Abstract
Simultaneous adsorption of phenol and phosphate(V) ions on the surfactant-modified clay from the binary mixture was studied and compared with the single phenol or phosphate(V) sorption. The maximum adsorption capacity of hexadecyltrimethylammonium–bentonite was 18.8 mg/g for phenol and 38.5 mg/g for phosphate(V) at simultaneous adsorption of both of them. The optimal pH values of adsorption on hexadecyltrimethylammonium–bentonite was found to be 7.0 and >7 for phosphate(V) and phenol, respectively. The kinetic studies showed that the adsorption followed a pseudo-second-order reaction for both. The equilibrium data were analyzed using the Langmuir, Freundlich, Temkin, and Dubinin-Radushkevich isotherm models.
Introduction
Several methods are available for removing phenol and phosphate(V) from aqueous solutions. These are chemical precipitation (Aksu and Akpinar, 2001; Aksu and Gonen, 2006), solvent extraction, and adsorption (Chitrakar et al., 2005; Ciesielczyk et al., 2015). Among them, adsorption is an attractive method (Zhang et al., 2008), due to its high efficiency (Ozacar, 2003), ease of handling, and availability of different adsorbents (Bartczak et al, 2016; Ciesielczyk et al., 2016; Klapiszewski et al., 2015; Saha et al., 2009). Various kinds of new adsorbents for removal (Navarro et al., 2009) and recovery (Tanada et al., 2003) of phosphate have been reported (Geelhoed et al., 1997), of which natural clays and their composites are considered as particularly effective (Yang et al., 2016), low cost (Xue et al., 2009), and characterized by chemical stability (Wang and Xing, 2004; Tanada et al., 2003). In this paper sodium bentonite was modified by hexadecyltrimethylammonium bromide (HDTMA–Br) to obtain a more efficient sorbent. The paper deals with the subject of common sorption of phosphates and phenol which should be removed from wastewaters due to their unfavorable effects on the environment. So far the literature data on this subject have not been available. The effects of various experimental parameters including pH of the solution, contact time, initial ions concentration, dose of sorbent, and temperature as well as adsorption kinetics of isotherm models were investigated.
Materials and methods
Adsorbents
Preparation of HDTMA–bentonite
In order to obtain HDTMA–bentonite, 1 g of Na–bentonite was shaken for 4 h at 60℃ with 100 cm3 of 0.01 mol/dm3 hexadecyltrimethylammonium–bromide (HDTMA–Br) solutions. Then the time samples of the sorbent were decanted, washed with distilled water, and finally dried in air.
Adsorption experiments
Phosphate and phenol adsorption experiments were conducted using the batch technique under the following (conditions) terms: T 20 or 40℃, pH range 2–11, shaking time 6 h, 0.4 g of adsorbent, and 100 cm3 of phenol and phosphate solution with the initial concentration of 0.5 mmol/dm3. Sorption experiments were performed with the solutions containing phosphate and phenol and also compared with a solution containing only phosphate or phenol. The concentration of phosphate and phenol in the equilibrium solutions was analyzed spectrophotometrically by the molybdenum blue (Marczenko and Balcerzak, 1998) or the 4-Aminoantipyrine method (Marczenko and Balcerzak, 1998), respectively. The amounts of phosphate and phenol adsorbed onto the HDTMA–bentonite at equilibrium were calculated from the following equation



The Temkin isotherm was represented by equation

The Dubinin–Radushkevich isotherm was given by the following equation (Itodo and Itodo, 2010)
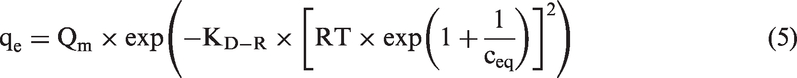
In the equations, qe and ceq denote the equilibrium concentrations of phenol or phosphate(V) in the clay (mol/g) and aqueous phase (mol/dm3); qmax, K, and n stand for adsorption maximum (mol/g), adsorption constants, heterogeneity parameter of the surface, respectively; Qm is the theoretical maximum capacity (mol/g); and bT is the Temkin constant (J/mol). The mean free energy of adsorption E (kJ/mol) from the Dubinin–Radushkevich isotherm was calculated using equation (6)

Results and discussion
Adsorption study
As follows from the graphs (Figure 1(a)) the adsorption of PO43− ions and phenol evidently increases with the contact time of the aqueous phase and the adsorbent. The adsorption equilibrium for both is determined after about 6 h. The pseudo-first-order model, pseudo-second-order model, and intraparticle diffusion model were tested for kinetic studies. The R2 values obtained for the pseudo-first-order model (0.73 for phosphate and 0.68 for phenol adsorption) and for intraparticle diffusion model (0.56 for phosphate and 0.51 for phenol adsorption) were low. The experimental data fitted very well the pseudo-second-order kinetic model and high correlation coefficients (>0.97) were obtained. These suggest that the adsorption data are well represented by pseudo-second-order kinetics (Abdelwaha, 2007). The constant Time (a), mass of adsorbent (b), and pH (c) influence on simultaneous adsorption of phosphate and phenol on HDTMA–bentonite. HDTMA: hexadecyltrimethylammonium.
Figure 2 presents the compared isotherms of phosphate and phenol adsorption at 20 and 40℃. The comparison of both isotherms confirms evidently that phosphate sorption on this adsorbent is much effective than phenol sorption. These diagrams along with those in Figure 2 show that the temperature increase of the process results in the increasing adsorption for phosphates and phenol. Figure 2 presents the adsorption of phosphate ions and phenol in their presence in the solution. As can be seen in the case of phosphates the presence of phenol deteriorates their sorption on HDTMA–bentonite though insignificantly. At the same time the addition of phosphate ions increases phenol adsorption significantly.
Adsorption isotherms of phosphate(V) (a) and phenol (b) on HDTMA–bentonite. HDTMA: hexadecyltrimethylammonium.
Parameters of isotherms for simultaneous adsorption of phenol and phosphate(V) ions onto HDTMA–bentonite.

Desorption of phosphate(V) and phenol from HDTMA–bentonite.

HDTMA: hexadecyltrimethylammonium.
The data collected in Table 2 prove that adsorption combination of HDTMA–bentonite–phosphates is more stable under any pH conditions than the phenol combination. In both cases the desorption in the acidic environment is the most intense and that in the neutral solutions is the least. As follows from the data presented in Table 2, desorption of phosphate ions is insignificant at pH 7 being about 8% whereas that of phenol is quite large, that is, about 40% at pH 7 and 50–60% at pH 3 or 9. Thus after earlier elution of phenol, it is possible to reuse HDTMA–bentonite as a sorbent for phosphate ions and phenol at pH close to 7 as under these conditions phosphate ions, which may be present on the sorbent, cannot desorb. However, this is the most favorable pH range for adsorption of both phosphate ions and phenol. Phosphate ions are strongly bonded with the adsorbate as the process is chemisorption. There desorption of these ions is insignificant even in alkaline or acid solutions. As phenol adsorption proceeds by means of weaker van der Waals binding, phenol can be washed away from the adsorbent surface using alkaline, acidic, or even of neutral pH solutions.
Conclusions
Simultaneous sorption of phosphate ions and phenol is possible on HDTMA–bentonite. The largest effectiveness of the process is observed at pH 7 for both ions. The values of the apparent free energy of adsorption from the Dubinin–Radushkevich isotherm and the heat of adsorption from the Temkin isotherm depict the physiosorption process for phenol and chemisorption in the case of phosphate ions.
Footnotes
Acknowledgements
First presented at the 15th Ukrainian–Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was carried out with the equipment purchased thanks to the financial support of the European Regional Development Fund in the framework of the Operational Program Development of Eastern Poland 2007–2013 (Contract No. POPW.01.03.00-06-017/09).
