Abstract
Pharmaceutical contamination of water sources presents serious risks to environmental sustainability and public health, requiring the development of affordable and effective wastewater treatment methods. Common treatment technologies often fail to fully remove pharmaceutical residues and are hindered by high energy use, operational costs, and the production of harmful by-products. Recently, research has increasingly focused on repurposing industrial wastes such as fly ash and sludge as low-cost, high-surface-area adsorbents for removing pharmaceuticals. This review thoroughly examines pharmaceutical pollutants, including their sources, environmental impacts, and concentration levels in wastewater. It assesses both traditional and emerging removal methods, with a particular emphasis on adsorption. The potential of raw and modified industrial waste materials as sustainable adsorbents is critically analyzed, considering key factors influencing adsorption, such as pH, temperature, initial concentration, as well as adsorption kinetics and isotherms. The review also emphasizes how these materials can be integrated into the circular economy by repurposing them for water purification. Recommendations for future research include enhancing adsorbent stability, reusability, and selectivity through modifications like magnetization, acid treatment, chitosan addition, and zeolitization, along with verifying their scalability. The results reinforce the importance of waste-based adsorbents in achieving sustainable water management.
Introduction
Wastewater treatment processes are responsible for playing a vital role in safeguarding public health, the environment, and ensuring the viable use of water resources by removing various pollutants, including pharmaceuticals in water. In developed countries, wastewater treatment plants have restrictive regulations and standards to enforce proper wastewater management and treatment in order to control the concentration of contaminants in effluents. Adherence to these restrictions is important for municipalities, industries, and other entities that generate treated wastewater. Therefore, proper wastewater treatment helps these entities to meet the regulatory guidelines, avoid penalties, and maintain their social and environmental responsibilities (Lourenco and Nunes, 2021). However, the current treatment processes are not efficacious, leading to the discharge of partially treated effluents into the environmental water sources. This discharge of inadequately treated wastewater containing pharmaceuticals into natural water bodies poses serious risks as it can exacerbate the spread of antibiotic-resistant bacteria and disrupt aquatic ecosystems. Moreover, it can cause long-term toxic effects on both wildlife and human populations who rely on contaminated water sources for drinking, irrigation, or recreation. These impacts not only threaten ecosystem integrity but also have significant social and economic consequences, including increased healthcare burdens and loss of valuable water resources. This can also cause other significant health risk concerns, such as waterborne diseases (Obaideen et al., 2022). Therefore, effective wastewater treatment processes are urgently needed to minimize and mitigate these detrimental effects and sustain the integrity of ecosystems and the environment (Mainardis et al., 2022).
Furthermore, efficient wastewater purification plays a significant role in supporting the principles of the circular economy through the use of solid waste such as coal fly ash from coal power stations and industrial sludge (Waleed, 2024). According to the circular economy, solid waste is recognized as a valuable resource that needs to be reused and reintegrated into the economy for further utilization as a secondary raw material. The circular economy is therefore defined as a skeleton designed to promote the extended retention of substances, resources, and products within the economic circular system to minimize waste production. Therefore, involving wastewater treatment plants in the economic system makes a valuable contribution to the sustainability of resources due to their capacity to reutilize water by extracting pollutants and harnessing energy resources (Guerra-Rodriguez et al., 2020). The reasons above have made the wastewater treatment topic attract huge attention from engineers, scientists in the water sector, environmental protection agencies, and academic institutions over the recent decades. The main benefits of proper wastewater treatment approaches are highlighted in Figure 1.

Benefits of proper wastewater treatment methods (Waleed, 2024).
Approximately half a billion people in the world are currently facing harsh, perpetual water scarcity, and the numbers are expected to increase up to 2.8 billion in 48 countries by 2050 (Chunyang et al., 2021). The world's supply and demand of fresh clean water is increasing exponentially with the rapid population growth, urbanization, industrialization, and over-exploitation of accessible water resources (Ahmad, 2023). Apart from the water scarcity point of view, the quality of the available water is deteriorating due to the multiple anthropogenic activities, producing and discharging an infinite amount of organic and inorganic pollutants (Sharabati et al., 2021). Water pollution, as a worldwide issue caused by the release of untreated and highly concentrated effluents, has led to the global contamination of water sources (Mainardis et al., 2022). Pharmaceuticals are among the organic pollutants that find their way to the environment mainly through sewage effluents, pharmaceutical discharges, dumping, excessive use of veterinary medicine in the agricultural industry, and as human medicine for therapeutic purposes (Kümmerer and Dionysiou, 2021; Muller and Padron, 2021). Virtually, half of the pharmaceuticals from multiple pathways end up in wastewater treatment plants with wastewater (Zhang, 2023). A recent review of existing literature on pharmaceuticals reveals that concentrations of 1.92–7.72 µg/L and 0.53–2.36 µg/L, 0.39–47.60 µg/L and 0.12–5.25 µg/L, 39–129 µg/L and 2.59–6.45 µg/L, 10960–12131 µg/L and 3980–19,540 µg/L, 0.016–1.48 µg/L and 0.023–0.81 µg/L, 0.056–0.22 µg/L and n.d–0.036 µg/L have been reported in influent and effluent wastewater from South Korea (Dong-Jin et al., 2023), South Africa (Kwangu et al., 2022; Hlengwa and Mahlambi, 2020), Nigeria (Olatayo et al., 2022), Ghana (Kodom et al., 2021), Russia (Perkons et al., 2021), and Italy (Massano et al., 2023), respectively. These findings indicate that water pollution by various pharmaceuticals, as well as their incomplete removal by wastewater treatment processes, is a global issue necessitating an urgent solution.
Multiple methods, including extraction, chemical precipitation, ozonation, electrochemical processes, filtration via membranes, coagulation, reverse osmosis, and advanced oxidation, have all been employed to remove pharmaceuticals from wastewater (Iqbal et al., 2024; Kamalesh et al., 2024; Shoma and Kansha, 2024; Prasad and Raval, 2024). However, while these procedures are effective in removing pharmaceuticals, they may include some major drawbacks, which include inflated costs, extreme conditions, and harmful chemicals. Adsorption as an improvement and convenient approach has been recognized as a potential alternative solution due to its low costs, high efficiency, and environmental friendliness. Even though various adsorbents have been used for the removal of pharmaceuticals in wastewater, their application is limited by their high synthesis costs, high operational costs, and limited reusability. As a result, industrial waste such as fly ash and sludge has been recently explored for the removal of pharmaceuticals from wastewater.
Therefore, the objective of this review is to provide insights into the recent relevant work reported in the literature on the utilization of fly ash and sludge as low-cost adsorbents for removing pharmaceuticals in wastewater. The review also discusses the critical directions from different standpoints on pharmaceuticals, including what they are, their uses, how they get into wastewater, their concentration levels, negative effects on the environment, and humans. Their adsorption process, how optimization parameters affect their removal efficiency, their performance as raw materials, and how they compare when they are functionalized. Merits such as kinetics and isotherms that describe their adsorption processes are also discussed. Furthermore, future directions and limitations are provided.
Background and literature review
Concept of pharmaceuticals and their contribution to environmental pollution
Pharmaceuticals are chemical products that have biologically active ingredients and possess specific positive modes of action in humans and animals for the treatment or disease prevention, and diagnosis. They also boost the skin beauty industry and are used as increments to stimulate the growth of livestock in order to maintain sustainability and profitability in the agricultural sector (Hejna et al., 2021; Monica et al., 2022). Their classes include antibiotics, nonsteroidal anti-inflammatory drugs, analgesics, and antiretroviral drugs, which are some of the most prescribed and used pharmaceuticals globally (Monica et al., 2022; Ahmad, 2023). According to the global market size, in 2024, the pharmaceutical market was valued at 3 trillion USD; simultaneously, the global prescription of pharmaceuticals was at 532 billion defined daily doses (Bajda, 2024). Pharmaceuticals such as nonsteroidal anti-inflammatory drugs, analgesics, and antibiotics are used to relieve pain and inflammation due to their antipyretic, anti-inflammatory, and analgesic properties (Hejna et al., 2021). Nonsteroidal anti-inflammatory drugs are regarded as the third most consumed pharmaceuticals globally, with consumption rates of 1280 tons/year in Germany, 2760 tons/year in Spain, 1800 tons/year in Canada, 3800 tons/year in Nigeria, and 2810 tons/year in South Africa (Monica et al., 2022).
Moreover, an estimated amount of 23,000 tons of antibiotics is consumed every year in South Africa, and 40,000 tons for antiretroviral drugs (Kwangu et al., 2022; Suranie et al., 2022). These pharmaceuticals are partially metabolized by the body and result in the excretion of up to 90% of the unused active pharmaceuticals as their metabolites or parental compounds through urine and fecal waste, where they may end up in wastewater (Suranie et al., 2022; Vinayagam et al., 2022). As conventional wastewater treatment plants are not effective in removing them, this results in their discharge into the environmental waters with effluents. Their disposal pathways also include the discharge of expired and unused drugs directly into the environment, where they get washed into environmental waters (Madikizela et al., 2022). In the United States of America, the total number of active pharmaceuticals was 11,926, and approximately 713 of those were discovered in wastewater (Femina-Carolin et al., 2021). Figure 2 shows the introduction pathways of pharmaceuticals into the environment.
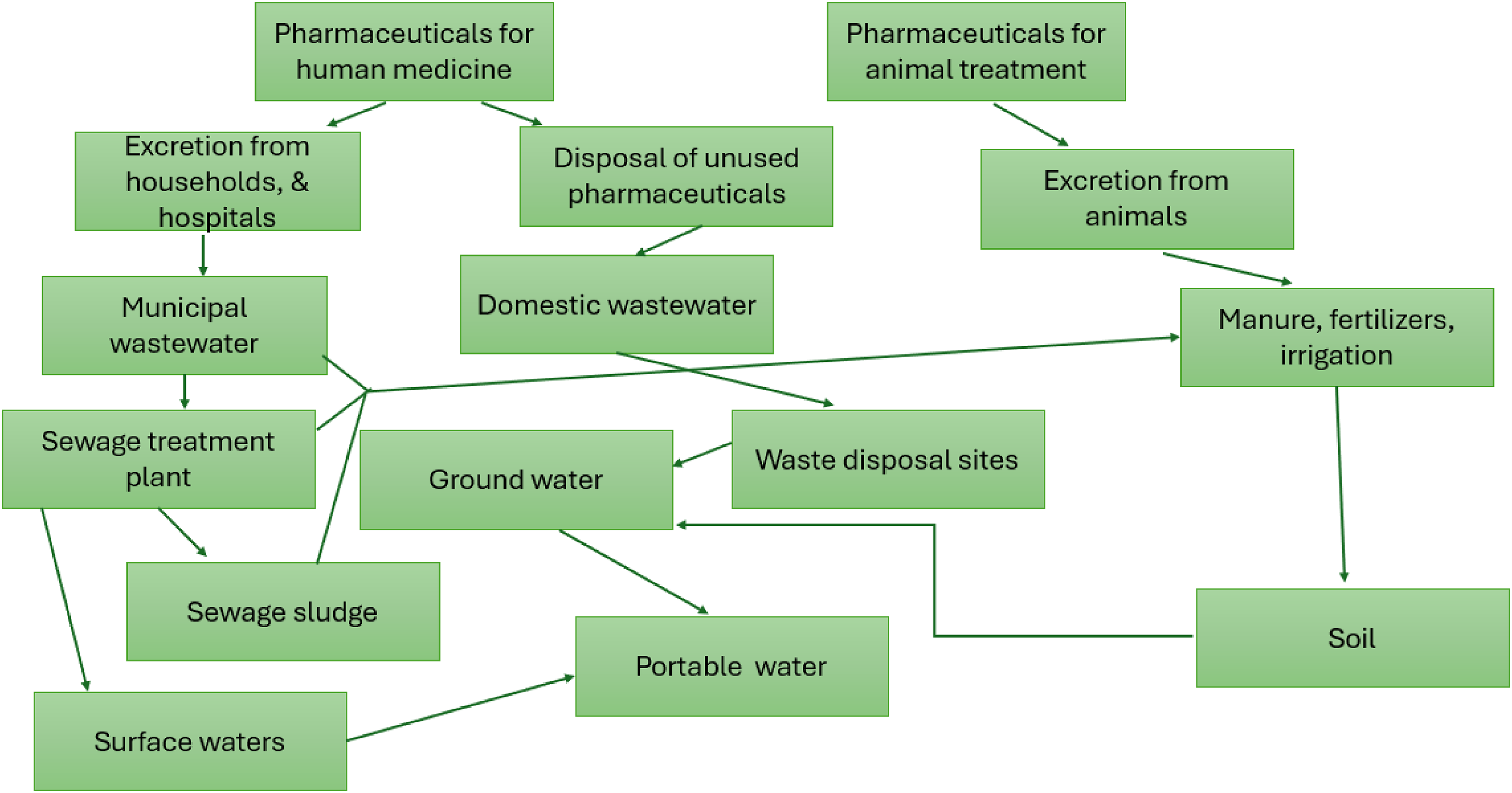
Pathways of pharmaceuticals into the environment (Vinayagam et al., 2022).
Negative effects of pharmaceuticals on humans, animals, and the ecosystems
There is an increasing concern among Ecotoxicologists regarding the global detection and quantification of pharmaceuticals in aquatic environments, as their long-term negative effects are being studied and reported in the literature (Selwe et al., 2022). Pharmaceuticals as emerging pollutants have been reported to cause harmful effects on the environment and human health, including impaired reproductive functions, mortality in aquatic animals, pollution of surface and groundwaters, and drug resistance in humans (Godlewska et al., 2021). Estrogens, as hormones that originate from livestock industries, are also important for human physiology but cause impaired reproductive functions and affect reproduction in humans when they enter the human food chains indirectly (Kusturica et al., 2022). These pollutants are also reported to cause higher chances of prostate and breast cancer in humans (Farounbi and Ngqwala, 2020). Drug resistance, which is a global concern, has increased exponentially and has been found to cause chronic diseases in humans, such as diabetes, asthma, and obesity (Kusturica et al., 2022). Simultaneously, antidepressants have been reported to cause bioaccumulation effects in aquatic species, which leads to increased cytotoxicity, liver and kidney damage (Akawa et al., 2021). Due to the interaction between humans and animal environments, antidepressant pollutants also cause reduced neurological development and psychiatric conditions.
Nonsteroidal anti-inflammatory drugs have shown to cause chronic ecotoxic effects, disorders in the organs of vertebrates and invertebrates by disturbing detoxification enzymes in the body (Mashile et al., 2021). Vinayagam et al. (2022) also reported that these pharmaceuticals cause hepatotoxicity and cardiovascular abnormalities in humans. Monica et al. (2022) observed a significant decline in the population of vultures in Asia, reduced prostate gland development, and damage to internal organs in aquatic species due to high concentrations of diclofenac. Furthermore, ibuprofen disrupts ovulation and motor behavior of aquatic species (Suranie et al., 2022). Aquatic plants and algae have also been found to be highly affected by concentrations of antibiotics in water, as they cause high oxidative stress and block photosynthesis (Femina-Carolin et al., 2021). Literature has also reported a huge increase in resistant strains of HIV, which are due to the concentrations of antiretroviral drugs in water (Eduardo et al., 2020). Figure 3 shows the health impacts of pharmaceuticals on aquatic species.
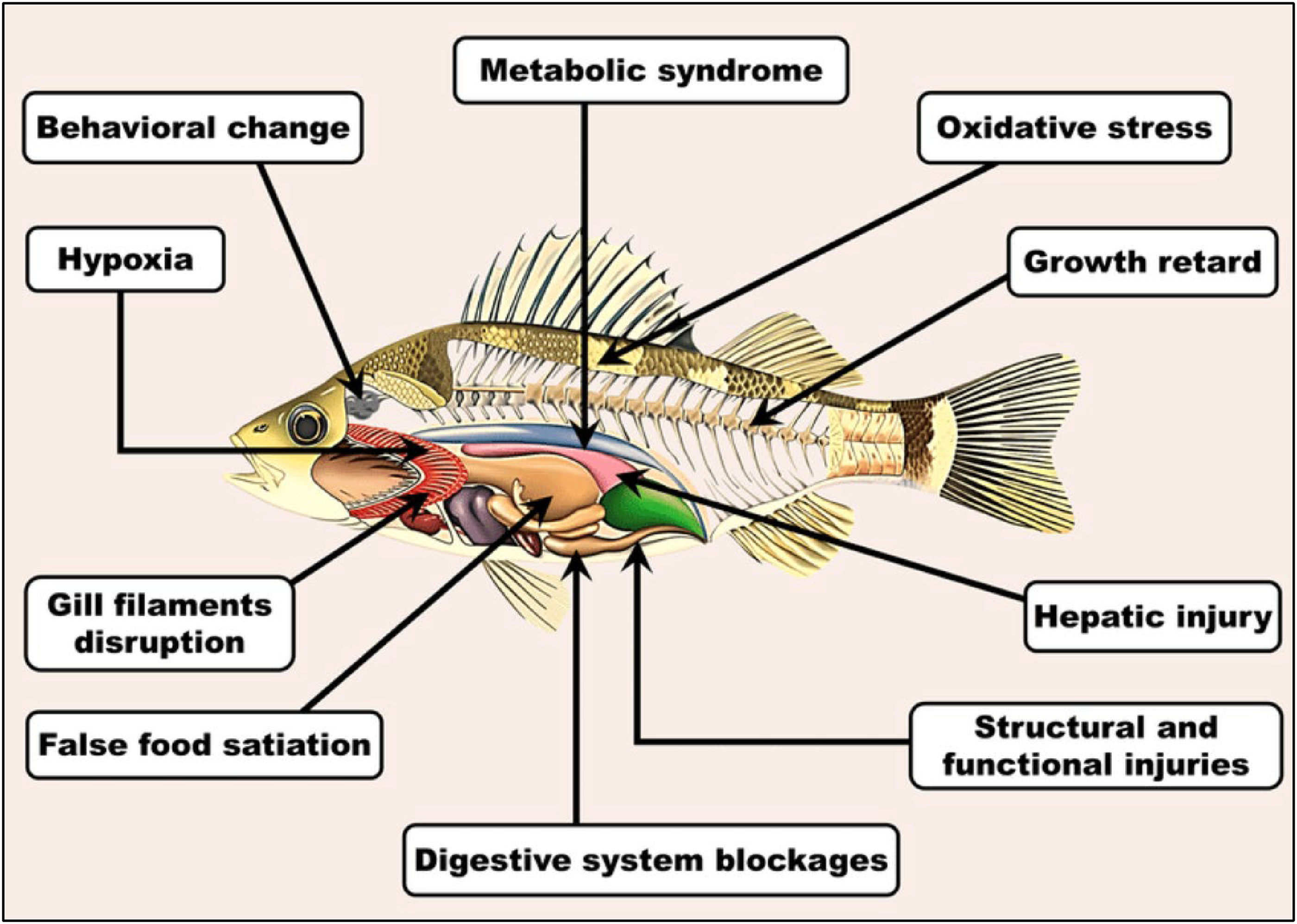
Effects of pharmaceutical pollution in aquatic species (Mahamud et al., 2022).
Various conventional methods for removal of pharmaceuticals
Cost-effective and efficient methods to address the global concern of pharmacyeuticals in effluent discharges into the environment remain a huge challenge in the water treatment sector (Alterary and Marei, 2021; Cuomo, 2023). Different strategies have been employed to treat wastewater contaminated with pharmaceuticals to protect human health and sustain various ecosystems. Filtration techniques such as membrane filtration, reverse osmosis, chemical oxidation approach, and electrochemical methods are some of the successful conventional methods used as removal strategies (Vinayagam et al., 2022). However, it is worth noting that these methods have significant disadvantages, such as managing operations, etc (Waleed, 2024). They also have drawbacks, such as huge power consumption, reduced effectiveness at low concentration levels of pollutants, production of harmful by-products, and disposal of the generated waste, which requires costly methods, as careless disposal leads to environmental pollution (Ahmad, 2023). In addition, filtration methods such as reverse osmosis and filtration via membranes have major disadvantages, such as membrane technology degradation due to exposure to extreme temperatures and other harsh conditions (Garcia-Avila et al., 2024). Oxidation processes such as ozonation and advanced oxidation have higher energy requirements to produce hydroxyl radicals that remove pharmaceutical pollutants in water; they produce ozone that has very low solubility in water, and the process results in the production of harmful side reaction products (Eduardo et al., 2020). Other methods include sedimentation, flocculation, and coagulation techniques, which are not efficient in removing pharmaceuticals due to the stability and mobility of some pharmaceuticals in wastewater (Wang et al., 2021; Bajda, 2024). To overcome the drawbacks mentioned previously, the adsorption method has shown good ability to remove pharmaceuticals efficiently in wastewater due to its simplicity, effectiveness, and minimized production of unwanted by-products (Vishwakarma, 2021; Seid et al., 2022).
Numerous conventional materials, such as activated carbon, alumina, and zeolites, have been effectively used as adsorbents for water purification purposes, which include the removal of pharmaceuticals in wastewater due to their characteristics such as polarity, large surface area, and high efficacy (Bajda, 2024; Cuomo, 2023). Furthermore, their performance can be enhanced by surface modification either through chemical or physical modification; however, their major drawback includes high costs and reduced reusability, even though they have multiple applications (Bajda, 2024). Additionally, in recent years, numerous natural-based adsorbents that can be obtained more easily, inexpensively, and in large quantities have been attempted as adsorbents. These include adsorbents derived from agricultural-based materials and industrial waste, focusing mainly on their attributes to remove pharmaceuticals in wastewater (Cuomo, 2023). Table 1 shows the various techniques used to remove pharmaceuticals in wastewater as reported in the literature, highlighting each method's advantages and disadvantages.
Strengths and weaknesses of various techniques used to remove pharmaceuticals from wastewater.

Industrial waste-derived low-cost adsorbents for removing pharmaceuticals
Industrial waste materials such as coal fly ash and sludge are generated by large-scale commercial operations and are often underutilized due to limited practical applications, leading to their disposal (Nippes et al., 2021). Because these materials are by-products, they are typically abundant, inexpensive, and readily accessible (Waleed, 2024). Many industrial wastes possess desirable physicochemical properties such as high surface area, the presence of pollutant-binding functional groups, and inherent adsorption efficiency that make them suitable candidates for use as low-cost adsorbents in wastewater treatment (Liu et al., 2022). Moreover, their surface properties can be improved through minor processing steps to further enhance their adsorption capacity (Selwe et al., 2022).
Various types of industrial waste, including fly ash, black liquor, sludge, and others, have been explored for their potential in removing pharmaceutical contaminants from wastewater. For instance, one study reported the use of biochar as an adsorbent for removing sulfamethoxazole, ibuprofen, and diclofenac, achieving removal efficiencies of up to 90%, with adsorption capacities of 24.06 mg/g, 569.6 mg/g, and 877 mg/g, respectively. These high removal efficiencies were attributed to favorable physicochemical interactions, specifically hydrogen bonding, hydrophobic interactions, and electrostatic forces between the biochar surface and the target pharmaceuticals (Mukarram et al., 2023).
Similarly, charcoal has been reported as an effective adsorbent for the removal of trimethoprim, with an observed adsorption capacity of 164 mg/g and a removal efficiency of 95%. The strong adsorption performance was attributed to electrostatic interactions facilitated by the amine group present in trimethoprim (Aderemi et al., 2023). In another study, pharmaceuticals containing hydroxyl and amine groups exhibited improved adsorption efficiency due to enhanced hydrogen bonding on the adsorbent surface, achieving removal percentages of up to 97% (Stojanovic et al., 2024). Although water is also capable of forming hydrogen bonds with the adsorbent surface, pharmaceutical molecules often form stronger or multiple hydrogen-bonding interactions, and additionally engage in hydrophobic and π–π interactions that water cannot participate in. These combined mechanisms allow the pharmaceuticals to compete effectively with water molecules, resulting in their high adsorption into the adsorbent surface. The adsorption mechanism is also affected by multiple factors. For example, while surface area is a fundamental factor influencing adsorption capacity, alone it does not fully determine the efficiency of pharmaceutical removal by industrial waste–derived adsorbents (Cuomo, 2023). Several other physicochemical properties of adsorbents and pharmaceuticals play critical roles in governing adsorption performance. The surface chemistry of the adsorbent, particularly the presence and type of functional groups such as carboxyl, hydroxyl, and amine groups, enables specific interactions like hydrogen bonding and electrostatic attractions with pharmaceutical molecules. These interactions often dominate the adsorption process by providing strong and selective binding sites. Additionally, the surface charge of the adsorbent and the pharmaceutical functional groups, which can vary with solution pH, influences electrostatic interactions, enhancing or hindering the adsorption of charged pharmaceutical species depending on their ionization states. The pore structure, including pore size distribution and porosity, also affects adsorption by controlling accessibility and diffusion pathways for pharmaceuticals of varying molecular sizes. Beyond these, hydrophobicity and π–π interactions significantly impact adsorption, especially for pharmaceuticals containing aromatic rings. Adsorbents with hydrophobic domains and aromatic structures facilitate strong hydrophobic and π–π stacking interactions, increasing adsorption affinity for such compounds. Moreover, surface heterogeneity and defects can create high-energy adsorption sites, further enhancing the binding capacity beyond what surface area alone can explain (Liu et al., 2022). These combined properties allow pharmaceuticals to be effectively adsorbed by adsorbents, resulting in high removal efficiencies from aqueous environments (Figure 4). Understanding and optimizing these multiple adsorbent characteristics is essential for developing more efficient and selective industrial waste–based adsorbents for pharmaceutical removal. Figure 4 shows how the characteristics of industrial waste contribute to their removal efficacy.

Adsorption process and different interactions involved in removing pharmaceuticals by industrial waste (Vinayagam et al., 2022).
Modifications of fly ash and sludge-based adsorbents
A recent study by Vukcevic and coworkers (2024) investigated the application of fly ash modified through alkali activation using sodium hydroxide, followed by incorporating chitosan coating to form a modified fly ash-chitosan adsorbent. Modification was performed by mixing 1 g of fly ash with 1 g of chitosan with 50 mL of 5% acetic acid. The mixture was then allowed to mix at room temperature for 24 h to ensure complete mixing. The resulting mixture was followed by injecting 1000 mL of 2 M sodium hydroxide and drying at 105 °C in an oven. Lastly, the product was washed with distilled water and allowed to dry at room temperature. In the study, the fly ash-chitosan and unmodified fly ash were used to adsorb erythromycin and doxycycline from wastewater, and the adsorbent displayed adsorption capacities of 17.74 mg/g and 314.21 mg/g, and percentage removals of 95% and 98.85% for erythromycin and doxycycline using the fly ash-chitosan. While the adsorption capacities were 12.21 mg/g and 201.45 mg/g, and percentage removals of 70% and 79% for the unmodified fly ash. The surface modification of the fly ash showed a remarkable surface area of 50.32 m2/g compared to the 20.32 m2/g of the unmodified, which showed that the modification was instrumental in the adsorption of the target compounds on the adsorbent surface (Vukcevic et al., 2024).
In another study published in the literature, an unmodified and sludge modified through thermal and pyrolytic treatment was used to remove carbamazepine and diclofenac from wastewater. The modified sludge was prepared by drying 10 g of sludge at 250°C in an oven for 2 h, the product was then cooled in a desiccator and used as an adsorbent. The modified adsorbent showed a remarkable percentage removal of 88% and 90% for carbamazepine and diclofenac, respectively, while they were 76% and 81% when the unmodified sludge was used (Varjuova et al., 2025).
Fly ash was also investigated as a potential adsorbent to remove tetracycline from wastewater. In the study, fly ash was modified through acidification, where 10 g of the fly ash was mixed with 50 mL of hydrochloric acid solution (30 w/w%) to form a slurry that was heated in an oven for 24 h at 100°C. The resulting product was then filtered using vacuum filtration and washed with deionized water multiple times and dried at 60°C in an oven to form an acid-modified fly ash. Simultaneously, a base-modified fly ash was prepared by mixing 20 g of fly ash with 160 mL of 3.5 M sodium hydroxide, and the slurry formed was dried at 100°C in an oven for 24 h. The resulting slurry was then washed with deionized water and dried at 60°C in an oven to form a base-modified fly ash. The adsorption showed that the acid-modified fly ash removed 35% of tetracycline with a surface area of 35.7 m2/g while the base-modified fly ash removed 76% with 110.8 m2/g surface area. These results show that base modification performed better than acid modification; however, it is worth noting that their performance is not consistently in that order (Houghton et al., 2025). The studies reported above show the importance and different methods of adsorbent modifications.
The reviewed studies demonstrate that the type of modification applied to fly ash and sludge strongly influences their adsorption performance for pharmaceutical pollutants. Chemical modifications, such as alkali activation combined with chitosan coating on fly ash, significantly enhanced adsorption capacity and surface interactions, often outperforming modifications that primarily alter physical structure, such as thermal treatment of sludge. Comparisons of acid- and base-modified fly ash further highlight that base treatment generally provides higher removal efficiency, likely due to increased surface area and generation of negatively charged sites, though effectiveness still depends on the specific target compound. Overall, these findings emphasize that the choice and combination of modification strategies, such as chemical, biological, or thermal, are critical in optimizing adsorption efficiency. They also indicate that low-cost, waste-derived materials like fly ash and sludge can be transformed into high-performance, environmentally and economically sustainable adsorbents.
Use of fly ash for pharmaceutical removals
Raw and modified fly ash
The use of fly ash as a cheap adsorbent for water remediation has gained global popularity (Ahmad, 2023). For example, a study by Maletic and coworkers reported the use of raw fly ash and chitosan-modified fly ash for the removal of multiple pharmaceuticals (diclofenac, ibuprofen, ketoprofen, naproxen, sulfamethoxazole, and ciprofloxacin) from wastewater. The adsorption capacities of 17.74, 23.12, 18.98, 18.23, 24.32, and 24.56 mg/g and percentage removals of 80%, 79%, 85%, 91%, 90%, and 79% were obtained for all the pharmaceuticals of interest for the chitosan-fly ash. However, the percentage removals were all less than 80%, with a maximum of 74%, and the adsorption capacity was reduced below 17.74 mg/g for raw fly ash, with a maximum of 15.23 mg/g. These results were all obtained at optimum conditions of pH 5.5, contact time of 60 min, adsorbent mass of 0.1 g, adsorbent dosage of 2 g/L, and room temperature. The adsorption process further showed that it conformed to pseudo-second-order kinetics, and equilibrium could be described by the Freundlich isotherm model for both the modified and raw fly ash (Maletic et al., 2024). This study has demonstrated that raw fly ash and chitosan-fly ash can be effectively used to remove pharmaceuticals from wastewater under optimum experimental conditions. Furthermore, it was shown that chitosan-fly ash outperforms the raw fly ash.
Tonglinxi and co-workers also reported the use of raw fly ash and microwave alkali-modified fly ash for the removal of ciprofloxacin from wastewater
A recent study on the removal of tetracycline antibiotic from wastewater using raw, acidified, basified, and zeolite-modified fly ash was reported in the literature. The adsorption from the used adsorbents showed a percentage removal of 35% for raw fly ash, 90% for zeolite-modified fly ash, 11% for acid-modified fly ash, and 76% for base-modified fly ash. The high percentage removals of base-modified and zeolite-modified fly ash were due to the fact that the adsorption mechanism was based on cation exchange rather than surface area. The zeolite-modified fly ash displayed high zeolite purity, a cation exchange capacity of 6.37 meq/g, and a surface area of 35.7 m2/g, while the base-modified fly ash had a higher surface area of 110.8 m2/g and a cation exchange capacity of 3.42 meq/g. The results for all the used adsorbents were obtained at a pH of 5, dosage of 7.5 g/L, temperature of 30 °C, contact time of 120 min, and 10 g of adsorbents. The adsorption isotherms indicated that adsorption conformed to the Langmuir model (Houghton et al., 2025). Zeolite-modified fly ash was the most effective, with removals of 90%, which could be attributed to the superior qualities of zeolites, such as high purity and ability to engage in cation exchange, further making them more reliable on cation exchange removal adsorptions rather than surface area.
Use of sludge for pharmaceutical removals
Various studies have shown that sludge can be used to adsorb pharmaceuticals from wastewater (Zhang, 2023; Muller and Padron, 2021). In a study by Qiu and colleagues, sludge modified into biochar was used to remove sulfamethoxazole from wastewater. In the study, the sludge biochar was modified at 300 and 800°C. Percentage removals of 95% were recorded with an adsorption capacity of 7033.4 mg/kg for sludge biochar at 800°C, while 75% and 100 mg/kg were obtained at 300°C. It was also reported that the high percentage efficiency and adsorption capacity at 800°C were due to the high accumulation of ash and formation of carbonate and organic anions during the high-temperature pyrolysis process, which increased its surface area. The results were obtained at an initial concentration of 10 mg/L for sulfamethoxazole, an adsorbent dosage of 1 g/L, a contact time of 24 h, and a sample pH of 7. The equilibrium obeyed the Freundlich isotherms (Qiu et al., 2023).
Another similar study reported the adsorption of ibuprofen, ketoprofen, and paracetamol from wastewater using sludge modified with biochar. Removal percentages of 85.16%, 90.76%, and 94.12% and adsorption capacities of 145.4, 105.9, and 57.6 mg/g were reported for paracetamol, ibuprofen, and ketoprofen, respectively. The high efficiency of the sludge-biochar was attributed to its high surface area and pore volume of 642 m2/g and 0.483 cm3/g, which promoted the adsorption of the pharmaceuticals. Experimental conditions were an adsorbent dosage of 1 g/L, a contact time of 180 min, an initial concentration of 50 mg/L, and sample pH of 8, 2, and 4 for paracetamol, ketoprofen, and ibuprofen. The experimental equilibrium data also conformed to the Langmuir isotherm (Streit et al., 2021).
Selected factors affecting adsorption efficiency
The effectiveness of the adsorption process is significantly affected by several optimization factors, which need to be optimized for enhanced efficiency and adsorption capacity (Singh and Srivastava, 2020). Figure 5 shows different factors that affect the adsorption process.

Factors affecting adsorption efficiency.
Effect of sample pH
Sample pH significantly affects pharmaceuticals’ adsorption due to its ability to influence the ionization of pharmaceuticals, the surface of the adsorbent, further affecting the removal efficiency (Waleed, 2024; Czech et al., 2021). During a pH optimization, a specific pH range is chosen, and the removal efficiency for the majority of the pharmaceuticals displays an increasing percentage removal trend as pH is increased until the removal efficiency is at its peak. Beyond this, the removal efficiency decreases. Hence, in a specific chosen range, there exists an optimal pH for adsorbing pharmaceuticals onto the adsorbent (Ma et al., 2020). However, it is also important to note that the effect of sample pH is also related to the point of zero charge of the adsorbent, which is the pH where the adsorbent has a neutral charge. The adsorbent displays a positive surface charge when the sample solution has a pH that is less than the point of zero charge of the adsorbent, while it displays a negative surface charge when the sample pH is greater than its point of zero charge. Therefore, the point of zero charge influences pharmaceutical removal efficiency by controlling the adsorbent's surface charge, which affects electrostatic interactions with charged pharmaceutical analytes (Chen et al., 2021; Li et al., 2020).
Streit and coworkers reported the adsorption of ketoprofen from wastewater using sludge modified with biochar. In their study, the percentage removals for all studied pharmaceuticals varied with pH from 2 to 10. The optimum percentage removal of ketoprofen was achieved at 90% at pH 6, and it was observed to be lower for pH values less than 4; however, when the pH was increased further from 6 to 10, the percentage removals declined sharply. This increase and decrease in percentage removals can be attributed to the pKa of ketoprofen (pKa of 4.6), sample pH, and the point of zero charge for the adsorbent (7.8). The high percentage removals are observed when the pKa is less than the sample pH (pKa 4.6 < pH 6), further making ketoprofen be in ionic form (negatively charged). Also, while the sample pH is less than the point of zero charge (pH 6 < pH0 7.8), the adsorbent surface ispositively charged. These cases allowed effective interaction of the negatively charged ketoprofen with the positively charged adsorbent surface, thus increasing the recoveries. Moreover, smaller recoveries were observed when the ketoprofen pKa was less than the sample pH (pH 8, 9, and 10 > pKa), and sample pH greater than the point of zero charge (pH 8, 9, and 10 > pH0 7.8), which resulted in the surface of the adsorbent being negatively charged while ketoprofen was also negatively charged. Furthermore, lower recoveries were obtained at pH values less than pKa (pH 1, 2, 3, and 4 < pKa), making ketoprofen positively charged, while the pH values were also less than the point of zero charge (pH 12, 3 and 4 < pH0 7.8), making the adsorbent surface to be positively charged resulting in repulsion (Streit et al., 2021).
The reviewed work highlights that sample pH is a major determinant of pharmaceutical adsorption, as it affects both the ionization state of the pharmaceutical and the surface charge of the adsorbent, governed by its point of zero charge (pH₀). The studies show that maximum adsorption occurs when the pharmaceutical and adsorbent carry opposite charges, for instance, when a negatively charged pharmaceutical (pH > pKa) interacts with a positively charged adsorbent surface (pH < pH₀), promoting strong electrostatic attraction. Conversely, adsorption efficiency decreases when both carry similar charges, leading to repulsion at very low or high pH. The reviewed work emphasizes that the optimal pH is system-specific, depending on the interplay between the pharmaceutical's pKa and the adsorbent's pH₀, and that careful pH optimization is essential for effective pharmaceutical removal in complex wastewater systems.
Effect of adsorbent dosage
An increase in the adsorbent dosage has been reported to increase the surface area and enhance the removal efficiency of the adsorbent. This is due to an increase in the number of active sites on the adsorbent, which improves the adsorption and further increases the removal efficiency (Zhao et al., 2021). However, it is also important to note the dosage at which the adsorbent reaches saturation.
A study on the removal of triclosan and ciprofloxacin using a sludge modified with biochar showed that their removal efficiency differs with different adsorbent dosages. In the study, it was shown that an increase in adsorbent dosage from 0.2 to 0.5 g/L increased the removal efficiency from 71.8% when 0.2 g/L was used to 91.2% when 0.4 g/L was used for ciprofloxacin, while a further increase resulted in a decline in the removal efficiency. Simultaneously, an increase in adsorbent dosage increased the removal efficiency of triclosan from 69% when 0.2 g/L was used to an optimum of 82.2% from an adsorbent dosage of 0.4 g/L which was the optimum, a further increase also resulted in a decrease of removal efficiency (Ihsanullah et al., 2022). The increase in removal efficiencies for both pharmaceuticals was due to an increase in the number of active sites available on the surface of the adsorbent, which favors adsorption, hence higher removals. Moreover, a reduction in removal efficiency may be due to competition between the two pharmaceuticals for an active site (Ihsanullah et al., 2022).
The removal efficiency of contaminants is clearly influenced by the amount of adsorbent used. Increasing the adsorbent dosage generally improves removal because more active sites are available for adsorption. However, this improvement is not indefinite; beyond a certain point, further increases in dosage can actually reduce efficiency. This decline likely occurs due to competition between different contaminants for the same adsorption sites and possible aggregation of adsorbent particles, which reduces the accessible surface area.
In my view, these observations highlight the importance of optimizing, rather than maximizing, adsorbent dosage. Determining the optimal dosage ensures the most effective removal of contaminants while avoiding unnecessary material use and costs. It also underscores the complexity of adsorption in multi-contaminant systems, where interactions between pollutants can influence overall performance. Therefore, careful consideration of dosage is essential to achieve both high efficiency and practical applicability in real-world treatment systems.
Effect of contact time
The effect of contact time on the adsorption removal efficiency for pharmaceuticals is a significant factor to optimize, as it influences the interaction between the adsorbent and the adsorbate. The studies revealed that an increase in contact time increases the removal percentage due to more time allowed for the pharmaceuticals to be adsorbed on the active sites of the adsorbent. However, there is also a point in time where the removal efficiency reaches equilibrium, and an increase in contact time no longer affects the removal efficiency (Sharabati et al., 2021; Chunyang et al., 2021).
In a recent study where fly ash was used to adsorb diclofenac from wastewater, it was shown that diclofenac removals vary with contact time when it was investigated between 0 and 15 h. The trend displayed that the diclofenac removals increase from 0 to 9 h with an optimal of 90% and reached equilibrium with a further increase in contact time, where the removal efficiency did not change (Magdalena et al., 2023). The increase in efficiency may be due to the efficient time given for diclofenac to adsorb to active sites of the adsorbent, while the constant removal efficiency after 9 h may be due to oversaturation of the active site, hence no impact on removals (Ponce-Robles et al., 2020; Kusturica et al., 2022; Kosma et al., 2020).
Contact time plays a crucial role in the adsorption of pharmaceuticals, as it dictates the interaction between the adsorbent and the target molecules. Initially, adsorption is rapid due to the abundance of available active sites, allowing efficient uptake. Over time, the process slows and eventually reaches equilibrium, reflecting the saturation of the adsorbent surface. This suggests that identifying this equilibrium point is essential for optimizing adsorption systems. Extending contact time beyond equilibrium provides no additional benefit and may reduce operational efficiency, highlighting the need to balance sufficient exposure with practical considerations in treatment design.
Effect of initial concentration of an analyte
The influence of the initial analyte concentration on the adsorption capacity and removal is significant and needs to be optimized. Generally, the removals increase with an increase in initial concentrations until an optimum initial concentration is reached, where the removals either decline or reach equilibrium.
Ihsanullah and coworkers reported the use of sludge to remove triclosan from wastewater, and the initial concentration was found to affect its removal. The concentration was varied from 10 to 60 mg/L, and the results showed an increase in the adsorption capacity (optimum of 80.89 mg/g) and removal percentage (maximum removal of 89%) as the initial concentration was increased from 10 to 60 mg/L (Ihsanullah et al., 2022). This trend was attributed to the increase in the number of available sites for triclosan to be adsorbed as the amount of triclosan is increased (Ihsanullah et al., 2022). However, it was expected that the efficiency was going to either decline or reach equilibrium as more initial concentration was increased, which was going to be due to a huge number of triclosan being adsorbed, resulting in saturation of the active sites.
The initial concentration of an analyte plays a pivotal role in determining both the adsorption capacity and overall removal efficiency. At lower concentrations, many adsorption sites remain underutilized, limiting the system's effectiveness. As the concentration increases, the driving force for mass transfer grows, allowing more molecules to interact with the adsorbent surface and thereby enhancing adsorption; however, this enhancement is not limitless. Beyond a certain point, the adsorbent surface approaches saturation, and additional analyte molecules can no longer be accommodated effectively. This leads to a plateau in removal efficiency or even a potential decline if overcrowding or competition for active sites occurs. From this perspective, the relationship between initial concentration and removal highlights the importance of identifying an optimum concentration that maximizes adsorption without overwhelming the system. Balancing these factors is essential for designing efficient and practical adsorption processes.
Effect of adsorption temperature
Optimizing adsorption temperature plays a crucial role in enhancing the efficiency of pollutant removal. If the adsorption process is endothermic, increasing the temperature typically improves adsorption capacity and removal efficiency by increasing molecular movement and promoting stronger interactions between the adsorbent and pollutant. This leads to higher percentage recoveries. If the process is exothermic, higher temperatures may reduce adsorption effectiveness by weakening the binding forces, resulting in lower removal rates (Ma et al., 2020). An exothermic adsorption process means heat is released when a substance (adsorbate) binds to the surface of an adsorbent, and an endothermic process means the opposite. The effect of temperature on the adsorption of pharmaceuticals using sludge-biochar as an adsorbent was assessed between 15 and 45°C, where the results showed that an increase in temperature also increases the adsorption capacity of the adsorbent. This trend was displayed by the adsorption of fluoroquinolone antibiotic using sludge-biochar, the removal efficiency increased from 15°C (65%) to 25°C (89%), then declined to 70% at 45°C (Ma et al., 2020). Another study reported by Shin and colleagues also showed an increase in adsorption capacity when the temperature was investigated from 15 to 35°C, where 25°C was the optimum with a percentage removal of 90%. The probable reason for the trends explained previously was found to be due to an increase in temperature, which results in a thermodynamically spontaneous system (negative Gibbs free energy) and an enthalpy change value that is between 0 and 40 kJ/mol. This led to the conclusion that the adsorption was due to van der Waals forces (Shin et al., 2020; Ma et al., 2020). Therefore, it is important to investigate the effect of temperature because, if not properly assessed, it will result in a lack of understanding of the mechanism of pollutant removal, which can be exothermic or endothermic. Studies have also shown that it is not advisable to investigate the effect beyond 50°C, as this could result in the possible decomposition of analytes (Madikizela et al., 2022). The results obtained from the effect of temperature are further employed to conduct thermodynamics studies. Thermodynamic studies evaluate Gibbs free energy (ΔG°), enthalpy (ΔH°), and entropy (ΔS°) changes. This helps to understand the spontaneity (gives information on whether adsorption occurs spontaneously or non-spontaneously), heat exchange (whether energy is absorbed or released during adsorption), and the disorder involved in adsorption processes. These further help in understanding the interaction between the adsorbent and the adsorbate on the adsorbent surface (Madikizela et al., 2022). The equations 1–4 are also used to calculate the thermodynamic parameters.




where
Different studies on pharmaceutical removals from wastewater using industrial waste (fly-ash, sludge).
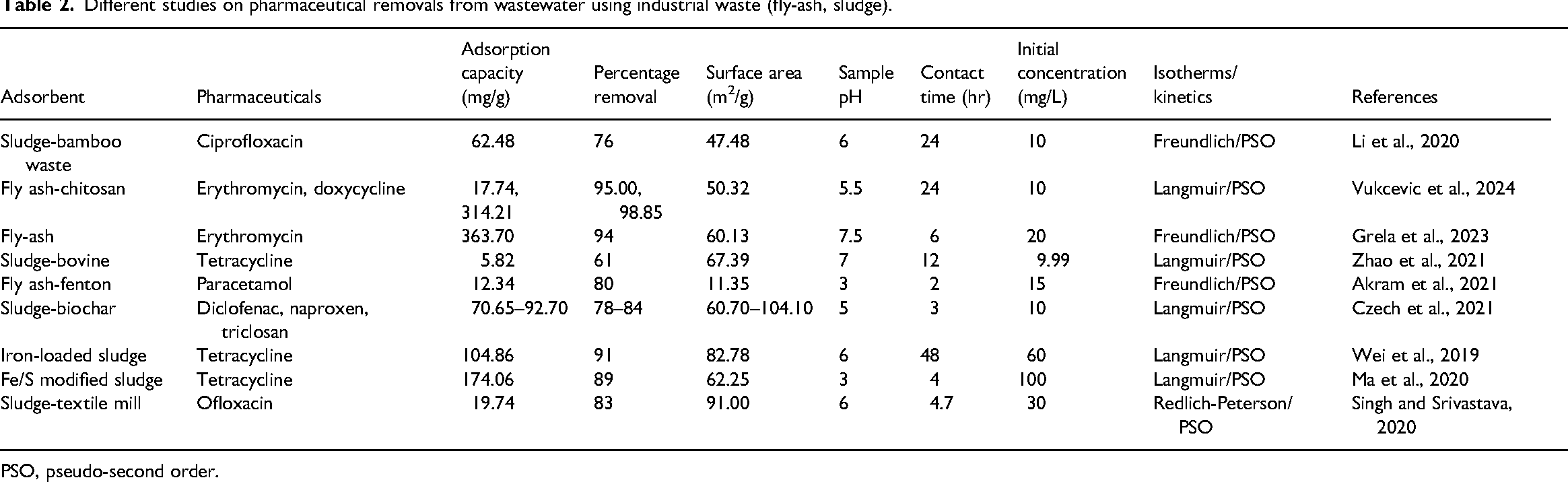
PSO, pseudo-second order.
Effect of pharmaceutical properties and adsorbent characteristics on adsorption efficiency
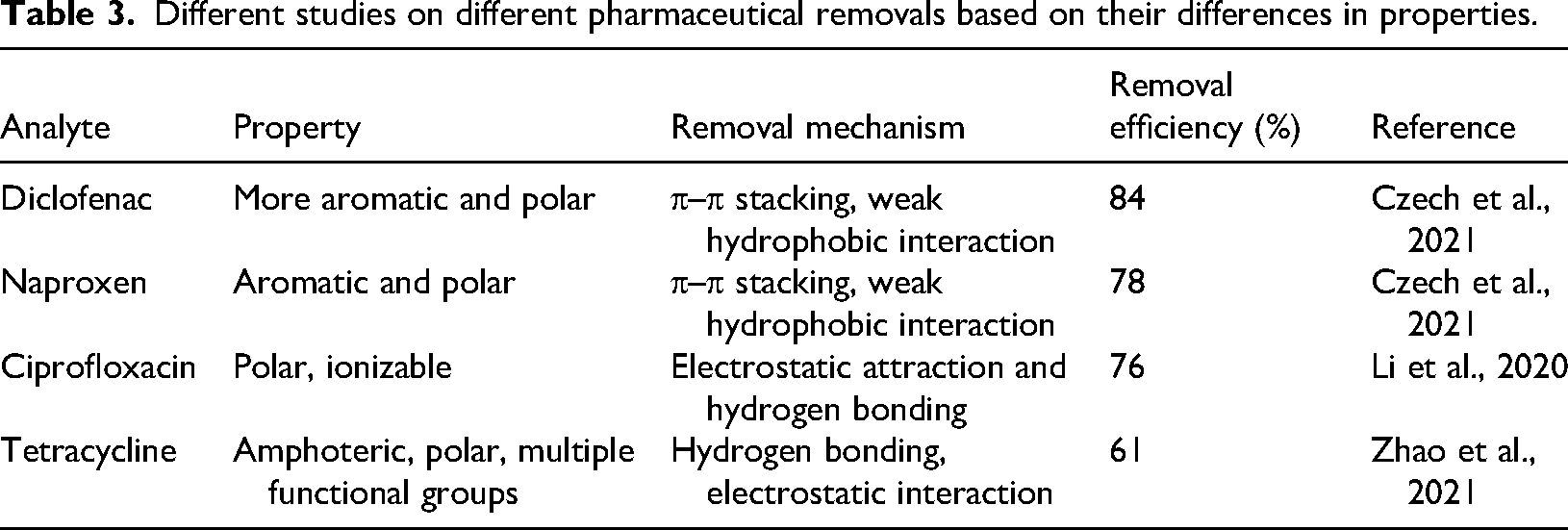
Pharmaceutical compounds have considerable diversity in terms of chemical structures, molecular size, polarity, and functional groups, all of which strongly influence their adsorption behavior (Vukcevic et al., 2024). For example, small molecular size pharmaceuticals that are also non-polar may adsorb primarily through hydrophobic interactions and π–π stacking, and they may diffuse more easily into the adsorbent's mesopores than large molecular-sized pharmaceuticals. Polar or ionizable compounds may interact via hydrogen bonding or electrostatic forces. These further result in different removal efficiencies and consequently, no single adsorbent can efficiently remove all pharmaceuticals. Industrial waste-based adsorbents, such as fly ash, have shown promise due to their large surface area, porosity, and reactive surface functional groups (Grela et al., 2023). However, adsorption efficiency depends on the compatibility between the adsorbent surface and the pharmaceutical molecule. Studies indicate that adsorbents can effectively remove certain pharmaceuticals, but the removal efficiency varies depending on the molecular properties of each compound. For example, in a study reported by Czech et al. (2021), diclofenac showed a slightly higher removal efficiency than naproxen under the same adsorbent, sludge-biochar. This was attributed to the high aromaticity of diclofenac, which facilitated π–π stacking, weak hydrophobic interaction. Table 3 shows how fly ash, as an adsorbent, removes different pharmaceutical compounds.
Different studies on different pharmaceutical removals based on their differences in properties.
Adsorption kinetics and isotherms
The adsorption kinetic studies are used to assess the adsorption rates, which are affected by different mass transfer conditions and depend significantly on temperature, pressure, and the type of adsorbent and adsorbate (Obaideen et al., 2022). Different models have been developed to assess the process and rate-determining steps. Adsorption kinetics for pharmaceuticals have generally been explained using either pseudo-first order or pseudo-second order. The pseudo-first order assumes that the number of active sites unoccupied on the adsorbent surface area is directly related to the adsorption rate. However, the second order assumes that the rate of adsorption is directly related to double the number of unoccupied sites or that the interaction between adsorbent and analyte is significant (Iqbal et al., 2024; Grela et al., 2023). Equation (5) describes the first order, while equation (6) describes the second order.

where
q(t) represents the amount of adsorbate adsorbed on the surface of the adsorbent at any time t (mg/g)
K1 represents the rate constant of the pseudo-first-order model (min−1)
K2 represents the rate constant of the pseudo-second-order model (min−1)
Qe represents the amount of adsorbate adsorbed into the adsorbent at equilibrium (mg/g)
t represents time (min). Literature has reported that adsorption of pharmaceuticals is commonly described by second-order kinetics (Czech et al., 2021; Ma et al., 2020). Singh and colleagues investigated the application of biochar-sludge to remove tetracycline from wastewater. In the study, an adsorption capacity of 112 mg/g and a removal percentage of 98% was obtained for the selected pharmaceutical. It was reported that the adsorption kinetics were described by pseudo-second order (Singh et al., 2021).
The adsorption isotherms also allow for the determination of equilibrium performance of the adsorbent at a constant temperature as well as the mechanism of adsorption between the adsorbent and the adsorbate (Shin et al., 2020). There are two adsorption isotherms: The Freundlich model
The equations below describe the models.

where:
Qm represents the maximum monolayer adsorption of the adsorbent (mg/g), and KL represents the Langmuir equilibrium constant (L.mg/g).
Regeneration and re-usability studies
Adsorbent reusability refers to the ability of an adsorbent material to be used in multiple adsorption cycles, while regeneration involves restoring its adsorption capacity after use. These processes are essential for cost-effective and environmentally sustainable treatment, as they reduce the need for continuous replacement of materials and minimize waste generation. Furthermore, successive applications of adsorbents depend on the ability of the adsorbent to be regenerated and recycled, together with their ability to adsorb different pollutants (Singh et al., 2021; Singh and Srivastava, 2020).
A recent study reported in the literature investigated the application of modified Fe/S-biochar for the removal of diclofenac from wastewater, and four successive regeneration cycles were performed on the adsorbent. The obtained results showed removal efficiencies of 95.7% and 78.2% for cycles 1 and 2, while they remained constant for 3 and 4 at 78.2%. A high decrease in the percentage removals for the four cycles was due to the desorption of loaded Fe/S and oversaturation of the adsorbent for cycles 3 and 4, while a decrease was observed for cycles 1 and 2 due to a reduction in active sites (Ma et al., 2020).
In summary, from the authors’ perspective, regeneration and reusability represent some of the most critical yet unresolved challenges in the application of adsorbents for water treatment. While many materials exhibit excellent initial adsorption performance, their ability to retain this capacity over repeated cycles is often limited. Significant reductions in removal efficiency after one or two regeneration cycles indicate that the functional groups or reactive components responsible for adsorption are not structurally stable during repeated use. This suggests that the adsorption mechanisms may rely heavily on surface-bound species that are altered, degraded, or lost during regeneration. Such instability raises practical concerns. In real-world treatment systems, adsorbents must withstand multiple cycles of adsorption and regeneration to be considered economically and environmentally viable. When a material shows substantial performance decay within the first few cycles, its long-term applicability becomes uncertain. Although some degree of performance reduction is expected, steep declines limit scalability and increase operational costs due to the need for frequent replacement.
The authors believe that improving regeneration stability requires a deeper understanding of the factors contributing to performance loss, whether due to structural changes, active-site depletion, leaching of modifying agents, or irreversible binding of pharmaceutical pollutants. Developing more robust material designs, along with regeneration protocols that preserve the integrity of active sites, will be essential for advancing these adsorbents toward practical implementation. In summary, while adsorbents hold significant promise for pollutant removal, their future applicability will depend strongly on achieving greater durability and stability across multiple regeneration cycles. Addressing this limitation should be a priority in ongoing and future research.
Leaching and environmental safety of industrial waste adsorbents
Sludge and fly ash have emerged as low-cost adsorbents for pharmaceutical removal; however, their environmental safety remains a critical concern. While both materials can effectively adsorb a range of pharmaceuticals, their composition differs significantly, influencing leaching behavior. Fly ash typically contains higher concentrations of heavy metals such as arsenic, lead, and cadmium, whereas sludge may have more organic-bound contaminants and variable metal content depending on its source (Singh et al., 2021). Comparative studies indicate that fly ash may pose a higher leaching risk under acidic conditions, whereas sludge leaching can be more variable but potentially mitigated through stabilization. Pre-treatment strategies such as calcination for fly ash or alkali/acid treatment for sludge have been shown to reduce leaching, but their effectiveness depends on specific material properties and operational conditions (Grela et al., 2023). Therefore, while both adsorbents are promising, a critical evaluation of their leaching potential, treatment history, and post-use disposal is essential to ensure environmental safety, highlighting that adsorption efficiency alone is insufficient to assess their suitability for wastewater treatment.
Recommendations and future work
Future work should focus on expanding the application of industrial wastes such as fly ash and sludge beyond their current applications, which are predominantly centered around the removal of heavy metals, dyes, and nutrients from different matrices. There is a huge gap in studies investigating their effectiveness in removing contaminants like pharmaceuticals. To enhance their performance and recovery potential, these wastes should be further modified through magnetization, zeolitization, and chitosan polymerization, as magnetized adsorbents allow for easier separation and reuse, while zeolite and chitosan structures improve porosity and selectivity. Additionally, current limitations in the use of industrial waste include limited practical applications in the water treatment sector, potential leaching of toxic elements, and structural instability. These must be systematically addressed through comprehensive risk assessments and standardized modification protocols. Future work should also aim to integrate these materials into real-world treatment systems and assess their long-term performance under operational conditions, as they show remarkable removal potential. Finally, life cycle assessments and techno-economic analyses are essential to determine the sustainability and economic feasibility of deploying these waste-derived adsorbents at scale, ensuring they offer a viable and environmentally responsible solution for water treatment.
Conclusions
In conclusion, the growing environmental and public health concerns related to pharmaceutical pollutants in wastewater highlight the urgent need for more efficient, cost-effective, and sustainable treatment methods. While common methods such as filtration and chemical oxidation have their limitations, the utilization of fly ash and sludge presents a promising alternative. These materials are abundant and relatively inexpensive and offer significant potential for pharmaceutical removal due to their high surface area and effective adsorption capabilities. However, their performance can be further enhanced through modifications, which improve their recovery and selectivity. Despite their advantages, challenges remain, including the potential leaching of toxic substances and structural instability, which must be addressed through further research and development. As the demand for clean water rises globally, particularly in regions facing water scarcity, the integration of waste-derived adsorbents into wastewater treatment systems offers a sustainable solution that aligns with the principles of a circular economy. Future research should focus on optimizing these materials, assessing their long-term performance, and conducting comprehensive life cycle and economic evaluations to ensure their feasibility and widespread implementation in tackling the global issue of pharmaceutical contamination in water.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of South Africa (Grant no. SRUG2204133298), Water Research Commission (Grant no. C2024/2025-01671).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that supports the findings of this study are available in the article.
