites of the aluminosilicate phases, act as active centers for Hg(II) uptake through surface complexation and electrostatic interactions. Additionally, in the FAB1.0 composite, cation-exchange processes associated with bentonite contribute significantly to mercury retention. The reversibility observed during acid-assisted regeneration indicates that these interactions are predominantly non-irreversible, consistent with surface complexation and electrostatic attraction rather than permanent chemical bonding. Overall, the synergistic contribution of the polymeric matrix and the inorganic phase accounts for the efficient Hg(II) adsorption and reusability of the floating composites.
Reuse cycles of floating composites
The analysis of the evolution of Hg(II) removal efficiency as a function of the reuse cycles of the evaluated adsorbent materials showed that composites FAB1.0 and FAZ1.0 exhibited a progressive increase in each cycle (Figure 7(a)). In particular, FAB1.0 displayed a sustained increase from 44% in the first cycle to 93% in the third, evidencing a progressive activation effect of the material. In the case of FAZ1.0, the efficiency rose from 37% to 71%, showing a remarkable improvement although tending to stabilize from the second cycle onwards. In contrast, the pure biopolymer (SA) recorded low and nearly constant values (≈19–26%), confirming its limited reusability. The analysis of the expansion of the performance area of each material (Figure 7(b)) further confirms the superiority of the composites compared to pure alginate.
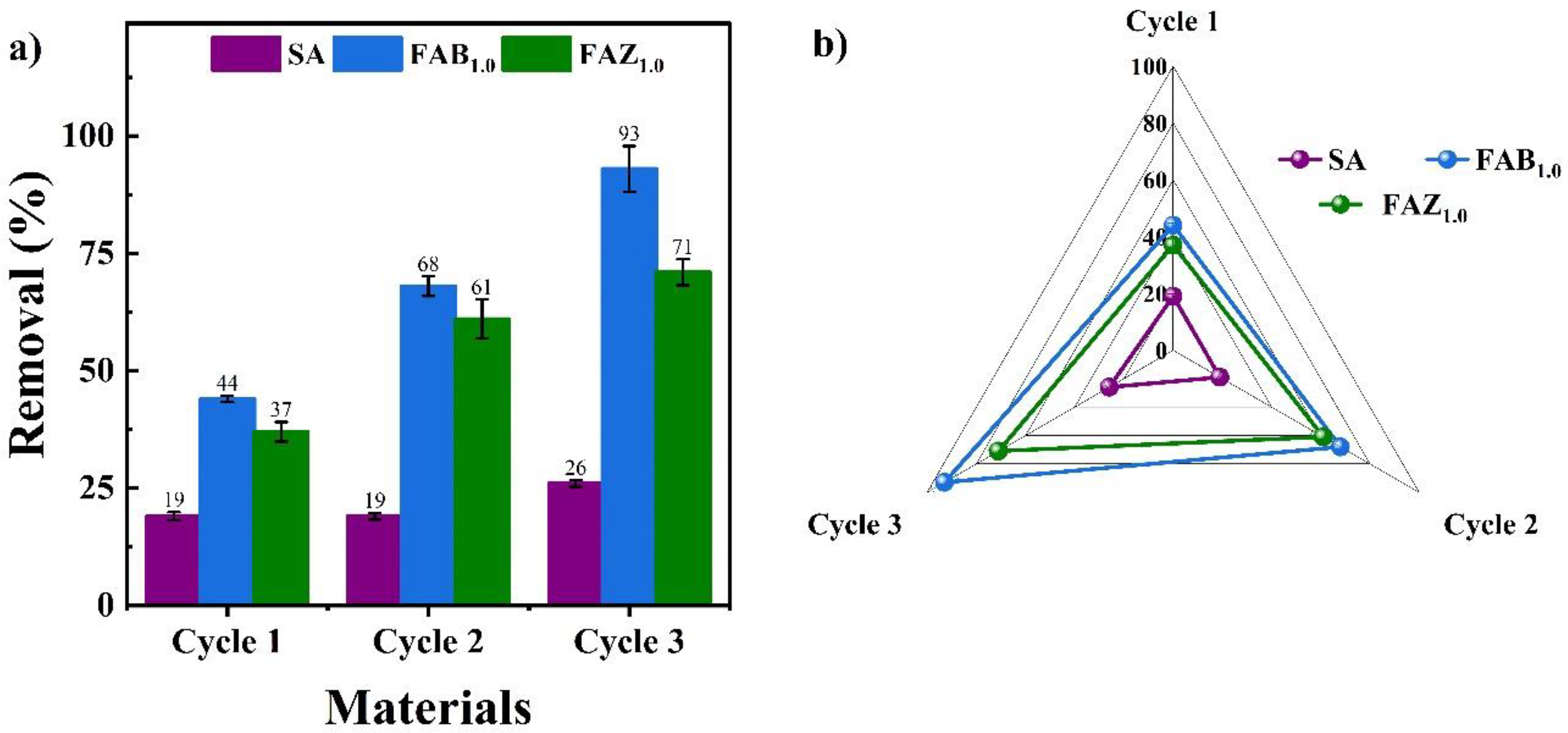
(a) Removal efficiency of Hg(II) over three reuse cycles for SA, FAB1.0, and FAZ1.0; (b) Radar plot comparison of cyclic performance. FAB: floating alginate–bentonite; FAZ: floating alginate–
The increase in removal efficiency observed in FAB1.0 and FAZ1.0 could be directly related to the HCl washing performed between cycles, since the acidic medium protonates the carboxylate groups of alginate and desorbs Hg(II) through proton competition, while also inducing the formation of soluble chloro-complexes (HgCl2, HgCl42−). This mechanism has been documented for polymeric and composite materials, where HCl at concentrations between 0.1 and 0.5 M efficiently regenerates adsorption capacity over several cycles, achieving Hg(II) desorption rates above 95% with moderate losses after multiple reuses (Alcalde-Garcia et al., 2023; Alguacil and López, 2020; Gupta et al., 2021). This phenomenon has also been observed in other studies using alginate–clay systems, where acid washing removes residual cations and surface precipitates, thereby improving wetting and pore accessibility for the following cycle; in this way, alginate–bentonite composites have been reported to retain more than 70% of their adsorption capacity after 6 reuse cycles (Tan and Ting, 2014; Wang et al., 2019).
Beyond simple regeneration, the progressive increase in Hg(II) removal efficiency suggests a conditioning of the polymer–aluminosilicate network during the first adsorption–desorption cycle. The composites are produced by freeze-drying Ca-crosslinked alginate matrices containing aluminosilicates; upon contact with aqueous solution, the structure rehydrates and swells. During the initial adsorption step, part of the Ca-alginate network remains relatively contracted, limiting diffusion toward internal adsorption domains located within the polymeric matrix and at the aluminosilicate surface. The subsequent acid washing promotes partial Ca2⁺/H⁺ exchange and relaxation of the crosslinked network, increasing water uptake and pore connectivity. Consequently, previously shielded carboxylate groups and aluminosilicate surface sites (Si–O⁻ and Al–O⁻) become more accessible to Hg(II) ions in the following cycles. Therefore, the observed increase in removal efficiency is attributed not only to desorption of residual Hg(II), but also to a structural conditioning of the adsorbent that improves mass transfer and accessibility of active sites. Similar structural relaxation and swelling effects of Ca-alginate matrices after ionic exchange and regeneration treatments have been widely reported (Lee and Mooney, 2012; Wang and Chen, 2014; Wang et al., 2019; Zhang et al., 2020b).
In this regard, several studies highlight that for aluminosilicates, acid washing must be mild and brief in order to clear the surface without causing structural damage. However, stronger or prolonged treatments with mineral acids may lead to dealumination of
In this context, the reuse cycle results of FAB1.0 and FAZ1.0 reflect a favorable balance between effective regeneration and structural preservation, which not only explains the progressive increase in Hg(II) removal after reuse cycles but also confirms their potential as stable, reusable, and high-performance adsorbents for the remediation of heavy metal contaminated waters.
While acid-assisted regeneration plays a key role in restoring and even enhancing the adsorption performance of FAB1.0 and FAZ1.0 over successive cycles, it also generates Hg(II)-containing acidic effluents that must be properly managed to ensure environmental safety. Therefore, the observed improvement in reusability highlights not only the functional robustness of the composites under mild regeneration conditions, but also the importance of integrating appropriate post-desorption handling strategies to prevent secondary contamination. In this sense, the regeneration step should be considered as part of a closed-loop adsorption–desorption process, in which performance recovery and environmental responsibility are addressed simultaneously.
Conclusions
The statistically optimized synthesis of floating alginate-based composites with bentonite (FAB1.0) and
In adsorption assays, FAB1.0 exhibited the highest Hg(II) removal capacity (≈33 mg g−1), followed by FAZ1.0 (≈28 mg g−1) and pure alginate (≈21 mg g−1). Kinetic fitting indicated a surface-controlled mechanism for FAB1.0, whereas FAZ1.0 showed diffusional limitations due to the zeolite's microporosity. The reusability of the floating composites, a key highlight of this study, showed increased removal efficiencies up to ∼93% for FAB1.0 and ∼71% for FAZ1.0 after three reuse cycles. This behavior was attributed to acid regeneration, which released active sites without compromising the structure. The reproducible adsorption–desorption behavior observed across reuse cycles confirms the stable retention of Hg(II) during each adsorption stage and its controlled release during regeneration, with no indication of uncontrolled metal leaching.
From a large-scale application perspective, the floating composites developed in this study offer clear operational and environmental advantages over conventional adsorbents. Their high buoyancy (>98%) enables straightforward physical recovery by surface collection or decantation, eliminating the need for energy-intensive separation processes such as filtration, centrifugation, or forced sedimentation, which often hinder the scalability of particulate adsorption systems. In addition, the structural stability maintained over multiple reuse cycles, together with effective regeneration under mild acidic conditions, suggests that these materials can operate within cyclic adsorption–desorption schemes with reduced generation of secondary solid waste. From an environmental standpoint, the use of alginate (a renewable biopolymer) combined with abundant and low-cost aluminosilicates, as well as the possibility of integrating controlled post-desorption effluent management strategies, reinforces the sustainability of the proposed approach. Overall, the results demonstrate that the optimized synthesis of FAB and floating –
Highlights
Statistical design optimized buoyancy (>98%) in floating alginate–clay composites. Optimized composites exhibited improved Hg(II) removal and cyclic reusability. Alginate–aluminosilicate interactions enhanced structural stability. Floating adsorbents enable easy recovery and sustainable Hg(II) remediation.
Supplemental Material
sj-docx-1-adt-10.1177_02636174261432740 - Supplemental material for Statistically optimized floating alginate–bentonite and β -zeolite composites with enhanced cyclic reusability for efficient Hg(II) removal from mining-impacted waters
Supplemental material, sj-docx-1-adt-10.1177_02636174261432740 for Statistically optimized floating alginate–bentonite and
Footnotes
Acknowledgments
Dr Ruiz thanks the SGR/MINCIENCIAS for its financial support under Project “Análisis del impacto socio ambiental del mercurio y tecnologías sostenibles para su remoción en la cuenca alta del rio Caquetá-Putumayo” with BPIN code: 2022000100050.
Ethical considerations
Not applicable. This research did not involve human participants, animals, or sensitive data.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by SGR/MINCIENCIAS under Project “Análisis del impacto socio ambiental del mercurio y tecnologías sostenibles para su remoción en la cuenca alta del rio Caquetá-Putumayo” with BPIN code: 2022000100050.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets generated and analyzed during the current study are not publicly available due to database license restrictions but are available from the corresponding author on reasonable request
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
