Abstract
Octafluoropropane (C3F8) is widely used as etching agent in refrigeration, air conditioning and semiconductor industries. However, due to its long lifespan and strong infrared absorbing ability, once C3F8 is emitted, the atmospheric radiation absorbing ability will be permanently altered, which will result in a serious greenhouse effect. Therefore, the efficient removal technology of C3F8 is crucial in protecting the environment and alleviating the greenhouse effect. In this work, a series of X-ETS-4 (X: Mg, Ca, Sr, Ba) molecular sieves were synthesized by solvothermal and ion-exchange method. The morphology and structure of the prepared X-ETS-4 were characterized by FT-IR, XRD, and SEM, etc. The adsorption performance of the X-ETS-4 on C3F8 are determined by fixed-bed adsorption breakthrough experiments and single-component isothermal adsorption experiments. The adsorption mechanism was investigated using different adsorption theoretical models. The results show that Ba-ETS-4 exhibits high adsorption capacity and high adsorption selectivity for C3F8, achieving C3F8 adsorption separation at very low concentration (C3F8/N2 volume ratio = 1:400). The saturated adsorption capacity of Ba-ETS-4 on C3F8 reaches 155.17 mg/g (298 K, 170 kPa), which is 15.49 times more than that of Na-ETS-4. In addition, the ideal adsorption solution theory (IAST) separation selectivity of C3F8/N2 reaches 341–941 in the pressure range of 0–170 kPa.
Introduction
Octafluoropropane (C3F8), also known as perfluoropropane, is characterized by high chemical stability, colorless and odorless, high electronegativity and insulation, etc. It is widely used as etching agent in refrigeration, air-conditioning industry and semiconductor industry. C3F8 is the fourth most abundant perfluorinated gas in the world. Refrigeration/air conditioning and semiconductor manufacturing are the main sources of C3F8 emissions. In addition, the aluminum industry is also a potential source of C3F8 emissions.
According to the United Nations Climate Assessment Report, C3F8 is a typical greenhouse gas with a GWP100 (Global Warming Potential) of 8,830, which is stable and has a lifespan of more than 2600 years, and is not easily degraded. Once emitted, perfluorocarbons gases (PFCs) permanently change the absorption capacity of the atmosphere due to their long lifetime and high infrared absorption capacity, which are associated with the extreme stability. According to some current emission reports, the global baseline mole fraction of C3F8 has increased nearly tenfold in the past 40 years. In addition, some literatures show that calculated emissions of PFCs are significantly higher than most previous estimates, suggesting that actual emissions of PFCs are significantly underestimated (Calvin et al., 2023; Mühle et al., 2010). Therefore, the development of efficient C3F8 removal technology is very important to protect the environment and alleviate the greenhouse effect.
The concentration of C3F8 in the air is very low, and the global baseline mole fraction of C3F8 is only at the ppt level (Say et al., 2021). The separation at the ppt level is obviously not desirable, so the removal of C3F8 can be focused on the removal of the exhaust gas from the relevant plant, usually below 2000 ppm. For this separation of components at very low concentration, the adsorption separation method has the highest operability due to low energy consumption, long adsorbent life, no long-term maintenance, and simple process. To meet the demands of industries such as semiconductors and electronics, enabling the recovery of valuable resources while reducing environmental burdens, developing an efficient physical adsorption process for the separation and recovery of C3F8/N2 at low concentrations is both essential and challenging.
Among the reported adsorbent materials, the fluorinated cage-structured Zn-bzc (Zheng et al., 2024) exhibits strong adsorption affinity for perfluorinated compounds. By introducing -CF₃ groups, not only is the material's hydrophobicity enhanced, along with its high chemical stability, but its adsorption selectivity for C₂F₆ is further improved. However, due to its complex preparation process, large-scale production remains challenging. In contrast, although Co0.2Cr-MIL-101 (Ye et al., 2023) demonstrates excellent C₃F₈ adsorption performance, the extensive use of heavy metal ions during its synthesis poses certain environmental risks. This limitation somewhat restricts its potential for widespread application.
Among many adsorption materials, molecular sieves are a very promising class of adsorption materials in the field of adsorption and separation, with relatively low costs. At present, a considerable number of them have been commercially applied, them have the advantages of high selectivity, high stability and mature preparation process, and are widely used in VOCs treatment, CO2 capture and adsorption of a series of nitrogen oxides (Peng et al., 2024; Zheng et al., 2024). However, there are various types of molecular sieves with different properties, and are greatly affected by working conditions. Currently, there are few literature reports on adsorption separation studies of C3F8/N2, it is of great significance to screen the adsorbents with superior performance among them for the adsorption of low concentration C3F8.
Based on the above considerations, Na-ETS-4 molecular sieves were synthesized by solvothermal method, and then modified by ion exchange to obtain a series of X-ETS-4 (X: Mg, Ca, Sr, Ba) molecular sieves, the adsorbent particles with the particle size of 20–40 mesh were screened out using an artificial break briquetting process, which was convenient for subsequent fixed-bed adsorption breakthrough experiments. The synthesized X-ETS-4 molecular sieves were characterized by X-ray diffraction (XRD), scanning electron microscope (SEM), Fourier transform infrared spectroscopy (FT-IR) and thermogravimetric analysis (TGA). The adsorption performance of X-ETS-4 was measured by fixed-bed adsorption breakthrough experiments. The adsorption heat, adsorption selectivity, desorption and regeneration performance and adsorption mechanism of C3F8 and N2 on X-ETS-4 were investigated in detail.
Materials and methods
Materials
The synthesis of ETS-4 was carried out using the following reagents: Silicon dioxide (SiO2, 100–200 mesh), Sodium hydroxide (NaOH, > 99%), Titanium butoxide (>99%), Hydrogen peroxide (H2O2, ≈30%).
The following reagents were used for the ion exchange modification of ETS-4: Magnesium chloride (MgCl2, > 99%), Calcium chloride (CaCl2, > 99%), Strontium chloride (SrCl2, > 99%), Barium chloride (BaCl2, > 99%) and Deionized water.
All the above reagents were purchased from the Shanghai Aladdin Biochemical Technology.
Experimental instrumentation
The experimental apparatus employed in this study is enumerated in Table 1.
List of experimental instrument names, models and manufacturers.

Synthesis of ETS-4
ETS-4 was prepared using a hydrothermal method according to the previous literature (Maghsoudi et al., 2025; Peng et al., 2023), with titanium butoxide and silicon dioxide serving as the titanium source and silicon source respectively.
First, two solutions were prepared separately as the silicon source and titanium source. Solution 1: Dissolve 2 g of NaOH in 100 mL of deionized water, then slowly add 24 g of silica gel under constant stirring. Solution 2: Dissolve 2.4 g of NaOH in 200 mL of deionized water and slowly add 12 g of titanium butoxide dropwise, then add 37 mL of H2O2 in three batches until stirring to obtain a bright yellow transparent solution.
Next, combine Solution 2 with 35 mL of deionized water and add it to Solution 1, stirring for 30 min. The mixture was then transferred into a 500 mL Teflon-lined autoclave, maintaining at 473 K for 48 h. The resulting product was washed with deionized water, filtered, and dried at 363 K for 12 h to obtain a white powder product, Na-ETS-4.
Synthesis of various single-cation exchanged ETS-4 materials
The synthesized through the above experiments was Na-ETS-4, which was subjected to ion exchange using 0.1 mol/L solutions of MgCl2, CaCl2, SrCl2, and BaCl2. The synthesized Na-ETS-4 was separately mixed with four different salt solutions at a ratio of 15 g Na-ETS-4 to 400 mL of salt solution. The mixtures were stirred at 70°C and 400 rpm for 8 h. Afterward, the solutions were centrifuged, and the adsorbent was washed with deionized water and dried at 90°C for 12 h. And the samples obtained were denoted as Mg-ETS-4, Ca-ETS-4, Sr-ETS-4, and Ba-ETS-4.
Granulation of powder X-ETS-4
To control the pressure drop during the dynamic adsorption process, the powdered zeolite samples were into compressed compacted blocks, followed by manual granulation and sieving to obtain granular samples with a particle size of 20–40 mesh. The granular samples were then stored in a desiccator for subsequent experiments.
Results and discussion
Structural analysis and characterization of X-ETS-4
The powder XRD patterns of the as-synthesized sample (Na-ETS-4) and the ion-exchanged samples (X-ETS-4) are presented in Figure 1. The main diffraction peaks are located at 2θ=7.6, 12.7 and 30.0°, which are coincident with the results reported in the literature (Lin et al., 2009). This agreement confirms the successful synthesis of the ETS-4 series samples in this work.
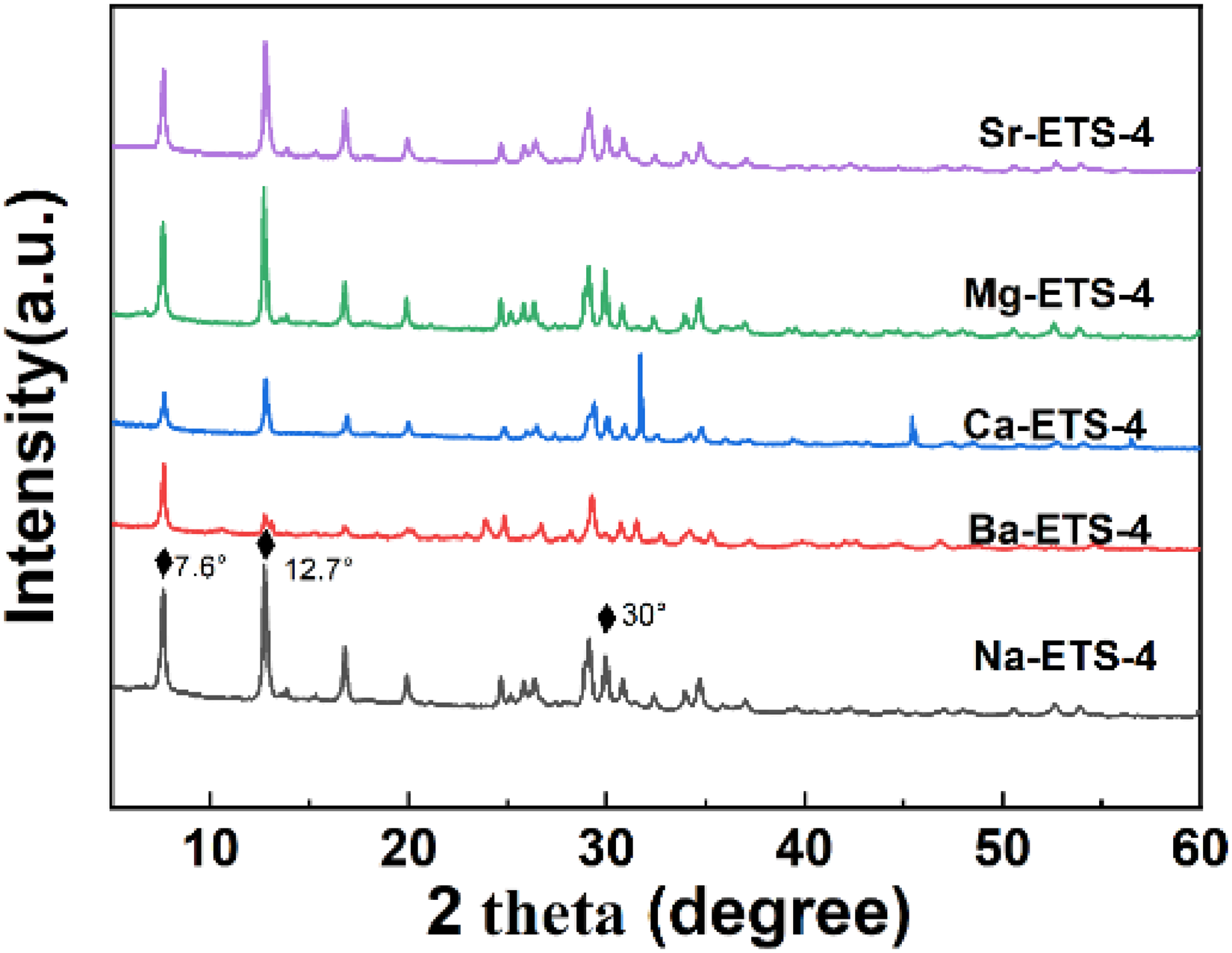
XRD patterns of X-ETS-4 samples.
Additionally, compared to Na-ETS-4,the XRD results for Ba-exchanged samples Ba-ETS-4 exhibit lower distinct peaks at 2θ=7.6, 12.7 and 30.0°, which is consistent with previous literature (Majumdar et al., 2011; Rashtchi et al., 2024; Vosoughi and Maghsoudi, 2022). This observation suggests that the incorporation of barium ions induces structural rearrangement, consequently affecting the crystallinity of Ba-ETS-4. The high polarizability of Ba²⁺ (α = 1.55 ų for Ba²⁺) enhances its interaction with polar C₃F₈ molecules. Additionally, Ba²⁺ exchange contributes to moderate enlargement of the material's pore channels, which facilitates C₃F₈ adsorption.
FTIR was used to test the functional groups of X-ETS-4 samples. As shown in Figure 2(a), these spectrograms are consistent with the patterns documented in the past literature (Maghsoudi et al., 2025). As seen from Figure 2, all the absorption peaks of the samples corresponded well, indicating that the ion-exchange-modified X-ETS-4 series samples retained the same functional groups as Na-ETS-4. The peaks at 2800–3600cm−1 corresponds to the symmetric and asymmetric stretching vibrational peaks of -OH. This extensive absorption peak may be due to the presence of silanol or titanol groups on the outer surface of the crystals as well as water molecules adsorbed on the crystal surface (Qiu et al., 2020; Rashtchi et al., 2024).

(a–b) FTIR spectra of X-ETS-4 samples (400–4000cm−1).
From Figure 2(b), the peak at 1650 cm−1 corresponds to the bending vibrational mode of -OH, which may be caused by water molecules adsorbed on the crystal surface. In addition, the peak at 1130 cm−1 is indicative of the presence of Si-O-Si bonds; The peak at 980 cm−1 indicated the presence of Si-O-Ti bonds; The peak at 700 cm−1 indicated the presence of Ti-O-Ti bonds; The peaks at about 450 cm−1 and 500 cm−1 are attributed to Si-O bonds and TiO4 tetrahedra. Infrared analysis showed that the metal ions were successfully coordinated to the ligands, and Na-ETS-4 and X-ETS-4 modified by ion exchange method had the same functional groups.
In order to explore the effect of different ion-exchanged on the morphology and structure of ETX-4, the crystal morphology and size of X-ETS-4 were observed and characterized by SEM. Figure 3 shows the SEM images of X-ETS-4 samples. We can see that after different cation exchanges the macrostructural lengths of Na-ETS-4, Ba-ETS-4, Ca-ETS-4, Mg-ETS-4, and Sr-ETS-4 are 22.36 μm, 16.66 μm, 26.89 μm, 19.01 μm, and 25.79 μm. There is no significant change in the macroscopic external shape of the adsorbent, but the crystal length of Ba-ETS-4 seems to be shorter compared to the other adsorbents, which is consistent with the literature (Rashtchi et al., 2024).

SEM images of X-ETS-4 samples: (a) Na-ETS-4; (b) Ba-ETS-4; (c) Ca-ETS-4; (d) Mg-ETS-4; (e) Sr-ETS-4.
From Figure 4, EDX spectra of Na-ETS-4, Ba-ETS-4, Ca-ETS-4, and Sr-ETS-4 demonstrate the presence of the ion-exchanged in the molecular sieve structure. In contrast, the presence of Mg2+ was difficult to detect in Mg-ETS-4. This analysis confirms that Na2+, Ba2+, Ca2+ and Sr2+ were successfully ion exchanged while Mg2+ was difficult to do so. This may be related to the properties (e.g., size and charge) of Mg2+ and the structure of the molecular sieves (Dyer, 2007).

EDX spectra of X-ETS-4 samples: (a) Na-ETS-4; (b) Ba-ETS-4; (c) Ca-ETS-4; (d) Mg-ETS-4; (e) Sr-ETS-4.
The thermal stability of the adsorbent material is an influential factor in the ability of the adsorbent to be widely used. The TGA curve of X-ETS-4 was determined from 303 K to 1073 K under N2 atmosphere. From Figure 5, compared to Na-ETS-4, the decomposition rate of Ba-ETS-4 and Mg-ETS-4 exchange slows down and the mass residue increases. The decomposition rates and mass residues of Sr-ETS-4 were essentially same as before. Due to the desorption of physically water, the Ca²⁺-exchanged sample exhibited accelerated decomposition rate and reduced residual mass (Panezai et al., 2016). Therefore, the Ba-ETS-4 has more excellent thermal stability.
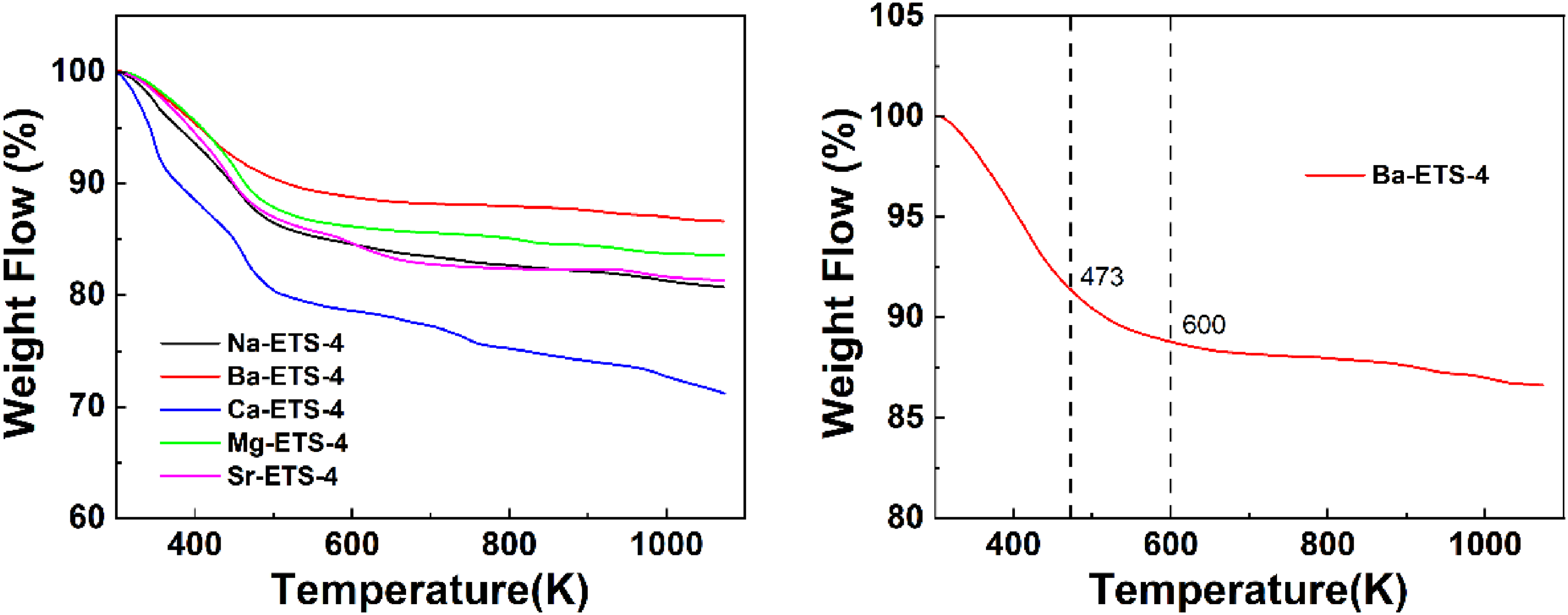
(a–b) TG curves of X-ETS-4 samples.
From Figure 5, the weight loss processes of the Ba-ETS-4 sample can be divided into three main stages: The first stage is 300–473 K. The mass loss in this stage is mainly due to the evaporation of residual solvent molecules from the material. The second stage is 473–600 K. The mass loss in this stage is mainly due to the decomposition and removal of some unstable groups or guest molecules on the surface of the skeleton, in addition to the continued evaporation of a small number of residual solvent molecules. The third stage is 600–1073 K. After the removal of solvent molecules and some unstable groups on the surface, the curve enters a plateau period and the rate of material decomposition becomes extremely slow due to the excellent thermal stability of the adsorbent material skeleton. Thermogravimetric results showed that Ba2+ ion exchange improved the thermal stability of the material (Rashtchi et al., 2024).
Adsorption performance of X-ETS-4
Breakthrough experiments of X-ETS-4
In order to investigate the adsorption breakthrough behavior of X-ETS-4 on C3F8/N2 mixtures, breakthrough experiments on five different molecular sieves were performed at low concentrations of octafluoropropane. Figure 6 shows the results of the breakthrough experiments of five different cationic X-ETS-4 at a flow rate of 1.0 L/min, temperature of 298 K, and pressure of 1.70 kPa in a gas mixture (V (C3F8:N2 = 1:400)). The experimental initial concentration and bed height were selected based on (Ye et al., 2023), while the flow rate of 1 L/min was determined by comprehensively considering both the mixed gas pressure of 1.7 MPa and fixed-bed pressure drop. Except for Ba-ETS-4, the rest of the molecular sieve adsorbent materials quickly permeate through and quickly reach adsorption saturation at the beginning of the experiment. In contrast, Ba-ETS-4 exhibited the longest breakthrough time (36.7 min) as well as the longest saturation time (71.1 min), which was about 8.88 times that of Na-ETS-4 before modification. This indicates that Ba-ETS-4 delivers a better dynamic adsorption performance of C3F8. This may be due to the introduction of barium ions, which increases the adsorption active site of C3F8 and effectively improves the adsorption performance of octafluoropropane (Kim et al., 2025).
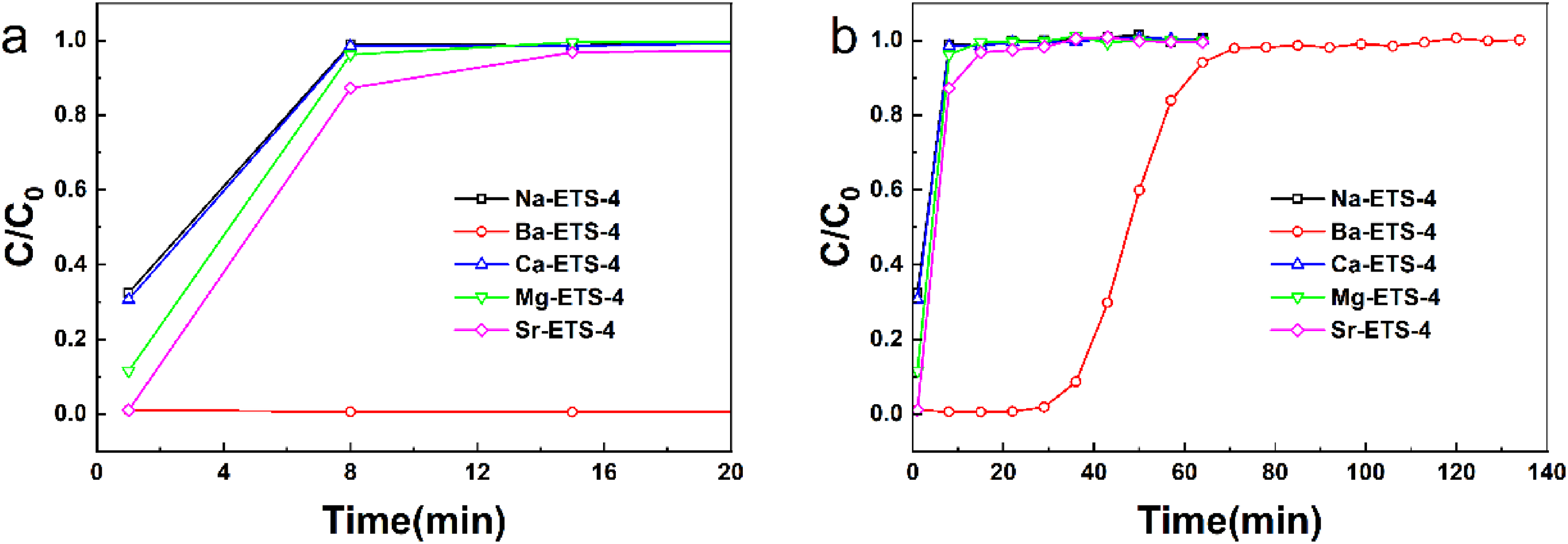
(a–b) C3F8 breakthrough curves for Ba-ETS-4 sample (conditions: T: 298 K, P: 170 kPa, flow rate: 1.0 L/min, V (C3F8:N2 = 1:400)).
Table 2 summarizes the calculation of dynamic saturation adsorption of different cations X-ETS-4. From the table, the dynamic saturated adsorption of C3F8 by Ba-ETS-4 reached 155.17 mg/g(0.83 mmol/g), which is 15.49 times higher than that before modification. The above analysis shows that the introduction of barium ions on the surface of ETS-4 by ion exchange can effectively improve the adsorption affinity of the adsorbent with octafluoropropane, increase the adsorption active sites, and effectively improve the adsorption performance of the material for C3F8. The Ba-ETS-4 with better adsorption performance will be further investigated in subsequent experiments.
Dynamic saturation adsorption of X-ETS-4 series samples.

Adsorption isotherms and adsorption model fitting of C3F8 and N2 on Ba-ETS-4
In order to investigate the adsorption separation mechanism of Ba-ETS-4 for separating N2/C3F8, isothermal adsorption experiments for Ba-ETS-4 were conducted. Figure 7 shows the single-component adsorption isotherms of N2 and C3F8 on Ba-ETS-4 material at 298, 303 and 308 K. Ba-ETS-4 exhibited high C3F8 adsorption, reaching 0.83, 0.77 and 0.72 mmol/g at 298, 303 and 308 K, respectively. Correspondingly, the amount of N2 adsorbed by the material was 0.27, 0.22 and 0.19 mmol/g. Moreover, the adsorption of N2 and C3F8 by the material decreased with the increase of temperature in the measured temperature interval, which indicates that the adsorption of N2 and C3F8 by the surface material is a typical physical adsorption process. At the same temperature, the adsorption of C3F8 by the material is significantly higher than that of its N2, which indicates its excellent separation potential.

Isotherms and model fitting of N2 and C3F8 adsorption on Ba-ETS-4.
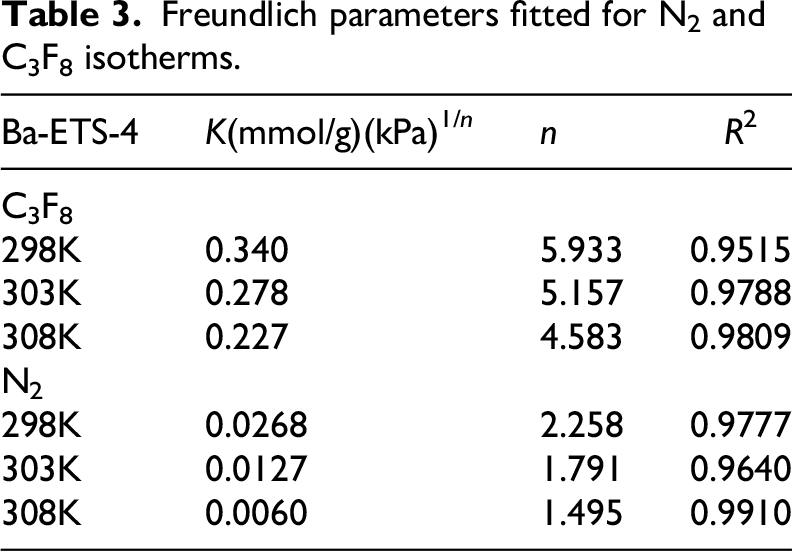
Langmuir (Alshammari, et al., 2024; Wu et al., 2024) and Freundlich (Wang, et al., 2022) adsorption models were used to fit the Ba-ETS-4 adsorption isotherms for C3F8 and N2 at different temperatures, and the Freundlich adsorption model, which was selected as the superior fit, is plotted in Figure 7. The fitted parameters are listed in Table 3. The adsorption of Ba-ETS-4 on C3F8 and N2 is in accordance with the Freundlich adsorption model, indicating that the adsorption process is characterized by multilayer adsorption, with a large number of inhomogeneous active sites on the surface of the material. These active sites are favorable to increase the adsorption affinity of the Ba-ETS-4 for C3F8. Summary of adsorption capabilities of C3F8 adsorbents at 298 K and 100 kPa is listed in Table 4.
Freundlich parameters fitted for N2 and C3F8 isotherms.
Summary of adsorption capabilities of C3F8 adsorbents at 298 K and 100 kPa.

Adsorption heat of C3F8 and N2 on Ba-ETS-4
In order to quantitatively study the difference in the adsorption force of Ba-ETS-4 on the two gases, we used the Virial equation to fit the adsorption isotherms of C3F8 and N2 of the material at three temperatures, 298, 303 and 308 K. And based on the obtained parameters, the equivalent heat of adsorption (Qst) of the material on C3F8 and N2 were calculated separately. The Virial equation (Czepirski and JagieŁŁo, 1989) is shown below: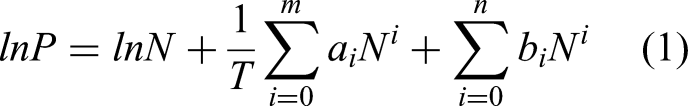
P(kPa) is the partial pressure of the adsorbed gas; N(mmol/g) is the adsorbed amount at the corresponding pressure; T(K) is the experimental temperature; ai, bi are the Virial coefficients, which denote the interactions between i molecules of adsorbate; m, n is the number of parameters needed to describe the fitting curve. Theoretically, as describing the equation of state of an ideal gas, the larger m and n are, the more accurate the fitting result will be. In practice, generally 4–6 can be taken to achieve a better fitting effect (Jalili et al., 2019).
After fitting the adsorption isotherms, the equivalent heat of adsorption of the material to C3F8 and N2 was calculated using the Qst equation based on the fitted virial coefficients ai. The calculated equations are as follows:
R is the gas constant; the remaining parameters are described with reference to the above equation.
Figures 8 shows isotherms and virial fitting of C3F8 and N2 adsorption on Ba-ETS-4. Table 5 shows the fitting parameters and correlation coefficients of the Virial equation fitting the adsorption isotherms of Ba-ETS-4 on C3F8 and N2, and R2 are 0.9842 and 0.9650, which indicate that the Virial equation can simulate the adsorption process of the material on the two gases to a certain extent. The equivalent heat of adsorption (Qst) calculated using its correlation parameters has a certain degree of confidence.
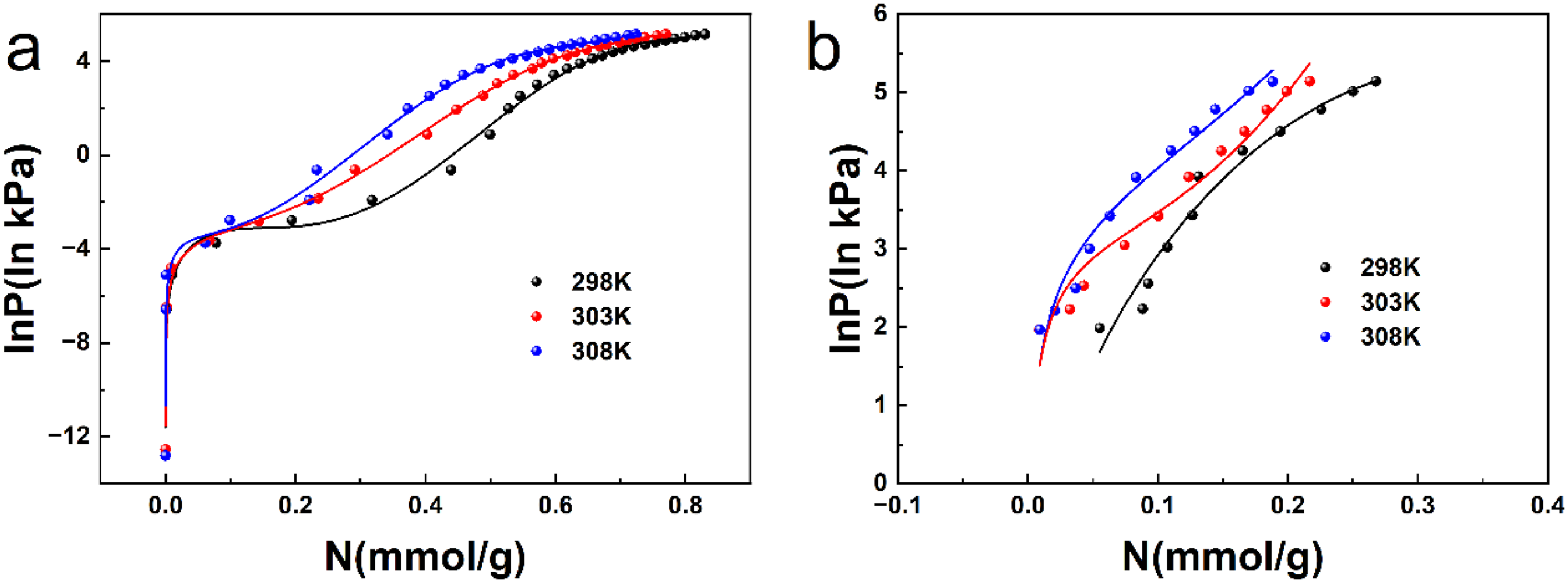
(a–b) isotherms and virial fitting of C3F8 and N2 adsorption on Ba-ETS-4.
Virial model fitting parameters and correlation coefficients.

Figure 9 shows the Adsorption heat of C3F8 and N2 on Ba-ETS-4. When the adsorption amount tended to be 0, the initial Adsorption heat of Ba-ETS-4 on C3F8 and N2 is 30.78 and 20.54 kJ/mol. The Adsorption heat of Ba-ETS-4 on C3F8 and N2 increased and then decreased with the adsorption amount, and finally tended to be 27.33 kJ/mol. And throughout the adsorption process, the Adsorption heat of Ba-ETS-4 on C3F8 (27.33–32.31 kJ/mol) was higher than that of its on N2 (16.36–20.54 kJ/mol), which indicated that the material possessed a stronger interaction force on C3F8. Besides, the Adsorption heat of both is less than 40 kJ/mol, which belongs to the category of physical adsorption, and the lower level of adsorption heat also makes the desorption process easier.

Adsorption heat of C3F8 and N2 on Ba-ETS-4.
Adsorption selectivity of C3F8 and N2 on Ba-ETS-4
Adsorption isotherms of Ba-ETS-4 for C3F8 and N2 at 298 K were fitted using the Dual-Site Langmuir-Freundlich (DSLF) model, The Adsorption selectivity of Ba-ETS-4 for a binary mixture of C3F8 and N2(1:400) was calculated using the parameters obtained from the fitting carried over to the Ideal Absorbed Solution Theory (IAST) model(Prausnitz, 1965; Walton and Sholl, 2015). The DSLF equations are as follows:
For the adsorption of components on solid surfaces in multicomponent mixtures, it is usually able to be described using IAST. In this paper, the adsorption selectivity of C3F8/N2 is calculated using the DSLF and IAST models based on the assumption that the adsorption of each component on the solid surface is independent of each other and the components do not react with each other. The equation for calculating the adsorption selectivity of IAST is as follows:
Table 6 shows the parametric results of fitting the adsorption isotherms of Ba-ETS-4 for C3F8 and N2 at 298 K by DSLF model. From the table, the fit of adsorption isotherms of Ba-ETS-4 for C3F8 and N2 are 0.9993 and 0.9811. This surfaces that the adsorption process of the material can be effectively fitted by using the DSLF model, and the fitting parameters will be utilized to further compute the IAST selectivity later.
DSLF model fitting parameters and correlation coefficients.
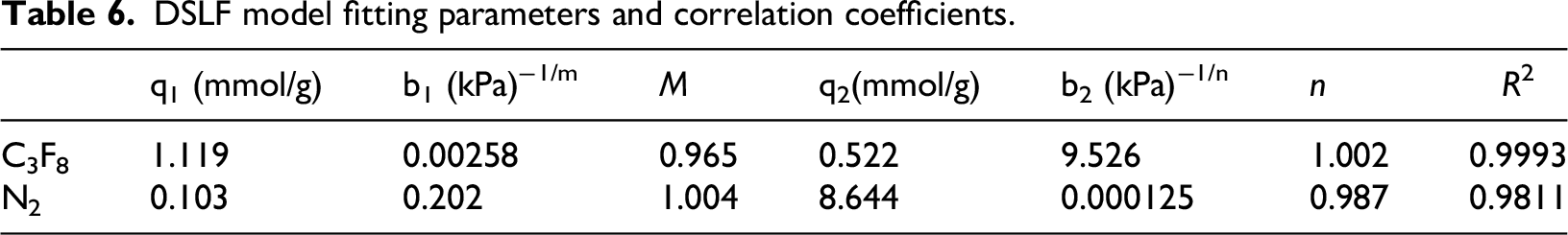
Figure 10 shows the calculated IAST selectivity of Ba-ETS-4 at 298 K for a binary mixture of C3F8/N2 (1:400). As can be seen from the figure, the selectivity of the material increases and then decreases with increasing pressure. In the range of 0–90 kPa, it gradually rises from 341 to 941. and then in the range of 90–170 kPa, it gradually decreases to 841. The higher selectivity of Ba-ETS-4 for C3F8 indicates its excellent separation capability, i.e., it can meet the separation demand at very low concentration of C3F8.
IAST selectivity of Ba-ETS-4 for C3F8/N2(1:400) binary mixtures at 298 K.
Desorption and regeneration properties of Ba-ETS-4
The desorption and regeneration performance are also an important part of evaluating the adsorption performance of the adsorbent. We tested the desorption properties and cyclic stability of Ba-ETS-4 materials by five consecutive fixed-bed adsorption breakthrough experiments with C3F8/N2. The experimental conditions were referred to 3.2.1(flow rate of 1.0 L/min, temperature of 298 K, and pressure of 1.70 kPa in N2), and the desorption process was carried out by blowing the fixed-bed with nitrogen.
From Figure 11, C3F8 was rapidly desorbed in the early stage of the desorption process, and C/C0 < 0.1 at 90 min. Until C3F8 was almost undetectable at 273 min, the desorption was complete. The transmission time and saturation time of five adsorption-desorption cycle curves are basically the same, and the curves have no obvious left shift, indicating that the Ba-ETS-4 material has high cyclic stability and good desorption and regeneration performance.
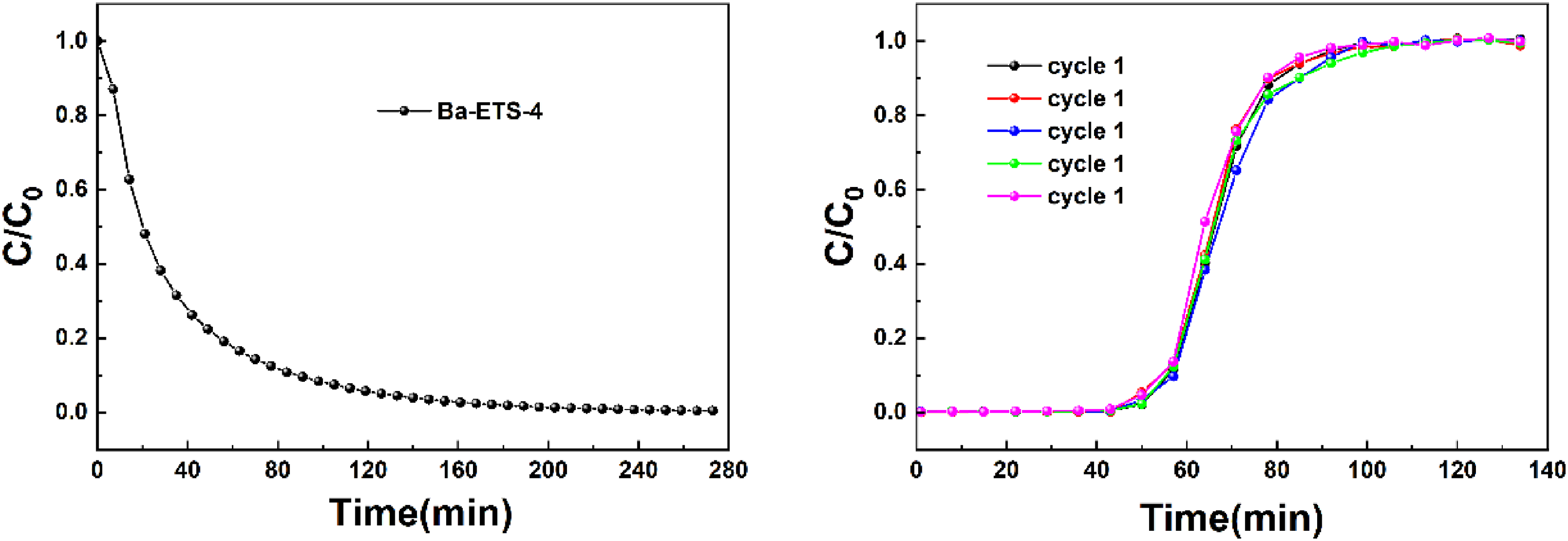
Desorption curves of Ba-ETS-4 on C3F8 at 298 K and Adsorption-desorption cycle curves in C3F8/N2 mixture.
Conclusion
In summary, Na-ETS-4 was synthesized by solvothermal method, and then a series of X-ETS-4 (X: Mg, Ca, Sr, Ba) molecular sieves were obtained by ion exchange modification for the adsorption separation of C3F8/N2. The results of XRD and TGA showed that the introduction of Ba2+ resulted in a greater deformation of the framework structure and higher thermal stability compared to other ions. Through the fixed-bed adsorption breakthrough experiment, the adsorption capacity of Ba-ETS-4 for C3F8 is greatly improved at very low concentration. The dynamic adsorption capacity of Ba-ETS-4 for C3F8 reached 0.83 mmol/g (155.17 mg/g) (298 K, 170 kPa, C3F8/N2 volume ratio = 1:400), which was 15.49 times higher than Na-ETS-4. The results of adsorption isotherm of Ba-ETS-4 on C3F8 and N2 conformed to the Freundlich isothermal adsorption model, indicating that the adsorption process was multilayer adsorption. The adsorption heat of Ba-ETS-4 on C3F8 (27.33–32.31 kJ/mol) was higher than that on N2 (16.36–20.54 kJ/mol), which indicated that the Ba-ETS-4 exhibited stronger interaction and higher adsorption selectivity on C3F8. The high selectivity of Ba-ETS-4 for C3F8(841) demonstrates its superior separation capability, enabling efficient C3F8 capture even at extremely low concentrations. In addition, Ba-ETS-4 also showed excellent desorption performance and cycle stability. As a result, Ba-ETS-4 exhibited excellent thermal stability and promising C3F8/N2 separation potential, making it a prospective adsorbent for highly efficient C3F8 removal.
The binary gas mixture system (C3F8/N2) adopted in this study differs from actual industrial gas compositions. Future research could incorporate additional fluorinated gases such as C3HF7, C2F6, and even H20 molecules to further investigate the adsorption and separation performance of multi-component gas mixtures.
With the advancement of molecular simulation technologies, combined computational-experimental approaches for crystal engineering studies have become increasingly prevalent. Subsequent work could integrate classical density functional theory (DFT) and Grand Canonical Monte Carlo (GCMC) methods to more accurately optimize material structures and predict material properties.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (grant number: 2024YFB4608600).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
