Abstract
The nano-ceramic which is mesoporous silica material was applied to test the removal efficiency of gas-phase Hg0 using a fixed-bed reactor. The physical and chemical properties of nano-ceramic were investigated by various techniques such as BET surface area (BET), X-ray diffraction, fourier transform infrared spectrometer (FTIR), and scanning electron microscope (SEM); then, the sample was tested for mercury adsorption under different conditions. The mercury adsorption tests shown that different Hg0 concentration, adsorption temperature, gas flow rate, and different gas components have significant effects on the mercury removal performance of nano-ceramic, and the adsorption removal rate of nano-ceramic can be 75.58% under the optimal experimental conditions. After fitting the experimental data to the adsorption model, it was found that the theoretical maximum mercury adsorption amount
Introduction
Mercury has become a pollutant that poses a great threat to the global ecological environment and human health due to its toxicity, volatility, persistence, and bioaccumulation in the environment. 1 Anthropogenic sources of mercury include industrial combustion such as coal combustion, nonferrous metal smelting, waste incinerators, and cement production. 2,3 Mercury and its compounds produced by anthropogenic and natural sources can circulate in the atmosphere for up to 1 year, so they can be widely distributed and transmitted thousands of kilometers away, and then transferred to surface waters and land through meteorological activities. 4 Mercury is mainly present in the atmosphere in the form of elemental form (Hg0) that can be directly inhaled. And it can be converted into bivalent mercury (Hg2+), especially methylmercury which can cause fatal damage 5 by the conversion of aquatic organisms in water.
At present, the most commonly used industrial flue gas mercury control technology is the use of raw/modified activated carbon catalytic oxidation adsorption technology. The modification method of activated carbon is mainly to add nonmetal elements such as sulfur and chlorine or metals such as Mn, Al, Ag, Cu, and their oxides on the surface of activated carbon by oxidation or impregnation to achieve the purpose of modification of activated carbon. 6 Vulcanization modification is an important method to improve the mercury adsorption capacity of activated carbon, because surface sulfur compounds can provide sufficient binding sites for mercury to form stable mercury sulfide. 7 Compared to some conventional adsorbents, metal sulfides have excellent Hg0 capture performance and resistance to H2O and SO2. 8 However, the cost of chemically modified activated carbon is higher than that of untreated activated carbon. The price of unmodified activated carbon is 1.1 US$ per kg, and the price of modified activated carbon is 1.9–2.6 US$ per kg. 9 Moreover, the treatment of mercury-containing high-temperature gas and mercury-containing wastewater generated after activated carbon regeneration is difficult, which also makes the regeneration cost of activated carbon high, and cannot be recycled well. Meanwhile, although the non-carbon-based adsorbents (such as zeolites and kaolins) are convenient and available in large price, they have poor adsorption performances, so they must be used by physical or chemical modification instead of original materials. 10 –12 Kwon and Vidic 13 found that the surface area of virgin and acid-treated bentonite increased from 86 m2 g−1 to 242 m2 g−1, virtually no adsorptive capacity was detected for the virgin bentonite, while some mercury removal was observed for acidified bentonite. This increase in capacity can be explained by an increase in surface area resulting from acid treatment creating mercury adsorption sites. Therefore, searching for high-efficiency, low-cost adsorbents has become a hot research topic. Nanomaterials have better adsorption performance than the original adsorbents because of their relatively large specific surface area and more active groups on the surface so become the most potential research direction of adsorbents.
Granite and Pennline 14 synthesized a nanocomposite adsorbent and tested its effect on removing coal-fired flue gas. At the same time, Granite and Pennline 14 and Pitoniak et al. 15 systematically carried out the kinetic experiments of titanium-based nanoparticles/fibers to remove Hg0 and investigated the effects of various atmospheres on mercury removal. The results show that the catalyst has good oxidation and removal properties for Hg0. Jeon et al. 16 used aerogel, xerogel, and impregnation methods to prepare nanoscale catalysts and used them in the field of mercury removal. It was found that the aerogel-prepared catalysts have higher BET and can form a single layer of vanadium oxide on the catalyst surface, and the adsorbent prepared by this method has higher activity against Hg0. The study also found that when the reaction temperature was 100°C, there was no significant difference in the removal of Hg0 by the adsorbent under a nitrogen atmosphere and an air atmosphere. Yang et al. 17 also loaded nano-sized TiO2 particles onto activated carbon, which showed good adsorption effect on pollutants.
As an important class of nano-metal oxides, nano-iron oxide is chemically stable due to the large number of lattice defects on its surface. Fe(III) in the defect is in an unsaturated state, it is easy to adsorb substances with excess electrons or form stable coordination compound, so that it has special adsorption potential and catalytic activity. Kong et al. 18 studied the mercury removal performance of nano-Fe2O3 and found that nano-Fe2O3-SiO2 has strong ability to remove elemental mercury when the optimal loading of Fe2O3-SiO2 is 10% and the optimal reaction temperature for removal of elemental mercury is 350°C. Borderieux et al. 19 studied the performance of high surface area nano-Fe2O3 to remove elemental mercury. It was found that nano-Fe2O3 acts as an adsorbent for elemental mercury under air atmosphere but as a catalyst in the presence of nitrogen dioxide. The average mercury absorption amount of nano-Fe2O3 is 1390 μg g−1, and the optimum temperature is 260°C. Abu-Daabes and Pinto 20 found that nano-silica materials have a capacity of 33 mg g−1 for HgCl2 at low temperatures. The nano-ZnS prepared by Li et al. 21 has a large surface area and has excellent mercury adsorption performance (497.84 μg g−1) compared with commercial activated carbon.
In addition to the properties of the adsorbent, the flue gas composition also has a significant effect on the capture of Hg0. These effects may be due to changes in the surface of the adsorbent and changes in mercury morphology caused by interactions with gas components including O2, SO2, and NO
In this article, we choose NC as a new type of adsorbent. NC is a modified mesoporous silica material and belongs to non-carbon-based adsorbent. Mesoporous silica materials have large surface area, good pore size and pore shape, and have great application potential in environmental and industrial processes. However, there are many industrial applications such as adsorption, ion exchange, and catalysis that require materials with specific properties such as specific binding sites, stereochemical structures, charge density and acidity, 29 –31 so a certain modification of the original mesoporous silica material is required. The cost of NC after modified is about 20 ¥ per kg. In actual engineering accounting, the cost of whole coupled system which combined the low-temperature plasma technology to treating the flue gas mercury is about 10 ¥ per 1000 m3, and the whole technology has the advantages of good treatment effect and low cost.
Applying an organic film on the surface of the silica pores, and using the surface of the micropores as the end group of the organic monolayer film, the hydrocarbon chains aggregated and formed close-packed arrays on the substrate. The siloxane groups then underwent hydrolysis and ultimately became covalently attached to the substrate and cross-linked to one another. This material, called functionalized monolayers on mesoporous supports (FMMS), can efficiently remove mercury and other heavy metals (such as lead and silver) from contaminated water and organic solutions. 32 This article will study its adsorption performance for Hg0 in flue gas from coal combustion and industrial production.
Experiment
Sample preparation
The preparation method of the NC material used in the experiment is as follows. Firstly, hexadecyl trimethylammonium chloride/hydroxide, silicate and medium trimethyltoluene solution were used as the precursors to prepare mesoporous silica materials, and then trimethoxy mercaptopropyl silane (TMMPS) was mixed with mesoporous silica, stirred at room temperature for 24 hours, and heated reflux for 4 hours. After cooling to room temperature, the mixture was filtered, washed copiously with 2-propanol to rinse away any surplus TMMPS, and then dried under vacuum. 32 –34
Sample characterization
The specific surface area (BET) and pore structure characteristics of the NC were measured by N2 adsorption–desorption method using a specific surface area and pore size distribution analyzer; the microstructure of the sample was scanned using Sigma 500/VP field emission SEM (Carl Zeiss, Germany) and the samples should be dried in an oven at 110°C for 12 h before being tested.
NC elements quantitative analysis was carried out under low vacuum conditions using X Ray Fluorescence (XRF) (ZSX Priums, RIGAKU, Japan).
The composition of NC was characterized by PANalytical X’Pert PRO (Max resolution ratio=3 kW) X-ray diffraction (XRD) using copper
The type of functional group in the experimental sample was characterized by FTIR (VERTEX 70, Bruker, Germany). The sample was prepared by KBr tableting method, and the wavelength range tested was 400–4000cm−1.
Experimental apparatus and methods
The experimental setup is shown in Figure 1. The whole system is mainly composed of gas source, mercury vapor generator, fixed bed adsorption reactor, and mercury vapor concentration analyzer. The mercury vapor is supplied by heating the liquid mercury in a constant temperature water bath.
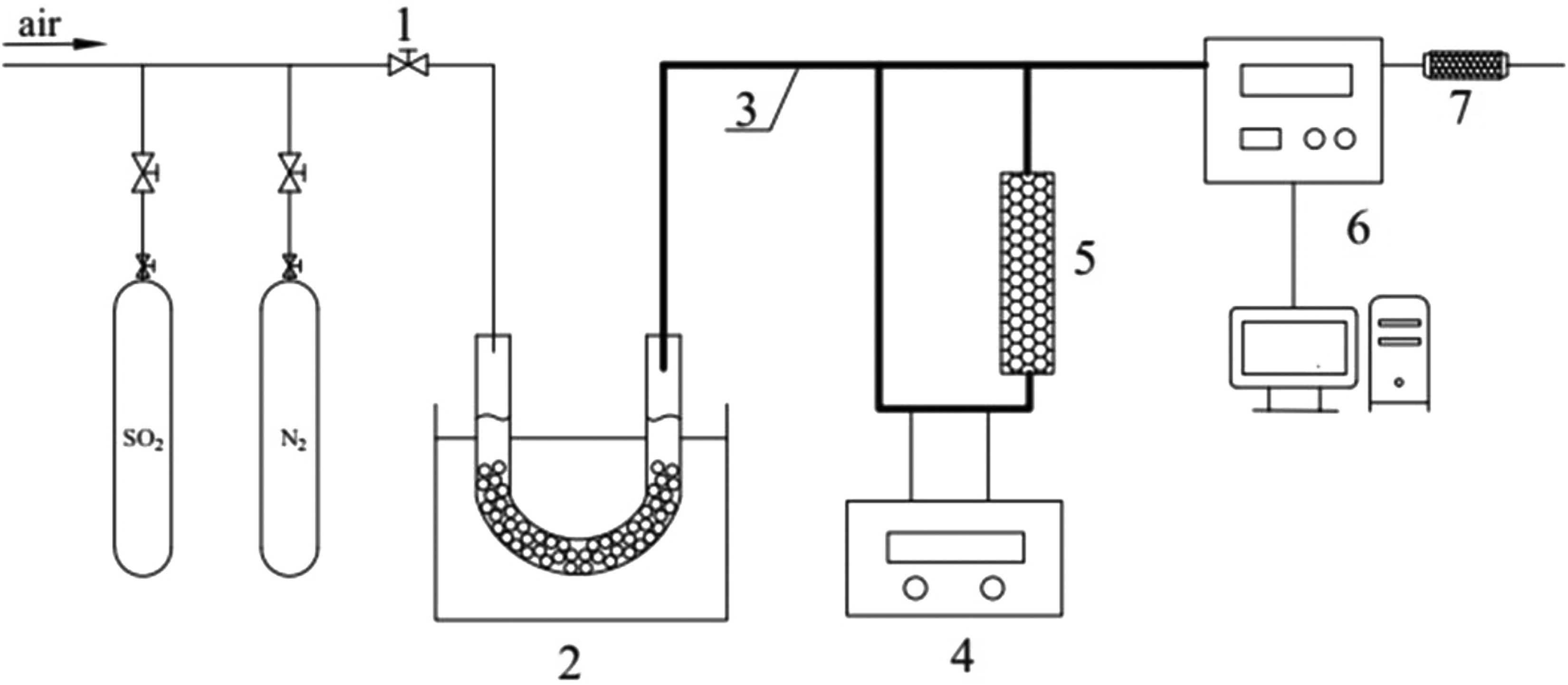
Schematic of the experimental setup. 1: valve; 2: mercury vapor generator; 3: heating band; 4: temperature control device of heating band; 5: adsorption bed reactor; 6: RA915+ mercury analyzer; 7: exhaust gas absorption device.
The Hg0 adsorption performance of NC is expressed by the removal rate (
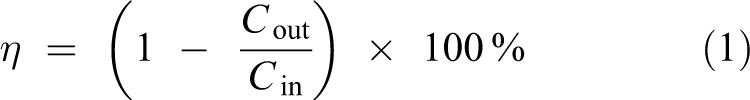
Results and discussion
Sample characterization
The results of the specific surface area (BET) and pore structure characteristics of the NC using the N2 adsorption–desorption method are shown in Table 1.
BET analysis of nano-ceramic.

BET: surface area; NC: nano-ceramic.
The NC has an average pore diameter of 8.482 nm and belongs to a mesoporous structure with a specific surface area of 122.28 m2 g−1. According to the classification of common adsorption isotherms, the adsorption isotherms of NC belong to class IV, which are characterized by obvious hysteresis loops. The adsorption mechanism corresponding to the IV adsorption line is as follows: firstly, a single layer of adsorption is formed, and the inflection point indicates the saturation of monolayer, and then multilayer adsorption is started, followed by capillary condensation. The starting point of the hysteresis loop indicates that the smallest capillary pores begin to condense, and the end point of the hysteresis loop indicates that the largest pore has also been filled with the condensed liquid. (Figure 2)
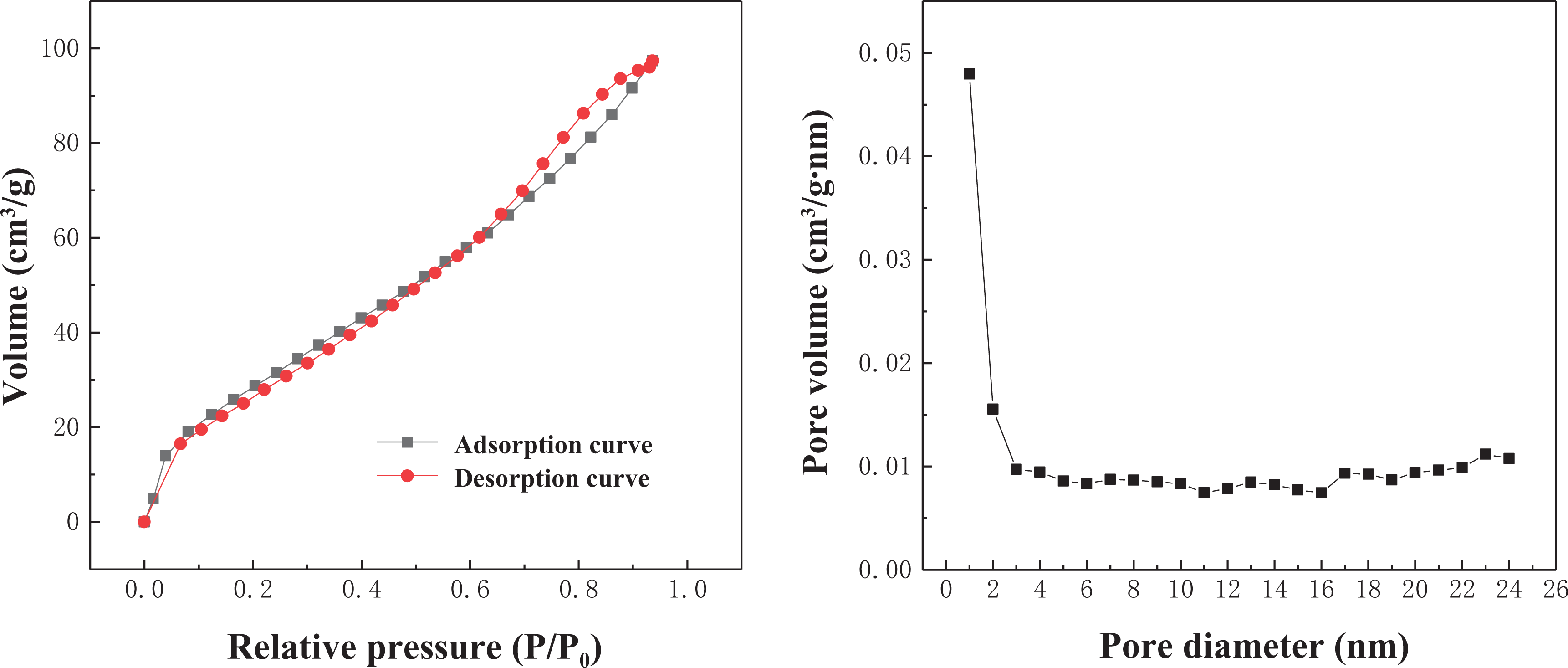
Adsorption/desorption curves and pore size distribution of nano-ceramic.
The main oxide types and contents in NC measured by XRF are given in Table 2.
Oxide types and contents in nano-ceramic.

NC: nano-ceramic.
The material sample was khaki. After XRD test, the main components of NC were quartz, dolomite, and gismondite (Figure 3).
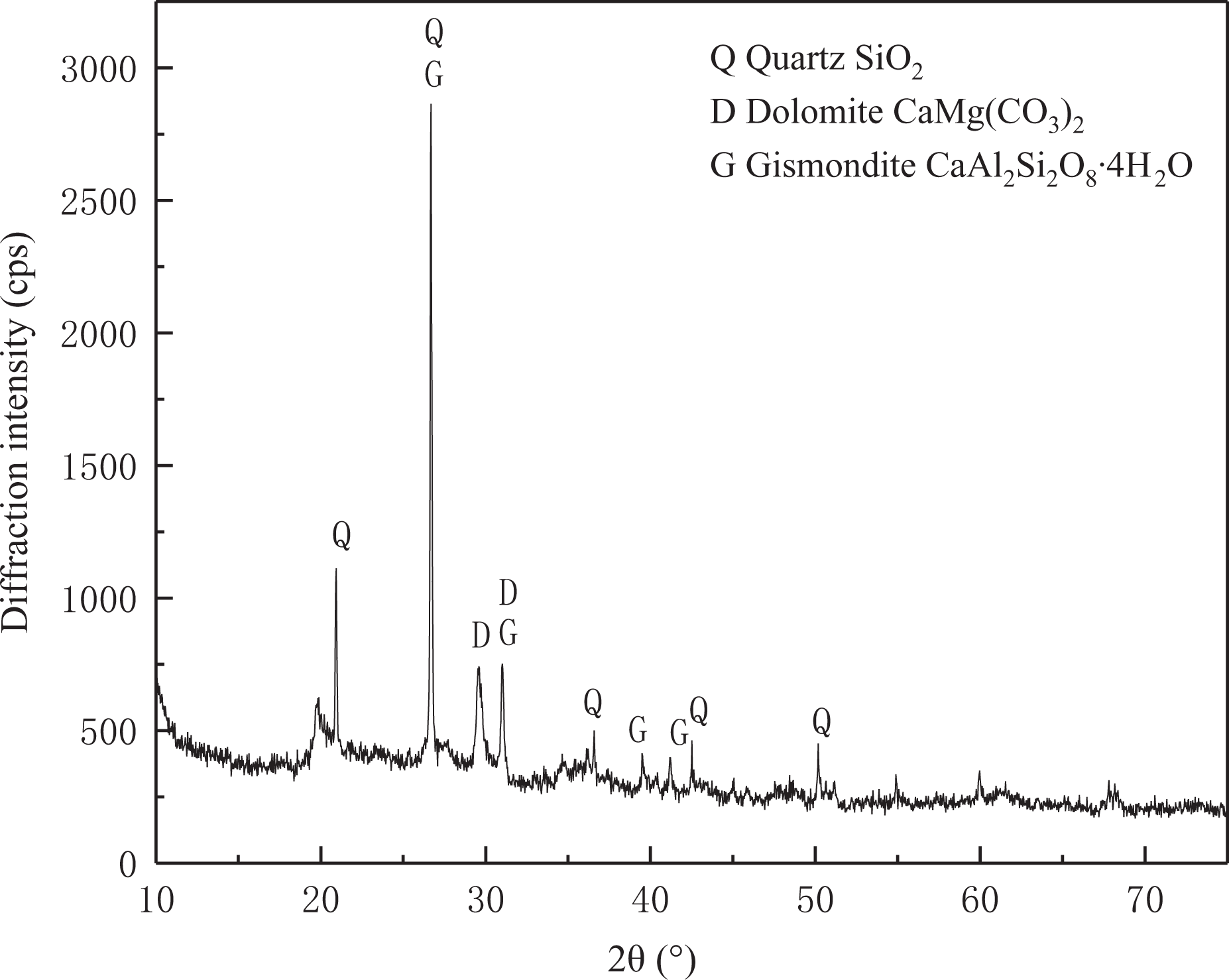
The XRD spectra of nano-ceramic. XRD: X-ray diffraction.
The comparison between the phase composition measured by XRD and the oxide type obtained by XRF shows that the NC used in the experiment is mainly composed of Si, Ca, Mg, Al, and other compounds and can be classified into non-carbon-based adsorbents.
The FTIR spectrum of the NC detected is shown in Figure 4.
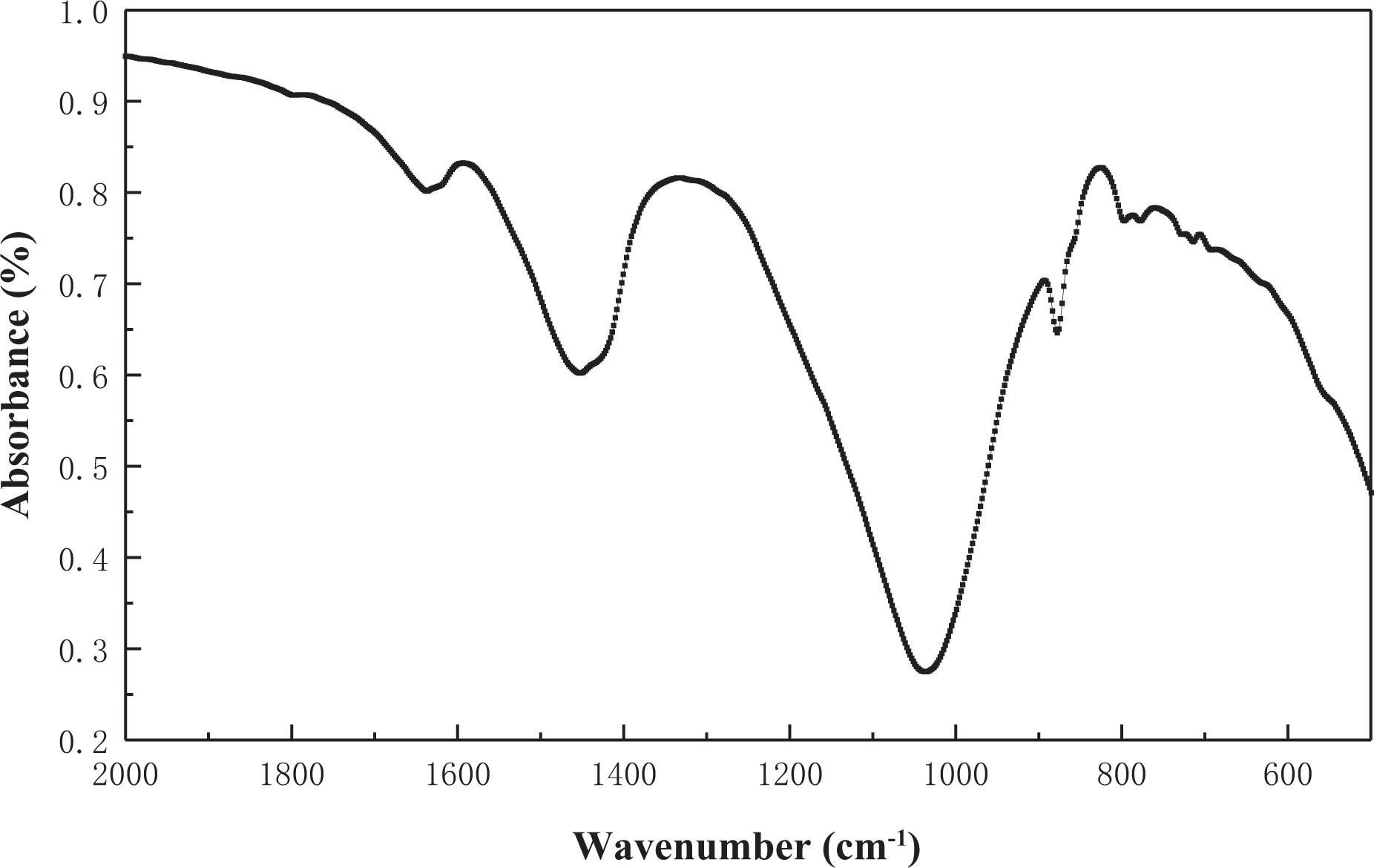
The FTIR spectra of the nano-ceramic. FTIR: fourier transform infra-red.
The type of functional groups on the surface of the adsorbent has an important influence on its adsorption properties. The researchers studied the mercury adsorption effects of different types of activated carbons and activated carbons modified by different methods. It is generally believed that the adsorption effect of activated carbon on mercury is determined by the different types of active sites on the surface. The research indicates that the oxygen-containing functional groups on the surface of activated carbon can provide active sites for the adsorption of mercury, which plays an important role in the adsorption of mercury by activated carbon. 35,36 The characteristic functional groups contained in the NC are presented in Table 3.
The functional groups contained in the nano-ceramic.
The microstructure of the adsorbent is generally considered to have influence on the adsorption process and mechanism. The SEM image of the nano-ceramic is shown in Figure 5, after magnifying 10,000 times, it can be found that the surface of the NC is a distinct layered structure with uniform surface and less dispersed particles.

The SEM of nano-ceramic. SEM: scanning electron microscope.
Sorbent activity test
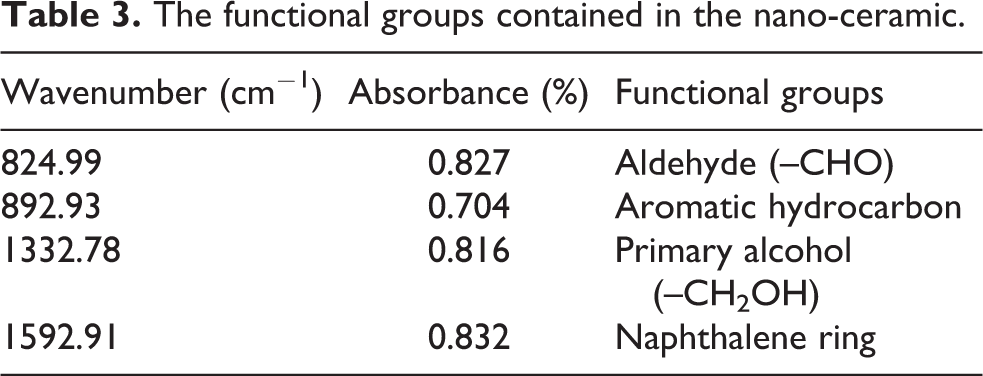
Effect of mercury concentration
The effect of mercury inlet concentration on removal efficiency is shown in Figure 6. The mercury concentration increases with the increase of the water bath temperature. When the water bath temperature is 40°C, 50°C, 60°C, 70°C, and 80°C, the inlet concentration of mercury is 205.2 μg m−3, 223.9 μg m−3, 269.4 μg m−3, 314.4 μg m−3, and 332.2 μg m−3. The adsorption temperature of 40–80°C was selected, the airflow rate was 1.5 L min−1, and the mass of NC was 2 g.
Efficiency–concentration curve of nano-ceramic.
With the increase of mercury inlet concentration, the mercury removal rate of NC first increases and then decreases. This is because the amount of NC used in the experiment is certain, so the number of adsorption active sites is constant, and the mercury concentration is constant. The larger the number of active sites and vacancies on the surface of the adsorbent required for the larger adsorption, the higher the concentration of Hg0 leads to the relative decrease of the active sites and the adsorption space in the NC, and the adsorption competition increases, so that the adsorption rate decreases and the adsorption performance is degraded. Jozewic and Gullet 37 suggested that the greater the concentration of mercury in the adsorbent surface will form a larger concentration gradient driving force, so that in the adsorption process, a greater adsorbed ability can be provided, and thus to enhance the rate of adsorption performance. Therefore, the increase of mercury concentration has a certain positive effect on the improvement of adsorption efficiency.
Effect of adsorption bed temperature
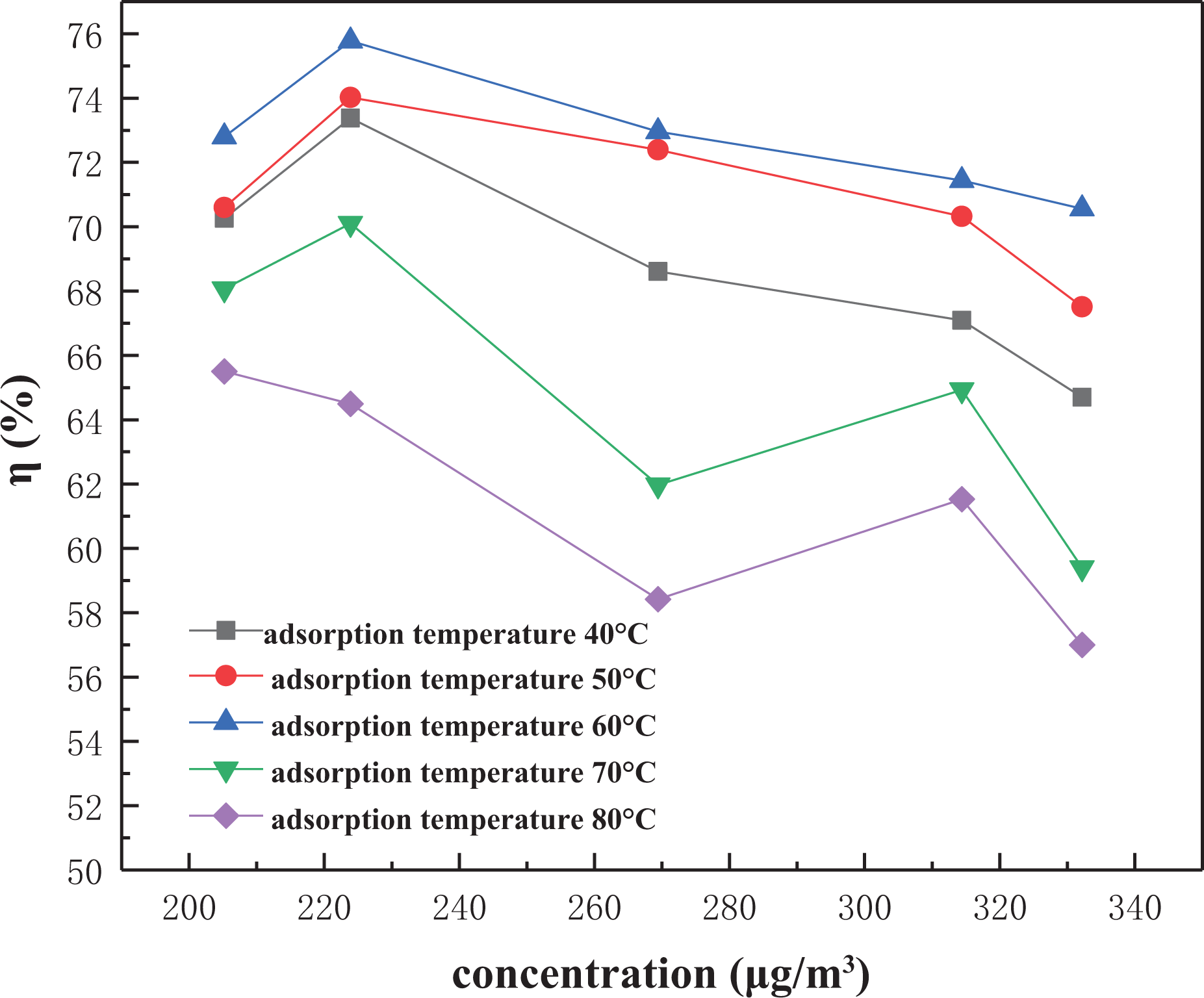
In the experiment, the effect of adsorption temperature on mercury removal efficiency was studied by changing the temperature of the heating band and changing the temperature of the region where the adsorbent was located. Due to the influence of the material properties of the tetrafluoroethylene tube, it could not be heated to a higher temperature. Therefore, the adsorption bed temperature selected in this experiment was 40–80°C, the airflow rate was 1.5 L min−1, and the mass of adsorbent was 2 g.
At lower adsorption temperatures, the adsorption rate of NC increases with the increase of adsorption temperature, and the higher the initial mercury concentration (the higher the water bath temperature), the more obvious the removal rate increases. On contrary, when the adsorption temperature is higher than 60°C, the removal rate begins to decrease as the adsorption temperature increases, as you can see the higher the
Efficiency–adsorption temperature curve of nano-ceramic.
This indicates that the adsorption of mercury by NC has both physical adsorption and chemical adsorption. First, the molecules of Hg0 are absorbed to the surface of NC by physical adsorption, then they are absorbed by chemical adsorption through slow reaction. At higher temperatures, the increase of temperature causes the physical adsorption performance to decrease, which leads to the decrease of mercury removal efficiency. At the same time, the H2O molecules and oxygen-containing functional groups (such as –CHO and –CH2OH) on the surface of the adsorbent also play an important role in the adsorption of mercury at normal temperature. As the temperature increases, the H2O molecules and oxygen-containing functional groups also decrease, which also affect adsorption properties of NC.
Effect of airflow rate
Changes in airflow cause changes in both Hg0 concentration and residence time. When the flow rate is small, the initial concentration
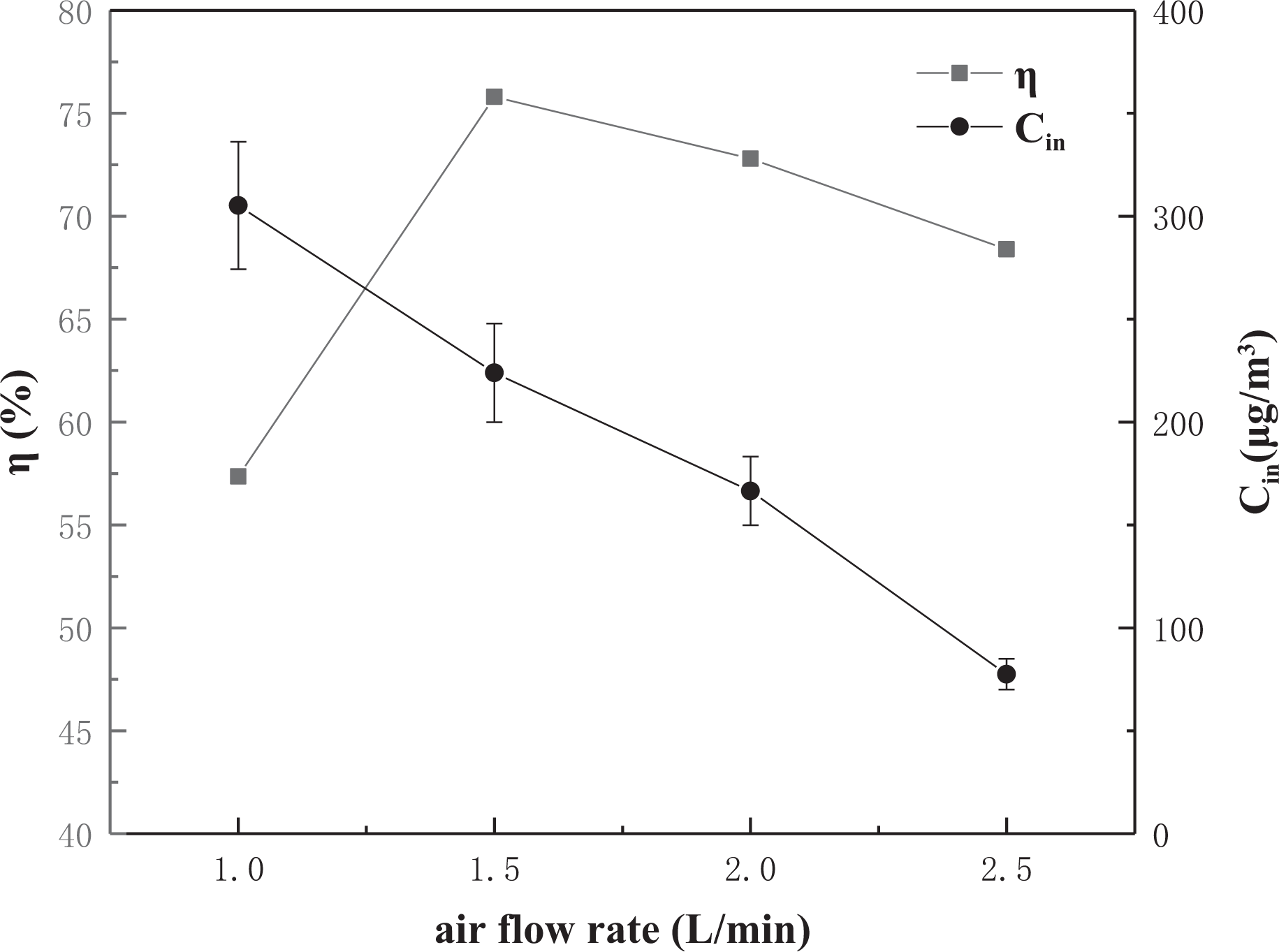
Efficiency–air flow rate curves of nano-ceramic.
When the gas flow rate is low, although the concentration of Hg0 is higher at the beginning of the experiment, the residence time is longer, and more Hg0 is in contact with the active sites on the NC per unit time, so the removal rate is relatively high. As the flow rate continues to increase, the mercury concentration
Effect of different atmospheric components
O2 has a non-negligible effect in the adsorption removal process of Hg0, because oxygen can oxidize Hg0 in solid or gas to oxidized mercury, and the higher the O2 concentration, the higher the possibility that Hg0 is oxidized. The adsorption of oxidized mercury is easier than the adsorption of elemental mercury, so the presence of oxygen can increase the adsorption efficiency of mercury. As can be seen from Figure 9, the removal rate of the NC was kept at a relatively low level when using N2 as carrier gas, and after switching the use of air as a carrier gas, the mercury removal rate began to rise rapidly and reached 63.4%. And the mercury removal rate declines to 14.9% when switching N2 as carrier gas again. This results that O2 has a significant effect on the Hg0 absorption performance of NC. This process mainly includes the following two reactions (“ad,” “g,” and “s” in the reactions formula represent “adsorbent,” “gas phase,” and “solid phase,” respectively) 38
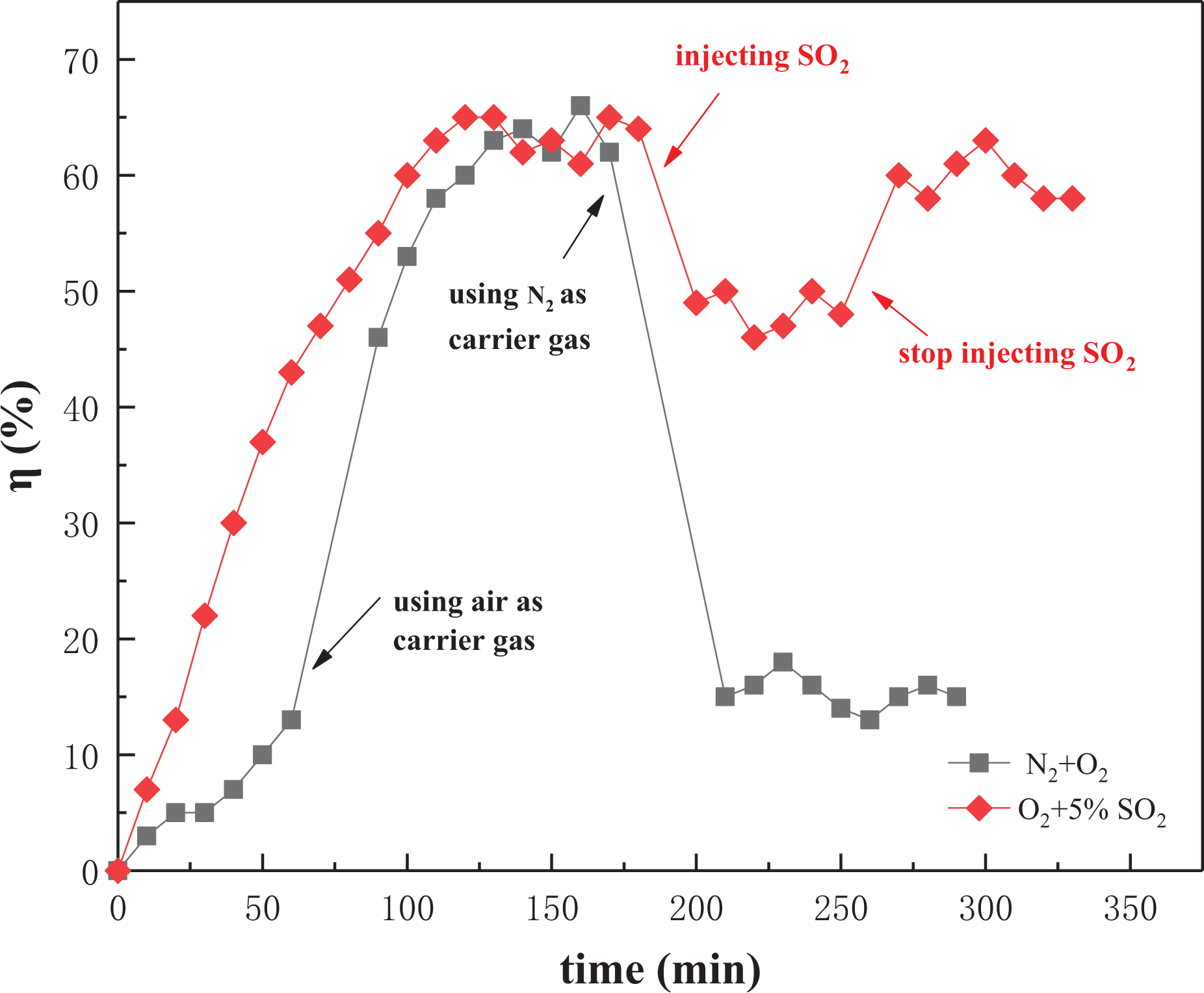
The effect of O2 and SO2 on mercury removal efficiency.
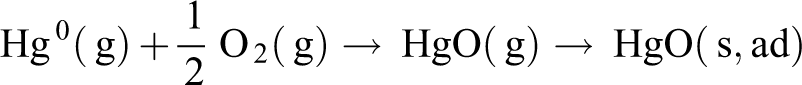
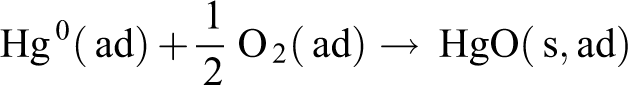
When using air and SO2 as carrier gas, it can be seen from Figure 9 that the mercury removal rate of the NC before the injection of SO2 is 64% on average, but
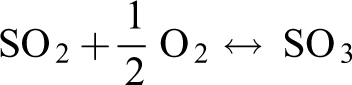
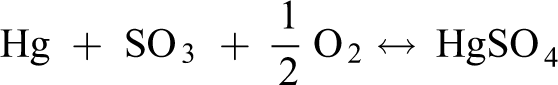
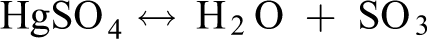
Olson et al. 40 and others believe that SO2 inhibits the oxidation of Hg0 mainly for two reasons: (1) SO2 reacts with O2 to form SO3 and HgSO4, which consumes reactive oxygen species that can oxidize Hg0 in the air. (2) Hg0 can be oxidized only after adsorption, but SO2 can inhibit the adsorption of Hg0. Relatively, the concentration of O2 in the air is much larger than the concentration of SO2 injected during the experiment. Therefore, even if SO2 consumes O2 and inhibits the oxidation of Hg0, the remaining O2 can still oxidize Hg0 to form oxidized mercury and then be adsorbed. This also explains why the removal rate of mercury after the injection of SO2 will decrease, but the decline will not be very large.
Adsorption model
The isotherm can account for the relationship between the equilibrium concentration of the adsorbate and the equivalent of the adsorbate on the adsorbent. Common adsorption isotherm models mainly include Langmuir and Freundlich adsorption equation, and they are expressed as follows
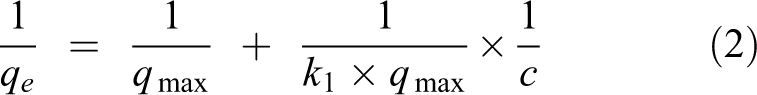
where
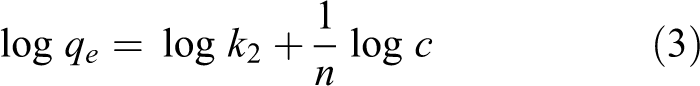
where
The linear adsorption of the Langmuir and Freundlich isotherms was carried out (Figure 10) using the adsorption data of the best removal efficiency of NC mercury (removal rate is 75.78%, water bath temperature is 50°C, adsorption temperature is 60°C, and the mass of NC is 2 g). The results of the parameter calculations in the equation are listed in Table 4.
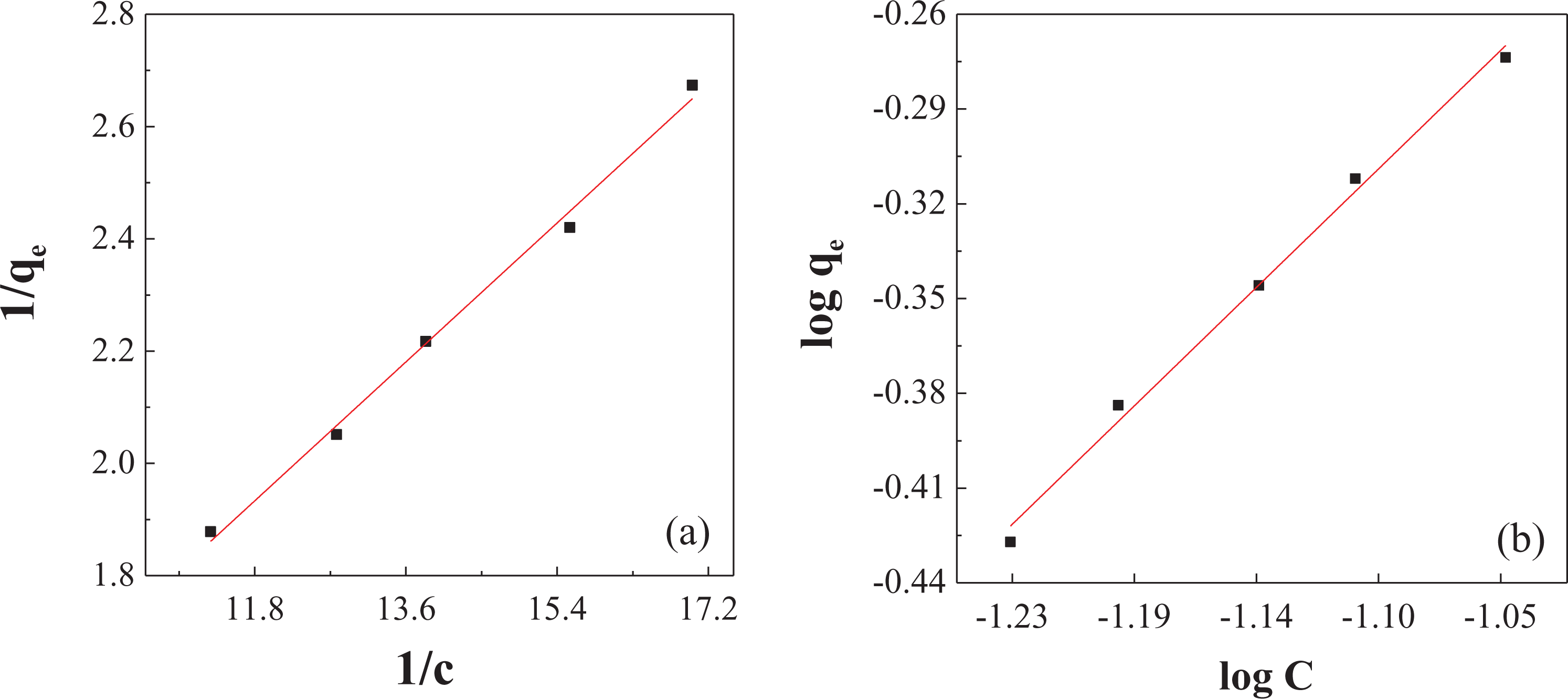
The fitting curves of (a) Langmuir equation and (b) Freundlich equation.
The parameters of adsorption isotherm equation.

NC: nano-ceramic.
From the results of Table 4, the fitting degree of the Langmuir equation and the Freundlich equation are basically the same (0.9931 and 0.9922). The Langmuir adsorption model is a single-layer physical adsorption, and the Freundlich adsorption model tends to be complex chemical adsorption. The similar fitting degree between the two shows that there are both physical adsorption and chemisorption processes for the NC, which is also the same as the speculation in the effect of adsorption temperature. First, the molecules of Hg0 are absorbed to the surface of NC by physical adsorption, then they are absorbed by chemical adsorption through slow reaction. The theoretical maximum mercury adsorption amount,
Kinetic analysis is a very important parameter to study the speed control steps in the adsorption process. Common adsorption kinetic models mainly include particle internal diffusion model, pseudo-first-order model, and pseudo-second-order model. The first two models which come from the mass balance equation are used to describe the internal diffusion and external mass transfer process. Pseudo-second-order model is mainly used to describe chemical adsorption process.
The kinetic rate equation for the quasi-first-order, quasi-second-order dynamic model and the intra-particle diffusion model is expressed as follows
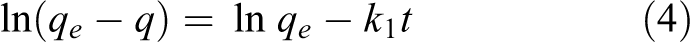
where
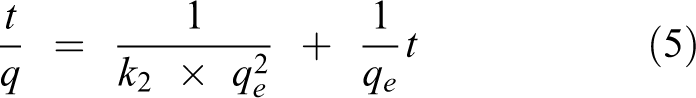
where

where
The fitting results of the quasi-first-order and the quasi-second-order dynamic model and the intra-particle diffusion model are shown in Table 5 and Figure 11.
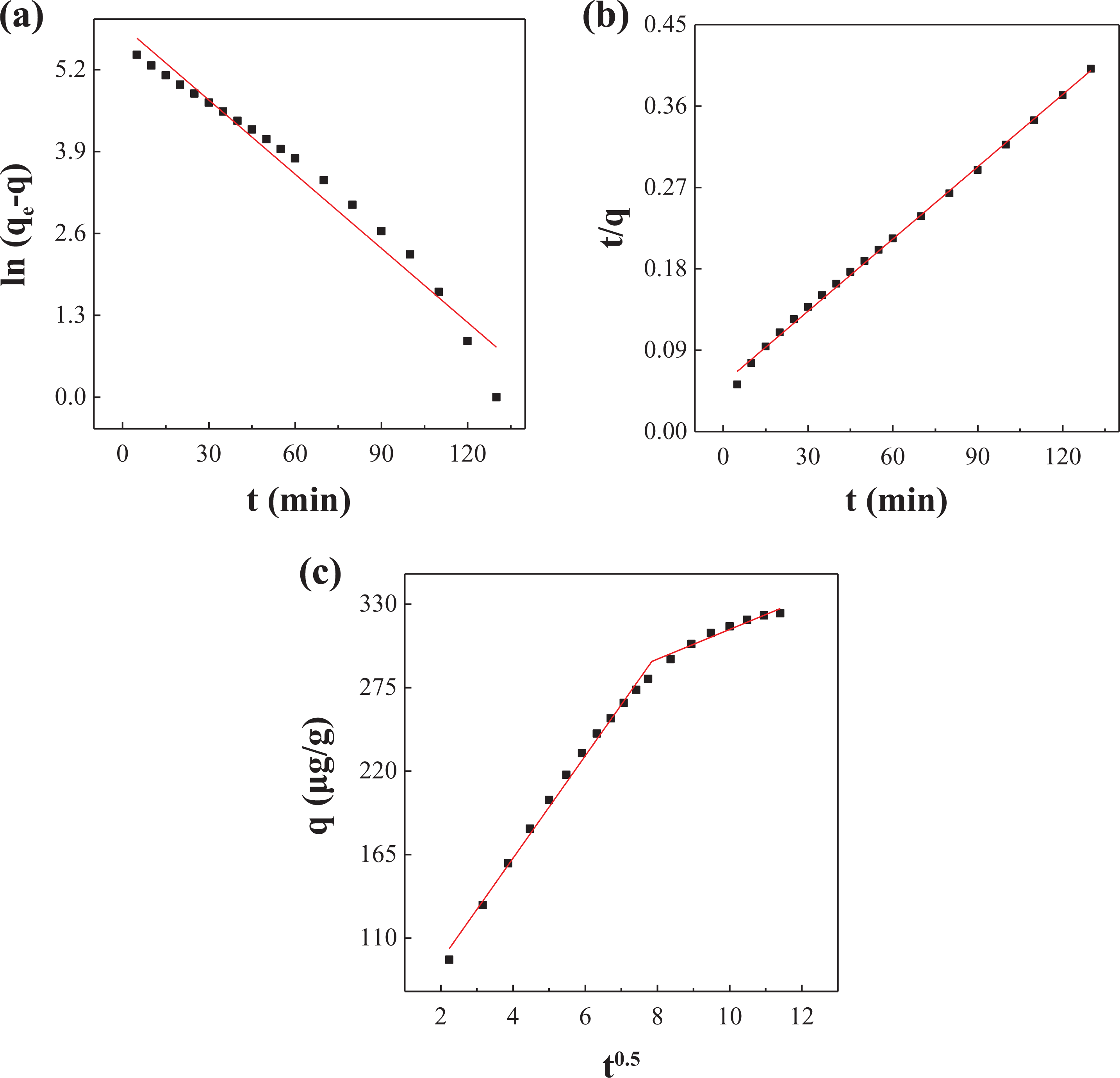
The fitting curves of (a) pseudo-first-order kinetics, (b) pseudo-second-order kinetics, and (c) intra-particle diffusion equation.
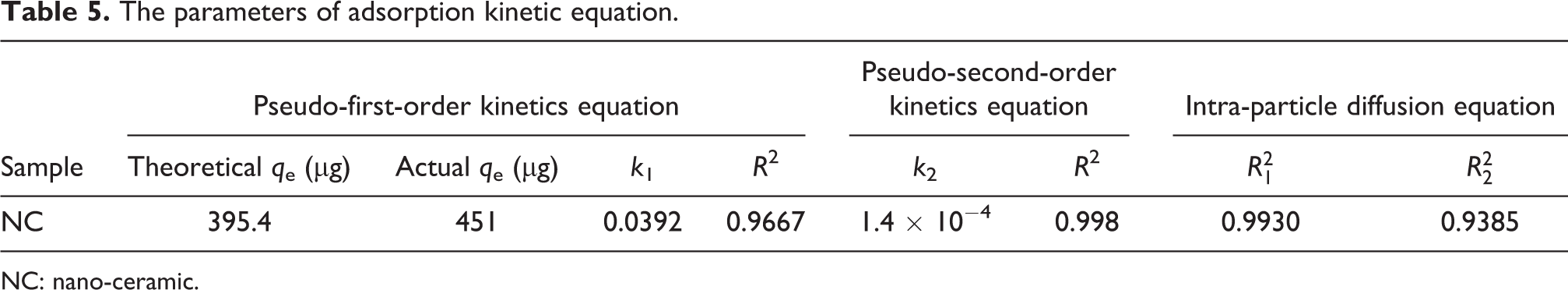
The parameters of adsorption kinetic equation.
NC: nano-ceramic.
It can be seen that the theoretical maximum adsorption amount calculated by the model is similar to the actual data. This also verifies the reliability of the first-order kinetic model. According to the fitting results, the adsorption of Hg0 by NC is more in line with the quasi-second-order kinetic equation. The second-order kinetic model considers that the adsorption process is controlled by the chemisorption mechanism and involves electron sharing or electron transfer between the adsorbent and the adsorbate and also reflects the composite effect of adsorption. Combined with the multi-segment fitting results of the intra-particle diffusion model, we conclude that the adsorption of Hg0 by nano-ceramics is divided into three stages: “transmembrane–diffusion–adsorption.” First, Hg0 diffuses from the gas through the gas film to the outer surface of the nano-ceramic; secondly, Hg0 enters the inner pore from the outer surface of the solid; finally, it is the adsorption phase, and Hg0 and NC surface are adsorbed by van der Waals force and chemical bond. In addition, the intra-particle diffusion model fit in Figure 11(c) shows that the linear relationship between NC and Hg0 adsorption does not pass through the origin, indicating that the adsorption rate control process is controlled by the out-of-particle diffusion process (such as surface adsorption).
Conclusions
NC is a mesoporous silica material which has been chemically modified to form an FMMS on the micro surface. In this article, NC is used as a new adsorbent to study the adsorption performance for gaseous Hg0 under different experimental conditions.
Different mercury inlet concentration and adsorption bed temperature have great influence on the adsorption performance of NC. With the increase of concentration and adsorption temperature, the removal rate of Hg0 shows a trend of increasing first and then decreasing, and the higher the concentration and the temperature, the effect of NC adsorption is greater. The removal rate of Hg0 of NC is 75.78% under the optimum mercury concentration (223.9 μg m−3) and optimal adsorption temperature (60°C).
The composition of carrier gas has a significant effect on the Hg0 adsorption performance of NC. When using O2 (air) as the carrier gas, the removal rate of NC can reach 63.4%, and the removal rate decreases rapidly as only 14.9% after switching back to use N2 as carrier gas. And the removal rate of Hg0 decreased from 63.6% to 49.4% after adding SO2 to the air.
In the adsorption model fitting of NC, the theoretical maximum mercury adsorption of NC is calculated to be 1.61 mg g−1. The similar fitting degree between the two shows that there are both physical adsorption and chemisorption processes for the NC. First, the molecules of Hg0 are absorbed to the surface of NC by physical adsorption, then they are absorbed by chemical adsorption through slowly reaction. The fitting results of the adsorption kinetics model show that the pseudo-second-kinetic equation has the best fitting effect. At the same time, combined with the intra-particle diffusion model, it is speculated that the adsorption of Hg0 on NC is a multi-segment of “transmembrane–diffusion–adsorption.”
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Open Foundation of Shaanxi Key Laboratory of Lacustrine Shale Gas Accumulation and Exploitation (under planning), the Major Science and Technology Projects of Shanxi Province (no. 20181102017), the Open Project Program of State Key Laboratory of Petroleum Pollution Control (no. PPC2017010), CNPC Research Institute of Safety and Environmental Technology, and the Fundamental Research Funds for the Central Universities (no. 2009QH03).
